Abstract
Introduction
Wearables have emerged as a transformative rehabilitation tool to provide self-directed training in the home. Objective. In this study, we examined the efficacy of a novel wearable device, “Smart Reminder” (SR), to provide home-based telerehabilitation for hemiparetic upper limb (UL) training in persons with stroke.
Methods
Forty stroke survivors from community support groups were randomized (stratified by the period after stroke onset and impairment severity) to either the SR group or the sham device group. Participants received either 20 hours of telerehabilitation using the SR device or training with pictorial handouts and a sham device over 4 weeks. In addition, all participants wore a standard accelerometer for 3 hours each day, 5 times a week, outside the prescribed training. Participants were assessed by a masked assessor at baseline, post-intervention (week 4), and follow-up (week 8). The outcome measures included Fugl-Meyer Assessment for Upper Extremity (FMA-UE), Action Research Arm Test, Motor Activity Log, muscle strength, active range of motion and amount of movement of the UL, and compliance rate of training.
Results
The SR group improved substantially in their FMA-UE scores after treatment (mean difference = 2.05, P = .036) compared to the sham group. Also, adherence to the training using the SR device was significantly higher, 97%, than the sham group, 82.3% (P = .038).
Conclusion
The 4-week telerehabilitation program using a “SR” device demonstrated potential efficacy in improving FMA-UE scores of the hemiparetic upper limb. However, it did not significantly enhance the performance of the affected limb in daily activities. The trial was registered on ClinicalTrial.gov (URL: http://www.clinicaltrials.gov) with the identifier NCT05877183.
Introduction
Stroke is a leading cause of disability, significantly impacting the quality of life of stroke survivors and imposing substantial socioeconomic burden on families and society. Upper limb (UL) impairment is a major contributor to the loss of independence in stroke survivors, 1 with its recovery rate generally poor. 2 A study by Kwakkel and Kollen 3 indicated that 30 to 66% of stroke survivors had little to no hand function. Current evidence recommended that more than 20 hours of training is needed for optimal rehabilitation of the hemiparetic arm after a stroke, 4 a requirement that is often unsustainable due to constrained health services.5,6 Furthermore, clinic-based treatment lacks ecological validity of how the patients might transfer their learned skills in their homes and communities. 7 More frequent and shorter intervals of exercise and motor skill training in the everyday environment may be more effective than individual clinic sessions.7,8 Therefore, self-directed programs that allow patients to practice their skills at home after clinic treatment are increasingly favoured.9,10
Wearable technologies, or wearables, have emerged as a transformative tool in rehabilitation, serving as activity trackers, reminders, and connectors. These devices often include sensors that enable remote clinical monitoring outside traditional healthcare settings. 11 This unique feature in wearables allows them to be a promising tool for self-directed training in the home environment.12,13 Wearables are electronic devices worn outside the body to track activity without restricting movement.12,14,15 Some wearable devices can be connected to smartphones, tablets, and computers, facilitating virtual reality (VR) training and telerehabilitation.16-18 Others provide augmented feedback essential for motor relearning, promoting the recovery of the hemiparetic UL.19-21 Augmented feedback is particularly beneficial for some stroke survivors with impaired intrinsic feedback mechanism (ie, proprioception and sensation).22,23 It can be delivered in single or multiple modes through visual, auditory, and haptic input.24,25 Some wearable can emit sound and vibration to encourage the use of the affected arm during daily activities in stroke survivors10,26 or integrate with VR systems to provide performance feedback to the users.16,18,27
In home-based stroke rehabilitation, wearable technologies are applied through VR, robotic, somatosensory stimulation, and accelerometer-based feedback. 13 Studies have demonstrated that the potential of wearables as an upper limb rehabilitation tool in improving motor performance of the hemiparetic UL.10,16,26,28-32 However, the earlier studies referenced 16,26,28-31 utilized a wide variety of interventions, such as VR, accelerometer-based feedback and somatosensory stimulation. Previous reviews21,23 highlighted that there was insufficient high-quality evidence concerning the use of wearable devices for home-based rehabilitation. Most research focused on the assessment capabilities of wearable sensors rather than their treatment potential.12,15,21 Therefore, the effect of wearable-based interventions in rehabilitating the hemiparetic ULs at home remains unclear in the current literature,15,21,32 necessitating the need for further research to establish the clinical impact in home-based rehabilitation.15,23
Our study introduces a wearable device called “Smart Reminder” (SR) with 2 unique new features not found in previous wearable studies.10,18,26,28-30,32,34,35 Unlike earlier studies10,26,34,35 that only used an accelerometer, the SR wearable has an accelerometer and gyroscope sensors to track rotational movement. The gyroscope measures angular acceleration exclusively and is useful in measuring rotational projections such as range of motion (ROM).36,37 In addition, the SR device connects to a smartphone telerehabilitation application (mobile application [app]) to offer interactive therapy and remote monitoring. The telerehabilitation app displays the prescribed exercises, the user’s real-time movements (ie, ROM of the UL), and completed repetitions.
The SR was developed based on our previous “Remind to Move” (RTM) concept,26,32 which prompts patients to use their affected arm for activities of daily living. Similar to RTM, the SR device vibrates and beeps to remind users to perform prescribed exercises at scheduled times and regular intervals customized by the therapist. Furthermore, the telerehabilitation app of the SR includes exercise videos featuring simple functional tasks like wiping a table, pouring water, and picking up a cube, etc., along with standard ROM exercises for the upper limb (see Supplemental S3). Notably, the SR intervention uses a smartphone as a portable visual display, a feature generally absents in the existing wearable-based VR studies according to our recent scoping review. 13 Smartphones are advantageous due to their portability and high global ownership rates. 38 Our preliminary studies on SR wearable17,39 have shown promising results on the usability and feasibility of the SR wearable to provide home-based UL telerehabilitation. However, a large-scale clinical trial is needed to confirm its efficacy.
In this study, we investigated the efficacy of an enhanced wearable device, SR,17,39 for home-based self-directed upper limb telerehabilitation in persons with stroke. The primary objective was to compare the effects of a 4-week SR telerehabilitation program to conventional training with a sham device on motor outcomes and arm use in the hemiparetic upper limb immediately after treatment. Additionally, we examined the long-term effects of SR telerehabilitation at a 1-month follow-up. We hypothesized that the multimodal augmented feedback (active ingredient) from the SR telerehabilitation (delivery mode) would be more effective than conventional training with pictorial handouts using a sham device (delivery mode) in promoting motor recovery and arm use of the hemiparetic UL in persons with stroke.
Methods
Study Design
This study was a single-blinded, randomized controlled trial. The Consolidated Standards of Reporting Trials (CONSORT) Checklist 40 guided this study (see Supplemental 1). Using block randomization, participants were randomly assigned to 2 arms—Group 1 (SR device group) or Group 2 (sham device group) by a non-team member. The randomization was stratified according to the participants’ baseline UL severity and stroke onset. A TIDieR checklist 41 was used in this study to detail the intervention, instrumentation, procedures, and outcome measures (see Supplemental 2).
Only the assessor who conducted outcome measures on participants was blinded to treatment allocation. The assessor was a qualified occupational therapist trained in using the outcome measure, not the attending therapist. Masking in treatment allocation for attending therapists and participants was not feasible due to the nature of the intervention.
Participants
Forty stroke participants were recruited from community self-help groups in Hong Kong using stratified convenience sampling from May to September 2023. The researchers screened the eligibility of the participants according to the inclusion criteria: (1) age >18 years, (2) unilateral hemispherical involvement, (3) diagnosis of stroke with onset >3 months, 42 (4) Functional Test for Upper Extremity-Hong Kong (FTHUE-HK) version 43 score between 3 and 6 (maximum of 7), (5) no complaint of excessive pain and swelling over the hemiplegic arm, (6) Modified Ashworth Scale ≤2, (7) Mini-Mental State Examination ≥19, 26 and (8) able to follow verbal instructions and 2-steps command in using the wearable device and smartphone.
Participants were excluded if they were: (1) participating in another similar form of experimental study during the study period, (2) had a history of botulinum toxin injection in the past 3 months, (3) had other significant UL impairment, that is, fixed contractures, frozen shoulder, and pain, (4) diagnosed with conditions that would interfere with device use, that is, visual impairment, active cardiac problems, and palliative treatment, and (5) not fully vaccinated against COVID-19. This study was approved by the Human Studies Committees of the Hong Kong Polytechnic University (HSEARS20230317001) and registered on ClinicalTrial.gov (URL: http://www.clinicaltrials.gov) with the identifier NCT05877183. Informed consent forms were signed by the participants before data collection. After completing the study, the participants were given a grocery voucher to compensate for their transportation expenses.
Sample Size Calculation
The sample size was calculated using the effect size of our previous feasibility study, 39 in which the effect size of the primary outcome measure (shoulder abduction ROM) was η2 = .193 using analysis of covariance (ANOVA). Considering that there would be 3 repeated measurements and an estimated 25% attrition rate, a sample size of 40 participants for 2 groups is needed to achieve 90% in power and Type I error set at a 0.05 significance level calculated using G*power, Version 3.1.9.7. 44 Therefore, a total sample of 40 participants was recruited in this study, with 20 per group.
Instrumentation
This study used 2 wearable devices—an SR and a triaxial accelerometer. The SR (Figure 1) is a lightweight wristwatch-like device measuring 6.5 cm × 6.0 cm × 2.5 cm and weighing 70 g, designed ergonomically with easily adjustable Velcro® straps. The SR had an in-built accelerometer, gyroscope sensors, and a rechargeable battery that could be used for up to 72 hours. The device vibrated and beeped to remind users to perform prescribed exercises at scheduled times and regular intervals, as customized by the therapist. Its Bluetooth function, connected to a telerehabilitation app on a mobile phone or tablet, allows participants to watch training videos, which include simple functional tasks and standardized upper limb ROM exercises (see Supplemental S3).
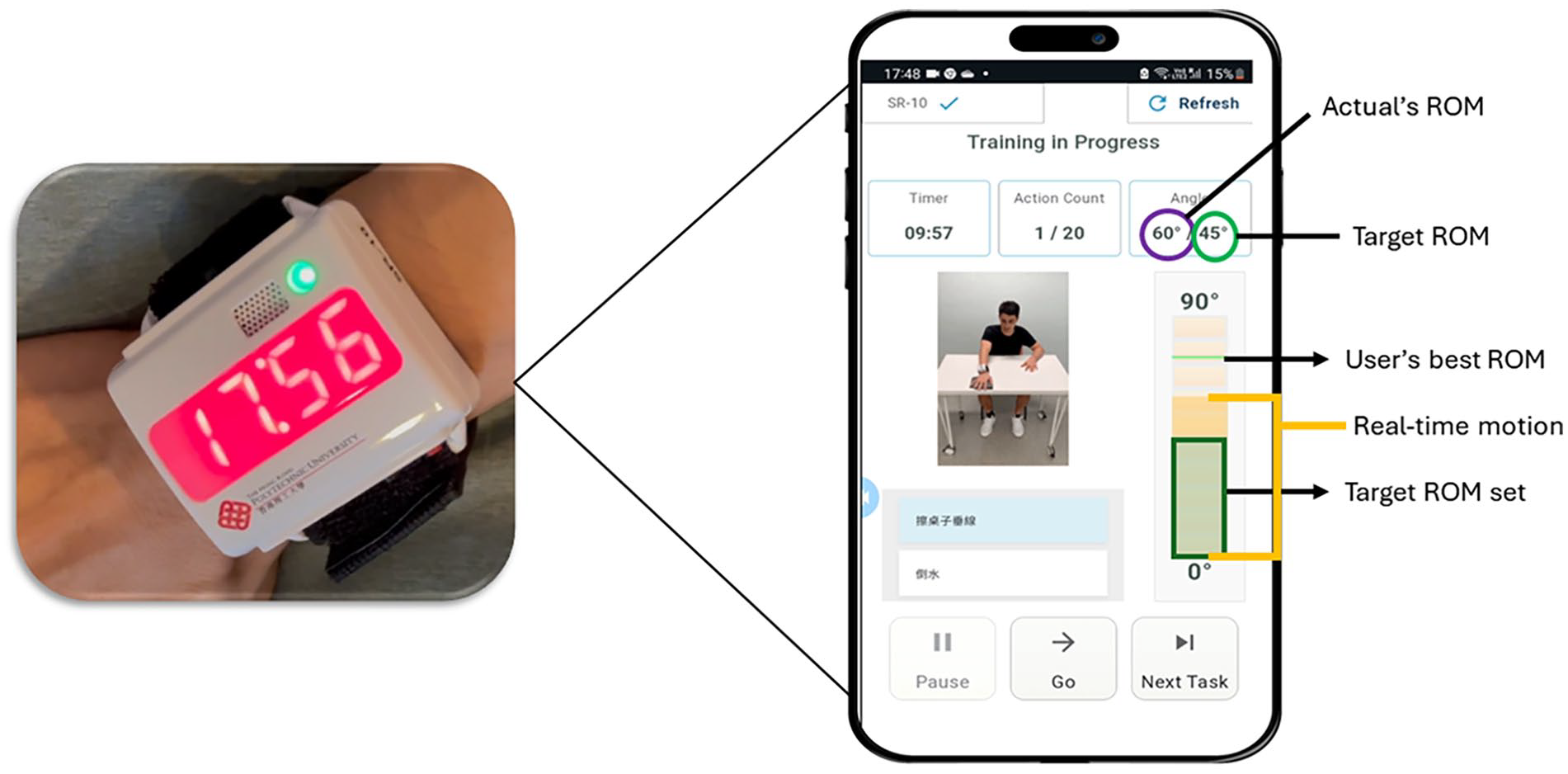
“Smart reminder” (SR) device.
During telerehabilitation, participants watched the prescribed exercise videos in the app on their mobile phones and performed the customized exercises accordingly. Concurrently, they received real-time feedback on their ROM performance from the app on their phones. The built-in sensors in the SR device detected and recorded the participants’ ROM and number of completed repetitions. This information was displayed on the mobile phone’s screen and uploaded to an encrypted web portal for the therapist to review remotely. The therapist could adjust the therapy parameters remotely based on the participants’ ROM performance and compliance rate. Some modifications on the current SR device were done after our usability study. 17 They included a user-friendly Velcro strap for single-handed use and added functional and fine motor tasks to increase exercise variety. One of the study’s aims was to objectively track the hemiparetic UL’s use outside of the “prescribed therapy” to encourage frequent use of the affected limb in the participants’ daily routine. One limitation of the current SR device was that it could not track the amount of UL movement outside the telerehabilitation sessions when the device was off. Therefore, another wearable device, a triaxial digital inclinometer accelerometer, WT901C, by WitMotion Shenzhen Co. Ltd., with a size of 5.13 cm × 3.6 cm × 1.5 cm, was used together with the SR in this study. This accelerometer served as the “sham device” in the study’s sham group. It resembled a wristband with a rechargeable battery that could last for 24 hours of continuous usage. The accelerometer tracked the participants’ arm movements but did not emit any reminder or connect to a telerehabilitation application on the mobile device. During arm movements, the accelerometer recorded acceleration in 3 orthogonal directions (x, y, z) 45 Additionally, the accelerometer had a sampling frequency range of 0.2 to 200 Hz and an acceleration measurement range of ±16g. There was a micro-standard deviation (SD) card in the accelerometer device for programming and recording of kinematic data.
Intervention Groups
Participants in the SR group wore the 2 wearable devices, the SR, and the triaxial accelerometer (the same one as the sham device group), during the 4-week intervention period. They used the SR device for telerehabilitation training and wore the accelerometer outside of the telerehabilitation training to monitor their daily arm movement. Participants were taught how to operate both devices and connect the SR device to a telerehabilitation app downloaded on their mobile phones. The attending occupational therapists prescribed appropriate exercises from the app tailored to the severity of the participants’ UL impairment. Also, the therapist set the frequency and duration of the exercise reminders emitted by the SR after discussion with the participants. The participants were instructed to engage in telerehabilitation training using the SR device for 1 hour per day, 5 times a week over 4 weeks. Outside the telerehabilitation session, the participants were asked to wear the triaxial accelerometer for 3 hours per day, 5 times a week and encouraged to use their affected arm in their functional activities over the intervention period. Previous studies26,31 suggested that wearing the wearable device for 3 hours was intensive and appropriate; hence, in this study we adopted the same wearing regime. The participants attended a 45-minute face-to-face session with the therapist at a local university laboratory once a week, as recommended from the findings of our previous usability study. 17 This interim review allowed tailored adjustments to the prescribed exercises based on participants’ progress and provided timely technical support. The therapist also reviewed the kinematic data from the SR device and accelerometer at least once before each weekly visit, ensuring continuous monitoring and timely adjustments to the therapy plan. During the visit, the therapist provided summary feedback on the participants’ performance.
The participants in the sham device group received conventional training using similar types of UL exercises adapted to the severity of their UL impairment as that of the SR group, but with the prescribed exercises presented as a pictorial handout (see Supplemental S2) rather than an in-app video. During the 4-week intervention, they were instructed to exercise for 1 hour per day, 5 times a week, and to record their exercise in an activity log. Additionally, they were asked to wear the triaxial accelerometer (“sham device”) during the prescribed 1-hour training and another 3 hours per day, 5 times per week outside training. Outside the prescribed training, they were encouraged to use their affected arm in their functional activities while wearing the accelerometer to track their arm use. Like the SR group, they also received a 45-minute consultation session at a local university laboratory once weekly with the therapist to modify the prescribed exercises according to their progress, except that no feedback was given on the amount of arm use.
Data Collection and Outcomes Measures
Participants were assessed by a blinded assessor at baseline before randomization (week 0), then at post-intervention (week 4), and at follow-up (week 8). The participants’ demographics, such as age, sex, the period after stroke onset, side of hemiparesis, hand dominance, and level of UL functioning based on the Functional Test of Hemiplegic Upper Extremity, Hong Kong (FTHUE-HK) version, 43 were collected at baseline. The FTHUE-HK version 43 is based on Brunnstrom’s developmental stages of stroke recovery and categorizes UL disability into 7 functional levels, with the highest level as the least severe. The primary outcome measures in this study assess the hemiparetic upper limb (UL) across various levels according to the International Classification of Functioning, Disability, and Health (ICF) 46 : impairment, activity, and participation. Impairment measures include the Fugl-Meyer Assessment for Upper Extremity (FMA-UE), active ROM (AROM) of shoulder, elbow, and forearm, and muscle strength of shoulder and elbow. Activity and participation are measured using the Action Research Arm Test (ARAT) and the Motor Activity Log (MAL), respectively. Secondary outcomes include the amount of UL tracked by accelerometer in daily routines and the compliance rate of training.
The FMA-UE measures the synergistic pattern and ability to make arm movements 47 and is recognized as the gold standard in stroke research. 48 The muscle strength of the hemiparetic elbow and shoulder of the participants was measured using a digital manual muscle tester (Nicholas manual muscle tester, Model 01160). The Nicholas manual muscle tester, a portable handheld dynamometer, measures force to 0.1 kg with a range of 0.9 to 199.9 kg. 49 It is ergonomically designed to support the tester’s hand and has a visible display to show the reading force output during a break test. 49 Additionally, a digital goniometer (Baseline 12-1027 Absolute Axis 360 Degree Digital Goniometer, Dedham, Massachusetts) was used to measure the active ROM of the hemiparetic shoulder (flexion/abduction), elbow, and forearm rotation (sum of supination and pronation).
Regarding the activity and participation measures, we assessed hemiparetic UL function using ARAT, the participants’ subjective and objective arm use of their hemiparetic UL in daily routine using MAL and the accelerometery arm movement, respectively. The subjective measure, MAL, a self-reported questionnaire, consists of 2 subscales to measure patients’ perceived Amount of Arm Use (MAL-AOU) and Quality of Arm Use (MAL-QOM). 50 Regarding the amount of movement, all participants wore the triaxial accelerometer on their affected arm for 3 hours per day, 5 times a week, to record the amount of arm movement. The raw acceleration data collected by the triaxial accelerometer was processed using 5 major steps described in Supplemental 4, S4 to calculate the amount of accelerometry arm movements, also termed “movement counts.” These 5 steps are frequency filtering, rectification, thresholding, downsampling, and activity accumulation. Firstly, a band-pass filter was applied with cut-off frequencies set at 0.25 and 2.5 Hz 51 to eliminate the effect of gravity and invalid activities such as tremors and to attenuate background signal noise. Rectification of the raw accelerations in the 3 axes, x, y, and z directions over the 3-hour wearing period was performed to convert into a single signal using Pythagorean mathematics (R = √[x2 + y2 + z2]). 45 Following the frequency filtering and rectification steps, upper and lower thresholds were applied to the rectified output. The upper threshold (set at 0.4g) removes outliners, such as instances of hitting, and the lower threshold (set at 0.09g) eliminates background noise in the time domain. Referencing previous literature,52,53 the output frequency was downsampled to 10 Hz to ensure consistent movement counting. For the last step, activity accumulation, the acceleration data was processed into “movement counts” using 1-second epoch time. 52 Lastly, the compliance rate of training for both groups was measured using the SR device’s data log for the SR group and the activity log for the sham group.
Data Analysis
Data analysis was conducted using SPSS software version 26.0 (SPSS Inc., Chicago, IL, USA), based on an intention-to-treat principle once the participants started treatment. The method of “Last Observation Carried Forward” (LOCF) was used if the drop-out rate did not exceed 10%. 26 Descriptive statistics on demographics and clinical scores of participants were presented in mean and SD, absolute number, or percentage. A comparison of baselines between the SR and sham device groups was performed using an unpaired t-test for normally distributed data, Mann–Whitney U test for non-normally distributed data, and Chi-square for nominal data. The treatment effect within each group (SR and sham groups) before/after treatment and at follow-up was assessed using single repeated measure of Multivariate Analysis of Variance (MANOVA) with Bonferroni post-hoc adjustment. For between-group differences, MANOVA was used to analyze the change scores of the outcome measures to compare the treatment effect of the telerehabilitation provided by SR and conventional training supported with a sham device. In the event of significant differences, Bonferroni post hoc test was performed.
The average movement counts of the participants’ hemiparetic UL per hour over the 4-week intervention period were calculated and compared between groups to assess the amount of arm movement in the participants’ daily routine. Lastly, the participants’ mean compliance rate of training was calculated using the percentage of the total completed exercise sessions over the prescribed session. Using 2-tailed analysis, statistical significance was set at a P value less than .05.
Results
Forty-eight stroke survivors from the community self-help groups in Hong Kong were screened. Forty participants met the eligibility criteria and were recruited and randomized into the SR and sham device groups. The remaining 8 stroke survivors did not fulfill the eligibility criteria because they did not meet the upper extremity functional levels (ie, FTHUE-HK levels 3-6), indicating that they had either mild or very severe UL disability. Two participants from the sham device group dropped out before the completion of the intervention. One participant withdrew due to Botox™ injection, and another dropped out due to poor exercise tolerance as the participant could not tolerate the study’s required exercise dose (ie, 20 hours in total). One participant from the SR group missed the follow-up assessment as he was overseas during the assessment period. The overall drop-out rate was 7.5%; therefore, all 40 participants were included in the data analysis based on intention-to-treat, and the LOCF method was used for the missing data. Figure 2 illustrates the CONSORT flow diagram of the study. The baseline demographic characteristics of the study participants are provided in Table 1, and there was no significant difference in the demographics between the SR and sham device groups.
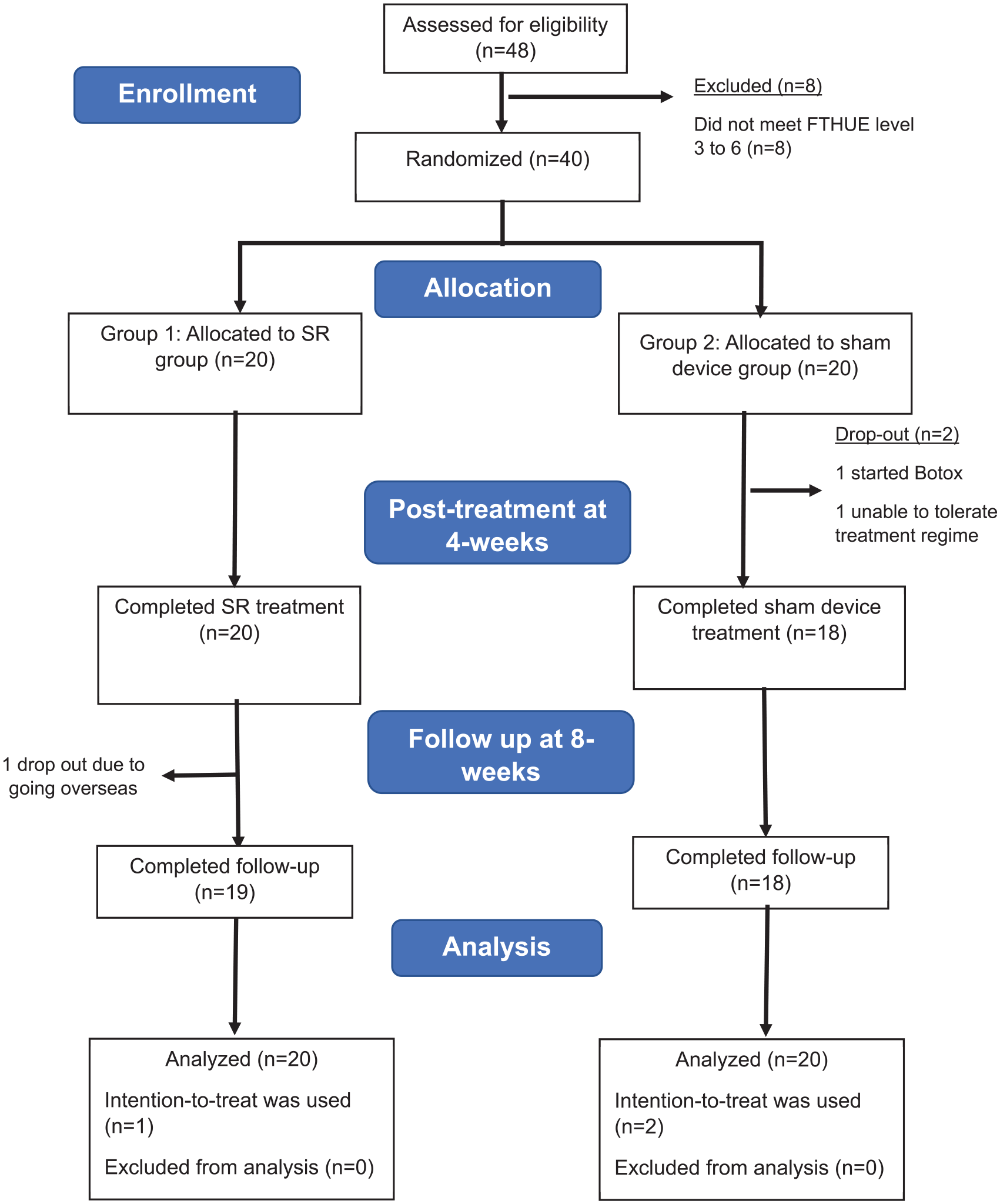
The CONSORT diagram.
Baseline Demographic and Clinical Characteristics.

Abbreviations: MMSE, Mini-Mental Status Examination; FTHUE-HK, Functional Test for Hemiplegic Upper Extremity-Hong Kong Version; ROM, range of motion; MS, muscle strength; FMA-UE, Fugl-Meyer Upper Extremity Assessment; ARAT, Action Research Arm Test; MAL-QOM, Motor Activity Log-Quality of Movement; MAL-AOU, Motor Activity Log-Amount of Use.
Effect on Upper Limb Motor Recovery
Table 2 presents the treatment effects within each group immediately after treatment (at 4 weeks) and at follow-up (at 8 weeks). Result from a single repeated measure MANOVA indicated a significant multivariate effect of time on the upper limb outcomes for both the SR (Wilks’ Λ = .195, F22, 58 = 3.213, P ≤ .001) and sham device (Wilks’ Λ = .303, F22, 56 = 2.079, P = .014) groups. Follow up univariate ANOVAs revealed that the SR group experienced significant improvements in the impairment measures. After 4 weeks of treatment, there were notable gains in AROM and muscle strength of the paretic elbow (elbow AROM = 7.52 ± 11.88°, P = .032; elbow flexion strength: 0.83 ± 1.32 kg, P = .035) and in FMA-UE scores (2.95 ± 3.55 points, P = .004). These improvements were sustained at the 8-week follow-up, with additional improvements in forearm rotation AROM (17.03 ± 28.89°, P = .049), muscle strength of the paretic elbow (0.84 ± 1.35 kg, P = .035), and FMA-UE scores (3.35 ± 3.42 points, P ≤ .001). In the sham device group, participants showed a significant improvement in muscle strength of the paretic shoulder immediately after the 4-week treatment, with shoulder flexion increasing by 0.76 ± 1.09 kg (P = .018) and shoulder abduction strength by 0.93 ± 1.35 kg (P = .018). At the 8-week follow-up, these participants exhibited further improvements in their paretic upper limb outcomes. Muscle strength of the paretic shoulder continued to increase, with shoulder flexion strength reaching 0.91 ± 1.34 kg (P = .020) and shoulder abduction strength rising to 1.36 ± 1.79 kg (P = .009). Additionally, their FMA-UE scores improved by 2.65 ± 3.38 points (P = .007), and their MAL quality of movement (MAL-QOM) scores increased by 0.29 ± 0.44 points (P = .026). No significant improvements in the ARAT were observed in either group.
Comparison of Upper Limb Outcomes Within Groups Throughout 8-week Period.

SD, standard deviation; AROM, active range of motion; FMA-UE, Fugl-Meyer Upper Extremity Assessment; ARAT, Action Research Arm Test; MAL-QOM, Motor Activity Log-Quality of Movement; MAL-AOU, Motor Activity Log-Amount of Use; MS, muscle strength; Pa, P-value for comparing within-group after treatment after treatment using single repeated measure MANOVA with Bonferroni adjustment. Pb, P-value for comparing within-group at 8-week follow-up using single repeated measure MANOVA with Bonferroni adjustment.
Mean (SD; n = 40).
P < .01. *P < .05.
Table 3 compares the treatment effects of both groups: the SR and the sham device groups. The results of the MANOVA indicated a significant multivariate difference between the groups on the upper limb outcome measures at post-treatment (Wilks’ Λ = .380, F13, 26 = 3.258, P = .005). Further univariate ANOVA revealed a significant group difference in the FMA-UE scores, with the SR group showing a greater improvement than the sham device group (F1, 38 = 4.71, P = .036, η² = .110), indicating a medium effect size. 54 Post hoc analysis confirmed that participants in the SR group had significantly improved FMA-UE performance compared to those in the sham device group (mean difference = 2.05 points, 95% confidence interval [CI] = 0.139-3.961). No significant differences were observed in other outcomes after 4-week treatment or at the 8-week follow-up.
Comparison of the Treatment Effects Between Groups Throughout 8-week Period.

Δ, change score; SD, standard deviation; AROM, active range of motion; FMA-UE, Fugl-Meyer Upper Extremity Assessment; ARAT, Action Research Arm Test; MAL-QOM, Motor Activity log-Quality of Movement; MAL-AOU, Motor Activity Log-Amount of Use; MS, muscle strength; Pc, P-value for comparing between-group after treatment at 4 weeks using MANOVA with Bonferroni adjustment; Pd, P-value for comparing between-group at 8-week follow-up using MANOVA with Bonferroni adjustment; Effect size, η2, partial eta squared.
Mean (SD; n = 40).
P < .01. *P < .05.
Given the differences in FMA-UE score improvements between the 2 groups, further sub-group analysis using Mann–Whitney U test was conducted to assess the impact of the UL’s severity on the treatment outcome. Participants were categorized into mild paresis with notable capacity (FMA-UE score ≥ 48) and moderate to severe paresis with limited capacity (FMA-UE score <48) based on their initial FMA-UE scores. 55 Figures 3 and 4 present the graphical displays for the changes in FMA-UE scores among the participants with moderate to severe and mild UL hemiparesis over time after the 2 treatments. For participants with moderate to severe UL paresis (FMA-UE score = 17-46), the results showed a significant difference between both groups (U = 37.5, Z = -2.61, P = .008). The SR group significantly improved in their performance in FMA-UE compared to the sham group immediately after 4-week treatment with a mean difference of 3.38 points (see Figure 3). At follow-up (eighth week), both groups continued to improve in FMA-UE, and there was no significant difference between them. However, as shown in Figure 4, no significant differences were found between the 2 groups for participants with mild UL paresis after treatment and at follow-up. At follow-up (eighth week), the sham group continued to improve in their FMA-UE scores while the improvement of the SR group was maintained.
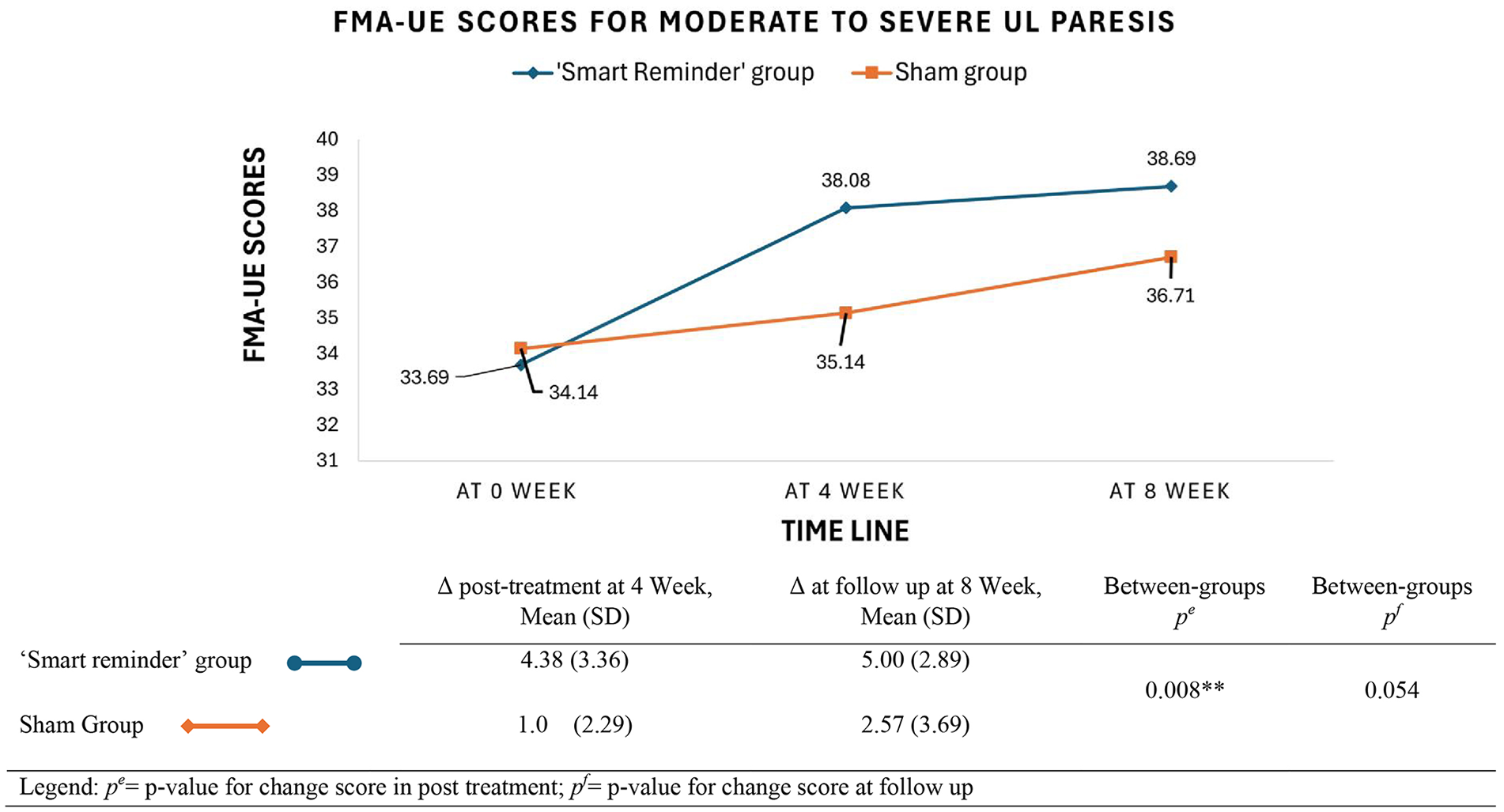
FMA-UE scores for participants with moderate to severe UL hemiparesis at baseline, post-treatment, and follow-up (n = 27; “Smart Reminder” device group: n = 13; sham device group: n = 14; FMA-UE < 48); **P < .01.
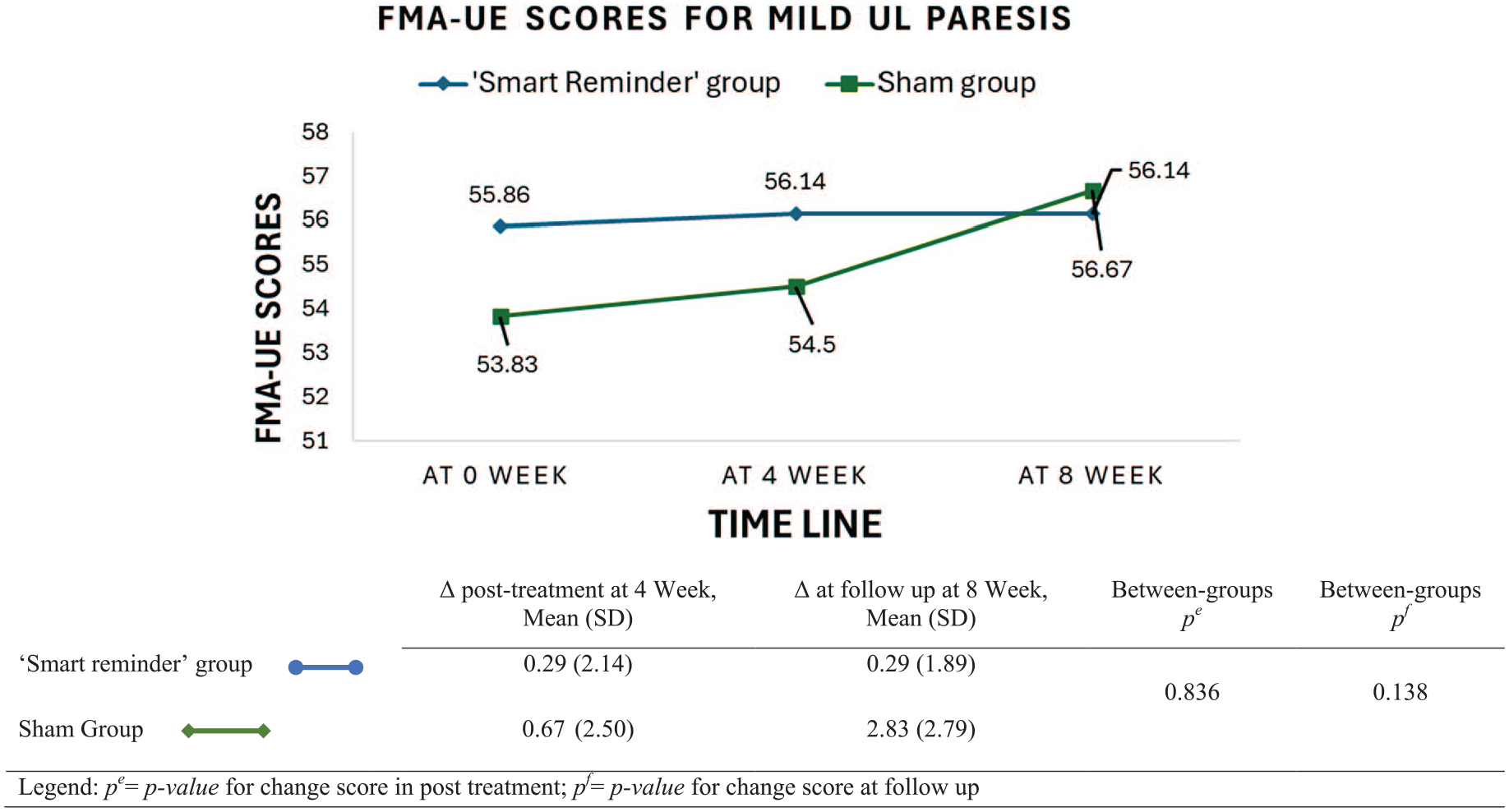
FMA-UE scores for participants with mild UL hemiparesis at baseline, post-treatment, and follow-up (n = 7; “Smart reminder” device group: n=; sham device group: n = 6; FMA-UE ≥ 48).
Therapy Compliance and Amount of Paretic Arm Use
As can be seen from Table 3, the SR group had a significantly higher compliance rate to the therapy program than the sham group (F1,39 = 4.62, P = .038, η2 = .108), with a medium effect size. 54 All the participants in both groups were given a standard accelerometer to wear for 3 hours outside the prescribed therapy exercises to collect their movement counts during their daily activities. Outside the prescribed therapy, the average movement counts per hour for the SR and sham groups showed no significant statistical difference (F1,39 = 0.82, P = .371, η2 = .021).
Discussion
In this study, we investigated the effects of a wearable device, SR, integrated with telerehabilitation to provide home-based UL rehabilitation for stroke survivors in the community. Results showed that the SR group exhibited more improvements in their FMA-UE scores after treatment compared to the sham group. Additionally, adherence to the SR telerehabilitation was significantly higher than that of the sham group. However, no significant differences were observed between the groups in other impairment outcomes or measures at the activity and participation levels of the ICF model, including the ARAT and the MAL. The study’s findings demonstrated that the intervention provided by SR seemed more efficacious in improving the FMA-UE scores of the hemiparetic UL than the conventional training using a sham device in the stroke participants. The favorable motor gain and therapy adherence in the SR group suggested that the multimodal augmented feedback from the SR device had an added therapeutic value for the UL recovery after stroke in the form of home-based rehabilitation. This observation was consistent with our feasibility study 39 on the SR device; in this feasibility study, apart from having a higher adherence rate, the AROM of the participants’ hemiparetic shoulders improved more significantly than conventional therapy after 4 weeks of telerehabilitation using SR. Furthermore, the positive effect of the SR treatment was consistent with the results of previous studies.16,56-58
Several studies41,56,59,60 echoed the effectiveness of using enhanced feedback such as knowledge of results (KR) and performance (KP) in VR training for UL rehabilitation after stroke. KR refers to the outcome of the movement while knowledge of performance provides information of the movement itself.20,22 Similarly, the participants received multimodal augmented feedback (visual and auditory) concurrently on their ROM KP and number of completed repetitions (KR) during the telerehabilitation using the SR device. Several pieces of evidence16,61-63 suggest that augmented feedback, such as KR and KP, can modulate sensorimotor cortical activity by facilitating learning in selected brain networks. Persons with stroke might benefit most from augmented feedback as their ability to generate intrinsic feedback to guide their performance was compromised due to neurological sensory impairments. 64 Hence, the multimodal augmented feedback provided during the SR seemed more beneficial for the stroke participants in motor learning than static pictorial exercise handouts used by the sham device group. Moreover, the interactive visual environment provided by the telerehabilitation training using SR and the remote monitoring by the therapist motivated the participants to engage in the therapy regime, accounting for the high compliance rate.
The mean difference in the FMA-UE change score between the SR and sham group was 2.05. Although statistically significant, the difference was considered modest and fell below the recommended MCID for the FMA-UE—a lower minimal clinically important differences (MCID) for the FMA-UE for patients with chronic severe hemiparesis, specifically at 3.5 points, 65 compared to 4.25 to 7.25 points for participants with mild to moderate paresis 66 and 12.4 points for participants with moderate to severe paresis in the early stroke phase. 67 In the subgroup analysis, the mean difference in the FMA-UE change score between the 2 groups was 3.38 points. This difference was statistically significant but marginally below the MCID for participants with chronic moderate to severe hemiparesis (FMA-UE <48). 65 Nevertheless, participants in the SR group with chronic moderate to severe hemiparesis demonstrated an average improvement of 4 points in their FMA-UE score immediately after treatment (Figure 3A), meeting the proposed MCID for this subgroup. This finding suggested that participants with more severe chronic hemiparesis might benefit more from the multimodal augmented feedback provided by the SR device during training.
This observation was supported by several studies,61,62,68,69 highlighting strong evidence to support the effectiveness of augmented feedback for motor training of the severely impaired UL. In another study, Fong et al 32 also found that the UL function of their participants with more severe hemiparesis significantly improved after 2 weeks of sensory cueing treatment using a wearable device. One possible explanation is that participants with more severe hemiparesis had lower motor skill levels and more impaired sensory functions, making augmented feedback, that is, ROM performance in this study, more useful. The stroke participants with more severe sensory impairments, such as proprioception, might rely more on extrinsic feedback to know what was needed to improve performance. 22 Furthermore, previous reviews19,23 also highlighted that concurrent feedback suits individuals with low skill proficiency. During telerehabilitation provided by the SR device, participants received concurrent visual and auditory feedback that raised their awareness of the performance, enhancing their motor learning process. Participants with mild paresis, on the other hand, might have intact intrinsic feedback systems and were less reliant on ROM feedback. Moreover, the ceiling effects of the FMA-UE in assessing persons with mild UL impairment 70 (ie, FMA-UE score ≥48) might have contributed to the absence of treatment difference observed in both study groups.
Another interesting finding was that, despite the motor gains in the SR group as reflected in their FMA-UE scores, there was no significant improvement observed in the participants’ ARAT and MAL scores. This observation suggests that the improvement in impairments was not substantial enough to create a change in the functional performance of the paretic UL or alter the participants’ perception of using the affected upper limb in their daily routine. One explanation is that most of the study participants had chronic severe hemiparesis, which previous studies have shown that these participants tend to correlate with low MAL scores and a reliance on the non-paretic upper limb for daily activities.71-73 Furthermore, this study used the MAL-30, which has excellent internal consistency for individuals with mild to moderate paresis but is less sensitive for those with severe paresis. 74
Lastly, the study’s results indicated that participants who received conventional training along sham device also improved in their hemiparetic UL function, but to a lesser extent. This finding contrasts with the author’s previous feasibility study, 39 where no improvement in the UL outcomes was observed in the conventional therapy group. One possible reason is that in the current study, the sham device group was provided with an accelerometer to track their daily UL movements. Despite the sham device not offering external feedback or connecting to a telerehabilitation app, its presence heightened participants’ attention and awareness of their hemiparetic arm, thereby creating a “treatment” effect on them. The knowledge that their UL movements were being monitored by their therapist could have motivated participants to exercise and use the affected arm in their daily activities. Supporting this notion, Sandra et al 75 found that the placebo effect increases when devices are perceived as personalized, which enhances treatment effectiveness. This is confirmed by the observation in this study that the SR and sham device groups did not differ significantly in their average hourly movement counts of the affected ULs during daily activities.
Limitations
This study had some limitations. Based on the recommendation by previous studies,26,31 participants were instructed to wear the accelerometer for 3 hours to capture their paretic UL movements in their daily routines. However, a 3-hour duration might be insufficient to accurately capture the actual real-life usage patterns for the paretic upper limb, potentially creating an upward biased as participants might intentionally increase their movement during the wearing period. Previous studies10,76 had adopted longer wearing durations of 12 hours or more to better capture the daily activity of the paretic upper limb. In addition, the SR device could not track the amount of hemiparetic arm use by the stroke participants in their daily routine outside of therapy. A wearable device with dual functions, such as supporting the “prescribed therapy” and tracking the hemiparetic arm’s use in the stroke survivors outside of therapy, would be ideal to encourage intensive use of the affected UL in their daily routine to sustain the therapeutic gains. 13 Lastly, this study did not assess the arm use of the non-hemiparetic upper limb in the participants. Recent research77,78 has indicated that participants with severe hemiparesis often exhibit significant motor deficits in their less-affected arm, which they heavily rely on for daily activities. Future studies may evaluate both the less-affected and the hemiparetic arms using wearable technology, particularly in persons with severe hemiparesis.
Conclusion
This study integrated wearable technology and smartphones to offer stroke survivors alternative methods for rehabilitating their hemiparetic UL at home. The findings demonstrated that the telerehabilitation provided by the SR device was more efficacious than conventional training using a sham device in improving the motor impairments of the hemiparetic UL measured using FMA-UE in persons with stroke. However, it did not significantly increase the participants’ perception in using their hemiparetic UL. Additionally, adherence to telerehabilitation with SR was excellent among stroke survivors. Further research is needed to explore the economic benefits of this novel device and its potential to offer valuable opportunities for self-directed rehabilitation, thereby easing the burden on conventional rehabilitation services.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683241283412 – Supplemental material for Effects of a Wearable-Based Intervention on the Hemiparetic Upper Limb in Persons With Stroke: A Randomized Controlled Trial
Supplemental material, sj-docx-1-nnr-10.1177_15459683241283412 for Effects of a Wearable-Based Intervention on the Hemiparetic Upper Limb in Persons With Stroke: A Randomized Controlled Trial by Fong Mei Toh, Winnie W. T. Lam, Pablo Cruz Gonzalez and Kenneth N. K. Fong in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-2-nnr-10.1177_15459683241283412 – Supplemental material for Effects of a Wearable-Based Intervention on the Hemiparetic Upper Limb in Persons With Stroke: A Randomized Controlled Trial
Supplemental material, sj-docx-2-nnr-10.1177_15459683241283412 for Effects of a Wearable-Based Intervention on the Hemiparetic Upper Limb in Persons With Stroke: A Randomized Controlled Trial by Fong Mei Toh, Winnie W. T. Lam, Pablo Cruz Gonzalez and Kenneth N. K. Fong in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-3-nnr-10.1177_15459683241283412 – Supplemental material for Effects of a Wearable-Based Intervention on the Hemiparetic Upper Limb in Persons With Stroke: A Randomized Controlled Trial
Supplemental material, sj-docx-3-nnr-10.1177_15459683241283412 for Effects of a Wearable-Based Intervention on the Hemiparetic Upper Limb in Persons With Stroke: A Randomized Controlled Trial by Fong Mei Toh, Winnie W. T. Lam, Pablo Cruz Gonzalez and Kenneth N. K. Fong in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-4-nnr-10.1177_15459683241283412 – Supplemental material for Effects of a Wearable-Based Intervention on the Hemiparetic Upper Limb in Persons With Stroke: A Randomized Controlled Trial
Supplemental material, sj-docx-4-nnr-10.1177_15459683241283412 for Effects of a Wearable-Based Intervention on the Hemiparetic Upper Limb in Persons With Stroke: A Randomized Controlled Trial by Fong Mei Toh, Winnie W. T. Lam, Pablo Cruz Gonzalez and Kenneth N. K. Fong in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors would like to thank the study participants for their participation.
Author Contributions
Fong Mei Toh: Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Writing—original draft; Writing—review & editing. Winnie W. T. Lam: Investigation; Writing—review & editing. Pablo Cruz Gonzalez: Investigation; Writing—review & editing. Kenneth N. K. Fong: Conceptualization; Funding acquisition; Investigation; Methodology; Project administration; Resources; Supervision; Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research project was funded by the Research Impact Fund (No. R5028-20F) of the Research Grants Council, University Grants Committee, Hong Kong SAR, and partially funded by external grants from Caspar Health Limited (No. ZDBD) and Hong Kong Young Women’s Christian Association (No. 5-ZH2X), awarded to KNKF.
Ethical Approval and Consent to Participate
This study (HSEARS20230317001) was approved by the Institutional Review Board of the Hong Kong Polytechnic University before its commencement.
Consent for Publication
All authors have approved this manuscript for publication. This manuscript has not been previously published and is not pending publication elsewhere.
Data Accessibility
There are no funders’ requirements that the data should be accessed.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
