Abstract
Background
It has long been of interest to characterize the components of the motor abnormality in the arm after stroke. One approach has been to decompose the hemiparesis phenotype into negative signs, such as weakness, and positive signs, such as intrusion of synergies. We sought to identify the contributions of weakness and flexor synergy to motor deficits in sub-acute stroke.
Methods
Thirty-three sub-acute post-stroke participants and 16 healthy controls performed two functional arm movements; one within flexor synergy (shoulder and elbow flexion), and the other outside flexor synergy (shoulder flexion and elbow extension). We analyzed upper limb 3D kinematics to assess both overall task performance and intrusion of pathological synergies. Weakness and spasticity were also measured.
Results
Both tasks produced similar impairments compared to controls. Analysis of elbow and shoulder multi-joint coordination patterns revealed intrusion of synergies in the out-of-synergy reaching task based on the time spent within a flexion-flexion pattern and the correlation between shoulder and elbow angles. Regression analysis indicated that both weakness and synergy intrusion contributed to motor impairment in the out-of-synergy reaching task. Notably, the Fugl-Meyer Assessment (FMA) was abnormal even when only weakness caused the impairment, cautioning that it is not a pure synergy scale.
Conclusions
Weakness and synergy intrusion contribute to motor deficits in the sub-acute post-stroke period. An abnormal FMA score cannot be assumed to be due to synergy intrusion. Careful kinematic analysis of naturalistic movements is required to better characterize the contribution of negative and positive signs to upper limb impairment after stroke.
Introduction
Approximately 80% of stroke survivors experience motor deficits, typically in the form of hemiparesis.1,2 Between 50% and 60% of patients with an initial arm paresis have arm disability at 6 months 3 and 1 year. 4 We have argued that the examination of the process of true motor recovery requires a focus on the impairment level. 1 Motor impairment in the arm after stroke has multiple components: weakness, reduced motor control or dexterity, sensory loss, spasticity, and intrusion of pathological synergies.1,5,6 These components may be associated with distinct neural substrates and recovery profiles.1,7,8 Twitchell and Brunnstrom first described pathological flexor and extensor synergies as patterns of limb movement at the joint level that emerge after stroke.5,6 In the clinical setting, a widely used motor impairment measure is the Fugl-Meyer Assessment (FMA), an ordinal scale that quantifies the abilities of the participants to make both isolated and coordinated joint movements. 9 The FMA was designed to emphasize the contribution of pathological synergies, flexor and extensor, to post-stroke limb deficits. For example, in one maneuver, participants are required to flex the shoulder (0°-90°) while maintaining a straight elbow (0°). In this case, any flexion at the elbow would indicate the intrusion of a flexor synergy and lead to a lower score.
Electromyography (EMG) can also be used to measure abnormal flexor and extensor muscle co-activation patterns during 2D isometric movements of the upper limb post-stroke.10-12 However, kinematic analysis is still required to quantify the effect of synergies as Brunnstrom 6 and Twitchell 5 described them, that is, demonstrating their intrusion into functional movements. This is because the quality of performance in a reaching task is assessed based on its kinematics: Did the participant make a normal-looking prehension movement or not? Indeed, kinematically normal movements can be made in the presence of significant co-contraction and high levels of stiffness.13,14 Thus, abnormal EMG is almost certainly necessary, but not sufficient, to generate abnormal joint kinematics; these need to be shown directly. In recognition of these issues, studies have designed and analyzed reaching tasks that promote the emergence of synergies. In one approach, participants were required to perform movements while bearing different shoulder loads,15-17 and in another, the kinematics of movements performed in and out of synergy were compared. 18 For example, Zackowksi et al 18 showed that in chronic stroke, reaching kinematics in a task that required elbow extension (out of flexor synergy) were much more impaired than in the case of a within-flexor synergy task. In addition, the stroke participants were also impaired in isolating movements around the wrist, elbow, and shoulder joints; flexion spilled over to the other joints, which led the authors to conclude that abnormal reaching kinematics in chronic stroke are driven by intrusion of the flexor synergy. While joint individuation was also examined in sub-acute stroke, 19 intrusion of pathological synergies during reaching was not. Here, instead of using the indirect measure of joint individuation, we devised two measures that directly detect flexor-synergy intrusion at the level of joint kinematics during 3D functional arm movements. Based on this novel approach, we were able to examine the contribution of flexor synergy intrusion to functional arm movements. We hypothesized that intrusion of flexor synergies would be detectable in sub-acute stroke and contribute to motor impairment.
Materials and Methods
Participants
Participants with either an ischemic or a hemorrhagic stroke (confirmed by imaging) were recruited by the Negev lab (a collaborative initiative of Ben-Gurion University and Adi Negev Nahalat Eran in Israel) between 2019 and 2022. Research protocols for both stroke and healthy participants were approved by Sheba Hospital Helsinki Committee and Ben-Gurion University Human Participants Research Committee, respectively. Inclusion criteria were: (1) Ability to give informed consent, (2) Intact cognitive and motor control abilities before the incidence, and (3) Sufficient active movement of the arm. Participants were excluded if they had a history of physical or neurological conditions that interfered with either the study procedures or the assessment of motor function (e.g. arthritis, neuropathy, Parkinson’s disease). The paretic side was identified by a clinician.
We analyzed data from 33 stroke participants in the sub-acute stage
20
(1-8 weeks post-stroke, aged 65.1 ± 11.8; 25 males; 32 right hand dominant; Supplemental Table 1) and 16 healthy controls (aged 67.1 ± 5.25; 6 males; 12 right hand dominant). Comparing the age distribution of the groups yielded no significant difference (
All participants were recorded while performing the two tasks. Four recordings—two stroke participants performing the cup-to-mouth task with their right arm and two others performing the same task using their left arm were lost due to technical difficulties.
Experimental Design
Two 3D arm tasks were recorded using markerless kine matics 21 : a cup-to-mouth task that required shoulder and elbow flexion, and a reaching task that required shoulder flexion and elbow extension. In the cup-to-mouth task, participants were instructed to perform a simulated cup-to-mouth motion from a side table to their mouth, holding a plastic cup in their hand. In the reaching task, participants were instructed to perform upward and forward reaching movements toward a suspended target. In both tasks subjects flexed their shoulder, but in one they had to extend the elbow (out of synergy reaching) and in the other they had to flex their elbow (within synergy movement). Thus, using simple functional tasks, we were able to elicit two qualitatively different coordination patterns: one within the flexor synergy and the other out of the flexor synergy. The instruction was to complete ten trials of each task. To compare the two tasks, in the cup-to-mouth task, only the movement segments of reaching from table to mouth were examined.
To quantify movement, we used a novel approach for analyzing markerless 3D kinematics (DeepBehavior) 21 using a convolutional neural network algorithm (OpenPose) 22 that was trained to detect 57 key points in the human body in each video frame (Figure 1). We compared the paretic and the non-paretic arms to the non-dominant arm of age-matched controls. The non-dominant arm was used to ensure that performance deficits in the participants with stroke could only be attributed to their stroke deficit and not to differences in hand dominance in those cases where the paretic deficit happened to be in the non-dominant arm.
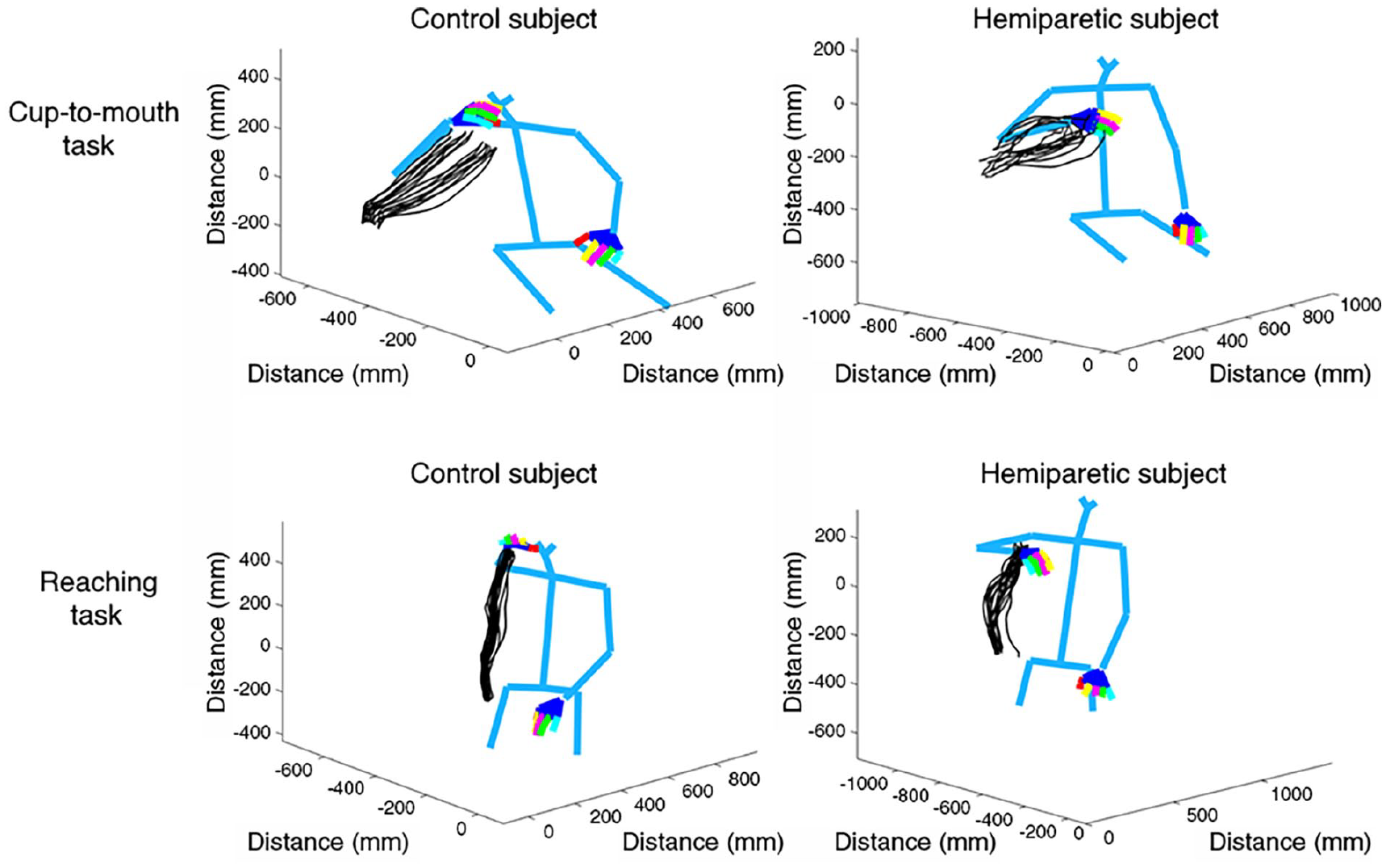
Kinematics of the arm and body during two 3D tasks. Estimation of 3D movements in two participants: blue lines connect the model’s joint positions, and the movement trajectories are marked by black lines (each participant performed ten repetitions of the task). Trajectories are shown for a healthy control participant (left column) and a stroke participant (right column) performing the cup-to-mouth task (top row) and the reaching task (bottom row).
Recording Setup
The tasks were recorded using a custom-made system comprising two high-speed cameras (150 frames per second, 1280 × 1024 pixels, Blackfly S Color 1.3 MP USB3 camera (FLIR Inc) with a Fujinon 1.5MP 6 mm C Mount lens), set on a custom-designed aluminum camera holder with a 66° angle between their axes. In this setup, cameras were positioned 120 cm in front of the participant, at a height of 95 cm, and placed at a set 45° angle toward the participant, which allowed us to produce the 3D kinematic data. In the cup-to-mouth task, participants faced the cameras at a 45° angle with the moving arm closer to the camera lenses and a side table with an empty cup placed to their side. In the reaching task, participants were recorded from the frontal angle while facing the cameras and reaching up toward a suspended object (~1.5 m above ground). Each participant attempted to perform ten iterations of each task in each hand (in separate blocks). Stroke participants that could only partially execute these tasks were included in the analyses if minimal paretic arm movement was detected at least twice in each task. All participants in our study were able to perform shoulder elevation (as indicated by a clinician). During the recordings, no markers were placed on the participants as the analysis algorithm enables markerless detection of joint positions.
Impairment and Functional Measures
The motor component of the upper limb FMA, 9 Action Research Arm Test (ARAT), 23 spasticity (Modified Ashworth Scale, MAS), 24 and weakness (grip dynamometer) scores were collected from all participants. Neither MAS nor weakness scores were collected when the arm was flaccid, or the participant couldn’t perform power grip.
Reported inter-rater and intra-rated reliabilities for the FMA are between 0.98 and 0.996, while test-retest reliability is 0.96.25,26 Reported inter-rater and intra-rated reliabilities for the ARAT are between 0.96 and 0.99, while test-retest reliability is 0.96. 27 The values obtained using the grip dynamometer were normalized into z-scores according to the age, gender, and dominant hand of each of the participants. 28
Data Analysis
The recordings resulted in two synchronized videos from two cameras. Each video was passed through the OpenPose algorithm to detect joint positions. Then, the corresponding 2D positions of joints from each video were stereo-triangulated to obtain the estimated 3D position.
29
To do this, a prior calibration using a checkerboard was obtained. This resulted in a list of 3D positions of all joints. These data were smoothened using a Savitzky-Golay filter with a window size of 57 and a polynomial degree of 3. Then, the joint tangential velocities were calculated. Movements were segmented based on the wrist velocity profiles (movement start and end were defined based on the crossing point of 10% of the peak velocity). Peak velocity detection and segmentation were automatic but verified and adjusted manually. The main performance measures calculated at the wrist joint were movement extent, peak velocity, movement duration, and smoothness. Extent was defined as the radial 3D position at the end of the reaching/cup-to-mouth task compared to the start position. Smoothness was defined as the minus log of the normalized integrated squared jerk (see equation (1); jerk is the third derivative calculated based on the position of the wrist during the movement;
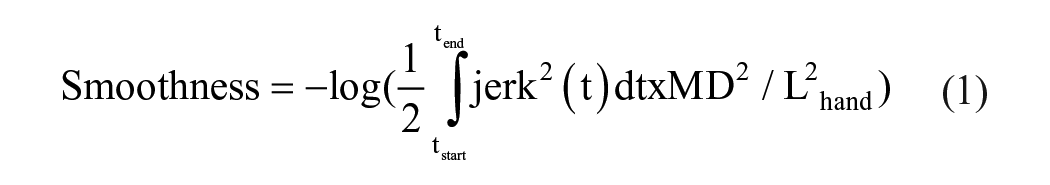
Joint angle data was calculated using an intrinsic (anatomical) coordinate system (angle calculated in relation to a specific joint; shoulder flexion angle is defined as the angle in degrees between the ipsilateral elbow joint, ipsilateral shoulder joint, and contralateral shoulder, projected onto the sagittal plane—defined by the torso and shoulder vectors created by the 3D model of the participant; elbow extension angle is defined as the angle in degrees between the ipsilateral wrist, elbow, and shoulder joints).
Flexion-flexion coordination was quantified based on the angular velocity of the elbow and shoulder joints in two ways: (1) flexion-flexion proportion was cal culated as the time spent while simultaneously flexing the elbow and shoulder, divided by the total time of the movement, and (2) flexion-flexion strength was the Pearson’s correlation coefficient of the shoulder flexion and elbow flexion angles, during the largest segment in the movement that the shoulder was flexing (determined based on their angular velocities). To deal with the skewed distribution of this measure, we performed a Fisher transformation by calculating the inverse hyperbolic tangent (arctanh) of the correlation coefficients.
Statistical Analysis
Between-group differences (control, non-paretic, and paretic stroke) were assessed using two-sample, two-tailed
Two-way mixed design analysis of variance (ANOVA) was perfor med to assess the differences in kinematic measures between groups (control and paretic stroke) and across tasks.
Furthermore, the contribution of different impairment measures (spasticity, weakness, and intrusion of synergy) to performance measures (e.g. smoothness and extent) in the data of stroke participants was assessed using linear regression analysis. Statistical and data analysis was performed via MATLAB 31 and JASP. 32
Power Analysis
Based on the final sample containing 33 stroke participants and 16 age-matched controls, the power to identify significant differences across groups, in a two-way ANOVA assuming a medium effect size of
Furthermore, our correlation analysis has a power of 94% to identify an effect of ρ = 0.5. The multiple linear regression analysis has a power of 92% to determine an effect of
Results
Performance Was Abnormal in Both 3D Arm Tasks
Participants performed a reaching task that required flexion at the shoulder and extension at the elbow (movement outside of flexor synergy) and a cup-to-mouth task that required flexion at the shoulder and elbow (movement within flexor synergy) (Figure 1 and Supplemental Figures 1–6).
Movements of stroke participants were slower and hypometric, showed increased jerkiness, and sometimes involved compensatory strategies. To go beyond observation, performance on the two tasks was quantified by measuring each participant’s peak velocity, movement duration, movement extent, and smoothness.
Performance in stroke participants was impaired compared to control participants (Figure 2). All effects were highly significant (
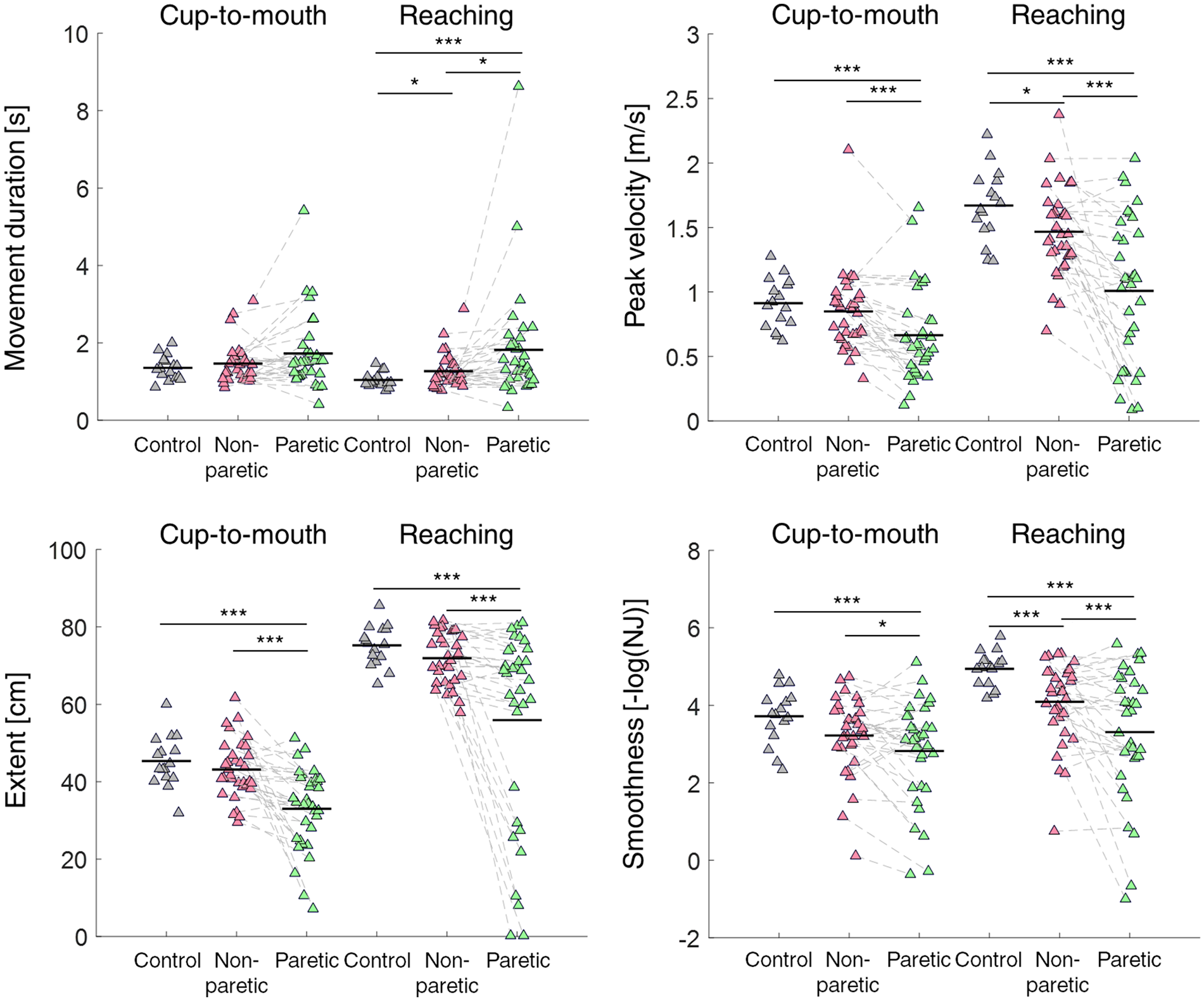
Kinematic measures of performance showed impaired performance in both the reaching and cup-to-mouth tasks after stroke. (A) Scatter plots of movement duration, (B) peak velocity, (C) extent, and (D) smoothness in each group, for both tasks. Each triangle represents the values of the measure for a single participant across all individual movements in the task. Horizontal lines represent averages. Significant differences across groups are denoted by asterisks (*
When comparing the performance of the non-paretic side with the performance of age-matched control participants, no significant differences were observed in any of the measures for the cup-to-mouth task (
An interaction analysis between damage (paretic hand and control) and task (cup-to-mouth and reaching; in and out of synergy, respectively) was significant for peak velocity (
Evidence for Intrusion of the Flexor Synergy
To further examine for intrusion of synergies into arm movements, we devised two novel approaches to measure the coordination of shoulder and elbow joint angles during performance. The first approach was to quantify the flexion-flexion pattern during arm movements in patients and healthy controls. To do this, we calculated the proportion of time that participants spent in flexion-flexion coordination during the movement by identifying the segments in which both the shoulder and elbow were flexing (“flexion-flexion proportion”; see Figure 3A). In neurotypical individuals, this proportion would reflect the amount of flexion-flexion that was required to complete the task. Any increase in flexion-flexion proportion compared with neurotypical behavior was deemed an abnormal synergy.
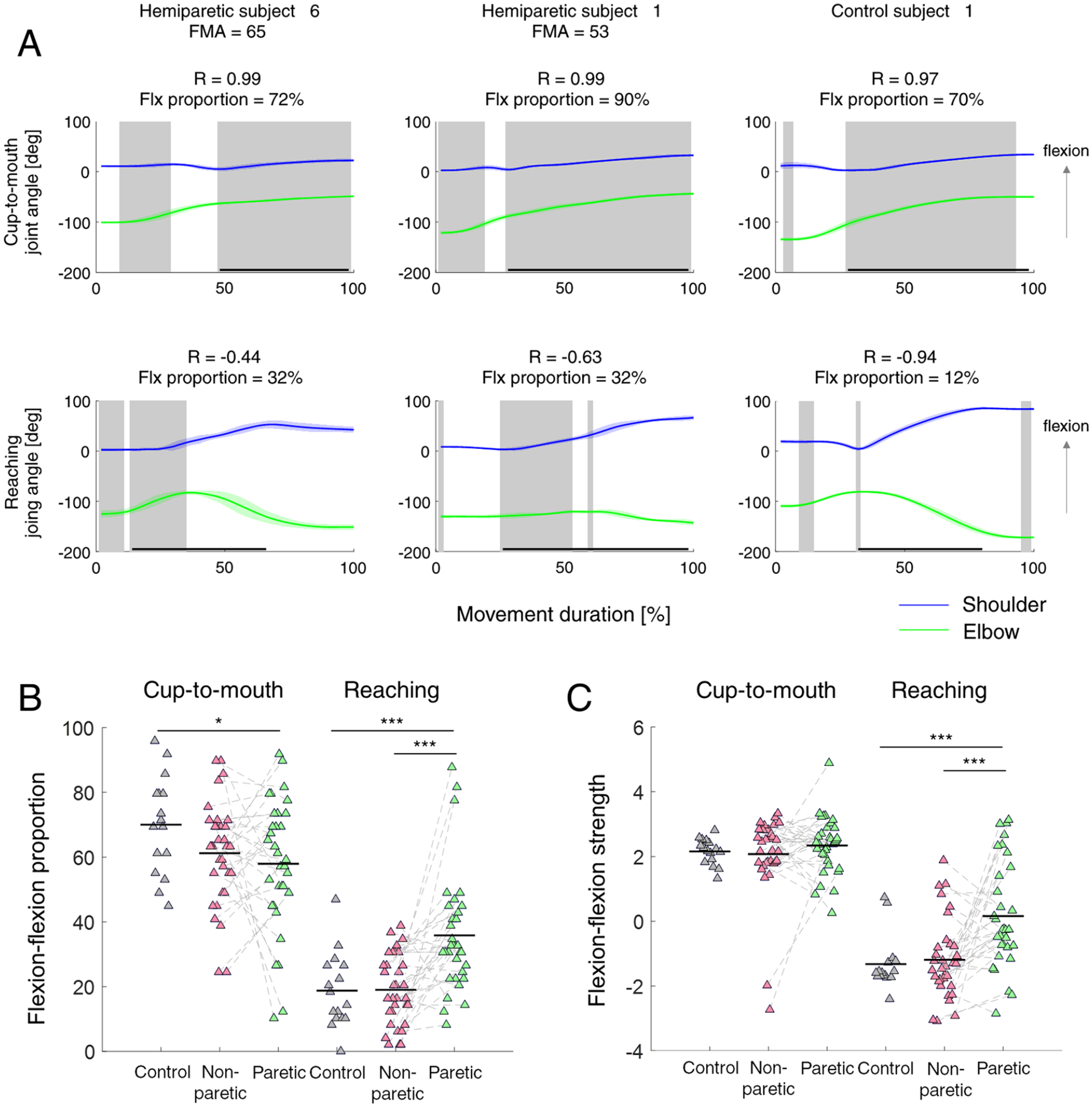
Two kinematic measures of flexor synergy intrusion. (A) Shoulder and elbow angular trajectories of two hemiparetic participants (left and middle) and a control participant (right) in both tasks: cup-to-mouth (top) and reaching task (bottom). Gray areas represent the time points in which both joints were flexing, based on which the proportion of time spent in flexor synergy was calculated (flexion-flexion proportion). Horizontal black lines represent the longest segment identified of shoulder flexion. Based on this segment, Pearson’s correlation coefficient on the angular trajectories of the shoulder and elbow was calculated (flexion-flexion strength). Angular trajectories are averaged across all individual movements in the task. (B) Scatter plot of the percentage of time spent within flexion-flexion movement pattern in each group. (C) Scatter plot of the correlation coefficient of the flexion-flexion angles in each group. Correlation coefficients were measured only in segments of shoulder flexion. Each triangle represents the values of the measure for a single participant. Significant differences across groups are denoted by asterisks (*
Indeed, in the cup-to-mouth task, the control participants spent a greater percentage of time in flexion-flexion coordination (mean ± std: 70.03 ± 15.1) than in the reaching task (mean ± std: 18.75 ± 11.6). In addition, the proportion of time control participants spent in flexion-flexion coordination was highly affected by the task (
We next examined if stroke participants show a general across-task increase in using a flexion-flexion pattern. Interestingly, this wasn’t the case - they spent significantly less proportional time than control participants in flexion-flexion coordination in the cup-to-mouth task (paretic vs controls:
Another way of assessing the level of intrusion of the flexor synergy was to assess the strength of the dependency between the degree of shoulder flexion and involuntary flexion at the elbow. To accomplish this, we analyzed the motion of the elbow when the shoulder was flexing. If abnormal synergy patterns interfered with the movement, this would mainly occur in the parts of the movement where the shoulder was actively flexing, enslaving the elbow into flexion. We, therefore, identified in each task the longest segment of shoulder flexion and calculated the Pearson’s correlation coefficient on the angular trajectories of the shoulder and elbow during that segment (Figure 3C). This measure, termed “flexion-flexion strength,” would reflect the extent of shoulder-elbow flexion dependency, and is expected to be positive in tasks requiring mostly a flexion-flexion pattern, and negative in tasks requiring a flexion-extension pattern. Here also, any deviation from neurotypical behavior, whereby the strength of the flexion-flexion pattern increased, was deemed a synergy.
In the control participants, flexion-flexion strength was positive in the cup-to-mouth task (mean ± std: 2.14 ± 0.40) and negative in the reaching task (mean ± std: −1.34 ± 0.82). As with the flexion proportion measure, significant between-group differences in flexion-flexion strength were observed in the reaching task, with stroke participants exhibiting increased flexion-flexion strength (
Notably, two control participants displayed slightly positive values in this measure in the reaching task (0.55 and 0.72). This can be explained by these participants’ idiosyncratic natural movement pattern that involved flexing their elbow before extending it to reach the target. Indeed, when calculating the Pearson Correlation coefficient of the entire movement, these participants produced negative values, indicating that overall, they extended their elbow while flexing the shoulder in the reaching task. Importantly, stroke participants showed increased flexor synergy strength in their paretic arm compared to their non-paretic arm (
To conclude, two direct kinematic measures of flexion-flexion coordination (proportion and strength) provided evidence for flexor synergy intrusion in movements requiring a flexion-extension coordination pattern.
Inter-Joint Coordination Was Abnormal Due to Both Weakness and Synergy Intrusion
So far, the results indicated impaired performance in both tasks, and intrusion of the flexor synergy in the reaching task. However, it is still unclear to what extent overall task performance could be attributed to flexor synergy intrusion.
To test this, we sought to evaluate the differential contribution of different types of impairment to task performance. We applied regression analysis with kinematic measures as the dependent variables, and the two synergy measures and grip dynamometry as independent variables (Table 1). All regression analyses were performed for the paretic arm of stroke participants.
Regression Analysis of Different Types of Impairment Measures of Stroke Participants.
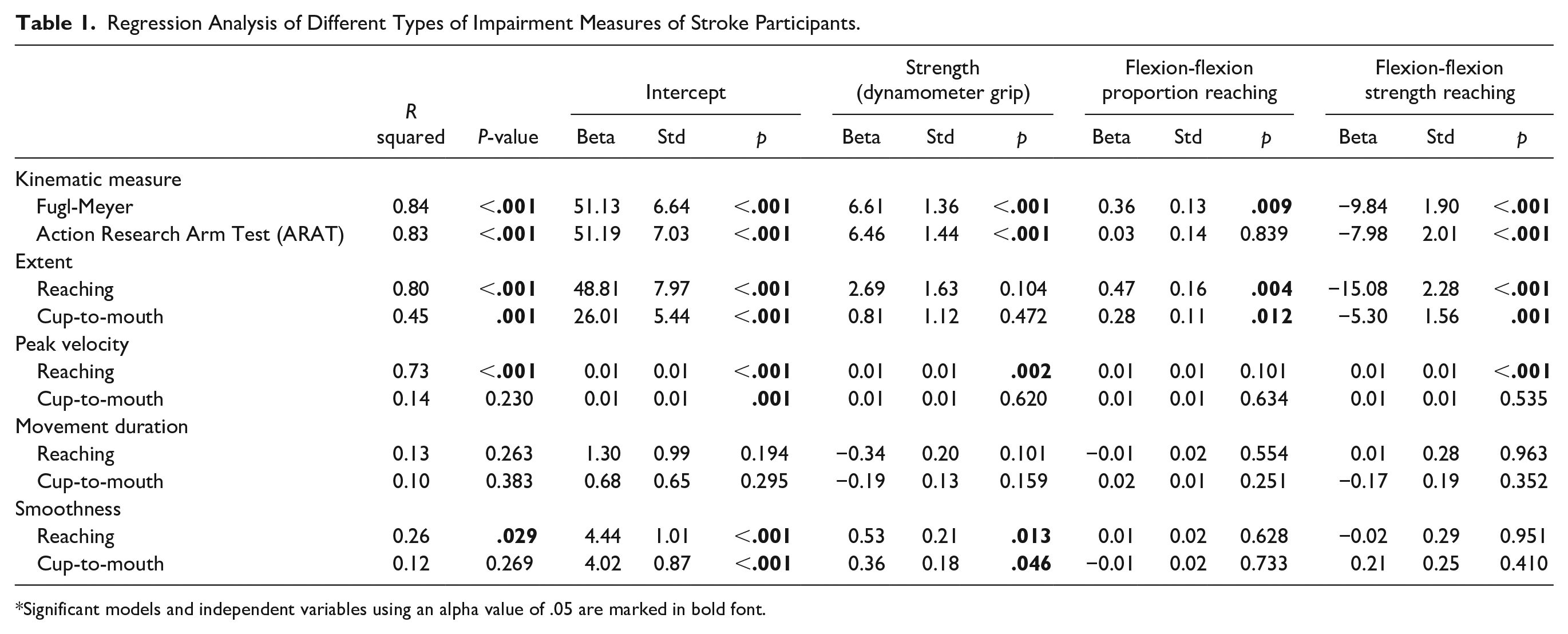
Significant models and independent variables using an alpha value of .05 are marked in bold font.
In the reaching task, weakness was a significant pre dictor of peak velocity and smoothness, whereas the flexor synergy measures (taken from the reaching task) were significant predictors for extent (strength & proportion) and peak velocity (strength; Table 1). Notably, the flexor synergy intrusion measures were also significant predictors of extent in the cup-to-mouth task. Since the impairment in the cup-to-mouth task was associated with a reduction in flexion-flexion coordination (Figure 3), we argue that, in this case, this measure reflects a joint-coordination deficit that is not related to synergy intrusion.
Next, we evaluated the differential contribution of types of impairment to clinical measures by applying the same regression analysis approach with either the FMA or ARAT as dependent variables and the two synergy measures (taken from the reaching task) and weakness as independent variables. Both ARAT and FMA scores were significantly predicted by the synergy measures and by weakness (
To test whether spasticity plays a significant role in explaining these scores, we ran an additional model with weakness, flexor synergy strength, and a spasticity measure (the MAS) (see Supplemental Table 4). Spasticity did not affect any of the dependent measures. Additionally, even though the MAS scores significantly correlated with these measures (FMA, ARAT, extent in both tasks and peak velocity in the reaching task) (Supplemental Table 5), adding spasticity as an independent variable didn’t improve the fit of the regression models.
Thus, our regression results suggest that intrusion of synergies and weakness affect task performance, and also FMA and ARAT scores (Table 1).
It is important to note that the FMA clinical scores were comparably affected by synergy intrusion and weakness. It is perhaps not surprising that the FMA was also affected by weakness. Notably, when examining the participants with severe impairment as defined by the FMA (FMA below 45), 34 we noticed that all 13 participants exhibited a significant level of weakness (lower than four standard deviations below the population mean). Most of these participants (11/13) also exhibited synergy intrusion (flexor synergy strength higher than zero; Figure 4A).
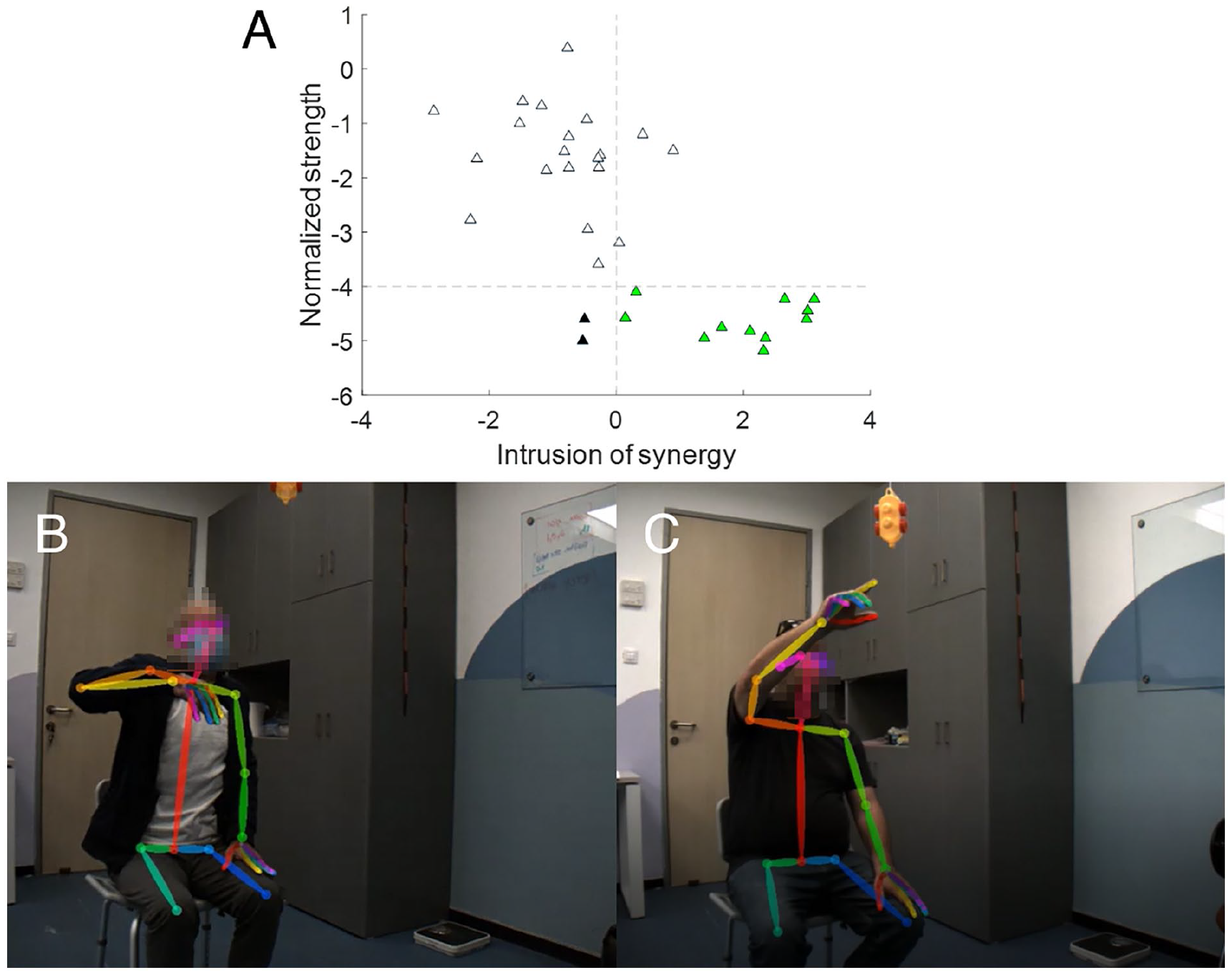
Impaired participants exhibit weakness and intrusion of synergies. (A) Scatter plot of the weakness measure compared to the flexor synergy strength measure taken from the reaching task. Each triangle represents the values of the measure for a single participant across all individual movements in the task. Triangles of participants with lower FMA scores than 45 are colored: green for participants with intrusion of flexor synergy and weakness and black for participants with weakness but without intrusion of flexor synergy. (B-C) Examples of participants performing the reaching task. Two participants are presented at the end of the upward movement in the reaching task, demonstrating their difficulty completing it. Movies depicting the 3D model of the participants performing the task are also available in the supplementary material (Supp Mov. A and B, corresponding to each of the participants presented in this figure).
Looking from another angle, 13 participants displayed positive values for flexion-flexion strength. Only two of these participants had normal strength and score on the FMA > 47 34 (Figure 4A).
Sources of Motor Impairment Through Observation
Though the contributions of flexor synergy and weakness were detected in the quantitative analysis, it is important to note that not all upper limb joint coordination deficits can be attributed to pathological synergies. To characterize the unique impairment profiles of the stroke participants, a group of researchers and clinicians examined video clips for each participant with stroke.
The videos of participants were watched by a neurologist (JWK) and two occupational therapists (RBN, SD). One of the dominant problems noted by the clinicians was weakness-induced compensatory movements around the shoulder. Specifically, weakness was evident in the difficulty participants had anteriorly flexing the shoulder and extending the elbow to its full capacity (Figure 4B and C). Compensatory abduction and hiking of the shoulder led to internal rotation of the arm, with the result that the elbow fell into flexion (Figure 4B). Weakness was also apparent in wrist drop (Figure 4B). The clinicians also noted evidence for muscle adhesion and scapula impingement. Therefore, we argue that weakness and peripheral secondary impairments made independent contributions to inter-joint coordination deficits that went beyond synergy intrusion.
Discussion
We sought to examine the underlying components of abnormal arm motor control after stroke with a novel approach for assessing the intrusion of the pathological flexor synergy to 3D arm movements using markerless pose estimation. We found that intrusion of the flexor synergy and weakness both contributed to poor kinematic performance in reaching. Abnormal clinical measure scores in the sub-acute stage were also attributable to weakness and synergy intrusion.
Here, we demonstrated the benefits of using 3D kinematics, which allows a suitably granular analysis of reaching performance in naturalistic tasks. Performance was abnormal, with significant differences in all kinematic measures (Figure 1 & Supplemental Table 2). Assessing kinematics in two tasks that differentially weight out-of-synergy movements allowed us to assess the presence of synergies, as has been done previously.18,35 Levin 35 showed that inter-joint coordination deficits during 2D planar arm movements in participants with chronic stroke were invariant to reaching direction, suggesting that intrusion of synergies is not a major contributor to 2D post-stroke reaching deficits. 35 In contrast, Zackowski et al 18 showed synergy intrusion by demonstrating greater deficits in 3D reaching requiring movement outside the flexor synergy than within the flexor synergy. They supported this conclusion by showing that participants with stroke could not make isolated movements of the wrist, elbow, or shoulder joints. 18 Our kinematic analyses revealed slightly greater deficits in the out-of-synergy task (reaching) and a significant interaction between the group and task for peak velocity (Supplemental Table 3). Thus, in the case of 3D movements, synergy intrusion differentially affects out-of-synergy movements in both chronic 18 and sub-acute stroke.
A crucial benefit of kinematic analysis is that it allows the quantification in functional tasks of both negative symptoms, such as loss of dexterity and the effect of weakness, and positive symptoms, such as intrusion of synergies.35-37 We developed two measures for flexor synergy intrusion during upper limb movements: “proportion” and “strength.” The proportion measure sought to capture the effect of synergies on the overall coordination of the movement, whereas the strength measure was designed to capture the amount of coupling between the joints (Figure 3). The fact that both measures were significant indicates that synergies affect both the strength of the coupling and its degree of intrusion during the movement. The increased inter-participant variability in the proportion measure suggests that this measure is more sensitive to an individual’s coordination style compared to the strength measure.
Given the clear indications of intrusion of flexor synergy in the flexion coordination analyses (Figure 3), we would have expected to find a more robust task-dependent difference at the kinematics level (Supplemental Table 3). This apparent discrepancy between the analyses may be driven by the fact that within-synergy movements are sensitive to other deficits, such as weakness and loss of dexterity. The reduction in flexion-flexion coordination in the cup-to-mouth task may be a marker of these additional contributors to the deficit.
Our results seemingly contradict those of Wagner et al
19
by showing a contribution of flexor synergy intrusion to reaching abnormalities in the sub-acute stage. Wagner and colleagues compared the ability to make isolated joint movements in the acute post-stroke stage (all participants were tested within 2 weeks of their stroke) to performance in a reaching task. Their results showed that although the ability to individuate joints was impaired in the early sub-acute phase, it didn’t explain more than marginal amounts of the variance of the reaching deficit (3%), while weakness explained the majority of variance (5%-30%).
19
In contrast, our study demonstrates that weakness and synergy together explain 53% to 71% of the variance of the FMA, ARAT, reaching extent, and peak velocity (Table 1), where each one of them explained at least 16% of the variance in kinematic performance measures (
Studies in the second half of the 20th century that attempted to formally characterize changes in the post-stroke arm paretic phenotype over the course of recovery noted that in addition to weakness, stroke participants also suffer from obligatory flexor and extensor synergies.5,6 The FMA 9 was designed, in part, to capture such synergy intrusion. The FMA correlates with other measures of synergies10,11,37 and is abnormal in acute, sub-acute, and chronic stroke.38-42 Our measures also correlated with the FMA (Supplemental Table 5). Surprisingly, despite the synergy-measurement rationale for the FMA, we found that it can also be scored low when only weakness is present, as demonstrated by those participants that did not display intrusion of synergies but had low FMA scores (Figure 4). Indeed, various studies have shown that the FMA correlates with weakness.7,37,43,44
These results, along with previous ones, have several implications. First, they emphasize the importance of going beyond clinical scales toward the characterization of neurological deficits with fine-grained kinematic analyses of natural movements. 45 Notably, our measures were designed to specifically quantify unique aspects of motor impairment (flexor synergy intrusion), but the same experimental design and kinematic approach could also be used to differentiate compensatory strategies from true recovery after stroke. Second, the results suggest that the cause of an abnormal FMA can be qualitatively different from one patient with stroke to another: in one it might primarily reflect weakness, and in another, the intrusion of synergies. Thus, response to an intervention when measured by the FMA may mean very different things, for example, when administered in sub-acute versus chronic stroke.46-48 Third, the fact that weakness plays such an important role in the motor control deficits in the acute and sub-acute stages post-stroke may be the reason that the reticulospinal tract (RST), which is critical to the generation of large forces, 49 gets upregulated in the chronic phase.50-52 Essentially, a weakness problem, especially when severe, gets replaced by a synergy problem. Novel interventions, likely best instigated in the acute and subacute stages, are going to be needed to mitigate this zero-sum game trade-off.
Limitations
One potential limitation of this study is that our cup-to-mouth task didn’t require as much shoulder flexion as the reaching task (see Supplemental Appendix). This could be why we couldn’t find consistent differences in how stroke participants performed both tasks. We think that this is unlikely due to the qualitative difference between the tasks (the reaching task requires primarily moving outside the flexor synergy: shoulder flexion and elbow extension, and the cup-to-mouth task does not). Furthermore, this limitation does not affect the flexor synergy strength measure since that was calculated in a continuous segment in which the shoulder was flexing. Additionally, it could be that elbow flexion in the reaching task was affected by crossing the stretch reflex threshold, 53 a manifestation of spasticity. While the level of spasticity cannot explain the variability in the clinical results beyond the contribution of synergies and weakness, a more refined measure of spasticity (such as the tonic stretch reflex threshold, TSRT) is needed to address this possibility in future studies.
We did not perform EMG, albeit purposefully. As stated in the introduction, while abnormal EMG co-activation may be considered a necessary condition for the emergence of synergies, isolating pathological synergies in non-isometric conditions, that is, during voluntary movements, using EMG is challenging and has not been attempted to our knowledge. In addition, abnormal EMG does not inevitably result in abnormal kinematics. 14 Our interest was more about whether the joint flexor synergy had a deleterious effect on reaching and less about its origin. Indeed, we would be hard-pressed to devise an alternative explanation for an unwanted flexor synergy that does not involve muscle co-activation.
Another possible limitation is the fact that this system has not been validated on stroke participants. We have, however, previously performed a validation study of this system compared to a gold-standard marker-based system in healthy adults. 54 Since the kinematic modeling approach is performed on single frames, and people after stroke in the sub-acute phase typically do not suffer from anatomical deformations, we do not think that validation in this population is essential.
Supplemental Material
sj-tif-1-nnr-10.1177_15459683241268535 – Supplemental material for The Kinematics of 3D Arm Movements in Sub-Acute Stroke: Impaired Inter-Joint Coordination is Attributable to Both Weakness and Flexor Synergy Intrusion
Supplemental material, sj-tif-1-nnr-10.1177_15459683241268535 for The Kinematics of 3D Arm Movements in Sub-Acute Stroke: Impaired Inter-Joint Coordination is Attributable to Both Weakness and Flexor Synergy Intrusion by Inbar Avni, Ahmet Arac, Reut Binyamin-Netser, Shilo Kramer, John W. Krakauer and Lior Shmuelof in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-2-nnr-10.1177_15459683241268535 – Supplemental material for The Kinematics of 3D Arm Movements in Sub-Acute Stroke: Impaired Inter-Joint Coordination is Attributable to Both Weakness and Flexor Synergy Intrusion
Supplemental material, sj-tif-2-nnr-10.1177_15459683241268535 for The Kinematics of 3D Arm Movements in Sub-Acute Stroke: Impaired Inter-Joint Coordination is Attributable to Both Weakness and Flexor Synergy Intrusion by Inbar Avni, Ahmet Arac, Reut Binyamin-Netser, Shilo Kramer, John W. Krakauer and Lior Shmuelof in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-3-nnr-10.1177_15459683241268535 – Supplemental material for The Kinematics of 3D Arm Movements in Sub-Acute Stroke: Impaired Inter-Joint Coordination is Attributable to Both Weakness and Flexor Synergy Intrusion
Supplemental material, sj-tif-3-nnr-10.1177_15459683241268535 for The Kinematics of 3D Arm Movements in Sub-Acute Stroke: Impaired Inter-Joint Coordination is Attributable to Both Weakness and Flexor Synergy Intrusion by Inbar Avni, Ahmet Arac, Reut Binyamin-Netser, Shilo Kramer, John W. Krakauer and Lior Shmuelof in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-4-nnr-10.1177_15459683241268535 – Supplemental material for The Kinematics of 3D Arm Movements in Sub-Acute Stroke: Impaired Inter-Joint Coordination is Attributable to Both Weakness and Flexor Synergy Intrusion
Supplemental material, sj-tif-4-nnr-10.1177_15459683241268535 for The Kinematics of 3D Arm Movements in Sub-Acute Stroke: Impaired Inter-Joint Coordination is Attributable to Both Weakness and Flexor Synergy Intrusion by Inbar Avni, Ahmet Arac, Reut Binyamin-Netser, Shilo Kramer, John W. Krakauer and Lior Shmuelof in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-5-nnr-10.1177_15459683241268535 – Supplemental material for The Kinematics of 3D Arm Movements in Sub-Acute Stroke: Impaired Inter-Joint Coordination is Attributable to Both Weakness and Flexor Synergy Intrusion
Supplemental material, sj-tif-5-nnr-10.1177_15459683241268535 for The Kinematics of 3D Arm Movements in Sub-Acute Stroke: Impaired Inter-Joint Coordination is Attributable to Both Weakness and Flexor Synergy Intrusion by Inbar Avni, Ahmet Arac, Reut Binyamin-Netser, Shilo Kramer, John W. Krakauer and Lior Shmuelof in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-6-nnr-10.1177_15459683241268535 – Supplemental material for The Kinematics of 3D Arm Movements in Sub-Acute Stroke: Impaired Inter-Joint Coordination is Attributable to Both Weakness and Flexor Synergy Intrusion
Supplemental material, sj-tif-6-nnr-10.1177_15459683241268535 for The Kinematics of 3D Arm Movements in Sub-Acute Stroke: Impaired Inter-Joint Coordination is Attributable to Both Weakness and Flexor Synergy Intrusion by Inbar Avni, Ahmet Arac, Reut Binyamin-Netser, Shilo Kramer, John W. Krakauer and Lior Shmuelof in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We thank Sandra Deluzio for the insightful clinical interpretation of the data.
We thank the recovering participants and families who agreed to participate in this study and acknowledge the medical and research staff members of Adi Negev Nahalat Eran who assisted with the entire operation.
Author Contributions
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by The United states-Israel Binational Science Foundation grants 2021248 (LS and AA) and 2015327 (LS and JWK).
Supplementary material for this article is available on the
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
