Abstract
Background
The cerebellar cortex has gradually become a promising therapeutic target for improving motor recovery post-cerebral infarction, potentially dependent on the structural integrity of motor-related corticocerebellar pathways (CCP). However, the relationship between the imaging markers of motor-related CCP and motor prognosis remains inadequately explored. Utilizing diffusion tensor imaging (DTI), this study aims to longitudinally assess the role of motor-related CCP in predicting motor recovery for both upper and lower extremities following cerebral infarction.
Methods
Twenty-nine patients with right middle cerebral artery (MCA) infarction underwent 2 DTI scans 7 to 14 and 30 days after onset, and 29 age-sex matched controls received 1 scan. Fractional anisotropy (FA) values were measured for corticospinal tract (CST) and CCP (cortico-pontocerebellar tract, CPCT; dentate-thalamocortical tract, DTCT; dorsal-spinocerebellar tract, DSCT). Multivariate regression analyses were performed to examine the relationships between DTI parameters and Fugl-Meyer Assessment (FMA).
Results
Compared to the control group, FA and FA asymmetry index (FA-AI) of CST, DTCT, and DSCT on the affected side were significantly reduced. In the linear regression model, the decreased FA-AI of DTCT was a strong predictor for upper FMA (R2 adj = 0.271, P = .022), while the FA-AI of DSCT independently predicted lower limb FMA (R2 adj = 0.400, P = .019).
Conclusions
In patients of MCA infarction, FA-AI of motor-related CCP may be a valuable imaging indicator for predicting motor outcomes. The DTI-assessed structural integrity of the cerebellar ascending fiber tracts (DTCT and DSCT) may correlate with the motor recovery of the upper and lower extremities, respectively.
Keywords
Introduction
Cerebral infarction is one of the most significant causes of disability among adults, with up to 50% of survivors experiencing long-term motor impairment.1,2 Neuroplasticity has been well documented to be related to progressive spontaneous or treatment-driven improvements in motor recovery following a stroke. 3 Notably, the cerebellum has gradually emerged as a promising therapeutic target for regulating limb movement through its motion-related afferent and efferent fiber tracts. 4 Recent studies have indicated that the cerebellar cortex can influence the remodeling of the cerebral cortex via the motor-related corticocerebellar pathway (CCP). Therefore, the structural integrity of motor-related CCP is regarded as crucial for effective motor rehabilitation after stroke.5,6
Anatomically, there are 3 primary fiber tracts relating to afferent and efferent functions of motor-related CCP. The cortico-pontocerebellar tract (CPCT) transmits motor commands from the sensorimotor cortex downward to the contralateral cerebellum via the middle cerebellar peduncle (MCP).4,7 The dorsal spinocerebellar tract (DSCT) of the spinal cord is responsible for the feedback and projection of motor signals generated in the primary motor cortex (M1). It primarily conveys somatosensory information from the spindles and Golgi tendon organs of the ipsilateral trunk and limb muscles through the inferior cerebellar peduncle (ICP) to the cerebellum. 4 The dentate thalamocortical tract (DTCT) originates from the dentate nucleus, ascends through the superior cerebellar peduncle (SCP), and crosses the midbrain to project to the contralateral thalamus. This tract is responsible for inhibiting downward impulses from the cortex, thereby facilitating the transmission of motor information to the cerebral cortex. 8 Nowadays, to assess the integrity of these fiber tracts in vivo, non-invasive neuroimaging methods are mainly recommended to provide valuable imaging markers.
Diffusion tensor imaging (DTI) is a clinically feasible and non-invasive method in many clinical neuroimaging studies. The fractional anisotropy (FA) value, obtained from DTI data, represents the degree of anisotropy of water molecules in the specific tract. It and its derivatives-FA asymmetry index (FA-AI) have been used to examine white matter integrity.9,10 After stroke onset, due to the occurrence of Wallerian degeneration secondary to the injury, FA values in both the injured region and the remote areas on the fiber decreased and were generally lower than those on the uninjured side. Prior DTI studies have reported that secondary degeneration of the contralateral cerebellar peduncle following supratentorial stroke correlates with the motor function of the hemiplegic limb.11,12 Yoo et al 7 conducted a retrospective study indicated that the structural integrity of the DTCT could predict long-term motor outcomes evaluated by the Fugl-Meyer Assessment Upper Extremity (FMA-UE). In another study, Kim et al 13 investigated the correlation between fractional anisotropy (FA) and its derivative indices with motor function scores in 23 patients with subacute middle cerebral artery (MCA) infarction. They discovered that the FA asymmetry index (FA-AI) of the ICP was positively correlated with impaired muscle movement. Liu et al 14 conducted a longitudinal study examining changes in the spinal cerebellar pathway among 33 patients with subacute MCA stroke. They found that the FA value of the contralateral ICP could serve as an indicator of post-stroke outcomes evaluated by the Fugl-Meyer Assessment Lower Extremity (FMA-LE).
To sum up, previous studies have suggested that the integrity of a single motor-related CCP tract may be correlated with motor recovery in patients with cerebral infarction. However, few studies have focused on (1) selecting suitable DTI parameters for the evaluation of motor-related CCP, (2) evaluating the correlations between the integrity of different tracts of motor-related CCP, and (3) comparing the predictive value of motor-related CCP tracts for the movements of different limbs. We suppose that the motor disorder of the upper or lower limb may be related to the integrity of different motor-related CCP tracts. Therefore, our study will first examine the structural integrity changes of all 3 motor-related CCP tracts using different DTI parameters (FA values and FA-AI) in patients with subacute MCA infarction, compared to healthy controls. Second, we will evaluate the correlations between the DTI parameters of CPCT, DTCT, and DSCT and investigate their relationships with motor function scores for both upper and lower limbs. Finally, we will establish multivariate linear regression models with DTI markers from baseline and 30 days later to identify early imaging indicators for predicting late clinical outcomes.
Methods
Study Design and Subjects
This prospective study received approval from the institutional review board of Xuanwu Hospital (No. 2021[139]), and each participant provided written informed consent. The study involved the prospective collection of patients with subacute cerebral infarction who were hospitalized at our institution between 2022 Feb and 2023 Dec. The criteria for enrollment included: (1) admission within 7 to 14 days after symptom onset, with a complete medical history and the ability to cooperate with DTI scan; (2) infarction confined to the right MCA territory, (3) modified Rankin Scale score of ≥3; and (4) the absence of other neurological or psychiatric disorders. The exclusion criteria included: (1) contraindications for MRI scans; (2) deafness and/or blindness, aphasia, or visual field deficits; (3) occlusion of the right internal carotid artery; (4) new-onset neurological impairment during the observation period, other neurological disorders aside from cerebral infarction, or an inability to receive standard rehabilitation treatment due to serious medical complications during their stay in the inpatient rehabilitation facility; and (5) diffuse white matter hyperintensities on T2-weighted images (Fazekas score ≥2 for periventricular and/or deep white matter) or any evidence of structural abnormalities on the scans.
Of the 40 patients recruited according to the inclusion criteria, 11 were excluded (1 patient underwent cardiac surgery, 2 patients were interrupted in a scan, and 8 were lost to follow-up). Consequently, 29 patients underwent 2 DTI scans and motor assessments during both the subacute phase (7-14 days after onset) and the 30 days after onset. All subjects received routine rehabilitation therapy, encompassing physical therapy, functional training, balance training, and gait training for 2 hours per day, 5 days a week, over 4 weeks. Additionally, 29 age-matched healthy controls were recruited from the cohort at the Health Promotion Center of our institution. Motor assessments were conducted using the FMA-T (total FMA, score range 0-100), FMA-UE (score range 0-66), and FMA-LE (score range 0-34).
Date Acquisition
All subjects underwent an MRI scan using a 3.0 T PET/MR system (Signa, GE Healthcare, WI, USA) equipped with a 19-channel head and neck coil. The scanning protocol encompassed the entire brain, utilizing the following sequence parameters: Sagittal T1-weighted 3-dimensional (3D) turbo field echo, repetition time (TR)/echo time (TE) = 8.5 ms/3.2 ms, flip angle = 15°, FOV = 256 × 256 mm2, matrix = 256 × 256, voxel = 1.0 × 1.0 × 1.0 mm3, several slices = 188, and scanning time = 318 s. DTI adopts a single spin echo plane imaging pulse sequence, TR/TE = 9231 ms/97.2 ms, FOV = 224 × 224 mm2, matrix = 112 × 112, voxel = 2.0 × 2.0 × 3.5 mm3, b values = 0 and 1000 s/mm2, several slices = 2960, non-collinear directions = 64, and scanning time = 692 s.
Image Processing and DTI Analysis
The lesion of each patient was drawn on the respective high-resolution T1-weighted images using MRIcron (nitrc.org/ projects/mricron), record lesion volume, and then, the binary lesion masks were spatially normalized to a Montreal Neurological Institute (MNI) brain template. Finally, the lesion masks were summed and overlapped on the MNI template (Figure 1).
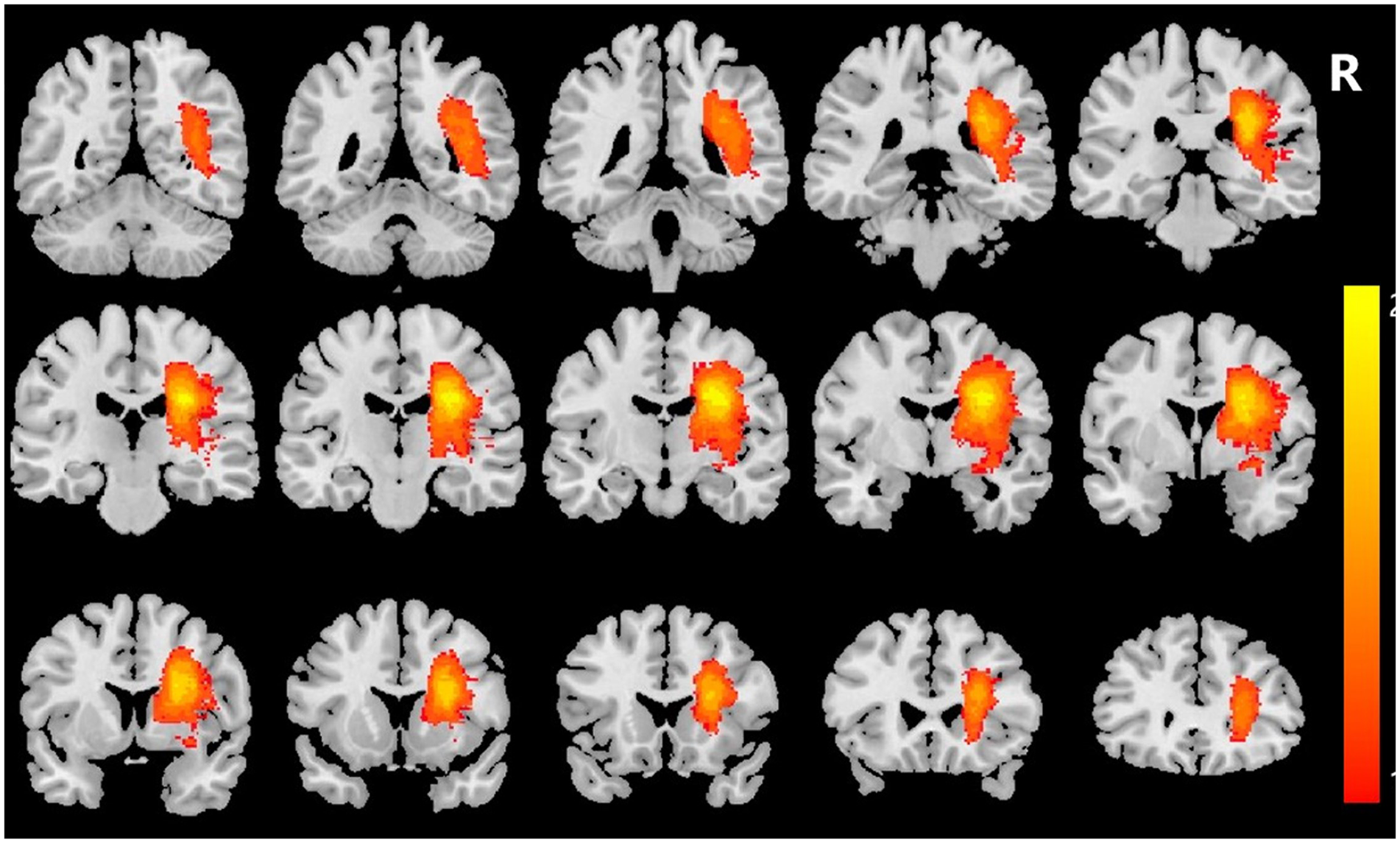
Lesion maps of the patients (L, left; R, right). All patients had lesions located in the right cerebral hemisphere. Colors represent the number of patients with a lesion at the given location.
DTI data preprocessing was performed with QSIPrep, including denoising, bias field correction, motion, and eddy current correction. FA maps were generated with FSL’s (FMRIB Software Library, version 6.0.0.1:57B01774) dtifit. 15 The regions of interest (ROIs) were manually defined on a voxel-by-voxel basis by 2 investigators (L.J. and S.Y.), each possessing over 5 years of experience in brain anatomy and DTI. These investigators were blinded to the motor function of the patients. The design of the ROIs at each location was informed by topographical maps from previous studies, as well as color FA maps, and was outlined bilaterally (Figure 2). The affected and contralateral ROIs for patients were delineated, along with the left and right ROIs for normal subjects. The ROI of CST defined the area represented by the blue fibers of the color FA map at the pontomedullary junction (PMJ) level on the axial plane (online-only Data Supplement). The ROI of CPCT is delineated in MCP, the ROI of DTCT is delineated in SCP, and the ROI of DSCT is delineated in ICP. All 3 ROIs are displayed in the coronal image, with the sagittal plane FA color map as a reference. The cyan fibers of the SCP are located on the rostral side of the transverse bridge fibers. The MCP is defined by a region of green fibers surrounding the transverse bridge fibers, and the ICP is defined by blue-purple fibers at the pontomedullary junction. Each ROI is sketched in voxels at the 3 levels that show the best. Quantitative values of FA were obtained by averaging all voxels within the ROI using MRIcroGL software. The asymmetry index of FA (FA-AI) was calculated using the following formula: (FAaffected − FAunaffected)/(FAaffected + FAunaffected). 13 A lower FA-AI indicates a lower FA value on the affected side and vice versa. The formula (FAright − FAleft)/(FAright + FAleft) was used to calculate the FA-AI in the control group. 16
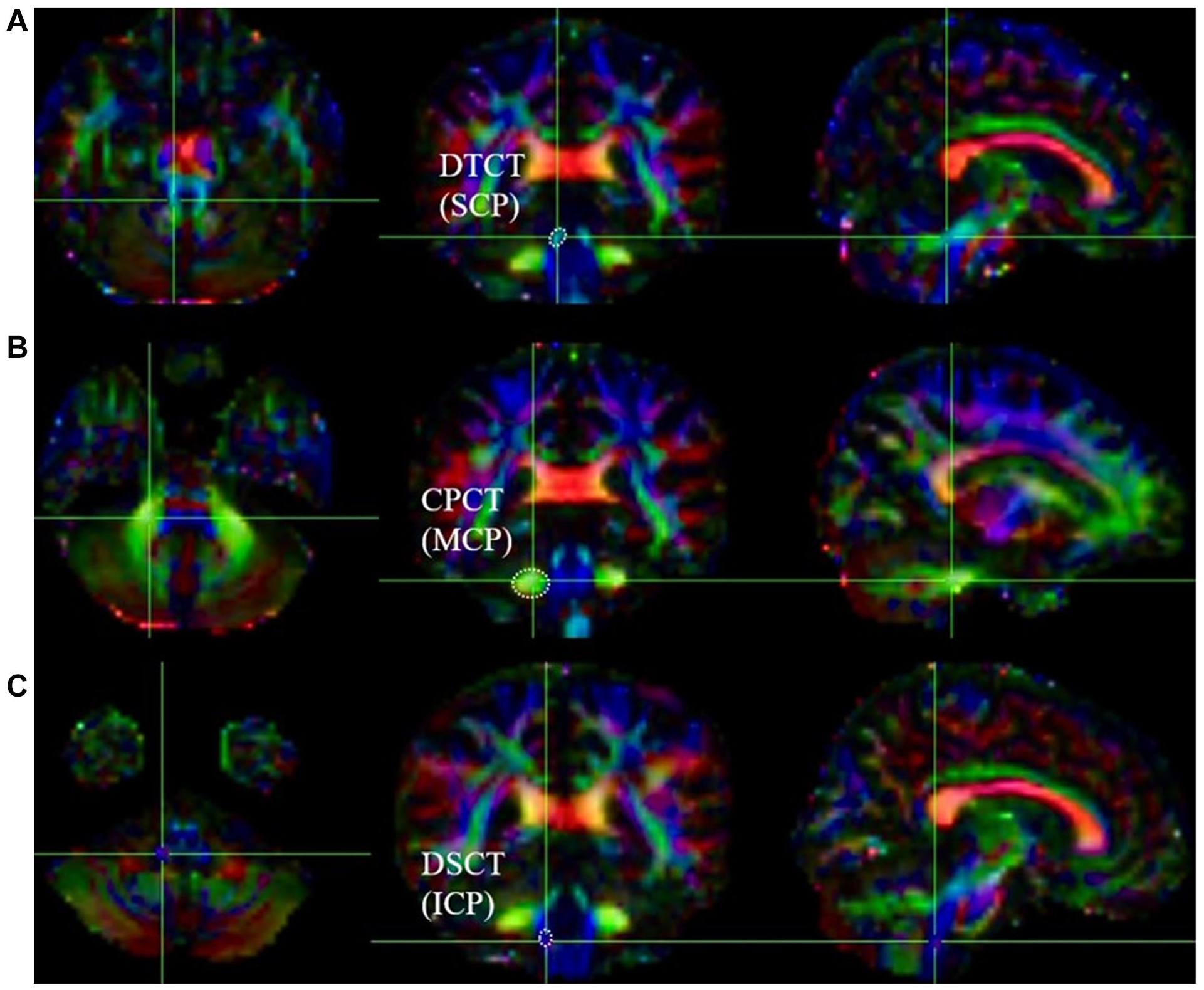
Color fractional anisotropy (FA) maps of diffusion-tensor imaging delineating the regions of interest (ROIs). Axial, coronal, and sagittal sections are shown from left to right. (A) The ROI of the dentate thalamocortical tract (DTCT) in the superior cerebellar peduncle (SCP) is indicated by a dotted line in the coronal image. (B) A dotted line in the coronal image indicates the ROI of the cortico-pontocerebellar tract (CPCT) in the middle cerebellar peduncle (MCP). (C) The ROI of the dorsal spinocerebellar tract (DSCT) in the inferior cerebellar peduncle (ICP) is indicated by a dotted line in the coronal image at the pontomedullary junction (PMJ).
Statistical Analysis
The Shapiro-Wilk test assessed the normality of the functional parameters and DTI-derived indices. The reliability of the ROI measurements between observers was evaluated by Intraclass Correlation Coefficient (ICC) with 95% confidence intervals (CI) using a mean-rating (k = 2), absolute-agreement, 2-way mixed-effects model was used to assess FA values for each ROI in healthy participants and patients. A paired t-test was employed to compare the FA values of CST and CCP’s affected and unaffected sides. The Wilcoxon signed-rank test was utilized for data that did not conform to a normal distribution. An unpaired t-test was used to compare the FA-AI values between the patient and control groups. Spearman correlation analysis assessed the correlation between each fiber and the FMA score. A multivariate linear regression model determined the association between the FA-AI and FMA-T, FMA-UE, and FMA-LE scores. Age, sex, and lesion volume were identified as confounding variables, whereas the FA-AI obtained at each anatomical location was used as the independent variable. All tests were 2-tailed, and a significance level of P < .05 was established.
Results
Table 1 summarizes the demographic and clinical characteristics of the patients. The lesions of 29 patients were located in the right MCA, without the involvement of the brain cortex, with an average lesion volume of 44.670 ± 25.466 mm3. The inter-rater reliability of the ROI measurements (ICC) between the 2 investigators (L.J. and S.Y.) was 0.881 (95% CI: 0.843-0.910).
Demographic and Clinical Characteristics of the Patients and Controls.
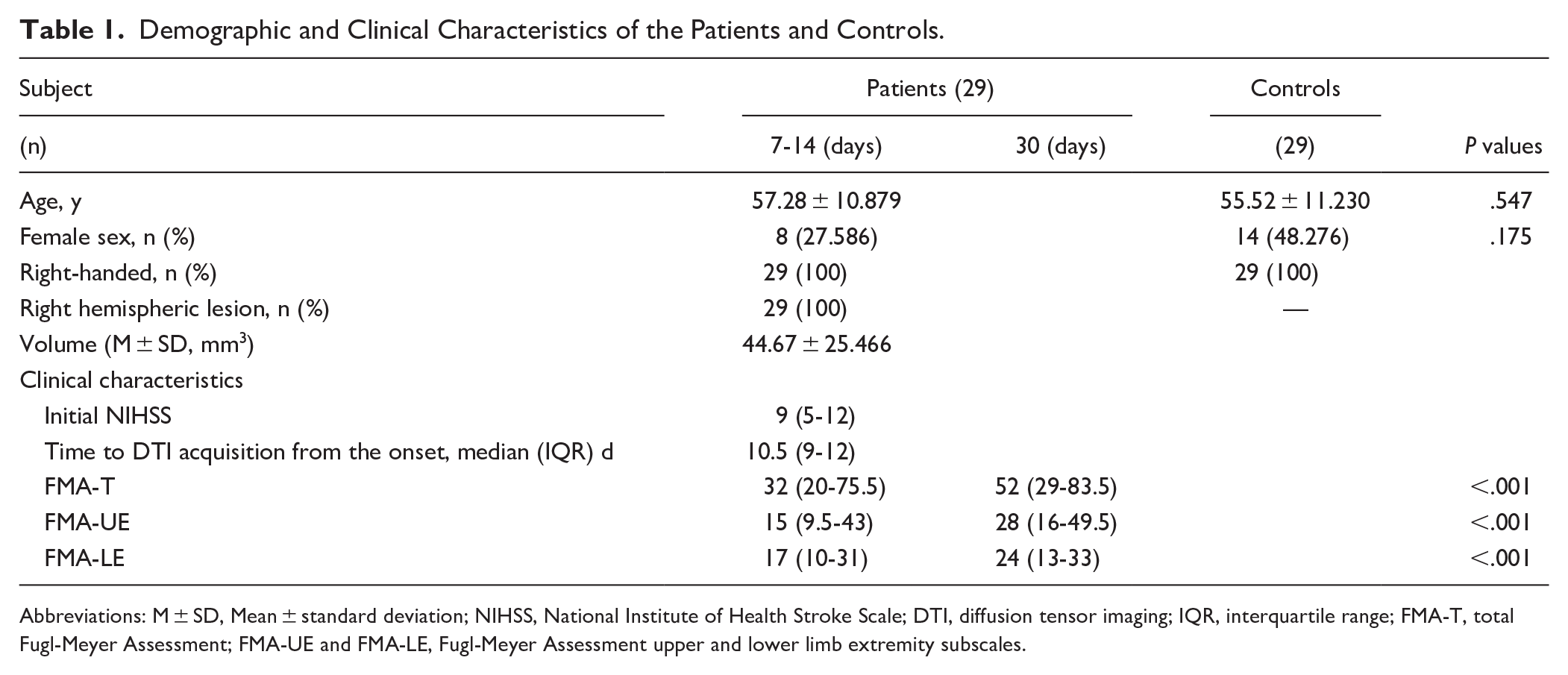
Abbreviations: M ± SD, Mean ± standard deviation; NIHSS, National Institute of Health Stroke Scale; DTI, diffusion tensor imaging; IQR, interquartile range; FMA-T, total Fugl-Meyer Assessment; FMA-UE and FMA-LE, Fugl-Meyer Assessment upper and lower limb extremity subscales.
Comparisons of CCP Integrity in Patients with Subacute MCA Infarction
Table 2 presents the mean FA values and FA-AI for CST and each CCP tract. In the control group, no significant differences were observed among the bilateral tracts (P > .05). In the patient group, the FA values of PMJ, SCP, and ICP on the affected side were significantly lower than those on the contralateral side (P < .001). However, no significant differences were found between the 2 sides of MCP (P = 0.712).
Comparison of Regional FA Between Affected and Unaffected Tracts and Comparison of FA Asymmetry Indices Between Patients and Controls.
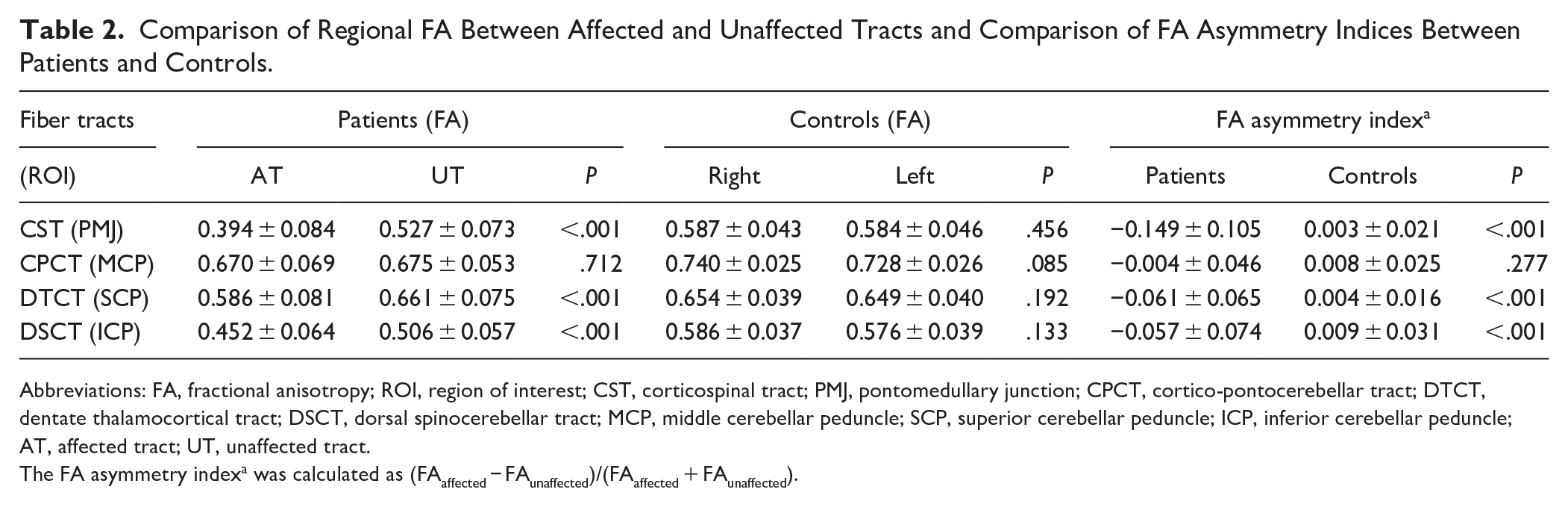
Abbreviations: FA, fractional anisotropy; ROI, region of interest; CST, corticospinal tract; PMJ, pontomedullary junction; CPCT, cortico-pontocerebellar tract; DTCT, dentate thalamocortical tract; DSCT, dorsal spinocerebellar tract; MCP, middle cerebellar peduncle; SCP, superior cerebellar peduncle; ICP, inferior cerebellar peduncle; AT, affected tract; UT, unaffected tract.
The FA asymmetry indexa was calculated as (FAaffected − FAunaffected)/(FAaffected + FAunaffected).
The FA-AI values were significantly lower in the patient group compared to the control group for PMJ, SCP, and ICP (P < .001, Bonferroni corrected in supplemental material Table 2). However, no significant difference was observed in the FA-AI for the MCP between the 2 groups (P = 1.00). Moreover, FA-AI of SCP was significantly positively correlated with FA-AI of ICP (r = 0.445, P = .016), while FA-AI of MCP was not significantly associated with the other 2 fiber tracts (P > .05).
Correlations Between the FA Asymmetry Index and Motor Function Assessments
The results of the correlation analysis are presented in Table 3. No significant correlation was found between the FA-AI of the PMJ and MCP and FMA-T, FAM-UE, or FMA-LE during both 7-14 days and 30 days after onset (P > .05). In the subacute phase, the FA-AI of the SCP was positively correlated with FMA-T, FAM-UE, and FMA-LE (r = 0.655, 0.624, and 0.591; P = .000, .000, and .001, respectively). FA-AI of the ICP exhibited positive correlations with FMA-T and FAM-LE (r = 0.431 and 0.582; P = .020 and .001, respectively), while no such correlation was found for FAM-UE (r = 0.260; P = 0.173).
Correlation Between the FA Asymmetry Index and Fugl-Meyer Assessment in the Patient Group.
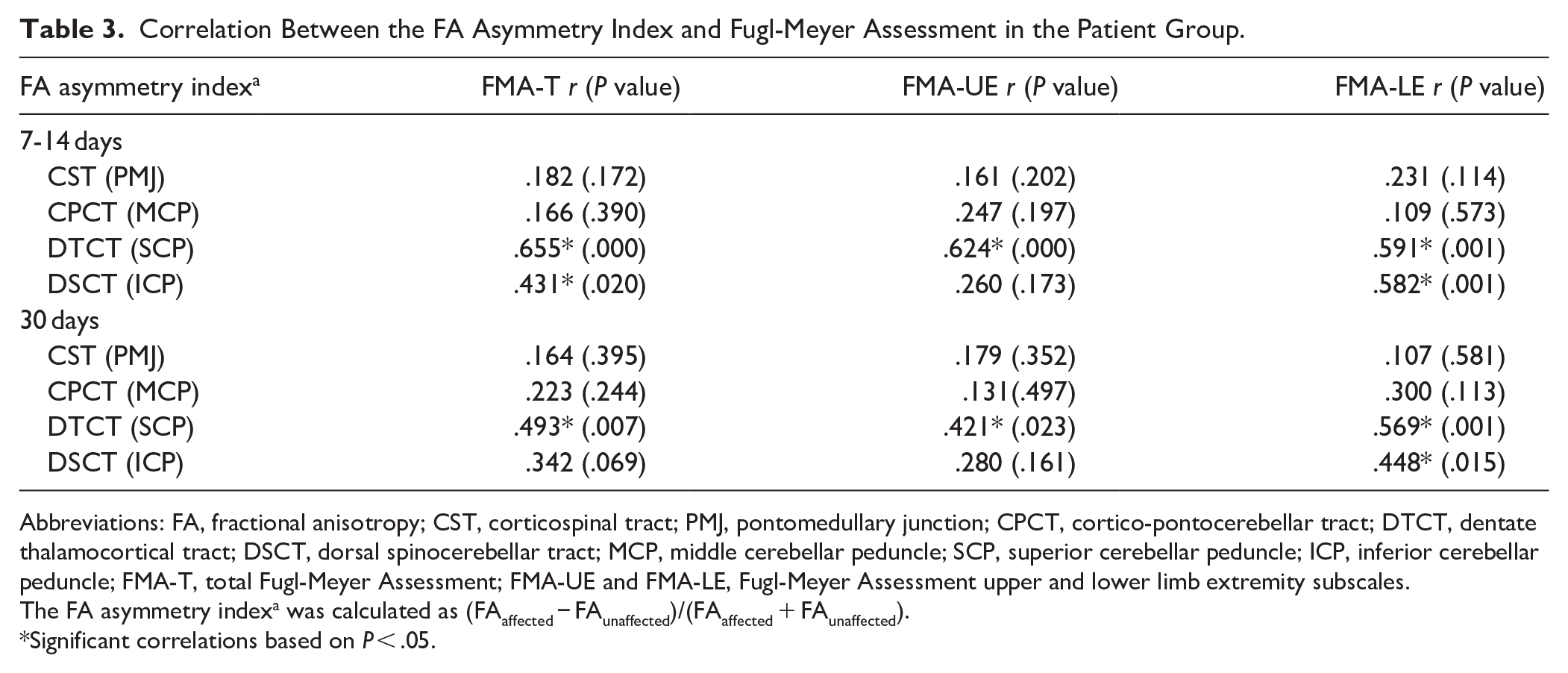
Abbreviations: FA, fractional anisotropy; CST, corticospinal tract; PMJ, pontomedullary junction; CPCT, cortico-pontocerebellar tract; DTCT, dentate thalamocortical tract; DSCT, dorsal spinocerebellar tract; MCP, middle cerebellar peduncle; SCP, superior cerebellar peduncle; ICP, inferior cerebellar peduncle; FMA-T, total Fugl-Meyer Assessment; FMA-UE and FMA-LE, Fugl-Meyer Assessment upper and lower limb extremity subscales.
The FA asymmetry indexa was calculated as (FAaffected − FAunaffected)/(FAaffected + FAunaffected).
Significant correlations based on P < .05.
Thirty days after onset, the FA-AI of the SCP exhibited positive correlations with the FMA-T, FAM-UE, and FMA-LE (r = 0.493, 0.421, and 0.569; P = .007, .023, and .001, respectively). In contrast, the FA-AI of the ICP demonstrated a positive correlation only with the FAM-LE (r = 0.448; P = .015), while no significant correlations were observed with FMA-T and FAM-UE (r = 0.342 and 0.280; P = .069 and 0.161).
Predictive Value of CCP Integrity Determined by Multivariate Linear Regression Models
The results of multiple linear regression analysis of FA-AI at 7-14 days and FMA scores at 30 days post-onset was presented in Figure 3. The FA-AI of the PMJ is excluded from any predictive models (online-only Data Supplement). The FA-AI of the SCP independently contributed to FMA-T, FMA-UE, and FMA-LE (R2adj = 0.301, 0.294, and 0.259; P = .006, .008, and .013, respectively). Additionally, the FA-AI of the ICP independently contributed to FMA-LE (R2 adj = 0.342, P = .003). In the intercept model of PMJ, SCP, and ICP, the FA-AI of SCP independently predicted FMA-UE (R2 adj = 0.271, P = .022), while the FA-AI of ICP independently predicted FMA-LE (R2 adj = 0.400, P = .019).

Multivariate linear regression models were employed to predict the FMA score at follow-up based on the FA-AI of each CCP tract at baseline. Models A and B represent predictions of FMA-UE and FMA-LE scores based on DTCT, respectively. Plot C Displays the prediction of the FA-AI of DSCT to FMA-LE. Plots D and E, the interaction model after controlling variables shows that FA-AI of DTCT predicts FMA-UE and FA-AI of DSCT predicts FMA-LE.
Discussion
Our study proposes that appropriate DTI indicators can predict motor recovery in both the upper and lower limbs, respectively. First, we found that the structural integrity, as indicated by FA values on the affected side and FA-AI between the bilateral sides, of the ascending pathways (DTCT and DSCT) was significantly compromised in patients with subacute MCA infarction, while no significant damage was observed in the CPCT. Furthermore, FA-AI values may serve as more valuable DTI parameters for assessing the integrity of the SCP and ICP, as they are correlated with one another and are continuously related to motor scores from baseline and 30 days later. Notably, the FA-AI of SCP measured during the subacute phase could independently contribute to predicting upper-limb movements 30 days later, whereas the FA-AI of ICP was associated with lower-limb movements but not with FA-AI of CST. This finding supports our hypothesis that the integrity of different motor-related CCP tracts plays distinct roles in motor recovery.
Reduced Integrity of DTCT and DSCT in Patients with MCA Infarction
The distal portion of the nerves undergoes progressive degeneration after transection of the nerves, after which the phenomenon was commonly referred to as Wallerian degeneration (WD). Ohara S. and Ikuta F. showed the early stages of WD in 1985 by studying distal segments of the phrenic nerve in mice after crush injuries. They found that axonal degeneration and myelin disintegration became evident from day 2 to day 6. In patients with cerebral infarction, WD can be detected by DTI within 7 days after infarction. Still, it is greatly affected by cell edema, cell edema resolution in 7-14 days, and FA value decreases most significantly. 17 In 14-30 days, FA value reduction gradually slowed down. In this study, the integrity of fiber tracts showed a similar trend of dynamic changes during stroke recovery. However, within the scope of this study, it is undetermined whether the FA decrease in the fiber tracts was affected by hemiplegic muscles or functionally impaired neurons due to ischemic stroke and subsequent transneuronal degeneration or both.
Originating in the dentate nucleus, DTCT receives afferent fibers from the premotor cortex, auxiliary motor cortex, and DSCT, which collectively target the ventral premotor area, dorsal premotor cortex, auxiliary motor area, and primary motor cortex.18,19 In patients with supratentorial cerebral infarction, an afferent block occurs alongside reduced cerebellar activity, leading to diminished excitatory output from the cerebellum to the affected cortex. 20 This process occurs via reciprocal dentate thalamocortical pathways and is accompanied by retrograde axonal degeneration. Buijink et al 21 demonstrated the retrograde degeneration of axons traversing the stroke lesion following thalamotomy, thereby supporting this hypothesis.
In healthy individuals, DSCT is responsible for transmitting proprioceptive information from the lower limb muscles to the ipsilateral cerebellum via ICP, playing a crucial role in controlling posture and movement.14,20 In patients with cerebral infarction, CPCT is impaired in the output of motor commands from the sensorimotor cortex to the contralateral cerebellum, and the peripheral proprioceptive input to the cerebellum in hemiplegic limbs is inadequate. 22 Finally, the afferent stimulation of ICP to the cerebellum is diminished, resulting in adverse changes in the spinal cerebellar pathway, manifested as significantly reduced FA value and FA-AI of ICP.
The CPCT originates from multiple regions of the cerebral cortex, including the sensorimotor cortex, and serves as the primary input pathway of the cerebrum (via MCP) to convey information regarding the coordination of autonomous motor activities to the dentate nucleus. 23 In this study, the structural integrity of the MCP remained unchanged, corroborating previous findings.5,11,12 This stability may be attributed to the fact that the CPCT, as a descending afferent pathway, encompasses frontal pontine tracts and occipital-temporal-parietal pontine tracts. Consequently, the reduction of certain fibers does not reach a threshold that would significantly impact the results, and the CPCT does not target the motor cortex, thereby rendering it less susceptible to cortical excitation. 12 It may also be due to the fact that the average FA value of the PMJ may not adequately represent the integrity of the CPCT pathway as a whole, especially the part between the cortical region and the pons.
Correlation Between FA Asymmetry Index of CCP and Motor Function Injury
The results of this study showed a positive correlation between the FA-AI of SCP in the subacute phase and FMA-UE in both the baseline and 30 days later, confirming the previous findings of Yoo et al 7 based on 25 patients with subacute MCA infarction. Previous studies that conducted a 12-week longitudinal follow-up of 18 patients with acute subcortical infarction indicated that the gradual increase in the FA value of the contralateral frontal cortical-thalamic junction positively correlated with the FMA. 24 Furthermore, recent studies have shown that intermittent theta-burst stimulation of the dentate nucleus in the contralateral cerebellar hemisphere enhances vision-motor learning and reduces spasticity in the affected upper limb of patients experiencing subacute to chronic supratentorial stroke.25,26 Li et al 27 studied the relationship between the change of FA value of CCP and motor rehabilitation after repetitive transcranial magnetic stimulation (rTMS) treatment in MCA infarction and found that the contralateral FA value of SCP increased after rTMS treatment, which was related to the FMA-UE.
In this study, the FA-AI of SCP and ICP were positively correlated with both the baseline and FMA-LE 30 days later. Soulard et al 28 conducted a longitudinal evaluation of the correlation between gait recovery over 2 years and the FA value of bilateral SCP in 29 patients with subacute cerebral infarction accompanied by gait disorders. They found that gait recovery gradually improved over time, reaching a plateau around 6 months, and that this recovery was significantly correlated with the FA value of the contralateral SCP. This suggests that the contralateral SCP may be a candidate imaging marker for assessing gait disorder recovery. Kim et al 13 studied the correlation between Functional Ambulation Category (FAC) and FA-AI of ICP in 23 patients with cerebral infarction accompanied by gait disorder and 23 normal subjects and found that FA-AI of ICP was positively and independently correlated with FAC at both admission and discharge. Additional studies have also shown that the FA change of contralateral ICP is associated with the change of FMA-LE of the patient’s affected limb, and the low FA-AI of ICP reflects the degree of degeneration, indicating poor walking function and severe weakness in the lower limb.14,29 The above findings provide strong support for the results of this study.
Notably, these studies found no association between the FA-AI of MCP and either the limb’s strength or general motor function, which aligns with the present study’s findings.7,11,12 However, other studies have demonstrated a significant correlation between FA-AI in MCP and fine motor skills, as measured by the 9-hole nail test or the Jepsen hand function test.5,30 Kim et al, 9 based on a study of 33 stroke patients and 17 healthy controls, found that MCP was significantly positively correlated with FA-AI of CST in patients with left and right hemisphere lesions. In contrast, FA-AI of MCP was negatively correlated with the muscle scale of the ankle. It is suggested that changes in CPCT depend on CST’s integrity and help predict motor function. All these findings indicate that the structural integrity of CPCT is poorly correlated with the general motor output of the limb. However, it may be associated with the fine motor skills of the hand. 30
Additionally, it has been reported that the FA value of the CST strongly predicts motor outcome after stroke. 31 In this study, although the FA value of the affected PMJ was lower than that of the unaffected, the FA laterality index of the PMJ was not correlated with FMA. The results of this study may be because all lesions involved the right CST, and there was no heterogeneity. It may also be that we did not follow up enough time and did not show an adequate level of recovery.
FA Asymmetry Index of CCP Predicts Upper and Lower Limb FMA
Whether the FA-AI of SCP alone is an independent variable model or the FA-AI of PMJ, SCP, and ICP interaction model, SCP shows good prediction capability for FMA-UE during the 30 days later. Previous research by Yoo et al 7 also found that SCP’s FA-AI as an independent variable model could better predict FMA-UE outcomes 6 months post-stroke. Furthermore, a study involving 18 patients with chronic supratentorial cerebral infarction revealed a connection between cortical excitability and corticocerebellar connections, particularly concerning FA values and FA ratios in SCP. 12 As a key component of DTCT, the thalamus acts as an essential relay station for processing sensory information beneath the cortex. It receives sensory signals from the spinal cord, brainstem, and the upper limbs and hands, which are subsequently processed and projected to the cerebral cortex to facilitate the regulation of movement. 32 Brain network studies have also confirmed that, under normal conditions, the basal ganglia and cerebellum activate concurrently during task performance. 19 The reduced structural integrity of the DTCT leads to impaired transport of ascending tracts and reduced excitability of cortico-cortical connections, thereby disrupting neuroplasticity within the cortical region and inhibiting upper limb movement and relearning following a stroke.
In our results, we found that both SCP and ICP’s FA-AI were able to predict FMA-LE outcomes in univariate models. In the intercept model of PMJ, SCP, and ICP, the ICP was identified as an independent influencing factor for FMA-LE, demonstrating greater predictive power compared to SCP. The non-conscious proprioceptive and tactile information from the lower extremities and lower trunk is transmitted to the cerebellum via the anterior and posterior tracts of the ICP. 13 Consequently, inadequate proprioceptive stimulation resulting from lower limb weakness and compromised walking ability may disrupt proprioceptive feedback from the hemiplegic muscles to the cerebellum. Increasing evidence suggests that non-invasive peripheral nerve or cerebellar stimulation, as well as deep cerebellar stimulation (including excitatory intermittent θ burst stimulation and inhibitory repetitive transcranial magnetic stimulation), can enhance movement and walking function in stroke patients with hemiplegia.20,33-35 Therefore, we can infer that the ascending cerebellar tract (DTCT and DSCT) may play a crucial role in promoting the excitability of the cerebellar cortical motor cortex, thereby facilitating appropriate motor output.
This study did not find any relationship between MCP integrity and motor function, which aligns with the findings of Yoo et al 7 However, previous research has indicated that the integrity of MCP is associated with fine hand activities.30,36 In terms of lower limb function, Wang et al 37 found that the FA ratio of MCP was more significantly correlated with the scores of 1-year paresis grading (PG), lower limb PG, upper limb PG, and FAC. This suggests that distal CPCT injury may be more closely related to gait recovery following hemiplegia. These findings may be attributed to the fact that the FMA score, while building a broad assessment system, has limitations in accurately capturing specific motor functions and needs to be further evaluated for fine hand movements and gait function in the future.
Limitations
In this study, the main method we used for DTI analysis (ROI-based analysis), although simple and clinically applicable, has some drawbacks. This method calculates the average value of fibers in every direction within the target area, thereby neglecting the influence of cross-fibers on the results. For example, although the SCP serves as the primary efferent pathway from the cerebellum to the thalamus, it also encompasses afferent and efferent pathways targeting other regions, such as the cerebellar tract and the spinocerebellar anterior tract. 38 To mitigate result bias, we have selected brain regions where the target fibers are densely concentrated as ROIs and conducted repeated measurements (2 times by 2 different doctors). Future studies may improve the accuracy of these findings through other DTI analyzing techniques, such as fiber tracing.
Conclusions
Our findings indicate that the structural integrity of the motor-related CCP measured by DTI in patients with subacute MCA infarction may serve as a significant imaging predictor for the outcome of motor recovery. Notably, the structural integrity of the cerebellar ascending fibers is more closely related to motor outcomes. Specifically, the FA-AI of DTCT (SCP) measured during the subacute phase independently predicts upper limb motor function, whereas the FA-AI of DSCT (ICP) correlates with lower limb motor recovery. These results support our hypothesis regarding the distinct roles of different motor-related CCP tracts in facilitating motor recovery.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683251351879 – Supplemental material for Prediction of Motor Recovery after Subacute Cerebral Infarction: Role of Corticocerebellar Pathway Integrity
Supplemental material, sj-docx-1-nnr-10.1177_15459683251351879 for Prediction of Motor Recovery after Subacute Cerebral Infarction: Role of Corticocerebellar Pathway Integrity by Jing Liu, Yi Shan, Bi-Xiao Cui, Shao-Zhen Yan, Lin-Lin Ye, Lei Cao, Miao Zhang and Jie Lu in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors thank Dr. Boyan Xu (MR Research, GE Healthcare, Beijing, China) for supporting the data analysis.
Author Contributions
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by the National Natural Science Foundation of China (Grant No. 82402499) and the Huizhi Ascent Project of Xuanwu Hospital (HZ2021ZCLJ005).
Disclosures
None.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
