Abstract
Background
Yi-Qi-Tong-Luo Granules (YQTLs) is a natural compound of Traditional Chinese Medicine authorized by China Food and Drug Administration (CFDA). These granules are employed in the convalescent stage of cerebral infarction and render notable clinical efficacy. This study aims to uncover the underlying mechanisms of YQTLs on remyelination after cerebral ischemia injury.
Materials and Methods
We established cerebral ischemia model in rats using microsphere-induced multiple cerebral infarction (MCI). We evaluated the pharmacological effects of YQTLs on MCI rats, through Morri’s water maze test, open field test, hematoxylin and eosin staining, and glycine silver immersion. We employed liquid chromatography mass spectrometry metabolomics to identify differential metabolites. Enzyme-linked immunosorbent assay was utilized to measure the release of neurotrophins, while immunofluorescence staining was used to assess oligodendrocyte precursor cells differences and myelin regeneration. We used Western blotting to validate the protein expression of remyelination-associated signaling pathways.
Results
YQTLs significantly improves cognitive function following cerebral ischemia injury. Pathological tissue staining revealed that YQTLs administration inhibits neuronal denaturation and neurofibrillary tangles. We identified 141 differential metabolites among the sham, MCI, and YQTLs-treated MCI groups. Among these metabolites, neurotransmitters were identified, and notably, gamma-aminobutyric acid (GABA) showed marked improvement in the YQTLs group. The induction of neurotrophins, such as brain-derived neurotrophic factor (BDNF) and PDGFAA, upregulation of olig2 and MBP expression, and promotion of remyelination were evident in YQTLs-treated MCI groups. Gamma-aminobutyric acid B receptors (GABABR), pERK/extracellular regulated MAP kinase, pAKT/protein kinase B, and pCREB/cAMP response element-binding were upregulated following YQTLs treatment.
Conclusion
YQTLs enhance the binding of GABA to GABABR, thereby activating the pCREB/BDNF signaling pathway, which in turn increases the expression of downstream myelin-associated proteins and promotes remyelination and cognitive function.
Introduction
In humans, stroke continues to be a leading global cause of significant disability and mortality. It is well-established that both gray matter (GM) and white matter (WM) sustain damage during hypoxic-ischemic injuries, with the latter further precipitating dementia. 1 The hallmark of WM stroke injury is rapid myelin breakdown, 2 and a failure in remyelination within WM lesions often results in substantial functional loss. 3 Oligodendrocyte precursor cells (OPCs) represent the immature stage of oligodendrocytes (OLs), the myelinating cells essential for rapid and efficient action potential conduction. The conversion from OPCs to OLs is a vital process for the repair of damaged WM following ischemic injury. 4 The subgranular zone of the dentate gyrus (DG) in the hippocampus is a primary site of adult neurogenesis. 5 Generation of various neural lineages stems from mature neural stem cells that can self-renew or differentiate into neurons, astrocytes, or OLs in response to specific stimuli. 6
The newly formed cells have the capacity to integrate into the hippocampal circuitry, where they exert influence over learning, memory, and overall cognitive function. 7 Within the DG, gamma-aminobutyric acid (GABA)—a neurotransmitter secreted by certain interneurons and OLs—modulates neural stem cell quiescence, neuron fate determination, and synaptic integration. 8 As a principal inhibitory neurotransmitter within adult brain neural circuits, GABA not only exerts a classical inhibitory effect, but also plays a pivotal role as a developmental signal during the early stages. 9 Its influence spans various biological processes, encompassing neuroblast proliferation and migration, synaptic plasticity, and remyelination. Importantly, GABAergic transmission remains essential for maintaining healthy brain development and function. 10
GABAergic transmission fosters the survival and morphological maturation of newborn neurons by enhancing the cAMP response element-binding (CREB) protein signaling pathway. 11 Within the DG, the expression of pCREB is highly time-sensitive, influencing numerous processes including proliferation, survival, neurite outgrowth, and dendrite branching. The brain-derived neurotrophic factor (BDNF), a target gene of CREB, holds a paramount role in inducing the expression of myelin basic protein (MBP) in the brain. 12 Enhanced activation of the CREB transcription factor facilitates BDNF expression and endogenous BDNF synthesis. The repair of WM hinges on the secretion of trophic factors that stimulate OPC proliferation and differentiation.
Yi-Qi-Tong-Luo Granules (YQTLs) hails from the renowned BuYangHuanWu decoction, targeting cerebral ischemia, particularly during the recuperative phase of cerebral infarction. Both Phase III clinical trials and observations in 2197 cases have illustrated the effectiveness and safety of YQTLs for the treatment of ischemic stroke.13,14 Pharmacological investigations have revealed the neuroprotective properties of this formulation, with its mechanism of action involving anti-apoptotic effects, the promotion of angiogenesis, and mitigation of cerebral ischemia-reperfusion injuries.15,16 Yet, the complete potential mechanisms of YQTLs remain uncharted. As such, our study was designed to evaluate the impacts of YQTLs on remyelination and neural repair post-stroke, and to investigate the gamma-aminobutyric acid B receptors (GABABR)/CREB signaling pathway as a potential mechanism of the drug’s action. A deeper comprehension of these processes is essential for developing therapeutic interventions targeting WM injury following cerebral ischemia.
Material and Methods
Animal Preparation
Sprague-Dawley male rats (170-210 g) were procured from Beijing Vital River Laboratory Animal Technology Co., Ltd. (Beijing, China, No. 110011221104951125). The rats were housed in a regulated environment with a consistent temperature of 21 ± 2°C and a 12-hour light-dark cycle. All procedures involving animals adhered strictly to the guidelines established by the Ethics Committee of Xi Yuan Hospital of China Academy of Chinese Medical Sciences (Protocol No. 2022XLC040-2) and the National Guidelines for Animal Protection, ensuring the welfare and ethical treatment of the animals throughout the study.
Establishment of Multiple Cerebral Infarction Model in Microsphere-Induced Rats
Rats were anesthetized via intraperitoneal injection of 1% pentobarbital sodium (40 mg/kg). Following the onset of unconsciousness, the right common, external, and internal carotid arteries were meticulously dissected and isolated. Temporary occlusion of the right internal and common carotid arteries was achieved using vascular clamps. Concurrently, a suspension of fluorescent PE microspheres (106-212 μm in diameter; UVPMS-BY2, Cospheric, USA) totaling 0.2 mL was introduced into the right external carotid artery using a 1 mL syringe, allowing the microsphere suspension to enter the right internal carotid artery and induce an embolic event by impeding cerebral blood flow. In the sham group, the rats only underwent superficial skin incisions and soft tissue dissections under anesthesia.
Drug Treatments in Rats
YiQiTongLuo Granules (YQTLs; Lot: 20051231) were kindly provided by Shineway Pharmaceutical Co., Ltd. (National number: Z20120001, Hebei, China). The granules comprise a variety of active ingredients, namely Asragalus mongholicus Bge. (Huangqi), Salvia miltiorrhiza Bge. (Danshen), Ligusticum chuanxiong Hort. (Chuanxiong), Carthamus tinctorius L. (Honghua), and Pheretima guillelmi Michaelsen (Dilong), with the botanical names being verified via MPNS (http://mpns.kew.org). High-performance liquid chromatography (HPLC) analysis confirmed the principal components of YQTLs. 17
Rats were allocated randomly to 3 groups: Sham, multiple cerebral infarction (MCI), and MCI treated with YQTLs (MCI + YQTLs). Each group consisted of 12 rats. The administration dose for YQTLs was established at 7.56 g/kg, as cited in previous research. 15 YQTLs were introduced via intragastric administration, commencing on the second day post-surgery and continuing until the point of euthanasia. Control groups received equivalent volumes of saline via oral gavage.
Behavioral Evaluation
Morris Water Maze Test
The spatial learning and memory performance were evaluated on day 28, employing the Morris Water Maze (MWM) in alignment with previously established methods. 18 The apparatus consisted of a stainless-steel circular tank (150 cm in diameter and 60 cm in height), divided into 4 quadrants and filled with water maintained at 22 ± 2°C. An obscured platform submerged 1.5 cm beneath the surface of the water with a non-toxic black dye. Memory acquisition trials were carried out 4 times daily over a span of 4 consecutive days. The rats were trained to locate the concealed platform, with manual guidance provided by the experimenter if the platform was not discovered within a 90-second window. On the fifth day, a single probe trial was performed where the platform was removed, and each rat was permitted a 60-second exploration of the pool. The movement of the rats was captured using a Morris water maze video analysis system provided by Beijing Zhongshi Dichuang Technology Development Co.
Open Field Test
The Open Field Test (OFT) was employed to evaluate the spontaneous activity of rats in a controlled environment. The testing arena was a quiet, open-field enclosure, measuring 50 cm × 50 cm × 50 cm and demarcated into 25 square grids. Each rat was gently positioned in the central grid at the onset of the test, with a 3-minute interval allowed for free exploration of the space. Two behavioral parameters were subsequently recorded: (a) horizontal movement, quantified as the number of grid-crossings achieved by the rat, with a complete crossing defined by the animal moving more than 3 limbs over the grid lines and (b) rearing frequency, interpreted as the number of times the rat stood upright on its hind limbs. Following each rat’s test, the enclosure’s surfaces were thoroughly sanitized with a 75% ethanol solution, ensuring a neutral environment for the subsequent test subject.
Histopathological Staining
Hematoxylin and Eosin Staining
To observe neuronal pathological changes, hematoxylin and eosin (H&E) staining was employed. On the 28th day post-operation, rats were anesthetized and their brains promptly removed and preserved in 4% paraformaldehyde for a minimum of 7 days. Following this, the brains were embedded in paraffin and sectioned into 7 µm thick slices. The sections underwent sequential staining with H&E solutions, and the results were photographed under a Nikon Eclipse E100 microscope.
Glycine Silver Immersion Plating Nerve Staining
Brain sections were introduced into the Glycine silver staining solution C for initial staining, followed by treatment with solution B. Post-treatment, sections were swiftly removed, the residual glycine silver staining solution B was shaken off, and the sections were immersed in glycine silver staining solutions AⅠ and AII. After subsequent dehydration and sealing, sections were examined using a microscope for image capture and analysis (Nikon Eclipse E100, DS-U3, Japan).
Luxol Fast Blue Staining
Paraffin sections underwent deparaffinization and rehydration. The sections were then introduced to luxol fast blue staining A solution for an hour, covered with a membrane, and subsequently immersed into luxol fast blue staining solutions B and C for a period of 15 seconds to allow for differentiation, which was halted with washing as soon as the myelin sheath appeared blue and the background was nearly colorless. Sections were then re-stained with eosin, dehydrated and sealed. As in previous staining procedures, the sections were scrutinized using a microscope for image capture and analysis (Nikon Eclipse E100, DS-U3, Japan). The severity of WM lesions was scored as previously described. The severity of the WM lesions was graded as normal (grade 0), disarrangement of the nerve fibers (grade 1), the formation of marked vacuoles (grade 2), and the disappearance of myelinated fibers (grade 3) as described elsewhere.19,20
No-Targeted and Targeted Metabonomics Profiling
Liquid Chromatography Mass Spectrometry Analysis and Data Preprocessing
Metabolic profiling was analyzed using a Q-Exactive plus quadrupole-Orbitrap mass spectrometer, paired with an ACQUITY UPLC I-Class system (Waters Corporation, Milford, USA). This was done in both ESI positive and negative ion modes. The ACQUITY UPLC HSS T3 (100 mm × 2.1 mm, 1.8 μm, Waters) was utilized for metabolite separation, operating with an injection volume of 2 μL and a flow rate of 0.35 mL/minute. The mobile phase consisted of 0.1% formic acid in water (A) and in acetonitrile (B). In terms of data preprocessing, the raw data was imported into Progenesis QI v3.0 software (Nonlinear Dynamics, Newcastle, UK) for processing that included baseline filtering, nonlinear peak identification, integration, retention time correction, peak alignment, and normalization. To discern overall differences in metabolic profiles between groups, Partial Least-Squares-Discriminant Analysis (PLS-DA) was employed. Utilizing a volcanic map, the visualization of P values (<.05) and Fold change values (<1 and >1) facilitated the screening of differential metabolites. Uniform examination of the specific quantities of different metabolites in each comparison group was accomplished through a statistical map. Venn was used for the expression levels of all significantly different metabolites and the top 50 significantly different metabolites ranked by Variable importance of projection (VIP). A P value of less than .05 and the top 20 represent the enrichment significance of the metabolic pathway. The entire experimental operation was also outsourced to Lu-Ming Biotech Co., Ltd., Shanghai, China.
Neurotransmitters Analysis
Metabolic profiling analysis was conducted using an AB SCIEX Triple TOF 5600 System (AB SCIEX, Framingham, MA), combined with the AB ExionLc system, operating in both ESI positive and ESI negative ion modes. An ACQUITY UPLC HSS PFP column (100 mm × 2.1 mm, 1.8 µm) was used for this purpose. The mobile phase comprised 0.1% formic acid in water (A) and in methanol (B), flowing at a rate of 0.3 mL/min. Metabolite quantification was accomplished by utilizing the multi-reaction detection mode of triple quadrupole mass spectrometry (MRM). The SCIEX OS-MQ software was responsible for automatically identifying and integrating the MRM transitions using default parameters. A mixed standard solution was prepared and subjected to gradient dilution to yield the calibration curve. Subsequent calculation of metabolite concentration information was conducted, based on sampling, dilution ratio, and other parameters, thereby procuring the absolute content information of each metabolite in the actual sample. The entire experimental operation was also performed by Lu-Ming Biotech Co., Ltd., Shanghai, China.
Determinations of the Contents of BDNF, VEGF, TGF-β, and PDGFAA
For the analysis of BDNF, vascular endothelial growth factor (VEGF), transforming growth factor β (TGF-β), and platelet-derived growth factor AA (PDGFAA) contents, all rat serum samples were utilized. Measurements were conducted employing commercial kits (INS-30640, INS-30908, INS-31072, INS-36177, Inselisa Biotechnology Co., Ltd, Hebei, China).
Immunofluorescence Assay
Brain tissue sections, fixed with paraformaldehyde, were incubated with a cocktail of primary antibodies: anti-Neurofilament, anti-MBP, anti-Olig2, and anti-BDNF antibodies (GB12144, GB11226, GB11766, GB11559, Servicebio respectively, all 1:500). Subsequently, sections were exposed to secondary antibodies, namely CY3 goat anti-rabbit IgG (GB21303, Servicebio, 1:300) and goat anti-mouse IgG conjugated with Alexa Fluor 488 (GB25301, Servicebio, 1:400). Following this, sections were stained with 4’,6-diamidino-2-phenylindole (DAPI). The resulting specimens were inspected under a fluorescence microscope (Nikon Eclipse C1, Japan). The positive signals were analyzed by ImageJ software (version 1.8.0).
Western Blot Assay
Protein extractions from cerebral tissue samples were executed employing RIPA lysis buffer, supplemented with phosphorylprotease and protease inhibitor (Wuhan Servicebio Technology Co. Ltd., China). Homogenates were subjected to gel electrophoresis on either 8% or 10% SDS-PAGE gels. Proteins were subsequently transferred from the gel to nitrocellulose membranes (WGPVDF45, Servicebio). These membranes were blocked with 5% skim milk for 30 minutes before being incubated overnight at 4°C with a suite of primary antibodies, each at a dilution of 1:1000. These antibodies included anti-GABAA receptor alpha 1 (GABRA1, GB113973, Servicebio), anti-GABBR2 (27567-1-AP, ProteinTech), anti-Phospho-CREB(S133; GB114684, Servicebio), anti-CREB (9197, CST), anti-Phospho-AKT (4060, CST), anti-AKT (9272, CST), anti-Phospho-ERK1/2 (GB113492, Servicebio), anti-ERK1/2 (GB12087, Servicebio), anti-MBP, anti-Olig2, and anti-β-actin (GB12001, Servicebio). Membranes were then incubated with HRP-conjugated secondary antibodies for 30 minutes. Protein bands were visualized using an enhanced chemiluminescence kit and quantified with AIWBwell™ software.
Statistical Analysis
All statistical computations were performed using the Statistical Product and Service Solutions16.0 software. Data comparisons among multiple groups were undertaken using 1-way analysis of variance, followed by post hoc Tukey or Dunnett test, as appropriate. Data are presented as mean ± SD, and the threshold for statistical significance was set at P < .05.
Results
YQTLs Improves Spatial Learning and Memory Deficits in MCI Rats
The MWM test gauged the spatial learning and memory capabilities of rats from different groups. Figure 1A illustrates the trajectories of the rats. During the training days (2-5), the MCI group exhibited a notable prolongation in escape latency time, in contrast to the sham group; this latency was significantly reduced in the MCI + YQTLs group compared to the MCI group (P < .05 and <.01; Figure 1B). During the spatial probe trial, both the number of platform crossings and time spent in the target quadrant by the MCI group diminished markedly when contrasted with the sham group (P < .01); these metrics were significantly improved in the MCI + YQTLs group compared to the MCI group (P < .01; Figure 1C and D). These findings demonstrate that MCI significantly impaired the memory abilities of the rats, a defect ameliorated by YQTLs treatment.
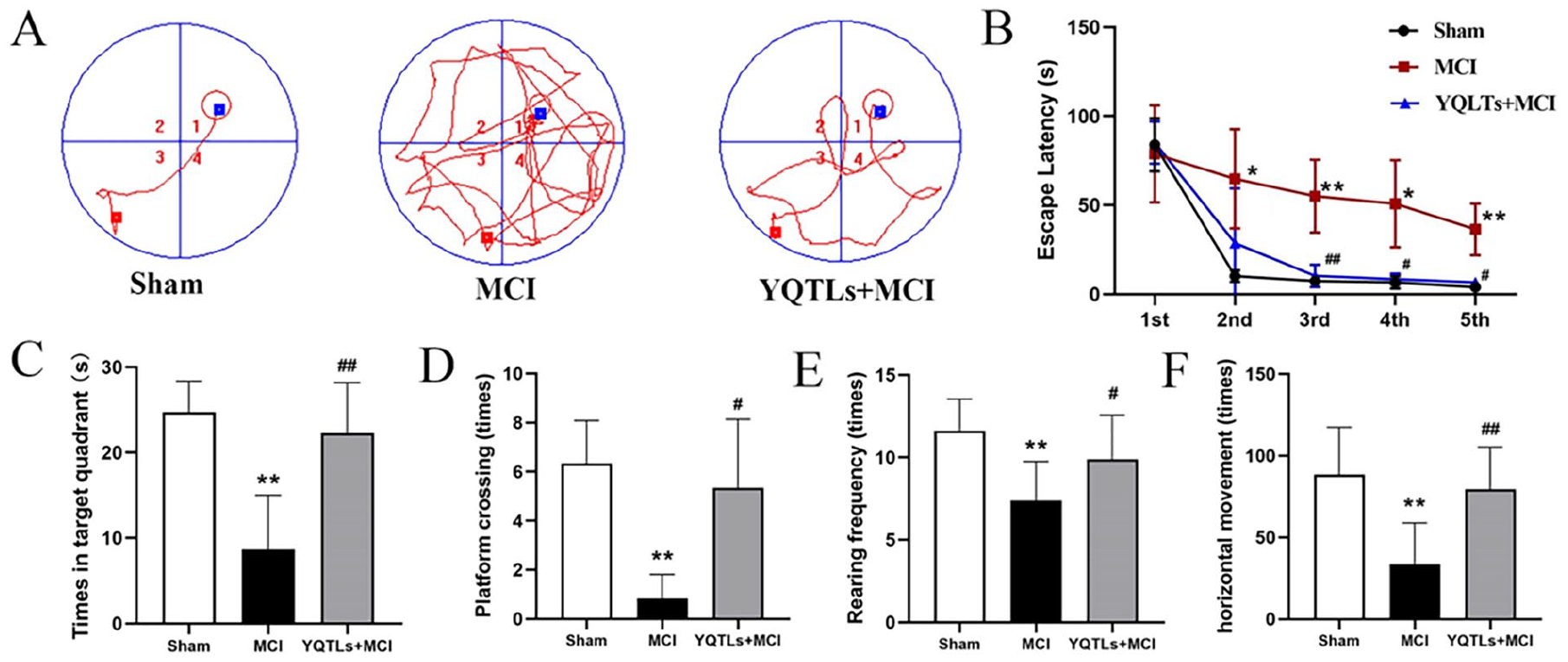
YQTLs facilitates cognitive recovery following MCI. (A) The typical trajectories of rats in different groups in the spatial probe trial. (B) Escape latency. (C) Target quadrant time. (D) Platform crossing. (E) Rearing frequency. (F) Horizontal movement. The data are presented as the Mean ± SD, n = 6; in comparison to the sham group, *P < .05, **P < .01; in comparison to the MCI group, #P < .05, ##P < .01.
In the OFT, the MCI group showed a substantial reduction in both the cumulative number of crossings and rearing movements compared with the sham group (P < .01), indicating a significant decrease in locomotor activity. However, the MCI + YQTLs group significantly ameliorated these behavioral changes, exhibiting an increased number of crossings and rearing movements compared to the MCI group (P < .05 and <.01; Figure 1E and F).
Impact of YQTLs on Histopathological Changes in the Hippocampus Post-MCI
Histological changes in the hippocampal region of rat brain tissue were observation in CA1, CA3, and DG. H&E staining revealed that in the sham group, pyramidal neurons exhibited a clear structure, arranged in multiple, neat layers, with round nucleoli and distinct, dark-blue staining. In contrast, the MCI group’s hippocampal CA3 region showed considerable neuronal damage, evident in significant nuclear contraction, irregular or triangular shape, and disorderly arrangement. However, the MCI + YQTLs group displayed ameliorated pathological damage. Most nerve cells appeared normal, while only a minority exhibited shrinkage and negligible necrotic nerve cells were observed (Figure 2A).
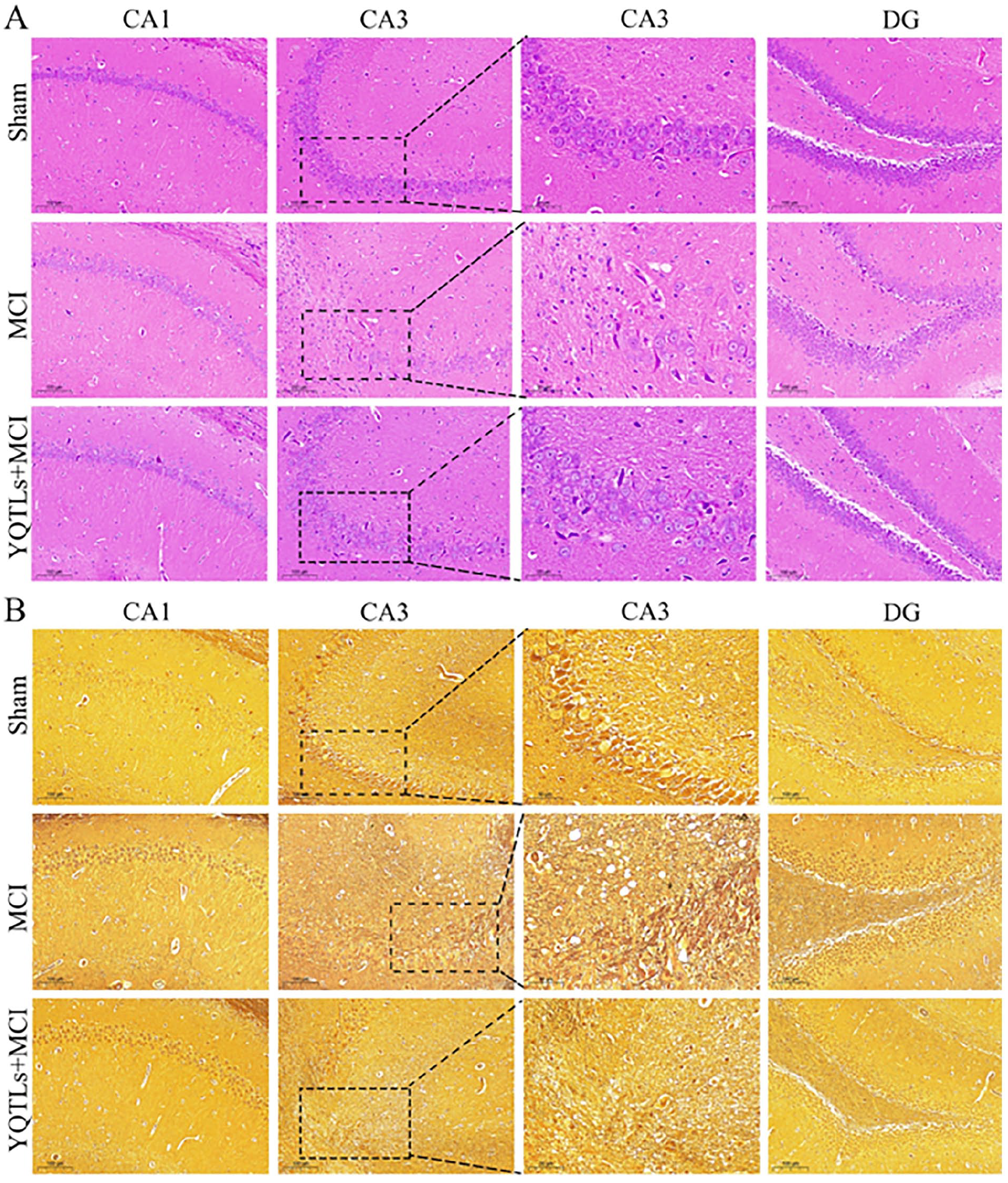
Histopathological staining of YQTLs on the hippocampus region. (A) HE staining. (B) Glycine silver immersion plating nerve staining. Three representative region CA1, CA3, and DG of hippocampus had been shown. CA1, CA3, and DG in scale bar:100 μm; CA3 in scale bar: 50 μm.
Silver staining, typically utilized to stain nerve axons, resulted in a golden yellow background. In the sham group, nerve fibers exhibited regular distribution, while in the MCI group, nerve fibers exhibited degeneration, fracture, and entanglement, with senile plaques and dendrites appearing black. However, in the MCI + YQTLs group, neurofibrillary pathological damage was largely reversed. Most nerve fibers returned to a normal state, with only a few displaying signs of degeneration and fracture, and neurofibrillary tangles were scarcely evident (Figure 2B).
YQTLs Intervention Produces Metabolic Differences in MCI Rats
Brain tissue samples underwent analysis via liquid chromatography mass spectrometry in positive ion mode. Multivariate statistical analysis was performed on the raw data from the sham, MCI, and MCI + YQTLs groups using PLS-DA, demonstrating distinct separations of metabolites amongst the groups (Figure 3A). Further segregation of metabolites across these groups is evident in Venn diagrams, volcano plots, and heatmaps (Figure 3B-D). This implies a considerable alteration in the expression levels of endogenous metabolites, particularly taurine, GABA, N-acetyl-L-aspartic acid, hypoxanthine, niacinamide, creatine, GABA glutamate, L-tryptophan, L-arginine, adenosine, adenosine monophosphate, among others, between sham rats, MCI rats, and YQTLs-treated rats (Figure 3D). Hierarchical clustering provided a visual representation of the top 50 significantly different metabolites, ranked by VIP (Figure 3E). Subsequently, neurotransmitters were scrutinized using targeted UPLC and MRM, with the total ions current (TIC) of standard and tissue samples depicted in Figure 3F. Relative to the sham group, the MCI group displayed a substantial decrease in GABA concentration (P < .01), while no significant change was observed in taurine and L-glutamic acid concentrations. However, the MCI + YQTLs group showed a significant resurgence in GABA and taurine levels compared to the MCI group (P < .01), with no substantial shift in L-glutamic acid concentration (Figure 3G).
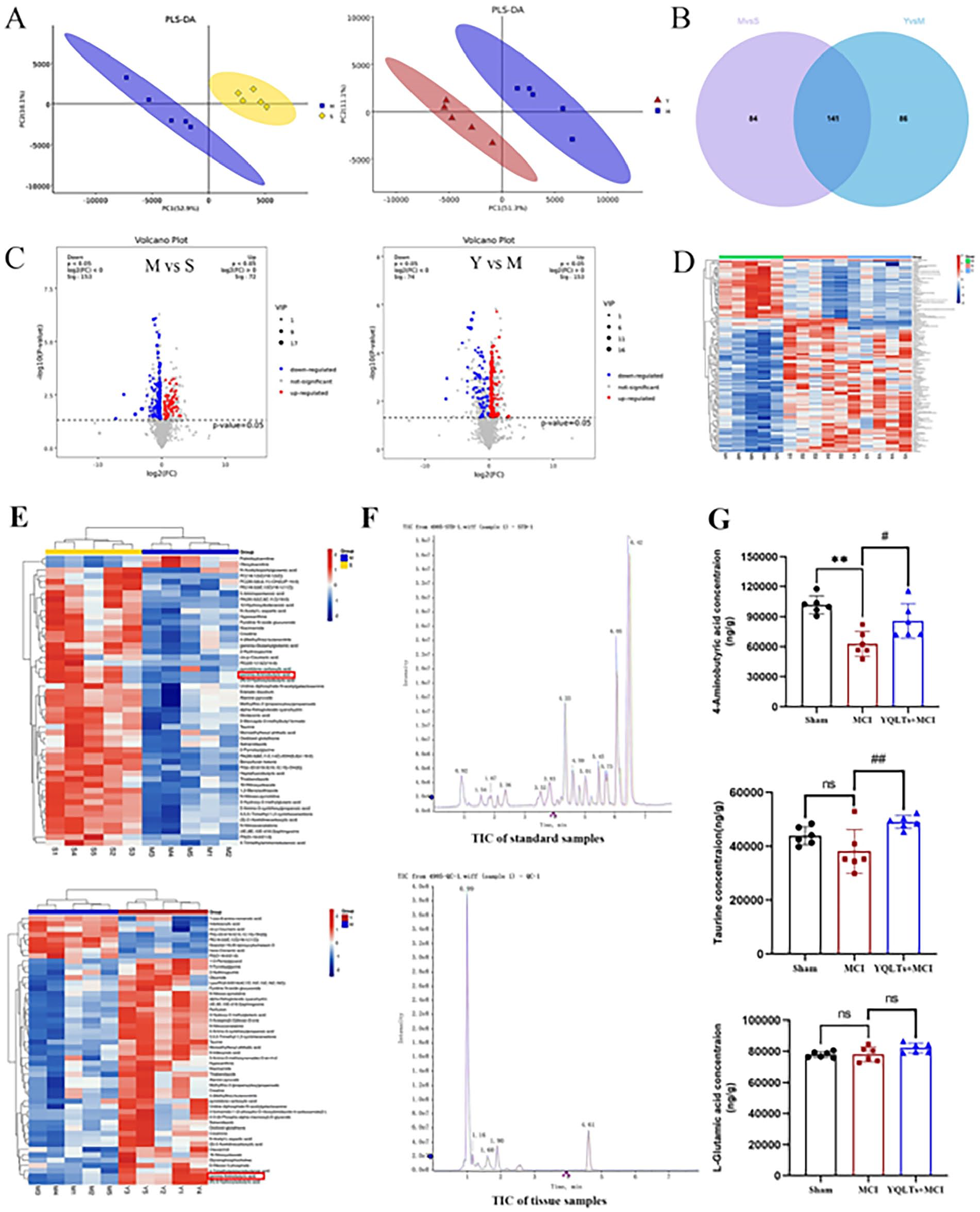
YQTLs on no-targeted and targeted metabonomics profiling. (A) PLS-DA score plots, (B) Venn, (C) volcano plot, and (D) the heatmap of all significant different metabolites. (A-D) Rat brain samples from sham, MCI, and YQTLs groups. (E) The heatmap of the top 50 significantly different metabolites from rat brain samples from sham, MCI, and MCI + YQTLs groups. (F) Total ions current; (G) The concentration of GABA, taurine, and L-Glutamic acid. The data are presented as the Mean ± SD, n = 6; in comparison to the sham group, *P < 0.05, **P < 0.01; in comparison to the MCI group, #P < 0.05, ##P < 0.01.
YQTLs Intervention Augments Neurotrophins Secretion in MCI Rats
The neurotrophin levels in the serum—namely BDNF, PDGFAA, VEGF, and TGF-β—were assessed using enzyme-linked immunosorbent assay kits, with the results illustrated in Figure 4A through D. A marked diminution in the levels of BDNF, PDGFAA, VEGF, and TGF-β was observed in the MCI group relative to the sham group (P < .01). Conversely, treatment with YQTLs led to a substantial upswing in the levels of these neurotrophins in the MCI + YQTLs group after MCI injury (P < .01). Immunofluorescent staining was employed to appraise BDNF expression within the DG region. The MCI group exhibited scarce BDNF positive cells staining, while in stark contrast, the MCI rats treated with YQTLs demonstrated robust and positive BDNF staining (P < .05; Figure 4E and F). These findings suggest that YQTLs intervention may foster neurotrophins production and facilitate neural repair following cerebral ischemic injury.
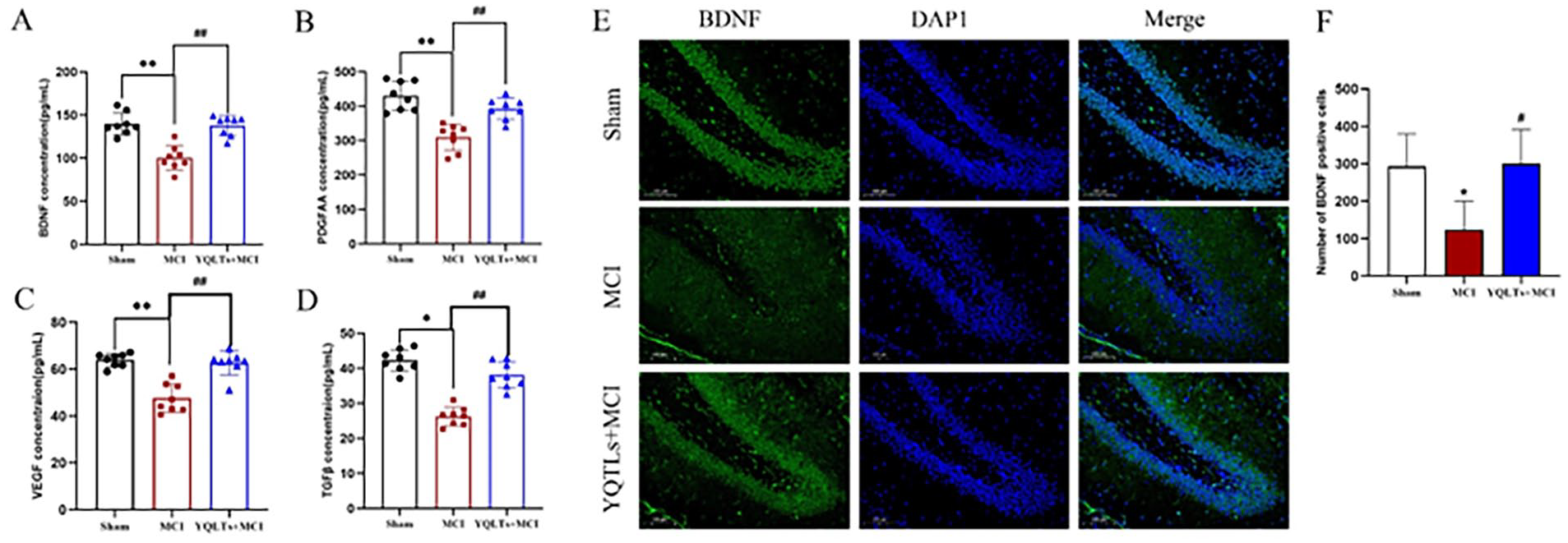
Effects of YQTLs on neurotrophin index in MCI injury rats. (A) BDNF, (B) PDGFAA, (C) VEGF, (D) TGF-β, (E) brain tissue was stained by BDNF (green) and DAPI (blue) and examined by fluorescence microscopy, and (F) BDNF positive cells were counted. The DG region was shown in scale bar:100 μm. The data are presented as the mean ± SD, (A-D) n = 8, (F) n = 3; in comparison to the sham group, *P < .05, **P < .01; in comparison to the MCI group, #P < .05, ##P < .01.
YQTLs Intervention Stimulates Remyelination in MCI Rats
Alterations in Olig2 were investigated using immunofluorescence and western blotting techniques (Figure 5A-C). The findings revealed that both the number of positive cells and protein expression of Olig2 in the DG region were markedly reduced in the MCI group compared to the sham group (P < .05). Conversely, a significant surge in Olig2+ cells and Olig2 protein expression was observed in the MCI + YQTLs group compared with the MCI group (P < .01).
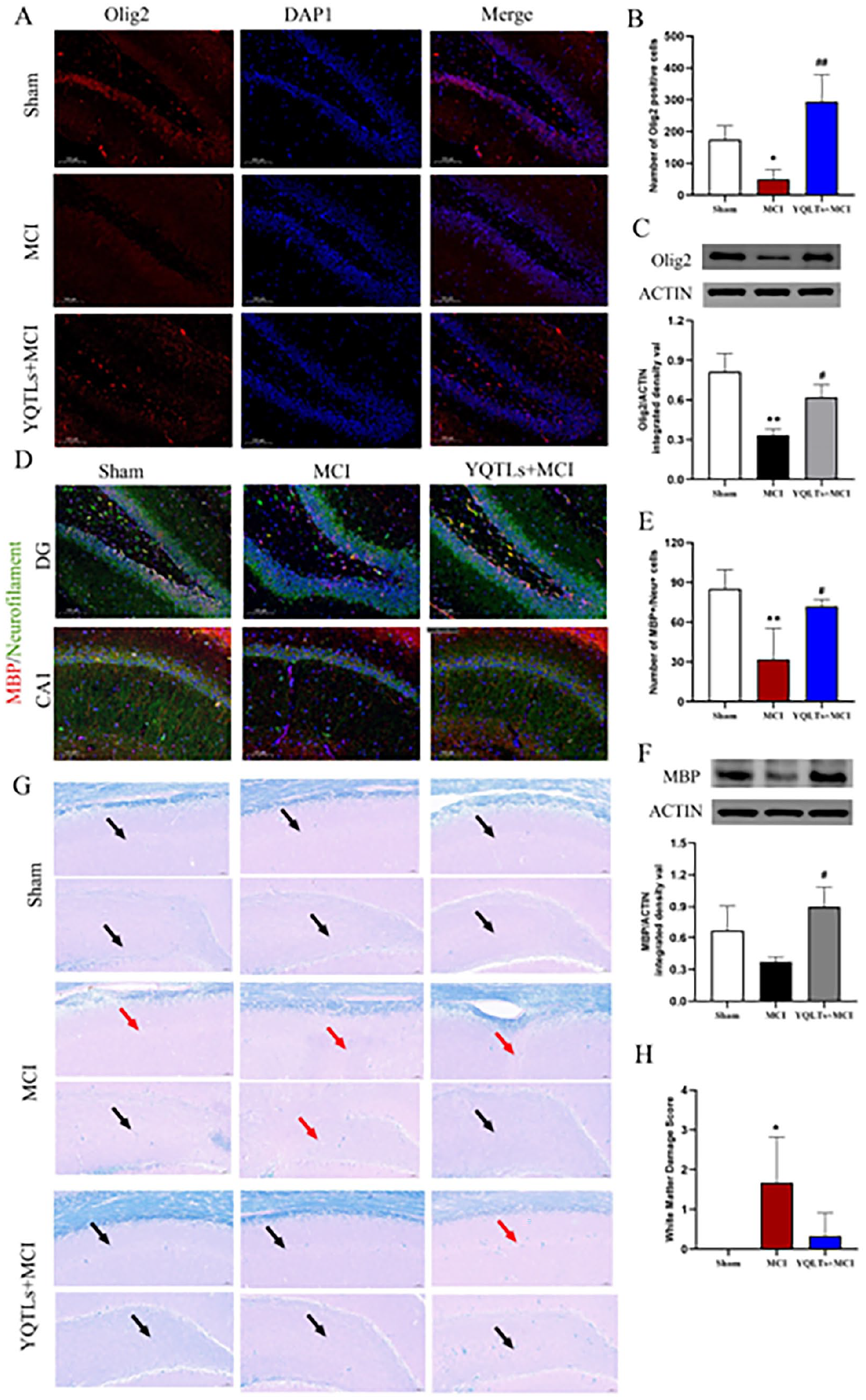
Changes of Olig2 and MBP immunoreactive in the hippocampus after ischemia. (A) Immunofluorescence for Olig2 in the DG region of 3 groups, scale bar: 100 µm. (B) The number of positive cells of Olig2. (C) The protein expression of Olig2 in all groups. (D) Immunofluorescence for MBP in the DG and CA1 region of 3 groups, scale bar: 100 µm. (E) The number of positive cells of MBP/Neurofilament + cells in DG. (F) The protein expression of MBP in all groups. (G) LFB staining in the DG and CA1 region of 3 groups, scattered nerve fiber myelin (black arrow), demyelinated (red arrow), scale bar:50 µm. (H) Quantitative analysis of white matter damage severity. The data are presented as the Mean ± SD, n = 3; in comparison to the sham group, *P < .05, **P < .01; in comparison to the MCI group, #P < .05, ##P < .01.
The extent of post-ischemic myelination was gauged by assessing MBP expression within the DG region (Figure 5D-F). As anticipated, a severe drop in MBP protein expression was noticed in the MCI group compared with the sham group (P < .05). Yet, the YQTLs intervention in the MCI + YQTLs group was found to boost the protein expression of MBP (P < .05). Myelinated fibers, as defined by MBP and neurofilament expression. In the sham group, MBP + myelinated fibers were readily apparent throughout all layers of the CA1 region. However, in the MCI group, the incidence of these fibers diminished. Treatment with YQTLs resulted in a marked visibility of MBP + myelinated fibers within the CA1 region and an upturn in the numbers of MBP + positive OLs in the DG region compared with the MCI group (P < .05).
Luxol Fast Blue (LFB) staining was employed primarily to stain the nerve’s myelin sheath. The myelin sheath, in normal tissue, enveloped the nerve axon, appearing intact and blue in the sham group. Conversely, the MCI group presented a colorless demyelination site at the lesion region and the severity of the WM lesions score was significantly increased (P < .05). YQTLs treatment was able to restore the myelin sheath back to its normal form and the WM lesions score was decreased (Figure 5G and H).
YQTLs Activates GABABR/CREB Signaling Pathway in MCI Rats
An enrichment map detailing metabolic pathways (P < .05) is presented in Figure 6A. Pathways showing significant differences include Alanine, Aspartate, and Glutamate Metabolism, GABAergic Synapse, Taurine and Hypotaurine Metabolism, as well as the cAMP, mTOR, and PI3K-AKT signaling pathways, among others. In the ischemic hemispheres of the MCI group, the levels of GABABR, pAKT/AKT, and pCREB/CREB were noticeably lower than those in the sham group (P < .01 and <.05). Upon administration of YQTLs, these declining levels were reversed with a significant increase in GABABR, pERK/ERK, pAKT/AKT, and pCREB/CREB compared to the MCI group (P < .05; Figure 6B and C). The expression of GABAAR, ERK/ACTIN, AKT/ACTIN, and CREB/ACTIN did not demonstrate any statistical significance across all groups (Figure 6B and C).
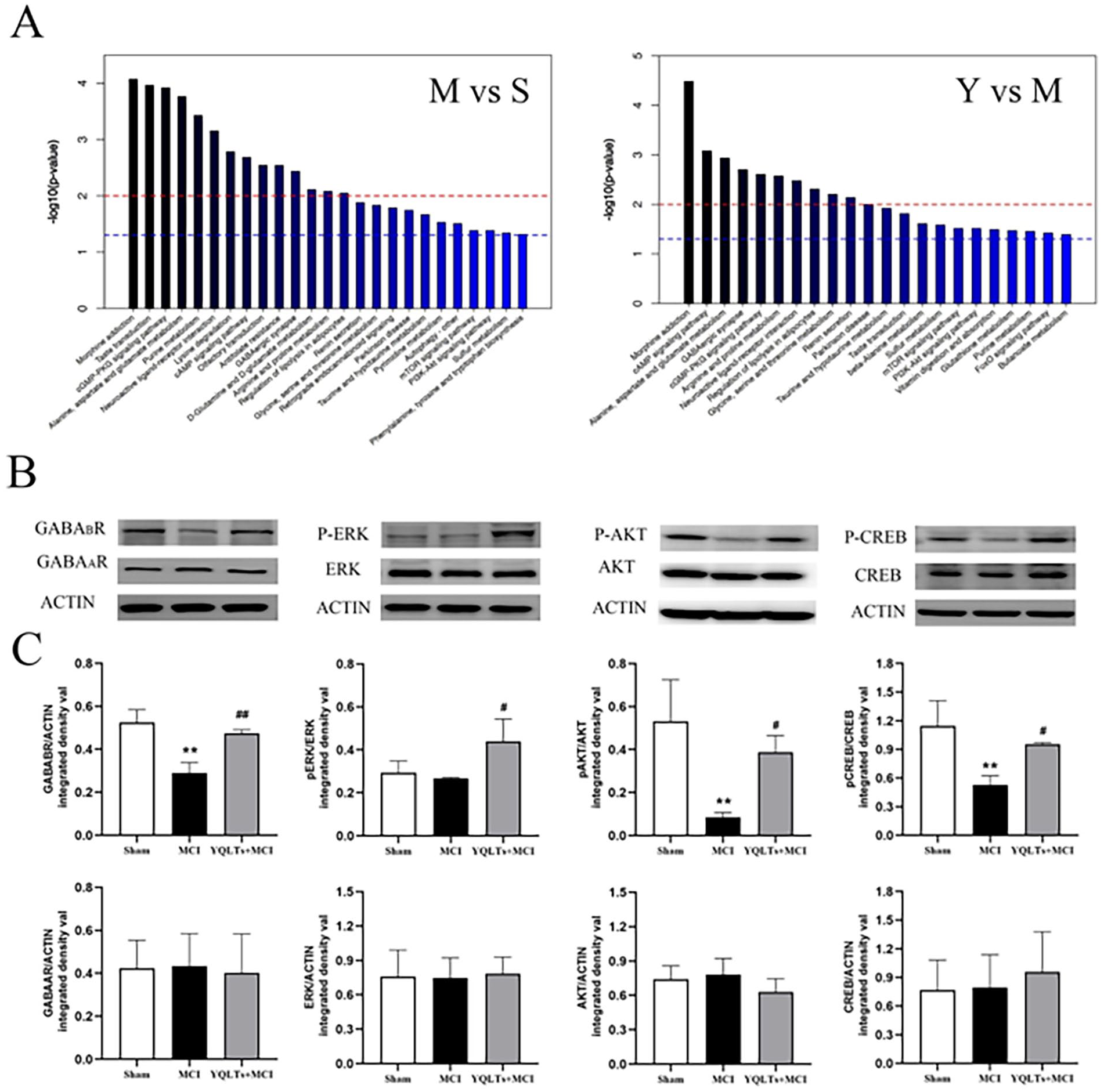
YQTLs activates GABABR/CREB pathway in the brain after ischemia. (A) Enrichment map of metabolic pathways (P < .05), including M versus S and Y versus M. (B) Western blot assays for the protein bands in the ischemic hemispheres of GABAAR, GABABR, pERK/ERK, pAKT/AKT, and pCREB/CREB. (C) The quantitative analysis of all the protein levels. The data are presented as the Mean ± SD, n = 3; in comparison to the sham group, *P < .05, **P < .01; in comparison to the MCI group, #P < .05, ##P < .01.
Discussion
Cardiovascular21,22 and cerebrovascular diseases 23 are 2 main killer worldwide. Among them, cerebral ischemia is a major cause of stroke and harm human health. Our research demonstrated that learning and memory functions in MCI rats were impaired, as evidenced by the MWM test and OFT. Due to the subjective nature of manual recording in open field experiment, compared with automatic data collection and analysis, this index was only used to roughly evaluate the sensorimotor function of ischemic rats, and the protective effect of drugs on cognitive impairment in ischemic animal models was evaluated in combination with water maze experiment. Nevertheless, MWM demonstrated YQTLs treatment post-MCI significantly enhanced spatial exploration ability and spontaneous activity. Spatial learning and memory are dependent on the physiological structure and function of the brain’s hippocampus. In our study, the morphological structure of the rat hippocampus was compromised by the MCI model, leading to neuronal degeneration and a reduction in the number of surviving neurons—consistent with findings from previous studies.15,24 Pathological staining observations revealed that YQTLs offered a notable protective effect against neuronal injury and displayed an inhibitory action on neurofibrillary fracture and entanglement.
Neurotransmitters hold a pivotal role in preserving the equilibrium between stimulation and inhibition within the brain. Previous research has established that YQTLs can stimulate the release of GABA and decrease Glu content to stave off the destruction of hippocampal neurons due to ischemia/reperfusion. 16 In our study, metabolites exhibiting significant differences were examined via non-targeted metabonomics profiling. Our results highlighted GABA—a top 50 differentiating metabolite—which displayed decreased levels in the MCI group and increased levels in the MCI + YQTLs group. Further, quantification of neurotransmitter metabolites, including GABA, was performed using MRM, revealing changes in alignment with the non-targeted metabonomics results. Myelination is stringently regulated by neuron–OL communication and necessitates the involvement of several signals, including neurotransmitters such as GABA. 25 By enhancing the availability of GABA through the administration of tiagabine and vigabatrin, the effects of hypoxia were mitigated, resulting in enhanced oligodendrogenesis and the progression of OPCs to myelinating Ols. 26 Recent investigations have revealed that GABA/GABARs-mediated signaling mechanisms bear considerable importance not merely in neurons, but also in the development and functioning of OPCs. 9
Neurotrophic factors such as BDNF, PDGFAA, VEGF, and TGFβ have been demonstrated to protect neuronal survival in stroke models. 27 Moreover, an increasing body of evidence, from both in vitro and in vivo studies, underscores the role of these factors in the proliferation and differentiation of OPCs and remyelination, particularly BDNF.27-29 For instance, in a mouse model of cuprizone-induced demyelination, BDNF elevation induced by reactive astrocytes was associated with an increase in myelin proteins, which assisted in reversing neurological impairment following demyelination. 29 Olig2, a crucial transcription factor, influences the differentiation and maturation of OLs. Olig2-deficient mice exhibit hypomyelination and direct impairment of neuronal functional development. 30 Furthermore, MBP exerts a positive impact on axonal function, neuronal survival, and cognitive function. 31 Consistent with previous reports, our study found that cerebral ischemia inhibited the secretion of these neurotrophic factors. However, YQTLs treatment induced their release and expression, particularly of BDNF. Concurrently, YQTLs amplified the expression of Olig2, suggesting the promotion of differentiation and maturation of OLs. Moreover, MBP staining and protein expression analysis showed a decrease in MBP expression in the DG and CA1 regions of the MCI group. Yet, following YQTLs administration, the expression level was largely restored, approaching that of the sham group. In LFB staining, we observed some restoration of myelinated nerve fibers in the DG and CA1 regions of the MCI + YQTLs group. These findings indicate a potential restorative effect on disrupted neural circuits in cerebral ischemia, which could aid in cognitive impairment recovery.
Illuminating research has indicated that the phosphorylation of CREB, and the subsequent activation of its downstream signals, may affect the survival, maturation, and integration of nascent granule cells (GCs) within the DG. 32 Concurrently with the occurrence and peak of CREB phosphorylation, GABA displays a depolarizing effect on GCs. Upon counteracting the depolarizing GABA effect with the shNKCC1 virus, CREB activation can mitigate structural morphological damage and cellular death. This observation intimates that GABA may instigate a signaling cascade culminating in CREB phosphorylation. 33 In our experimental findings, we observed inhibited GABA content and CREB phosphorylation in MCI rats. However, YQTLs were shown to enhance GABA content and ultimately stimulate the phosphorylation of its downstream effector protein, CREB, following cerebral ischemic injury.
GABA operates primarily through ionotropic GABAA and metabotropic GABAB receptors. While the expression of GABAA subunits fluctuates with time and differentiation, both GABAB subunits are constantly expressed throughout the developmental process. 34 In DRG-OPC co-cultures, the expression of GABAA decreased in OLs interacting with axons and undergoing myelination, but GABAB expression persisted as OPCs matured into Ols. 35 The role of GABAB in OPC function remains somewhat elusive, with conflicting reports on its role in OPC development. While 1 recent study reported no observable response in OPCs to a GABAB agonist, 36 others noted divergent results. Namely, in vitro activation of GABAB led to increased OPC proliferation, 37 and a GABAB agonist expedited differentiation and myelination in vivo. In contrast, a GABAA antagonist had no impact on OPC differentiation. 38 In line with these findings, we observed a decrease in GABABR in the MCI group, an effect modulated by YQTLs. This observation suggests that YQTLs play a crucial role in regulating the regeneration of the myelin sheath by promoting GABABR.
Despite uncertainty surrounding the downstream proteins affected by GABAB activation, there’s evidence to suggest that CREB might orchestrate myelin gene activation via the MAPK, PI3K, and FAK pathways. 25 Our preceding study employed proteomics and the STRING database to map the protein network interacting with CREB. The resulting analysis suggested close associations between CREB and the cAMP-PKA, Ras-MAPK, and PI3K–AKT pathways. Furthermore, we observed that PKA, AKT, and MAP2K2 protein expression was downregulated in the model group, and this effect was reversed by treatment with BuYang HuanWu decoction. 39 A recent investigation demonstrated that a combination of Astragalus membranaceus and ligustrazine (Ast + Lig) mitigated cerebral I-1 hour/R-48 hour injury by reducing neuroexcitatory toxicity and fostering neuronal survival. This neuroprotective mechanism was partially attributed to NR2B-ERK/CREB signaling. 40 ERK, a serine/threonine-protein kinase, physiologically modulates cell proliferation and differentiation pathways. It can trigger nuclear CREB phosphorylation and the subsequent expression of downstream genes. 41 Moreover, the AKT/PKB pathway, a major effector of nerve growth factor and BDNF, presents an attractive therapeutic target for neurodegeneration. 42 Hence, we assessed the expression of pERK and pAKT. We found that MCI group presented a conspicuous reduction in pAKT expression. YQTLs treatment amplified ERK and AKT phosphorylation, suggesting that YQTLs may enhance pCREB levels possibly via pERK or pAKT pathways. This implicates a potential close relationship between GABABR and pERK and pAKT.
Strength and Limitations
While we have detailed the interventional impact of YQTLs, further exploration is necessary, necessitating the application of target protein inhibitors or small interference RNA in both in vitro and in vivo studies. A comprehensive understanding of how YQTLs influence the associated downstream pathways to control myelin gene activation remains a matter of future determination. We plan to pursue this line of investigation in forthcoming research.
Conclusion
In this study, we demonstrates that YQTLs administration can potentiate OLs differentiation and MBP expression, stimulated by GABA. This appears to be predominantly mediated via the GABABR/CREB/BDNF signaling pathway, thereby contributing to myelin regeneration and cognitive function improvement.
Footnotes
Acknowledgements
This article was polished by Englishgo Editing.
Author Contributions
Xiaodi Fan: Writing—original draft. Min Zhan: Writing—review & editing. Wenting Song: Writing—review & editing. Mingjiang Yao: Writing—review & editing. Guangrui Wang: Writing—review & editing. Tian Li: Writing—review & editing. Yehao Zhang: Writing—review & editing. Jianxun Liu: Writing—review & editing.
Data Availability
Data is available from corresponding authors on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Scientific and Technological Innovation Project of China Academy of Chinese Medical Sciences (CI2021A04617, CI2021A01304), the National Natural Science Foundation of China (82104444), the Fundamental Research Funds for the Central Public Welfare Research Institutes (ZZ15-YQ-008), and National Science and Technology Major Projects (2018ZX09737-009).
