Abstract
Background:
The peripheral nervous system (PNS) exhibits remarkable regenerative capability after injury. PNS regeneration relies on neurons themselves as well as a variety of other cell types, including Schwann cells, immune cells, and non-neuronal cells.
Objectives:
This paper focuses on summarizing the critical roles of immune cells (SCs) in the injury and repair processes of the PNS.
Results:
During peripheral nerve injury, macrophages infiltrate the site under the induction of various cytokines, primarily accumulating at the dorsal root ganglia (DRG) and the nerve distal to the injury site, with only a small number detected at the nerve proximal to the injury site. The phenotype of macrophages during injury remains controversial, but recent single-cell sequencing analyses may provide new insights. In peripheral nervous system injury, macrophages participate in Wallerian degeneration as well as in the reconstruction of nerve bridges and angiogenesis during axonal regeneration. Neutrophils appear early in the injury process and are primarily present at the injury site and the distal segment. After peripheral nervous system injury, immature neutrophils from the peripheral blood play a major role. Although lymphocytes constitute only a small fraction compared to macrophages and neutrophils after peripheral nervous system injury, they still play important roles, including Treg cells, B cells, and NK cells. A large number of immune cells accumulate at the injury site, contributing not only to Wallerian degeneration but also to axonal regeneration.
Conclusion:
In conclusion, this paper summarizes current knowledge regarding the mechanisms of immune cell infiltration after PNS injury, providing new insights for future research on the role of immune cells in peripheral nerve injury.
Keywords
Introduction
Unlike damaged neurons in the central nervous system that typically cannot regenerate, axons in the peripheral nervous system (PNS) can regenerate after injury. The peripheral nerve has a connective tissue sheath, primarily composed of epineurial mesenchymal cells, with nerve bundles inside, each bundle being surrounded by specialized perineurial mesenchymal cells, and the Schwann cells (SCs) within the bundles wrapping around the axons, while the SCs are enveloped by endoneurial mesenchymal cells (Figure 1). 1
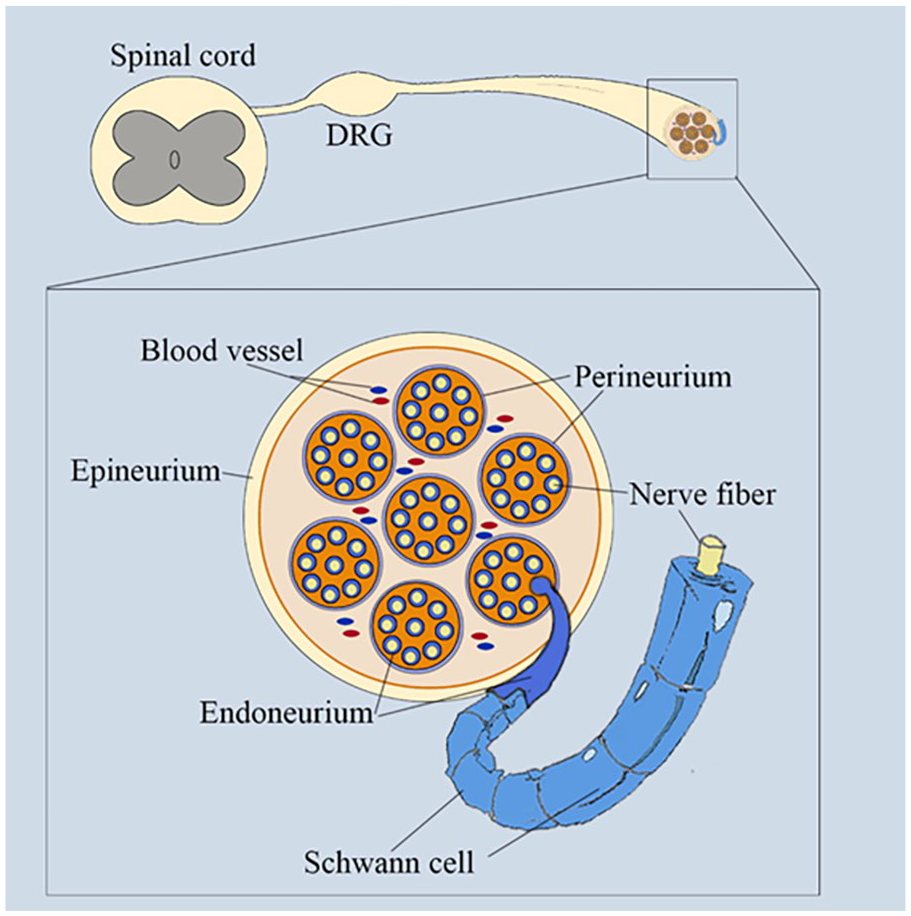
The structure of the sciatic nerve.
After the axonal injury, both myelinating and non-myelinating SCs undergo extensive reprogramming to promote and guide axonal repair. During demyelination, SCs inhibit expression genes of promyelinating genes, such as Erg2 or Krox20, as well as myelin genes, including myelin basic protein, myelin protein zero, peripheral myelin protein 22, and myelin-associated glycoprotein (MAG), to promote SC detachment from the distal stump axons and demyelination, while also re-expressing molecules that were inhibited in immature SCs during the onset of myelination, including L1, NCAM, p75NTR, and glial fibrillary acidic protein.2,3 After peripheral nerve injury, repair phenotype SCs mainly participate in: (1) SC autophagy, myelinating SCs initiate the breakdown of their own myelin4-6; (2) promote neuronal survival and axon regeneration, such as through factors GDNF, artemisinin, BDNF, NT3, NGF, VEGF, erythropoietin, trophic factors, upregulation of p75NTR and N-cadherin proteins; (3) promote macrophage invasion and activation, vascular formation, and myelin breakdown, such as upregulation of TNFα, leukemia inhibitory factor Il-1α (LIF Il-1α), Il-1β, LIF, and monocyte chemoattractant protein-1 (MCP-1, also known as chemokine [C–C motif] C–C chemokine ligand 2 [CCL2]) genes7-9; (4) participate in the formation of nerve bridges, in cases of nerve transection, c-Jun, SRY-box 2, and neuregulin 1 regulate SC morphological changes and axon-glial interactions, promoting the formation of nerve bridges and axon regeneration pathways; and (5) participate in remyelination of regenerating axons, such as upregulation of zinc finger E-box-binding homeobox 2, nuclear factor κB, and histone deacetylase 1 and 2 proteins.10-14
In addition to the synergistic effect of SC on regeneration, injured neurons themselves also have a strong regenerative response to regeneration. After peripheral nerve injury, genes related to pain perception in the dorsal root ganglia (DRG) decrease, including Trpv1, Scn9a, Scn10a, and Scn11a, but P2rx3 is upregulated, and the downregulation of P2rx3 can reduce neuropathic pain behavior. 15 In addition, there is an upregulation of inflammation-related genes associated with pathological pain, including Csf1 and Fam19a4, and an upregulation of the expression of genes related to axon regeneration, such as Atf3, Sox11, Sprr1a, and Gap43. 15 CCL2 in damaged DRG neurons can promote neuropathic pain through upregulation of ELF2-NIS-lncRNA-CCL2 axis expression, and it binds to the receptor CCR2. 16
In fact, in addition to the synergistic effect of SCs and injured neurons themselves, successful axon regeneration also depends on the coordinated efforts of various immune cells, which release extracellular matrix molecules, cytokines, and growth factors that support axon regeneration.17,18 Many studies have reported the involvement of immune cells in axon regeneration after peripheral nerve injury. In this review, we summarize the roles of immune cells, including macrophages, neutrophils, and lymphocytes, in peripheral nerve injury, aiming to better understand the role of immune cells in peripheral nerve injury.
The Biology of Macrophages After PNS Injury
The Localization of Macrophages in PNS Injury
Macrophages are a type of mononuclear phagocyte, typically originating from precursor cells in the bone marrow. 19 Monocytes originate from monocyte precursors in the bone marrow, mature, enter the bloodstream and tissues, and after antigen exposure, migrate to lymph nodes or differentiate into macrophages in tissues. According to the traditional Th1/Th2 dichotomy, macrophage phenotypes can be classified into pro-inflammatory and neurodegenerative (M1) and anti-inflammatory and pro-repair (M2) phenotypes, with M2 macrophages further divided into 4 different subtypes, including M2a, M2b, M2c, and M2d.20,21 LPS and IFNγ can induce the activation of M1 macrophages, which express inducible nitric oxide synthase (iNOS) and secrete TNFα, IL-6, ROS, IL-1β, NO, and matrix metalloproteinases (MMPs), while IL-2 and IL-4 can promote the activation of M2 macrophages, which express arginase-1 (Arg1) and secrete IL-10, TGF-β, and extracellular matrix (ECM).22,23 In addition to the M1 and M2 macrophage classifications, macrophages in most tissues, including the nervous system, can be divided into 2 types: resident macrophages and infiltrating macrophages. 24
After sciatic nerve transection, macrophages accumulate in the distal segments on day 3, with higher concentrations in the epineurium than in the endoneurium, reaching a peak around day 14, at which point the concentration in the epineurium drops below that in the endoneurium. 24 Additionally, a small accumulation of macrophages is also observed in the proximal end. 25 On day 4, macrophages accumulate in the lumbar DRG and can still be detected on day 32. 26 However, Kwon et al 27 reported that the number of macrophages in the DRG gradually increased from day 7 and continued to increase until day 28. Following sciatic nerve crush injury, flow cytometry revealed an increase in Monocytes/Macrophages (Mo/Mac) at the site of nerve injury and in the distal segment starting on day 1, peaking on day 3, and decreasing by day 7, but still higher than in the control group. In the proximal segment of the sciatic nerve, Mo/Mac did not increase on days 3 and 7, while in the DRG, Mo/Mac peaked on day 3, with no significant changes on days 1 and 7 compared to the control group. 28 Notably, despite the rise in the number of macrophages in the peripheral axons and DRG, the source of macrophages in the 2 was different. After injury, macrophages in axons are mainly derived from circulating monocytes in blood, 29 while resident macrophages are mainly used in DRG.28,30
Macrophage accumulation in peripheral axons and DRG is associated with enrichment of monocyte chemoattractant protein-1, LIF, IL-1α, IL-1β, and pancreatitis-associated protein III (PAP-III).29,31-34 It is noteworthy that, in addition to peripheral neurons and SC being able to produce CCL2, macrophages are both senders and receivers in the CCL signaling pathway, further promoting the recruitment of Mo/Mac, but CCL2 is not essential for macrophage accumulation, myelin clearance, and axonal regeneration in the PNS.30,35,36 Furthermore, there is substantial evidence that macrophage infiltration involves more than just these factors. ECM protein type VI collagen regulates peripheral nerve regeneration by modulating macrophage recruitment and polarization. 37 Galectin-1 promotes macrophage accumulation and enhances monocyte migration through the p44/42 MAP kinase pathway.38,39 Complement depletion can inhibit macrophage infiltration after sciatic nerve injury. 40 Mouse TLR2, TLR4, and MyD88 can inhibit the production of CCL2 and IL-1β, as well as macrophage infiltration. 41 The TLR4-S100A8/A9 signaling pathway plays an important role in monocyte/macrophage recruitment after nerve injury. 42 Macrophage infiltration and the production of pro-inflammatory cytokines TNF-α and IL-6 are reduced in P-selectin-deficient mice. 43 Intercellular Adhesion molecule-1 knockout inhibits macrophage infiltration after sciatic nerve transection injury. 44 Lack of prostaglandin receptors reduces the number of macrophages, while local administration of prostaglandin receptor agonists promotes the accumulation of IL-1β and prostaglandin receptor-expressing cells at the injury site. 45 Serum amyloid A (SAA) participates in the accumulation of macrophages in injured nerves through the IL–6–SAA–CCL2 signaling pathway. 46 These results demonstrate the diversity of macrophage infiltration pathways and indirectly prove the importance of macrophages in peripheral nerve injury.
Macrophage Phenotype in PNS Injury
Macrophages exhibit different phenotypes in peripheral nerve injury. Nadeau et al 47 found through flow cytometry that after sciatic nerve ligation, the number of M1 macrophages peaked at 1 to 2 days and gradually decreased by day 7, while the number of M2 macrophages gradually increased and peaked at day 3. Tomlinson et al 48 found that following sciatic nerve transection, the M1 phenotype marker iNOS was strongly expressed at day 3, decreased by day 5, and continued to decline until day 28, while the M2 marker Retnla increased from day 5 to 14, but Chi3l3 and Arg1 decreased from day 5 to 14. These results suggest that M1 and M2 may be regulated by different mechanisms and coexist in the injured sciatic nerve.
Ydens et al 49 found that after sciatic nerve axotomy, M1 markers such as iNOS, IL-12p40, and IFNγ were not induced at any time point (3, 7, and 14 days) in the distal segment of the sciatic nerve, while M2-related genes Arg1 and Ym1 were significantly induced and peaked at 1-day post-axotomy, returning to baseline by day 7. Kalinski et al 28 performed single-cell RNA sequencing (scRNA-seq) of the injured site and distal segment of the sciatic nerve at least 3 days after nerve crush and identified 5 macrophage clusters, referred to as Mac1-5. Cells within clusters Mac1-3 expressed high levels of Rbpj, which is required for M2 macrophage polarization and mediates expression of M2 gene subgroups. 50 Mac4 cells expressed transmembrane glycoprotein NMB (Gpnmb), a negative regulator of inflammation that has a protective effect after tissue injury. 51 Furthermore, Arg1 was mainly expressed in some cells within Mac1, Mac3, and Mac4, limited to the compressed nerve site and barely detectable in the distal nerve stump, in contrast to F4/80 and CD68 macrophages found in the injured site and distal nerve. 28 Additionally, Mac1, Mac3, and Mac4 strongly express TRs Maf/c-Maf and Mafb/MafB, where c-Maf is crucial for acquiring an immune-suppressive phenotype, while MafB promotes macrophage reprogramming toward an M2-like state.52,53 Mac5 represents a group of blood-derived proliferative bone marrow cells with stem cell-like characteristics. 28 Zhao et al 54 analyzed the site of sciatic nerve injury and the distal segment at 1, 3, and 7 days post-crush using single-cell sequencing, revealing that macrophages strongly expressed Arg1 on days 1 and 3 post-injury, accompanied by elevated levels of the glycolytic regulator Hif1a, which returned to normal levels by day 7, with no involvement of inducible nitric oxide synthase 2 in this process. It is worth noting that although glycolysis is considered a key feature of M1 macrophages, Wang et al 55 demonstrated its significant role in M2 macrophage activation. These studies indicate that M2 macrophages play a dominant role in peripheral nerve injury, with no detection of M1 macrophages. One possible reason is that M1 macrophages occur earlier post-injury, as in the zebrafish model, monocytes infiltrate within 120 minutes after nerve transection, and macrophages begin engulfing residual axonal and myelin debris between 145 and 200 minutes. 56 Early M1 macrophages can transition to M2 macrophages through increased expression of arginase 1 (Arg1), IL-10, and TGF-β after engulfing neutrophils.57-59 Another reason is that in the damaged nerves, the inflammatory environment shaped by mesenchymal stem cells (MES) and nerve sheath fibroblasts (Fb) may enhance sustained phagocytic activity in macrophages by metabolizing arginine and ornithine derived from engulfed apoptotic cells into polyamines. 60 Furthermore, the inflammatory environment in damaged nerves may be shaped by (MES and nerve sheath Fb, while the lower immune gene activity exhibited by repair SCs indicates a less significant role in shaping nerve inflammation. 28
In conclusion, there is still debate about the phenotypic changes of macrophages after peripheral nerve injury. The macrophage phenotype is suitable for study in vitro, but it is not applicable in complex tissues. Mokarram et al 61 found that INFγ and IL4 could significantly change the polarization state of macrophages in vitro, but INFγ could not significantly increase the number of M1-type macrophages in injured sciatic nerve tissues. However, the research by Ydens et al 62 may provide new insights for us. They believe that using the M1/M2 phenotype classification of macrophages in vivo is not applicable. This is because, in the steady state, the macrophages located in the epineurium around the nerve bundle express high levels of Retnla (encoding Relma), Clec10a (encoding Mgl1), Cbr2, Mgl2, and low levels of Ccl12. MacsThe macrophages located in the endoneurium have low expression of Retnla, Clec10a, and high expression of Cbr2, Mgl2, and Ccl12. In the steady state, the macrophage in the epineurium already expresses the so-called “M2 markers” Relmα and Mgl1. After sciatic nerve crush injury, the resident macrophages in the nerve sheath show no obvious response to the nerve injury, while the resident macrophages in the endoneurium will generate a new group of macrophages in a self-renewing manner after the first day of injury, which may be the reason for the local proliferation of resident macrophages after injury. These endoneurial macrophages produce monocyte chemoattractants to recruit rapidly acquired Arg1, Chil3, and Vegf-producing monocyte-derived macrophages, which play a dominant role in the repair process. Interestingly, these monocyte-derived macrophages entering the endoneurial compartment already express Arg1, while Mo in the peripheral blood does not express Arg1,54,62 indicating a rapid transition of macrophages from blood to the endoneurial compartment. According to another study, there are 2 subsets of Mo in the blood: Mo1 (Cx3cr1hi, Csf1rhi, Ear2hi, Cd36hi, Ccr2neg, Ly6c2neg, and Nr4a1hi), Mo2 (Ly6c2hi, Chil3hi, Ccr2hi, and Ear2neg) and a Mac cluster (Prg4/proteoglycan4hi, Ccl24hi, Ltc4shi, and Fn1hi), among which Mo2 is more similar to the Mo in the nerve on the first day after sciatic nerve injury, indicating that they are the main source of Mo entering the nerve at the time of injury. It is worth noting that these Mo2 express low levels of Hif1a, Pfkl, Pgk1, Ldha, and Slc16a3 in the blood, but once they enter the injured tissue, they are activated and undergo rapid metabolic reprogramming to produce glycolytic energy. 54
Mechanisms of Macrophages Promoting Myelin Debris Clearance and Axonal Regeneration After PNS Injury
Peripheral nerve injury or metabolic and mechanical damage can induce various changes including axonal degeneration, demyelination, proliferation of glial cells, blood–nerve barrier disruption, and infiltration and activation of macrophages, collectively referred to as Wallerian degeneration. 21 After peripheral nerve injury, the removal of myelin debris is a critical step in peripheral nerve regeneration, as myelin fragments can express inhibitory molecules for axonal growth, such as NI250 (Nogo) and MAG.63-65 In addition, the recombination of damaged ECM is also a prerequisite for axon regeneration, and MMP are the main driver of ECM degradation. 66 Tissue inhibitor of MMPs (TIMP-1), secreted by macrophages and SCs, regulates MMP activity and prevents collagen IV degradation in the sciatic nerve basal lamina. 67
In the early stages of Wallerian degeneration, SCs play a critical role, while macrophages are the main cells involved in the clearance of myelin and axonal debris in the late stages of Wallerian degeneration (WD).68,69 Monocyte-derived macrophages are responsible for efficient demyelination and clearance during Wallerian degeneration, while resident macrophages are involved in axonal regeneration. 70 Macrophages play a role in demyelination and clearance of myelin debris. On the one hand, this requires the involvement of complement component 3 (C3) and its receptor, complement receptor 3 (CR3) on macrophages.71,72 On the other hand, IL-1β, TNFα, MCP-1, collagen VI, Nrf2, phospholipase A2, MMP-2, and MMP-9 are also involved in macrophage-mediated Wallerian degeneration. 21
During axon regeneration, fibronectin initially binds the proximal and distal nerve segments, followed by collagen secretion from Fb and laminin production from SCs. These ECM molecules orient longitudinally to form a tubular bridge, which reassembles the severed nerve ends. Subsequently, the lumen of the bridge can be filled with neurotrophic factors to promote vascular development, axon growth, and cell survival. 73
After sciatic nerve transection injury, macrophages are mobilized before SCs in the nerve bridge, participating in the reconstruction of the regenerative pathway. On the one hand, the upregulation of surface receptor Plexin-B2 guides them to avoid collision with growth cones, resulting in parallel alignment after collision, which helps align ECM deposition and direct SC migration to reinforce the regenerative pathway. 74 On the other hand, the hypoxic environment in the transected tissue triggers macrophages to secrete VEGF-A, inducing vascular polarization in the transected tissue. Subsequently, these newly formed blood vessels are used by SC as a guiding path for invasion and crossing the bridge, promoting axonal regeneration. 75 However, a recent paper by Talsma et al 76 suggests that macrophage depletion is not necessary for rapid regeneration after injury and that it may be the role of other cells to compensate for the role of macrophages. Apart from changes in macrophage protein expression regulation, alterations in macrophage metabolism are also crucial for axon regeneration after peripheral nerve injury. The loss of macrophage-specific monocarboxylate transporter (MCT) 1 results in impaired glycolysis and mitochondrial metabolism, increased pro-inflammatory expression in macrophages, and reduced phagocytic capacity, impairing axonal regeneration. 77 After sciatic nerve crush injury, macrophages not only upregulate hypoxia-inducible factor 1α and glycolysis-related enzyme gene expression to promote glycolysis to meet their bioenergy demands but also inhibit OXPHOS, promoting intracellular lactate accumulation. 54 The increase in lactate levels will enhance the phagocytic capacity of macrophages. 78 Furthermore, macrophages after injury cause a sharp increase in Slc16a3, which encodes MCT4 used to shuttle lactate out of cells, 54 Extracellular lactate has been shown to have immunosuppressive functions, promoting angiogenesis, axon growth, and neuronal health.79-81
The Biology of Neutrophils After PNS Injury
The Localization of Neutrophils in PNS Injury
There have been many reports about the distribution of neutrophils in the peripheral nerve: in the naive nerve, less than 1% of the nerve-resident immune cells express markers for granulocytes (GCs), monocytes, or B cells. 54 Zhao et al 54 performed single-cell sequencing analysis of the site of sciatic nerve injury and the distal segment, and found that the number of GCs significantly increased 1 day after sciatic nerve crush, decreased sharply on day 3 compared to day 1, and further decreased on day 7, with GCs mainly composed of neutrophils and a small number of eosinophils. Kalinski et al 28 found through flow cytometry that GCs were absent in the naive nerve, increased to 7800 ± 300 on the first day of injury, decreased to less than 1000 on the third day, and even fewer on the seventh day. In contrast, the number or composition of immune cells at the proximal nerve stump did not change significantly. In the DRG, although the number of GCs showed an increasing trend, no significant changes in the immune cell spectrum within the ganglia were observed except for Mo/Mac. However, another study found no neutrophils in DRG. 82 Nadeau et al 47 found through flow cytometry that neutrophils had already infiltrated the nerve at 3 hours and observed a peak of infiltration 12 to 24 hours after nerve ligation at the injury site and distal segment. In contrast, Yamamoto et al 83 found through H&E staining that neutrophils in the WD area began to accumulate at 6 hours post sciatic nerve crush, peaked at 12 hours, almost disappeared at 1 day, and only accumulated in the epineurium, not in the parenchyma. The discrepancy between the results of Yamamoto et al and the former may be due to the absence of neutrophils at the injury site and the histological judgment used by the former, while the latter used flow cytometry and single-cell sequencing.
Mechanisms of Neutrophils in PNS Injury
After nerve injury, neutrophil recruitment occurs early, while the infiltration of macrophages into the injured nerve becomes apparent from 2 to 3 days, peaking at day 7. 21 The role of neutrophils in peripheral nerve injury has been widely reported, for example, neutrophil peptide-1 (NP-1) derived from neutrophils is a member of a class of peptides called defensins, responsible for signaling related to wound and tissue healing in the body.84-86 NP-1 has been reported to promote sciatic nerve regeneration or clearance of axonal debris,22,85 likely stemming from the role of NP-1 in recruiting monocytes and macrophages. 87 In addition, treatment with the gut bacterial metabolite indole-3-propionic acid (IPA) increased the number of neutrophils in the DRG after sciatic nerve axonal injury, promoting axonal regeneration. 88 It is noteworthy that IPA treatment does not increase the number of neutrophils in the injured axon, and the literature has not yet explained how neutrophils promote axonal regeneration in DRG, which requires further study. In addition, neutrophils that promote axonal regeneration may be low expression Ly6G high expression CD14 (Ly6Glow CD14hi), because Sas et al89,90 reported that immature Ly6GlowCD14hi neutrophils stimulated axonal growth of primary DRG neurons and expressed high levels of Arg1, while mature Ly6GhiCD14low neutrophils lacked neuroprotective/neuroregenerative properties. This result is consistent with Zhao et al’s 54 study: they found that there were mainly 2 granulocyte clusters (GCs) in the blood, containing mature GCs (mGCs, Ly6ghi, Ngphi, Lcn2hi, Il1bneg) and immature GCs (iGCs, Cxcr2hi, Csf3rhi, Ly6glow, and Il1bhi). One day after the sciatic nerve crush injury, the GCs in the nerve were Csf3rhi and Ngplow, indicating that iGCs entered first. These iGCs already expressed high levels of Hif1a, Pfkl, Pgk1, Ldha, and Slc16a3 in the blood, which seemed to promote glycolysis and may produce lactate, and further increase the expression of these gene products upon entering the nerve.
In stark contrast, it has been reported that myelin clearance is unimpeded after macrophage depletion, while a compensatory increase in neutrophils, and depletion of neutrophils using Ly6G antibodies significantly inhibits myelin clearance.47,91 Additionally, Yamamoto et al 83 found that at the onset of sciatic nerve crush, neutrophils first accumulate in the epineurium of the WD region, at which point they do not recruit macrophage accumulation. Instead, neutrophils inhibit macrophage infiltration through NET formation, which seems to be a salvage process for the injury. Later, neutrophils disappear, and macrophages begin to accumulate from the epineurium to the parenchyma.
The Biology of Lymphocytes After PNS Injury
After peripheral nerve injury, there are also changes in the expression of lymphocytes. Kalinski et al 28 used flow cytometry to detect lymphocytes and found that only a few lymphocytes exist in the naive sciatic nerve. Compared with the control group, the number of lymphocytes in the injured site and distal end of the sciatic nerve increased significantly at 1, 3, and 7 days after injury, but there was no significant difference in the number of lymphocytes in DRG. Zhao et al 54 found through single-cell sequencing of the sciatic nerve that only a few T cells exist in the naive sciatic nerve, and they gradually increase after injury, accounting for approximately 10% of all immune cells by the seventh day, and the increase in T cells is mainly due to neural infiltration. In comparison to T cells, natural killer (NK) cells also gradually increase after injury, but are almost undetectable by the seventh day. Davoli-Ferreira et al 92 found, through flow cytometry, that after ligation of the sciatic nerve, no infiltration of regulatory T cells (Treg) cells was observed in the DRG, while at the injury site, the expression of T cells remained upregulated 14 days after the injury. However, compared to 7 days after the injury, the expression of T cells decreased. In contrast, the number of Treg gradually increased within 14 days after the injury.
In conclusion, we can understand that lymphocytes also participate in the regulation after peripheral nerve injury, and different lymphocytes play different roles. After transection of the sciatic nerve, activated T lymphocytes and B lymphocytes restrict motor recovery, while the lack of mature T and B lymphocytes improves the survival rate of motor neurons and myelin regeneration. 93 Depletion of B cells slows down macrophage infiltration and delays myelin clearance and axon regeneration. 71 After ligation of the sciatic nerve in T lymphocyte-deficient mice, neuropathic pain is reduced and recovery is faster. 94 In aged mice (20-22 months) after crush injury to the sciatic nerve, the enrichment and increase of inflammatory cytokines (including lymphotoxins) activated the NF–κB–CXCR13 pathway in the DRG, recruiting nearby CXCR5CD8 T cells. The binding of CXCR5CD8 T cells to MHC I-expressing sensory neurons activated caspase 3, damaging pAKT and pS6 signaling, and inhibiting axon regeneration. 95 Treg also play a positive role in peripheral nerve injury. After peripheral nerve ligation injury, although no Treg infiltration was observed in the DRG after peripheral nerve injury, Treg can inhibit the activation of resident macrophages in the DRG, reducing neuronal damage. 92 Further research is needed on how Treg inhibits the activation of resident macrophages in the DRG. Recruitment of Treg cells at the injury site can reduce the development of neuropathic pain, partly by inhibiting the recruitment and activation of CD4+ Th1 cells and the production of IFNγ, and partly through IL-10 secretion. 92 This result is consistent with the conclusion of the study by Austin and others that Treg can reduce the number of infiltrating T cells, macrophages, and antigen-presenting cells, improving the severity of the disease and mechanical pain hypersensitivity. 96 In addition, NK cells can promote Wallerian degeneration and reduce mechanical hypersensitivity after injury, partly due to the early induction of retinoic acid early inducible protein 1 (RAE1) mRNA expression in the DRG 3 days after crush injury to the sciatic nerve, but RAE1 protein aggregation can only be detected at the proximal end of the sciatic nerve. RAE1 protein, as a membrane-bound ligand for the activating NK cell receptor NKG2D, attracts NK cells. 97
Conclusions
Peripheral nerve injury involves significant biochemical changes in both the axon itself and the surrounding cells. Following peripheral nerve injury, although the neurons themselves showed enhanced neurite growth for a few hours, they no longer showed enhanced neurite growth after a few days. 98 The self-regulation of neurons alone is insufficient to promote self-repair, requiring coordinated regulation from surrounding cells. This review focuses on the role of the immune system in peripheral nerve injury, summarizing the localization and function of macrophages, neutrophils, and lymphocytes in peripheral nerve injury (Figure 2).
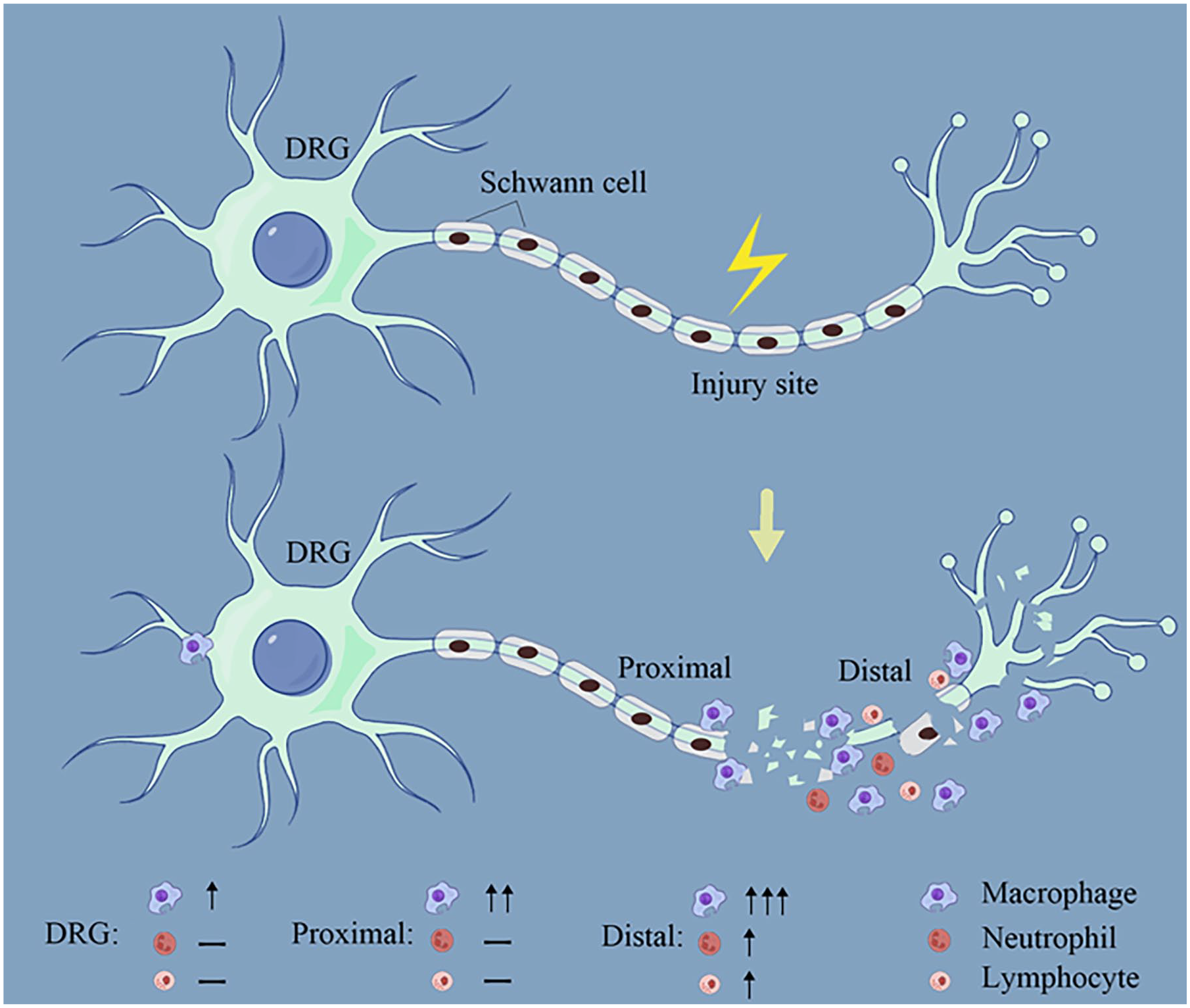
The hypothesized peripheral nerve diagram after axonal transection or crush shows that macrophages mainly accumulate at the distal axonal segment, with enrichment also seen at the proximal axonal segment and DRG, while neutrophils and lymphocytes only show a significant increase at the distal axonal segment, with no significant change at the proximal axonal segment and DRG.
Macrophages play a crucial role after injury, with their highest abundance found in the injured sciatic nerve, constituting 83% of all white blood cells. 54 After sciatic nerve injury, macrophages derived from peripheral blood play a predominant role in the injured sciatic nerve. The recruitment of these macrophages is maintained by multiple signaling pathways, suggesting that inhibiting a single pathway may not affect macrophage recruitment, highlighting their crucial role after injury. Macrophages primarily accumulate at the injury site and the distal segment, with a small accumulation in the DRG and proximal nerves, but their numbers decrease shortly after. In contrast to the sciatic nerve, macrophages in the DRG primarily consist of resident macrophages. Despite literature reporting M1 and M2 phenotypes in macrophages after sciatic nerve injury, single-cell sequencing studies reveal that the M1 and M2 phenotype distinction does not apply universally. Macrophages in the sciatic nerve exist in the epineurium and endoneurium, exerting distinct roles after injury. Upon entering the injury site, macrophages, on one hand, participate in Wallerian degeneration, responsible for the clearance of myelin sheaths and axon debris, and on the other hand, facilitate nerve repair and regeneration through protein regulation and metabolic changes.
Compared to the recruitment of macrophages, neutrophils enter the injured site earliest after sciatic nerve injury, primarily enriching at the site of nerve injury and the distal segment, while there are almost no neutrophils in the proximal segment and DRG. However, the studies indicate that increasing neutrophil numbers in the DRG by applying IPA can promote axon regeneration. After sciatic nerve injury, blood neutrophils exhibit 2 main phenotypes: mature neutrophils and immature neutrophils, with immature neutrophils being the first to enter the injured site and exert their effects. There are not many reports on the role of neutrophils in Wallerian degeneration and nerve repair and regeneration. According to current literature, neutrophils may promote Wallerian degeneration. However, the role of neutrophils in promoting nerve repair and regeneration, especially regarding whether neutrophil depletion affects axon regeneration, is yet to be established. Another study suggests that NP-1 expressed by neutrophils can promote axon growth, but NP-1 can recruit monocytes and macrophages, indicating that the promotion of axon growth by neutrophils may depend on the actions of monocytes and macrophages.
Despite the low proportion of lymphocytes in sciatic nerve injury, they still play a crucial role. T lymphocytes, such as CD8 T cells, contribute to neuropathic pain and hinder nerve recovery, while Treg cells regulate the aggregation of these cells, reducing the development of neuropathic pain. Endogenously produced antibodies by B cells can facilitate rapid clearance of myelin debris and effective axon regeneration after nerve injury. In addition, NK cells can promote Wallerian degeneration and alleviate mechanical hypersensitivity after injury.
In summary, both innate and adaptive immune responses, after peripheral nerve injury, interact or counteract each other, playing a crucial role in promoting the regeneration of the peripheral nerves.
Footnotes
Acknowledgements
Not applicable.
Author Contributions
Yiming Xia: Investigation; Methodology; Resources. Min Cai: Formal analysis; Investigation; Methodology; Validation. Yiyue Zhou: Methodology; Software; Visualization. Yi Yao: Data curation; Methodology; Visualization. Maorong Jiang: Data curation; Methodology; Resources; Software. Dandan Gu: Data curation; Software; Validation. Dengbing Yao: Conceptualization; Supervision; Writing—review & editing.
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the National Natural Science Foundation of China (Grant No. 31971277; 31950410551; Dengbing Yao); Scientific Research Foundation for Returned Scholars, Ministry of Education of China (Dengbing Yao); a project funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD; Dengbing Yao); and Jiangsu College Students’ Innovation and Entrepreneurship Training Program (202213993005Y; Yi Yao).
Consent for Publication
All the authors of this manuscript have approved the manuscript and consented to its publication.
Ethical Approval and Consent to Participate
Not applicable.
