Abstract
Background
Slowed balance and mobility after stroke have been well-characterized. Yet the effects of unilateral cortical lesions on whole-body neuromechanical control is poorly understood, despite increased reliance on cortical resources for balance and mobility with aging. Objective. We tested whether individuals post stroke show impaired cortical responses evoked during reactive balance, and the effect of asymmetrical interlimb contributions to balance recovery and the evoked cortical response.
Methods
Using electroencephalography, we assessed cortical N1 responses evoked over fronto-midline regions (Cz) during backward support-surface perturbations loading both legs and posterior-lateral directions that preferentially load the paretic or nonparetic leg in individuals’ post-stroke and age-matched controls. We tested relationships between cortical responses and clinical balance/mobility function, as well as to center of pressure (CoP) rate of rise (RoR) during balance recovery.
Results
Cortical N1 responses were smaller and delayed after stroke (P < .047), regardless of perturbation condition. In contrast to controls, slower cortical response latencies associated with lower clinical function in stroke (Mini Balance Evaluation Systems Test: r = −.61, P = .007; Timed-Up-and-Go: r = .53, P = .024; walking speed: r = −.46, P = .055). Paretic-loaded balance recovery revealed slower CoP RoR (P = .012) that was associated with delayed cortical response latencies (r = −.70, P = .003); these relationships were not present during bilateral and nonparetic-loaded conditions, nor in the older adults control group.
Conclusions
Individuals after stroke may be limited in their balance ability by the slowed speed of their cortical responses to destabilization. In particular, paretic leg loading may reveal cortical response impairments that reflect reduced paretic motor capacity.
Introduction
Increasing evidence suggests there are age-related shifts from subcortically- to more cortically-mediated balance control, yet little is known about how stroke, prevalent in older adults, affect these cortical mechanisms involved in balance control. Slower motor reactions after stroke contribute to lower resilience to postural perturbations.1-4 An impaired ability to rapidly and effectively use the paretic leg may require compensatory use of the nonparetic leg for balance and walking after stroke.5-10 From a neurophysiologic perspective, greater asymmetry in corticomotor excitability between paretic and nonparetic lower limbs, assessed in seated positions, is associated with greater reliance on the nonparetic leg to increase walking speed. 11 However, it is unclear whether this asymmetry in corticomotor neurophysiology persists during functional tasks involving time-synchronized control of whole-body movements. Recordings of brain activity during reactive balance may provide a well-controlled, whole-body movement paradigm to assess neuromechanical interactions between cortical activity and balance control in post-stroke lower limb hemiparesis.
Lesions affecting cortical and subcortical pathways in older adults after stroke may compromise the ability to engage cortical resources for rapid balance recovery following destabilization. Using electroencephalography (EEG) during standing balance recovery reactions, we recently found that balance destabilization elicited greater cortical beta activity during balance recovery in neurologically-intact older adults with lower balance function. 12 This finding in older adults suggests greater sensorimotor cortical reliance for postural stability in individuals with lower balance function. Likewise, in neurotypical younger adults, cortical compensation during balance recovery may be reflected in larger cortical evoked responses during reactive balance in individuals with relatively poor balance ability, 13 when taking compensatory steps following challenging balance perturbations,14,15 and when perturbations are perceived as more threatening.16,17 Supporting this notion, individuals with lower post-stroke mobility commonly engage expansive cortical networks spanning sensorimotor and frontal regions during continuous walking tasks. 18 In stroke, those with lower mobility function also reached a “ceiling effect” of lower cortical activity compared to higher-functioning individuals when presented with more challenging dual-task walking conditions. 18 Together, these findings suggest individuals with stroke may increase reliance on cortically-mediated strategies for balance control,12,18 which may be compromised by lesions affecting cortical and subcortical structures.
Reactive balance control is essential to walking and mobility, 19 but cortical responses during rapid corrective balance reactions has not been characterized after stroke. Here, we measure the cortical N1 response, a large negative-going peak in the EEG signal over midline sensorimotor areas ~150 ms after a sudden disturbance to standing balance. 20 The N1 response is thought to reflect detection of a sudden error to balance or posture, 21 and has been localized to the supplementary motor area,22,23 with contribution from multiple sources including the supplementary motor area, the anterior cingulate cortex, sensorimotor areas, and parietal cortex.24-26 Temporal features of cortical responses and relevance to balance recovery responses may also be necessary to develop effective balance rehabilitation approaches.
Delineating differences in cortical function involving paretic versus nonparetic leg use during bipedal behaviors such as walking and balance is challenging. Individuals with lateralized cortical lesions due to stroke commonly present with limb hemiparesis, causing interlimb motor control deficits.7,19 Differential cortical mechanisms during paretic and nonparetic leg motor activity have been identified during seated and isometric lower limb muscle contractions in individuals with chronic stroke.11,27-29 In particular, plantarflexor muscles play a key role in post-stroke mobility function,5,30-32 and show more severely impaired corticomotor excitability (ie, lower motor evoked potentials) compared to dorsiflexors. 27 Further, individuals with greater nonparetic corticomotor excitability show greater biomechanical reliance on the nonparetic leg to generate propulsive forces during walking, suggesting a link between corticomotor function and whole-body behaviors. 12 Reactive balance paradigms may provide a method to assess neural contributions to whole-body behaviors through use of external perturbations that elicit a time-locked behavioral response that successively recruits subcortical followed by cortical contributions to lower limb motor reactions.33-35 Lateralized balance recovery may provide neuromechanical insight into paretic motor deficits that appear to contribute to post-stroke falls.1,10,19,36 A case-series study reported direction-specific spatial and spectral components in perturbation-evoked EEG cortical responses in stroke (n = 3) and younger adults (n = 6). 37 However, whether these differences in spectral features relate to clinical ability or balance impairment or to differences in the timing or magnitude of evoked cortical responses after stroke has not been investigated.
Our central hypothesis is that stroke impairs rapid evoked responses of the cerebral cortex during balance-correcting behavior, reflected in the cortical N1 response, and is linked to asymmetrical interlimb contributions to post-stroke mobility behavior. We used multidirectional standing balance perturbations to differentially challenge balance control between the legs. We further tested the effect of mechanical balance perturbations loading either the paretic or nonparetic leg during balance recovery on evoked cortical N1 responses and kinetic reactions and relationships to clinical balance and mobility function. We predicted that (1) stroke survivors would have later and attenuated perturbation-evoked cortical N1 responses during balance recovery compared to neurotypical, age-matched controls, with the most impaired cortical N1 responses during paretic-loading conditions and (2) that longer latencies of perturbation-evoked cortical N1 responses would be associated with clinical balance deficits and slower kinetic reactions after stroke.
Methods
Study Design and Participants
Eighteen community-dwelling individuals with chronic (>6 months) stroke (Table 1) and 17 age-matched controls were recruited from local outpatient and community clinics. Inclusion criteria included above the age of 21 years, the ability to walk at least 10 m without the assistance of another person, the ability to stand unassisted for at least 3 minutes, and the cognitive ability for informed consent. Participants were excluded for any diagnosed neurologic condition other than stroke or pain affecting standing or walking. Age-matching of the control group was performed such that the mean age of each group was within 5 years. The experimental protocol was approved by the Emory University Institutional Review Board and all participants provided written informed consent.
Stroke Participant Characteristics (n = 18).

Abbreviations: PSD, post stroke duration; TUG, Timed-Up-and-Go; min-iBESTest, Mini Balance Evaluation Systems Test; m/s, meters per second; M1: primary motor cortex; S1: primary somatosensory cortex; IC: internal capsule; BG: basal ganglia; ACA: anterior cerebral artery; MCA: middle cerebral artery; PLIC: posterior limb IC; CR: corona radiate; N/A, not available.
Stroke occurred near corticomedullary junction, potentially explaining ipsilateral limb deficits.
Participants completed a single visit of clinical balance and mobility testing (ie, Mini Balance Evaluation Systems Test [mini-BESTest], 38 Timed-Up-and-Go [TUG], 39 and 10-m walk test) following standard clinical practice procedures and administered by the same licensed physical therapist. Participants were then subjected to a series of support-surface translational perturbations to assess EEG measures of evoked cortical activity and biomechanical reactions during standing balance recovery.
Standing Balance Perturbations
Participants stood barefoot on a moving platform (Factory Automation Systems, Atlanta, GA, USA) with embedded force plates and were subjected to anterior, posterior, and left-ward and right-ward posterolateral support-surface translational perturbations that served to preferentially load either the paretic leg, the nonparetic leg, or equal legs during balance recovery. Participants wore a safety harness anchored to the ceiling to reduce fall risk and no body weight was supported by the harness. Participants were instructed to stand with feet at shoulder-width apart and evenly placed directly under the hips, but received no other instruction regarding baseline standing posture. During the paretic-loaded condition, the support-surface moves posterolaterally toward the nonparetic leg, shifting a greater proportion of body weight support onto the paretic leg (Figure 1). Likewise, the nonparetic-loaded condition shifts a greater proportion of body weight support onto the nonparetic leg. In contrast, in the bilateral condition, the support-surface moves in the posterior direction with no lateralization (Figure 1), targeting plantarflexor agonist muscles to correct for postural destabilization. Anteriorly directed perturbations were also included to discourage participants from leaning backward in anticipation of posteriorly directed perturbations used in the analyses. Participants were instructed to stand with their typical, self-selected posture and foot placement at approximately shoulder-width, but baseline stance width was not precisely standardized as similar motor response latencies are observed across a range of narrow and wider stances. 40 Twenty-four perturbations (7.5 cm, 16.0 cm/s, 0.12 g) within each of the 4 directions (total of 96 perturbations) were delivered in a pseudorandomized order at unpredictable inter-trial intervals (15-60 s). Participants received instructions to recover balance with a feet-in-place strategy if possible and to keep arms folded at their chest. We selected this relatively low-level perturbation level because it could be successfully completed by most participants using a feet-in-place strategy. However, 2 participants in the stroke group and 1 control participant were unable to recover balance at this perturbation level with feet-in-place and maintaining arms crossed at their chest; for these participants the magnitude of balance perturbation was scaled down to (6.0 cm, 12.0 cm/s, 0.08 g) while maintaining the same temporal characteristics of the perturbation such that each perturbation reached peak acceleration at 100 ms across all participants. The perturbation series was delivered in a pseudorandomized order, where a perturbation of the same direction had no more than 2 consecutive occurrences. Real-time EEG and electromyography (EMG) activity from bilateral lower limbs (eg, tibialis anterior, soleus, and medial gastrocnemius) and sternocleidomastoid muscles were visually monitored by the experimenters to ensure that the participant’s cortical and muscle activity levels returned to baseline standing levels after each trial before the next perturbation was delivered. Additionally, real-time kinetics were monitored between trials to ensure that participants maintained an upright body position relative to the vertical in the sagittal plane that was consistent with their baseline standing posture. If real-time kinetics deviated from the vertical between trials, participants were verbally cued to stand upright and correct for postural lean. If a participant took a reactive step during the trial, the experimenter ensured that feet placement was returned to baseline prior to the start of the next trial. Instances of stepping were noted in real-time for offline confirmation and excluded from analyses based on ground reaction forces. 15 Participants took a seated rest break every 8 minutes during balance perturbation testing, or more frequently if the participant requested a break or reported or showed signs of fatigue (physiologic, observed, and participant-reported) during testing.
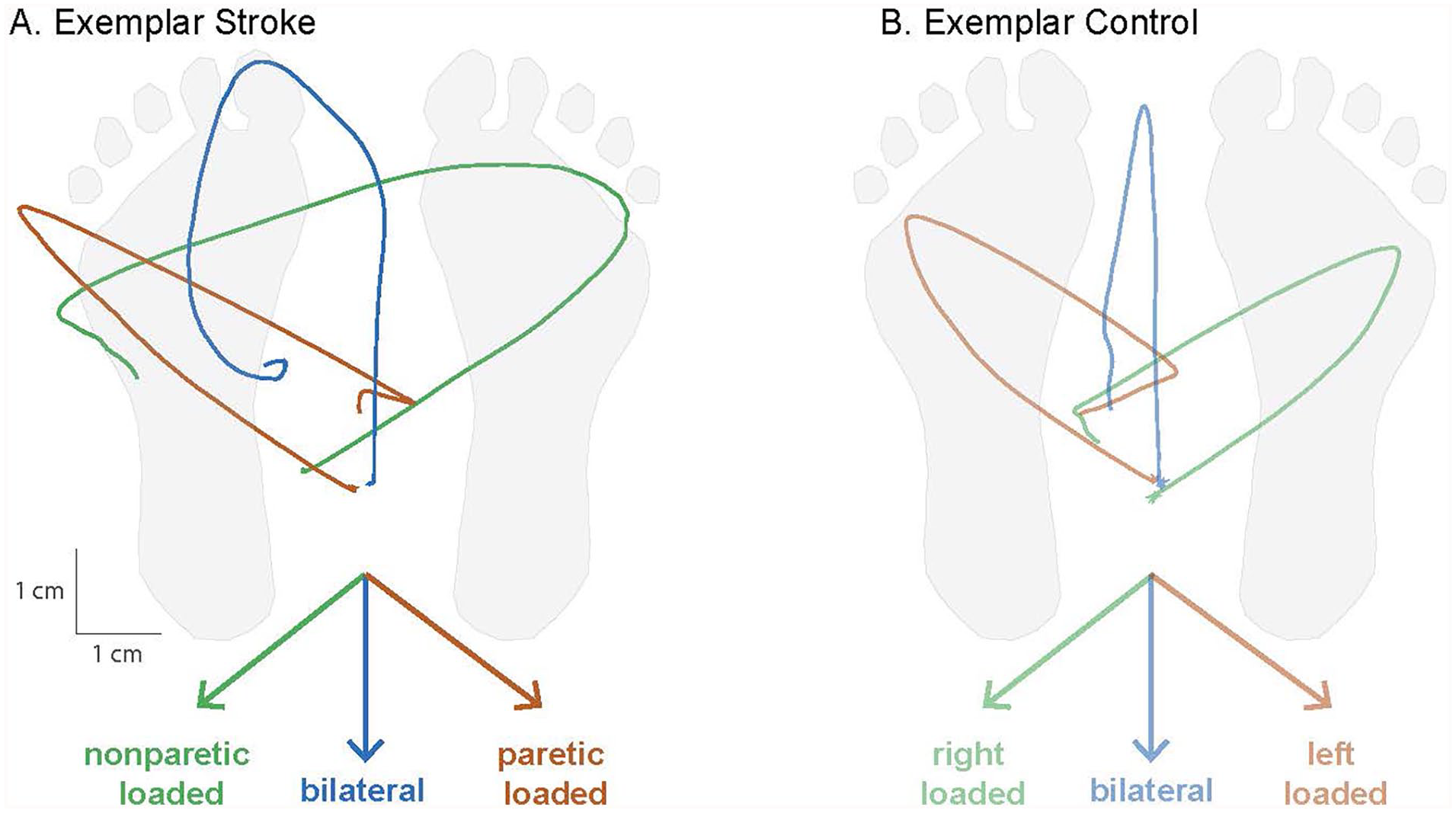
Perturbation conditions and resulting kinetic reactions for balance recovery. Across each of the perturbation conditions, the support surface moved in the direction indicated by the colored arrows necessitating a corrective shift in the center of pressure (CoP) toward the paretic leg, bilateral legs, or nonparetic leg to prevent imbalance. Mechanical effects of each condition on CoP displacement trajectories (condition-averaged across trials) and postural loading are depicted in an exemplar stroke (A) and control (B) participant. In lateralized conditions, the CoP trajectory was shifted toward the paretic or nonparetic legs (green and orange), while the bilateral condition (blue) showed no lateralized bias in the perturbation. Note that the paretic-loaded condition refers to movement of the support surface and feet in the direction of the nonparetic limb, consequently shifting the CoP beneath of paretic limb and loading the paretic limb during the rapid kinetic reaction. During the bilateral condition, individuals after stroke commonly displayed asymmetric balance recovery strategies (A) but were effectively forced to unilateral leg loading during lateralized perturbation conditions. Condition-averaged CoP trajectories are shown from an example participant in each group.
EEG Data Acquisition and Analyses
During balance perturbations, cortical activity was continuously recorded from EEG signals using a 64-channel active electrode cap (actiCAP, actiCHamp amplifier, Brain Products, GmbH, Gilching, Germany). EEG signals were digitized with a 24-bit analog-to-digital converter and an online 20 kHz low-pass filter and before sampling at 1000 Hz and storing for offline analysis. All EEG data were preprocessed using freely available functions from the EEGLAB toolbox and custom MATLAB scripts. 41 Continuous data time-locked to the perturbation onset were imported into EEGLAB. Trigger labels for successful feet-in-place trials (ie, no reactive step taken) were selected across all conditions. Continuous EEG data were high-pass filtered (cutoff 0.5 Hz, finite impulse response, and filter order 3300) and downsampled to 500 Hz. Bad channels were identified as channels having excessive noise or artifact (muscle activity, motion, and electrical) at baseline (eg, >50 µV) through visual inspection. Excessively noisy channels were removed and missing channels were interpolated for component analyses as part of the preprocessing pipeline. Data were re-referenced to an average reference. Line noise was removed using the Cleanline plugin. 41 Data were then epoched −2 to 2 seconds around each perturbation (platform onset at t = 0 s), and decomposed into maximally independent components (ICs) using adaptive mixture component analysis algorithm (AMICA). 42 ICs from AMICA were categorized using the ICLabel plugin, an automated algorithm that identifies nonbrain sources (eg, eye, muscle, and cardiac activity) and brain sources, 43 and confirmed with visual inspection. Nonbrain sources were removed. The remaining brain ICs were projected back into channel space. For 1 participant with a lower number of non-stepping trials (S12), we aimed to maximize the number of trials for AMICA by including all trials (step and no step) in this part of the preprocessing pipeline before removing all stepping trials for N1 waveform computation and all subsequent analyses. Data were visually inspected and trials with excessive signal drift were removed.
EEG data from the midline sensorimotor region (Cz) for posterior and postero-laterally directed perturbations were selected and low-pass filtered at 30 Hz for evoked cortical event-related potential analyses.20,44 The peak latency and amplitude of the cortical N1 response were extracted from the mean waveforms within and across conditions after baseline (−150 to −50 ms) subtraction. The cortical N1 response was defined as the first local minimum point of negative value in the EEG waveform within 100 to 300 ms post-perturbation. As individuals with stroke tended to show polyphasic perturbation-evoked cortical responses (Figure 3C), this automated selection criteria enabled consistency in selection of the cortical N1 response within-participants (between conditions) and between-participants. Two participants (S12 and S19) required an extended time window of 100 to 350 ms post-perturbation because the first local minimum in the cortical response waveform occurred >300 ms post-perturbation (316 and 314 ms post-perturbation for all conditions collapsed, respectively).
Individuals show motor adaptation effects to balance perturbations, in which people after stroke show the greatest adaptation during the first 6 to 8 balance perturbations.8,45 We confirmed that the N1 amplitudes and latencies correlate with r > .89, P < .000001 between datasets that include versus exclude the first 8 trials (within and across conditions for both groups), and as such, any effects of initial adaptation will have negligible impact on the present results.
Kinetic Data Acquisition and Analysis
Kinetic (2 AMTI OR6-6 force plates, each 40 cm × 60 cm) and kinematic (10-camera Vicon Nexus 3D motion analysis system) data were recorded during balance perturbations (100 Hz sampling frequency). Reflective markers were placed on anatomical landmarks on the legs and trunk (eg, head, neck, hips, knees, ankles, and feet) and were used as inputs to Vicon’s plug-in-gait model to compute the body’s center of mass velocity and displacement throughout balance recovery.
The corrective kinetic reaction during balance recovery following perturbations was quantified as the CoP RoR. During the perturbation, the CoP initially moves passively as a result of the perturbation, and the individual must then rapidly counteract this effect to slow and reverse the direction of CoP movement to maintain upright stability (Figure 1). 46 The CoP RoR in the later 150 to 300 ms phase of balance recovery occurs during a timeframe in which an individual’s active contribution to reactive balance is possible and necessary for a successful feet-in-place balance recovery. As such, the average slope of this later phase CoP position trajectory indexes how quickly an individual generates corrective responses to the loss of balance. A slower CoP RoR relative to the support surface movement would result in a less effective neuromechanical stabilization strategy that may lead to loss of balance. 46 The CoP position was used to assess CoP RoR in the direction parallel to support-surface movement quantified as the linear slope (ie, rate of displacement in CoP position) between 150 and 300 ms post-perturbation onset.
Statistical Analyses
We confirmed normality and heterogeneity of variance of all data used for analyses using Kolmogorov–Smirnov and Levene’s tests, respectively. We matched lateralized balance conditions of paretic-loading to left-loading and nonparetic-loading to right-loading in controls. First, we compared cortical N1 response latency and amplitudes collapsed across all conditions between stroke and control groups using independent t-tests. We then tested group (control, stroke) and condition (bilateral, paretic-loaded, nonparetic-loaded) main effects and interactions between group and condition for each cortical N1 metric (peak latency, peak amplitude) using a 2-way analysis of variance (ANOVA). We used Pearson product moment correlation coefficients to test for associations between cortical N1 peak metrics and clinical and biomechanical metrics. We used multiple linear regression (MLR) analyses (factors: group, N1, and group-by-N1) to test whether the relationship between cortical N1 peak metrics (latency or amplitude) and clinical metrics (walking speed, TUG, or mini-BESTest) differed as a function of group in the collapsed N1 waveform and across each condition. Condition-specific cortical N1 responses were used for all kinetic-based analyses when results differed by condition and due to the temporal synchrony of EEG and kinetic data collection. We used a 2-way ANOVA (factors: group, condition, and group-by-condition) to test for group-by-condition interaction and main effects on CoP RoR. We used MLR analyses (factors: group, N1, and group-by-N1) to test whether the relationship between cortical N1 peak metrics (latency or amplitude) and CoP RoR differed as a function of group across each condition. All analyses were performed using Statistical Package for Social Sciences version 27 (IBM Corp., Armonk, VY, USA) with an a priori level of significance set to 0.05.
Results
One participant in the control group withdrew from the study due to fear of falling; this participant was unable to complete balance perturbation testing and was excluded from analysis. Due to fatigue and increased time necessary for balance testing, 8 participants in the stroke group completed a shortened protocol (range of perturbations completed: 60-86 out of 96 perturbations in full protocol). Including those adopting the shortened protocol, EEG recordings from 18 individuals’ post-stroke and 16 controls were included in analyses. Participants in the stroke group had lower clinical measures of balance function on the mini-BESTest (P < .001), slower performance on the TUG-test (P < .001), and slower walking speed (P < .001; Table 2). Technical issues involving biomechanical force data acquisition occurred in 3 participants (stroke, n = 2; control, n = 1); these participants were excluded from analyses involving CoP RoR but are included in all other analyses.
Participant Group Characteristics.

Abbreviations: TUG, timed-up-and-go; m/s: meters per second.
Excludes control participant (n = 1) who withdrew from the study and was not included in analyses. Values are depicted in mean ± standard deviation [range: minimum-maximum].
Fisher’s exact test.
Effect of Stroke on Cortical N1 Response and Relationship to Clinical Balance and Mobility
After stroke, cortical responses were impaired and associated with post-stroke clinical dysfunction. Individuals post stroke exhibited delayed latencies to the cortical N1 peak (stroke = 219 ± 39 ms; control = 196 ± 22 ms, P = .025) and reduced N1 amplitudes (stroke = 14.9 ± 11.9 µV; control = 21.7 ± 11.0 µV, P = .047) compared to age-matched controls across all conditions (Figure 2A). The relationship between N1 latencies and behavioral outcomes varied between groups. In the stroke group, delayed N1s correlated with lower mini-BESTest scores (r = −.61, P = .007), slower Timed-Up-and Go-(TUG) test performance (r = .53, P = .024), and exhibited a trend with reduced walking speed (r = −.46, P = .055; Figure 2B). In the control group, delayed N1s were similarly associated with slower TUG test performance (r = .508, P = .045) and showed no correlation with mini-BESTest scores (r = −.274, P = .304) or walking speed (r = −.001, P = .997). We observed that the outcomes did not differ across the conditions and therefore the N1 responses were collapsed across all conditions and used in the present analysis. While the stroke group consistently showed stronger relationships between cortical N1 latency and clinical metrics compared to controls (Figure 2B
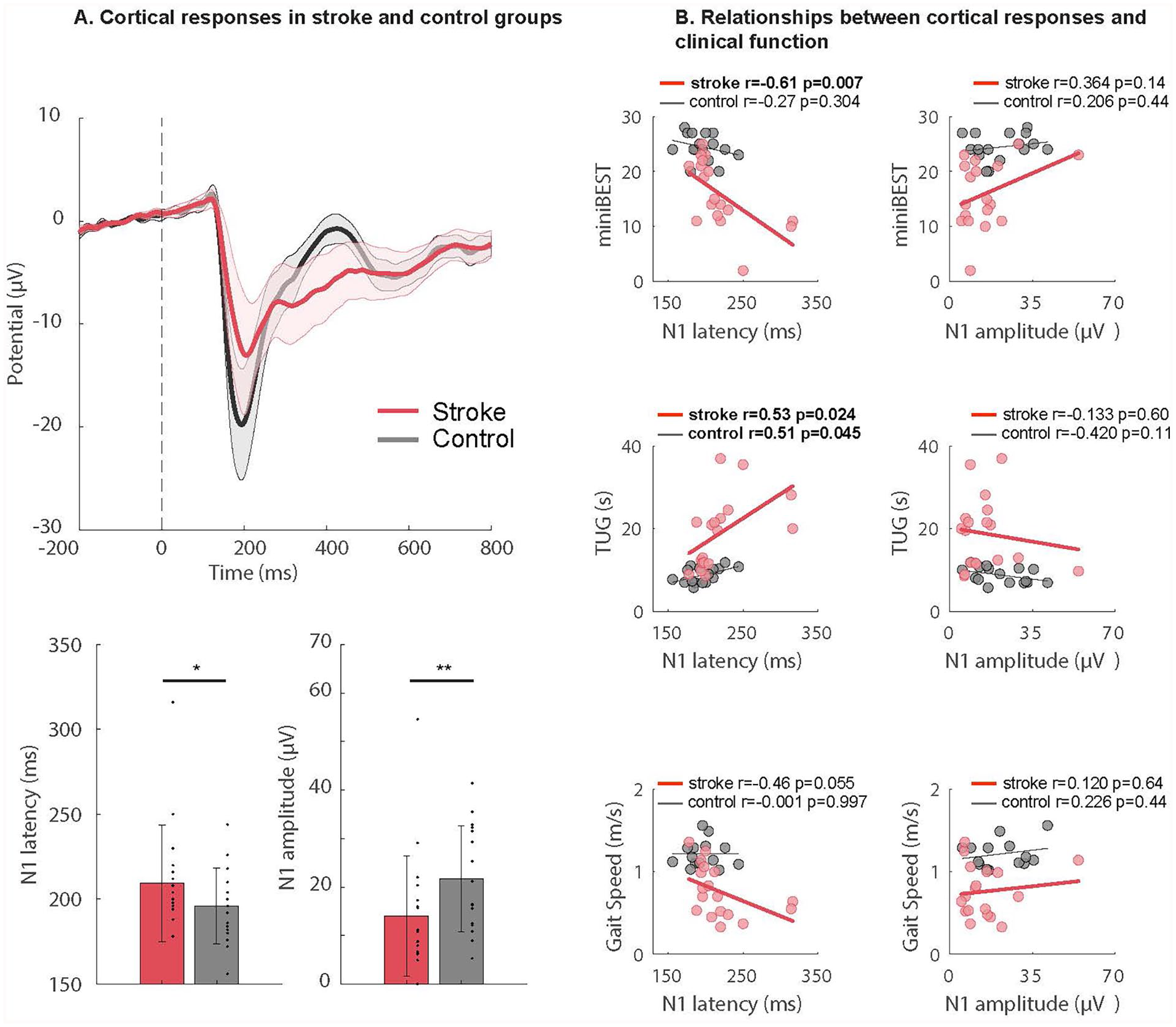
Impaired cortical responses associated with clinical balance and mobility after stroke. (A) Evoked cortical N1 response waveforms during balance recovery are shown collapsed across all perturbation conditions in each group. Mean ± standard error of the mean are depicted in the shaded regions (top). In participants after stroke, cortical N1 responses showed later peak latencies (stroke = 219 ± 39 ms; control = 196 ± 22 ms, *P = .025) and attenuated peak amplitudes (stroke = 14.9 ± 11.9; control = 21.7 ± 11.0, **P = .047) compared to age-matched controls (bottom). (B) Relationships between evoked cortical N1 peak latency (left column) and peak amplitude (right column) during reactive balance across all conditions versus mini-BESTest score (top row), single-task timed-up-and-go (TUG) test (middle row), and walking speed (bottom row), in individuals post stroke and age-matched controls. For the mini-BESTest, slower cortical N1 response latencies were associated with lower mini-BESTest score in the stroke group (r = −.61, P = .007) while showing no relationship in controls (r = −.274, P = .304). (A). For the TUG, both groups showed an association between later cortical N1 latency and slower TUG performance (stroke: (r = .53, P = .024; controls: r = .508, P = .045). For gait speed, slower cortical N1 response latencies showed a trend for association with slower gait speed in the stroke group (r = −.46, P = .055), a relationship that was absent in controls (r = −.001, P = .997). Cortical N1 peak amplitude (right column) was not significantly associated with any clinical balance or mobility function metric in the stroke and control groups (P > .11).
Effect of Lateralization of Balance Perturbations in Stroke and Controls
Contrary to our initial hypotheses, the lateralization of balance perturbations did not have any discernible impact on cortical N1 response latency or amplitude in either the stroke or control groups (Figure 3A and B). Despite considerable between-individual variability (Figure 3C), no significant interaction effects were observed for N1 latency (F2,64 = 0.463, P = .722) or amplitude (F2,64 = 0.624, P = .516; Figure 3B).
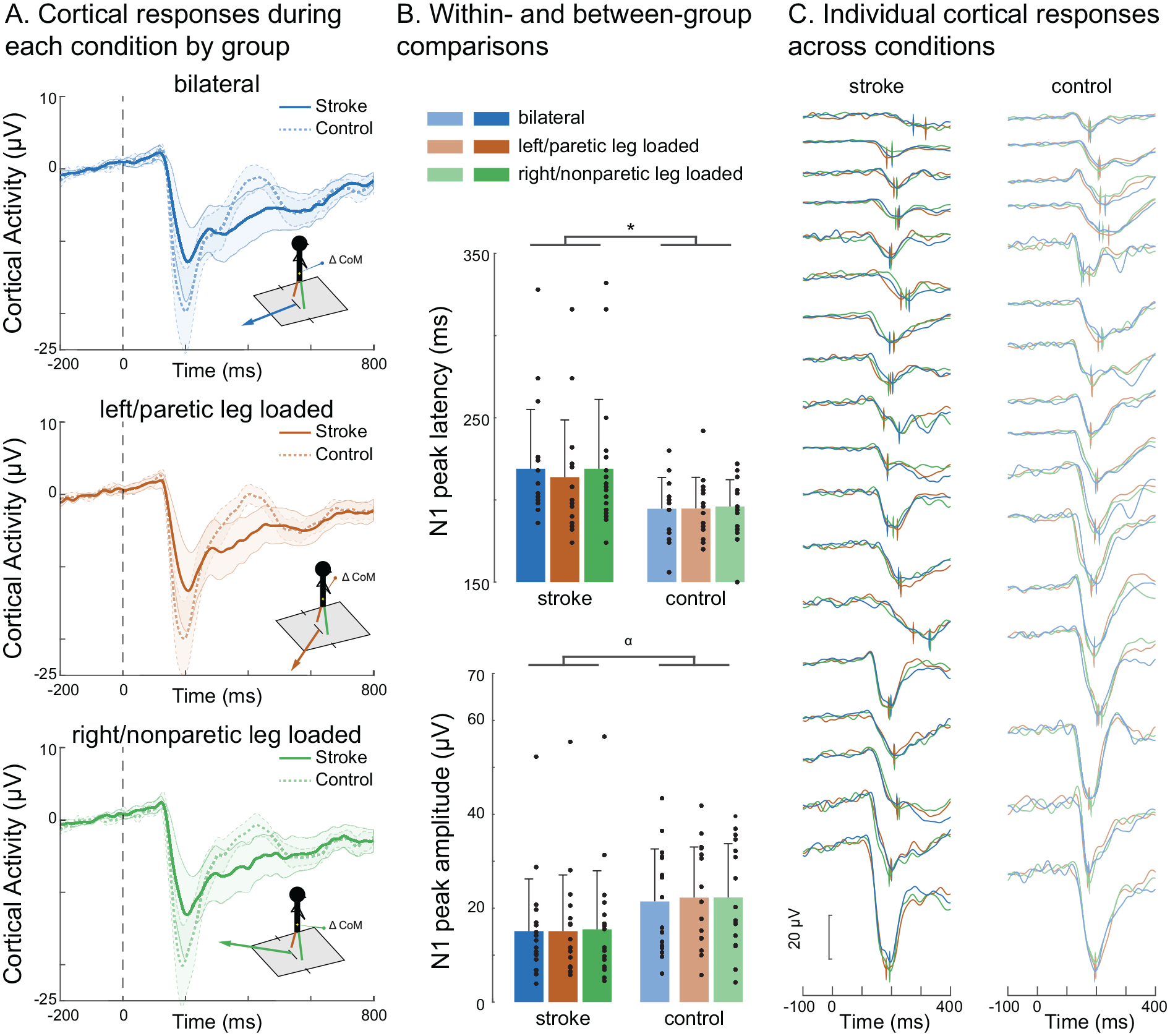
Cortical N1 responses within each balance perturbation conditions. (A) Cortical N1 response waveforms evoked during symmetrical bilateral (top) and lateralized perturbation conditions loading each the left/paretic (middle) and right/nonparetic legs (bottom) are shown in each the stroke and control groups. Mean ± standard error of the means are depicted in the shaded regions. (B) There was a main effect of group, in which stroke showed later N1 peak latencies (F1,32 = 5.27, *P = .028; top) and a trend toward lower N1 peak amplitudes (F1,32 = 2.932, α = .097; bottom) compared to controls. There were no main effects of condition for N1 peak latency (F1,32 = 1.063, P = .310; top) or amplitude (F1,32 = 1.47, P = .292; bottom) during the paretic-loaded condition (214 ± 35 ms), nonparetic-loaded (219 ± 42 ms) condition, or bilateral condition (219 ± 42 ms) within the stroke group among high between-participant variability (C) Means ± SDs depicted in figure bar plots.
Cortical N1 peak latency showed a main effect of group (F1,32 = 5.27, P = .028) but not for condition (paretic-loaded: 214 ± 35 ms; nonparetic-loaded 219 ± 42 ms; bilateral (219 ± 42 ms; F1,32 = 1.063, P = .310; Supplemental Table; Figure 3C). Regarding N1 amplitude, a non-significant trend toward a main effect of group (F1,32 = 2.932, P = .097) was observed, with no main effects of condition (F1,32 = 1.47, P = .292; Figure 3B).
Kinetic Reactions During Balance Recovery and Associations With Cortical N1 Response
There was a group-by-condition interaction effect on the CoP RoR (F2,29 = 3.054, P = .026), in which the stroke group had a slower CoP RoR than the control group only within the paretic-loaded condition (P = .012; Figure 4A and B). This group difference remained significant when excluding participants who completed the protocol with smaller perturbations (P = .029). Within the stroke group, the CoP RoR was faster in the nonparetic-loaded condition compared to each the paretic-loaded condition (P = .004) and the bilateral condition (P = .002), with no difference between the paretic-loaded and bilateral conditions (P = .312). Within the control group, the CoP RoR was slower in the bilateral condition compared to each the lateral loading conditions (left-loaded, P < .001; right-loaded, P = .012). The lateral loading conditions in controls were not different from each other (P = .312)
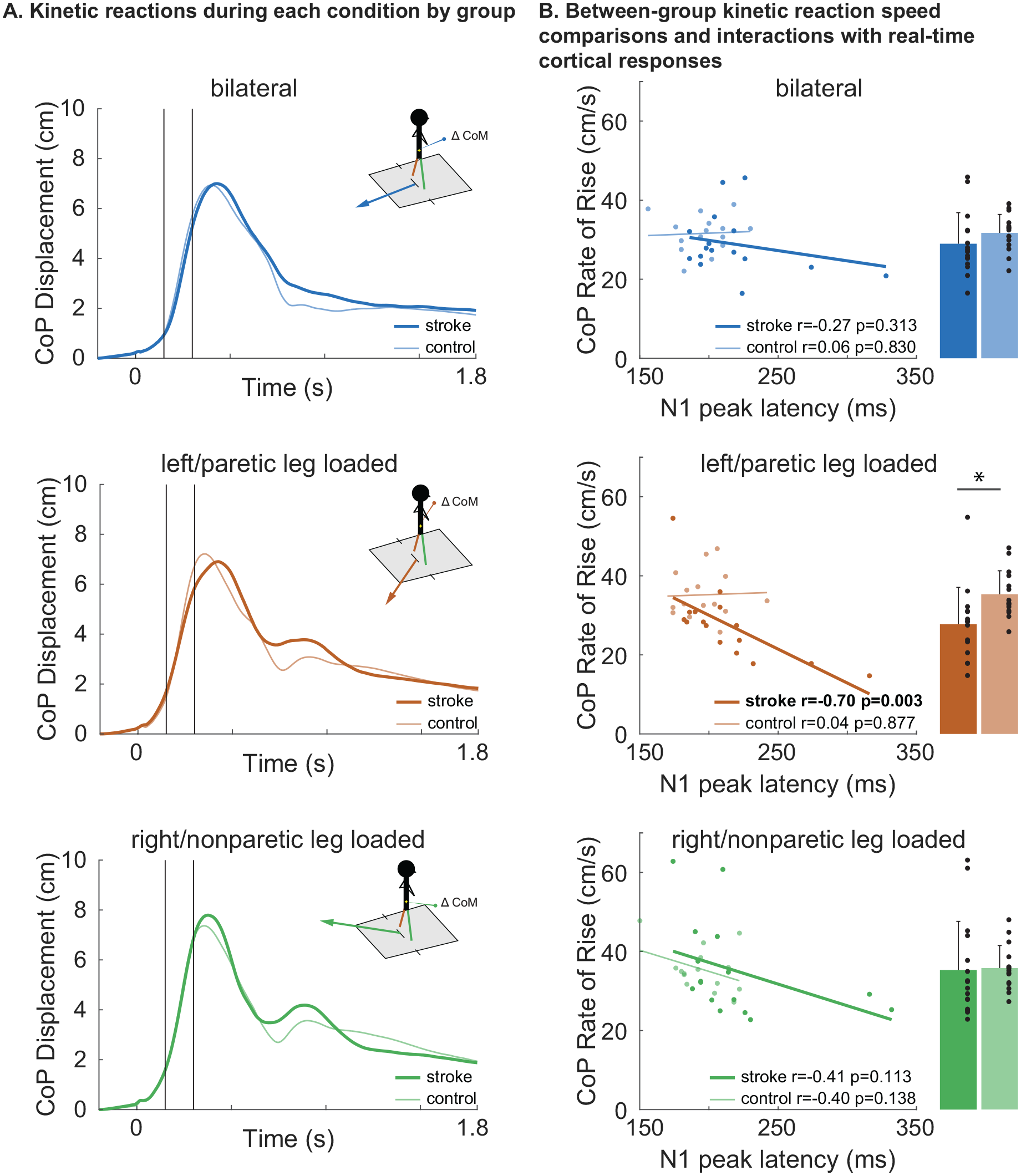
Balance recovery kinetics and relationship to time-synchronized cortical N1 response latency. (A) Center of pressure (CoP) displacement (y-axis) over time (x-axis) across bilateral, nonparetic-loaded, and paretic-loaded perturbation conditions are shown as mean waveforms for each group. The CoP rate of rise (RoR; CoP RoR) was calculated as the linear slope of the CoP displacement between 150 and 300 ms (black vertical lines) post-perturbation onset (time = 0). During paretic-loaded balance recovery (middle), participants with stroke showed slower CoP RoR compared to nonparetic-loaded recovery (bottom; P = .004) and controls (P = .012) and no difference compared to the bilateral condition (top). During nonparetic-loaded balance recovery, there was no difference in CoP RoR between groups (bottom). Mean ± SD are shown for CoP RoR. (B) During paretic-loaded balance recovery (middle), there was a relationship between later cortical N1 peak latencies and slower CoP RoR in stroke (r = −.70, P = .003) while no effect was observed in controls. During nonparetic-loaded recovery (bottom) and bilateral conditions (top), there was no relationship between cortical N1 latency and CoP RoR. No relationships were observed between N1 amplitude and CoP RoR in any condition or group (not shown).
Within the stroke group, later N1 peak latencies were associated with slower CoP RoR within the paretic-loaded condition (r = −.70, P = .003), but not in the nonparetic-loaded condition (r = −.41, P = .11) or the bilateral condition (r = −.27, P = .313; Figure 4B). These results are reported separately for each condition because (unlike the clinical metrics) the results differed across conditions. None of these associations were observed in the control group (all P > .138). When testing for group-by-N1 latency interaction effects on CoP RoR, there was a trend for interaction within the paretic-loaded condition (t = −1.803, P = .083) but not the nonparetic-loaded (t = −0.012, P = .991) or bilateral conditions (t = −0.705, P = .487). For N1 amplitude, there were no group-by-N1 amplitude interaction effects in any balance condition (all P > .598) or relationships with CoP RoR for any condition in either group (all P > .491).
Discussion
The observed findings are the first to demonstrate that clinical and kinetic balance dysfunction in people after stroke is related to delayed cortical N1 responses evoked during reactive balance recovery. Our reactive balance paradigm provided a well-controlled probe of cortical reactivity during a functionally-relevant, whole-body behavior, demonstrating that balance perturbations elicit slower, smaller cortical responses after stroke compared to age-similar controls. These findings suggest delayed and impaired cortical error assessment during balance reactions in people after stroke. While balance conditions loading the paretic leg resulted in slower kinetic reactions for balance recovery compared to controls,2-4 balance conditions that positioned the nonparetic leg for recovery enabled individuals post stroke to achieve similar kinetic reactions to their age-matched peers. Relationships between time-synchronized cortical response speed and kinetic reactivity during paretic-loaded balance recovery may reflect the constraints of rapid cortical engagement at the limits of paretic motor capacity (eg, paretic leg loading) that are masked when the nonparetic leg contributes to compensatory balance control (ie, nonparetic leg loading and bilateral loading conditions). Thus, individuals after stroke may be limited by their ability to process cortical responses to postural perturbations, which may encourage a shift from rapid reactions to anticipatory strategies for postural control that are largely ineffective for preventing falls.47-49
Impaired Cortical Responses With Dynamic Balance Deficits Post Stroke
Longer latencies and smaller amplitudes of peak cortical N1 responses in people with stroke (Figure 2A) are consistent with the presence of impaired cortical responses during balance recovery, with temporal cortical-behavioral relationships being driven by individuals with the most impaired balance and mobility function (Figure 2B). Cortical N1 peak latencies >300 ms in 2 individuals after stroke were longer than expected. Longer cortical response latencies could be explained by many factors, for example (1) stroke-induced disruptions in the propagation of somatosensory information and greater biomechanical displacement required to elicit similar somatosensory signaling to the cortex, (2) the stroke lesion disrupts cortical integration of actual somatosensory information with predicted somatosensation (ie, error assessment), or (3) a different neural source operating at longer latencies (eg, cerebello-cortical networks) 50 may predominantly contribute to the error-related negativity of cortical N1 responses in the most severely affected individuals. Consistent with this notion, at least 1 of the 2 individuals in the present study with the longest cortical N1 response latencies >300 ms showed more severe clinical impairment (Fugl-Meyer [LE] = 14) and both individuals showed lower balance and mobility function (mini-BESTest = 11 and 10; TUG = 20.04 and 28.19 s). Smaller and slower cortical responses, potentially reflecting impaired cortical error assesssment, 51 may be particularly detrimental during abrupt and challenging balance perturbations that elicit greater corticomotor drive for balance recovery compared to less abrupt balance perturbations. This is consistent with neurotypical individuals who display increases in functional connectivity between cortical activity and reactive lower limb motor responses with more challenging perturbations. 52 The present results indicate that even relatively small amplitude perturbations with feet-in-place responses elicit differences between stroke and control groups, which is advantageous for the study of individuals with severe poststroke impairment. Together, these findings suggest that the speed and effectiveness of sensorimotor error detection and information processing during balance recovery behavior is compromised in individuals with cortical and subcortical lesions.
The presence of brain-behavioral relationships primarily in the stroke group suggests that slowed cortical activity may play an increased role in balance behavior after stroke. Notably, controls also demonstrated a relationship between delayed cortical N1 response latencies and slower TUG performance (Figure 2B), suggesting the behavioral significance of the speed of cortical activity with speed-dependent balance control in older adults, and that stroke-related deficits may amplify these effects of aging on delayed cortical error assessment during balance. Interestingly, while prior studies found that cortical N1 response amplitudes were attenuated in older compared to younger adults,53,54 there were no reported differences in cortical N1 response latencies as a function of age within older adults. 54 Likewise, cortical N1 response magnitude but not latency associated with clinical balance confidence and cognitive function in neurologically-intact older adults.20,44 Similarly, we recently observed unique relationships between measures of N1 timing and amplitude and measures of balance and mobility in a group of individuals with Parkinson’s disease, but notably the cortical N1 response magnitudes and latencies were not different between older adults with and without Parkinson’s disease. 44 The network of brain areas contributing to the N1 response has not been assessed in stroke or older populations, which often recruit the cortex more broadly for challenging tasks requiring postural control. 18 While conclusions based on lesion location are limited in the present study, our results suggest that direct damage to regions contributing to the cortical N1 response (eg, SMA, anterior cingulate, parietal, sensorimotor cortex) or the interconnected brain regions represented by participants in the present study (Table 1) could affect balance error assessment and may play a limiting role in balance function after stroke. Together, the present findings reveal neurophysiologic features of cortical slowness in the context of balance behavior may be amplified in stroke neuropathology and is linked to balance and mobility dysfunction.
Lateralization of Balance Perturbations Did Not Affect Cortical Responses
Similar cortical N1 responses elicited during paretic versus nonparetic-loaded balance recovery conditions may reflect different neuromechanical features of balance recovery after stroke. While the lateralization of perturbations toward paretic and nonparetic legs generated asymmetrical limb loading during balance recovery (Figure 1), it was surprising that perturbation loading condition did not affect cortical responses (Figure 3A and B). While nonparetic-loaded cortical N1 response speeds may reflect relatively faster somatosensory error assessment, neural conduction, and/or cortical processing within the cortex (Figure 4), paretic-loaded cortical N1 response speeds may reflect heightened surprise or threat responses17,21 that occur with increased loading toward the less desirable leg for weight bearing and motor control.
7
The latter may explain the (non-significant) tendency for individuals post-stroke to show faster cortical N1 response latencies during paretic-loaded conditions (Figure 3B
Impaired and Compensatory Post-Stroke Kinetic Reactions Revealed During Lateralized Balance Recovery
The present findings provide evidence that people post stroke can achieve kinetic reactive balance performance that is not different from their age-matched peers when they are mechanically positioned to compensate with the nonparetic leg. While the stroke group demonstrated slower kinetic balance reactions during paretic loading compared to controls (Figure 4), they showed faster and comparable kinetic balance reactions to their age-matched peers during nonparetic loading. This finding builds upon previous research showing slower paretic leg balance reactions,2-4 yet effective compensation through increased nonparetic leg postural reliance after stroke. 7 Controlling for biomechanical differences and balance challenge presented by medial-lateral and anterior-posterior directional postural perturbations 56 (eg, adopting tandem stance in baseline standing posture) could exaggerate the effect of paretic loading in anterior-posterior conditions. The present findings provide a foundation for future studies to test whether therapeutic strategies aimed at accelerating nonparetic leg balance reactions could effectively improve the post-stroke balance recovery ability, particularly in individuals with limited recovery potential of the paretic lower limb. Future studies are needed to test whether the relatively small perturbations and feet-in-place reactions used in the present study extend to balance reactions requiring reactive stepping,14,15 particularly in individuals after stroke who may present with conflicting preferences to both weight bear and step with the nonparetic leg.
Paretic-Loaded Balance Recovery Reveals Cortical-Kinetic Interactions After Stroke
Paretic-loaded balance recovery revealed time-synchronized relationships between delayed cortical N1 responses and slower speed of corrective kinetic reactions (Figure 4). Cortical-kinetic relationships were absent in controls, which could potentially be explained by differences in anticipatory posture between groups. For example, people with stroke may increase their reliance on anticipatory postural control given their delayed cortical responses during reactive balance. However, although participants in the present study were expecting an upcoming perturbation occurrence and were aware of the possible directionality (eg, either leg loaded and/or spatial predictability), they were unlikely able to utilize anticipatory control to effectively adjust posture in accordance with the precise timing that varied between trials and pseudorandomized direction of each perturbation. Thus, each of the perturbation conditions likely present similar anticipatory control within-participants and increased demand on reactive strategies for balance recovery. The specificity of the observed cortical-kinetic relationships during paretic-loaded balance recovery therefore suggests that positioning the paretic leg for balance recovery may reflect a difference in the neural strategy of reactive postural control during a more difficult condition compared to conditions that may be less difficult post stroke. A wide range of factors may explain within-group differences in cortical-kinetic relationships in the stroke group, including between-limb differences in somatosensory input, neural conduction, multisensory integration, cortical processing speed, specific psychological or cognitive constructs, or specific changes in neuromechanical strategies (eg, optimizing stability versus efficiency).
57
The absence of these relationships in controls suggest that brain lesions could impact cortical recruitment and activity in a manner that affects balance behavior, and that this might be easier to observe in balance behaviors that are more difficult. While not statistically significant (P = .11), a similar relationship between cortical N1 responses and kinetic reactions during the nonparetic-loaded condition is interesting because it suggests that individuals post stroke may achieve similar reactive balance performance through different neuromechanical strategies compared to their age-matched counterparts, potentially with compensatory use of their nonparetic leg (Figure 4B
Limitations
The balance perturbations in the present study were delivered with temporal unpredictability (ie, participants did not know the exact time of the perturbation occurrence) and spatial unpredictability (ie, direction of the perturbation was unknown), there is a possibility that participants increased anticipation of upcoming perturbations. The down-scaling of perturbation parameter magnitudes, as was necessary in the present study for severely balance-impaired participants, elicits slower kinetic balance responses. While we previously observed no effect of perturbation magnitude on N1 latencies when scaled in this manner in older adults, 20 the lower acceleration of balance perturbations in some participants could have influenced the speed of kinetic and cortical responses. There is limited clinical information regarding impairment level (eg, Fugl Myer scores) in the stroke group in the present study, which could potentially affect balance responses and features of the evoked N1 response through differences in standing posture. Standing postural symmetry was not standardized in the present study due to the severity of lower limb hemiparesis in some participants and the maintenance of an uncomfortable and non-preferred posture could introduce a dual-task effect, which could confound the cortical N1 amplitude. 58 While it is challenging to control for postural asymmetries in individuals after stroke, investigation of the within-session effects of changes in postural symmetry on cortical and biomechanical responses to balance perturbations could shed light on the effects of postural asymmetry on neural and biomechanical balance control, particularly as maintaining symmetry may introduce a cognitive dual-task challenge in some people after stroke. While the average ages of participants in each the stroke and control groups did not differ (Table 2), participants in the stroke group presented with a wider age range than those in the control group which should be considered in interpretations of the present results. Real-time EEG, EMG, and kinetic data were monitored visually in real-time in the present study to ensure participants returned to a relaxed and stable baseline postural state before each trial. Nonetheless, if participants were able to predict upcoming perturbations, then this would likely result in anticipatory changes in pre-perturbation postural stability, as has been shown in prior studies involving balance and gait.47-49 Subtle trial-to-trial differences could have gone undetected by experimenters monitoring these data in the present study and others that have implemented similar perturbation-based protocols.13-15,20,37,44,53,59
Conclusions
Our results demonstrate a link between delayed cortical activity and slowed kinetic and clinical balance function after stroke, particularly under challenging postural conditions that necessitate the paretic leg for balance recovery. These findings highlight temporal features of evoked cortical N1 responses as a biomarker of clinically-relevant balance behavior, offering a possible neural target for rehabilitation efforts during stroke recovery. Future studies may reveal that slower cortical and kinetic reactivity necessitate a shift from rapid reactions to anticipatory control for balance recovery behavior after stroke-induced lesions.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683241282786 – Supplemental material for Delayed Cortical Responses During Reactive Balance After Stroke Associated With Slower Kinetics and Clinical Balance Dysfunction
Supplemental material, sj-docx-1-nnr-10.1177_15459683241282786 for Delayed Cortical Responses During Reactive Balance After Stroke Associated With Slower Kinetics and Clinical Balance Dysfunction by Jacqueline A. Palmer, Aiden M. Payne, Jasmine L. Mirdamadi, Lena H. Ting and Michael R. Borich in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We would like to acknowledge Michelle Goto, Pam Inglett, and Annie Goldman for their assistance with data collection for this project.
Author Contributions
Jacqueline A. Palmer: Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Software; Writing—original draft; Writing—review & editing. Aiden M. Payne: Data curation; Investigation; Methodology; Software; Writing—review & editing. Jasmine L. Mirdamadi: Formal analysis; Writing—review & editing. Lena H. Ting: Conceptualization; Funding acquisition; Writing—review & editing. Michael R. Borich: Conceptualization; Funding acquisition; Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health [F32HD096816 and K99AG075255] to JP [F32HD105458] to JLM [F32MH129076, Udall Pilot award P50NS098685] to AMP [K12HD055931] to MRB [5T90DA032466, 1P50NS098685, and R01 HD46922] to LHT and [R01 AG072756] to LHT and MRB.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
