Abstract
Background1
Despite a growing interest in gaming rehabilitation for upper limb (UL) recovery post-stroke, studies investigating the effects of game-based rehabilitation incorporating functional games are lacking.
Objective
To investigate the efficacy of an intensive, functional, gamified rehabilitation program compared to task-based training on UL motor function in acute/sub-acute stroke survivors.
Methods
This randomized, multicenter, single-blind, clinical trial comprises 120 participants with unilateral stroke who were randomized to receive either gamified training (n = 64) using the ArmAble™ [experimental group (EG)] or task-based training (n = 56) in conjunction with conventional therapy for 2 hours per day, 6 days per week for 2 weeks, followed by UL rehabilitation for another 4 weeks at home. Primary outcomes evaluated by a blinded assessor included the Fugl-Meyer Assessment-Upper Extremity (FM-UE), and Action Research Arm Test (ARAT). Data were analyzed using a linear mixed-effect regression model.
Results
The mean (standard deviation) age of the participants was 54.4 ± 11.7 years (78.1% men) in the EG and 57.7 ± 10.9 years (73.2% men) in the comparator group (CG). The median (interquartile range) time since stroke was 30.0 (54.0) days in the EG and 22.5 (45.0) days in the CG. Following the 2-week intervention, a statistically significant improvement was observed in the EG for the FM-UE [between-group mean differences (95% confidence interval): −3.9 (−6.5, −1.3);
Conclusion
Gamified rehabilitation using the ArmAble™ device has shown immediate and short-term improvement in UL function after acute/sub-acute stroke.
Clinical Trials Registry Number:
CTRI/2020/09/027651
Keywords
Introduction
Upper limb (UL) motor impairment occurs in 50% to 80% of people with an acute stroke and 40% to 50% of those with a chronic stroke, 1 out of which only 5% to 34% eventually gain full UL function. 2 As time progresses, patients’ inability to use the affected arm can contribute to anxiety, poor self-perceived quality of life, and decreased societal participation. 3 Therefore, to improve a person’s capacity post-stroke for engaging in regular everyday activities, UL rehabilitation is crucial across the stroke continuum of care and is of utmost significance.
In order to enhance neurological recovery through neuroplasticity and thus improve UL motor recovery, task-based, intense, and repetitive training is essential.4,5 Traditional conventional therapies have several shortcomings such as lack of active participation from participants, 3 monotonous nature of practices, 6 increased treatment cost, and unavailability of rehabilitation specialists.6,7 Game-based rehabilitation has been suggested as a novel therapeutic approach to overcome these shortcomings and to supplement existing treatment regimens. 8 Game-based rehabilitation delivers intensive training, allows interaction, provides real time feedback, and cues to participants to improve their UL function following stroke.2,9 Incorporating gaming elements into rehabilitation can promote motivation, engagement, active participation, satisfaction, and thereby foster the adherence of participants to the intervention. 10 These features of the game-based rehabilitation program may allow individuals to devote extra time carrying out UL movements for the chosen tasks and mitigate issues related to boredom, frustration, or adherence.11,12 This help participants to achieve a sizeable number of repetitions that are necessary for neuro-plastic changes as well as motor adaptation to take place with rehabilitation.9,13
The use of virtual reality (VR)-based gaming interventions was highlighted in the guidelines provided by the American Heart Association and the American Stroke Association with class IIa evidence on improving UL function following stroke. 14 Moreover, recent meta-analyses have demonstrated significant improvements (with a moderate or large effect size) in UL motor function (the Fugl-Meyer Assessment-Upper Extremity [FM-UE]) following VR-based gaming interventions compared to conventional therapies in individuals recovering from sub-acute stroke. However, no such improvements were evident for UL activity when assessed with the Action Research Arm Test (ARAT), Box and Block Test, Wolf Motor Function Test, and Barthel Index following VR-based gaming interventions in this population.11,15,16
Post-stroke rehabilitation should be commenced as early as possible after the injury, preferably within the first 6-months when plasticity is known to be heightened.17,18 A few small-scale studies have substantiated the positive effect of game-based rehabilitation on enhancing UL motor function after acute/sub-acute stroke.19,20 However, studies with a larger sample size often employed commercial gaming systems or non-functional games were used, which challenges the ecological validity of games used for rehabilitation purposes in the acute/sub-acute phase of post-stroke recovery.21-24 Commercial games and gaming devices may be a suitable option as they are extensively available and come at a reasonable cost. 25 However, it should be taken into consideration that commercial games are stereotypically designed for healthy individuals and thus may not meet the requirements of patients with neuromusculoskeletal impairments. 2 To better meet patient’s diverse needs and support therapeutic goals, dedicated game-based programs, which provide functional games emulating activities of daily living, while allowing therapists to provide tailor-made therapeutic components (feedback and difficulty level) centered around each patient’s ability, are required.26,27
An innovative, custom-built, interactive arm rehabilitation device called the “ArmAble™” (BeAble Health Pvt Ltd., IIT Hyderabad, Telangana, India) has been recently developed, which provides intensive, repetitive, functional task-based gaming therapy while incorporating motor learning and neuroplasticity principles.28,29 The ArmAble™ is a portable “Plug & Play” device consisting of a computer monitor, a gaming software, and a platform with a game console. The game console allows the participants to move their arm in the anteroposterior and/or mediolateral directions while playing interactive functional games.29,30 In this device, the difficulty levels (easy, medium, and hard) of games are chosen prior to the commencement of the intervention based on the motor capacity of participants.
The ArmAble™ device can be expended from the very early stage of recovery post-stroke, even if there is no movement possible in the affected UL, because the interface or gaming console allows bimanual training. There is also an option of attaching various types of handles to the gaming console based on the requirements of each game. The games installed in the device have been specifically aimed at rehabilitating the UL and are functionally relevant, mimicking daily activities, along with being progressively challenging, thereby promoting adherence. The ArmAble™ device enables one to execute the closed kinetic chain form of exercises, which might be comparatively simpler in the initial stages post-stroke for avoiding synergistic movement patterns. The device also allows therapists to supervise participants’ movement performance while playing games in real-time (knowledge of performance) as well as at the end of each gaming session (knowledge of result). All participants will perceive the knowledge of performance and results from the therapist’s and device’s feedback.
This study is the first randomized clinical trial investigating the effectiveness of game-based rehabilitation using the ArmAble™ device. To establish the effectiveness of an intervention, it is imperative that, we compare novel treatment techniques with a dose and context-matched treatment group as this is a major determinant of rehabilitation outcome. 31 In fact, task-based training is superior to usual care for improving UL motor outcomes by providing functional, task-based training using motor learning and motor control principles such as intensive training, intermittent feedback, and variable practice.32,33 Thus, the aim of this study was to investigate the effectiveness of an intensive, functional, gamified UL rehabilitation program compared to a task-based training in improving UL motor function and activity, performance of activities of daily living, and the quality of life in people with acute/sub-acute stroke.
Methods
The EnteRtain trial was a multicenter, single-blinded, randomized clinical study with a 1:1 participant allocation ratio. After receiving an approval from the institutional ethics committee, Kasturba Medical College and Kasturba Hospital (IEC-328/2020), the trial was registered in the Clinical Trials Registry of India (CTRI/2020/09/027651). The trial was carried out in accordance with a previously published protocol. 28
Participants
Participants were recruited from the physical therapy departments of 4 stroke rehabilitation centers located across India. Individuals diagnosed with acute/sub-acute unilateral stroke and admitted to the participating rehabilitation center were screened for eligibility by the research staff at each center. Individuals aged 18 to 80 years, of either sex, who are hemodynamically stable, those with a Brunnstrom Motor Recovery Stage (BMRS) in the range of ≥1 to ≤ 5, a stroke severity score >6 on the National Institute of Health Stroke Scale (NIHSS), a Montreal Cognitive Assessment score ≥26 points, and a score ≤3 in modified Ashworth Scale in the affected UL muscles were eligible to participate in the trial. Individuals were excluded if they had global or receptive aphasia, complex regional pain syndrome, major medical issues or co-morbidities that prevented them from being a part of the study, vestibular dysfunction and a functional vision, and/or auditory loss that limit computer use. Before enrolling, a written informed consent was obtained from all participants, and all procedures followed the Helsinki declaration. 34
Sample Size
Sample size was obtained using the comparison of mean formula. According to the previous studies, we expected a minimal clinically important difference (MCID) of 10 points in the FM-UE total score23,35 and 12 points in the ARAT total score36,37 between the 2 groups. Sample size has been computed with an estimated effect size (Φ
Randomization and Masking
One of the investigators located at the primary site (Manipal) administered the centralized block randomization (20 permuted blocks of 6 participants) through a computer-generated random numbers table. The group allocation was concealed with the help of Sequentially Numbered Opaque Sealed Envelopes method. Recruited participants were stratified based on age and stroke severity. Trained research staff blinded for the allocation of research participants conducted all the assessments in all the centers.
Intervention
Participants randomized to the experimental group (EG) received a gamified UL training program using the ArmAble™ device. The participants were provided with both functional games (eg, delicious dosa, strike the mosquito, and champion cleaner) and fun-based/commercial games (eg, bouncing the ball and fresh fruit catcher). The console of the device keeps a control on these games via movements of the shoulder, elbow, forearm, and hand. Progression made by the therapist based on the motor capability of participants included shifting bilateral handle to unilateral handle, adding resistance either by tilting the table vertically (anti-gravity movements) and/or attaching a resistance band to the console. Game parameters adjusted for the progression included object intervals, object speed, and object dispersion to the left and right (easy/medium/hard levels). Participants assigned to the comparator group (CG) received an UL task-based training that included daily function tasks (eg, lifting up the UL and positioning it on the table, pushing a chair with the hand, opening up the fingers to grasp a cup and navigating it toward the mouth). Each participant completed at least 3 tasks 100 times per session (for a total of 300 repetitions/session). Both groups received the interventions for 45 to 60 minutes per day, 6 days per week for 2 weeks under the supervision of a trained therapist. More information about the device, EG/CG intervention, and the details of progression are available in the “Template for Intervention Description and Replication checklist” table included in our protocol and in the preliminary study.28,29 The games and tasks were chosen based on the interests, context, and motor capacity of the participants. In addition to the EG/CG intervention, all participants received the same conventional therapy in all 4 participating centers. Conventional therapy consisted of facilitatory/inhibitory techniques, strength training, passive and active limb mobilization, balance, and ambulatory training for at least 45 to 60 minutes per day.
Following the 2 weeks intervention period, participants in both groups received a functional UL rehabilitation program to complete at their home without the supervision of a therapist. The UL program was for 30 minutes per day for 6 days per week for 4 weeks and there was no difference in either content or amount of training between the groups. The purpose of this home training was to maintain and retain the effects of game-based UL rehabilitation, therefore, no active game-based training was received by the EG during this period. All participants were given a logbook to keep track of their therapy sessions at home during this follow-up period. Participants were contacted by phone, once a week, to ensure that they were adhering to the therapy program at home.
Outcome Measures
Participants were assessed at 3 time points: at the beginning of the study, second week (immediately after the intervention), and sixth week (follow-up assessment). The study outcomes such as the FM-UE and motricity index (MI)-arm component falling under the International Classification of Functioning, Disability, and Health (ICF) body structure and function domain, the ARAT and modified Rankin Scale (mRS) under the ICF activity domain, and the Stroke Impact Scale—version 3.0 (SIS-V 3.0) under the ICF participation domain were assessed.38-40 The primary outcomes were UL function and activity measured with the FM-UE (maximum score: 66) and ARAT (maximum score: 57). The secondary outcome measures included the MI-arm component for assessing UL strength, the mRS to assess the degree of disability, and the SIS-V 3.0 to assess the impact of the consequences of stroke on different domains of health and life. Furthermore, at the termination of the 2 weeks intervention, game user experience was assessed using the game user experience satisfaction scale (GUESS) 41 only for participants in the EG. All outcome assessments were completed by a blinded assessor (a qualified physiotherapist) except for the SIS-V 3.0 and GUESS which were self-reported in English/local language by the participants themselves. Throughout the study, any adverse events were documented.
Statistical Analysis
R-studio was used to perform statistical analysis. We performed data entry twice to reduce transcription errors. Data skewness was determined using the
Results
Participant recruitment commenced from October 2020 and was carried out up to December 2022 until reaching required samples for the study. The follow-up period of this clinical trial ended in January 2023. A total of 910 participants were screened on admission to the respective rehabilitation centers. Of those, 765 participants did not fulfill the desired eligibility criteria and 25 refused to participate in the trial. A total of 120 participants were included in the present study, and 115 completed the 2-week intervention. Seventeen participants missed the follow-up assessment at the end of 6 weeks. However, we have imputed the missing values using the LOCF method. Therefore, 120 participants (EG: 64; CG: 56) were included in the final analysis (Figure 1).
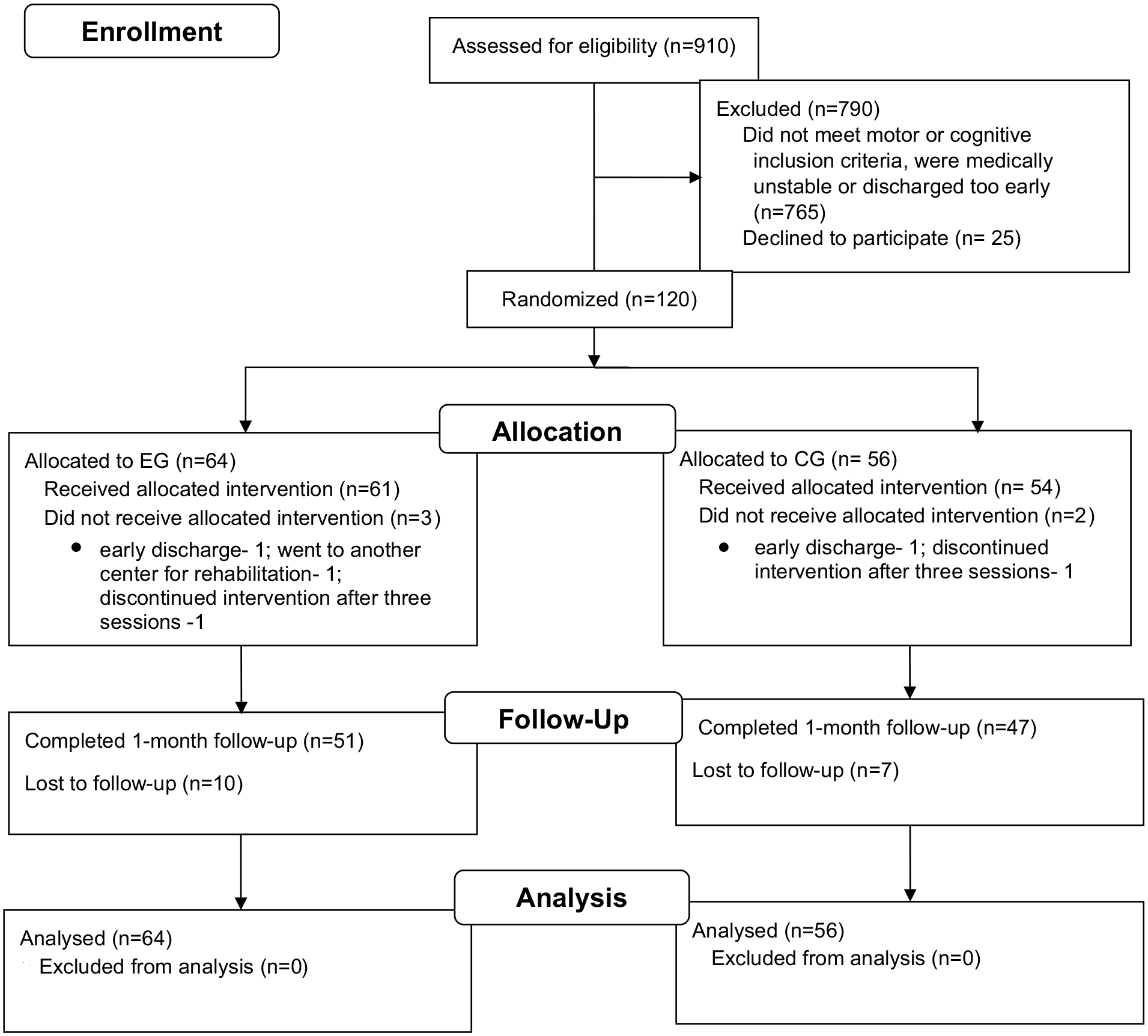
CONSORT flowchart of participants through the trial.
The mean age of the participants was 54.4 ± 11.7 years in the EG (n = 64, 78.1% men) and 57.7 ± 10.9 years in the CG (n = 56, 73.2% men). The EG and CG groups were comparable at baseline except for BMRS-UL (
Demographics and Clinical Characteristics of the Study Participants at Baseline (n = 120).
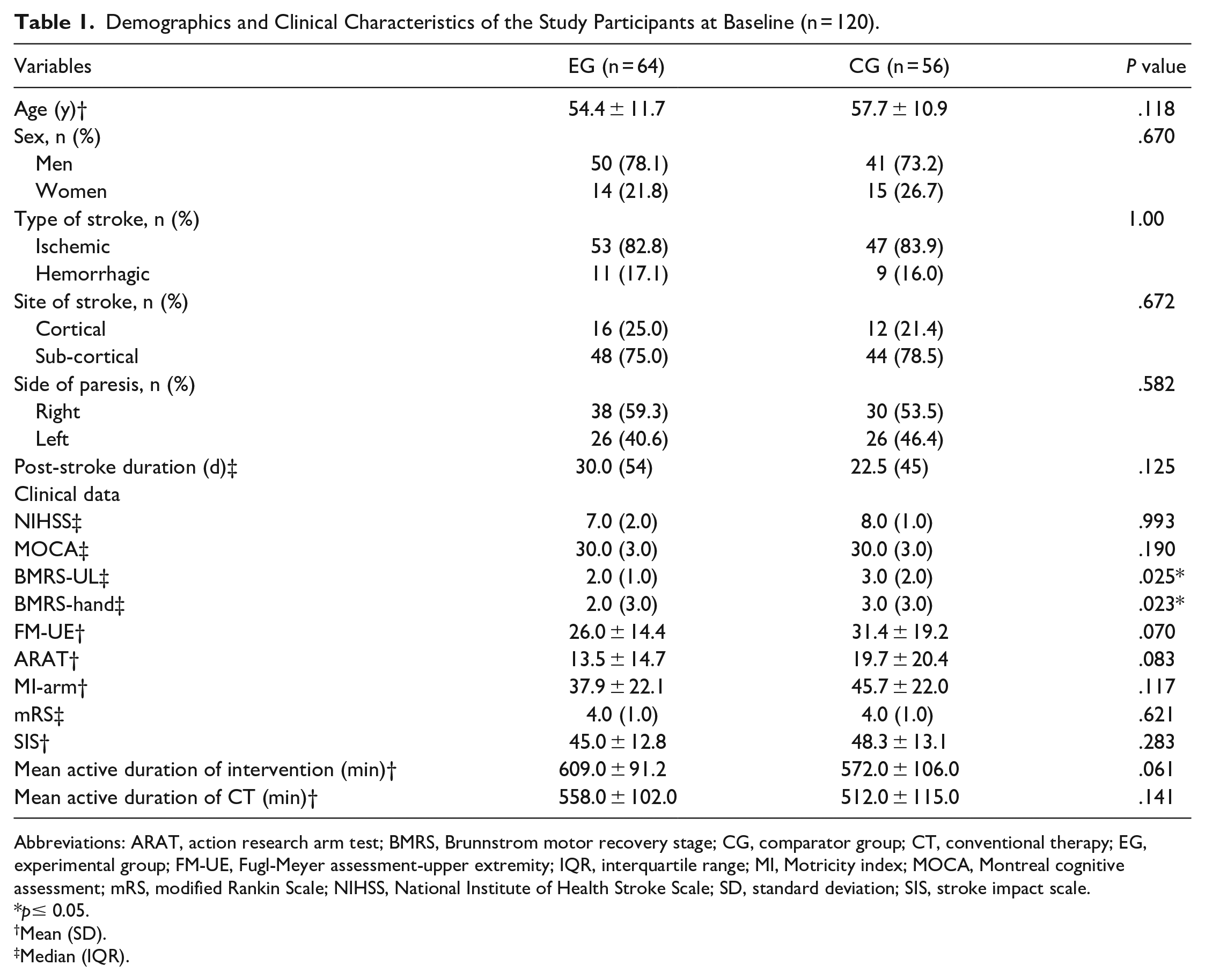
Abbreviations: ARAT, action research arm test; BMRS, Brunnstrom motor recovery stage; CG, comparator group; CT, conventional therapy; EG, experimental group; FM-UE, Fugl-Meyer assessment-upper extremity; IQR, interquartile range; MI, Motricity index; MOCA, Montreal cognitive assessment; mRS, modified Rankin Scale; NIHSS, National Institute of Health Stroke Scale; SD, standard deviation; SIS, stroke impact scale.
Mean (SD).
Median (IQR).
The active duration of intervention was 609.0 ± 91.2 minutes in the EG and 572.0 ± 106.0 minutes in the CG. None of the participants reported any serious adverse events. However, some participants reported fatigue (visual analog fatigue scale score >7; EG = 6; CG = 5); shoulder pain (EG = 3; CG = 1); dizziness (EG = 2); and mild back pain (EG = 2) during and/or after the trial period. The observed adherence to exercise training at home during the 4-week period was 89.1% in the EG and 85.8% in the CG.
Effects of Game-Based Rehabilitation on UL Function, Activity, and Participation Domain
Table 2 displays the outcomes of the linear mixed model analysis and displays measured mean values at 3 different time intervals. At 2 weeks post-intervention, a statistically significant improvement was observed for EG in UL function measured with the FM-UE [between-group mean differences (95% CI): −3.9 (−6.5, −1.3); Cohen’s
Gains in Upper Limb Function, Activity, and Participation Outcomes at 2- and 6-wk Post-intervention in the EG and CG (n = 120).
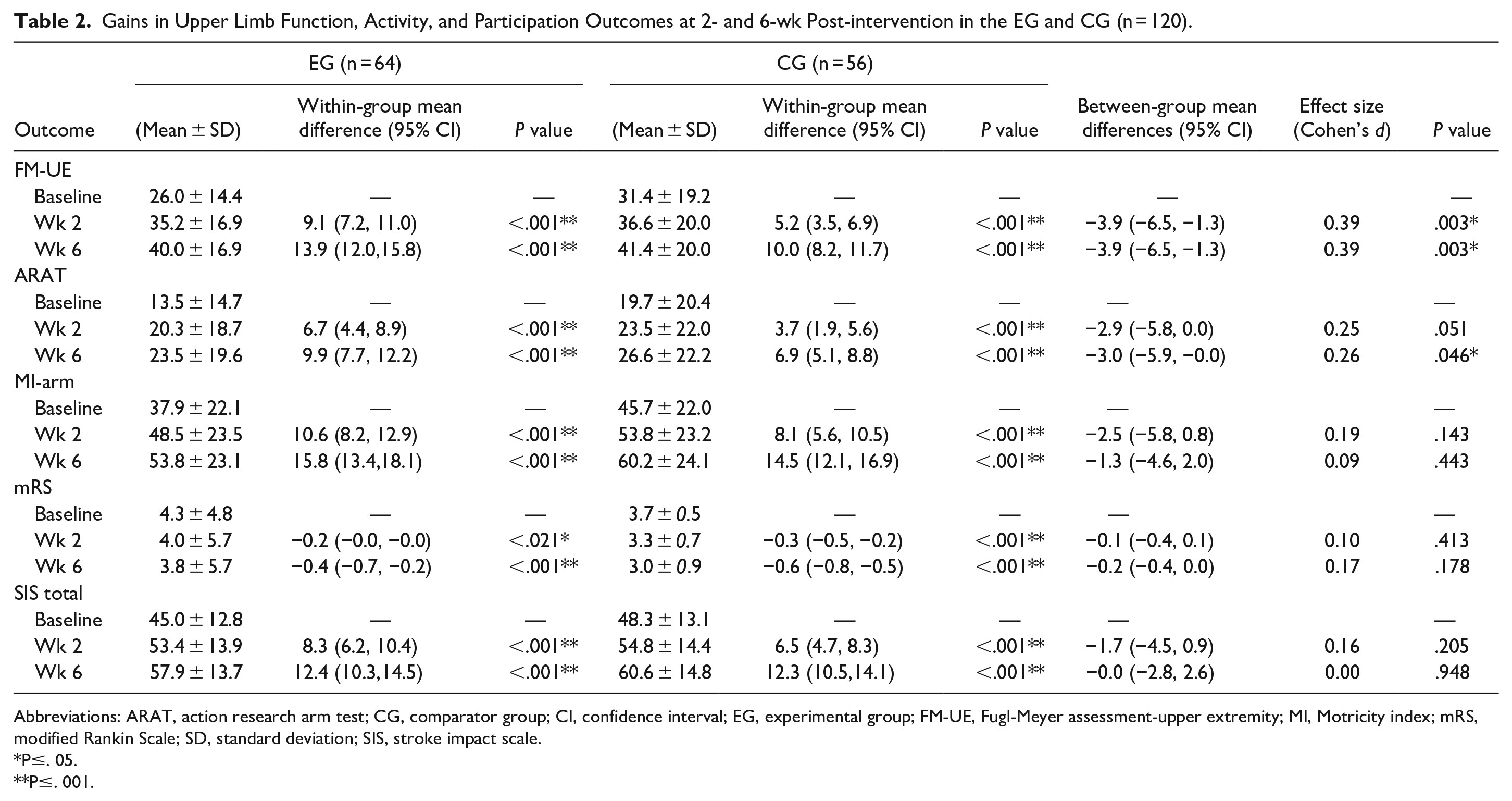
Abbreviations: ARAT, action research arm test; CG, comparator group; CI, confidence interval; EG, experimental group; FM-UE, Fugl-Meyer assessment-upper extremity; MI, Motricity index; mRS, modified Rankin Scale; SD, standard deviation; SIS, stroke impact scale.
P≤. 05.
P≤. 001.
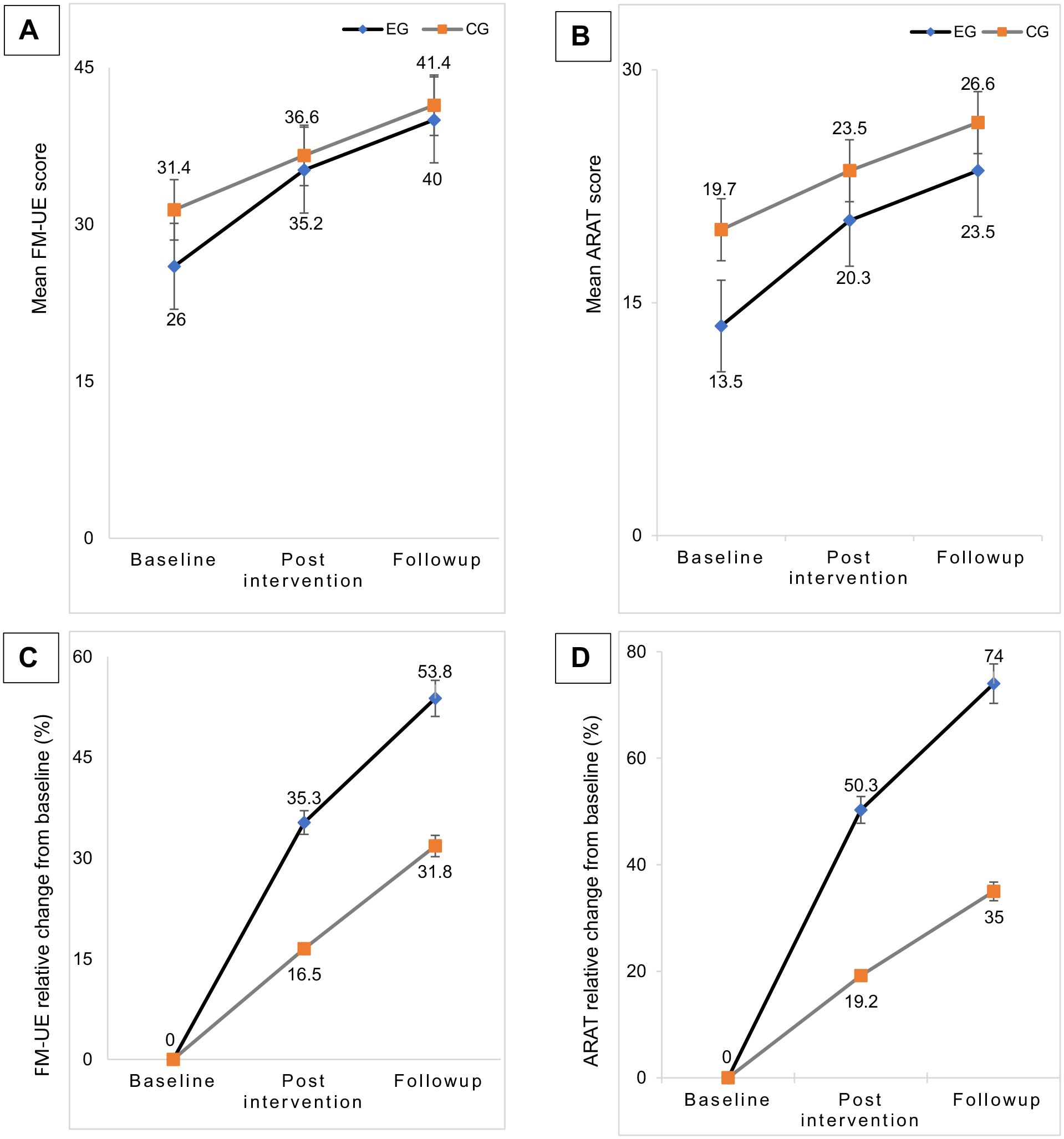
Mean UL motor function and activity (A and B) and percentage change in UL motor function and activity (C and D) from baseline to post intervention and follow-up as measured with FM-UE and ARAT, respectively.
Sub-Group Analysis Based on the UL Severity Level
The sub-group analysis based on the severity level of UL paresis revealed a significant between-group difference for FM-UE and ARAT scores in participants with mild-to-moderate UL paresis (Table 3). In the EG, FM-UE scores improved 15.1% after intervention and 20.6% at follow-up from baseline. Similarly, ARAT scores also improved 19.6% after intervention and 24.9% at follow-up from the baseline score. On the other hand, there was no significant improvement in participants with severe UL paresis.
Subgroup Analysis of Participants With Mild-to-Moderate and Severe UL Paresis (n = 120).
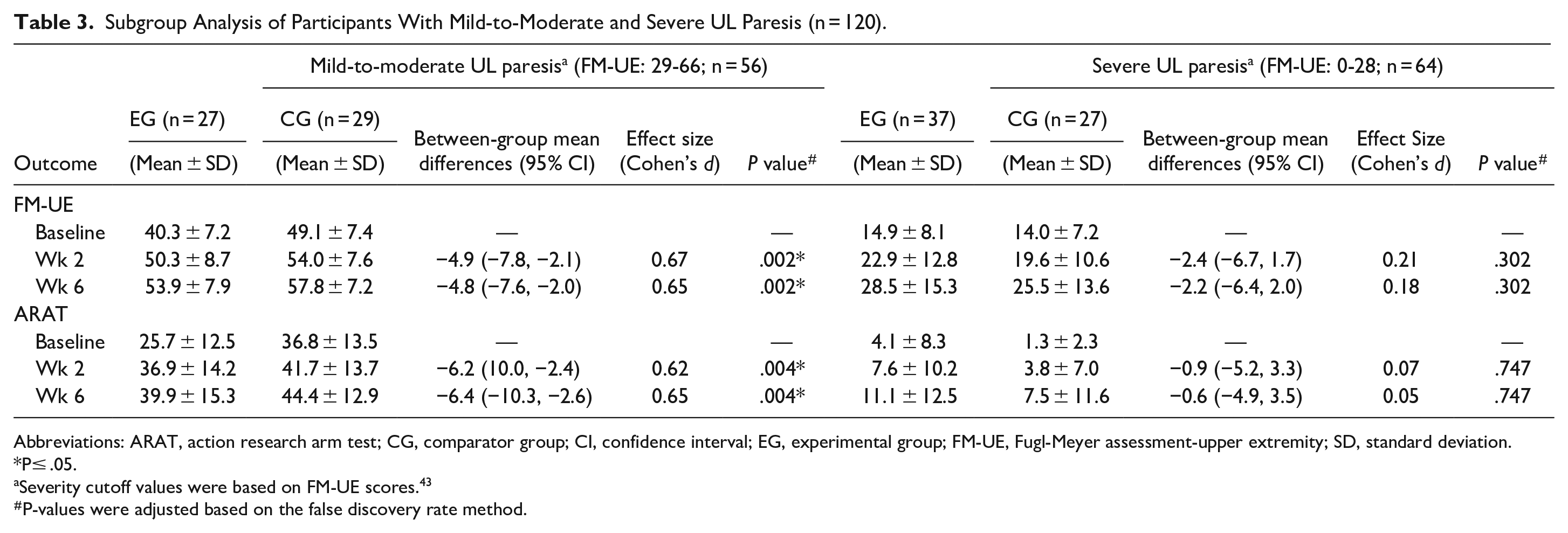
Abbreviations: ARAT, action research arm test; CG, comparator group; CI, confidence interval; EG, experimental group; FM-UE, Fugl-Meyer assessment-upper extremity; SD, standard deviation. *P≤ .05.
Severity cutoff values were based on FM-UE scores. 43
P-values were adjusted based on the false discovery rate method.
Game User Experience Satisfaction
All participants responded positively to all scoring items of the GUESS scale. The mean composite score was 54.6 ± 2.9 out of a maximum score of 63, which showed a high level of participants’ satisfaction following the rehabilitation program. When the subscales were examined, the major contributors to a high satisfaction level were enjoyment, usability, visual esthetics, and personal gratification with the games and device (Figure 3). Our participants reported that the ArmAble™ system was easy to use, and they enjoyed well-developed characters and visual graphics of the games available on the device. Aside from this, the participants felt that they were able to achieve their goals and thereby improve skills through active participation. They perceived the games as creative, enjoyable, and meaningful, felt successful when they overcame obstacles/challenges, and were constantly motivated to advance to the next level.
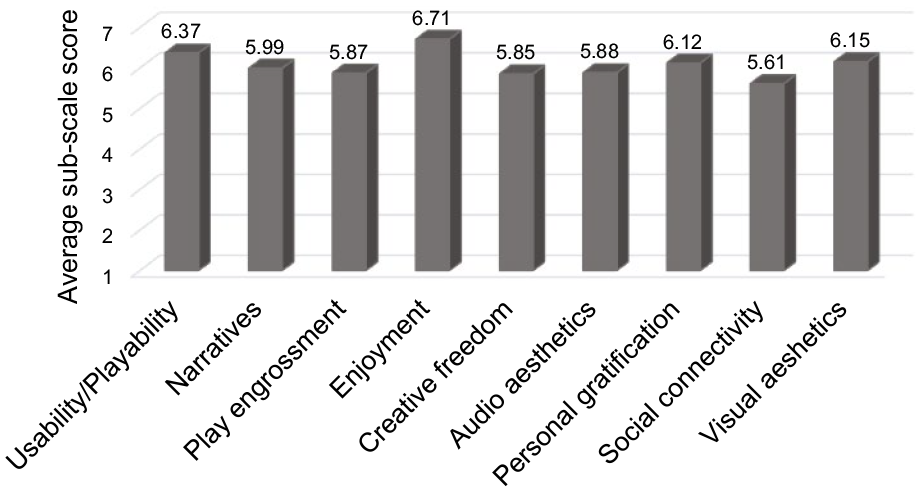
Game user experience satisfaction sub-scale scores of participants in the experimental group (n = 61).
Discussion
The EnteRtain trial is a randomized, multicenter, single-blind clinical trial designed to compare the effectiveness of a gamified rehabilitation program using a customized arm rehabilitation device, the ArmAble™ with conventional therapy for people with stroke in the acute/sub-acute phases of recovery. In our study, the CG received task-based training, which is a highly customized, client-centric, functional task-based intervention incorporating motor learning and motor control principles. Participants in the EG significantly improved their UL function immediately after the gaming intervention (at the second week) with an effect size of 0.39 and maintained the effects at the sixth week with an effect size of 0.39. Despite the small effect size, participants in the EG improved their motor functions from baseline by 9.1 (13.8%) and 13.9 (21.0%) on average on the FM-UE scale at the second week and sixth week respectively, which is greater than the established MCID of 9 (13.6%). 35 However, the improvement found in the UL activity (ARAT) with a small effect size of 0.25 and 0.26 at the second week and sixth week assessment, respectively, was not more than the reported MCID value of 12 (21.0%). 36 Since there was a lower baseline value for the EG compared to the CG for certain variables (Tables 2 and 3), the effect sizes reported for between-group comparisons may be interpreted with caution.
The improvements noted in the primary outcomes (FM-UE and ARAT) can be explained by several reasons. In our trial, participants in the EG were trained using a system with games designed specifically for UL rehabilitation that were functional (mimicking the activities of daily living), and the training was intensive and repetitive. In the ArmAble™ device, the challenge or difficulty level of each game were altered to better suit the motor skills of the participants. Based on responses to the GUESS questionnaire, 41 participants experienced a sense of mastery and active participation in the functional games which they perceived as meaningful, purposeful, goal-oriented, and yet to be within their ability. Additionally, positive rehabilitation outcomes in our study could be due to the increased excitement of participants throughout the experimental intervention, which contributes to increased motivation. 44 Consequently, participants were comparatively less focused on their physical impairments and were more focused on their gaming experience. Game-based rehabilitation has the potential to stimulate both supervised and reinforcement learning, owing to the augmented visual, acoustic, and sensory feedback, 45 which are concurrently available in the ArmAble™ device. The participants were getting both knowledge of performance (in terms of rewards, individual game time, change in the difficulty level, and game speed) and knowledge of results (total distance covered, total game score, total game time, graphical representation of movement trajectory) from both the device and the therapist, since it was a supervised rehabilitation program.
Since the ArmAble™ device in our study permits the use of bilateral handles in a closed kinetic chain pattern, experimental intervention for participants with severe UL impairment was made possible with a limited stereotyped synergistic movement pattern and with minimal assistance from a therapist. Furthermore, in the EG, there was a need for interaction with the objects appearing on the computer screen, which necessitated the use of an input or interface devices. Most games primarily required gross motor skills to grasp and navigate the console for interaction, which may have contributed to increased UL function following the gamified intervention. Usage of specially designed handles as per the requirement of the context-specific games (eg, mug, plate, spherical, and pinch grip handles) would have activated distal extensors and pinch muscles to control movement in the game space during the training. The effect was not seen immediately after the intervention for the activity outcome (ARAT). However, the exercise training at home had tasks similar to the context of functional games, and patients might have practiced concurrently the same tasks at home. This may have contributed to improvement in the activity domain at the end of 6 weeks. Overall, these findings are consistent with preceding reports that VR-based gaming approaches improve UL function after stroke, as measured by common UL instruments such as the FM-UE and ARAT.31,46-48 A recent meta-analysis also reported that when compared to conventional therapy, VR-based gaming intervention resulted in enhanced UL motor function following sub-acute stroke. 49 However, this finding was deemed insignificant after trim and fill adjustment of risk of bias. However, the study suggested that VR-based gaming intervention might be used in conjunction with conventional therapy to restore UL motor function. Customizing game-based rehabilitation systems and adjusting them according to patients’ needs could enhance results of rehabilitation by employing rehabilitation principles (eg, supportive feedback, adjusting for task difficulty, task-based training, and the use of the affected limb). 50
It is possible that game-based rehabilitation given at the initial stages of stroke recovery would have improved UL function, which is in accordance with findings on neuroplasticity-induced motor recovery. 48 According to a post-hoc analysis of a study by Laffont et al, 23 early UL video game-based rehabilitation may be more efficacious if commenced within the first 30 days post-stroke. In our study, the median (interquartile range) post-stroke duration for EG was 30.0 (54.0) days. As brain plasticity is critical during the first month after stroke, 51 early training done in the proper way and at the right amount will help recovery when used during this period. 52
The changes reported in the sub-group analysis (based on the severity level of UL paresis) were higher than the reported MCID values for the FM-UE and ARAT,35,36 except for the post intervention ARAT scores in people with mild-moderate stroke. Given that more than half of our participants suffered from severe UL impairment (53.0% based on the FM-UE score <28), and greater limitation in UL activity (mean baseline ARAT score of 7.2 ± 12.7), 12 sessions of gaming intervention over a comparatively short period of 2 weeks might be considered insufficient to elicit significant changes in UL activity, UL strength, disability level, and the impact of consequences of stroke on various domains of health and life post intervention when compared to an active control group. Furthermore, it has been reported that functional recovery for people with severe UL paresis is poorer than for those with mild to moderate UL paresis.53,54 Previous trials involving people with severe UL paresis did not show promising results for VR-based gaming therapy.21,23 We hypothesized that a high dose administered over a prolonged period may generate a distinctive result on these outcomes. Another possible explanation could be the complexities of treating UL dysfunction especially in people with severe UL paresis. Following stroke, there may be changes in muscle synergies due to disrupted motor pathways, sensory impairments, tonal abnormalities, and altered neural reorganization, which lead to stereotyped voluntary movements, making rehabilitation difficult for regaining performance in daily activities. 45
People with stroke in the EG reported a high level of satisfaction with the game-based rehabilitation program provided through the ArmAble™ device incorporating functional games with ecological validity. The high level of satisfaction with the games reported by our participants could be due to the following factors: 1. the positive feedback provided by the therapist and device, 2. incorporation of functional games (which emulate the activities of daily life), 3. the motivating and challenging environment (easy, medium, and hard), 4. the selection of games based on the individual motor capacity of participants which helps them achieve their goals, and 5. a rich sensory experience (with visual, auditory, and tactile feedback). Previous studies have reported that game-based rehabilitation leads to a high level of satisfaction and motivation, thereby increasing adherence to the therapy.20,55-58 However, these studies have used a 5-point or 7-point Likert scale to assess the level of satisfaction or enjoyment. In our study, we have used the GUESS scale, which is a valid and reliable scale that has multiple components to assess user satisfaction and experience. Game-based training with the ArmAble™ allows active participation of individuals with stroke in therapy, which leads to a high number of movement repetitions and thereby promotes motor learning and augments UL recovery.56,58
Our participants (mean age of 55.7 ± 11.5 years) were relatively younger as compared to those included in a systematic review on VR-based gaming interventions. 59 This might have improved the extent of acceptance of the study intervention, as evidenced by less drop-outs (n = 3) from the EG. Some of our participants complained about shoulder pain due to increased usage of the hemiparetic side, and fatigue resulting from intensive training during the intervention sessions. These findings are consistent with the previous studies that used wearable hand sensors for training games in spaces without support surfaces (open kinetic chain exercise).60,61 Even so, despite fatigue and/or UL pain, none of our participants dropped out of the training sessions, and their attendance rate for these sessions was also high.
Strengths and Limitations
The strengths of this study include the high rate of adherence coupled with the allocated interventions that were harmonized for dosage, the engagement of an active CG, and the use of outcome measures that are both reliable and valid, across all domains of the ICF. Based on the FM-UE scores, 53.0% of the participants in our study had severe, 19.1% had moderate, and 27.8% had mild UL impairment. As a result, our findings broaden the applicability of game-based rehabilitation with the ArmAble™ to patients with a wide range of motor impairments. The relatively large sample size and multicenter nature of the study would proliferate the generalizability of the study findings to various rehabilitation set-ups in low- and low-middle income countries.
Our intervention could be considered short, reflecting upon the current actuality of brief hospital stays due to planned early discharges and lack of insurance coverage for some participants in our setting. It is reported that the median length of hospital stay following acute stroke is 7.3 (2.8, 23.1) days globally 62 and 10 to 13 days in India 63 including our hospitals and clinical setups. A thorough sensory assessment was not done to evaluate the sensory status of the participant, even though, as per NIHSS scoring, only 2 participants had severe to total sensory loss and 6 participants had mild-to-moderate sensory loss. We have not assessed the daily hand use and it may not be possible to ascertain the improvement in UL motor function with the improvement in FM-UE alone. In addition, the duration of their home exercise program during the follow-up period was self-reported during our telephone follow-up.
Future Recommendations
Our study findings have implications for stroke rehabilitation. Given that the majority of strokes occur in low- and middle-income countries with limited resources and access to technology as well as rehabilitation therapists, 64 simple, low-cost, task-based, high-intensity, game-based therapies may be considered in optimizing motor recovery. 65 Furthermore, remote game-based telerehabilitation strategies for patients at home using the low-cost gaming device ArmAble™, could fulfill the needs of patients in rural or remote sites and reduce travel burden and costs, merits further investigation. The cost-effectiveness of this model of rehabilitation delivery is worth investigating. Future research should assess the effectiveness of this particular intervention with the chronic stroke population and also include outcomes related to daily hand use, cognitive, and sensory function since these aspects will affect the rehabilitation outcomes.
Conclusion
Our results showed statistically and clinically significant improvement for UL function (FM-UE) immediately after the 2-week intervention program and statistically significant improvement for UL function (FM-UE) and activity (ARAT) during the follow-up assessment at 6 weeks. People with stroke reported a high level of satisfaction with the game-based rehabilitation program provided through the ArmAble™ device incorporating functional games with ecological validity. Thus, from the initial phase of stroke recovery, game-based training with the ArmAble™ device might be utilized to provide an intensive, repetitive therapy for enhancing UL motor function.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683231222921 – Supplemental material for Effectiveness of an Intensive, Functional, and Gamified Rehabilitation Program on Upper Limb Function in People With Stroke (EnteRtain): A Multicenter Randomized Clinical Trial
Supplemental material, sj-docx-1-nnr-10.1177_15459683231222921 for Effectiveness of an Intensive, Functional, and Gamified Rehabilitation Program on Upper Limb Function in People With Stroke (EnteRtain): A Multicenter Randomized Clinical Trial by A. Sulfikar Ali, D. Senthil Kumaran, Amritha Unni, Sanjukta Sardesai, Vasudeva Prabhu, Punitha Nirmal, Aparna R. Pai, Vasudeva Guddattu and Ashokan Arumugam in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We would like to thank the people who took part in our study, and acknowledge Mr. Viral Shah, Mr. Habib Ali, and Mr. Sreehari K G for their assistance with patient recruitment and technical assistance.
Author Contributions
A Sulfikar Ali: Formal analysis; Investigation; Methodology; Writing—original draft. D. Senthil Kumaran: Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Supervision; Writing—review & editing. Amritha Unni: Investigation; Writing—review & editing. Sanjukta Sardesai: Formal analysis; Writing—review & editing. Vasudeva Prabhu: Methodology; Project administration; Resources; Writing—review & editing. Punitha Nirmal: Methodology; Project administration; Resources; Writing—review & editing. Aparna Pai: Methodology; Resources; Writing—review & editing. Vasudeva Guddattu: Formal analysis; Software; Writing—review & editing. Ashokan Arumugam: Conceptualization; Formal analysis; Investigation; Methodology; Supervision; Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by BeAble Health Pvt Ltd., IIT Hyderabad, Telangana, India; and the Society of Indian Physiotherapists [grant number: SIP_2021_YR01]. However, the funders played no part in the development, planning, execution, data analysis, interpretation, or publication of this study’s conclusions.
Supplementary material for this article is available on the
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
