Abstract
Background
Repetitive transcranial magnetic stimulation (rTMS) is a nonpharmacological and noninvasive brain stimulation technique that has been proven to be effective in Parkinson’s disease (PD). The combination of rTMS and treadmill training improved gait function in PD greater than treadmill training alone.
Objective
The aim of our study was to evaluate the combination of a novel high-intensity, short intervention rTMS treatment and a multimodal treatment protocol including of physiotherapy, occupational therapy and language therapy, the so-called Parkinson’s Disease Multimodal Complex Treatment (PD-MCT), to improve motor function.
Methods
In this randomized double-blind sham-controlled trial rTMS with 48 Hz or sham was applied over the cerebellum 3 times a day for 5 consecutive days. Patients were assessed at baseline (V0), after 5 days of treatment (V1), and 4 weeks later (V2). The primary clinical outcome measure was the motor sum-score of the Unified PD Rating Scale (UPDRSIII), secondary clinical outcomes were quantitative motor tasks.
Results
A total of 36 PD patients were randomly allocated either to rTMS (n = 20) or sham (n = 16), both combined with PD-MCT. rTMS improved the UDPRSIII score comparing baseline and V1 in the treatment group by −8.2 points (
Introduction
Physiotherapy and pharmacological therapy are the 2 main pillars of PD treatment, the latter being limited by the occurrence of side effects. There is a need for expansion of disease management to new effective therapeutic strategies with less adverse events. The idea of combining different treatment modalities to improve PD symptoms has triggered the development of multidisciplinary team approaches with physiotherapy, occupational therapy, and speech and language therapy. The so-called Parkinson’s Disease Multimodal Complex Treatment (PD-MCT) improves motor function and quality of life.1,2 PD-MCT was established in Germany in 2008. 3
Repetitive transcranial magnetic stimulation (rTMS) is a noninvasive brain stimulation technique that has been proven to be effective in many neurological disorders including motor symptoms in PD. 4 . Interestingly, the combination of rTMS and treadmill training improves gait function in PD greater than treadmill training alone.5,6 To date the combination of PD-MCT and rTMS was not tested, probably due to the fact that most rTMS protocols require long treatment periods of approximately 3 weeks or longer, 7 which exceeds the average duration of 10 days for PD-MCT.
Meta-regression analyses demonstrated that a greater number of pulses (per session or across sessions) yields larger rTMS effects, whereas the number of session and their intensity were not predictive for efficacy.8,9 In their 2020 study, Cole et al 10 investigated the effect of rTMS in patients with major depression with an exceptionally high number of 10 sessions per day, while 1 session per day is usual. Unfortunately, this study was designed without a sham-control. This so-called accelerated protocol was well tolerated with only minor adverse events. Therefore, we designed a novel high-dose protocol with 3 sessions per day within 5 days of treatment
The involvement of the cerebellum and the cerebellar-thalamo-cortical (CTC) pathway in the pathophysiology of PD has been overlooked for many years. 11 Recent studies demonstrated that cerebellar activity and connectivity is increased in PD patients compared to controls.11-13 This hyperactivation may represent an adaptive mechanism that compensates and bypasses the disease-related impairment of basal ganglia function.11,12,14-16 Nowadays cerebellar functional alterations in basal ganglia diseases are a “hot topic” in this field 17 and several studies used rTMS cerebellar stimulation as a treatment option for movement disorders.18-20
The aim of our study was to develop and test a novel high-intensity, short intervention rTMS protocol that fits to the schedule of the PD-MCT. The cerebellum was chosen as target for rTMS stimulation. We hypothesize that increasing the suggested compensatory mechanism of the cerebellum via rTMS improves motor function in PD patients and that the combination of rTMS and PD-MCT has additional benefits in comparison to PD-MCT alone.
Methods
Patients
A total of 44 patients with the diagnosis of idiopathic Parkinson’s disease according to the UK Brain Bank Criteria 21 were enrolled consecutively from the Department of Neurology, University Clinic Bonn, Bonn, Germany. The inclusion criteria were age 40 years or older and stable antiparkinsonian therapy for at least 1 month. Exclusion criteria were a history of seizures, frequent headaches, head injury or any neurosurgical intervention, dementia, treatment with neuroleptics, the presence of metallic particles in the head, and implanted cardiac pacemaker or neurostimulators. Changes of antiparkinsonian medication were not allowed during the trial.
Patients were interviewed for a history of falls during the past year and classified as faller or non-faller. 22 A fall was defined as “Unintentionally coming to rest on the ground, floor, or other lower level.” 23 Subjects were classified as fallers if they reported that they had fallen more than once during the previous 12 months. 24
This study was performed in accordance with The Code of Ethics of the World Medical Association (Declaration of Helsinki) and approved by the Ethics committee of the University hospital of Bonn, Germany (No. 211/18, date 10/22/2019). All enrolled subjects gave written informed consent for participation. This trial was registered at the German Clinical Trial Registry, https://drks.de (DRKS-ID: DRKS00022358), the study design flowchart is shown in Figure 1.
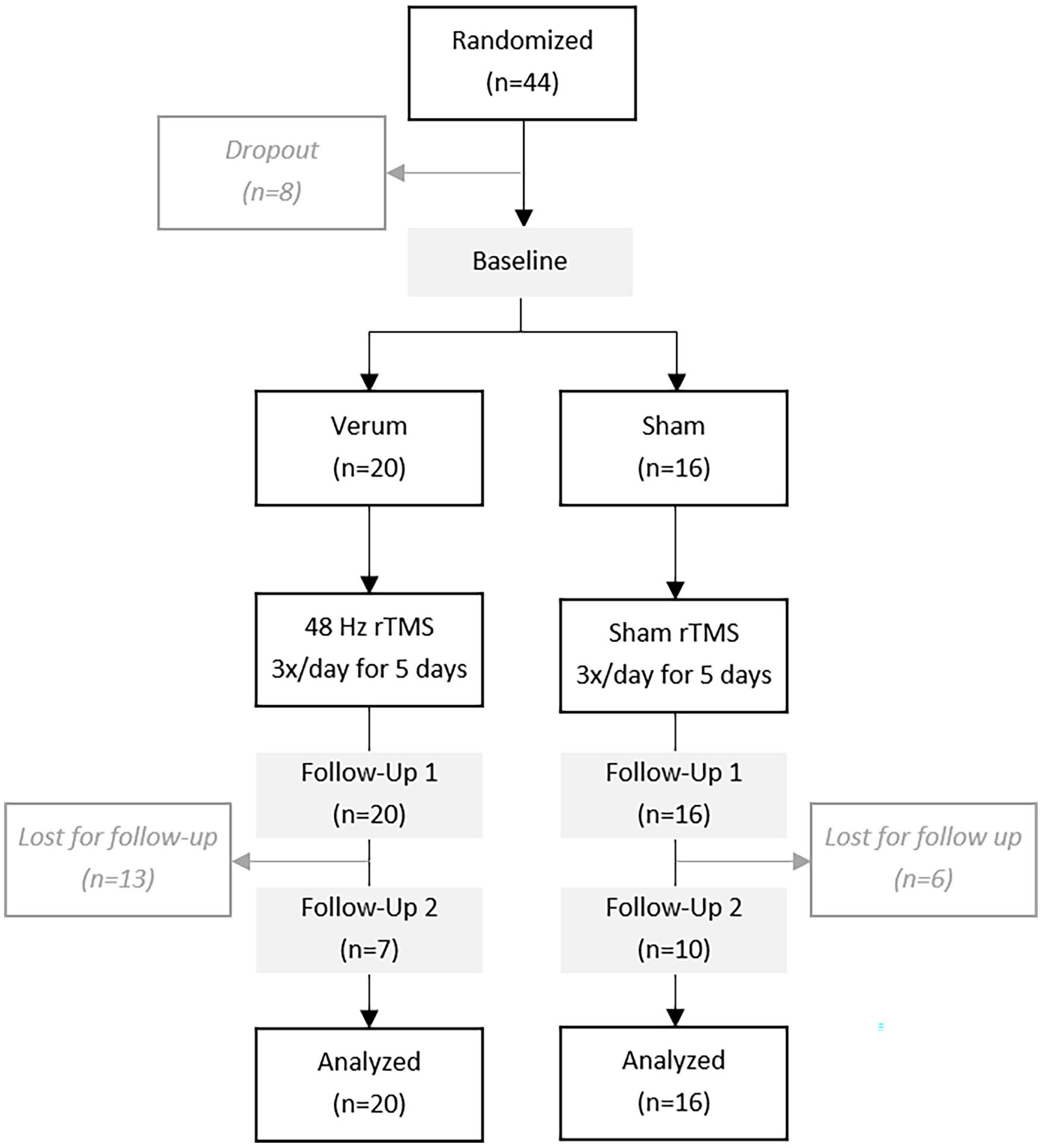
Study flow chart.
Study Design
The study design was a double-blind, randomized sham-controlled, with a 1:1 allocation ratio.
Participants were allocated to either the verum (rTMS 48 Hz) or the control group (sham) using a block randomization with an AABB distribution model (A = experimental; B = control). Neither the patient, nor the involved investigators were aware of the assigned treatment group. Treatment allocations were kept in sequentially numbered envelopes and opened only at the time of enrolment. The medication remained unchanged throughout the study and stimulation was administered in the “on medication” state. Motor function was assessed at baseline and 3 to 4 hours after completion of the last rTMS treatment. The treatment consisted of 15 rTMS sessions during a 5-day period (3 sessions/day on 5 consecutive days), stimulation sessions were delivered hourly.
Clinical Assessments
Study participants were assessed at baseline (V0), at the end of the treatment session (V1 at day 5) and at the end of the trial (V2 at day 30) in the on-medication state. The primary outcome measures were the change in the motor sum-score of the Unified PD Rating Scale, part III (UPDRSIII) 25 from V0 to V1. Secondary outcome measures included the 3 m “Timed Up and Go” (TUG) test measured in seconds, 26 the time needed to walk 8 m (8MW), the 9-Hole Peg Test (NHPT), and summed scores for UPDRSIII items 18 and 19 (speech and facial expression), 20 and 21 (tremor), 23–26 and 31 (bradykinesia), and postural instability (pull-test, item 30).
Device-Based Assessment of Postural Control
Computerized dynamic posturography was performed at V0, V1, and V2 using a standardized balance perturbation method, which measured the degree of medial-lateral and antero-posterior sway using an ultrasound-based measuring system with a movable and adjustable plate (PosturoMed CMS10; zebrisMedical GmbH, Isny im Allgäu, Germany) as described before. 27 Higher values correspond to greater sway, meaning worse clinical performance and impaired postural stability.
Intervention
The rTMS intervention consisted of 15 sessions (3 sessions/day) over 5 consecutive days. A Magstim Rapid 2 (Magstim Co. Ltd, Whitland, UK. Device number P/N: 3012-00) with an air-cooled figure-of-8 coil delivered stimuli with the coil held tangentially over 3 regions: the inion, 4 cm lateral to the left of the inion, 4 cm lateral to the right of the inion. Placebo treatment was performed with a placebo coil (Magstim) with identical appearance, sound emission, stimulation of superficial tissue (muscles), and operation of the TMS coil without stimulating cortical tissue. Due to a lack of evidence that individual adjustment of stimulator output via determination of motor threshold (MT) correlates with stimulation efficiency in distinct brain regions, 19 we used a fixed output intensity of 50%.
The cerebellum was chosen as stimulation target, because of growing evidence for pathophysiological involvement of the cerebellum in PD.28,29 New findings in anatomical connections linking the cerebellum and the basal ganglia support this hypothesis. 30 We stimulated 3 cerebellar sites, because this protocol was effective in patients with spinocerebellar ataxia (SCA) in previous studies.19,31,32 Patients laid their head down on a pillow on a small table in front of them, and the TMS coil was held facing upwards. The coil was held in place against the patient’s head with a mechanical arm. The participants were provided with ear plugs.
The experimental group received rTMS similar to a standard intermittent theta burst stimulation (iTBS) paradigm consisting of 3 stimuli bursts at 48 Hz, in contrast to standard protocols consisting of 50 Hz, repeated at 5 Hz frequency. Approximately 50% of maximum stimulator output intensity was delivered. One train consisted of 15 bursts with a 5.4 second inter-train interval, for each region 32 clockwise trains were delivered. Stimulation sessions were done hourly, 3 sessions were applied per day resulting in 64 800 pulses in total for 1 participant.
Statistical Analysis
Standard descriptive statistics were provided. Normal distribution was tested by the Kolmogorov–Smirnov test. Outliers were checked for plausibility prior to inclusion in analysis. Subjects that received at least 1 stimulation were included in statistical analysis.
A 2-way repeated measures analysis of variance (ANOVA) with, if applicable, Greenhouse–Geisser sphericity correction was used to investigate interaction effects and simple main effects of timepoint and treatment. In-group differences between V0 and V1 were analyzed using the paired Student’s
Results
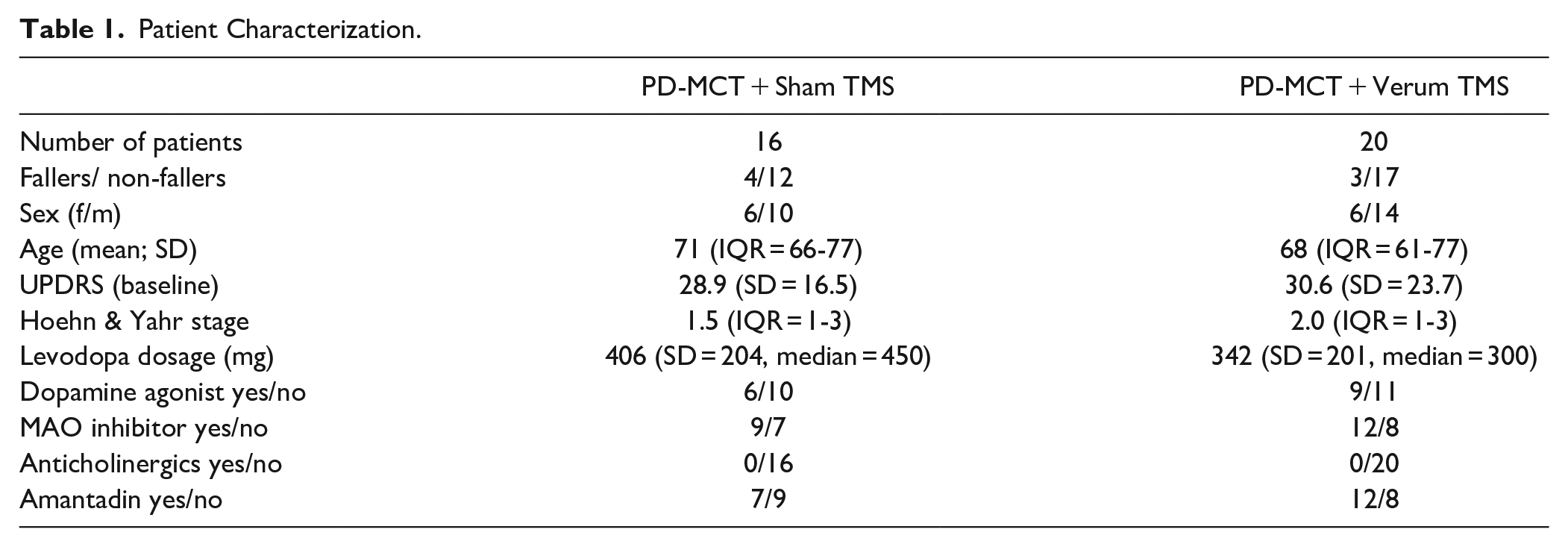
44 patients were randomized for this study. The verum group was slightly larger (20 vs 16 patients) and had fewer females (30% in the verum vs 37.5% in the sham group). Mean age was similar between the groups. There were more patients in Hoehn & Yahr stage II in the verum group (n = 6) as compared to the sham group (n = 2), but disease severity as assessed by UPDRS motor score was similar among both groups. Patients in the sham group took slightly higher average daily doses of levodopa, but the frequency of additional Parkinson medication was similar between both groups. Per study protocol, medication remained stable throughout the study. For detailed description of patient characteristics, see Table 1. Eight patients dropped out prior to first stimulation: 5 due to necessity of adapting medication according to treating physicians, 1 patient due to concurrent coronavirus infection, 1 patient due to a preexisting diagnosis of epilepsy, and 1 patient due to doubts about the diagnosis of idiopathic Parkinson’s disease.
Patient Characterization.
Of the 36 patients that received either verum or sham rTMS, 100% completed a follow-up visit directly after end of the 5 days stimulation protocol. Willingness to return for a second follow-up visit 1 month after end of stimulation was low in both groups. Only 10 patients (62.5%) of the sham group and 7 patients (35%) of the verum group completed the second follow-up visit. Due to the small number of long-term follow-up visits, statistical analysis beyond the first follow-up visit was rendered impossible. Adverse events were limited to headache reported by 1 patient and subjective worsening of gait disturbance reported by another.
The results of the 2-sided repeated measures ANOVA of UPDRS motor scores indicated a significant main effect for timepoint (
Results From 2 × 2 Repeated Measure ANOVA With Timepoint and Treatment as Independent Factors.
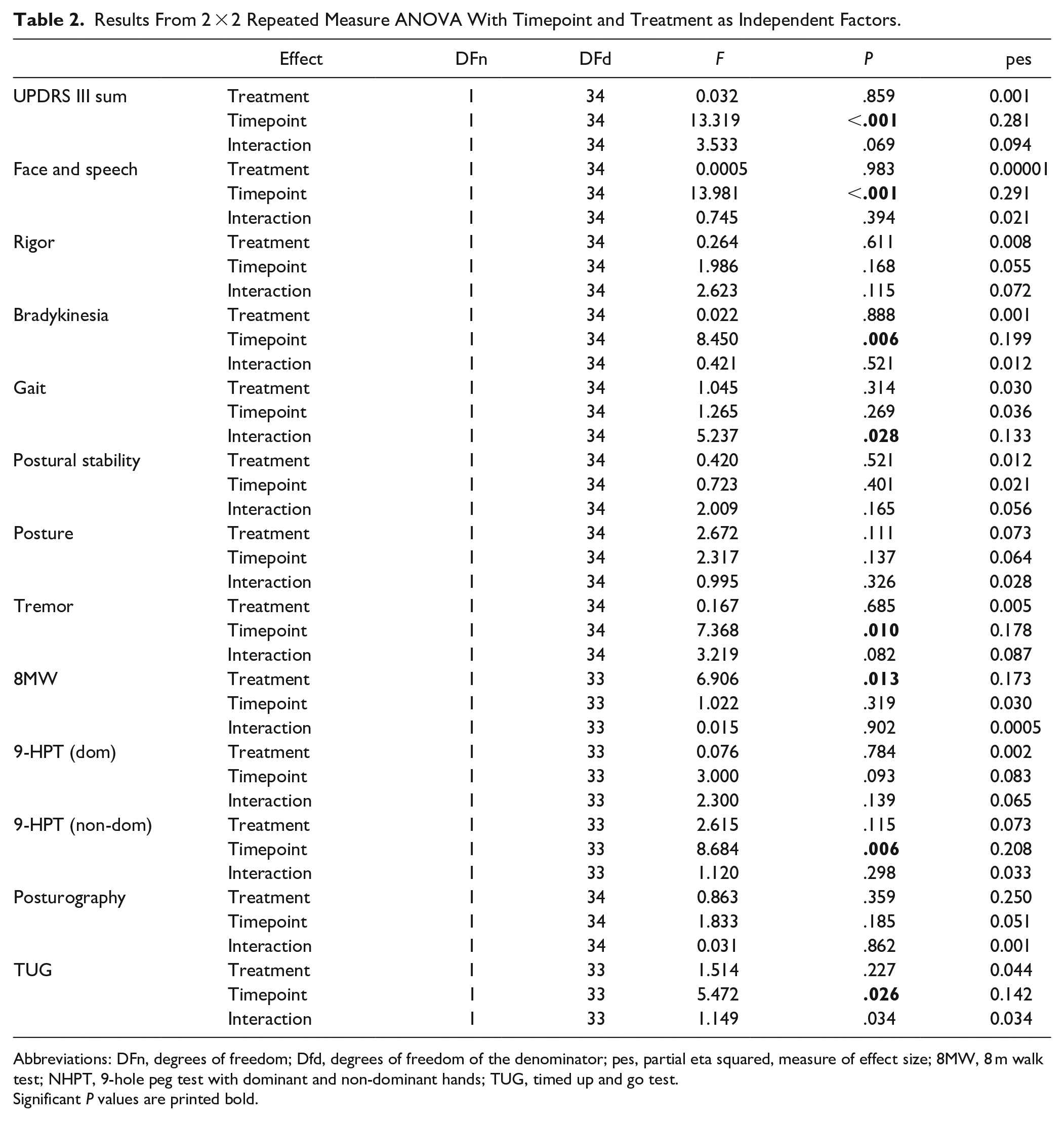
Abbreviations: DFn, degrees of freedom; Dfd, degrees of freedom of the denominator; pes, partial eta squared, measure of effect size; 8MW, 8 m walk test; NHPT, 9-hole peg test with dominant and non-dominant hands; TUG, timed up and go test.
Significant
Comparison of Outcome Measures From Patients in the Sham and Verum Group Before (V0) and Directly After (V1) Cerebellar rTMS and 1 Month After Treatment (V2).
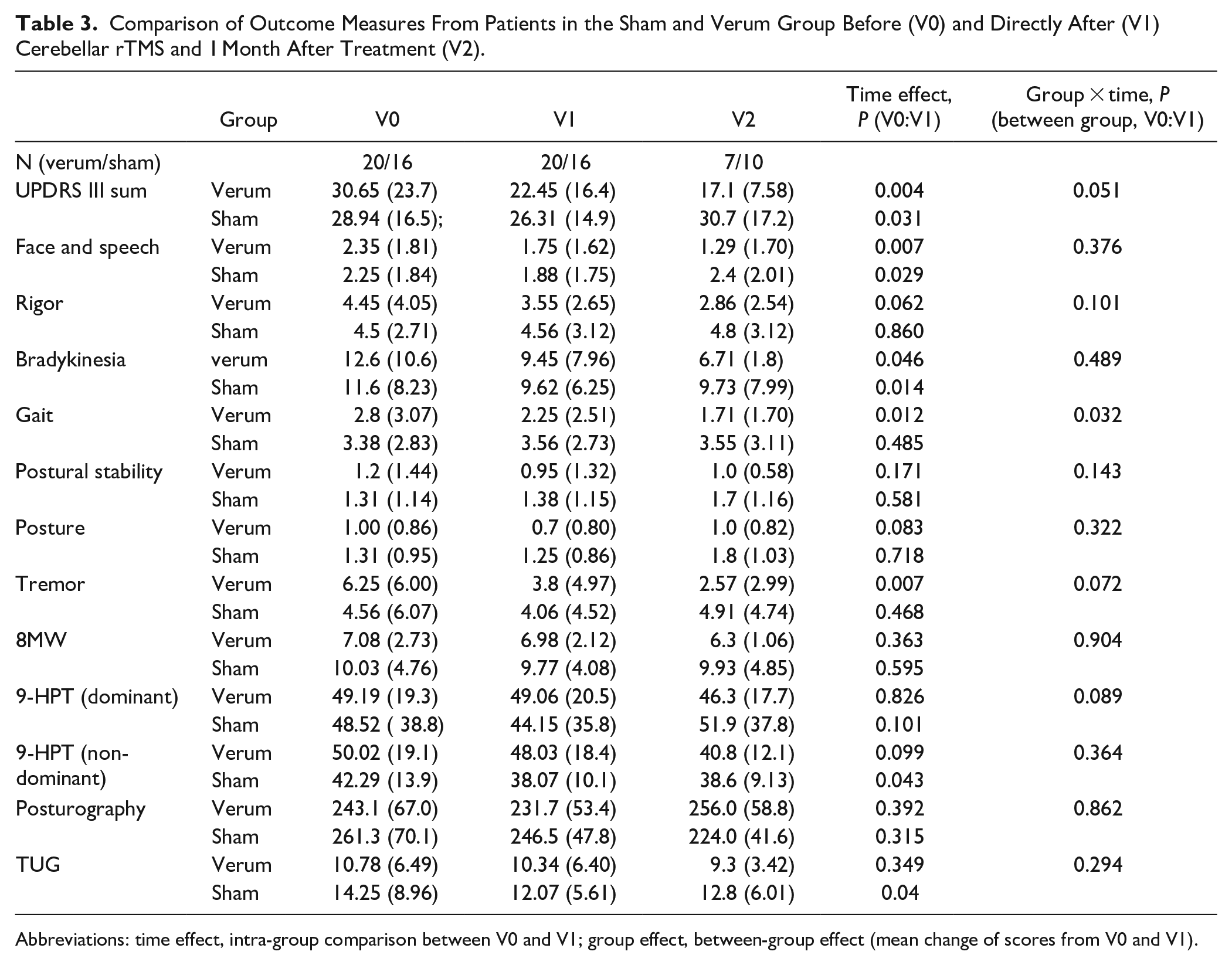
Abbreviations: time effect, intra-group comparison between V0 and V1; group effect, between-group effect (mean change of scores from V0 and V1).
To analyze which domains contributed most to a reduction of UPDRS motor score, we divided the score into subdomains as followed: face and speech (items 1-2), rigor (item 3), bradykinesia (items 4-8 and 14), gait (Items 9-11), postural stability (item 12), posture (item 13), and tremor (items 15-18).
The ANOVA indicated a significant interaction effect for the gait subdomain (
To fully investigate specific group differences, we conducted post-hoc
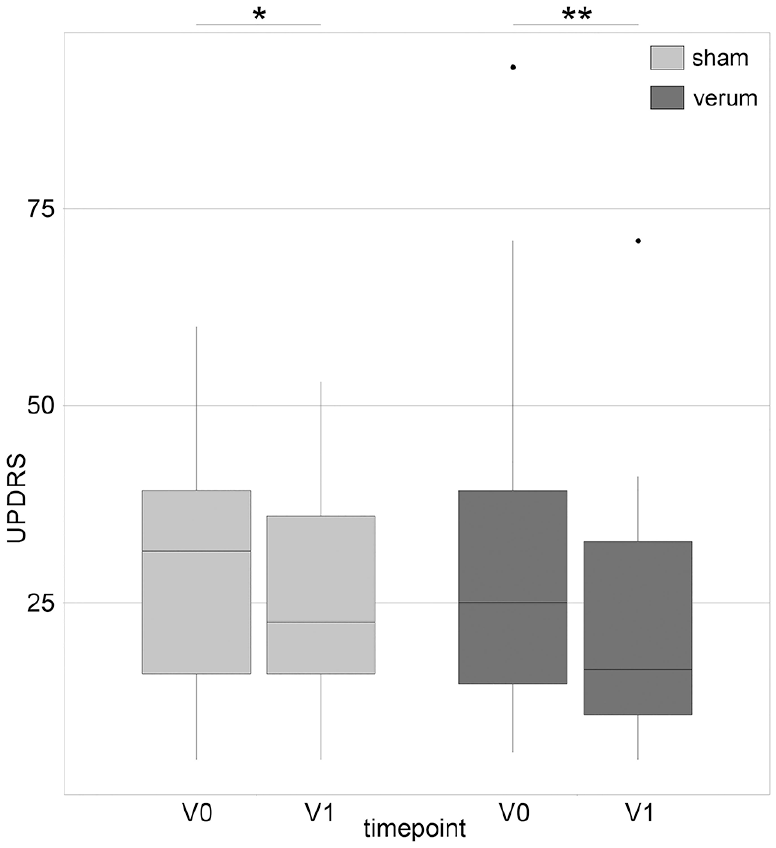
UPDRS motor score before and after cerebellar rTMS in sham and verum group.
There was a significant reduction of UPDRS subscores in both groups for the domains face and speech (
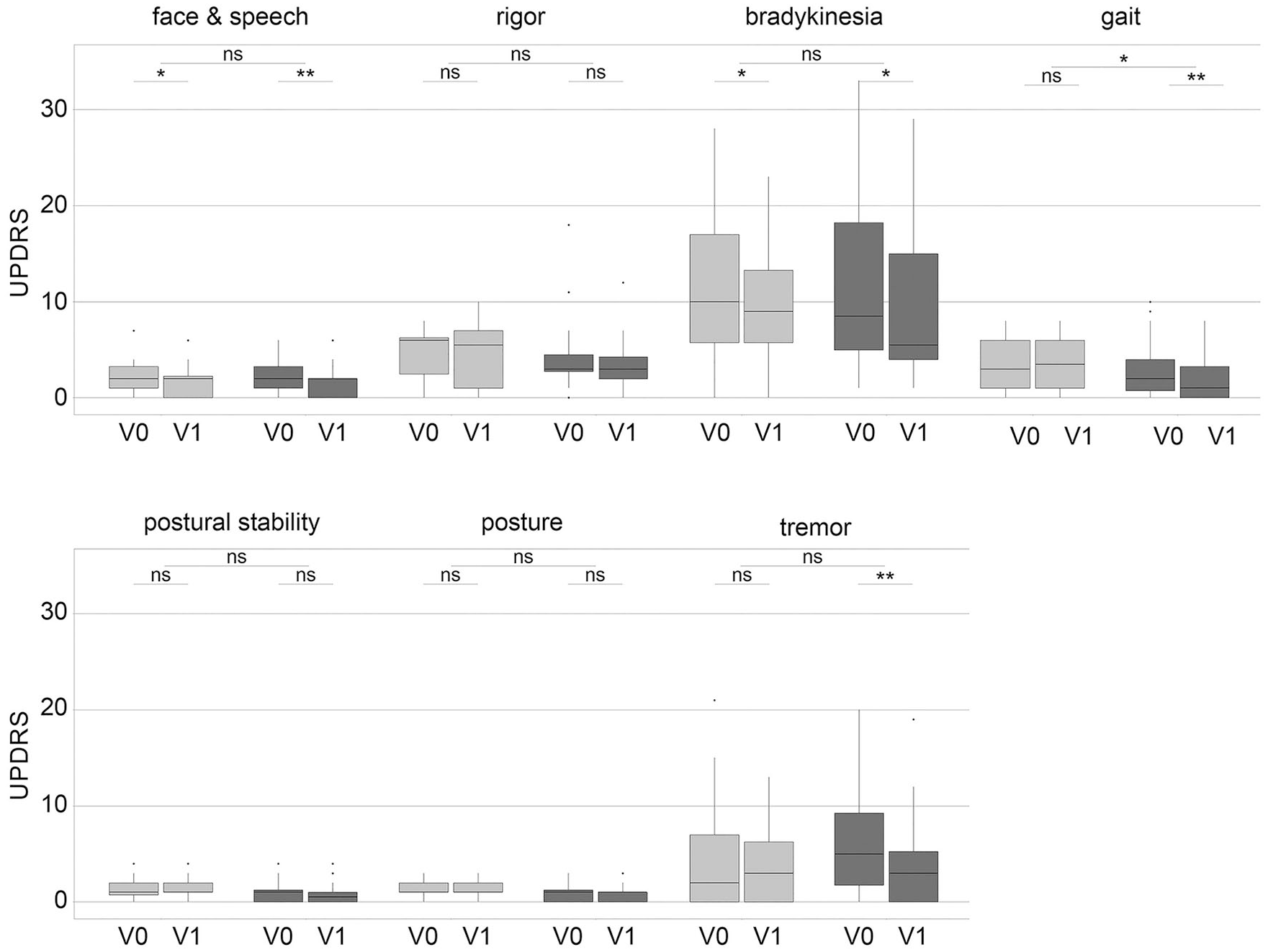
Reduction of UPDRS subdomains before and after cerebellar rTMS in sham and verum group.
The inter-group analysis of UPDRSIII reduction was significant for the gait domain only (
Analysis of the change in UPDRS motor sum-score in the subgroups of “non-fallers” also did not reach significance (
Of the secondary outcome parameters, only the intra-group analysis of the 9-hole peg test of the non-dominant hand and the TUG test in the sham group reached statistical significance (
Discussion
In this study, we found that both the PD-MCT alone and PD-MCT combined with rTMS can improve motor function in PD as measured in UPDRSIII in a randomized controlled trial design. There was a trend toward more improvement in the rTMS-group with a marginally significant reduction of the UPDRS motor score in the inter-group analysis (
Furthermore, only the rTMS-group showed a significant improvement in the tremor subdomain, but inter-group analysis did not reach significance. The subdomains face and speech, and bradykinesia showed significant improvement in both groups.
In this study, we demonstrate that the tremendous expansion of the total number of rTMS stimuli to 64 800 pulses within 5 days was feasible, effective, and tolerable with only minor adverse events. To our knowledge, this is the first study in the field of movement disorders to use such an ambitious stimulation protocol.
Effect of PD-MCT on PD Motor Symptoms
Recent investigations described that PD-MCT improves PD motor symptoms and of quality of life.1,2 Longer duration of therapy improves treatment effects.1,2,33 In Germany, the majority of PD patients is treated for a period of at least 14 days. 34 In our study PD-MCT duration was 8 days, but still showed beneficial treatment effects as measured by UPDRSIII. PD-MCT usually permits adjustments in patients’ PD medication. In this study, changes of medication were not allowed to prevent interaction with treatment protocol and maintain comparability of the intervention groups. This is a methodological strength of this study in comparison to other clinical studies focusing on PD-MCT. Interestingly, even without adaption of PD medication we observed improved motor function in the PD-MCT group, but not all UPDRS subitems responded, for example, tremor. This is in line with the findings of Hartelt et al 2 showing that tremor responded less than non-tremor symptoms. In general, we want to point out that our study design regarding PD-MCT lacks a control group and that a causal relation between PD-MCT and the observed improvements cannot not be proven.
Effect of rTMS on PD Motor Symptoms
We partly confirmed our hypothesis that the combination of rTMS and PD-MCT exerts additional benefits in comparison to PD-MCT alone. This finding was not unexpected, because 2 treatments should be superior to 1 single treatment. In this study we chose to test PD-MCT against the combination of PD-MCT plus rTMS, because of the rather time intensive study protocol with 3 stimulations on 5 consecutive days. While in hospital for PD-MCT, this protocol is feasible for most patients, but the same protocol in an outpatient setting might have caused difficulties for many patients. This is also reflected by the high dropout rate after discharge from hospital in both groups, while initially, all patients had intended to return for a follow-up visit 1 month after stimulation. The combination of intensified physiotherapy and rTMS is therefore of special interest in the context of neuro-rehabilitation, because this gives the opportunity to greatly expand duration of intensified physiotherapy. This may help to fully exploit the therapeutic potential. A similar concept has been used to treat patients with hemispheric strokes and led to improved gait and balance function in these patients. 35
The statistical analysis of change of the UPDRSIII sum score showed a trend favoring the rTMS treatment in comparison to sham treatment, but marginally missed the level of significance. We cannot rule out that elimination of the PD-MCT from the study design would have led to more items with significant differences between rTMS and sham group. A clear-cut superiority over sham was observed for the subdomain gait as measured with the sum of subitems 9 to 11 of the UPDRSIII score in the inter-group comparison. We also tested the gait function using other scores like the 8 m walk and the timed up and go-Test, but did not observe the same improvement of gait.
The extent of the improvement of the UDPRS-sum score in the rTMS-group of 8.2 points in our study is similar to other studies. Chung et al 6 observed 8.0 points reduction in UPDRS motor score after 25 Hz stimulation of the M1-cortex in PD patients.
Postural stability measured with the subitem of the UPDRSIII and dynamic posturography was unchanged after rTMS treatment in our study. This was unexpected, because cerebellar rTMS treatment in SCAs improved postural control and gait significantly. 19 Still, it concurs with a recent meta-analysis focusing on rTMS activation of the primary motor cortex (M1) or the supplementary motor area, 7 where postural instability was almost unchanged after stimulation.
Only a limited number of PD studies investigated cerebellar stimulation protocols so far. In the study by Janssen et al, 18 the ipsilateral cerebellar hemisphere, corresponding to the body side most affected by PD, was stimulated using TBS. This led to increased gait speed when walking with small steps, but decreased maximum gait speed with a normal step size. The overall changes in gait were only mild, probably due to exclusive recruitment of patients showing “OFF” state with freezing of gait (FOG); this study was designed to test TBS effects on FOG.
Bilateral continuous theta burst stimulation (cTBS) of the cerebellum in PD patients with levodopa-induced dyskinesia mediated dyskinetic effects, but did not reduce UPDRS scores. 20 The anti-dyskinetic effects of cTBS was later on confirmed by another study. 36
Resting tremor of PD patients objectively assessed by means of kinematic techniques was found not to be reduced after cTBS delivered over the cerebellar hemisphere.
37
In our study, tremor was significantly reduced in the intra-group comparison (
We assume that the positive effects observed in this study are, at least in part, derived from the intensified stimulation protocol. The stimulation field composed of 3 different sites of the cerebellum (bilateral and medial), rTMS was performed 3 times a day and the total number of pulses was 64 800. Previous cerebellar TMS studies in PD patients did not include the median cerebellum. This region includes the vermis that is involved in gait and balance control.38-40. For this reason, the medial cerebellum has already been recommended as promising target location for non-invasive brain stimulation. 18 TMS studies in patients with cerebellar atrophy (eg, SCAs and sporadic ataxia) started including the medial cerebellum and both cerebellar hemispheres since 1999, with positive effects on gait and stance.32,41,42
However, the presented study was not designed to investigate effects of stimulation of different cerebellar regions, and the conclusions on the role of the medial cerebellum remains speculative at this stage. Furthermore, we cannot assure with certainty that our magnetic field reached the medial region sufficiently. The anatomical scalp-to-cortex distance has great influence on the electric field generated by the TMS coil, such that the induced electric field strength decreases with increasing distance from the coil, resulting in less currency reaching the cortical tissue. 43 Thus TMS is restricted to superficial cortical targets, around 2 to 3 cm in depth. 44 Particularly in the vermis region of the cerebellum is the decay of TMS intensity with depth relevant, because in this region the scalp-to-cortex distance is greater (3 cm) than in the lateral cerebellum or neocortex. We cannot rule out that the positive effects observed in our study are based on the lateral stimulation sites solely without impact of the medial stimulated target. A computational modeling of the electric field would have clarified this issue, but was not done in this study.
Putative Neurophysiological Mechanisms Underlying the Observed Changes
The CTC circuit suppresses the contralateral primary motor cortex (M1), which is termed cerebellar inhibition (CBI). In Parkinson’s disease the CBI is reduced in comparison to healthy individuals, suggesting that the excitability of the CTC pathway is decreased.
45
This impaired CTC inhibitory interaction cannot be restored by standard dopaminergic medication.
46
It has been shown before that rTMS using TBS is able to modulate the excitability of CTC circuits in healthy subjects.
47
There is growing evidence to support the idea that the CTC circuit is involved in the pathophysiology of PD tremor and that CTC acts as a primary oscillator of tremor.
48
The “dimmerswitch” model postulates that the basal ganglia induce tremor (“light switch”) and the CTC pathway modulates the tremor amplitude (“light dimmer”).
48
In this study, tremor was significantly reduced in the intra-group analysis of the rTMS-group (
Recently, the discovery of a disynaptic connection between the cerebellum and the basal ganglia has opened a novel field in research about neuromodulation of the cerebellum. 49 And there is a direct connection between the dentate nucleus and the basal ganglia. 50 That means that basal ganglia and cerebellar circuits can act independently from the level of the cerebral cortex, 51 possibly this pathway may also be involved in rTMS-induced amelioration of PD symptoms. Cerebellar iTBS increases cortical activation during visuo adaptive motor tasks, which is a neuronal correlate of cerebellar involvement in adaptive motor learning. 52 This effect is illustrated by improved error reduction in a visuo-motor adaption task in healthy subjects. 52 There is evidence that such effects are not transient.52,53 Following this line of arguments, improved motor learning, induced by cerebellar TMS, may have boostered the effects of intensified physiotherapy in our study. This synergistic effect can be utilized by combining physiotherapy with cerebellar rTMS and is highly relevant in the context of neuro-rehabilitation, where patients have to re-learn motor functions or at least compensate for acquired motor deficits.
Innovativity of the Stimulation Protocol
The novelty of our study is based on several innovative advancements of the stimulation protocol. We extended the total number of TMS pulses to 64 800, which was never used before in movement disorder investigations. We condensed the treatment duration to 5 days with 3 daily sessions, whereas most rTMS studies are designed over 3 to 5 weeks with 1 session per day only. It has been previously shown that multiple daily stimulation sessions with inter-session intervals of 50 to 90 minutes have a cumulative effect on synaptic strengthening.54,55
Despite this high intense and ultra-short treatment protocol, the side effects and tolerability were identical to other TMS interventions with only few reported minor adverse events. To strengthen the feasibility in the clinical application, we used the same stimulator output intensity for all patients without prior determination of the MT as described before. 19 With respect to the use of rTMS in the cerebellum, there is no evidence in the literature that adjustment of TMS power using MT is correlated with clinical outcome and that the MT of the neocortex predicts the stage of neuronal excitability of a distinct brain region like the cerebellum. Notably, the power has to be chosen high enough to penetrate the bone thickness.
Limitations
Due to the small number of long-term follow-up visits and a high dropout rate after discharge from hospital in both groups, statistical analysis was limited to baseline and the first follow-up visit. Thus, we cannot provide information about long-term effects. However, on a purely descriptive basis, most score reductions remained stable 1 month after treatment. This may hint toward treatment effects for a duration of more than 1 month.
During stimulation, all patients were hospitalized for PD-MCT. This may have led to recruitment from a larger radius with correspondingly longer travels to the study site, which may not have been tolerated by some patients in an outpatient setting. Given the difficult journey due to motor impairment for many patients with movement disorders, this is a possible explanation for the low number of patients in both groups returning to the follow-up visit 1 month after discharge.
Another limitation is caused by the stimulation at 3 locations that was performed in all patients (bilateral cerebellum and medial cerebellum). This does not allow identification of the most effective target position and we cannot rule out that a unilateral stimulation would have been sufficient.
Conclusions and Clinical Implications
To conclude, we demonstrated that the combination of rTMS with a multi-modal complex physiotherapy augments the effects of physiotherapy to improve gait function. Furthermore, using a sham-controlled double-blinded study design we demonstrated that rTMS plus PD-MCT improves key symptoms of Parkinson’s disease. We extended previous studies and previous stimulation protocols showing that an ultra-short and high-intensive intervention is tolerable, feasible with a good safety profile and yields similar effects as longer treatment periods over several weeks. This may lead to advanced stimulation protocols with improved feasibility, as 5 days of treatment are easier to handle for patients with movement disorders and impaired mobility.
In line with Wagle Shukla et al 56 we believe that rTMS has great potential to be applied as an adjunct therapy for the treatment of PD for in- and outpatients and can increase the efficacy of neuro-rehabilitation in general.
Footnotes
Acknowledgements
We would like to thank the Institute for Medical Statistics of the University Hospital Bonn, Germany for their statistical advice.
Author Contributions
Grobe-Einsler M: Formal analysis; Investigation; Methodology; Supervision; Writing—original draft. Lupa A: Formal analysis; Investigation; Writing—review & editing. Weller J: Formal analysis; Writing—review & editing. Kaut O: Conceptualization; Supervision; Writing—original draft.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MGE receives research support from the German Ministry of Education and Research (BMBF) within the European Joint Program for Rare Diseases (EJP-RD) 2021 Transnational Call for Rare Disease Research Projects, from the National Ataxia Foundarion (NAF), and from Ataxia UK, and received consulting fees from Healthcare Manufaktur, Germany, all unrelated to this study. MGE is member of the European Reference Network Rare Neurological Diseases (ERN-RND). All other authors declare no potential conflict of interest with respect to the research, the authorship, and publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability
The data that support the findings of this study are available upon reasonable request.
