Abstract
Background
Moderate-severe traumatic brain injury (TBI) has been associated with progressive cognitive decline in the chronic injury stages in a small number of studies.
Objective
This study aimed to (i) replicate our previous findings of decline from 1 to 3+ years post-injury in a larger, non-overlapping sample and (ii) extend these findings by examining the proportion of decliners in 2 earlier time windows, and by investigating novel predictors of decline.
Methods
N = 48 patients with moderate-severe TBI underwent neuropsychological assessment at 2, 5, 12 months, and 30+ months post-injury. We employed the Reliable Change Index (RCI) to evaluate decline, stability and improvement across time and logistic regression to identify predictors of decline (demographic/cognitive reserve; injury-related).
Results
The proportions of patients showing decline were: 12.5% (2-5 months post-injury), 17% (5-12 months post-injury), and 27% (12-30+ months post-injury). Measures of verbal retrieval were most sensitive to decline. Of the predictors, only left progressive hippocampal volume loss from 5 to 12 months post-injury significantly predicted cognitive decline from 12 to 30+ months post-injury.
Conclusions
Identical to our previous study, 27% of patients declined from 12 to 30+ months post-injury. Additionally, we found that the further from injury, the greater the proportion of patients declining. Importantly, earlier progressive hippocampal volume loss predicted later cognitive decline. Taken together, the findings highlight the need for ongoing research and treatment that target these deleterious mechanisms affecting patients in the chronic stages of moderate-severe TBI.
Introduction
Moderate to severe traumatic brain injury (TBI) is associated with significant impairments to cognitive domains, including attention, speed of processing, memory, and executive functioning. These impairments disrupt work, school, and social activities.1,2 For many, these impairments are enduring, causing life-long disability, which is in turn associated with a heavy social and economic burden. On the other hand, others may experience only minimal ongoing impairments.3-6 Prognosticating how an individual will fare in the chronic stages of injury is needed to optimize treatment and clinical management; however, our ability to do so is very limited, despite extensive research into TBI recovery and predictors of outcome.2,7,8 The current paper examines patterns and predictors of cognitive recovery in moderate-severe TBI and focuses on treatable causes of poor outcomes. The current study was designed to replicate and extend our prior research on long-term cognitive recovery in moderate-severe TBI. 9
The majority of longitudinal studies of cognitive recovery in moderate-severe TBI have found that recovery is asymptotic, with a steep early period of improvement.10-13 followed by a gradual plateauing at a level that is less than that of pre-injury functioning. 11 These studies typically examine patients at the group-level; however, group-based studies can mask more granular features of recovery trajectories. Indeed, across studies that have been conducted at the individual-level, a more complicated picture of recovery emerges, where for some individuals, cognition worsens in the months and years following injury.5,14-18 Such findings are consistent with the growing evidence that moderate-severe TBI is a progressive and neurodegenerative disorder.9,19,20–27
In our 2008 study, 16 we prospectively investigated long term cognitive changes from 12 to 30+ months post injury in 33 individuals with moderate and severe TBI. To evaluate clinically significant changes at the individual level, we used the Reliable Change Index (RCI). 28 We found that while group level changes over time were negligible, a subgroup of 27% of participants showed a significant decline (using RCI) on at least 2 subtests of a 12-test neuropsychological battery post-injury.
These findings are consistent with those from other groups.5,29 For example, Millis et al 5 examined neuropsychological test performance in a cohort of complicated mild to severe TBI patients at 1 and 5 years post-injury; they found that while most individuals (63%) showed no change in functioning or improved (22%), 15% of the sample declined on 2 or more tests of their 15 test battery. Himanen et al 14 examined cognitive changes in a TBI cohort tested over a period of 30 years. They found a significant decrease in a global index of cognitive function in 56% of patients, controlling for aging-related decline, with improvement in 23% and no change in 21%. More recently, Sigurdartdottir et al 29 examined 79 patients across multiple time points on memory and executive function tests, including 1 to 10 years following moderate-severe TBI (N = 48 in this subset). In the 1 to 10 year window, 11% of patients declined on memory (but not executive functioning).
As a result of considerable methodological variations across the handful of studies that have been conducted at the individual level—with differences in test batteries, times post-injury, and predictors—the proportion of patients showing declines, the relevant risk factors, and the timing of declines remains uncertain. Moreover, the question whether neurodegeneration is predictive of cognitive decline has yet to be examined (but see Farbota et al 23 who found that performance on cognitive tests at 2 months post-injury was predictive of subsequent volume loss in several associated structures).
Our first objective was to look at patterns of change across time, both by patient and by task. In objective 1a, we were interested in replicating our prior study to provide more conclusive evidence of the proportion of decliners across the 12 to 30+ month post-injury time window. We were also interested in whether there were variations in the number of decliners as a function of time post injury. Thus, in a non-overlapping sample to that used in our previous research, we examined change in cognitive function across 3 time windows: 2 to 5 months, 5 to 12 months, and 12 to 30+ months post-injury (as in Till et al 16 ). We developed a framework for categorizing patients as either “declined,” “stable,” or “improved,” and compared the proportion of patients falling into these categories over each period. For the purposes of this study, we operationalized the categories in a very specific and narrow sense, as opposed to global or general functional decline, for example, decline was defined as worse performance on 2 or more (of 12) tests; see Methods section for detailed explanation. We predicted that the proportion of patients demonstrating decline from 12 to 30+ months post-injury would be comparable to that observed in Till et al, 16 (ie, 27%) given the similarity in methods (ie, same clinical setting, same timelines, same approach to classification of decline) used with this non-overlapping sample. Since the period of most rapid recovery occurs in the first months of injury, 11 and Himanen et al 14 found much greater incidence of decline compared to patients in other studies when they examined out to 30 years post-injury, we predicted there would be an increasing proportion of patients declining from the early to the later windows. In objective 1b, we explored whether any cognitive domains are more vulnerable to decline than others by profiling the patterns of change—decline, stability, and improvement—for each cognitive test in the battery.
The second objective was to comprehensively examine predictors of cognitive decline. We examined variables found to be predictive in prior studies of individual recovery trajectories, including age5,14 and out-patient therapy hours16,30 along with other cognitive reserve and injury-related variables known to influence TBI cognitive outcomes.31-33 Given evidence of neurodegeneration in the chronic stages of TBI from our own research and that of others, 26 we extended previous research by examining whether reductions in brain volume would predict cognitive decline. In our previous study, we found that tests of verbal retrieval showed the most decline from 12 to 30+ months post-injury. 16 Therefore, we predicted that hippocampal volume loss, measured from 5 to 12 months post-injury, would be predictive of subsequent cognitive decline from 12 to 30+ months post-injury.
Methods
Participants
The current sample comprised N = 48 participants with moderate to severe TBI (defined as having a Glasgow Coma Scale (GCS) score of 13 or less and/or a length of post-traumatic amnesia of 1 hour or more) who were part of a larger, prospective, longitudinal study of recovery (the Toronto Rehab TBI Recovery Study 34 ) and had been referred to the in-patient Neurorehabilitation Program at the Toronto Rehabilitation Institute. Participants in the larger study underwent neuropsychological testing at 2, 5, and 12 months post-injury. As described in Till et al, 16 the participants met the following inclusion criteria: age 17 years and older, positive computed tomography or magnetic resonance imaging (MRI) findings or evidence of a moderate to severe TBI, proficiency in English as judged by the treating speech language pathologist and/or psychometrist, and functional use of 1 or both upper extremities. Individuals were excluded from the study if they had sustained their TBI secondary to another neurologic event (eg, a fall caused by a stroke), had a history of psychotic disorder, had another neurologic disorder or systemic disorder known to affect cognitive functioning or recovery (eg, Lupus, Korsakoff encephalopathy, sleep apnea), were known to be actively engaged in alcohol or substance abuse at time of testing, or failed the test of symptom validity at any of the assessments. An additional inclusion criterion of the current study was that participants had to be enrolled in the Toronto Rehab TBI Recovery Study after the enrollment window was closed for the Till et al study in order to create a non-overlapping sample for replication. At the time of this investigation, 69 participants were eligible for inclusion. Of these, 3 were deceased, 9 could not be reached, 8 declined to participate due to lack of interest, and 1 participant recently had a neuropsychological assessment in the community. The new sample therefore comprised 48 participants corresponding to 69.6% of the 69 eligible participants. The study sample and the eligible patients who did not participate in the study (“attrition group”) did not differ on any relevant demographic or injury variables, with 1 exception: the attrition group had a higher socioeconomic status (SES) that was approaching significance (P = .08) as compared to the study sample group.
Injury and demographic characteristics are presented in Table 1 for the Till et al 16 sample (n = 33) and the current study sample (n = 48). The two samples were similar overall, but the current sample was marginally older, t(79) = −1.942, P = .056, and had more years of education, t(78) = −3.112, p = .003 and a higher pre-morbid IQ, t(59) = −2.184, P = .033. In addition, there was a significant difference between samples in the mean time post-injury for assessments 1 (t(79) = −2.714, P = .008), 2 (t(79) = −2.370, P = .021), and 4 (t(74) = 2.412, P = .021). There was no significant difference in the mean time post-injury for assessment 3. The mean time post-injury for the assessments in both studies were approximately 2 months, 5 months, 1 year, and 2.5 years post-injury (current study) and 3 years (previous study).
Demographic and Clinical Characteristics of Till et al 16 Versus Current Sample.
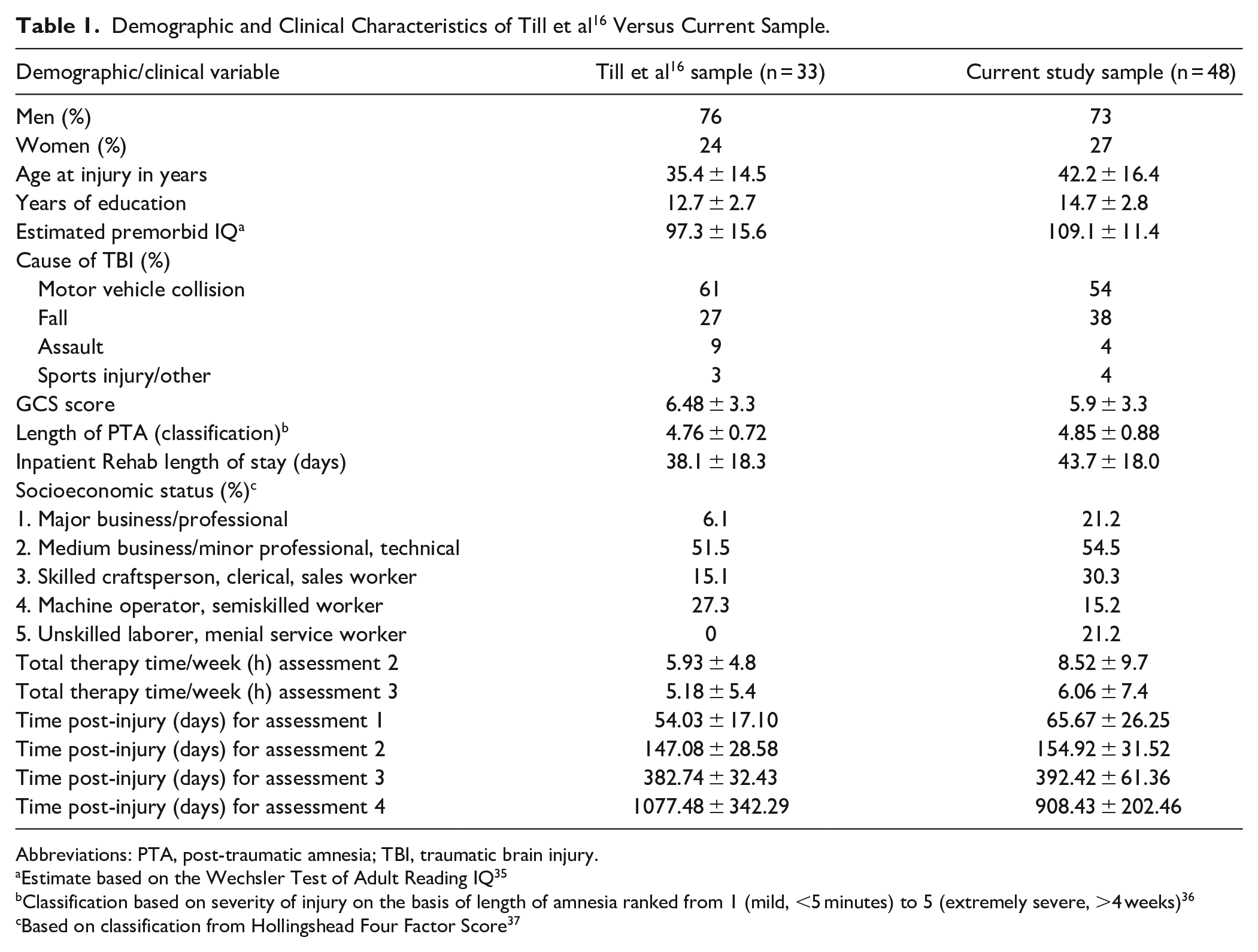
Abbreviations: PTA, post-traumatic amnesia; TBI, traumatic brain injury.
Estimate based on the Wechsler Test of Adult Reading IQ 35
Classification based on severity of injury on the basis of length of amnesia ranked from 1 (mild, <5 minutes) to 5 (extremely severe, >4 weeks) 36
Based on classification from Hollingshead Four Factor Score 37
Design and Procedures
The parent study employed a longitudinal design with prospective neuropsychological assessments at approximately 2, 5, 12, and 30+ months post-injury. Assessments were completed by a trained psychometrist and supervised by a Clinical Neuropsychologist. All participants received a clinical neuropsychological feedback and written report, contributing to the high retention rate of this sample in the parent study (77.8%). Prior to recruitment, each participant had already undergone 1 to 2 days of assessment that included comprehensive neuropsychological and mood testing with a trained psychometrist at 3 time points: 2 months, 5 months, and 12 months post-injury. For the current study, all participants received an additional neuropsychological assessment. While our eligibility criteria stated participants must be at least 24+ months post-injury, in our sample, the 4th assessment for all participants occurred at least 30+ months post-injury.
Objective 1a: We examined the proportion of patients showing cognitive decline, stability and improvement at 3 time windows: 2 to 5, 5 to 12, and 12 to 30+ months post-injury. As in Till et al, 16 we operationalized as a “decliner” a participant that declined on 2 or more neuropsychological tests using the RCI. “Improvers” were operationally defined as those who improved on 2 or more tests using the RCI (and did not decline on 2 or more of the remaining tests that comprised our cognitive battery). In the event that patients improved on 2+ cognitive domains, but also declined on 2+ others (4-8% of our patient sample), they were categorized as a “decliner.” Those patients not categorized as an improver or a decliner were classified as “stable.” Of note—Logical Memory I and Logical Memory II were not treated as separate tests, nor were the RAVLT total learning and delayed recall scores. Thus, if patients declined on both sub-tests for either Logical Memory or RAVLT, this was counted as a single test decline, so as to avoid overestimation of decline and provide a conservative estimate.
Objective 1b: For each of the 12 tasks in the battery, we examined the proportion of patients showing decline, stability, and improvement from 12 to 30+ months post-injury because the number of decliners was greatest at this time window. Here, we included all patients in the parent study, that is, the 33 patients from Till et al, 16 and the 48 patients from the current study in order to increase analytic stability.
Objective 2: To examine predictors of decline, we again examined patients from 12 to 30+ months post-injury only, and we again collapsed patients from our previous and current study to increase power and analytic stability.
Outcomes
Neuropsychological Test Battery
Please see Till et al 16 for a full description of the tests in the current study, which are identical to those of Till et al. 16 In brief, the twelve neuropsychological tests encompassed the domains of manual motor speed and dexterity, attention and speed of processing, verbal learning and memory, visual-spatial ability, and executive function. The test battery comprised: Block Design and Digit Span (including Digit Span Forwards and Digit Span Backwards) from the Wechsler Adult Intelligence Scale—3rd edition (WAIS-III) 38 ; and Wechsler Abbreviated Scales of Intelligence (WAS) 39 ; Symbol Digit Modalities Test (SDMT, oral only) 40 ; Trail Making Test (TMT)—Parts A and B41,42; Logical Memory I and II subtests (LM-I/II) from the Wechsler Memory Scale—3rd edition (WMS-III) 43 ; Rey Auditory Verbal Learning Test (RAVLT, total learning and delayed recall scores) 44 ; Controlled Oral Word Association Test (COWAT, phonemic fluency) 45 ; and Grooved Pegboard (dominant hand only).41,42
Hippocampal Volume Acquisition and Processing
As part of the larger study, participants underwent MRI scanning after completing an MRI screening protocol. All MRIs were acquired on a General Electric (GE) Signa Echospeed 1.5 Tesla HD scanner (SIGNA EXCITE, GE Healthcare, Milwaukee WI), using an 8 channel head coil. Sequences included sagittal T1 (TR (repetition time)/TE (echo time) = 300/13 ms), slice thickness = 5 mm, space 2.5 mm, matrix 256 × 128 axial gradient recalled echo TR/TE = 450/20, flip angle = 20◦, slice thickness = 3 mm no gap, matrix 256 × 192 axial fluid attenuated-inversion-recovery TR/TE = 9000/45 ms, TI (inversion time) = 2200 ms, slice thickness = 5 mm no gap, matrix 256 × 192 axial fast spin echo proton density/T2 TR/TE 5500/3090 ms, slice thickness = 3 mm no gap, matrix 256 × 192. All above mentioned sequences were obtained with a 22 cm field of view. For complete image acquisition, processing and analysis protocol, please refer to Green et al. 19
The hippocampi were manually outlined using Analyze TM 8.1 Brain Imaging Resource, Mayo Clinic, MN) by an experienced tracer from coronally orientated MR images in the anterior-posterior direction. Calculations of volumes were computed automatically by multiplying the number of voxels traced in each slice, by their depth (ie, slice thickness). The hippocampi were measured according to the method described by Watson et al46,47 (see Maller et al, 48 for a more detailed description of this procedure).
Total percent change in hippocampal volume from 5 months to 12 months post-injury was then computed for volumes of the total left (HPCLTOT) and total right (HPCRTOT) hippocampi taking into account baseline volumes. The formula for change in total right hippocampal volume is presented below.
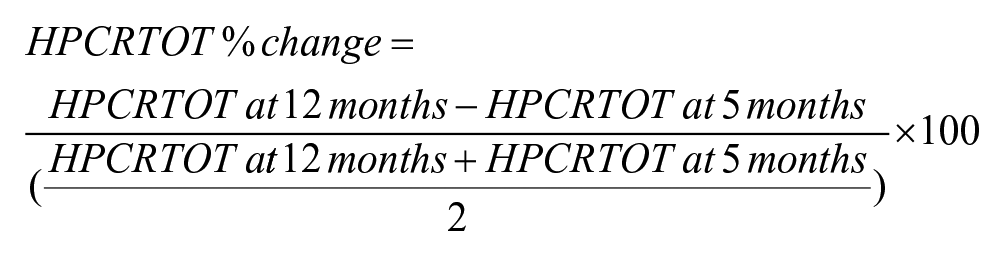
Statistical Analyses
Objective 1
Following the methods of Till et al, 16 we obtained test-retest reliability coefficients for the RCI calculations from Levine et al 49 for TMT (Parts A and B 41 ; Digit Span tests, 38 SDMT, 40 COWAT, 45 and Grooved Pegboard. 41 Reliability for RAVLT (Schmidt 44 ) was taken from Geffen et al 50 ; the WAIS-III 38 and WMS-III 43 manuals were used for the Block Design and Logical Memory subtests. Reliability coefficients ranged from 0.67 (Grooved Pegboard) to 0.82 (Block Design).
To conduct these analyses, normed scores for neuropsychological data were used to rule out potential confounds such as age, and to allow for direct comparison of groups.
The RCI allows for an understanding of change in individual subjects that is considered clinically meaningful, when sources of measurement error (including test–retest practice effects) are taken into consideration. 28
RCI = (x2 – x1) / Sdiff, where x1 represents the subject’s baseline score, x2 represents the subject’s follow-up, and Sdiff is the standard error of that difference. The Sdiff was computed from the standard error of measurement (Se), which is a function of the initial standard deviation of the measure and its reliability according to the following formula: Sdiff = √ [2(Se)2, where Se = s√ (1 − rxx). Reliable change is established when the difference between the follow-up and baseline scores exceeds the 90% confidence interval for the predicted score (ie, ±1.64 × Sdiff).
Objective 2
In preliminary testing of the models the following predictor variables were included: demographic/cognitive reserve variables including, sex, age, years of education, SES, 37 and estimated pre-morbid IQ on the Wechsler Test of Adult Reading35,51; severity of injury variables including, GCS, 52 and length of post-traumatic amnesia (LPTA 53 ); percent-change in left and right hippocampal volumes from 5 to 12 months post-injury (ie, hippocampal atrophy), 19 mean therapy hours at 5 and 12 months post-injury; and self-reported symptoms of depression from the Beck Depression Inventory, 54 and anxiety from the Beck Anxiety Inventory. 55 Only progressive hippocampal atrophy (left and right) 5 to 12 months post-injury was found to be significantly correlated with decliner status, and LPTA showed a trend.
Logistic regression analyses were conducted for Objective 2 in which we regressed decliner status (ie, decline vs no-decline) from 12 to 30+ months post-injury against progressive hippocampal atrophy (left and right) 5 to 12 months post-injury. The model controlled for LPTA because of the possibility of it being a confounding variable in the relationship between decliner status and atrophy. In addition, we regressed on left and right hippocampal atrophy from 5 to 12 months post-injury (controlling for LPTA) and we regressed on left hippocampal atrophy from 5 to 12 months post-injury (controlling for LPTA and right hippocampal atrophy from 5 to 12 months post-injury (and ipso facto vice versa).
IBM SPSS statistical package (Version 25, 2019, were used for all t-test comparisons and R 56 was used for the Objective 2 logistic regression analyses.
Results
Objective 1. Pattern of Cognitive Decline Across Time by Patient and by Neurocognitive Task
Figure 1 presents the incidence at each time window of those who declined, improved or remained stable. Grey lines represent movement of participants to different categories across time. At each time window, the majority of patients either remain stable or improve. Nonetheless, overall, 58.3% patients were classified as having declined in at least 1 time window. The proportion of patients classified as a decliner during 1 window-only was 29.2%. The proportion of patients classified as a decliner at 2 time windows was 12.5% (N = 5), with 4 of these 5 patients showing decline from 2 to 5 months and from 12 to 30+ months post-injury rather than at contiguous time windows. Only 1 patient showed decline at all 3 time windows.
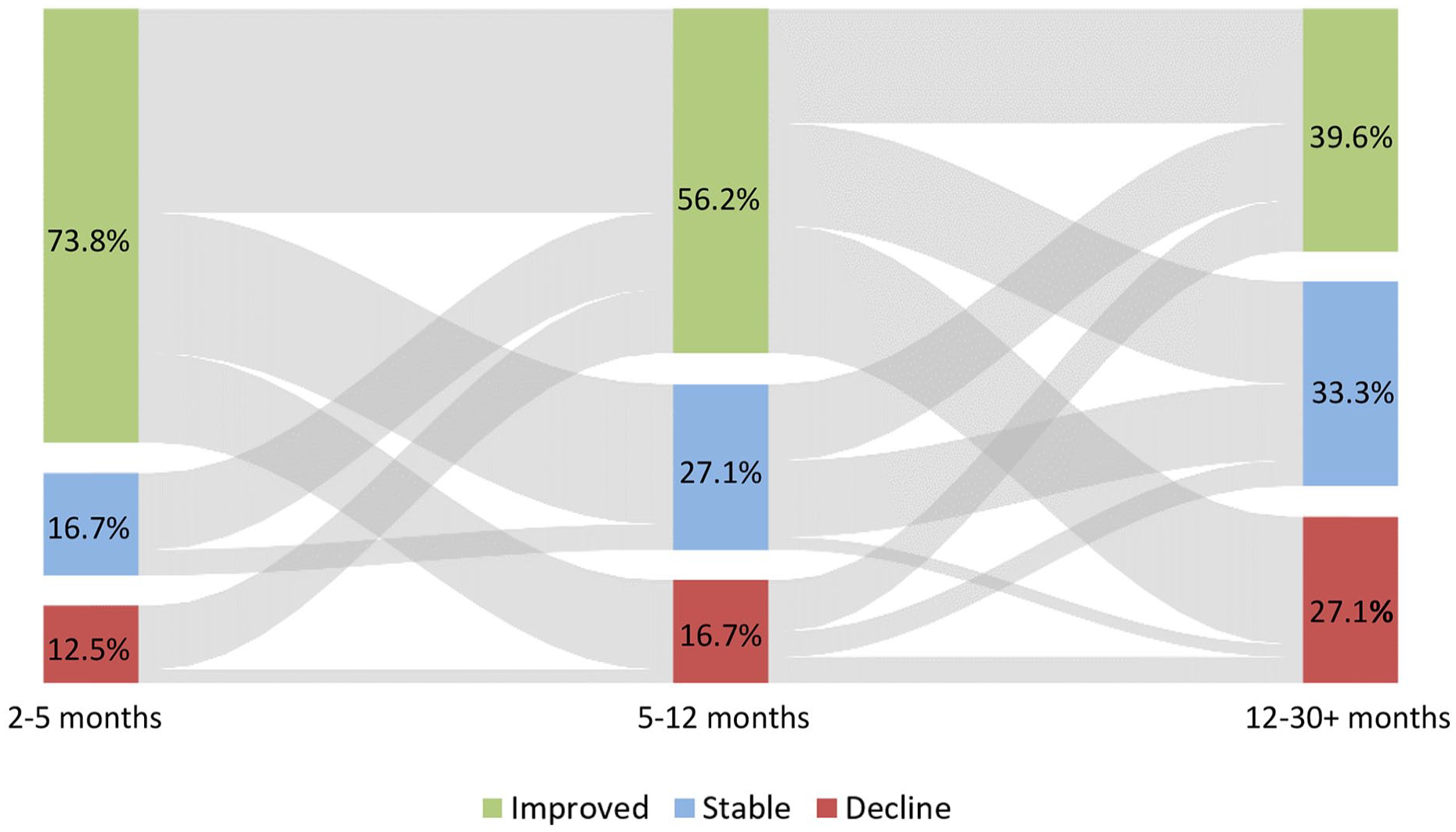
Clinically significant decline, improvement and stability (arranged bottom to top, respectively) using RCI from 2 to 5, 5 to 12, and 12 to 30+ months post-injury time windows.
Figure 1 can also be used to directly compare the findings of the replication sample to that of the original sample studied by Till et al. 16 The proportion of decliners in the replication sample between 5 and 12 months post-injury was 27.1%, which is nearly identical to that of the Till et al 16 sample, which was 27.3% over the same time period. The figure also indicates a greater proportion of patients showing declines the further post-injury, which was consistent with our hypothesis. From 2 to 5 months post-injury, 12.5% show decline; from 5 to 12 months, 17% showed decline.
We collapsed the data for patients classified as decliners from both the Till et al 16 sample and the current sample to look at the pattern of decline across neuropsychological tests. The results are illustrated in Figure 2. Delayed recall on the RAVLT test (RAVLT-dr) and on Logical memory (LMII), and verbal retrieval (COWAT) had the greatest number of decliners, with greater than a third of decliners declining on these measures.
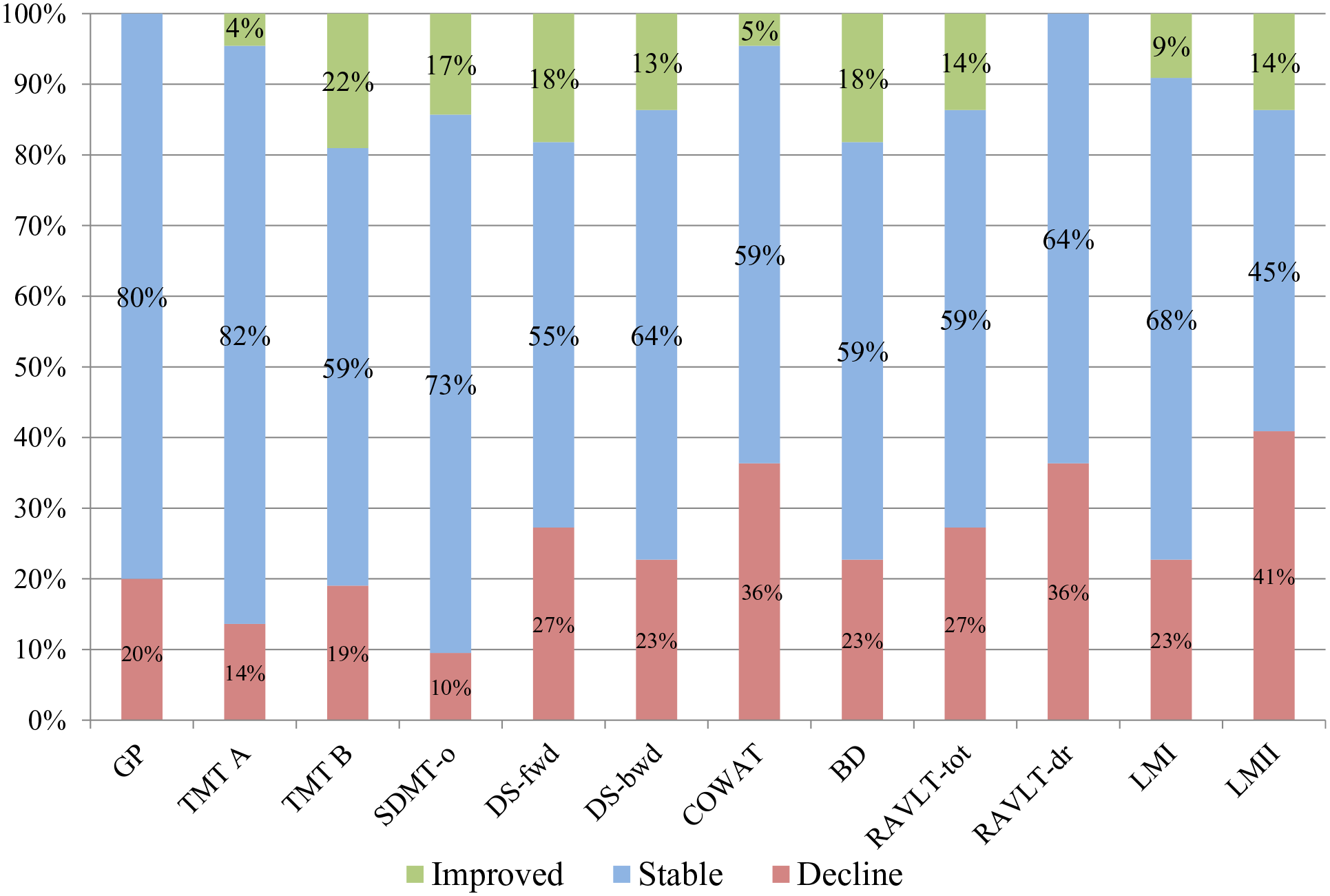
Decline, stability, and improvement (arranged bottom to top, respectively) by neuropsychological task.
Objective 2. Predictors of Cognitive Decline
In our regression models only left hippocampal atrophy from 5 to 12 months (controlling for LPTA) was predictive of decliner status from 12 to 30+ months post-injury (regardless of controlling for right hippocampal atrophy from 5 to 12 months) z = −2.371, P = .018 (see Tables 2 and 3).
Logistic Regression Results for Cognitive Reserve Variables, Therapy Hours, and LPTA as Predictors of Decliner Status 12 to 24 months Post-Injury.

Standard errors are reported in parentheses.
Abbreviations: WTAR, Wechsler Test of Adult Reading; LPTA, length of post-traumatic amnesia.
Logistic Regression Results HPCLTOT and HPCRTOT Decline From 5 to 12 Months Post-Injury as Predictors of Decliner Status 12 to 24 Months Post-Injury Controlling for Age and Estimated Pre-Morbid IQ.

Standard errors are reported in parentheses. Significance code: *0.05. Estimated Pre-morbid IQ is based on the WTAR score collected at 5 months post-injury.
Abbreviations: HPC, hippocampus; WTAR, Wechsler Test of Adult Reading.
Discussion
The first aim of the current study was to provide a more granular understanding of cognitive recovery and decline from 2 months to 30+ months post-injury, both at the level of the patient and the task. In measuring performance at 12 and 30+ months post-injury, we found that 27.1% of our sample declined, which replicated our previous study (Till et al 16 ) that had found a decline rate of 27.3% across nearly the identical timeliness. Given the consistent methodology across the 2 studies, the findings underscore that more than a quarter of patients are at risk of cognitive decline in the first year after moderate-severe TBI. Looking across all 3 time windows of the study, there was an increasing proportion of decliners the more remote patients were from injury (ie, from 12.5% in the first window to 27.1% in the third). To our knowledge, no previous study has examined decline across more than 1 time window within the same cohort. Although the majority of the patients are remaining stable or improving, the findings nonetheless point towards a cognitive picture that worsens as patients move further from injury. Generating hypotheses for future research, we looked at the specific tests on which patients showed the largest number of declines. We found that over a third showed declines on measures of verbal retrieval (ie, RAVLT-dr; Logical Memory-II; COWAT). This finding also guided the second objective of the study, which was to examine “atrophy” (ie, progressive volume loss) of brain structures as a predictor of cognitive decline. In our previous research, we have examined atrophy of the whole brain, grey and white matter structures, and found ubiquitous declines. 33 We therefore assessed whether progressive volume losses in the hippocampi might explain the progressive cognitive declines observed here, and we found that, indeed, left hippocampal volume loss was a predictor of decliner status. The current results highlight the need to continue research and clinical care well into the chronic stages of injury, and to better understand what is contributing to declining function.
Our current findings are compatible with previous research. When comparing our current findings with our own past findings (Till et al 16 ), we very closely replicated: with 27.1% (current) versus 27.3% (previous). Patients in both studies were drawn from the same large, urban setting in Canada’s largest TBI rehabilitation setting. Thus, the findings offer some degree of confidence in the rate of cognitive decline during this window, at least in Canadian samples. With regard to the increasing number of patients declining the further post-injury, no study has previously compared time windows. However, taking all prior studies together, our findings converge somewhat with the literature. For example, Himanen et al 14 studied patients more than 30 years post-injury and found the greatest rate of decline (ie, 56%) of all studies; Ruff et al, 15 studied decline in verbal memory earliest post-injury and found the least amount of decline of 20%. However a more recent study examining patients up to 10 years post-TBI found significant declines only in memory, when examining several cognitive domains, and only in 11% of patients. 29
The overall pattern observed in the current study raises the question whether it reflects the product of multiple beneficial and deleterious mechanisms that operate concurrently after moderate-severe TBI, but with slightly differing timelines. Specifically, while TBI results in a period of spontaneous cognitive recovery, moderate-severe TBI also appears to set in motion acute and long-term processes that are harmful to the brain, including necrosis, apoptosis, diaschisis, protracted neuroinflammation, damage to microtubular transport, and possibly overuse.57,58 The pattern of increasing declines across time may reflect spontaneous recovery reaching coming to an end, as more deleterious and neurodegenerative processes are commencing or continuing. In the absence of spontaneous recovery, the deleterious processes may now play a more predominant role in clinical presentation. Consistent with this pattern, in a group longitudinal study examining recovery of executive function, we observed evidence of early recovery (on a measure of intra-individual variability) followed by later decline. 17
When we explored the tests that were most sensitive to decline (focusing only on decline from 12 to 30+ months post-injury, and collapsing the data from the current study with those from Till et al 16 ) the 3 tests showing the maximum number of decliners were tests of verbal retrieval, delayed recall and verbal fluency. Those tasks with the least number of patients showing decline were tests of attention and speed of processing, namely Symbol Digit, and Trails A and Trails B, 2 of which are also considered measures of executive function (ie, Symbol Digit and Trails B). Our findings add to a growing evidence base that long-term TBI outcomes are not uniform across different cognitive domains, and that verbal retrieval may be particularly susceptible to persisting impairment and perhaps decline. 11 For example, a longitudinal study by Marsh (2018) 59 that found that by 5 years post-injury, while 80% of patients were unimpaired on measures of general intelligence, simple attention and visual perception, 60% demonstrated impairments specific to verbal memory, and 28% demonstrated impairment in verbal fluency. Sigurdardottir et al 29 found declines in memory, but not other cognitive domains in their recent long-term follow up study. As well, a meta-analysis that looked at over at over 70 studies of episodic memory changes following moderate to severe TBI implicated verbal memory as being more sensitive to ongoing injury effects. 60
Our second objective was to examine predictors of decline. Here, we again collapsed the current sample with those from Till et al 16 in order to increase power and analytic stability, and we focused on decliner status from 12 to 30+ months post-injury where we had observed the largest number of decliners. In our first model we examined cognitive reserve variables (age, years of education, estimated pre-morbid IQ), total therapy hours at 5 months post-injury, and LPTA as a measure of injury severity. None of these variables was predictive of decliner status from 12 to 30+ months post-injury. Notably, therapy hours at 5 months—for which the decliner group had significantly less therapy hours than the non-decliner group in our previous study—was not a significant predictor of cognitive decline.
Given the growing evidence of degeneration in the chronic stages of moderate-severe TBI,9,19,22,24-27 we were interested in the possibility that the cognitive declines we observed may be attributable to neural degeneration. Therefore, we examined whether progressive atrophy of the hippocampi—due to the predominance of verbal retrieval measures on which patients showed decline—might be predictive. We found left hippocampal volume loss was a significant predictor of decline. More specifically, when each of the hippocampi were examined while controlling for influences of the contralateral hippocampus, only atrophy of the left hippocampus was significant. This is the first study to our knowledge examining neural, and in particular neurodegenerative predictors of cognitive decline at the individual subject level.
The above findings support the idea that moderate-severe TBI must be considered not only a chronic disorder, but also a progressive one. 19 They also underscore that multiple mechanisms underlie behavioral change during the first years of injury, those that are beneficial to recovery (eg, functional reorganization, restitution, unmasking, resolution of diaschisis61-64 and those that may be deleterious (eg, delayed apoptosis and neuroinflammation)57,58—as both recovery and decline are observed within and between patients at each time point. Additional neurobiological mechanisms may also be at play long-term, such as chronic neuroinflammation, axonal injury, and tauopathy.19,65-68 TBI disrupts the glymphatic system, leading to impaired waste clearance and toxic substance buildup. 69 Excitotoxicity and blood-brain barrier dysfunction further contribute to neural damage. 70 Negative neuroplasticity may result in maladaptive changes, negatively impacting cognitive function.71,72 These interrelated mechanisms highlight the complex nature of TBI-related cognitive decline, emphasizing the need for further research to develop a greater understanding of these mechanisms, and how they may influence one another, in order to better predict outcomes and mitigate decline.
The current study shows that hippocampal volume loss predicted subsequent cognitive decline. Further research is needed to understand how to protect the brain from such declines. The current results highlight the need for ongoing clinical management of patients into the chronic stages of injury, and to deliver clinical strategies that promote brain health, such as intensive physical and cognitive exercise, and mindfulness meditation.73-76
A limitation of the current study, one that would affect the generalizability and reliability of the findings, is the relatively small sample size, particularly given the heterogeneity of the moderate-severe TBI population. Mitigating against this concern, however, is the consistency in prevalence of decliners across the Till et al, 16 and the current study. Another concern regarding the generalizability of the findings is the difference between the Canadian healthcare system for brain injury and that of other contexts. Within the Canadian healthcare system, the large majority of patients are receiving some form of rehabilitation therapy at 5 months post-injury. One might argue, therefore, that our findings underestimate the extent of decline occurring in contexts where patients receive less rehab at 5 months post-injury. However, we did not find evidence of a relationship between therapy hours at 5 months post-injury and decline at 12 to 24 months post-injury. This finding suggests that early therapy might not be protective of later decline, though, our findings are quite limited in the ability to answer this question in that the content of therapy varied considerably across patients. Lastly, absence of a healthy control group is another limitation of our study. In the absence of controls, alternative explanations for the declines include: (1) new onset of a neurological disorder (eg, Alzheimer’s disease); (2) aging-related declines; (3) normal test-retest variability in performance. 49 Onset of new neurological disorder was an exclusion criterion, although undiagnosed dementia in a participant cannot be conclusively ruled out. With regard to aging-related decline, the magnitude of decline observed across the 7-month time window (ie, 5-12 months post-injury) and approximately 18 month time window (ie, 12-30 months post-injury) is not compatible with our understanding of the typical rate of cognitive decline in adults of this age-range. 77 With regard to the third alternative explanation, the use of RCI is intended to mitigate against measurement-related test-retest variability. However, it is important to note that recent studies have shown transient declines in healthy adult cognitive performance. Therefore, it is possible that the prevalence of decline observed in these studies is an overestimate.
Conclusions
The current results, replicating past results, suggest that cognitive decline in the chronic stages of injury (ie, 12–30+ months post-injury) affects at least a quarter of patients, and with an increasing proportion of patients showing decline across time windows (ie, 2-5 months, 5–12 months, and 12–30+ months post-injury)—though for the most part, it is not the same patients declining at each window. Tests of verbal retrieval appeared to be most sensitive to decline, and our findings suggest that hippocampal volume loss—within the left hippocampus in particular—may explain this decline. No other predictors of decline, offered clear prognostic value in our sample. Further research is needed to better understand who is at risk of decline, and critically, to determine how to prevent decline.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors report no conflicts of interest and assert that all procedures contributing to this work comply with the ethical standards of the University Health Network Research Ethics Board on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Authors would like to acknowledge financial support from the Canadian Institutes of Health Research (grant# MOP 86704), The Walter and Maria Schroeder Foundation, The Joseph & Antoinette Sorbara Foundation, the Canada Research Chairs Program, and the Physicians’ Services Incorporated Foundation (12-43).
