Abstract
Background
Frequent and objective monitoring of motor recovery progression holds significant importance in stroke rehabilitation. Despite extensive studies on wearable solutions in this context, the focus has been predominantly on evaluating limb activity. This study aims to address this limitation by delving into a novel measure of wrist kinematics more intricately related to patients’ motor capacity.
Objective
To explore a new wearable-based approach for objectively and reliably assessing upper-limb motor ability in stroke survivors using a single inertial sensor placed on the stroke-affected wrist.
Methods
Seventeen stroke survivors performed a series of daily activities within a simulated home setting while wearing a six-axis inertial measurement unit on the wrist affected by stroke. Inertial data during point-to-point upper-limb movements were decomposed into movement segments, from which various kinematic variables were derived. A data-driven approach was then employed to identify a kinematic variable demonstrating robust internal reliability, construct validity, and convergent validity.
Results
We have identified a key kinematic variable, namely the 90th percentile of movement segment distance during point-to-point movements. This variable exhibited robust reliability (intra-class correlation coefficient of .93) and strong correlations with established clinical measures of motor capacity (Pearson’s correlation coefficients of .81 with the Fugl-Meyer Assessment for Upper-Extremity; .77 with the Functional Ability component of the Wolf Motor Function Test; and −.68 with the Performance Time component of the Wolf Motor Function Test).
Conclusions
The findings underscore the potential for continuous, objective, and convenient monitoring of stroke survivors’ motor progression throughout rehabilitation.
Introduction
Stroke is a major cause of impairments, affecting nearly 800 000 individuals in the United States each year. 1 Following a stroke, about 80% of survivors experience some degree of upper-limb paresis, 2 which typically affects the limb contralateral to the lesioned brain hemisphere. 3 Post-stroke motor impairments are characterized by weakness with limited capacity to perform functional movements necessary for activities of daily living (ADLs), which diminishes independence and health-related quality of life. 4
In conventional clinical settings, monitoring the recovery of motor ability is often achieved by assessing motor impairment and/or functional limitations using validated tools, such as the Fugl-Meyer Assessment of the Upper Extremity (FMA-UE) and Wolf Motor Function Test (WMFT). However, these assessments have been largely criticized for evaluating patients’ motor ability within the structured clinical setting, without incorporating observations of their motor skills in naturalistic environments. 5 Additionally, the ordinal scales of these standardized tools often suffer from ceiling and floor effects, making it challenging to study individuals with mild or severe impairments. 6 More importantly, these tools are impractical to administer on a regular basis, as they are time-consuming (eg, may take an entire clinical visit covered by payers) and clinically burdensome (eg, requiring the presence of trained clinicians to administer these assessments, with ongoing training necessary to maintain accuracy).7 –9 As a result, particularly in the United States, these outcome measures are too often collected only at the beginning and end of an intervention covered by third-party payers. 10 This presents a critical problem as rehabilitation specialists obtain only a significantly under-sampled view of their patients’ recovery trajectories,8,11 impeding their ability to evaluate patients’ responses to treatments12,13 and thus, hindering the support of more personalized interventions to each patient’s unique circumstances.8,14
To reconcile these challenges, researchers have investigated various technology-driven approaches to facilitate more frequent and objective assessments of motor ability outside the clinical setting. For example, prior in-laboratory studies have demonstrated that kinematic information extracted from video cameras or inertial measurement units (IMUs) during patients’ performance of a predefined set of motor tasks exhibits robust correlations with validated clinical measures, such as the FMA-UE11,15 and the Functional Ability component of WMFT (WMFT-FA). 7 These investigations have envisioned scenarios where patients could perform these motor tasks in their home settings, allowing for more frequent and objective assessment of motor capacity throughout the rehabilitation process. However, while these studies demonstrate technical feasibility, their application in real-world situations, especially in the absence of trained personnel, may be impractical and could result in inaccurate assessments. Even minor deviations from the prescribed methods of performing motor tasks, such as misconfiguring the distance or direction of the target during point-to-point reaching movements (eg, a task pertaining to WMFT), could impact the precision of kinematic variables and, consequently, the accuracy of the assessment. Hence, researchers have been advocating for methods supporting task-free evaluations of motor ability, utilizing continuous inertial data collected during patients’ naturalistic engagement in ADLs. 14
Unfortunately, much of the research on wearable-based, task-free motor assessment has predominantly centered on evaluating limb activity, quantifying the intensity of stroke-affected limb movement, often in comparison to the less-affected limb, using wrist-worn accelerometers.16–19 However, these measures exhibit only moderate correlations with and limited responsiveness to standardized assessments of motor capacity. 20 Researchers have explained this disparity by highlighting that measures of limb activity solely represent a facet of patients’ motor performance (ie, how much patients actually move their arms), whereas standardized clinical assessments evaluate patients’ motor capacity/ability (ie, what patients are capable of doing). 5 To overcome this limitation, a few studies have attempted to explore kinematic variables obtained from an array of wearable IMUs during real-world ADLs that may be more closely linked to patients’ motor capacity.13,21–23 Unfortunately, the majority of these studies involved a large number of wearable sensors on the body, typically ranging from 12 to 17 IMUs, making the application of these studies rather impractical in a clinical context. Additionally, these studies have primarily focused on relatively simple, high-level upper-limb kinematic characteristics, such as the range of motion 21 and the distribution of the hand position, 24 for processing the complex movements involved in ADLs. Consequently, these variables exhibited only modest correlations with standardized measures of motor capacity.
In this study, we investigate a novel method to objectively and reliably assess upper-limb motor ability in stroke survivors by analyzing the kinematics of upper-limb movements performed during ADLs using a single IMU on the stroke-affected wrist. Analyzing upper-limb kinematics from continuous inertial data during uncontrolled and naturalistic upper-limb movements poses a significant challenge due to the diverse and variable ways in which patients perform their ADLs.25,26 To address this challenge, we focus our analysis on three-dimensional (3D) linear point-to-point upper-limb movements occurring during ADLs, a fundamental building block of goal-directed upper-limb activities that are common across various types of ADLs.27,28 To validate our approach, we collected data from 17 stroke survivors, who performed a set of unscripted ADLs naturalistically and continuously within a simulated home setting. Our primary hypothesis is that kinematic analysis of point-to-point movements (ie, a form of motor performance) offers insights closely linked to patients’ motor capacity. We further hypothesize that the extracted kinematic variables would demonstrate strong internal reliability, construct validity, and convergent validity with respect to established clinical assessments of motor capacity.
Methods
Participants
Participants were recruited from Spaulding Rehabilitation Hospital in Boston, USA. The study inclusion criteria were as follows: (1) age between 18 and 80 years, (2) experienced a stroke more than 6 months prior to the visit, and (3) exhibits moderate-to-mild impairment in the upper limb affected by the stroke (ie, FMA-UE score >35). This cut-off point for FMA-UE was selected because the conventional cut-off points for moderate impairment, as suggested by Woodbury et al. (ie, 33 out of 66), 29 might inadvertently include individuals who would likely encounter difficulties with many ADLs involving distal functionality (see Experimental Protocols for details). Therefore, we referenced the midpoint of the moderate range identified by Woodbury et al, while assuming full points for two reflex items (ie, biceps and triceps reflexes), as we anticipated that most within this range would achieve maximum scores, resulting in a total of 35 points. The study excluded individuals who (1) were incapable of raising the upper limb against gravity (ie, >30° of flexion and abduction), (2) exhibited severe upper-limb spasticity hindering passive finger movement (ie, Modified Ashworth Scale >3), (3) were unable to don and doff wearable sensors, either independently or with the assistance of a caregiver, and (4) showed significant cognitive impairments (Mini-Mental Status Examination <23). All participants provided written informed consent, which was approved by the Institutional Review Board of Mass General and Brigham Hospital (Protocol # 2019P002612).
Clinical Assessment of Upper Limb Motor Capacity
The primary clinical measures of this study were the FMA-UE and WMFT scales. The FMA-UE assesses the overall motor impairment severity in the stroke-affected upper limb. 30 The WMFT encompasses two components: Performance Time (PT) and Functional Ability (FA), which evaluate the extent of functional capacity and quality of movement, respectively. 31 As secondary measures, participants’ self-reported motor performance was collected using the Motor Activity Log (MAL), 32 consisting of two items: the Amount of Use (MAL-AoU) and Quality of Movement (MAL-QoM).
Experimental Protocol
Research staff helped study participants wearing two IMUs on their wrists (Shimmer3, Shimmer Research Inc., USA). We opted to collect data from both wrists to calculate benchmark limb activity measures for comparative analysis (see Wearable-Based Measures of Upper-Limb Activities for details). However, it is important to note that we only utilized the data acquired from the stroke-affected limb for our analysis aiming to identify kinematic variables closely linked to motor capacity. This decision was made because the clinical measures of motor capacity (ie, FMA-UE and WFMT) are also primarily conducted on the stroke-affected limb. Each IMU sampled three-axis acceleration and three-axis angular velocity at 50 Hz. Participants performed up to 11 unscripted ADLs within a simulated home environment at Spaulding Rehabilitation Hospital, as summarized in Table 1. The layout of the simulated home is depicted in Supplemental Figure S1. The selection of these ADLs was informed by prior studies that have also explored the validation of wearable devices for analyzing upper-limb performance within controlled laboratory settings.18,19 Specifically, these ADLs were chosen to encompass a broad range of upper-limb movements required during motor tasks, including both bimanual and unimanual activities, as well as gross-arm and fine-hand movements. Participants were not obligated to perform ADLs that they did not regularly undertake in their real-world scenarios. They received instructions on how to find the necessary materials to perform these ADLs within the simulated home (eg, locating a grocery bag in the kitchen, finding a toothbrush in the bathroom, etc.). Each participant received a sheet of paper listing the 11 ADLs they were expected to complete. Participants were directed to perform the ADLs naturally and independently, mirroring their behaviors in their own homes. The research staff intentionally refrained from providing any guidance on how to perform the ADLs to avoid influencing their inherent motor behavior. The participants’ motor performance was also recorded using a set of four wide-angle cameras, which included two egocentric and two exocentric views. The positions of the two exocentric cameras within the simulated apartment are illustrated in Supplemental Figure S1.
Motor Tasks Resembling ADLs Performed by Study Participants.
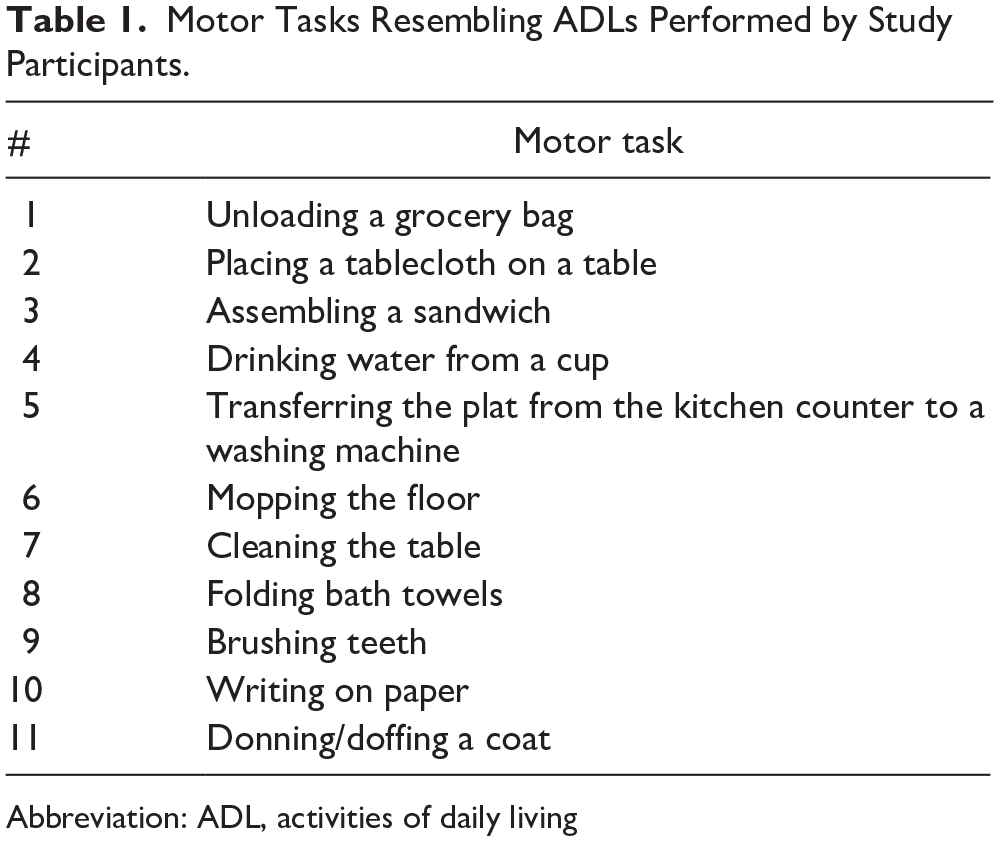
Abbreviation: ADL, activities of daily living
Analyzing Kinematics of Real-World Upper-Limb Movements
In this study, we utilize point-to-point movements performed during naturalistic ADLs to analyze the upper-limb kinematics. Point-to-point upper-limb movements refer to a multi-joint 3D movement that involves linear motion of the wrist from one position to another.27,28,33 It is one of the most frequently executed actions of the arm34,35 and serves as a fundamental building block of daily activities involving goal-directed interactions with the environments (eg, reaching to grab an object, transferring the object, and repositioning the hand back to the body after the interaction).27,28 More importantly, point-to-point movements possess kinematic characteristics that remain consistent across different ADLs in neurologically intact humans and even in other species.36,37 This implies that these movements could be detected irrespective of the specific type of ADL being performed or individual variations in their execution.27,28
Point-to-point movements are also an ideal candidate for wearable-based kinematic analysis in the context of stroke rehabilitation. The wrist kinematics of point-to-point movements, which can be captured using wrist-worn inertial sensors, have revealed distinct motor phenotypes related to stroke motor impairment, although the studies have been confined to laboratory settings.38–40 This high information density in wrist movements is because the hand can considered as the end effector that the central nervous system aims to control. 41 As a result, abnormal movements of proximal body parts and joints, such as the trunk, shoulder, and elbow, have a direct effect on the kinematics of the wrist. 11
Our analysis encompassed the complete and uninterrupted sensor data, capturing not only the patients’ execution of motor tasks but also their organic movements during transitions between these tasks. These transitional movements, such as walking from the kitchen to the bathroom or performing sit-to-stand movements from a chair, were included as they naturally occur in real-world scenarios. Two researchers reviewed the video recordings of study participants performing the ADLs and annotated the occurrences of point-to-point movements. To analyze the kinematics of point-to-point movements, we derived three-axis, gravity-free acceleration data in a global coordinate system using a Kalman filter-based sensor orientation tracking algorithm that fuses the local-coordinate acceleration and gyroscope data. 42 To minimize the effect of orientation drifts over time, we rotated the coordinate system for each point-to-point movement, aligning the x-axis with the direction of the motion. This direction was computed by applying Principal Component Analysis on the 3D velocity time-series and selecting the first principal component, which encapsulates the most significant variation in movements (ie, the point-to-point dimension).43,44 The global-coordinate acceleration time-series along the selected dimension was numerically integrated to yield the 1D velocity time-series.
The 1D velocity time-series was then segmented at zero crossings to generate movement segments.11,45 The velocity profiles of these movement segments exhibit varied morphology and temporal patterns in stroke survivors 38 due to impaired motor planning and control (eg, feedforward and feedback control).35,46 We extracted kinematic variables relevant to stroke motor impairments from each point-to-point movement. These variables include peak, average, root mean square, variance, and coefficient of variation of traveled distance, absolute velocity, acceleration, and jerk.11,38 We also investigated the ratio between average and peak velocity, skewness of velocity distribution, number of peaks of velocity, duration of movement segments, and number of movement segments.11,38 Subsequently, these variables from point-to-point movements were aggregated for each patient using statistical functions, such as mean, standard deviation, interquartile range, median, minimum, maximum, and 10th and 90th percentiles. 11 This comprehensive process yielded a total of 110 variables.
Investigation of the Reliability of Kinematic Variables
To analyze the reliability of the extracted kinematic variables, the performed reaching movements were divided into two groups, alternating their occurrence during the motor tasks. For instance, one group encompassed odd repetitions of the performed reaching movements (eg, first, third, fifth reaches, etc.), while the other comprised even repetitions (eg, second, fourth, sixth reaches, etc.). This data division approach was chosen over the conventional splitting of data into the first and second halves of the time duration in order to mitigate the impact of patients’ fatigue on kinematic variables. Each kinematic variable was derived from the two groups and subsequently analyzed for reliability using the two-way mixed intra-class correlation coefficient (ICC(3,1)). We used a threshold of 0.90 for ICC to identify kinematic variables demonstrating excellent reliability. 47
Statistical Analysis for Convergent Validity
Within this pool of reliable variables, we sought to identify those consistently demonstrating strong correlations with the clinical measures (ie, convergent validity). The primary analysis was to evaluate the associations using Pearson’s correlation analysis with standardized clinical measures of motor impairment (ie, FMA-UE), movement quality (WMFT-FA), and functional capacity (WMFT-PT). As a secondary objective, we examined the associations with the measures of motor performance (ie, MAL and sensor-based limb activity measures). The overall data analytic pipeline is depicted in Supplemental Figure S2.
Wearable-Based Measures of Upper-Limb Activities
We derived the established measures of limb activity, namely the intensity ratio and use ratio,16 –19 using the continuous accelerometer data collected throughout the experiment. Detailed computational derivations for these measures are provided in the Supplemental Materials. These measures are designed to assess the level of engagement of the stroke-affected limb during ADLs, reflecting a distinct construct from our primary focus on motor ability. Although our primary objective differs from what these measures are intended to capture, their inclusion allows for a more comprehensive analysis of the kinematic variables.
Results
Initially, 19 stroke survivors were enrolled in the study, a sample size chosen for convenience, drawing from prior research that similarly explored correlations between upper-limb kinematics and motor capacity. 11 However, data collected from two participants were compromised due to technical complications. Consequently, the final analysis included data gathered from 17 subjects. Table 2 summarizes the demographic and clinical characteristics of the study participants. Study participants performed 10.3 ± 0.82 motor tasks over a duration of 49.7 ± 10.4 minutes (mean ± standard deviation). Throughout the experiment, each participant performed 374.2 ± 235.6 point-to-point movements using the stroke-affected limb. The average duration of these movements was 0.48 ± 0.09 seconds.
The Demographic and Clinical Characteristics of the Study Participants.
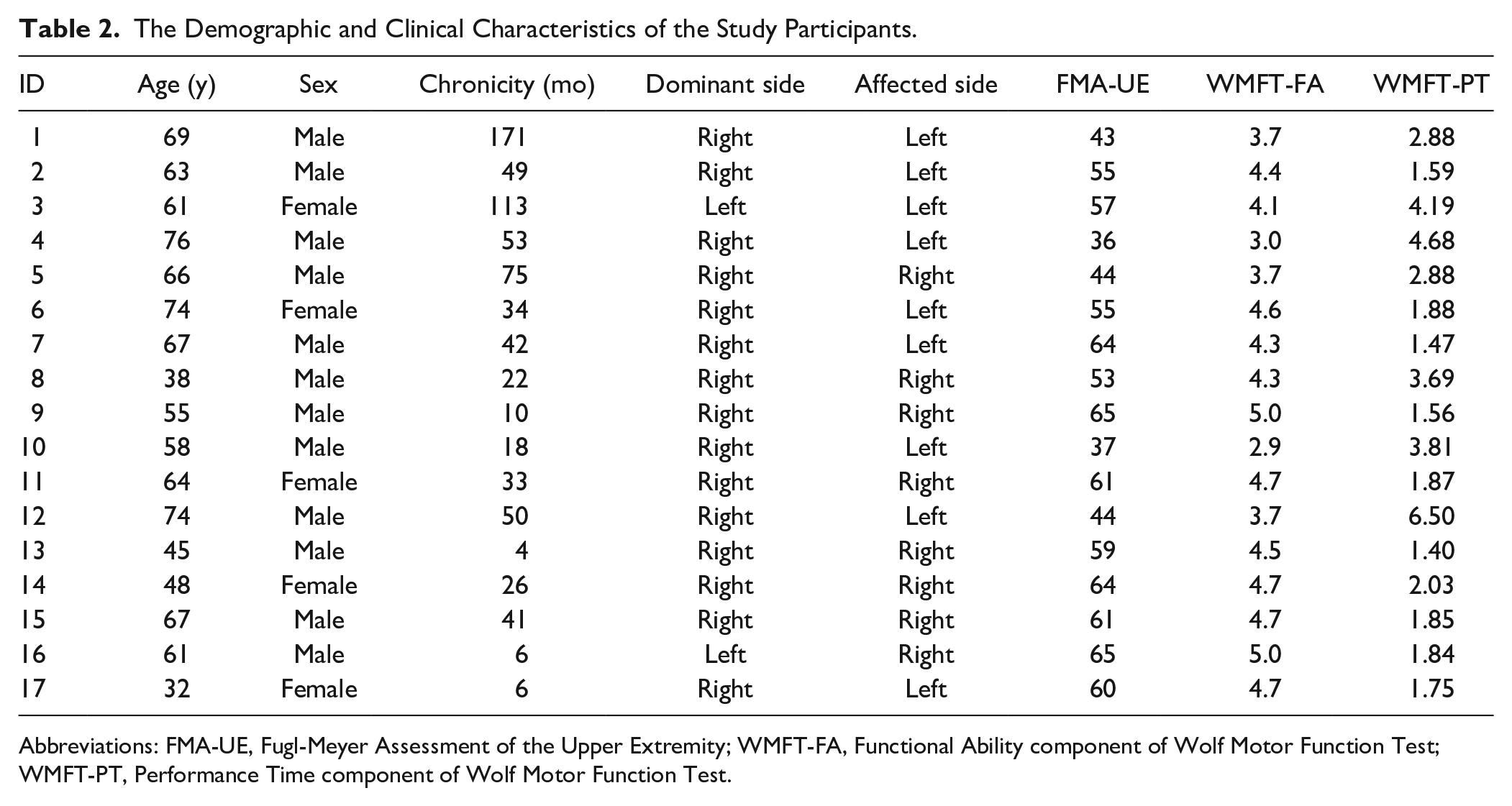
Abbreviations: FMA-UE, Fugl-Meyer Assessment of the Upper Extremity; WMFT-FA, Functional Ability component of Wolf Motor Function Test; WMFT-PT, Performance Time component of Wolf Motor Function Test.
Kinematic Analysis of Point-to-Point Movements
Among the 110 kinematic variables considered in this study, 25 variables exhibited excellent test-retest reliability (ie, ICC > 0.90).
47
Within this pool of reliable variables, we identified a particular variable that consistently demonstrated strong correlations with the primary and secondary clinical measures. The variable identified, denoted as
Table 3 presents a summary of correlations observed between
Pearson’s Correlation Coefficients Between the Clinical Assessments (ie, FMA-UE, WMFT, and MAL) and the Identified Kinematic Measure (
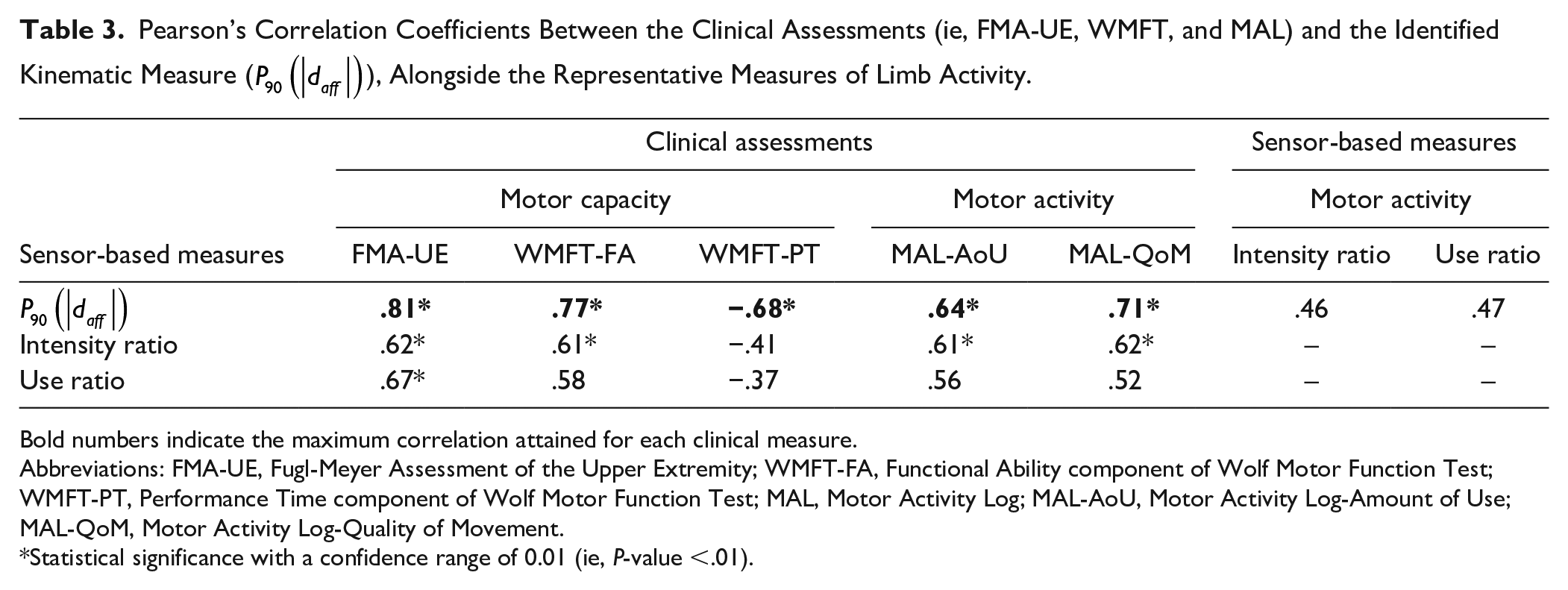
Bold numbers indicate the maximum correlation attained for each clinical measure.
Abbreviations: FMA-UE, Fugl-Meyer Assessment of the Upper Extremity; WMFT-FA, Functional Ability component of Wolf Motor Function Test; WMFT-PT, Performance Time component of Wolf Motor Function Test; MAL, Motor Activity Log; MAL-AoU, Motor Activity Log-Amount of Use; MAL-QoM, Motor Activity Log-Quality of Movement.
Statistical significance with a confidence range of 0.01 (ie, P-value <.01).
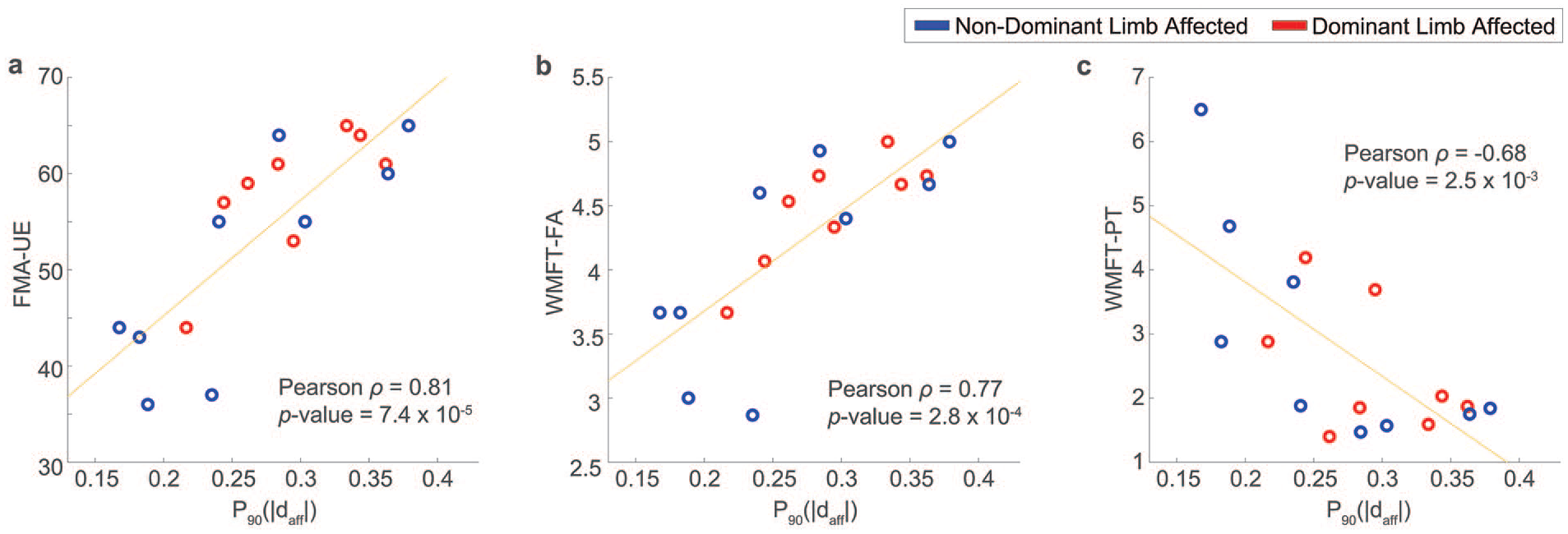
Correlations between
In Figure 2, the reliability of
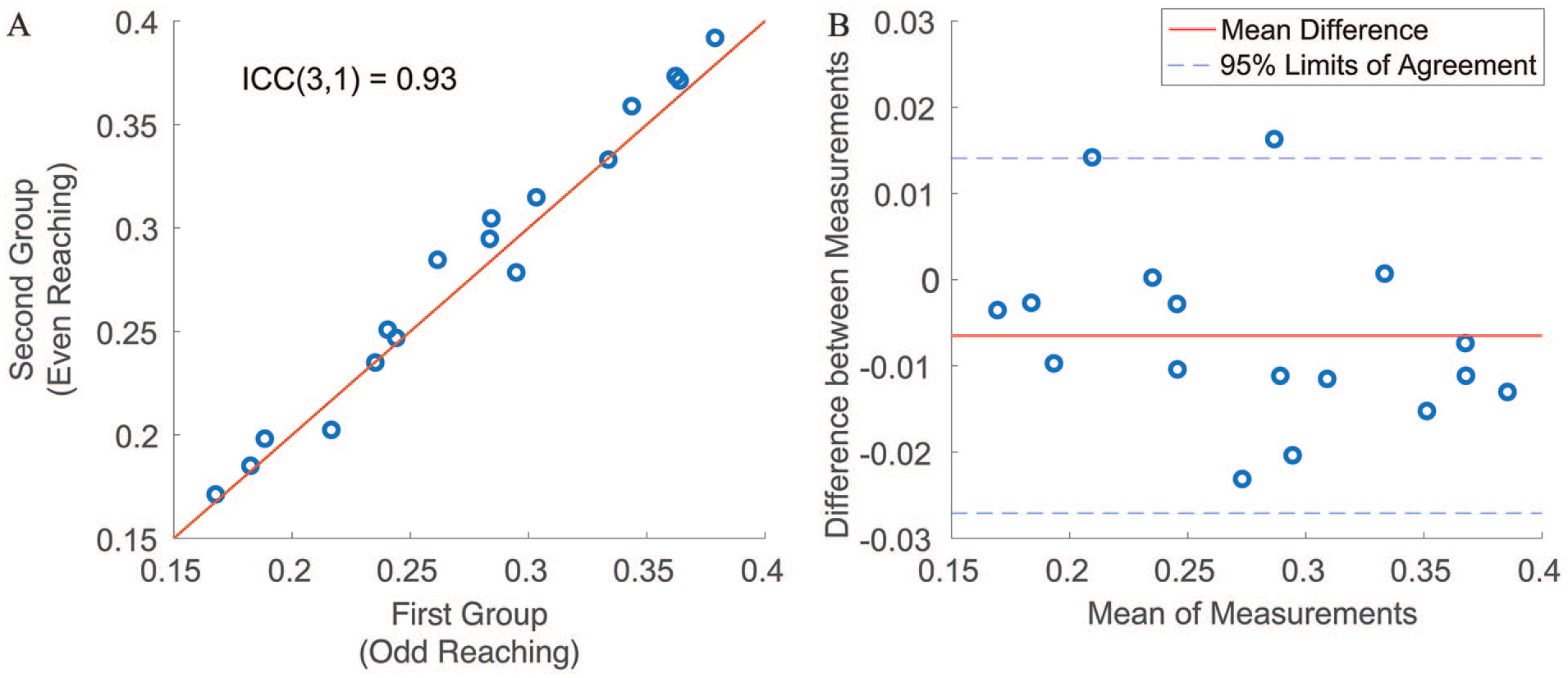
(A) A scatter plot to show the reliability of
Figure 3 illustrates the distributions of the traveled distance of movement segments (ie,
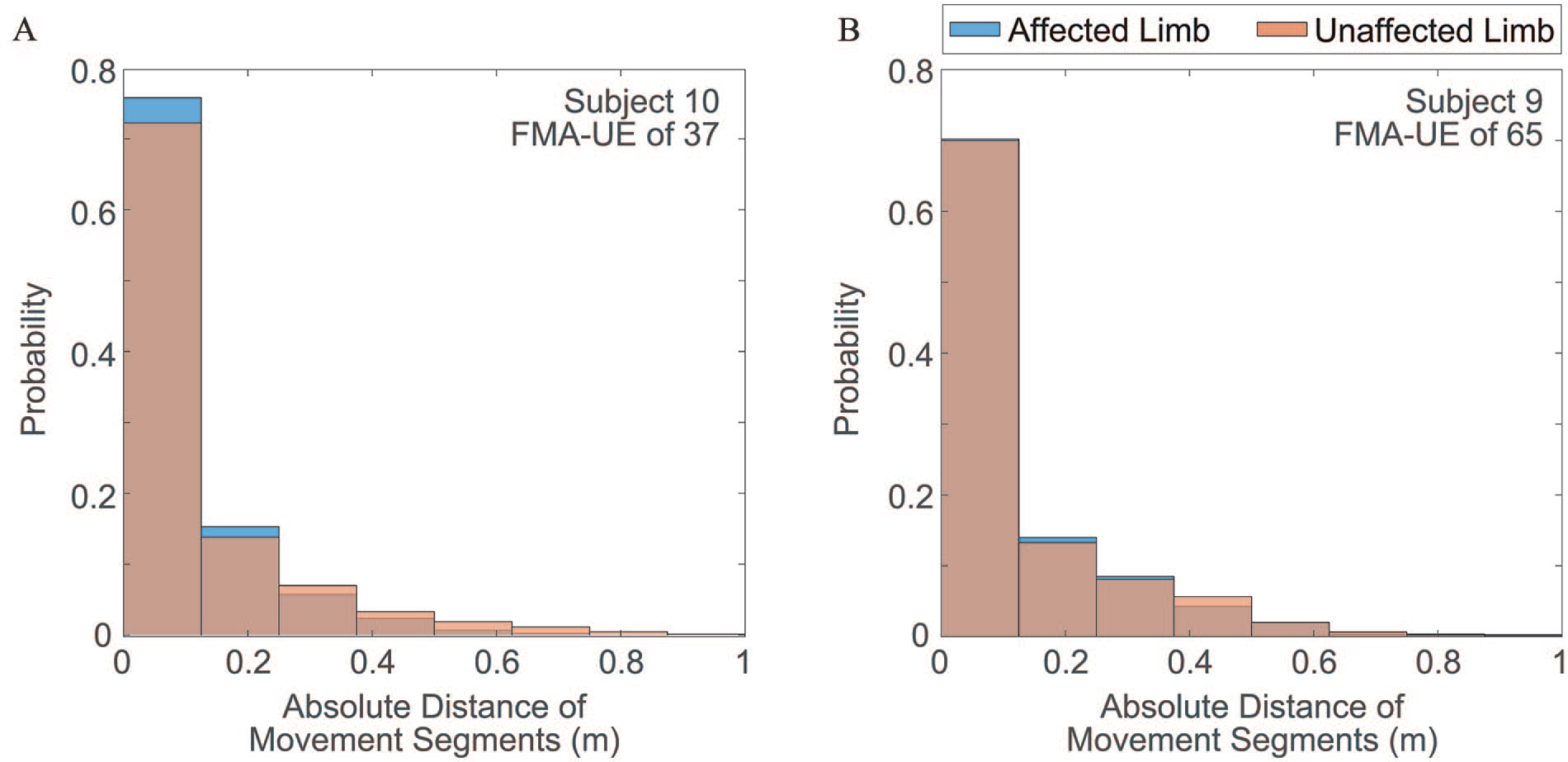
Distributions of movement segment distance extracted from the upper limbs of two participants: (A) one moderately impaired participant with an FMA-UE score of 37 and (B) one mildly impaired participant with an FMA-UE score of 65.
Conversely, Figure 3B, derived from a mildly impaired subject, displays comparable distributions between the two limbs, with an effect size of −0.022 (CI: −0.11 to 0.062), although the distribution of the limb affected by the stroke appears marginally lower than that of the less-affected limb. This suggests that the kinematic characteristics of the two limbs, as represented by the distribution of movement segment distance, become more alike with milder motor impairments. Interestingly, the disparity between the less-affected limbs of the moderately impaired and mildly impaired individuals was also small, with an effect size of −0.043 (CI: −0.12 to 0.034). This is intuitive as the kinematic characteristics of the less-affected limbs of the two subjects, resembling almost normal characteristics, should exhibit similar distribution profiles. Conversely, as expected, the effect size between the affected limbs of the two subjects was considerably large, measuring at −0.18 (CI: −0.29 to −0.077). Taken together, these results significantly bolster the evidence supporting the strong construct validity of
Discussion
This study introduces a new approach with potential application to assessing upper-limb motor ability in stroke survivors using an ecologically valid kinematic measure of real-world upper-limb movements. Notably, the identified kinematic variable,
In Table 2, we observe that
Table 2 also highlights an interesting finding that
The findings presented herein also align with prior studies on kinematic characteristics of point-to-point motions in stroke survivors, although these studies were constrained to analyzing 2D point-to-point movements in the transverse plane within controlled laboratory settings, rather than examining naturalistic 3D motions occurring during ADLs. A pivotal kinematic indicator of motor impairment following a stroke is the smoothness—or conversely, jerkiness—of movements.38
–40 Specifically, spasticity in stroke survivors with severe motor impairments restricts the range of motion and speed of upper-limb movements.51,52 As a consequence, the resulting upper-limb movements become fragmented, with smaller movement segments with reduced traveled distance, as shown in Figure 3A, demonstrating jerkiness in the performed movements. Conversely, stroke survivors with milder impairments tend to produce movement segments with larger distances, as observed in Figure 3B. This leads to smoother point-to-point motions with fewer movement segments and a greater range of motion and speed. In fact, we have noted that the traveled distance of movement segments (ie,
Leveraging kinematic variables of performed upper-limb movements to aid in the assessment of motor recovery offers a clear advantage. Firstly, due to its continuous nature,
This study presents several limitations, which also highlight important directions for future research. Firstly, it is important to acknowledge that our study had a relatively small sample size and did not examine the responsiveness of the proposed upper-limb kinematic measure to changes in patients’ motor ability over time. To ensure generalizability and responsiveness, future investigations should encompass a longitudinal study with larger and more diverse cohorts. Moreover, with such an expanded dataset, a future study could explore the use of machine learning algorithms to integrate multi-dimensional kinematic variables and other relevant clinical variables to improve the validity of the wearable-based measure. Secondly, while the study participants performed the ADLs in a naturalistic manner, it is essential to recognize that the collected data might not fully reflect their real-world motor performance. This underscores the necessity to gather data within naturalistic environments. Thirdly, the point-to-point movements used for the kinematic analysis were manually annotated by research staff using video recordings. However, future investigations require the development of an algorithm capable of automatically detecting point-to-point movements using inertial data for practical application. A promising direction for this lies in the application of deep learning-based methodologies, which have demonstrated significant potential in accurately detecting specific movement patterns, such as 3D point-to-point movements, within unstructured inertial data streams gathered during real-world ADLs. 55 Finally, the proposed method does not offer detailed information about the quality of performed movements, such as inter-joint coordination or compensatory movements, which could be clinically important. Inferring these aspects based on the kinematics of wrist movements remains an important research area.
Conclusion
This study investigated a reliable, objective, and continuous measure of upper-limb kinematics to aid in monitoring the stroke motor recovery process. To achieve this, we examined the kinematic characteristics of point-to-point upper-limb movements performed during naturalistic ADLs using a six-axis IMU on the wrist affected by stroke. The study highlights the potential of ecologically valid kinematic analysis to offer additional insights into evaluating patients’ impairment, thereby enhancing our understanding of their response to prescribed treatment regimens. The findings of this research could open new clinical and research opportunities for a more comprehensive and multi-dimensional perspective on stroke survivors’ motor progression through rehabilitation.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683241270066 – Supplemental material for Wearable-Based Kinematic Analysis of Upper-Limb Movements During Daily Activities Could Provide Insights into Stroke Survivors’ Motor Ability
Supplemental material, sj-docx-1-nnr-10.1177_15459683241270066 for Wearable-Based Kinematic Analysis of Upper-Limb Movements During Daily Activities Could Provide Insights into Stroke Survivors’ Motor Ability by Sunghoon Ivan Lee, Yunda Liu, Gloria Vergara-Díaz, Benito Lorenzo Pugliese, Randie Black-Schaffer, Mary Ellen Stoykov and Paolo Bonato in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-2-nnr-10.1177_15459683241270066 – Supplemental material for Wearable-Based Kinematic Analysis of Upper-Limb Movements During Daily Activities Could Provide Insights into Stroke Survivors’ Motor Ability
Supplemental material, sj-tif-2-nnr-10.1177_15459683241270066 for Wearable-Based Kinematic Analysis of Upper-Limb Movements During Daily Activities Could Provide Insights into Stroke Survivors’ Motor Ability by Sunghoon Ivan Lee, Yunda Liu, Gloria Vergara-Díaz, Benito Lorenzo Pugliese, Randie Black-Schaffer, Mary Ellen Stoykov and Paolo Bonato in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-3-nnr-10.1177_15459683241270066 – Supplemental material for Wearable-Based Kinematic Analysis of Upper-Limb Movements During Daily Activities Could Provide Insights into Stroke Survivors’ Motor Ability
Supplemental material, sj-tif-3-nnr-10.1177_15459683241270066 for Wearable-Based Kinematic Analysis of Upper-Limb Movements During Daily Activities Could Provide Insights into Stroke Survivors’ Motor Ability by Sunghoon Ivan Lee, Yunda Liu, Gloria Vergara-Díaz, Benito Lorenzo Pugliese, Randie Black-Schaffer, Mary Ellen Stoykov and Paolo Bonato in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-4-nnr-10.1177_15459683241270066 – Supplemental material for Wearable-Based Kinematic Analysis of Upper-Limb Movements During Daily Activities Could Provide Insights into Stroke Survivors’ Motor Ability
Supplemental material, sj-tif-4-nnr-10.1177_15459683241270066 for Wearable-Based Kinematic Analysis of Upper-Limb Movements During Daily Activities Could Provide Insights into Stroke Survivors’ Motor Ability by Sunghoon Ivan Lee, Yunda Liu, Gloria Vergara-Díaz, Benito Lorenzo Pugliese, Randie Black-Schaffer, Mary Ellen Stoykov and Paolo Bonato in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-5-nnr-10.1177_15459683241270066 – Supplemental material for Wearable-Based Kinematic Analysis of Upper-Limb Movements During Daily Activities Could Provide Insights into Stroke Survivors’ Motor Ability
Supplemental material, sj-tif-5-nnr-10.1177_15459683241270066 for Wearable-Based Kinematic Analysis of Upper-Limb Movements During Daily Activities Could Provide Insights into Stroke Survivors’ Motor Ability by Sunghoon Ivan Lee, Yunda Liu, Gloria Vergara-Díaz, Benito Lorenzo Pugliese, Randie Black-Schaffer, Mary Ellen Stoykov and Paolo Bonato in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors would like to thank the research staff in the Motion Analysis Laboratory at Spaulding Rehabilitation Hospital for their invaluable assistance in conducting data collection among stroke survivors.
Author Contributions
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the National Institute of Biomedical Imaging and Bioengineering (grant number R01EB027777) and the Eunice Kennedy Shriver National Institute Of Child Health & Human Development of the National Institutes of Health (grant number P2CHD101899).
Supplementary material for this article is available on the Neurorehabilitation & Neural Repairwebsite along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
