Abstract
Background
A growing number of studies has combined transcranial direct current stimulation (tDCS) with other non-invasive non-pharmacological therapies (NINPT) to enhance effects in pain reduction. However, the efficacy of these combined approaches in treating chronic primary pain (CPP) warrants thorough investigation.
Objective
This study aims to evaluate the efficacy of tDCS in conjunction with other NINPT in alleviating pain severity among CPP patients.
Methods
We conducted a systematic search for randomized controlled trials (RCTs) comparing the efficacy of tDCS combined with NINPT against control treatments in adult CPP patients. The search spanned multiple databases, including PubMed, EMBASE, LILACS, Scopus, Web of Science, and CENTRAL.
Results
Our systematic review included 11 RCTs with a total of 449 participants. In our meta-analysis, which comprised 228 participants receiving active-tDCS and 221 receiving sham-tDCS, we found a significant reduction in pain intensity (Standard Mean Difference = −0.73; 95% Confidence Interval (CI) = −1.18 to −0.27; P = .002) with the use of active-tDCS combined with NINPT.
Conclusion
These findings substantiate the therapeutic potential of combining tDCS with other NINPT, highlighting it as an effective treatment modality for reducing pain intensity in CPP patients.
Introduction
Chronic primary pain (CPP) is characterized by pain in 1 or more anatomical regions that persists or recurs for more than 3 months, as classified by the International Association for the Study of Pain and detailed in the eleventh edition of the International Classification of Diseases (ICD-11). This pain is notably accompanied by significant emotional distress or functional impairment and cannot be better attributed to another chronic pain condition.1,2
Nociplastic pain, frequently observed in various forms of CPP, plays a significant role in numerous chronic pain syndromes.3,4 This pain category arises from alterations in pain processing, predominantly due to central sensitization. Central sensitization amplifies neural signaling and modulates pain processing, leading to an altered perception of pain. 5 This shift in pain modulation can be attributed to either an increase in excitability or a decrease in inhibition within specific neural pathways. 6 Moreover, these changes involve epigenetic processes across various tissues, which adapt to these alterations in the nervous system. 7
In CPP management, neuromodulation strategies has garnered attention, 8 especially given the limited efficacy of pharmacological treatments in addressing nociplastic pain. 3 Among these strategies, transcranial direct current stimulation (tDCS) stands out as a non-invasive neuromodulatory technique that delivers direct currents through electrodes placed on the scalp. tDCS is a viable option due to its portability, accessibility, and promising results in pain relief.9-12 As a novel application, tDCS is used as a priming tool before the administration of another non-invasive non-pharmacological therapies (NINPT). This priming effect seems to enhance the therapeutic outcomes of subsequent NINPT. 13 Along those lines, recent studies that have used tDCS in addition to other NINPT have shown improved individual benefits of NINPT, with encouraging results in pain reduction.13,14-16 NINPT encompasses various interventions such as physical modalities, psychological interventions, complementary and alternative therapies, and other innovative techniques. In chronic pain conditions, NINPT modulates pain through peripheral, spinal, and supraspinal mechanisms. These mechanisms include the modulation of nociceptive signaling, facilitation of neuroplastic changes in pain pathways through processes like long-term potentiation or long-term depression, and the limitation of central sensitization underlying chronic pain. 17
The current literature reveals a gap in systematic reviews with meta-analyses evaluating the clinical effects of combining tDCS with other types of NINPT in chronic pain conditions (CPP). To address this, we conducted a systematic review of randomized controlled trials investigating the combined use of tDCS and various NINPT modalities for alleviating pain in patients with CPP. We hypothesize that the synergistic effects of active tDCS and NINPT will enhance pain management effectiveness compared to sham tDCS.
Methods
This study was carried out in accordance with the guidelines of the Cochrane Handbook for Systematic Reviews of Interventions and the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement.18,19 Furthermore, the study protocol was registered in the PROSPERO database under the protocol number CRD42023416391.
Eligibility Criteria
Types of Study
For this review, we included randomized clinical trials, published in English, that compared the efficacy of the tDCS combined with another NINPT. The control group consists of either the tDCS sham or the NINPT placebo. To ensure the rigor of our analysis, we excluded case studies and uncontrolled trials from our review.
Participants
This review focused on adult individuals diagnosed with any form of CPP (MG30.0) as defined in the ICD-11. Inclusion was specifically limited to the following subcategories:
Chronic widespread pain (MG30.01), including fibromyalgia syndrome;
Chronic primary musculoskeletal pain (MG30.02), encompassing conditions such as primary chronic low back pain, chronic primary neck pain, chronic primary chest pain, and primary chronic pain in the limbs;
Chronic primary headache or orofacial pain (MG30.03), which covers chronic primary orofacial pain and chronic pain from primary temporomandibular disorder;
Chronic migraine (8A80.2).
Exclusion criteria were set to omit trials involving participants with neuropathic pain, as well as studies focusing on children, adolescents, and animals.
Types of Interventions
Clinical trials offering any type of NINPT within the scope of behavioral and psychological therapy, as well as physical activity, physiotherapy, and acupuncture combined with tDCS, were eligible for inclusion. Only trials with interventions administered by qualified professionals at least once a week were included. Trials involving invasive interventions were excluded.
Types of Comparisons
Clinical trials comprising an active control group (either an active single intervention or standard treatment) and/or an inactive control group (no intervention, waitlist, or placebo-type intervention) were accepted.
Types of Outcome Measures
The primary outcome of our study was pain intensity. This was quantitatively assessed using continuous scales, including the Visual Analog Scale (VAS), which consists of a 100-mm line ranging from “no pain” to “the worst possible pain,” and the Numeric Rating Scale (NRS), an 11-point numeric scale where 0 represents “no pain” and 10 represents “the worst pain imaginable.” 20 Additionally, percentage improvements (eg, 50% pain reduction) and qualitative changes in pain state (eg, “mild pain”) were also considered as measures of pain severity. 21
As for the secondary outcome, it focused on the quality of life, based on 2 instruments: (1) Fibromyalgia Impact Questionnaire (FIQ), which includes scores ranging from 0 to 100, with higher scores indicating negative impact on life quality, 22 (2) Short-Form Health Survey Questionnaire (SF-36), which comprises 36 items across 9 subcategories, including functional capacity, physical role limitations, bodily pain, general health perception, vitality, social functioning, emotional role limitations, and mental health. In the SF-36, higher scores are indicative of a better quality of life.23,24
Search Methods for Identification of Studies
Information Sources
Two independent researchers (DDAS and RELBA) conducted a systematic search in relevant electronic databases for medical and related health literature: PubMed, EMBASE, LILACS, Scopus, Web of Science, and the Cochrane Central Register of Controlled Trials (CENTRAL). The literature search cutoff date was June 1, 2023.
Search Strategy
Medical Subject Heading terms and natural language terms were combined in the search strategy. The search terms included population, type of intervention, type of study, and outcomes. The following search terms were combined using the Population, Intervention, Control, and Outcomes strategy:
Population
chronic pain OR pain
Intervention
acupuncture OR cognitive therapy OR biofeedback OR mindfulness OR physical therapy OR manual therapy OR exercise AND neuromodulation OR tDCS OR transcranial direct current stimulation
Comparison
Sham-tDCS
Outcome
pain intensity OR quality of life OR adverse effects of tDCS OR patient-reported symptoms
The search was limited using filters for “humans” and “clinical trials.” The search strategy used in PubMed is presented in Table 1.
PubMed Search Strategy.

Study Selection Process and Data Extraction
First, 2 independent researchers (DDAS and RELBA) conducted a comprehensive search in the databases. Titles and abstracts were selected based on the predefined eligibility criteria. In the subsequent phase, full texts were evaluated, reapplying the eligibility criteria.
The inter-reviewer agreement on study eligibility showed strong concordance (Cohen’s kappa coefficient, k = 0.83; P < .001). 25 In cases of disagreement between the 2 primary reviewers, a third reviewer (JDSN) was consulted to reach a consensus.
Following the study selection, we used a standardized data extraction form for the chosen articles. This form captured crucial details such as author(s), publication year, study location, overall sample size, study population characteristics (including age range, diagnosis, and sex), outcome measures, definitions of intervention groups, and descriptions of the non-invasive intervention protocols. Additionally, we detailed the tDCS protocol, including time of application, stimulation site, parameters, number of sessions, and results (primary outcomes for pain intensity and secondary outcomes for quality of life, expressed as a function of the mean and standard deviation). Studies with incomplete descriptive outcome data were excluded from the meta-analysis.
Risks of Bias
Risk of bias assessment was independently conducted by 2 authors (RELBA and JDSN). Any disagreements were resolved through discussions among the reviewers. The assessment of bias was conducted using the Physiotherapy Evidence Database (PEDro) scale, which assigns methodological quality scores as follows: less than 4 (poor), 4 to 5 (fair), 6 to 8 (good), and 9 to 10 (excellent). 26
Summary Measures and Evaluation of the Quality of Evidence
The quality of evidence from studies included in the meta-analysis was analyzed, considering the risk of bias, results inconsistency, imprecision, and publication bias, with the final classification presented in 4 levels (high, moderate, low, and very low). 27 We followed the recommendations described in the “Grading of Recommendations, Assessment, Development, and Evaluation” (GRADE) available in the GRADEpro GDT software (https://gdt.gradepro.org).
Data Analysis
The meta-analysis was conducted using Review Manager software (v.5.4). We used the mean values, standard deviations, and sample sizes obtained for each group in the included studies. Measurements in centimeters were converted to millimeters. For the studies that reported results graphically, we used http://plotdigitizer.com to extract mean and standard deviation (SD) values. The SD for each intervention group was calculated from their confidence intervals using the formula: SD = √N × (upper limit − lower limit)/3.92. When standard error (SE) was reported, it was converted to SD using: SD = SE
To assess homogeneity, we employed the heterogeneity test. A P-value greater than .05 and an I2 index up to 25% were considered indicative of homogeneity. In cases where heterogeneity exceeded 25%, we applied a random-effects model. Conversely, if the I2 index was 25% or less, a fixed-effects model was used. We also conducted pre-specified subgroup analyses based on the timing of tDCS application (priming or additive).
For the meta-analysis, we compared the means from the VAS, NRS, and Verbal Numeric Scale (VNS) of 2 groups: those receiving active-tDCS with another NINPT and those receiving sham-tDCS with another NINPT, immediately post-intervention. A random-effects model and the Standardized Mean Difference (SMD) were used to assess effects on pain severity, with a 95% confidence interval (CI), following the guidelines in Chapter 10 of the Cochrane Handbook. 18 For interpreting SMD, we referred to Cohen’s d values, considering an SMD of 0.2 to 0.3 as a “small” effect, around 0.5 as a “medium” effect, and 0.8 as a “large” effect. 28
Results
Identification and Selection of Studies
Figure 1 illustrates the flowchart of the study selection process for our meta-analysis. Initially, our search strategy yielded 1,343 articles. Upon removing duplicates, the number decreased to 1,099 articles. A detailed review of titles and abstracts led to the selection of 23 manuscripts for full-text evaluation. Out of these, 11 studies met our criteria and were included in the quantitative analysis. These studies, published between 2011 and 2022, involved a total of 449 subjects, with 228 receiving active-tDCS combined with other non-invasive interventions and 221 receiving sham-tDCS with another non-invasive intervention. We observed that most of the included studies applied the anode to the primary motor cortex (M1), with only 1 study choosing the dorsolateral prefrontal cortex (DLPFC) region. No crossover between these studies was identified. Detailed sample characteristics and baseline measures are outlined in Table 2. The protocol characteristics of the included studies are presents in Table 3 and summarized in Figure 2.
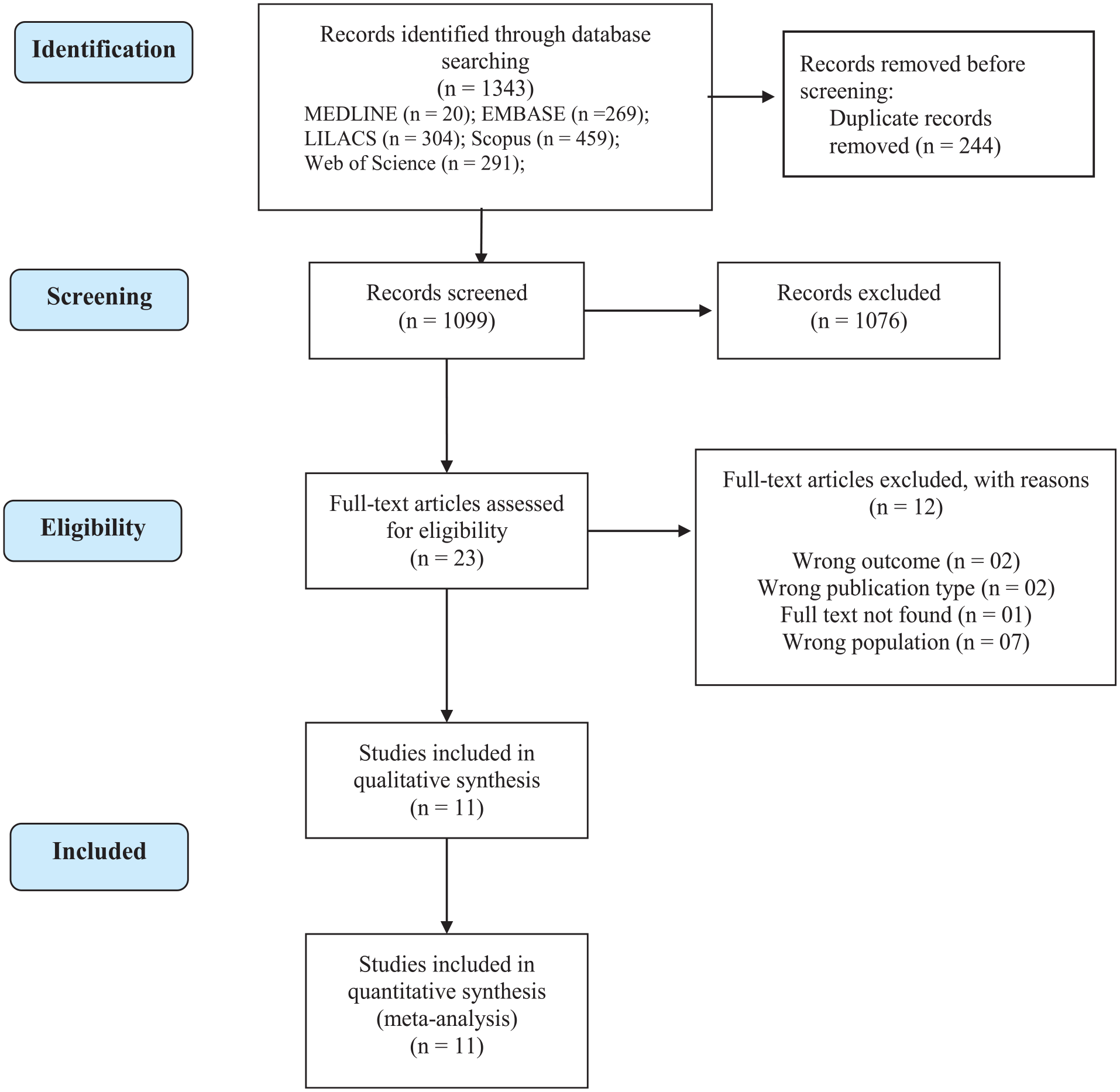
Flowchart of literature search and selection process.
Characteristics of the Patients at the Baseline.
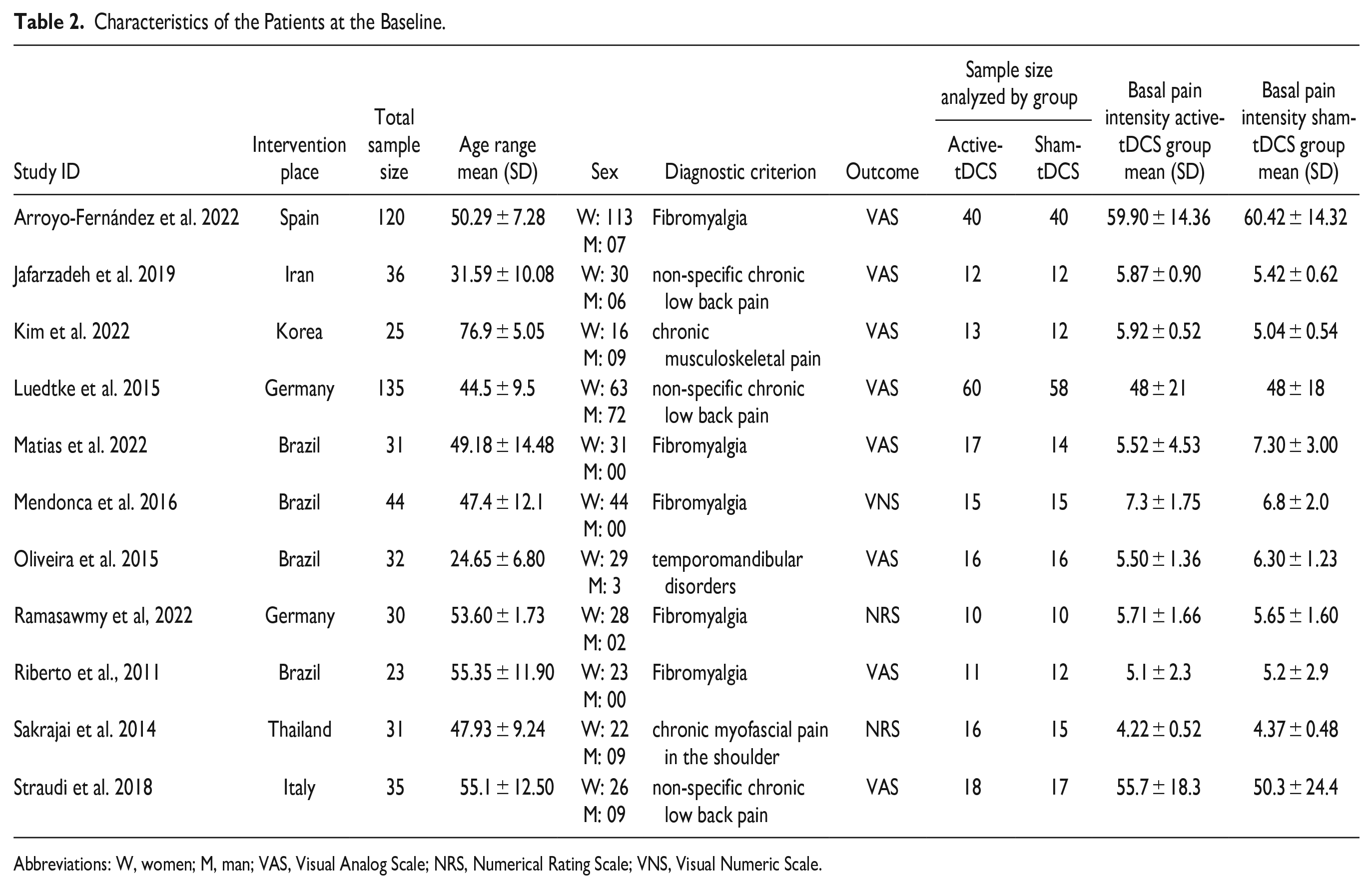
Abbreviations: W, women; M, man; VAS, Visual Analog Scale; NRS, Numerical Rating Scale; VNS, Visual Numeric Scale.
Characteristics of the Studies.
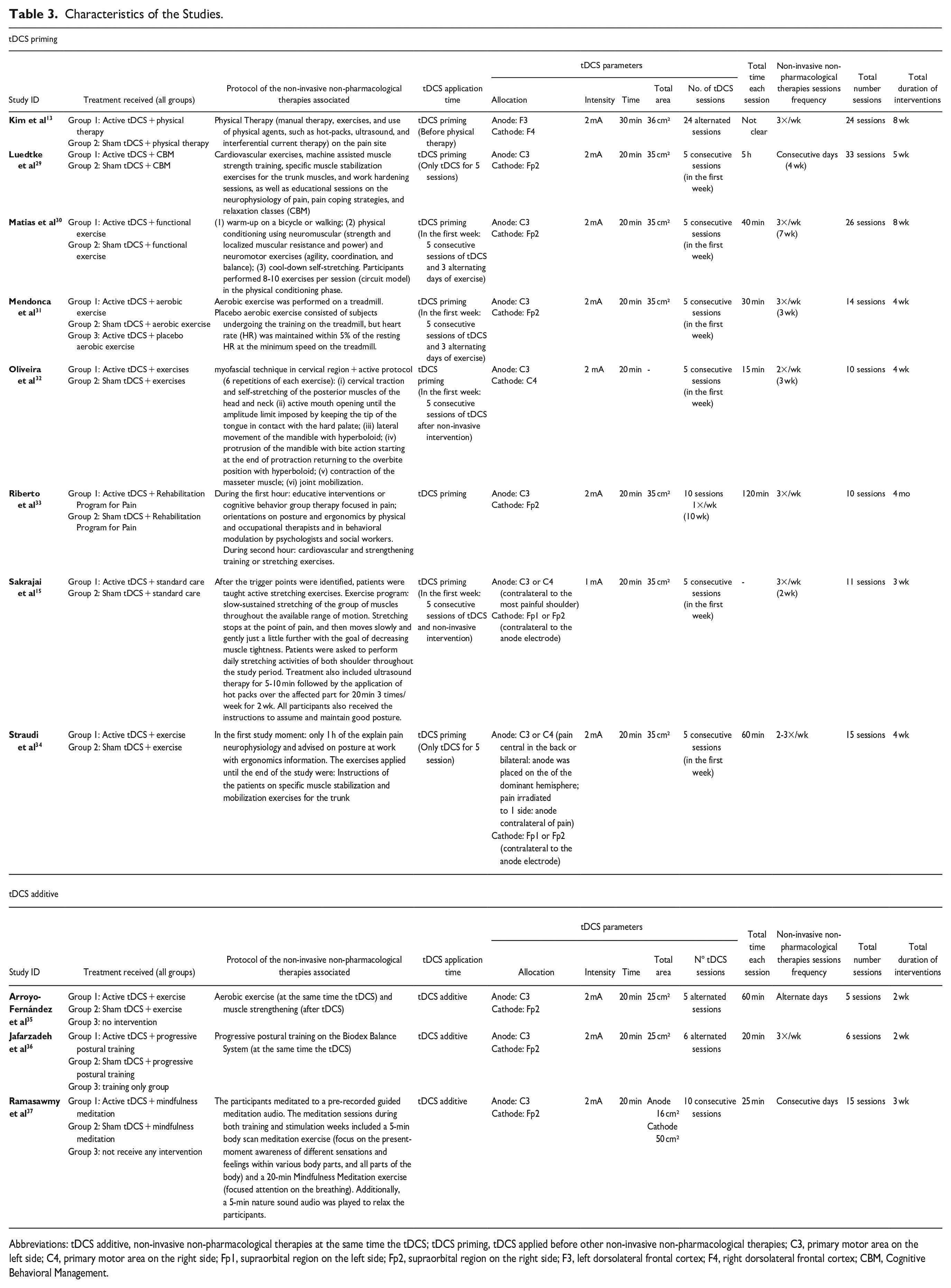
Abbreviations: tDCS additive, non-invasive non-pharmacological therapies at the same time the tDCS; tDCS priming, tDCS applied before other non-invasive non-pharmacological therapies; C3, primary motor area on the left side; C4, primary motor area on the right side; Fp1, supraorbital region on the left side; Fp2, supraorbital region on the right side; F3, left dorsolateral frontal cortex; F4, right dorsolateral frontal cortex; CBM, Cognitive Behavioral Management.
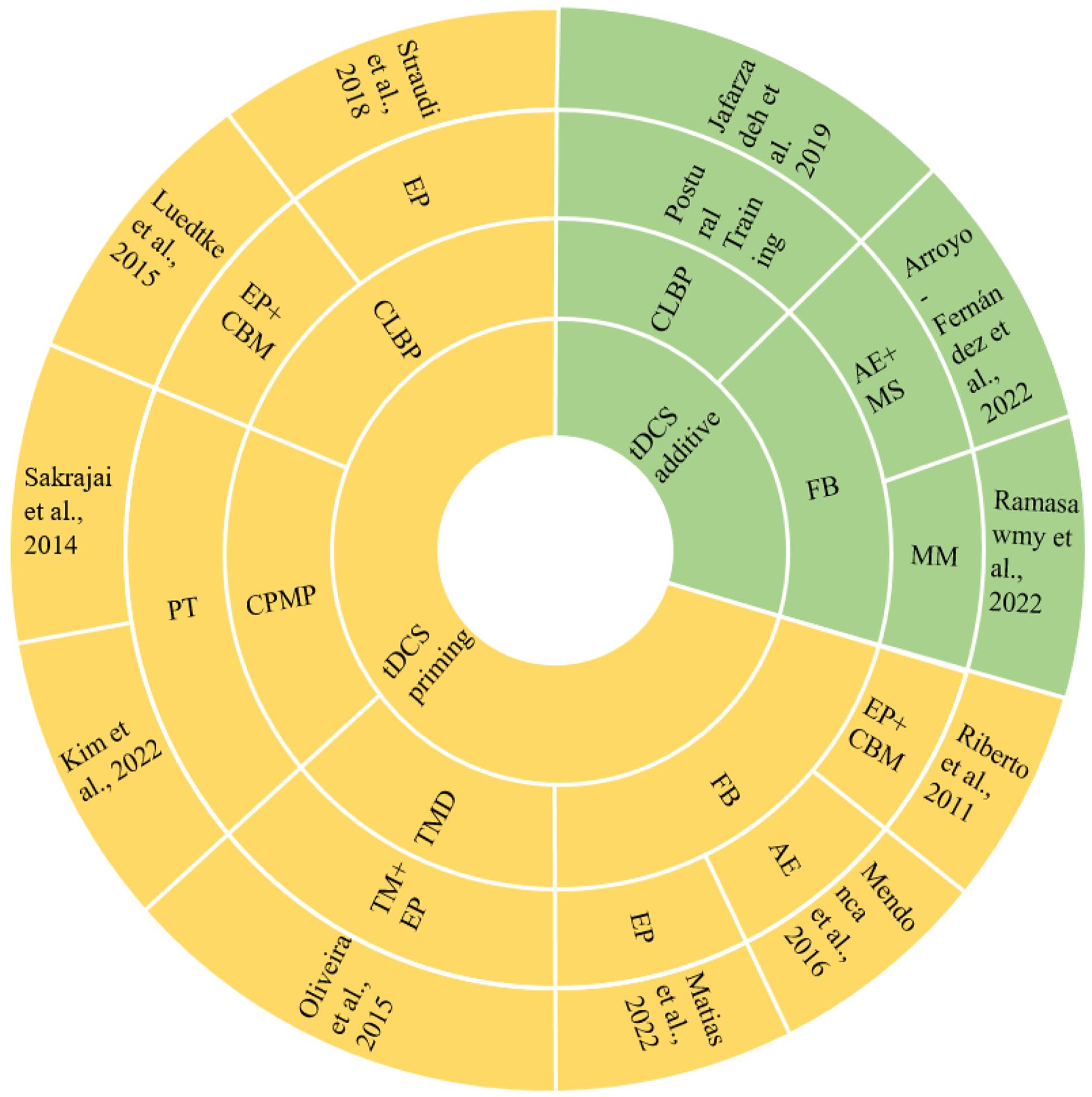
Hierarchical representation of the characteristics of the studies. The order of significance from inside to outside is the timing of tDCS application (additive or priming), followed by the sample’s diagnostic subcategory. The outermost layer is the study itself.
Risk of Bias
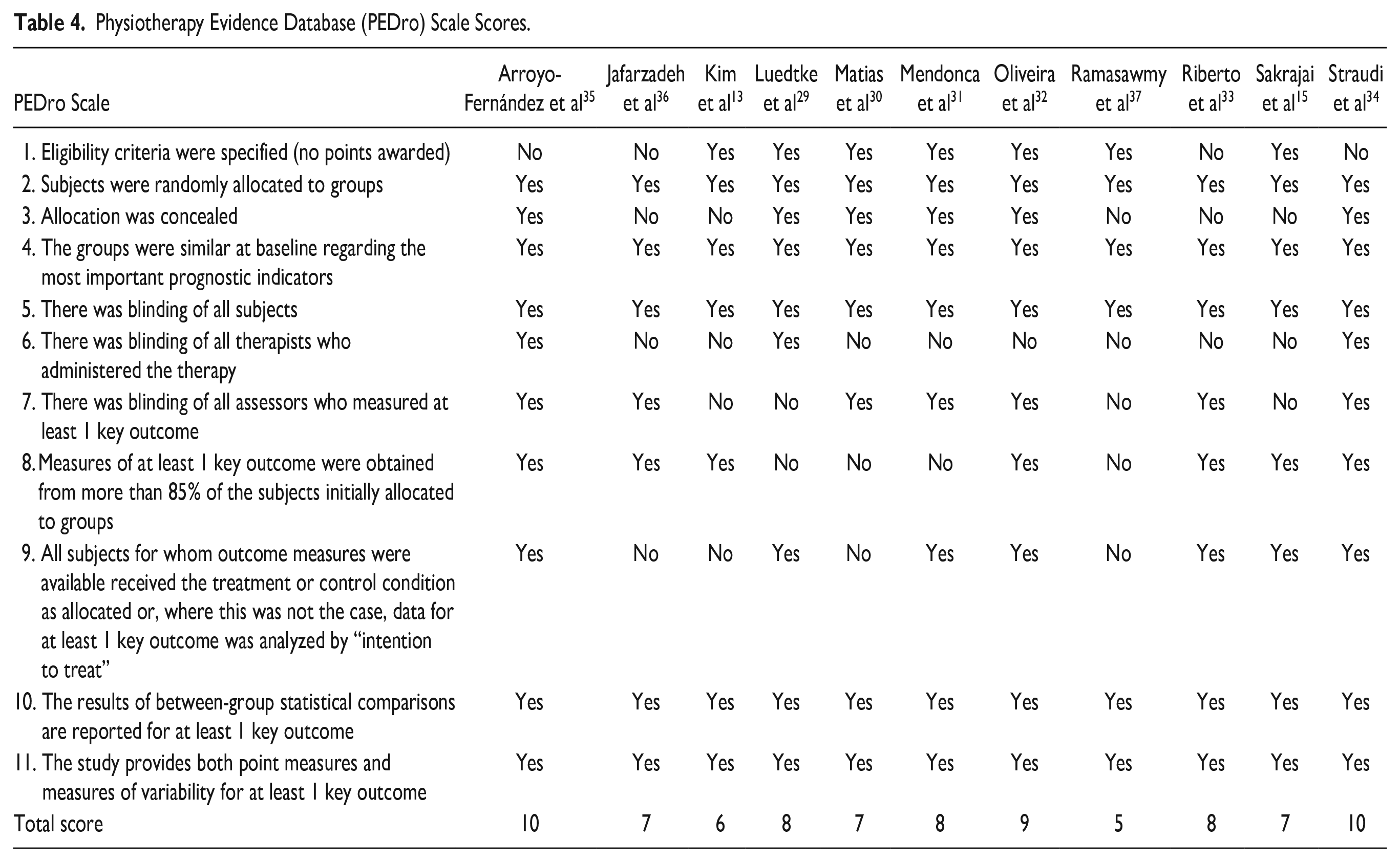
Table 4 details the risk of bias assessment for each included study. Within this assessment, only 1 study was identified as having reasonable methodological quality. 36 The rest of the studies were classified under higher quality categories, being categorized either as “good” or “excellent” in terms of their methodological rigor. This classification indicates a generally high standard of research quality across the majority of the studies included in the analysis.
Physiotherapy Evidence Database (PEDro) Scale Scores.
Pain Intensity
The analysis focused on the impact of combining tDCS with other types of NINPT on pain intensity. Results indicated a significant reduction in pain intensity, demonstrating a large effect size, when both interventions were actively used, irrespective of the tDCS application timing. The statistical outcomes were: Standard Mean Difference (SMD): −0.73; 95% Confidence Interval (CI): −1.18 to −0.27; Z-value = 3.12; P = .002; Tau² = .45; Chi² = 48.99 with a P = .001, and I² = 80% (as illustrated in Figure 3).
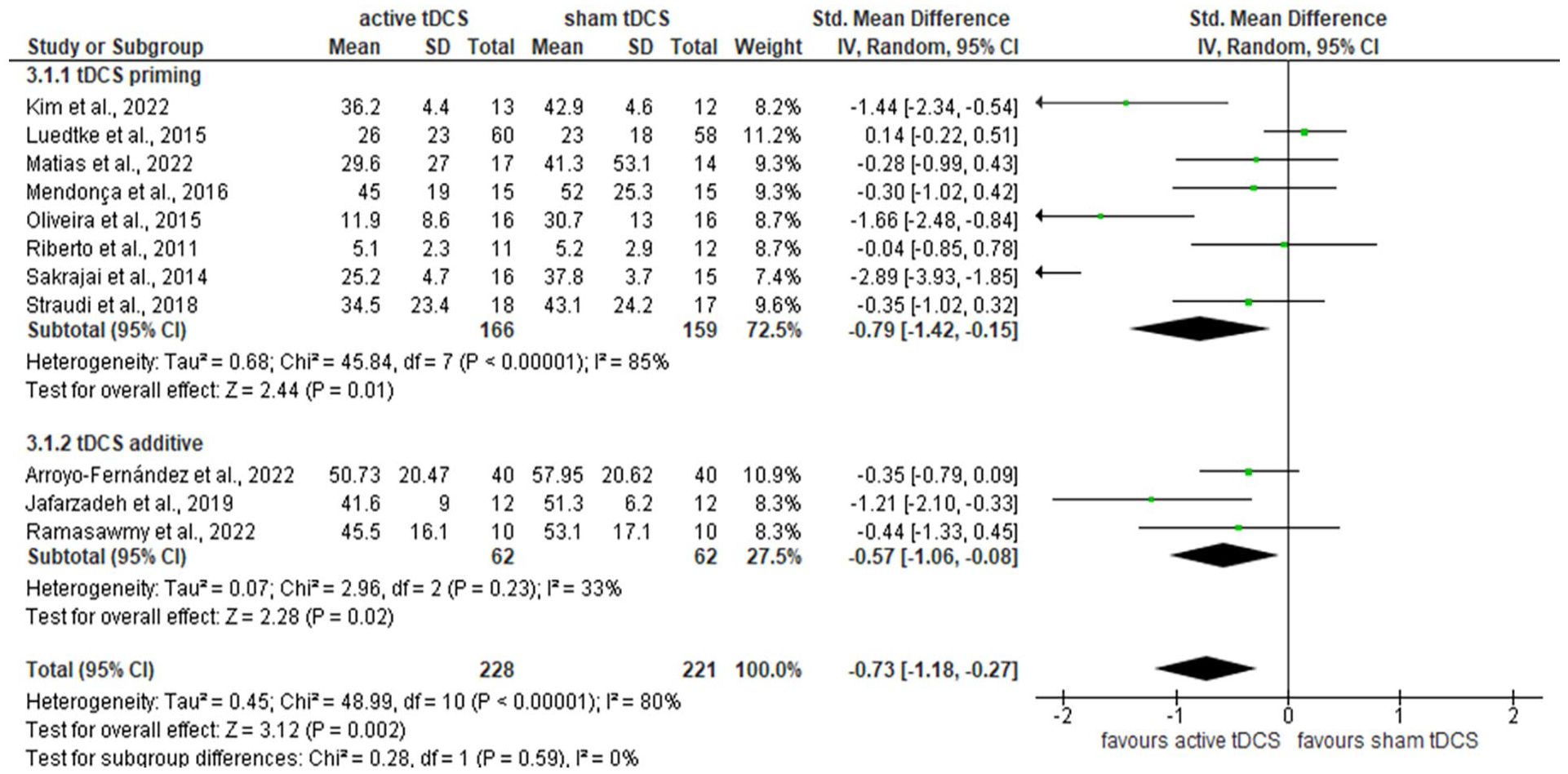
Forest plot of comparison: active tDCS associated non-invasive non-pharmacological therapies versus sham tDCS associated non-invasive non-pharmacological therapies for intensity of pain.
Subgroup analyses based on the timing of tDCS application yielded further insights. When tDCS was applied prior to another NINPT (referred to as “tDCS priming”), there was a notable reduction in pain intensity, also with a large effect size. The relevant statistics were: SMD: −0.79; 95% CI: −1.42 to −0.15; Z = 2.44; P = .01; Tau² = .68; Chi² = 45.84 with a P = .001, and I² = 85%.
Additionally, when tDCS was applied simultaneously with another NINPT (termed “tDCS additive”), there was a significant reduction in pain intensity, but with a moderate effect size. The statistical results for this scenario were: SMD: −0.57; 95% CI: −1.06 to −0.08; Z = 2.28; P = .02; Tau² = .07; Chi² = 2.96 with a P = .23, and I² = 33% (refer to Figure 3 for details).
Quality of Life
Eight studies were reviewed to compare quality of life across various groups. Among these, 3 studies utilized the SF-36. However, the inconsistency in data presentation hindered a meta-analysis. Specifically, 2 studies provided the mean and standard deviation for the overall SF-36 score. Only the study by Mendonca et al 31 detailed results for the recommended subcategories, 31 as per guidelines. 38 In contrast, 2 other studies employed distinct questionnaires. Additionally, a meta-analysis was conducted on 3 studies that used the FIQ. This analysis revealed no significant impact of combining tDCS with other non-invasive interventions on quality of life. The statistical results were as follows: Standard Mean Difference (SMD): −1.17; 95% Confidence Interval (CI): −8.06 to 5.72; Z-value = 0.33; P = .74; Tau² = .001; Chi² = .82 with a P = .66, and I² = 0% (refer to Figure 4 for more details).
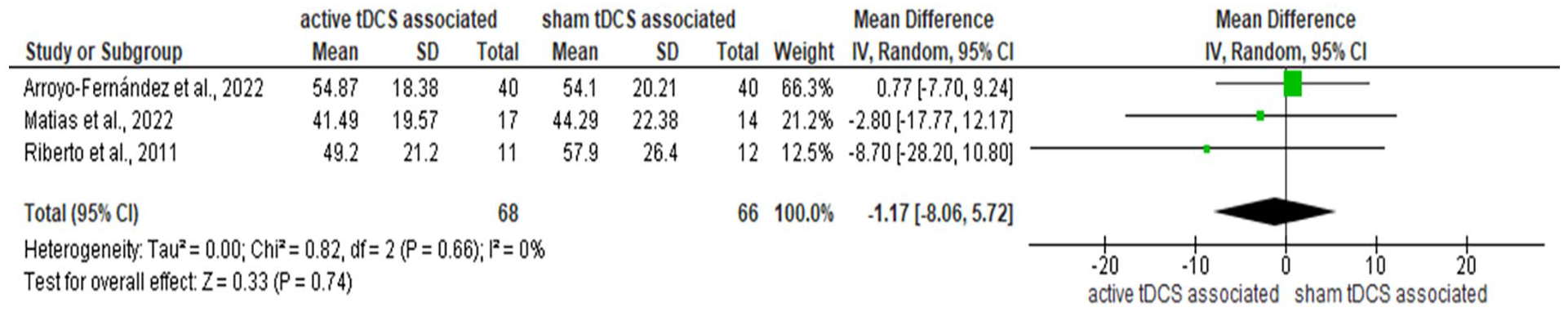
Forest plot of comparison: active tDCS associated non-invasive non-pharmacological therapies versus sham tDCS associated non-invasive non-pharmacological therapies for quality of life.
Patient-Reported Symptoms and Adverse Effects of tDCS
Out of the 11 studies reviewed, only 615,30,31,34,36,37 provided detailed accounts of the adverse effects observed in participants. The study by Jafarzadeh et al 36 found itching as a frequent side effect. They found that 20 minutes of active-tDCS applied over the primary motor cortex (M1) was generally well-tolerated, with only minimal adverse effects reported. 36 Matias et al 30 identified headache and tingling as the most common adverse effects in their study. In the research conducted by Mendonca et al, 31 participants experienced skin redness, drowsiness, and mild headaches, with these symptoms present in both active and sham groups. 31
The study by Ramasawmy et al 37 revealed that mild headache was a prevalent adverse effect, affecting 80% of participants in the active-tDCS group and 70% in the sham-tDCS group. This was followed by fatigue, reported by 70% of the active group and 50% of the sham group. 37 In Sakrajai et al, 15 a transient erythematous rash without itching or pain under the anode was observed in 2 patients from the active group. This adverse reaction resolved on its own within an hour, and no other adverse events were noted in the intervention groups. 15 Finally, the study by Straudi et al 34 reported that skin redness and tingling were the most commonly observed adverse effects in both treatment groups. 35
GRADE Analysis
Table 5 illustrates the evidence found in this review, providing the quality of this evidence considering 8 aspects suggested by GRADE. The quality of evidence was considered moderate for the effect of tDCS associated with another type of NINPT on pain intensity in CPP.
Active tDCS Associated With Non-Invasive Interventions Compared to Sham tDCS Associated Non-Invasive Interventions for CPP.
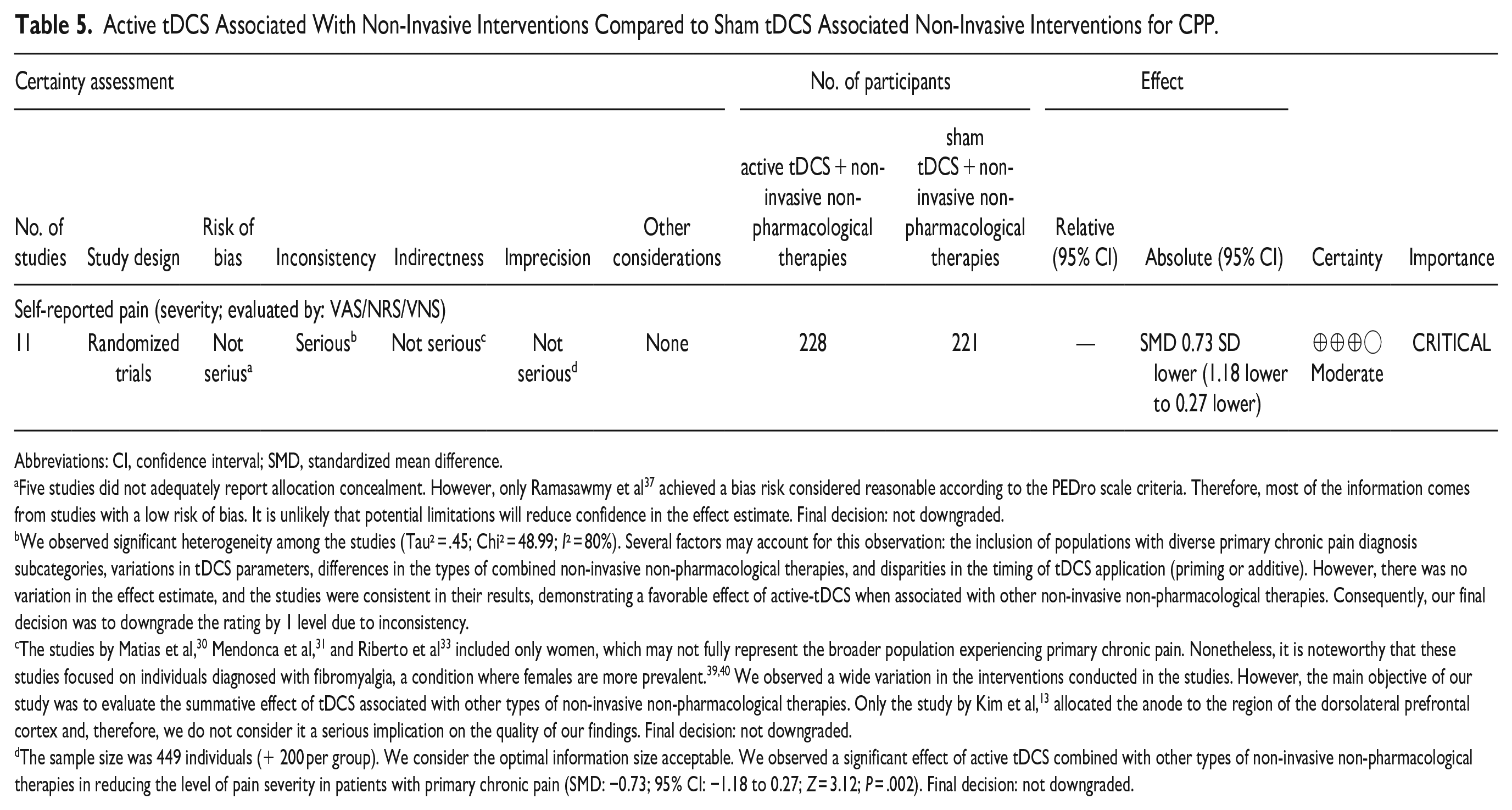
Abbreviations: CI, confidence interval; SMD, standardized mean difference.
Five studies did not adequately report allocation concealment. However, only Ramasawmy et al 37 achieved a bias risk considered reasonable according to the PEDro scale criteria. Therefore, most of the information comes from studies with a low risk of bias. It is unlikely that potential limitations will reduce confidence in the effect estimate. Final decision: not downgraded.
We observed significant heterogeneity among the studies (Tau² = .45; Chi² = 48.99; I² = 80%). Several factors may account for this observation: the inclusion of populations with diverse primary chronic pain diagnosis subcategories, variations in tDCS parameters, differences in the types of combined non-invasive non-pharmacological therapies, and disparities in the timing of tDCS application (priming or additive). However, there was no variation in the effect estimate, and the studies were consistent in their results, demonstrating a favorable effect of active-tDCS when associated with other non-invasive non-pharmacological therapies. Consequently, our final decision was to downgrade the rating by 1 level due to inconsistency.
The studies by Matias et al, 30 Mendonca et al, 31 and Riberto et al 33 included only women, which may not fully represent the broader population experiencing primary chronic pain. Nonetheless, it is noteworthy that these studies focused on individuals diagnosed with fibromyalgia, a condition where females are more prevalent.39,40 We observed a wide variation in the interventions conducted in the studies. However, the main objective of our study was to evaluate the summative effect of tDCS associated with other types of non-invasive non-pharmacological therapies. Only the study by Kim et al, 13 allocated the anode to the region of the dorsolateral prefrontal cortex and, therefore, we do not consider it a serious implication on the quality of our findings. Final decision: not downgraded.
The sample size was 449 individuals (+ 200 per group). We consider the optimal information size acceptable. We observed a significant effect of active tDCS combined with other types of non-invasive non-pharmacological therapies in reducing the level of pain severity in patients with primary chronic pain (SMD: −0.73; 95% CI: −1.18 to 0.27; Z = 3.12; P = .002). Final decision: not downgraded.
Discussion
This study conducted a systematic review and meta-analysis to assess the effectiveness of the tDCS combined with another NINPT in reducing pain severity in patients with CPP. We hypothesized that patients with CPP receiving active-tDCS in conjunction with another NINPT would experience a more significant reduction in pain intensity than those receiving sham-tDCS with an NINPT. In total, 11 studies were carefully analyzed, encompassing a combined sample of 449 subjects.
Our meta-analysis revealed a notable reduction in pain intensity when active-tDCS was paired with other NINPT, whether applied as a priming or additive technique, in patients with various forms of CPP. This finding aligns with those of a prior systematic review focused on chronic pain patients, which demonstrated enhanced pain reduction in groups receiving combined tDCS and NINPT treatments compared to control or solo intervention groups. 41 However, those earlier findings were not subjected to meta-analysis. Additionally, other existing systematic reviews have not fully explored the range of NINPT used in conjunction with tDCS, nor have they comprehensively addressed all subcategories of CPP.9,10 They typically evaluated different types of diagnoses separately and included secondary chronic pain subcategories in some cases. Our study, therefore, provides a more inclusive examination of tDCS and NINPT protocols employed in the treatment of various CPP types.
Timing of tDCS Application in Chronic Pain
The present review indicates that active-tDCS, irrespective of its timing (priming or additive), significantly reduces pain intensity. Non-invasive and non-pharmacological interventions, targeting various mechanisms, can normalize pain processing and act on central sensitization, effectively reducing pain intensity in patients with nociplastic pain.3,34 tDCS, particularly, modulates spontaneous neuronal activity leading to long-term neuroplastic changes.41,41 The therapeutic outcomes were significantly enhanced when interventions focused on the ascending (bottom–up) pathway were integrated with those influencing the descending (top–down) pathway.43,44
In our subgroup analysis, active-tDCS used as priming in combination with another NINPT significantly reduced pain intensity in various types of CPP. This could be attributed to tDCS priming protocols modulating cortical excitability, neural network connectivity, and synaptic input cooperation, influencing subsequent tDCS effects.42,45 These changes suggest metaplastic interactions, where the brain’s state at 1 moment can influence future outcomes, with synaptic plasticity depending on timing, presynaptic synchronization, and postsynaptic activity levels.43,46 This provides a unique opportunity for tDCS application at optimal times for synaptic activity modification. 46 The included studies showing superior pain reduction effects when tDCS priming is combined with physiotherapy for chronic musculoskeletal pain, 13 aerobic exercises in fibromyalgia, 31 manual therapy and exercises in temporomandibular disorders, 32 and various treatments in chronic myofascial shoulder pain. 15
However, some studies, like Matias et al 30 and Riberto et al, 33 observed no significant difference in pain reduction between groups with different tDCS applications, potentially influenced by factors like the tDCS repetition rate. Daily tDCS applications over a short period have shown cumulative effects on neuronal excitability. 47
The study also explored tDCS in conjunction with NINPT for non-specific chronic low back pain (CLBP). While standalone tDCS showed limited effects in CLBP patients,48,49 combinations with other bottom-up NINPT, like Transcutaneous Electrical Nerve Stimulation, yielded positive responses.50,51 However, results varied depending on the types of NINPT used, as seen in studies by Luedtke et al 29 and Straudi et al 34 suggesting the need for further research to identify optimal NINPT combinations with tDCS in CLBP treatment.
Regarding additive tDCS, used simultaneously with other interventions, a moderate effect was noted. This finding is supported by studies demonstrating pain intensity reduction in chronic pain patients when tDCS is combined with physiotherapy, 52 exercises, 44 manual therapy,53,54 cognitive-behavioral therapy, 55 mindfulness, 37 and biofeedback. 56
Our review highlights a gap in research regarding tDCS combined with various NINPT across different CPP diagnoses. Despite this, existing studies show good methodological quality and indicate that the association of active-tDCS with other NINPT has moderate evidence supporting its efficacy.
Locus of Stimulation for Chronic Pain
Among the studies reviewed, only the study by Kim et al 13 employed tDCS targeting the dorsolateral prefrontal cortex (DLPFC). This limited representation makes it challenging to compare the efficacy of different tDCS protocols. The primary motor cortex (M1) and the prefrontal cortex, including the DLPFC, are frequently chosen targets in brain stimulation for chronic pain management. 57 There is emerging evidence suggesting that stimulation of the DLPFC region may effectively reduce pain severity in CPP.58-60 Contrastingly, tDCS targeting the M1 region is more commonly used in CPP treatments, 61 with studies indicating its short, medium, and long-term effects on pain intensity. 62
A recent study highlighted the superior effectiveness of tDCS when applied to the M1 region over the DLPFC in patients with fibromyalgia. 63 However, another robust study showed no superiority of tDCS effects when applied to the M1, DLPFC, or operculo-insular cortex compared to the placebo group. 64 Determining the optimal site for stimulation in chronic pain treatment remains an open question. 61 This gap underscores the need for more research to directly compare the effects of stimulating between brain regions in individuals with chronic pain. Additionally, it is crucial to explore other types of NINPT that enhance the effects of tDCS.
Evidence of Intervention Associations in Quality of Life in CPP
In evaluating quality of life outcomes, we encountered notable inconsistencies in the presentation of SF-36 results, and only 3 studies30,33,35 could be statistically analyzed. The limited sample size in our analysis may have affected our ability to detect a significant impact of tDCS, combined with other NINPT, on the quality of life in CPP patients. For instance, a clinical trial involving 130 fibromyalgia patients reported a nonspecific effect, possibly attributable to a placebo response. In this trial, active tDCS did not demonstrate superiority over sham stimulation in enhancing quality of life. 65 However, it’s worth noting that tDCS, when applied to either the M1 or DLPFC regions, has shown to improve short-term quality of life. Specifically, stimulation of the M1 region was found to have medium-term effects on quality of life in fibromyalgia patients. 62
Adverse Effects of tDCS
Our findings regarding adverse effects are consistent with previous studies.66,67 We observed no severe adverse events linked to tDCS. The most common side effects were sensations of tingling, burning, and itching, with no significant difference in frequency between the active-tDCS and sham-tDCS groups. 67
Limitations and Perspectives
This systematic review and meta-analysis should be considered in light of certain limitations. Firstly, the subcategory of chronic primary visceral pain was not included due to diagnostic uncertainty based on the involved pathophysiological mechanisms. 68 Thus, we selected only CPP subcategories with well-established diagnostic criteria, ensuring more reliable results due to the similarity in the pathophysiological mechanisms of the chosen categories. Secondly, there was significant heterogeneity among the included studies, necessitating cautious interpretation of our results. This heterogeneity was anticipated, given the diverse types of NINPT assessed, in line with our objective to evaluate the impact of tDCS in conjunction with conventional CPP interventions. Another factor that may explain this heterogeneity could be the variability among tDCS protocols or the initial state of brain activation. Thirdly, our analysis was confined to the immediate effects post-intervention, omitting long-term outcomes. Future research should explore these long-term effects and also include an evaluation of other important outcomes, such as anxiety and depression. Finally, not including other types of non-invasive transcranial stimulation, such as transcranial magnetic stimulation, was another limitation of our study. We suggest that future studies include other types of non-invasive brain stimulation. Further, we recommend that subsequent studies strive to identify the most effective type of NINPT to be paired with tDCS for different CPP diagnoses.
Conclusion
Our study concludes that the combination of active-tDCS with other non-invasive interventions, regardless of application timing, is effective in reducing pain intensity in patients diagnosed with primary chronic pain, primarily when targeting the primary motor cortex. However, the impact of this combined approach on quality of life remains inconclusive.
Footnotes
Author Contributions
Renata Emanuela Lyra de Brito Aranha: Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Software; Validation; Writing—original draft; and Writing—review & editing. José Diego Sales do Nascimento: Conceptualization; Supervision; Validation; Visualization; and Writing—review & editing. Danielle Dorand Amorim Sampaio: Data curation; Investigation; and Resources. Nelson Torro-Alves: Supervision; Validation; and Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by funding from Coordination for the Improvement of Higher Education Personnel—CAPES; Brazilian National Council for Scientific and Technological Development—CNPq (313414/2021-1, 404988/2021-0), and Grant 008/2019, Pronex, Paraíba State Research Foundation—FAPESQ.
