Abstract
Introduction
Fibromyalgia (FM) is a condition characterized by generalized chronic pain associated with fatigue, sleep disorders, and comorbidities, including anxiety and depression. 1 Chronic pain is the primary symptom of FM and may lead to functional work incapacity and a decline in the quality of life. 2
According to the American College of Rheumatology (ACR), FM involves generalized chronic pain in the axial and peripheral skeleton, and is diagnosed by the presence of pain symptoms in at least 11 of 18 painful spots in the body. 3 In 2010, the ACR proposed complementary criteria for the diagnosis of FM. The affected individuals should meet 3 criteria: (1) widespread pain index (WPI) ≥ 7 and symptom severity (SS) score ≥ 5 or WPI of 3 to 6 and SS score ≥ 9; (2) presence of symptoms for at least 3 months; and (3) absence of another disorder that could explain the pain symptoms. 4
The pathophysiology of FM is still unknown, but this disease may be associated with changes in the central mechanism of pain control due to dysfunctions in neurotransmission, 5 leading to a deficiency in inhibitory neurotransmitters at the spinal or supraspinal levels (serotonin, enkephalin, and noradrenaline), or the hyperactivity of excitatory neurotransmitters (substance P, glutamate, and bradykinin). 6
The source analysis of EEG recordings shows the presence of hyperexcitability in the areas involved in pain processing, and this mechanism may be important for sustained pain in FM. 1 In this context, changes in EEG could serve as a physiological biomarker for different pain syndromes. 7 EEG profiles of populations with chronic pain are being evaluated to elucidate the pathophysiology of pain and to allow assessing it and monitoring its management. 1
Although there is a previous review of electroencephalographic patterns in chronic pain, the findings are heterogeneous and mostly unexplored for FM. 7 In addition, little was explored about the protocols used in FM. Some studies that evaluated chronic pain using EEG showed similar characteristics among individuals suffering from different pain syndromes, but the data from these studies remains inconclusive.1,7 Thus, changes in cortical activity in different regions of the brain, including frontal, parietal, and occipital lobes, as well as in the sensorimotor and somatosensory regions, were associated with chronic pain. 7
In this respect, the objective of this study was to investigate the main EEG findings in the literature on FM concerning (a) the use of EEG in the evaluation of individuals with FM, (b) differences in protocols for the use of EEG in FM, (c) the main electroencephalographic findings in individuals with FM, and (d) the EEG analysis method.
Methods
For this study, a systematic review of the literature was performed on the use of EEG in individuals with FM. A search was conducted in the PubMed, Lilacs, and SciELO databases using the following keywords: “fibromyalgia” AND “electroencephalogram”; “fibromyalgia” AND “electroencephalography.”
The inclusion criteria were studies on the topic; publications in English, Portuguese, or Spanish; with descriptors in the title or abstract; human populations over 18 years; and cross-sectional studies, case–control studies, cohort studies, pilot studies, or clinical trials available in full from 2001 to 2020; and studies with primary or secondary outcomes based on baseline electroencephalographic data at rest. The exclusion criteria were the use of EEG to evaluate clinical conditions other than FM, review articles, and duplicate studies. The systematic review complied with the PRISMA criteria.
The database search was carried out by 2 independent evaluators, who performed a preliminary screening of the studies by analyzing the titles and abstracts. A third evaluator examined the selected articles, identified duplicates, and applied the exclusion criteria.
After the final selection, a thematic analysis was carried out, in which the 19 chosen articles were examined by the 2 independent evaluators, who filled out a form for each article designed to select the data most relevant to the review. At this stage, the previously defined categories were the objectives, methods, EEG protocols, and main EEG findings.
Results
A total of 136 articles were identified after a database search using the keywords “fibromyalgia” AND “electroencephalography”, and 131 articles were found using the keywords “fibromyalgia” AND “electroencephalogram”. In the end, 20 articles remained after applying the exclusion criteria, 14 of which were case–control studies and 6 were clinical trials. The flowchart of the phases of the systematic review is shown in Figure 1. General information from the 19 articles is summarized in Table 1.
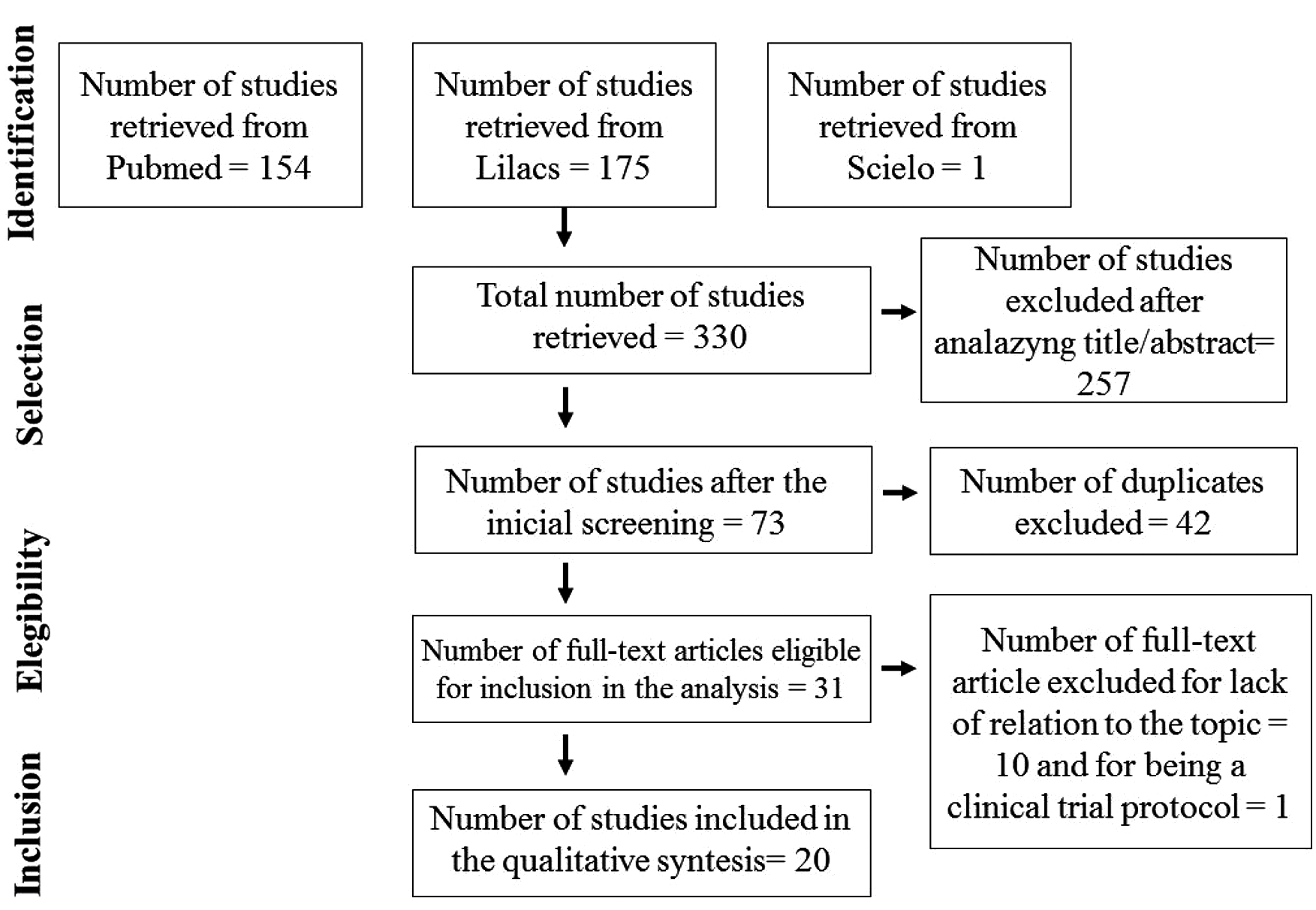
Flowchart of the phases of the systematic review according to the preferred reporting items for systematic reviews and meta-analyses criteria.
General Information From the Articles Included in the Systematic Review.
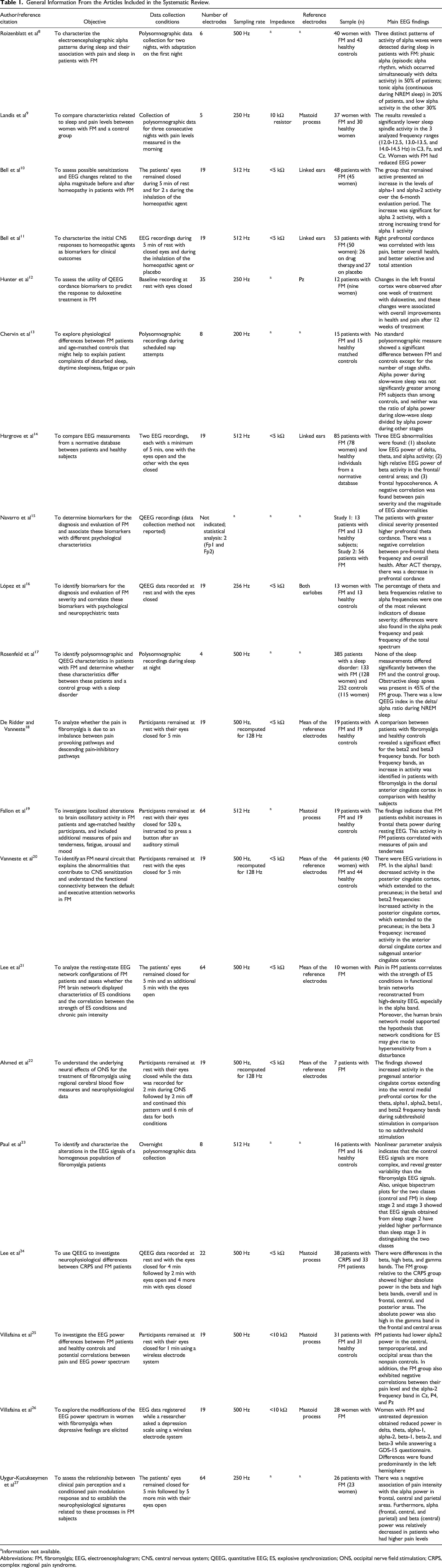
Information not available.
Abbreviations: FM, fibromyalgia; EEG, electroencephalogram; CNS, central nervous system; QEEG, quantitative EEG; ES, explosive synchronization; ONS, occipital nerve field stimulation; CRPS, complex regional pain syndrome.
Of the 20 included articles, 11 studies were published in the past 5 years, demonstrating the increased interest in the subject in recent years, 4 studies were published from 2000 to 2005, and 16 studies were published on or after 2006. With regard to the study origin, 9 studies were conducted in the United States, 4 in Spain, 3 in Belgium, 1 in the United Kingdom, 1 in India, 1 in South Korea, and 1 in Brazil. The main topics addressed in these studies were sleep disorders in FM patients using polysomnography, the effect of FM treatment in this population, neurophysiological differences in FM patients compared to other populations and the relationship between clinical measures and EEG spectral power differences.
Discussion
The systematic review indicated that most of the publications on the use of EEG for the electrophysiological evaluation of individuals with FM from 2001 to 2020 had the main objectives of identifying biomarkers for pain, performing a polysomnographic analysis, and correlating EEG findings with responses to different types of treatments.
Data from the literature was classified into 3 subcategories: (a) the use of EEG in the evaluation of individuals with FM, (b) differences in protocols for the use of EEG in FM, (c) the main electroencephalographic findings in individuals with FM, and (d) the EEG analysis method.
Use of EEG in the Evaluation of Individuals With FM
From a general perspective, research using EEG aims to explore the neural network configurations in FM. For this purpose, studies have investigated different variables, including the polysomnographic characteristics of this population,8,9,13,17,23 the relationship between a specific frequency band and its changes in response to treatment,10-12,15,22 physiological biomarkers related to clinical characteristics,16,19,21,25-27 and neural circuits characteristic of patients with FM.14,18,20,24
Lee et al 24 used EEG to relate the basal neurophysiological activity of patients with FM and with complex regional pain syndrome to quantify the differences between neural networks in FM and other chronic pain conditions. In this way, it was possible to access alterations proper to the condition in question.
Besides the resting state, studies involving other modalities of EEG applications in individuals with FM have been conducted. In this respect, the use of EEG biofeedback is critical because FM is a central sensitization syndrome with specific EEG abnormalities. 28 These authors affirm that therapeutic alternatives based on the guided exploration of brain waves have been sought for the amelioration of neurosomatic symptoms. 28 Other studies have evaluated responses to the observation of emotional images and scenes.1,29 Attention deficits have also been studied in individuals with FM, and correlations between these dysfunctions and somatic symptoms were established. 30 However, studies that present the activity component during EEG data collection configure specific alterations in neural networks, oriented to the task in question, and different from those proposed in this review.
Differences in Protocols for the Use of EEG in FM
Most of the studies included in the review analyzed the basal brain activity in FM, particularly with regard to alterations in frequency bands as specific markers of FM14-16,19-21,24-27 and other markers during sleep.8,9,13,17,23 Thus, the EEG parameters described were mostly oriented to patients at rest and polysomnography. However, different protocols were used in data collection, influencing the variety of findings that will be described later.
In general, studies evaluating cortical electrical activity in individuals with FM have used an adjustable cap containing electrodes distributed on the scalp according to the international 10 to 20 EEG system.9-12,16,18-22,24-26 The most commonly used electrodes were wet electrodes, which require an electrolyte gel or other material to transmit the signal from the scalp to the electrode, which is coated with Ag-AgCl. This coating, together with the conductive gel, decreases the resistivity between the skin and the electrode. 31
In a different scenario, Villafaina et al25,26 used a wireless electrode system, the Enobio device, for data collection in the EEG, whose reliability is validated even in a medium with dry electrodes. Although the use of fixed electrodes, concomitant to the conductive gel, allows better recording quality, the wireless electrodes in question have shown to be a good alternative for enabling a shorter preparation time for data collection and faster stabilization. 32
The number of EEG electrodes varied; some studies used10,11,14,16,18-20,22,25,26 32 electrodes and others 22 eletrodos 24 or 64 electrodes.19,21,27 The polysomnography studies have used a reduced number of channels, ranging from 4, 17 6, 6 and 8 electrodes.13,23 Reis et al 31 reported that the number and spatial distribution of electrodes affected the spatial resolution and accuracy of source localization. Thus, studies with a larger number of channels present a better accuracy on the oscillatory activity of a specific cortical region.
The included studies used an impedance of 5 kΩ10,11,14,16,20-22,24 and 10 kΩ9,25,26. The acquisition rate followed the value established in the Nyquist equation, which estimates that the sampling rate should be higher than twice the frequency range of interest. A low impedance, mainly between 1 and 5 kΩ, is essential to obtain better electroencephalographic data, and it can substantially reduce the electrical noise. 33 However, some studies did not give data about the used impedance,8,12,13,15,17,19,23,27 thus impairing the reliability regarding the signal quality of the collection.
Therefore, the sampling rates used were 200, 13 250,9,12,27 256, 16 500,18,20-22,24-26 and 512 Hz.10,11,14,19,23 The sampling rate usually ranged from 250 to 2000 Hz, which should be more than twice the Nyquist frequency to ensure adequate sampling and minimize aliasing. 31 However, data obtained by some of the included studies performed signal resampling during data analysis.18,20,22
Another critical point is the location of reference electrodes. In this respect, the sites included the mastoid process,9,19,24-26 earlobes, 16 linked ears,10,11,14 and the calculation of the overall mean ofelectrodes.18,20-22 Such diversity in findings corroborates with the general literature regarding EEG protocols, where there is still no consensus on the best location for reference electrodes. 33
Furthermore, EEG data were recorded following different approaches according to the purpose of each analysis. Most of the studies acquired data with the participants at rest and with the eyes closed.10-12,16,18-21,25 Although there is a greater possibility of the existence of artifacts, due to the occurrence of blinking, capable of affecting EEG data, some studies performed data collection in the opened eye condition.14,21,24,27 However, Hargrove et al 14 highlighted that the 2 types of collection provide similar data. As a strategy to reduce the possible drowsiness caused by the closed eye data collection, Fallon et al 19 presented auditory stimuli at intervals of between 35 and 45 s using external speakers. The participants should react to the stimuli by pressing a computer mouse button to maintain the waking state only.
The different collection methods were applied for different purposes, such as to identify more objective indicators in the FM diagnosis and correlate these indicators with psychological tests, 16 determine possible changes in the EEG before and after the homeopathic treatment of patients with FM,10,11 and assess whether a brain EEG analysis could predict responses to antidepressants for FM symptoms. 12
The data recording performed by Ahmed et al 22 was obtained during occipital nerve stimulation in the participant; however, it was also collected at rest, for baseline activity analysis. Thus, the use of the EEG was intended for analysis of neurophysiological differences in these 2 scenarios.
Main Electroencephalographic Findings in Individuals with FM
The objective of these studies was to determine the main electroencephalographic findings in individuals with FM. Therefore, the main electroencephalographic findings were variable according to the objectives and method of each study. One of the critical parameters analyzed was the variation in electrophysiological patterns, which are usually altered in individuals with FM. 16 Despite the difficulty in identifying pathological conditions in patients using only EEG, it is possible to find correlations between the disease and EEG changes.
Frequency bands are known to reflect behavioral states. The theta activity is usually associated with somnolence in adults and with higher-order cognitive activity, whereas alpha activity is usually related to a state of relaxation. 30 Increased theta activity has been observed in the frontal cortex and is related to the measures of sensitivity, pain, and fatigue, and can contribute to the persistent perception of pain in FM. 19 Navarro et al 15 also explored theta activity, from the brain cordance, finding it increased in the frontal region and related to the clinical severity of FM. In theory, manipulation of the amplitude of the alpha and theta bands would lead to behavioral changes. 30
The beta frequency band has been shown to be increased in FM patients.14,18,20,24 De Ridder and Vanneste 18 found significant changes for the beta2 and beta3 bands in the dorsal anterior cingulate cortex compared with healthy individuals. In addition, they found a significant decrease in functional connectivity between the dorsal anterior cingulate cortex and the pregenual anterior cingulate cortex in patients with FM compared with healthy controls in the beta2 and beta3 frequency bands, which are part of the medial pain pathway that encodes the motivational and affective pain component. Such a change in these regions suggests that FM is related to increased pain awareness rather than decreased pain inhibition. 18
The beta relationship with other frequency ranges was discussed by Lee et al, 24 when comparing the neurophysiological activity of participants with FM and regional complex pain syndrome, another condition that has chronic pain as the main symptom. In this study, the authors found a higher beta wave activity in the frontal, central, and parietal regions, which could reflect the central sensitization observed in FM. In addition, a lower proportion between theta and beta was found, associated in the literature with lower cognitive and attentional performance. 24
The findings on beta frequency band corroborate with the study of González-Villar et al, 34 which have observed increased connectivity over different brain networks at beta band, and differential microstate dynamics during resting state, suggesting a relationship with the subjective complains about deficits in attentional processes and cognitive functioning in FM.
Some studies have pointed out that changes in cortical activity would be related to clinical characteristics in FM.16,20,25,27 Evaluation of brain function at rest can be of great importance to reveal physiological biomarkers, especially for the graduation of pain, currently based on subjective criteria.
In this context, Villafaina et al 25 found that alpha2 activity in the central and parietal regions was related to pain scores on the visual analog scale, a method for subjective measurement of painful sensation. Uygur-Kucukseymen et al 27 reported that alpha (frontal, central, and parietal) and β (central) power decreased relatively in patients with higher pain levels.
On the basis of the studies analyzed, alpha oscillations have been related to brain processing in FM. Lee et al 21 observed the characteristics of explosive synchronization, the property of a brain synchronization network that differs from the normal rest state. Such conditions were found in the alpha frequency range and may be a potential mechanism of pain hypersensitivity in FM. In this sense, patients with FM have a low alpha activity, which may result in deficits in the pain inhibitory system. López et al 16 have shown that a lower level of alpha activity is an indicator of decreased sensorimotor integration in brain processing and that extra effort was necessary to attenuate the sensation of chronic pain.
The decreased power values of both alpha-1 and alpha-2 subbands also were assessed by Nir et al, 35 which showed the alpha-1's predictive potential for individual experiences of tonic pain, suggesting that alpha-1 oscillations recorded during rest may reveal an individual predisposition of pain responsiveness and may be an experimentally stable measure of cognitive and sensory processes in FM.
Other clinical variables were also accessed in the studies, such as depression, 26 tension, 19 and the impact of FM on the daily life of participants 20 . Villafaina et al 26 found that women with FM and untreated depression demonstrated a reduced power in delta, theta, alpha1, alpha2, beta1, beta2 and beta3, mainly in the left hemisphere, compared with the control group. Fallon et al 19 found a relationship between stress measures and an increase in the tetha activity in the frontal region of TM. Regarding the impact on daily living activities, Vanneste et al 20 reported a positive relationship between alpha2 and beta1 activity in the anterior dorsal cingulate cortex region and the score of the impact measurement questionnaire in FM.
In general, studies that performed a general analysis of cortical activity at rest found significant differences in FM, with absolute power of delta, theta and alpha decreased, increased beta activity in the frontal and central areas, and frontal hypercoherence.14,20 Furthermore, Vanneste et al 20 emphasized the decrease in alpha1 activity in the posterior cingulate cortex extending into the precuneiform and increased beta1 and beta2 activity in the posterior cingulate cortex up to the precuneus.
An analysis of possible sensitizations related to changes in the magnitude of alpha activity in the EEG after homeopathic treatment indicated significant progressive increases in the magnitude of alpha1 and alpha2 activities over a 6-month period. 10 The same authors reported that the change in the relative magnitude of alpha activity during the sessions was time dependent and not correlated with clinical improvements.
During the occipital nerve stimulation, Ahmed et al 22 observed a significant increase in the anterior cingulate pregenus cortex, extending to the medial ventral prefrontal cortex for tetha, alpha1, alpha2, beta1, and beta2. In the study by Hunter et al, 12 using cordance analysis to predict the therapeutic response to duloxetine, changes were found in the left frontal cortex after 1 week of treatment. Both studies found changes in the cortical oscillatory activity related to a decrease in painful complaints.
The study by Roizenblatt et al 8 characterized the electroencephalographic patterns of alpha activity during sleep and their association with pain and sleep in patients with FM. The results indicated the presence of 3 distinct patterns of alpha activity during sleep in FM: phasic alpha, an episodic alpha activity co-occurring with delta activity in 50% of patients; tonic alpha, which was continuous during NREM sleep in 20% of patients; and low alpha activity in the remaining 30% of patients. Therefore, the alpha activity pattern is associated with an extended duration of pain symptoms, the perception of poor sleep, and morning pain, which are the primary symptoms of FM.
Studies using polysomnography have reported a decreased number of sleep spindles, which might result in deficiencies in thalamic-cortical oscillatory networks that are crucial in inducing and maintaining sleep. 9 These results may indicate altered sensory processing in the thalamus as a component of the hyperalgesia state. 9 A practical implication of these findings is that sleep is more easily disturbed by endogenous and environmental stimuli. 9
Paul et al, 23 after performing a nonlinear analysis to extract data from the EEG signal, identified that participants with FM, compared with the control group, demonstrate lower energy and power characteristics. These findings indicate that normal EEG signals are more complex and reveal greater variability than EEG signals in patients with FM.
In contrast to the studies mentioned above, Chervin et al 13 found no significant difference between FM and controls for the standard polysomnographic measurements, except for the number of sleep-stage changes. However, limitations on the sample size may have influenced the results.
In this sense, the inconsistent findings on oscillatory activity at rest in FM, previously pointed out in the literature, 19 occur because of the use of different protocols and objectives of the studies that use EEG as an evaluation tool. Thus, although the EEG presents itself as a promising tool in treating patients with FM, there is still a need for research with consistent findings using standardized collection protocols.
EEG Analysis Method
We observe a wide variability in the method of EEG data analysis, such as the software used for preprocessing and analysis, the method of removing artifacts, and the type of analysis performed. First, regarding the data analysis software, most studies used EEGlab, a Matlab toolbox for preprocessing, and analysis of EEG data.9,21,25-27 The second most used program was the Eureka software,18,20,22 followed by the Neuroguide software,14,24 and MATLAB10. Other methods were the software Meditron8, QEEGT® (Quantitative EEG Tomographic)15,16 software and the analysis by visual techniques to determine sleep stages. 23 A single study reported using a locally developed software. 9
It is important to note that some studies, most of them conducted before 2009, did not specify the EEG data analysis program. These authors only explained that the EEG data was submitted to the Fourier fast transform.10-12,17 Thus, we emphasize that the specification of the program used for preprocessing and analysis of the EEG signal is of the utmost importance, as it makes the method reproducible and clear.
Regarding the removal of artifacts from the EEG signal, most studies used independent component analysis (ICA).21,22,25-27 Some used only a manual inspection of artifacts10,11,16,18,20 and other studies used a toolbox from the software Neuroguide.14,24 Only one study cited that it used infraorbital electrodes to detect ocular muscle artifacts. 12 Several studies have not made clear the method used to remove noise from EEG.8,9,13,15,17,19,23
However, we emphasize that some studies, besides using some software to remove artifacts, also performed a visual inspection.12,14,21,22,27 We understand that this is a more effective way to avoid the presence of artifacts in the EEG signal, helping to ensure that the data collected reflects a record of brain activity.
The analysis of the spectral of frequency bands was the most widely used.8-14,16,19,21,25,27 Comparisons between on and off 22 conditions, coherence analysis between different spatial locations,14,18,20 cordance analysis,10-12,15 and n-REM sleep assessments from the frequency domain were also performed.17,23 The majority of them were the spectral analysis of frequency bands. In this case, power was the measure of amplitude, i.e. the greater the amplitude, the greater the amount of power in the EEG signal. Thus, it represents the amount of energy in a given frequency band on the scalp. 36
Finally, some studies used the software sLORETA18-20,26 to identify the corresponding estimated neuronal activity generators of each frequency band. In this case, sLORETA showed that it can be an efficient tool for functional mapping consistent with the physiology. 37 sLORETA has also been used to localize generators of scalp-recorded potentials, based on the use of laser-evoked potentials, including those related to pain processing and modulation. This fact may point in the direction of future studies. 38
Although all the criteria of a systematic review were followed, we highlight some limitations, such as the conclusions based on the qualitative analysis of the studies. A quantitative analysis of the studies was not possible because of the heterogeneity of the results. We suggest the use of similar protocols, whenever possible, to allow greater comparability of results.
Conclusion
This systematic review provided evidence that the interest in the electroencephalographic investigation of individuals with FM is increasing. Although there are different approaches to this method of studying the population with FM, the findings are discordant and insufficient for thoroughly assessing this clinical condition. The considerable variation in the protocols followed in the studies may explain the inconsistencies in the findings.
Analysis of cortical electrical activity using EEG recordings is a promising method for the identification and characterization of biomarkers in FM. In this respect, EEG protocols should be standardized to reduce possible biases among studies, facilitating the comparison of results.
Footnotes
Author Contributions
GAM and NT conceived the experiments; GAM and MLLHM analyzed and interpreted the data; NT designed the work; All authors reviewed the manuscript. All authors have approved the submitted version and have agreed both to be personally accountable for the author's own contributions.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by funding from Coordination for the Improvement of Higher Education Personnel - CAPES; Brazilian National Council for Scientific and Technological Development - CNPq (311910/2017-3), and Grant 008/2019, Pronex, Paraíba State Research Foundation - FAPESQ.
