Abstract
Background
Visual cues can improve gait in Parkinson’s disease (PD), including those experiencing freezing of gait (FOG). However, responses are variable and underpinning mechanisms remain unclear. Visuo-cognitive processing (measured through visual exploration) has been implicated in cue response, but this has not been comprehensively examined.
Objective
To examine visual exploration and gait with and without visual cues in PD who do and do not self-report FOG, and healthy controls (HC).
Methods
17 HC, 21 PD without FOG, and 22 PD with FOG walked with and without visual cues, under single and dual-task conditions. Visual exploration (ie, saccade frequency, duration, peak velocity, amplitude, and fixation duration) was measured via mobile eye-tracking and gait (ie, gait speed, stride length, foot strike angle, stride time, and stride time variability) with inertial sensors.
Results
PD had impaired gait compared to HC, and dual-tasking made gait variables worse across groups (all P < .01). Visual cues improved stride length, foot strike angle, and stride time in all groups (P < .01). Visual cueing also increased saccade frequency, but reduced saccade peak velocity and amplitude in all groups (P < .01). Gait improvement related to changes in visual exploration with visual cues in PD but not HC, with relationships dependent on group (FOG vs non-FOG) and task (single vs dual).
Conclusion
Visual cues improved visual exploration and gait outcomes in HC and PD, with similar responses in freezers and non-freezers. Freezer and non-freezer specific associations between cue-related changes in visual exploration and gait indicate different underlying visuo-cognitive processing within these subgroups for cue response.
Keywords
Introduction
Freezing of gait (FOG) is a common motor symptom affecting people with Parkinson’s Disease (PwPD), 1 which commonly leads to falls. 2 FOG is defined as short, episodic periods of reduced forward motion of the feet, despite the intention to walk. 3 FOG often occurs during situations like gait initiation, turning, and dual-task walking,4,5 with occurrence increasing with disease progression. 6 Falls can result in injury and contribute to increased fear of falling, reduced activity levels, poor quality of life, and caregiver stress. 7 Thus highlighting the importance of, and need for, strategies and therapies to reduce falls and fall risk factors (ie, FOG). 8 Pharmacological treatments such as dopaminergic medication have been shown to reduce the duration and occurrence of FOG. 9 Despite this, evidence for the use of medication for FOG is limited and inconclusive, 10 with many patients displaying no improvement. 11 Implementation of non-pharmacological intervention for FOG, such as cueing has been shown to reduce falls and improve mobility in PwPD. 12 Visual cues (normally a transverse line to step over) are thought to be the most effective strategy for people with FOG, as they are more visually dependent for effective gait than those without FOG. 13 This is due to impairment in their proprioceptive feedback from the lower limbs.14,15 Visual cues provide spatial information used for the initiation or ongoing facilitation of gait, 16 which helps those with FOG to overcome gait deficits. Specifically, visual cueing can enhance various gait characteristics such as step length17-19 and improve gait initiation. 20 However, the underlying mechanisms for cue response remain poorly understood. 21 This has led to variation in intervention responses 18 and selective short-term improvements in selective gait outcomes (ie, only step length or reduced variability). 22
Underlying and integrated visual and cognitive (termed visuo-cognitive) mechanisms have been implicated in visual cue response in PwPD.23-25 Indeed, our recent research has shown that cortical brain regions involved in visuo-cognitive processing (ie, parietal and occipital cortices) are involved in visual cue response in those with FOG compared to those without FOG. 26 Visual cues may help to focus visuo-cognitive processing on specific task relevant regions of interest (eg, the lines to step over27,28), as real-world environments are increasingly visually complex and those with FOG struggle to inhibit visual distractors (ie, they look at thing unrelated to the task they are doing). One method of measuring visuo-cognitive processing in real-time is by recording saccadic (fast, jerk-like) eye movements, 29 as eye movements are a proxy measurement for visuo-cognition. Saccades are the movements between eye fixations, and form the basis for visual exploration and are influenced by visuo-cognitive neural processes, 30 subsequently, providing a means to explore the mechanisms behind visual cue responses. The frequency of saccades (visual exploration) is reduced in PwPD during walking compared to healthy older adults, which has been linked to deficits in visuo-cognitive, particularly attentional, processes.31,32 However, research has illustrated that visual cueing can ameliorate deficits and improve both saccade frequency (ie, the number of saccades per second) 25 and fixation number (ie, number of pauses of areas of interest)19,33 when walking in PwPD, which suggests that visual cueing may drive visual exploration and be underpinned by visuo-cognitive (particularly attentional) processes. However, to date there have been no studies that have investigated comprehensive visual exploration (both saccadic and fixation outcomes) and its response to visual cueing in individuals with and without FOG. 34 The lack of comprehensive outcomes likely relates to the poor sampling frequency (ie, 30-50 Hz) of previous eye-trackers that only allow for saccadic or fixational counts to be obtained accurately. 35 Advances in eye-tracking technology now allows for comprehensive eye-tracking outcomes to be provided at high resolution (ie, >100 Hz) during mobile tasks, such as walking. Comprehensive visual exploration outcomes will allow for greater understanding of visuo-cognitive processes underpinning cue response, as well as exploration of whether improvements in visual exploration are related to gait improvements in PwPD, 23 which should allow tailoring of future visual cue interventions. 23
This study aims to: (1) examine comprehensive visual exploration (saccades and fixations) response to visual cues during walking in PwPD and healthy controls (HC), and in PwPD who do and do not report FOG; and (2) investigate the relationship between visual exploration and gait response to visual cues in PwPD compared to HC, and in PwPD with and without FOG. We hypothesised that selective aspects of visual exploration will change with visual cues while walking, with those who report FOG experiencing the greatest improvement. Additionally, we hypothesised that changes in visual exploration will relate to gait improvement in PwPD, particularly within those that report FOG.
Methods
Participants
This interventional study examined visual exploration and gait response to visual cues during walking in PwPD that do and do not report FOG. Forty-three PD participants (n = 22 with self-reported FOG and n = 21 without FOG) and 17 age-matched HC participants at the Oregon Health and Science University (OHSU, Portland, Oregon, USA) Movement Disorders Clinic. Self-reported FOG was based upon question 1 of the new FOG Questionnaire after seeing the short clip related to the questionnaire. Subjects were categorized as “freezers” if they had experienced such a feeling or episode over the past month. All subjects provided their written informed consent prior to the experiment, and the study was approved by an Institutional Review Board (IRB) at OHSU, Portland, USA (#9903).
PwPD were included if they had clinical diagnosis of idiopathic PD by a movement disorder specialist according to UK brain bank criteria, a Hoehn and Yahr (H&Y) rating of stage II to III, 36 aged ≥50 years, adequate vision and hearing (Snellen chart visual acuity ≥12/18), and able to walk and stand unaided. PD participants were excluded if they had; (1) cognitive impairment (ie, Montreal Cognitive Assessment [MoCA] score <21) 37 ; (2) unstable medication for 1 month prior to study; (3) psychiatric co-morbidity; (4) acute lower back or lower/upper extremity pain; (5) peripheral neuropathy; (6) unable to comply with protocol; (7) rheumatic or orthopedic diseases affecting balance and gait.
Data Collection
Data were collected at the Balance Disorders Laboratory, Oregon Health and Science University, Portland, Oregon, USA between July 2018 and November 2019. Each participant attended a 2-hour session at the laboratory. Participants were tested in their usual ON medication state (within 60 minutes of taking anti-Parkinsonian medications).
Clinical Assessment
Participants underwent a series of demographic, clinical, and cognitive assessments. The following tests were administered; the Movement Disorder Society Unified Parkinson’s Disease Rating Scale Motor Subscale (MDS-UPDRS III), 38 a 15-minute assessment of motor signs related to PD severity. Global cognition was assessed with the MoCA. 37 Attention was measured with a computerized button pressing battery, involving simple reaction time (SRT), choice reaction time (CRT), and digit vigilance (DV). 39 Executive function was measured using Royall’s clock drawing (CLOX 1&2). 40 Working memory and visuo-spatial ability were measured through seated forward digit span and judgment of line orientation tasks, 41 respectively. Basic visual functions of acuity and contrast sensitivity were assessed using standardized charts (logMar and logCS).
Gait Assessment
Participants walked, at self-selected comfortable pace, over a 9 m straight path (with a 180° turn at each end) for 2 minutes. Four conditions were performed in a randomised order and included single and dual-task walking without, and with, visual cues. Under dual-task conditions, participants completed the forward digit span test whilst walking.
Initially, 2 numbers were played over a loudspeaker at a rate of 1 per second for the participant to recall and continued to a maximum of 9 numbers. 42 For the visual cue condition, there were transverse black lines on the floor (at a distance 20% larger than participant baseline step length), which participants were asked to step over. Gait was measured using wearable inertial measurement units (128 Hz, Mobility Lab version 2, APDM, Inc.), which consisted of a gyroscope, accelerometer, and magnetometer. Inertial sensors were worn, on the wrists (x2), feet (x2), shanks (x2), sternum (x1), and lumbar spine (x1) attached via straps.
Gait outcomes were provided through the Mobility Lab proprietary software, and included stride length (m), gait speed (m/s), stride time variability, stride (s) time, and foot strike angle (°). These gait characteristics were chosen as they are valid 43 and have previously been used in visual cueing studies.24,44,45 Only straight walking metrics were included. Turning was not examined, as the transverse nature of the visual cues along the 10 m pathway means that they are only used for straight walking (ie, there were no cues used specifically for the 180° turns).
Visual Exploration
Mobile infrared eye-tracker (Tobii Pro Glasses 2, Tobii, Inc.) was used to objectively measure visual exploration during walking. Saccades (frequency, number, duration, amplitude) and fixations (duration) were measured during walking tasks. Raw eye-tracker data were analyzed with a validated custom-made Matlab algorithm 43 with findings of this validation study showing good to excellent agreement Intraclass correlation coefficient (ICC > 0.75) across various gait features such as gait velocity, stride length, cadence and stride time in older adults, younger adults, and individuals with PD. The use of the mobile infrared eye tracker was previously validated by Stuart et al. 35 and was found to be relatively robust, with it correctly detecting 85% of saccades made by participants during a walking task.
Statistical Analysis
Statistical analysis was performed with IBM SPSS Statistics version 26. Normality of data was checked with Kolmogorov–Smirnov tests and visually assessed using box and scatterplots. Parametric statistics were used to assess descriptive demographic data (via Means, Standard Deviations, T-tests), and to examine visual exploration and gait changes in response to visual cueing. Separate linear mixed-effects models were conducted for each metric to examine the effect of intervention (cue, no-cue) and task (single, dual-task), with group (freezers, non-freezers) as a between-subjects factor. Further Pearson’s correlational analysis was conducted within each group (freezers, non-freezers, HC) to examine relationship between change scores (cue—no-cue) of visual exploration and gait. This was done for both single and dual task. Due to the exploratory nature of the study, significance level was set to P < .05 for statistical interpretation.
Results
Participants
Demographic, cognitive, visual, and clinical demographic data are displayed in Table 1. FOG, non-FOG, and HC were matched for age, height, weight, education, gender and across MoCA, CLOX1, CLOX2, and visual acuity scores. Non-freezers were more greatly impaired in terms of cognition, attention, executive function, working memory, visuo-spatial ability, and visual function than HC. This impairment was even greater in FOG in all domain’s bar the CLOX2 assessment as part of executive function. Freezers were significantly impaired when compared to HC in SRT (P = .022), CRT (P = .017), DV (P = .001), contrast sensitivity (P = .027), and Geriatric Depression Scale (GDS-15) (P = .007). Additionally, FOG had significantly more falls over the past 12 months (P = .031), and significantly worse attention (CRT [P = .029] and DV [P = .018]) compared to non-FOG.
Demographic, Cognitive, Visual, and Clinical Characteristics.
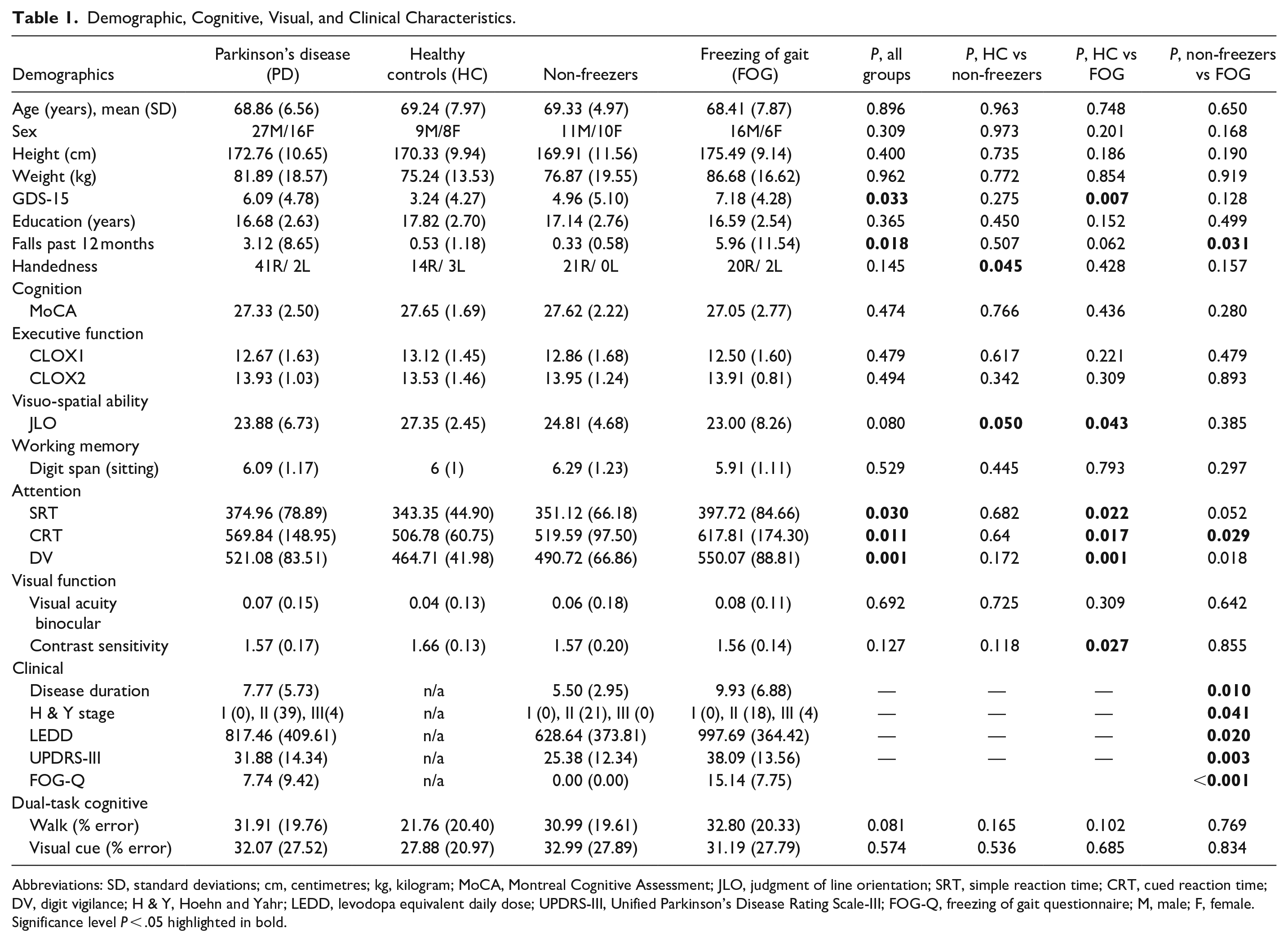
Abbreviations: SD, standard deviations; cm, centimetres; kg, kilogram; MoCA, Montreal Cognitive Assessment; JLO, judgment of line orientation; SRT, simple reaction time; CRT, cued reaction time; DV, digit vigilance; H & Y, Hoehn and Yahr; LEDD, levodopa equivalent daily dose; UPDRS-III, Unified Parkinson’s Disease Rating Scale-III; FOG-Q, freezing of gait questionnaire; M, male; F, female.
Significance level P < .05 highlighted in bold.
Eye Movement Outcomes: Effect of Visual Cues
In general, there were no significant differences in saccadic activity between the groups (Table 2) and dual tasking was found to have no significant effect on the eye movement outcomes (P < .01, Table 2). However, Table 2 shows that visual cues significantly increased saccade frequency (P ≤ .01), but reduced saccade peak velocity and amplitude in all groups (HC, freezers, non-freezers, P ≤ .01). Additionally, a cue by group interaction showed significant reduction in saccade peak velocity (P = .03) and increased saccade duration (P = .05) with visual cues in HC and non-freezers compared to freezers (Table 2). Post hoc analysis also showed specific reduction in saccade peak velocity in non-freezers with visual cues compared to without visual cues (P = .006), whereas HC and freezers did not significantly reduce their saccade peak velocity.
Linear Mixed Effects Model Results for Gait and Eye Tracking Variables Across All Participants Groups With and Without Visual Cues and During Single and Dual Tasks.
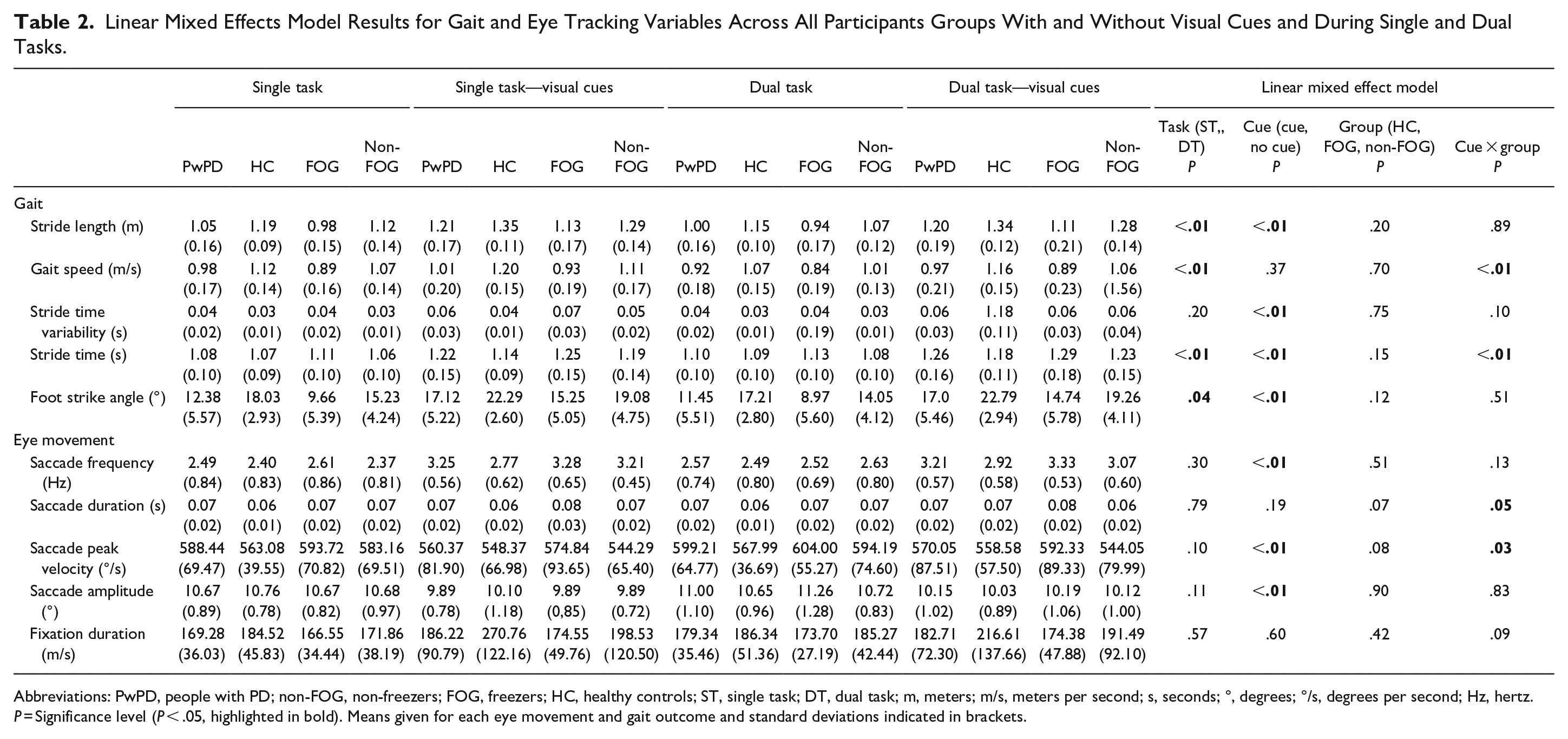
Abbreviations: PwPD, people with PD; non-FOG, non-freezers; FOG, freezers; HC, healthy controls; ST, single task; DT, dual task; m, meters; m/s, meters per second; s, seconds; °, degrees; °/s, degrees per second; Hz, hertz.
P = Significance level (P < .05, highlighted in bold). Means given for each eye movement and gait outcome and standard deviations indicated in brackets.
Gait Outcomes: Effect of Visual Cues
PwPD generally had worse gait than HC, and dual tasking worsened all gait variables apart from stride time variability (all P ≤ .05). Visual cues improved stride length, foot strike angle, stride time, and stride time variability in all groups for both single and dual task (P ≤ .01). A cue by group interaction showed that stride time increased significantly with visual cues for all 3 subgroups (post hoc analysis: freezers, non-freezers, and HC; P ≤ .001, P ≤ .001, P = .003 respectively), but freezers had the greatest increase. Post hoc analysis did not detect a significant intragroup change in gait speed across walking conditions.
Visual cues did not influence cognitive dual-task outcomes. Specifically, there was no significant difference in dual-task cognitive task percentage error score for any of the groups during walking without or with the visual cues (Table 1). Additionally, there was not a significant difference between dual-task cognitive task error score from walking without and with visual cues for HC (P = .077), PD (P = .966); non-freezers (P = .715) or freezers (P = .744).
Association Between Cueing-Related Changes in Visual Exploration and Gait
Significant relationships between cue-related changes in visual exploration and gait with visual cues were observed for PwPD under both single and dual-task conditions, but not for HC (Tables 3 and 4). Specifically, under single-task conditions, greater stride length related to less change in saccade duration (r = −.5, P = .042), foot strike time related to less change in saccade amplitude (r = −.5, P = .033), and stride time variability related to less change in saccade velocity and saccade amplitude (r = −.5, P = .040; r = −.6, P = .008 respectively) with visual cues in non-freezers. Additionally, more frequent saccades with visual cues related to increased stride length in freezers (r = .5, P = .040) with visual cues under single task. Under dual-task conditions, increased speed with visual cues related to greater saccade frequency (r = −.7, P = .002) in non-freezers. In freezers, increased stride length and foot strike angle related to increased saccade frequency (r = .7, P = .002; r = .5, P = .024 respectively) with visual cues. Furthermore, increased stride length and foot strike angle related to increased saccade duration with visual cues in freezers (r = .5, P = .017; r = .5, P = .034). Overall, this evidence suggests that there are PD and freezer specific associations between cue-related changes in visual exploration and cue-related changes in gait.
Relationships Between Cueing-Related Changes in Visual Exploration and Gait in Single Task.
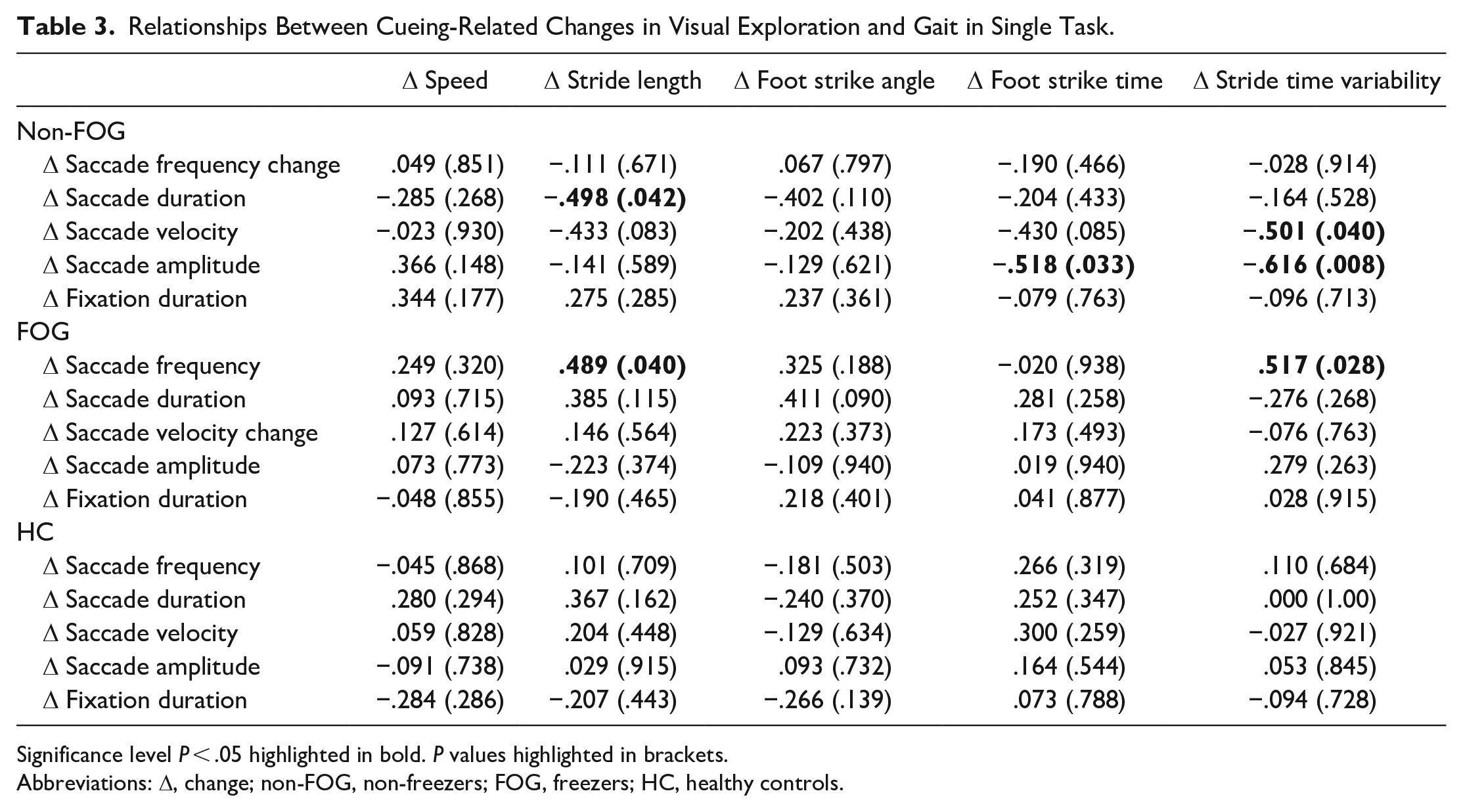
Significance level P < .05 highlighted in bold. P values highlighted in brackets.
Abbreviations: Δ, change; non-FOG, non-freezers; FOG, freezers; HC, healthy controls.
Relationships Between Cueing-Related Changes in Visual Exploration and Gait in Dual Task.
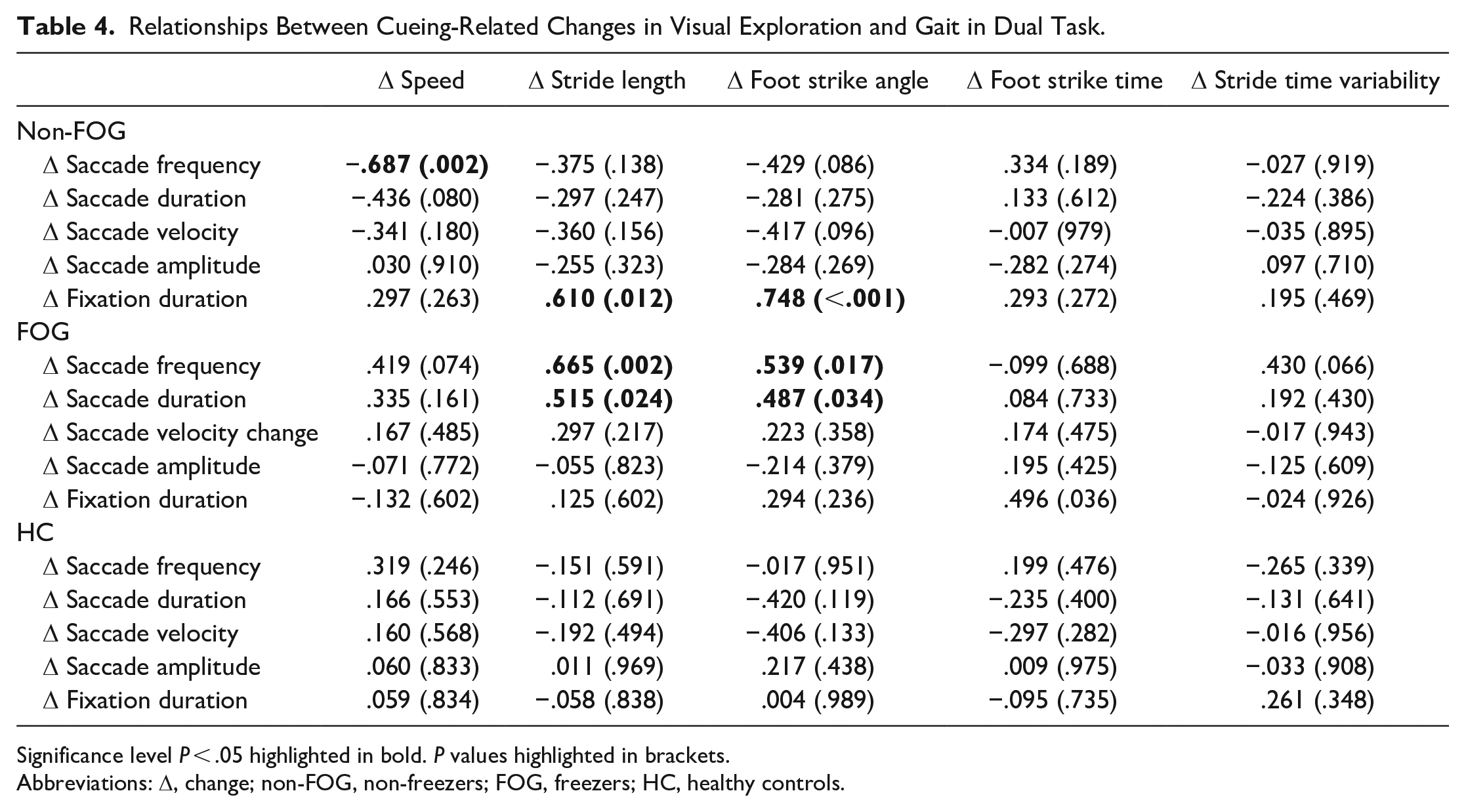
Significance level P < .05 highlighted in bold. P values highlighted in brackets.
Abbreviations: Δ, change; non-FOG, non-freezers; FOG, freezers; HC, healthy controls.
Discussion
To our knowledge, this is the first study to comprehensively explore the influence of visual cues on visual exploration (saccade and fixation outcomes) during gait in healthy older adults and PwPD who do and do not report FOG. Unsurprisingly, visual cues produced significant improvements in gait compared to usual walking across all groups. In line with our hypothesis, visual cueing significantly influenced selective visual exploration outcomes compared to usual walking with increased saccade frequency, decreased saccade peak velocity and amplitude observed across all groups, which demonstrates that visuo-cognitive processes underpin cue response regardless of group. However, in contrast to our hypothesis, saccadic peak velocity had significant reduction with visual cues in non-freezers compared to HC and those with freezing, which highlights that selective visuo-cognitive processes may response differently in non-freezers compared to other groups. Additionally, in line with our hypothesis, changes in visual exploration outcomes related to gait improvements with cueing in PwPD, but in contrast to our hypothesis relationships were selective across freezers and non-freezers. Our results highlight that visuo-cognitive processes underpin visual cue response in PwPD, with processes being subtly different across those who do and do not self-report FOG.
Visual Exploration When Walking: Response to Visual Cues
This study demonstrates that visual cues were found to significantly increase saccade frequency during gait across all groups, which is in keeping with the findings of Stuart et al. 25 and Vitorio et al. 19 where visuo-cognitive, particularly attentional, mechanisms were reported to underpin cue response. Similarly, Horowitz et al. 46 also found that highly visible targets and specific task goals resulted in improved saccadic activity in PwPD during static tasks. The current study went beyond previous work by recording comprehensive visual exploration with high resolution (>100 Hz) mobile infra-red eye-tracking, allowing additional measures of velocity, duration, and amplitude of eye movements to be measured. Previous research has been unable to provide comprehensive visual exploration outcomes, as prior studies have lacked a validated algorithm to obtain outcomes from eye-tracking data, and they also had lower sampling frequency (30-50 Hz) eye-tracking devices.14,19,25 It is known that frequencies ≥100 Hz are required to accurately attain comprehensive visual exploration outcomes (saccades and fixations), 35 which has limited previous studies due to the technological limitations and limits direct comparison to this study.
Visual cues significantly reduced saccade peak velocity and amplitude in all groups (HC, freezers, non-freezers), which may relate to enhanced goal directed visuo-cognitive processing. Reflexive saccades (triggered via visual distraction) typically have greater velocity that voluntary attentionally controlled saccades, 47 therefore the reduction in saccade velocity and the narrower amplitude of eye movements suggests more voluntary saccades were elicited with visual cues (ie, focus on the visual cues and avoidance of distractors). 28 Interestingly, saccade peak velocity was particularly reduced in non-freezers, with HC and freezers responding in a similar manner, which may indicate that visuo-cognitive processes underpinning cue response are similar in HC and freezers. Visuo-cognitive processes, particularly attentional processing, has been implicated in visual cue response in PwPD in our previous research.23,25,26,28,48 To examine the influence of attention, the present study used attentional distraction through dual tasking, which was found to have no significant effect on any of the eye movement outcomes when walking with visual cues compared to single-task conditions, which is similar to findings by Stuart et al. 25 Visual cues may reduce the visuo-cognitive processing of the environment that is required when walking in PwPD which allows specific focus on task relevant areas of interest (lines on the floor) and frees attentional resources to be applied to concurrent tasks (ie, the dual-task). 49
Gait Outcomes When Walking: Response to Visual Cues
PwPD (freezers and non-freezers) were found to have significant impairment in gait speed, stride length, and foot strike angle compared to HC. These gait impairments amongst PwPD are already well established having been highlighted previous research.50-52 Additionally, dual-tasking made these gait variables worse across all groups, particularly stride time, in line with previous literature. 53 As expected, visual cues increased stride length, foot strike angle, and stride time in all groups, similar to previous research in PwPD.17,18,20 Slower stride time with visual cues occurred across all groups (HC, freezer, non-freezer) but was particularly slow in freezers, which may indicate a more cautious gait approach with cues.
Our findings show significant relationship between gait outcomes and visual exploration outcomes in PwPD but not HC, which highlights the increased visuo-cognitive processing required for visual cue response in PwPD. More specifically, relationships existed between gait and visual exploration changes with visual cues for freezers and non-freezers. Results highlighted that improved gait (increased speed, stride length, etc.) was related to improved visual exploration (more frequent saccades, etc.) with visual cues, but these were selective to group (freezer vs non-freezer) and task (single vs dual). This is an important finding as it shows that underlying visuo-cognitive mechanisms may be different in Parkinson’s compared to controls and within freezer versus non-freezer sub-groups.48,54
Limitations
There are several limitations to highlight. First, this study only examined immediate effect of visual cues on visual exploration and gait outcomes, rather than continuous home-based cue use as performed in clinical practice. This gives no indication of whether any improvements in these outcomes from visual cues are lasting or whether they continue to occur in the absence of visual cues. Secondly, this study only examined information collected about eye movements and does not investigate how this information is processed through brain activity, or how freezers and non-freezers may process information differently. This could potentially be explored in further research and contribute to our understanding of the mechanisms behind visual cue response (ie, mobile brain imaging studies26,55-57) and aid the development of therapeutic interventions. Finally, there is a lack of contextual analysis whereby there is no means of determining where or what people look at whilst walking. This could, for example, be used to determine how far ahead individuals look while walking and a validated algorithm has been presented in previous work to semi-automate this. 27 Contextual analysis would be useful in future investigation as PwPD have been shown to visually explore their environments with less efficiency than HC. 28
Conclusion
This study provides valuable insight into the visual cue response of visual exploration during walking in HC and PwPD, who do and do not display FOG, which may improve mobility and reduce falls risk. Overall, visual cues significantly influenced several visual exploration and gait outcomes in HC and Parkinson’s participants, which highlights the requirement for visuo-cognitive processing to respond to cues in PwPD. Additionally, there were freezer and non-freezer specific associations between cueing-related changes in visual exploration and gait that suggests different visuo-cognitive mechanisms may underpin cue response in these 2 sub-groups.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a Parkinson’s Foundation postdoctoral fellowship for basic scientists (PI: Samuel Stuart) (PF-FBS-1898), a Parkinson’s Foundation clinical research award (PI: Samuel Stuart) (PF-CRA-2073), and a National Institutes of Health (NIH) career development award (PI: Martina Mancini) (5R00HD078492-04). Lisa Graham is supported through a collaborative PhD studentship between Northumbria University and Head Diagnostics (PIs: Dr Morris & Dr Stuart). Julia Das is supported through a collaborative PhD studentship between Northumbria University and Senaptec Inc. (PI: Stuart).
