Abstract
Background
Early mobilization (EM) within 24 to 72 hours post-stroke may improve patients’ performance and ability. However, after intravenous thrombolysis (IVT) or mechanical thrombectomy (MT), the increased risk of hemorrhagic complications impacts the implementation of early out-of-bed mobilization. Few studies have investigated EM after IVT or MT for acute ischemic stroke (AIS), and its impact in these patients is unknown.
Objective
To investigate the effect of EM on AIS treated with IVT or MT.|
Methods
We recruited 122 patients with first AIS; 60 patients were treated with IVT, and 62 patients were treated with MT. For each IVT and MT cohort, the control groups received standard early rehabilitation, and the intervention groups received an EM protocol. The training lasted 30 minutes/day, 5 days/week until discharge.
Main Outcomes Measures
The effectiveness of the interventions was evaluated using the motor domain of the Functional Independence Measure (FIM-motor) and the Postural Assessment Scale for Stroke Patients (PASS) at baseline, 2-week, 4-week, and 3-month post-stroke, the Functional Ambulation Category 2-week post-stroke, and the total length of stay at the stroke center.
Results
Both IVT and MT treatment groups showed improved FIM-motor and PASS scores over time; however, only the IVT EM group had significantly improved FIM-motor performance within 1 month after stroke than the control group. Conclusion. An EM protocol with the same intervention time and session frequency per day as in the standard care protocol was effective in improving the functional ability of stroke patients after IVT.
Keywords
Introduction
In recent years, treatments for ischemic stroke have progressed significantly and now include intravenous thrombolysis (IVT) using recombinant tissue-type plasminogen activator (rtPA) and mechanical thrombectomy (MT). IVT with rtPA is the mainstream medical treatment; however, MT with or without thrombolysis is recommended by national guidelines for patients after acute ischemic stroke (AIS) with large vessel occlusion, within 4.5 hours of symptom onset.1,2 Many studies have shown that IVT and MT can improve neurological outcomes in patients with AIS.3-8 While some studies support the use of rtPA, there are concerns about the quality and reliability of the evidence, particularly regarding the time window, dosage, and treatment outcomes of rtPA.9-13 The inconsistency in clinical effects is a significant issue. This variation in outcomes can be attributed to several factors, such as patient selection criteria, treatment strategies, and potential risks, including the risk of increased bleeding. 14 However, the functional independence of stroke survivors still affects their ability to fully integrate into social life. 15 Therefore, early rehabilitation is a core element in acute stroke care alongside medical and pharmaceutical interventions.16,17 However, one of the factors that influences the application of early mobilization (EM) in the acute phase is administration of IVT or MT due to increased risk of hemorrhagic complications.18,19
EM is defined as a program of task-specific sitting out of bed, standing, and walking as soon as possible after stroke; it can improve patient outcomes in activities of daily living (ADLs).20-22 Based on endogenous repair-related events reaching peak levels 7 days after stroke onset,23,24 EM can modify and boost neuronal plasticity processes to improve performance through the promotion of angiogenesis, inhibition of acute inflammatory re-sponses and neuronal apoptosis, and protection of the blood–brain barrier.23,25 The American Stroke Association does not recommend getting out of bed, standing up, or walking immediately, ≥3 times per day, or for a total duration of mobilization sessions more than the usual care protocol within 24 hours of a stroke.26-28 Many studies have examined the benefits of EM within 24 to 72 hours post-stroke.29-33 Despite the current lack of specific data to guide EM, one controlled study suggested that an early out-of-bed mobilization protocol with a 30-minute intervention time and once daily session in a stroke center within 24 to 72 hours after intracerebral hemorrhage resulted in greater benefits in early functional performance than an early in bed rehabilitation protocol. 34 The EM protocol in this study may represent a better option for physiotherapy intervention after AIS in the stroke center. 34
There is limited research regarding EM for AIS patients mobilized after IVT with rtPA or MT.35-38 One study reported that EM for AIS patients mobilized after IVT with rtPA was well tolerated considering the proportion of patients who experienced an adverse response. 39 However, most studies are retrospective, observational, or correlation studies and cannot make definitive causal claims about the impact of EM.35-37 Furthermore, most participants who undergo MT are not included in these studies.35-37
To the best of our knowledge, no study with a balanced number of stroke subtypes based on the treatment method (IVT with rtPA or MT) has explored the impact of treatment methods after AIS on a rehabilitation plan (ie, standard early rehabilitation [SER] vs EM protocols). Therefore, the purpose of the present study was to assess the impact of early out-of-bed mobilization on functional outcomes in AIS patients who underwent IVT with rtPA or MT. We hypothesized that out-of-bed EM with a duration and daily session frequency similar to those of standard care would lead to greater early functional independence benefits in the IVT group compared to the SER protocol at a stroke center. Conversely, considering that previous studies on post-stroke patients have shown that those receiving MT showed greater neurological improvements,40,41 we postulated that out-of-bed EM may have a lower impact on the MT group.
Methods
Study Design
A prospective, randomized, assessor-blinded trial was conducted for 3 months after stroke onset. Participants were recruited from AIS patients hospitalized in the Stroke Center of National Taiwan University Hospital (NTUH) within 24 hours of stroke onset between October 2018 and July 2022. All research procedures were conducted in accordance with ethical standards of the Declaration of Helsinki. The participants provided written informed consent to participate in the protocol, which was approved by the Institutional Review Board of NTUH (201807045RINB) and registered at ClinicalTrials.gov (trial registration ID: NCT03680469).
The primary endpoint was total score on the motor subscale of the Functional Independence Measure (FIM-motor) assessed 1 month post-stroke. Power estimates were based on a prior study investigating the effect of improvements in FIM-motor, 34 which revealed that a sample size of 56 patients for each treatment group would be necessary to achieve an 80% chance (effect size d = 0.6, α = .05, power = 0.80). 34 A total of 60 patients for each treatment group were included to allow for a dropout rate of approximately 5%.
Recruitment Process and Randomization
To evaluate the influence of treatment methods after AIS, the study generated 2 homogeneous groups to be tracked from 1 day after stroke. Randomization was stratified by the treatment method used after AIS: IVT with rtPA or MT. Each randomization block in each stratification group was designated to randomly assign the patients (in a 1:1 ratio using blocks of 4 to 1) to either the SER + EM or the SER group. Allocation was performed by a computer software and was concealed by a research assistant who was not involved in the study. The details of group allocation were placed into opaque, sealed envelopes to be handled only by the research staff. Upon each new entrant into the trial, the next envelope in the sequence was opened, indicating which treatment the patient would undergo. Patients and physical therapists (PTs) were not blinded to the intervention; however, blinded assessment was ensured as assessments were conducted by a different evaluator PT from the treating PT.
The assessments of FIM and PASS for the same patient were performed by the same blinded PT.
Participants
Eligible patients were aged 20 years or older, had confirmed first infarct stroke, and were admitted to a stroke unit within 24 hours of stroke onset. The inclusion criteria were as follows: (1) first ischemic stroke episode treated with either IVT with rtPA within 3 hours of onset or endovascular therapy within 6 hours of onset (including IVT following MT) 42 ; (2) ADLs independent before the stroke; (3) age 20 to 85 years; (4) stroke with unilateral hemiparetic lesion(s) confirmed using magnetic resonance imaging or computed tomography perfusion; (5) no other peripheral or central nervous system dysfunction; (6) no active inflammation or pathological changes in the joints; (7) no other active medical problems that require bed rest; and (8) systolic blood pressure between 120 and 160 mmHg at rest, oxygen saturation >92% (with or without supplementation), heart rate <130 beats per minute at rest, and temperature <38.5°C.21,29 The exclusion criteria were as follows: (1) medical conditions unrelated to the stroke that affect motor performance or the occurrence of major diseases during the study period, such as myocardial infarction, gastrointestinal bleeding, respiratory failure, or pulmonary embolism; (2) any other cognitive, emotional, or behavioral impairments resulting in insufficient comprehension, understanding, or collaboration; (3) inability to provide informed consent; (4) acute deterioration within 24 hours or symptomatic intracerebral hemorrhage defined by a type 2 parenchymal hemorrhage within 24 hours, with an increase of 4 points or more on the National Institutes of Health Stroke Scale (NIHSS); or (5) acute hydrocephalus within 24 hours of stroke onset.
Intervention
All patients in both groups received physical therapy in the early stroke period. According to the protocol, physical therapy began once the patient was randomized, within the first 24 to 72 hours of stroke onset. All the participants received early intervention training for about 30 minutes per day, 5 days a week, until discharge. Considering our hospital’s policy goal of early mobility 43 and patients’ physical abilities and comprehension, priority is given to physical therapy within the first 7 days at the stroke center, and occupational therapy and speech therapy are typically initiated 7 days after stroke or when patients are transferred to general wards post-stroke.
In the SER group, patients were advised to engage in activities while remaining in bed within 24 to 72 hours of experiencing a stroke. Out-of-bed activities were gradually introduced 3 days after admission to the stroke center, usually when the patient was getting ready for transfer to the general ward. Within 24 to 72 hours of stroke onset, in-bed activities were performed in the SER program. These activities included joint range of motion exercises and stretching; bridge exercises and straight leg raising to strengthen core and lower limb muscles; facilitation techniques based on the Bobath or Proprioceptive Neuromuscular Facilitation concept, 44 as well as functional training such as turning over or sitting supported in bed at angles less than 60° 29 to enhance postural control, following the motor relearning programme.44,45 Therapists implemented these exercises in at least 1 set of 10 repetitions, 45 making adaptations based on patients’ daily progress, as evaluated through ongoing reassessment. The progressed plans, which included modifications in the number of sets, movement angles, exercise positions, resistance levels, or the degree of therapist assistance, were tailored to maximize training effectiveness. For patients unable to complete an exercise, therapists adapted to an active assisted or passive manner, performing repetitive activities and utilizing sensory stimulation techniques based on Rood’s approach, such as touch, pressure, and vibration, to promote muscle responses.44,46
The participants receiving the EM protocol were defined as patients with AIS who receive EM treatment in addition to SER care within 24 to 72 hours of stroke onset, as described in the An Early Mobilization for Acute Cerebral Hemorrhage trial. 34 The EM protocol included: (1) sitting on the edge of bed for a minimal duration of 5 minutes (we applied same protocol during the patient’s first time out of bed, with 15 minutes sitting on the edge of bed) and (2) standing at the side of the bed for a minimal total duration of 5 minutes, or (3) side walking and ambulation. All activities were evaluated and supervised by a licensed PT in the stroke center. While sitting on the edge of the bed, sitting reaching training was employed; while in a standing position, pre-walking activities were performed, such as weight-shifting exercises and stepping in place alongside the bed. The use of electronic equipment was prohibited. Close monitoring of blood pressure and heart rate was ensured before, during, and after mobilization. Rest was allowed at any time, if needed, but was not considered when calculating the total training duration. The total intervention was conducted within a 30-min timeframe; therefore, the overall rehabilitation duration did not differ between the groups.
Outcome Assessment
Patient demographics and comorbidities were recorded at the time of enrollment. Baseline characteristics, including age, sex, duration of stay, lesion side, time to initial intervention after onset, treatment method, initial NIHSS score, and initial outcome variables, were recorded.
The primary outcome was a favorable outcome 1 month after stroke measured via the FIM-motor. 47 The FIM-motor is a continuous interval measure that ranges from 13 to 91 points; it consists of eating, grooming, bathing, upper-body dressing, lower-body dressing, and using the toilet, sphincter control, transfers (from bed or chair to toilet, bath, or shower), walking or wheelchair mobility and stair climbing. Each item is rated from 1 (requiring complete assistance) to 7 (completely independent). The sum of the results of the 13 items is the FIM-motor score. A high score indicates that the patient experiences high independence in ADLs.
The secondary outcome was the Postural Assessment Scale for Stroke Patients (PASS). 48 The PASS is a continuous interval measure that ranges from 0 to 36 points. It is obtained by totaling the scores of 12 four-level items, each of which is given a score from 0 to 3 points. A high score reflects the ability to maintain or change a lying, sitting, or standing posture. FIM-motor and PASS scores were recorded at baseline and at the 2-week, 4-week, and 3-month follow-up after stroke onset.
In addition, the Functional Ambulation Category (FAC) 2 weeks after the acute stage 49 and total length of stay in the stroke center were analyzed. The FAC is a 6-point ordinal scale ranging from 0 for nonfunctional to 5 for independent ambulation without an aid. Navigating stairs and indoor walking for a 10-m distance are needed to discern between categories 4 and 5. All adverse events during the intervention period were recorded.
Statistical Analyses
The results were analyzed using Statistical Package for the Social Sciences version 22.0 (IBM Corp., Armonk, NY, USA). We assessed all patients who underwent at least 1 treatment session, intention-to-treat analysis was used for all outcomes, and the carry-forward method was used to account for missing data. Descriptive statistics were used to report participant characteristics at baseline. Depending on the type of distribution, either Student’s t test or the Mann–Whitney U test was used to compare baseline characteristics. Normality was confirmed for all continuous variables using the Shapiro–Wilk test. Consequently, differences in the efficacy of 2 intervention methods were evaluated in the 2 treatment groups: EM group and SER group.
To evaluate the effects of EM on functional outcomes, namely mean FIM-motor and PASS scores, a linear mixed model of covariance was conducted for each intervention method, including improvement in NIHSS score within 72 hours of treatment and baseline NIHSS score as covariates. 36 In the analysis for each treatment type (IVT vs MT), we included treated group (EM vs SER), time (baseline, 2 weeks, 4 weeks, and 3 months post-stroke), and the group × time interaction term as fixed effects and baseline of the outcome of interest as random effects. If significant interactions and main group effects were noted, the appropriate post-hoc analysis was performed. Differences in categorical data were analyzed using Pearson’s chi-square test. All hypothesis tests used a significance level of P < .05.
Results
A CONSORT diagram is shown in Figure 1. According to the inclusion and exclusion criteria, 122 out of 151 patients with AIS were eligible for participation in the study and were randomized in the final groups. The 60 patients who underwent IVT with rtPA were randomized into SER and EM groups, with 30 patients in the SER group and 30 patients in the EM group. After 3 months of follow-up, there was 1 dropout in the SER group due to family reasons; however, all patients were included in the final analysis. The 62 patients who underwent MT were randomized into 2 groups, with 31 patients in the SER group and 31 patients in the EM group; after 3 months of follow-up, there were no dropouts.
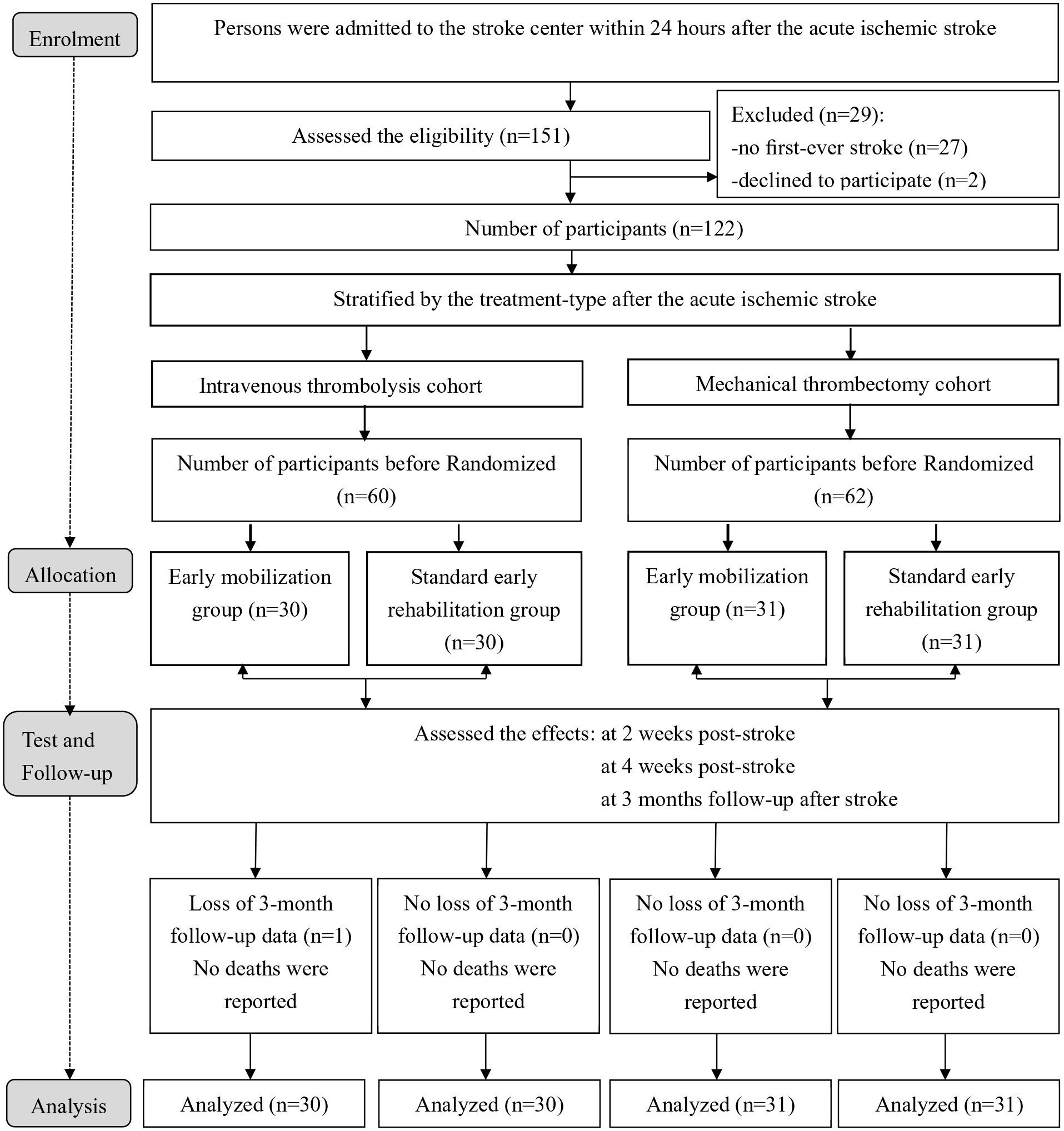
Flow diagram of patients based on CONSORT.
For participants who underwent IVT with rtPA, their demographic and clinical characteristics at study enrollment and their outcome measures are shown in Table 1. These variables were equivalent in the 2 groups after IVT, except for 1 outcome measure; the mean time to first mobilization after symptom onset (defined as time elapsed from admission to achieving out-of-bed unsupported sitting) was 46.2 hours (SD = 12.0) in the EM group and 109.0 hours (SD = 39.1) in the SER group (P < .001). For participants who underwent MT, the mean time to first mobilization after symptom onset was 53.83 hours (SD = 15) in the EM group and 106.03 hours (SD = 31.39) in the SER group (P < .001). At baseline, the 2 treatment-type groups were similar in age, height, weight, sex, the time of starting physical therapy, initial FIM-motor score, and PASS score. However, the NIHSS score of the cohort receiving MT showed significantly greater improvement than that of the cohort receiving IVT 24 hours after treatment (8.2 [SD = 5.98] after MT vs 4.5 [SD = 4.42] after IVT, P < .001). Moreover, none of the participants experienced adverse effects, such as neurological deterioration, hypotension, falls, or line dislodgements, during early rehabilitation or mobilization.
Patient Demographic and Clinical Characteristics.
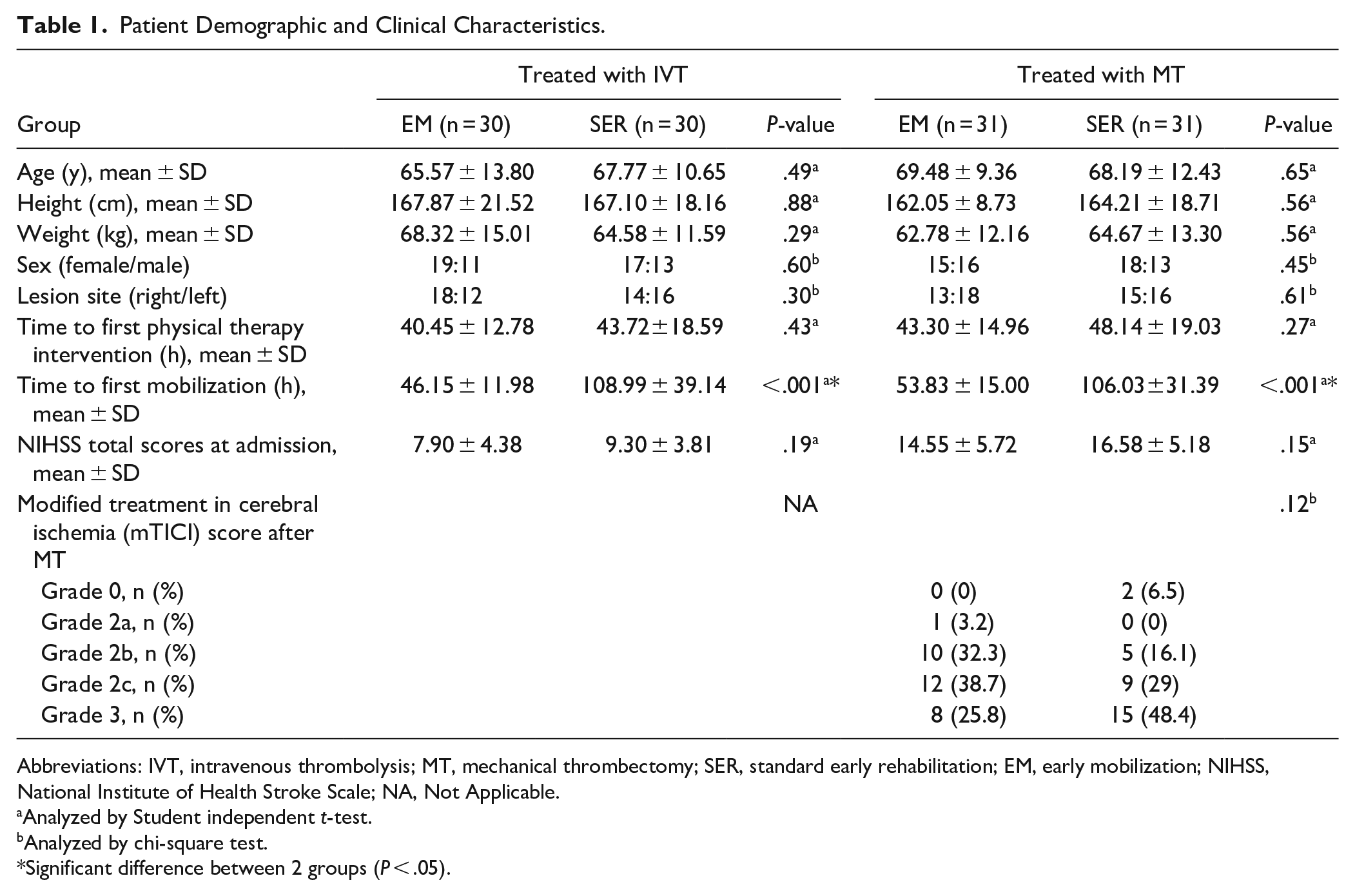
Abbreviations: IVT, intravenous thrombolysis; MT, mechanical thrombectomy; SER, standard early rehabilitation; EM, early mobilization; NIHSS, National Institute of Health Stroke Scale; NA, Not Applicable.
Analyzed by Student independent t-test.
Analyzed by chi-square test.
Significant difference between 2 groups (P < .05).
Table 2 shows the total PASS scores and total FIM-motor scores of the 2 groups that underwent IVT at baseline and 2 weeks, 4 weeks, and 3 months after stroke. After controlling for initial outcome value, significant group-by-time interaction was observed in the PASS (F = 3.437, P = .018) and FIM-motor (F = 4.201, P = .007) scores. There were no significant differences between the groups regarding total PASS scores; however, a significant main group effect was observed for total FIM-motor score. For the FIM-motor score, a post-hoc analysis of the group effect showed that the total scores in the EM group were significantly greater than those in the SER group at the 2-week (F = 5.586, P < .021, partial eta squared (ηp2) = .088(medium effect size)) and at the 4-week follow-up (F = 4.086, P = .048, ηp2 = .066 (medium effect size)).
The Total Scores of the FIM-Motor and PASS Measures at Baseline and Each Follow-Up Assessment in Both Groups Treated With IVT.
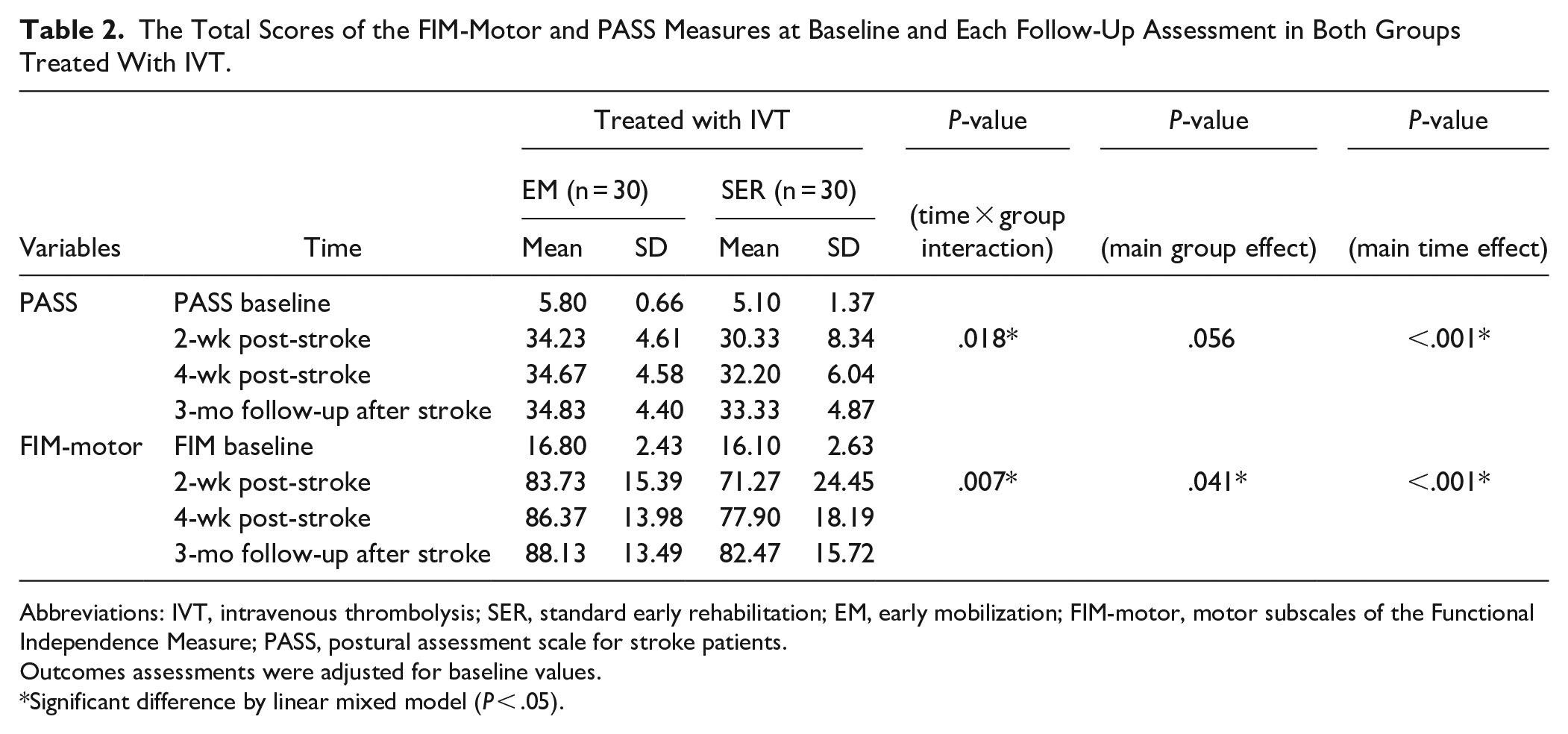
Abbreviations: IVT, intravenous thrombolysis; SER, standard early rehabilitation; EM, early mobilization; FIM-motor, motor subscales of the Functional Independence Measure; PASS, postural assessment scale for stroke patients.
Outcomes assessments were adjusted for baseline values.
Significant difference by linear mixed model (P < .05).
Table 3 shows the total PASS scores and total FIM-motor scores in the 2 groups that underwent MT at baseline and 2 weeks, 4 weeks, and 3 months after stroke. After controlling for initial outcome value, no significant group-by-time interaction was observed in the PASS and FIM-motor scores. No significant group effect was observed for PASS and FIM-motor scores at all evaluation times; however, the PASS and FIM-motor scores improved significantly over time (Table 3).
The Total Scores of the FIM-Motor and PASS Measures at Baseline and Each Follow-Up Assessment in Both Groups Treated With MT.
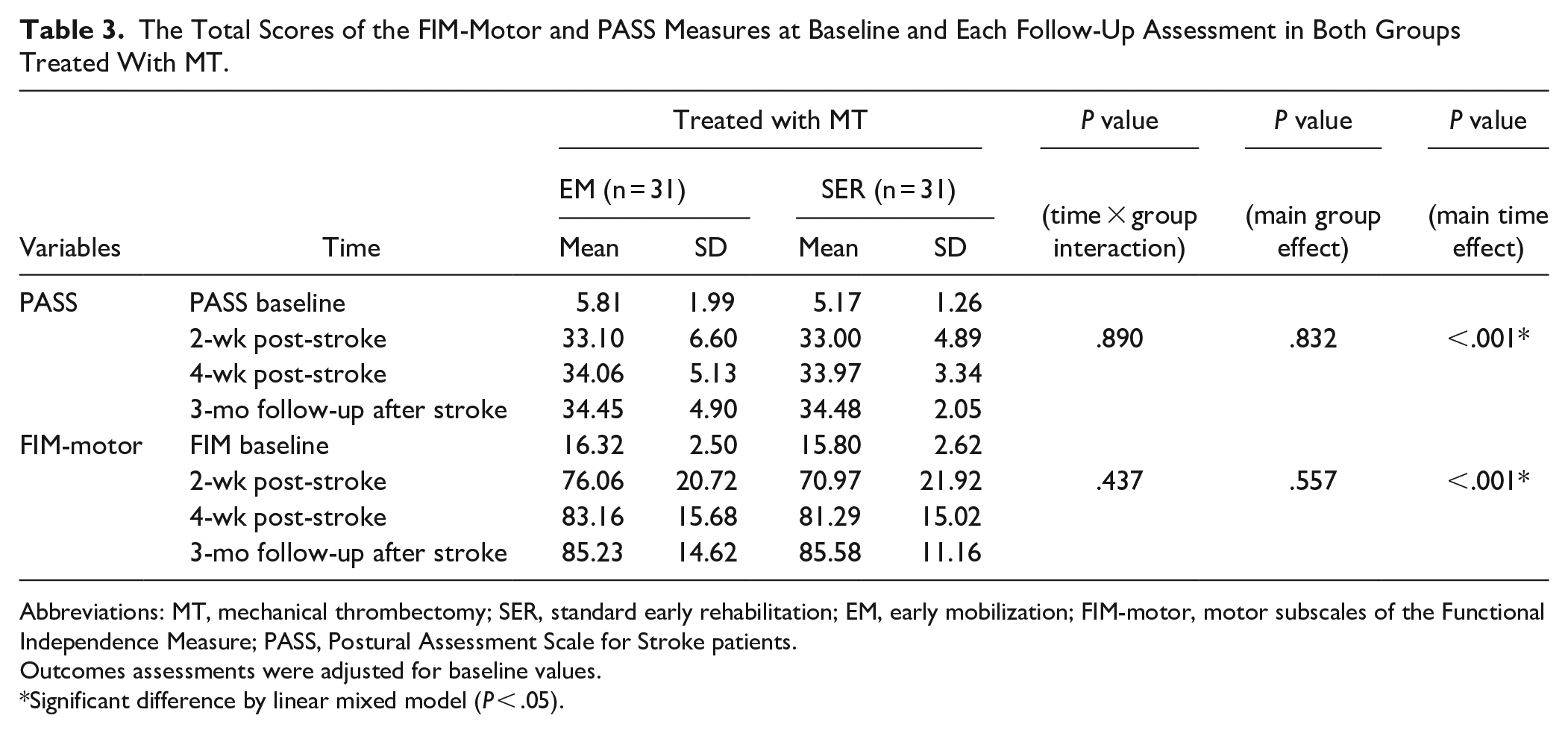
Abbreviations: MT, mechanical thrombectomy; SER, standard early rehabilitation; EM, early mobilization; FIM-motor, motor subscales of the Functional Independence Measure; PASS, Postural Assessment Scale for Stroke patients.
Outcomes assessments were adjusted for baseline values.
Significant difference by linear mixed model (P < .05).
Figure 2 shows the percentage of patients in each group following IVT (A) and MT (B) who achieved the FAC 0 to 5 level 2 weeks after stroke onset. The percentage of patients who achieved the FAC level in the EM groups was significantly higher than in the SER groups following IVT (χ2 = 14.75, P = .011) or MV (χ2 = 11.09, P = .05). However, there were no significant differences in length of stay at the stroke center between the EM and SER groups for either the IVT or MV cohorts (87.7 hours (SD = 43.3) vs 92.5 hours (SD = 41.5 ); 95.3 hours (SD = 52.1) vs 101.5 hours (SD = 43.76), respectively).
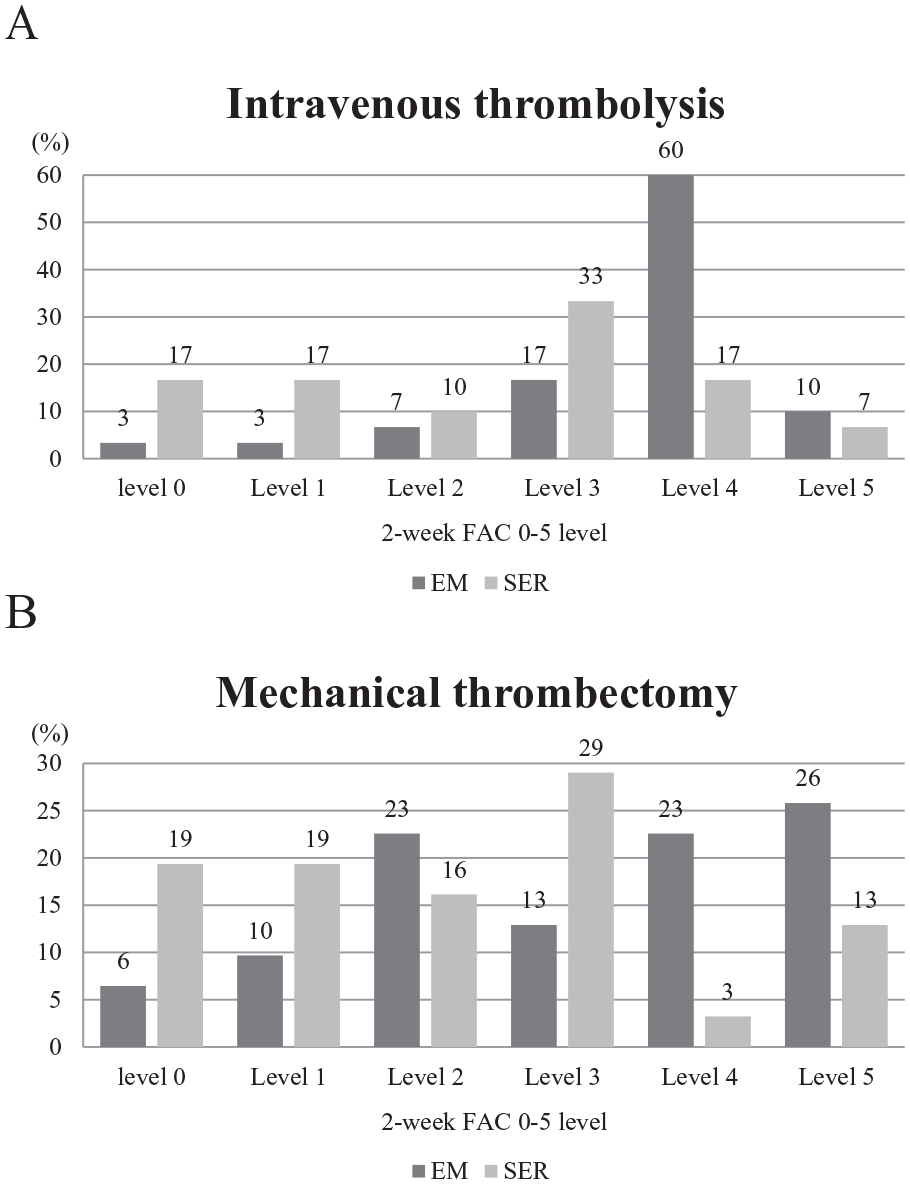
The percentage of each level of the functional ambulation category (FAC) at 2 weeks after stroke in standard early rehabilitation (SER) and early mobilization (EM) groups following intravenous thrombolysis (A) or mechanical thrombectomy (B)
Discussion
In our study, we observed significant differences in NIHSS scores at admission between the MT and IVT groups, highlighting the distinct nature of these 2 stroke treatment therapies. MT, involving direct physical removal of clots, presents different treatment conditions and criteria compared to IVT. The therapeutic window for IVT, as per local clinical guidelines, typically ranges from 3 to 4.5 hours after stroke onset. In contrast, MT offers a broader window of 6 to 24 hours, extendable based on clinical judgment and patient-specific factors.4,50 IVT is primarily aimed at AIS patients without specific contraindications, such as active bleeding or recent intracranial surgery, 50 whereas MT is indicated mainly for acute strokes from large vessel occlusion. These variances in treatment approaches and eligibility criteria may explain the differences in patient characteristics observed within our study cohort. The results suggested that the EM group had better FIM-motor scores within 3 months of AIS than the SER group in the cohort of patients who underwent IVT with rtPA. However, our results demonstrated that the EM versus SER protocol produced similar PASS and FIM-motor scores within 3 months of AIS for the patients who underwent MT.
A large-scale randomized controlled trial (AVERT) that focused on patients who received rtPA could not demonstrate the superiority of very EM compared with usual care. 39 The AVERT study initiated very EM within 24 hours of stroke onset, but the sample size was not large enough to detect a difference in the subgroup analysis. Another Japanese study on a hospital-based cohort of 6153 AIS patients treated with IVT reported that initiating early rehabilitation within 3 days of admission was associated with a significant increase in the proportion of patients who achieved functional independence. 35 However, in the Japanese study, the early rehabilitation group was defined as those who received any type or intensity of rehabilitation, and it provided no detailed information regarding the rehabilitation programs used. Another Yen’s study concluded that EM may be associated with an acceleration of functional improvement assessed by Barthel Index 4 weeks after stroke onset in AIS patients who underwent IVT with rtPA. 36 However, the study was a retrospective correlation study, no long-term follow-up was performed, and all patients had experienced a middle cerebral artery infarction stroke. 36
Only 1 study determined the feasibility of early rehabilitation implemented with out-of-bed activities in AIS patients who had undergone MT. 38 However, the primary outcome was mortality rate, and the results showed that EM was not better in decreasing mortality. Although the results of this previous study suggested that significant differences in the rate of mild dependence or independence regarding Barthel Index at the 3-month and 1-year follow-ups were different between the EM and conventional rehabilitation groups, the EM group had received 5- to 10-minute out-of-bed mobilization therapy with 4 sessions per day (high-frequency intervention design) for 4 days a week until discharge. 38 The frequency of intervention was quite different from our study; therefore, direct comparison with our results was difficult.
Our study indicated that starting EM training within 24 to 72 hours is superior to >72 hours after the onset of AIS in patients who underwent IVT with rtPA. In this cohort, the FIM-motor score at 2 and 4 weeks after stroke, as well as FAC performance 2 weeks after stroke onset, was significantly better in the EM group compared to the SER group. A previous study reported that early exercise after ischemic stroke improved the early survival of brain cells by increasing cerebral blood flow, resulting in reduced ischemic necrosis of neurons in the penumbra zone. 51 Sensory impulses, such as adding verticalization of sitting or standing activities, may be repeatedly introduced into the central nervous system, which facilitates axon sprouting of neurons, transmission of latent pathways, and synapses. 51 Therefore, EM may prevent brain cell injury and misuse and may promote the reconstruction of normal motion. Our results suggest that early initiation of out-of-bed mobilization treatment is more conducive to producing favorable functional outcomes within 1 month after stroke following IVT with rtPA. The EM group did not perform better than the SER group in FIM-motor at the 3-month follow-up and in posture stability measures. Most patients who received IVT with rtPA were categorized as having mild stroke (NIHSS 0-6) after 24 hours of onset in our study. 52 The recovery of postural control in the EM group was comparable to that in the SER group, possibly due to the ceiling effect of the assessment for mild stroke. 53
Controversially, in the case of patients who underwent MT in our study, while the average FIM-motor score in the EM group was higher than in the SER group, the between-group difference was not significant. After MT, patients showed an improvement of approximately 8 to 9 points in NIHSS scores at 24 hours. Most patients in both EM and SER groups achieved low NIHSS scores of around 3 to 4 after MT, indicating substantial amelioration of global neurological and motor impairment. The goal of thrombectomy is to achieve both reperfusion and recanalization 54 ; recanalization refers to the degree of arterial patency or clot burden, 55 while reperfusion refers to the restoration of blood flow in the downstream territory of the symptomatic artery and restoration of blood flow at tissue level. The modified Treatment in Cerebral Ischemia (mTICI) is the preferred measure of angiographic reperfusion, with grade 0 indicating no perfusion and grade 3 representing full perfusion of all visualized distal branches. 56 Previous studies found that patients showing large improvements in the NIHSS (87% of patients in the endovascular treatment and greater than 30% improvement) were more likely to exhibit favorable outcomes after 90 days, whereas those showing minimal improvement or no improvement were more likely to have unfavorable outcomes. 57 Another study showed that 83.3% of participants who underwent MT with mTICI 3 reperfusion achieved a favorable outcome (defined as a modified Rankin Scale score of 0 to 2 at 3 months); however, a favorable outcome was achieved by 43% of participants after IVT, with complete recanalization of the occlusion with distal flow.55,58 EM can enhance brain cell survival by improving cerebral blood flow and reducing penumbra, but the impact of mobility-induced cerebral blood flow is less significant compared to near-total reperfusion after MT. 59 In our study, patients who underwent MT achieved 95% reperfusion and substantial NIHSS improvement within 24 hours after AIS, which possibly explain the lack of significant differences in posture control and functional outcomes between the EM and SER groups. However, our study did not include information on instrumental ADL (IADLs), which assess cognitive abilities and patients’ capacity for independent living, financial management, and transportation. 60 Evaluation of IADLs could provide valuable insights into cognitive changes. Long-term assessment of IADLs should be considered in future research.
Limitations
This study had some limitations. First, the sample size of the study was relatively small, which might have increased the risk of type II error and might have been insufficient to detect small differences. In this study, a priori sample size estimation was performed based on similar previous clinical studies, and used a significance threshold of .05 and statistical power of 80%. However, it is important to note that this study is the first randomized controlled trial conducted on this topic, which may impose certain limitations on sample size estimation. In addition, the subjects were recruited from a single center; however, we analyzed a homogeneous group of patients admitted to a stroke center. Second, we excluded patients older than 85 years of age. Older age is associated with an increased severity of weakness and impairment and more negative outcomes. This exclusion may have introduced selection bias. Moreover, it should be acknowledged that patients in both the SER group and the EM group in this study might not have achieved the desired or optimal treatment effects from either IVT with rtPA or MT administration, which could have influenced the outcomes. Finally, the post-stroke outcome assessments selected do indeed have their advantages, disadvantages, and limitations. In the study, while the FIM scores offer vital information about patients’ functional independence in daily living, their interpretation may be affected by the extent of the patients’ use of wheelchairs, assistive devices, and other tools. This implies that observed improvements in FIM scores could partially reflect the use of these assistive tools, rather than an exclusive enhancement in physical functioning. Therefore, future studies should perform other prognostic assessments of long-term outcomes and incorporate relevant objective measurement instruments to fully characterize the impact of implementing EM for AIS patients who have undergone IVT with rtPA or MT.
Conclusion
An early out-of-bed mobilization protocol that combined standard established interventions with 30 minutes of rehabilitation intervention per day was effective in improving the functional ability recovery of AIS patients who had undergone IVT within 1 month after stroke. Further investigations are required to add advanced functional parameters to understand the effect of EM on AIS patients who have undergone MT.
Footnotes
Author Contributions
Hsiao-Ching Yen: Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Supervision; Validation; Writing—original draft; and Writing—review & editing. Guan-Shuo Pan Data curation; Investigation; Methodology; Validation; Writing—original draft; and Writing—review & editing. Jiann-Shing Jeng Conceptualization; Funding acquisition; Project administration; Resources; Software; Supervision; and Writing—review & editing. Wen-Shiang Chen Conceptualization; Project administration; Software; Supervision; and Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National Taiwan University Hospital (NTUH: 109-004497; 112-O0018). We also thank staff at the NTUH stroke center (4D1) for supporting this study.
