Abstract
Background
Evidence showed that patients with Parkinson’s disease (PD) who have a history of freezing of gait (FOG) have hypometric anticipatory postural adjustment (APA) during gait initiation (GI) compared to PD without FOG.
Objectives
This study aimed to test the feasibility of center of pressure (COP) displacement during GI as the measure of APA in PD with and without a history of FOG.
Methods
Patients with PD underwent COP trajectory measurements, including duration, length, velocity, and acceleration in different phases of APA (APA1, APA2a, APA2, and LOC), as well as evaluation of New Freezing of Gait Questionnaire (NFOG-Q), Tinetti balance and gait score, and Postural Instability and Gait Difficulty (PIGD) score in the on and off medication states.
Results
The duration (seconds) of APA2a, APA2b, and LOC were highest while velocity in mediolateral direction (X) (m/s), including APA1, APA2a, APA2b, and LOC showed lowest in PD with FOG. Velocity in the mediolateral direction in different phases of APA increased in patients with FOG after dopaminergic therapy. APA2a (seconds) and APA2b (X) (m/s) were significantly associated with NFOG-Q part II, APA2b (X) (m/s) was significantly associated with NFOG-Q part III, and APA2a (seconds) was significantly associated with Tinetti balance and gait and PIGD score.
Conclusions
PD with FOG history showed a favorable response of APAs to dopaminergic replacement. The APA parameters by COP trajectory, especially lateral COP shift toward the stance foot (APA2b (X) (m/s) and APA2a (seconds)) are surrogate markers to assess PD with FOG history.
Keywords
Introduction
Freezing of gait (FOG) is an episodic sudden cessation of locomotion, characterized by the feeling of the feet being glued to the ground and an inability to generate effective forward-stepping movements. 1 It is common in many forms of atypical parkinsonism and advanced Parkinson’s disease (PD).1,2 It is a distressing symptom that can lead to falls, decreased mobility, and a decreased quality of life. Therefore, investigating and predicting FOG episodes provides an opportunity for its prevention.
Anticipatory postural adjustment (APA) has gained attention because it indicates that a physiological mechanism is crucial for initiating a step. Therefore, APA is considered an indicator of balance control ability during gait initiation (GI). 3 Patients with PD and FOG have difficulty in GI, a longer movement preparation period, and a shorter forward thrust. The dissociation between APA and step onset in these cases raise the possibility that FOG may be caused by decoupling between postural preparation and GI. 4 Therefore, we can reasonably infer that APA is abnormal in PD patients with FOG.
Clinical studies showed patients with PD are hypometric, have smaller APA amplitude, longer APA duration during GI, as well as a reduced first-step speed and relatively short first-step length compared to those of healthy individuals,5,6 and hypometric APA also contribute to FOG in PD. 7 In addition to FOG, APA measurements may also be affected by levodopa therapy,8,9 weight loading, 10 and initial stance condition. 11
During quiet standing, the vertical projection of the center of mass (COM) onto the ground falls at the center of pressure (COP). When the subject lifts the foot to step forward, a gap between the COP and the COM may occur, causing a disequilibrium toward the stance leg. 12 To minimize the risk of disequilibrium, the central nervous system utilizes APAs and activates the trunk and leg muscles before the onset of postural disturbance.
Several methods to record APAs include recording the excursions of the COP of the subject standing on a force plate,13,14 electromyography of the leg muscles,7,15 or accelerometers.5,14 In the present study, only the excursions of the COP displacement trajectories were considered. GI is a transient procedure that involves anticipatory anteroposterior and lateral movements. A postural model for COP displacement trajectories in GI can divide the task into postural and locomotor phases (LOC). 16 The postural phase refers to the APA that occurs before the movement phase, while the locomotor phase supports the first step changes. The postural phase can be further divided into 2 sub-phases (APA1 and APA2) which require coordinated actions of 2 postural mechanisms as follows: APA1 represents the translation of the COP in the lateral and posterior directions together toward the swing foot heel (“ankle strategy” dominated by ankle muscles) and APA2 represents a lateral CoP shift toward the stance foot (“hip strategy” dominated by hip abductor/adductor muscles). 17 APA2 is further divided into 2 subphases: APA2a and APA2b.
To this end, we investigated 2 alternative hypotheses for APA measurement in PD patients with FOG. The first hypothesis was that a longer APA duration and decreased COP displacement velocity may be associated with worse FOG and contribute to falling risk and postural instability. The second hypothesis was that APA measurements can be affected by levodopa therapy in patients with FOG. In this study, we also attempted to determine which parameters of COP displacement during the APA subphase could serve as surrogate biomarkers for the FOG severity and postural instability in PD with FOG history.
Patients and Methods
Study Design and Patient Selection
We prospectively evaluated 145 patients who met the International Parkinson and Movement Disorder Society clinical diagnostic criteria for idiopathic PD as well as the magnetic resonance imaging diagnostic criteria for PD.18,19 Patients were included in this study only if their daily anti-Parkinsonian agents were titrated to a steady dose, and the Hoehn and Yahr stage was 1 to 3 and therefore, could walk independently.20,21 Patients were excluded if they had advanced PD stage (Hoehn and Yahr staging ≥4), cognitive decline that precluded following our instructions, any etiology that could interfere with balance (eg, visual, vestibular, and proprioception problems), and lower limb weakness. This study was approved by the Institutional Review Committee on Human Research of the hospital (IRB 201901802B0). All participants received verbal and written information regarding the purpose of this study and signed an informed consent form. Finally, 91 patients were included in the analysis. For clinical comparisons, 23 age- and sex-matched healthy volunteers were included in the control group.
Clinical Assessment of PD
Clinical assessments, including clinical rating scale assessments, were performed during both off- and on-medication status. 22 Patients underwent an evaluation in the off status after an overnight fast of at least 12 hours from dopaminergic medications. The time between taking dopaminergic medications and the “on” status to undergo testing was averaged at 1 to 1.5 hours. All patients underwent complete history taking for their age at disease onset, sex, body mass index (BMI), disease duration, and levodopa equivalent daily dose (LEDD). 23 The clinical severity of PD was assessed using the Unified Parkinson’s Disease Rating Scale (UPDRS) and Hoehn and Yahr stages.20,21 The total UPDRS score was computed as the sum of the UPDRS subscores I, II, III, and IV. UPDRS-derived Postural Instability and Gait Difficulty (PIGD) score, 24 which comprised UPDRS items 13, 14, 15, 29, and 30, fulfills the criteria for a “recommended” scale and has adequate clinometric characteristics in posture, gait, and balance measure in PD. It ranges from 0 to 20, with higher scores reflecting greater PIGD severity also assessed.25,26 The Cognitive Abilities Screening Instrument (CASI C-2.0) consists of 20 items divided into 9 domains, and the sum of the scores ranges from 0 to 100, with higher scores indicating better cognitive ability. 27 The presence of FOG was assessed using the New Freezing of Gait Questionnaire (NFOG-Q). 28 The NFOG-Q is a widely used and valid tool to quantify FOG severity 28 and it is constructed in 3 parts as follows: part I classified patients as a freezer (FR) or a non-freezer (NFR) based on whether they had experienced FOG episodes during the past month, part II (items 2-6, scoring range 0-19) rated the severity of FOG based on its duration and frequency in its most common manifestation, and part III (items 7-9, scoring range 0-9) rated the severity of FOG on daily activity. Participants were classified into having FOG if they answered “yes” in item 1 of the NFOG-Q. Furthermore, we used the Tinetti gait and balance assessment tool to assess fall risk in patients with PD. 29 It consists of 2 parts: balance score (score = 0-16) and gait score (score = 0-12). The Tinetti total score ranged from 0 to 28 points by summing the points obtained for each of the 2 tests.
Measurement of Parameters Obtained by the COP Trajectory
The COP trajectory was determined using a TekScan MatScan pressure mat model 3150 (TekScan Inc., South Boston, USA). The participants were asked to stand barefoot on a TekScan MatScan pressure mat in a relaxed posture on both legs, in a fixed and parallel position. The task was assessed after verbal instructions were administered to initiate gait and start with the most involved leg in patients with PD while starting with the right leg in healthy controls. In total, there were 6 tests for each patient (off state [n = 3] and on state [n = 3]). The mean values of 3 successive recordings were averaged and used in the analyses.
Method for Data Acquisition and Analysis
The TekScan MatScan pressure mat records parameters of COP displacement trajectories during GI as follows. First, the data of the COP displacement trajectories were collected every 0.03 seconds, all the rows, and columns at each point were prepared, and the changes in the tracks of COP in every sample were obtained. Second, the Ramer–Douglas–Peucker algorithm was used to simplify the points and find the turning points. 30 The turning points identified by the Ramer–Douglas–Peucker algorithm mean the turning point between 2 phases (eg, the turning point between APA1 and APA2a). The program can find 5 turning points in COP displacement trajectories using the algorithm (Figure 1A). If 5 turning points were not detected, we used several methods to adjust the turning points to 5. After obtaining rows and columns at each turning point, the length of 1 segment line between 2 turning points, that is, APA1, APA, APA2a, and LOC, respectively, was calculated. Moreover, the velocity and acceleration were calculated. Finally, the turning points for every sample were also evaluated by professionals for the proper segment in turning points for each sample. The turning points were artificially adjusted by professionals if there was no proper segment for the turning points. During the entire process, we used Python to automatically find the turning points and calculate the duration, length, velocity, and acceleration of APAs and LOC. In this study, the parameters of COP displacement trajectories, including duration, length, and velocity (track velocity [m/s] in the segments in the anteroposterior [Z] and mediolateral direction [X]), and acceleration of the COP trace in these phases (APA1, APA2a, APA2b, and LOC) among patients with and without FOG and healthy controls were calculated and compared (Figure 1B).
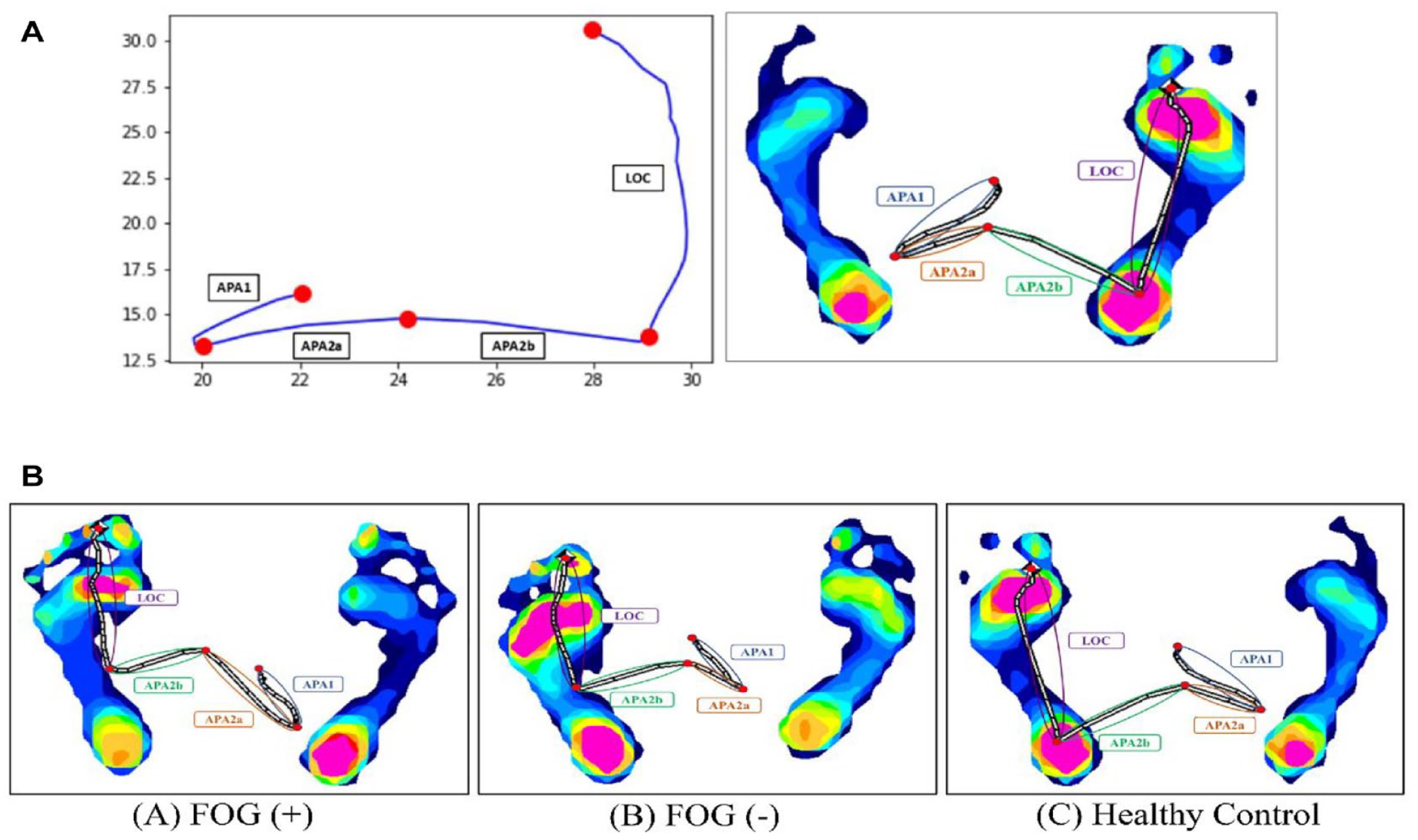
Method for calculating APA parameters during COP displacement and analysis by the Ramer–Douglas–Peucker algorithm (Figure 1A). (A) Center-of-pressure displacement trajectories during GI in the horizontal plane in 1 subject with PD with FOG, (B) 1 subject with PD without FOG, and (C) 1 healthy control.
Statistical Analysis
Data are expressed as mean (standard deviation [SD]). Baseline continuous variables between the 2 groups were compared using the mean of the independent t-test. Parameters of COP displacement measurements in different sub-phases of APA and LOC among the 3 groups (healthy controls, PD with FOG, and PD without FOG were compared using 1-way analysis of variance (ANOVA). Clinical scores and parameters of COP displacement measurements between the 2 groups (PD with and without FOG) before and after dopaminergic therapy (off and on status) were compared using repeated-measure ANOVA. Correlation analysis was used to determine the relationship between the parameters of COP displacement in different sub-phases of APA and NFOG-Q Part II, NFOG-Q Part III, and Tinetti total score. Multiple linear regression analysis using a stepwise procedure was performed to evaluate the influence of independent variables on NFOG-Q Part II, NFOG-Q Part III, and Tinetti total score (dependent variable). All statistical analyses were conducted using the IBM SPSS Statistics v23 statistical software (IBM, Redmond, WA, USA).
Results
General Characteristics of Patients With PD
A total of 91 patients with PD were included in this study. Of these, 31 patients had FOG (mean age, 65.2 ± 9.1 years) and 60 patients did not (mean age, 69.2 ± 10.0 years). Patients’ baseline characteristics are presented in Table 1. PD with FOG history had a longer disease duration (years) and higher LEDD (mg); UPDRS II, III, and IV; total scores; and Tinetti balance and gait scores (al1 P < .0001), and higher Hoehn and Yahr staging (P = .001) than those who did not have FOG history.
Baseline Characteristics of Patients.
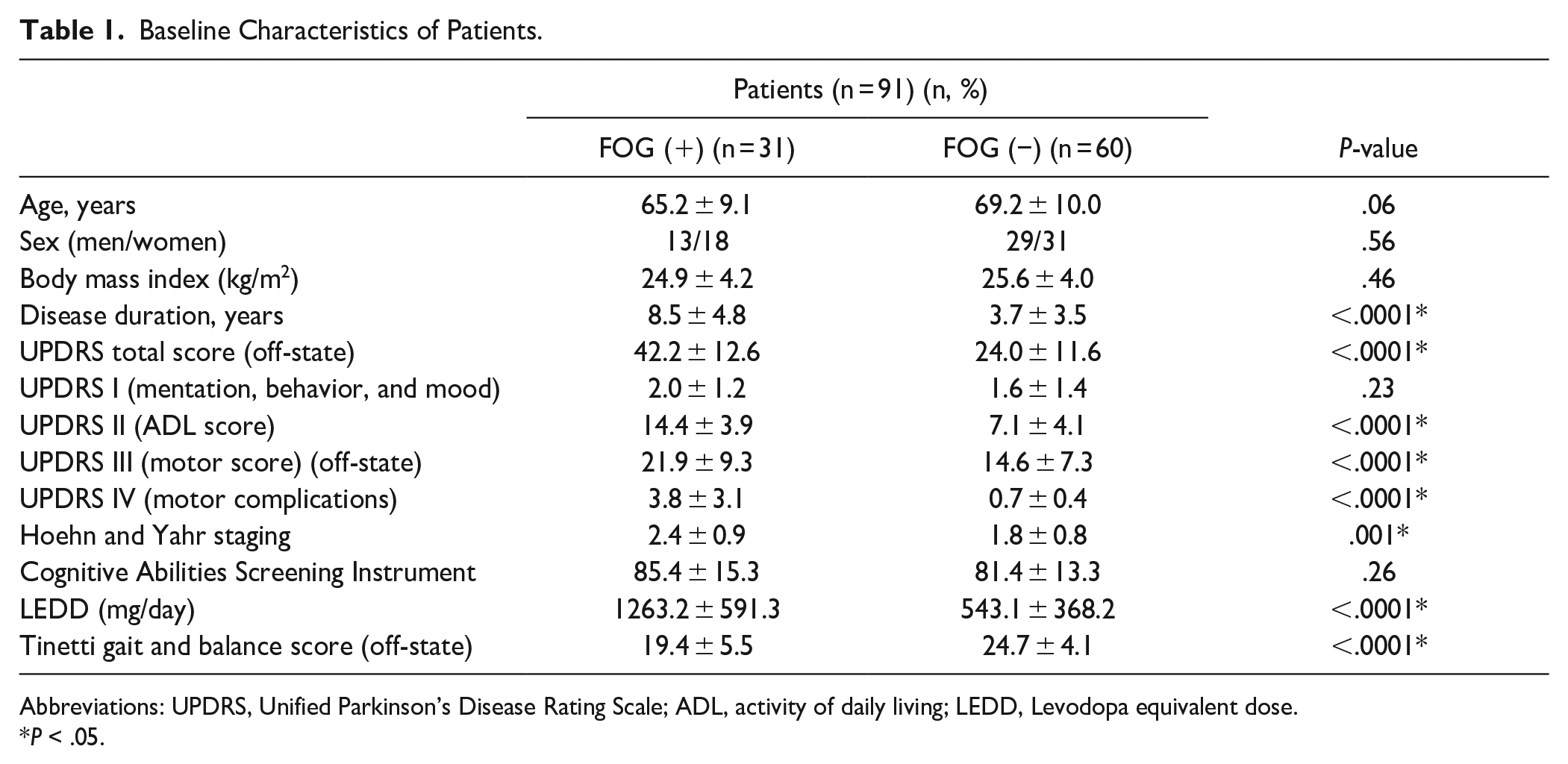
Abbreviations: UPDRS, Unified Parkinson’s Disease Rating Scale; ADL, activity of daily living; LEDD, Levodopa equivalent dose.
P < .05.
UPDRS Total Score and Tinetti Balance and Gait Score Before and After Dopaminergic Therapy
The UPDRS total score and Tinetti total score between PD with and without FOG history during the off and on states were listed in Table 2. The UPDRS total score and Tinetti total score before and after dopaminergic therapy in PD with FOG history showed a significant difference (P < .0001 and P < .0001, respectively), and UPDRS total score and Tinetti total score before and after dopaminergic therapy in PD without FOG history also showed a significant difference (P < .0001 and P < .0001, respectively). Furthermore, statistical significance was observed in the UPDRS and Tinetti total scores between PD with and without FOG history before and after dopaminergic therapy using repeated measures ANOVA (P < .0001 and P < .0001, respectively).
COP Displacement Parameters in Patients With PD With or Without FOG and Healthy Control.
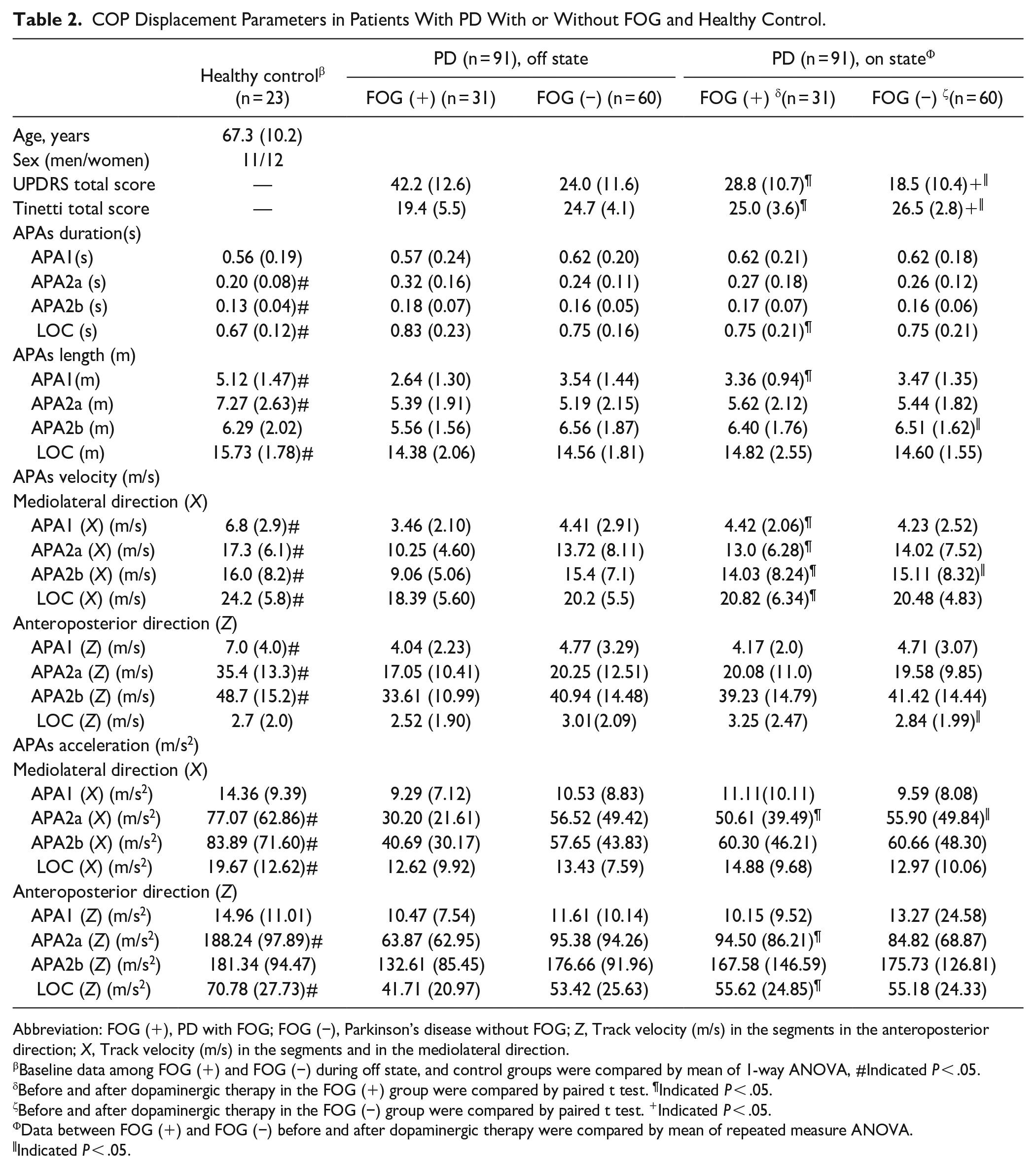
Abbreviation: FOG (+), PD with FOG; FOG (−), Parkinson’s disease without FOG; Z, Track velocity (m/s) in the segments in the anteroposterior direction; X, Track velocity (m/s) in the segments and in the mediolateral direction.
Baseline data among FOG (+) and FOG (−) during off state, and control groups were compared by mean of 1-way ANOVA, #Indicated P < .05.
Before and after dopaminergic therapy in the FOG (+) group were compared by paired t test. ¶Indicated P < .05.
Before and after dopaminergic therapy in the FOG (−) group were compared by paired t test. +Indicated P < .05.
Data between FOG (+) and FOG (−) before and after dopaminergic therapy were compared by mean of repeated measure ANOVA.
Indicated P < .05.
Parameters Obtained by the COP Trajectories During GI Among Patients With PD With and Without FOG History and Healthy Controls
The duration (seconds), length (m), velocity (m/s), and acceleration (m/s2) of the baseline APAs and LOC among 3 groups (PD patients with and without FOG history during off-state and healthy controls) were listed in Table 2 and Figure 2. The duration (seconds) of APA2a (seconds), APA2b (seconds), and LOC (seconds) were higher in PD with FOG history, followed by PD without FOG history and healthy controls (APA2a [s], P = .001, APA2b [s], P = .006, and LOC [s], P = .004). The length (m) of APA1(m), APA2a (m), and LOC (m) were significantly lower in PD with FOG history, followed by PD without FOG history and healthy controls (APA1[m], P < .0001, APA2a [m], P = .001, and LOC [m], P = .02); Velocity (m/s) in mediolateral direction (X), including APA1 (X) (m/s), APA2a (X) (m/s), APA2b (X) (m/s), and LOC (X) (m/s) showed significantly higher in healthy control, followed by PD without FOG history and PD with FOG history (APA1 [X] [m/s], P < .0001, APA2a [X] [m/s], P = .002, APA2b [X] [m/s], P < .0001, and LOC [X] [m/s], P = .001); Velocity (m/s) in anteroposterior direction (Z), including APA1 (Z) (m/s), APA2a (Z) (m/s), and APA2b (Z) (m/s) showed significantly higher in healthy control, followed by PD without FOG history and PD with FOG history (APA1 [Z] [m/s], P = .004, APA2a [Z] [m/s], P < .0001, and APA2b [Z] [m/s], P = .001); The other parameters including acceleration (m/s2) in the mediolateral direction (X) and the anteroposterior direction (Z) among 3 groups were listed in Table 2.
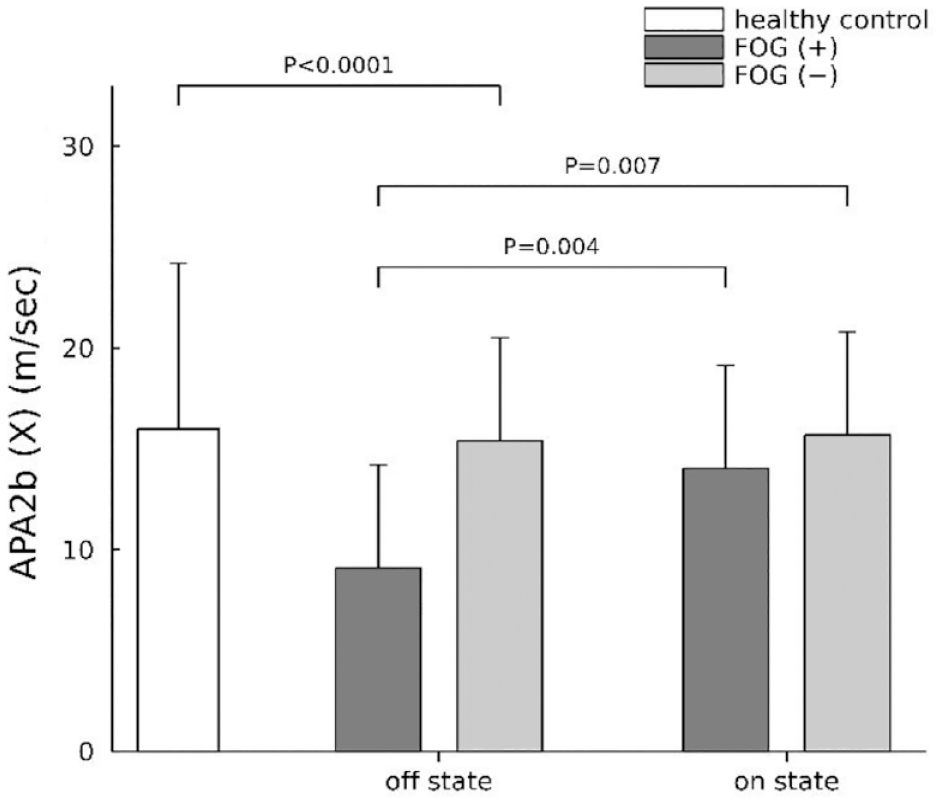
APA2b (X) velocity (m/s) among patients with and without FOG, and healthy control before and after dopaminergic therapy.
Parameters Obtained by the COP Trajectories During GI Among Patients With PD With or Without FOG Before and After Dopaminergic Therapy
The APA1 length (m) and the velocity in the mediolateral (X) direction, including APA1 (X) (m/s), APA2a (X) (m/s), APA2b (X) (m/s), and LOC (X) (m/s) before and after dopaminergic therapy in PD with FOG history showed a significant increase (APA1[m], P = .002, APA1 [X] [m/s], P = .01, APA2a [X] [m/s], P = .002, APA2b [X] [m/s], P = .004, LOC [X] [m/s], P = .008, APA2a [X] [m/s2], P = .002, APA2b [Z] [m/s2], P = .04, and LOC [X] [m/s2], P = .004). In contrast, the APAs and LOC duration (seconds), length (m), and velocity (m/s) in the mediolateral (X) and anteroposterior direction (Z), and acceleration (m/s2) in the mediolateral (X) and anteroposterior directions (Z) before and after dopaminergic therapy in PD without FOG did not show any statistical significance. Furthermore, after controlling for age and height, statistical significance was observed in APA2b (m), APA2b (X) (m/s), LOC (Z) (m/s), and APA2a (X) (m/s2) between PD with and without FOG history before and after dopaminergic therapy (repeated measures ANOVA, APA2b [m], P = .02; APA2b [X] [m/s], P = .007; LOC [Z] [m/s], P = .049; and APA2a [X] [m/s2], P = .04).
Correlation Analysis Between APA Parameters and the NFOG-Q Parts II and III, Tinetti Balance and Gait Score, and UPDRS PIGD Score
The correlation analyses used to test the influence of the APA parameters during COP displacement in the off state, NFOG-Q parts II and III, Tinetti balance and gait score, and UPDRS PIGD score are listed in Table 3. APA duration in APA2a (seconds) was significantly correlated with the NFOG-Q parts II and III, Tinetti balance and gait score, and PIGD score (P = .001, P = .005, P < .0001, and P < .0001, respectively). APA length in APA1 (m) during the off state was significantly correlated with the NFOG-Q parts II and III, Tinetti balance and gait score, and PIGD score (P = .001, and P = .005, P = .007, and P < .0001, respectively). APAs velocity (m/s) in mediolateral direction (X), including APA2a (X) (m/s) and APA2b (X) (m/s) during the off state, was significantly correlated with the NFOG-Q parts II and III, Tinetti balance and gait score, and PIGD score (P = .02, P = .04, P = .0006, and P < .0001 in APA2a (X), and P < .0001, P < .0001, P = .007, and P = .007, respectively in APA2b (X)). APA2b (Z) (m/s) during the off state, was significantly correlated with the NFOG-Q parts II and III, Tinetti balance and gait score, and PIGD score (P = .004, P = .02, P = .0007, and P < .0001, respectively). Other correlation analyses are listed in Table 3 and the Supplemental Figure.
Correlation Analysis Between Parameters of COP Displacement Measurements and the NFOG-Q, Tinetti Total Score, and UPDRS PIGD Score.
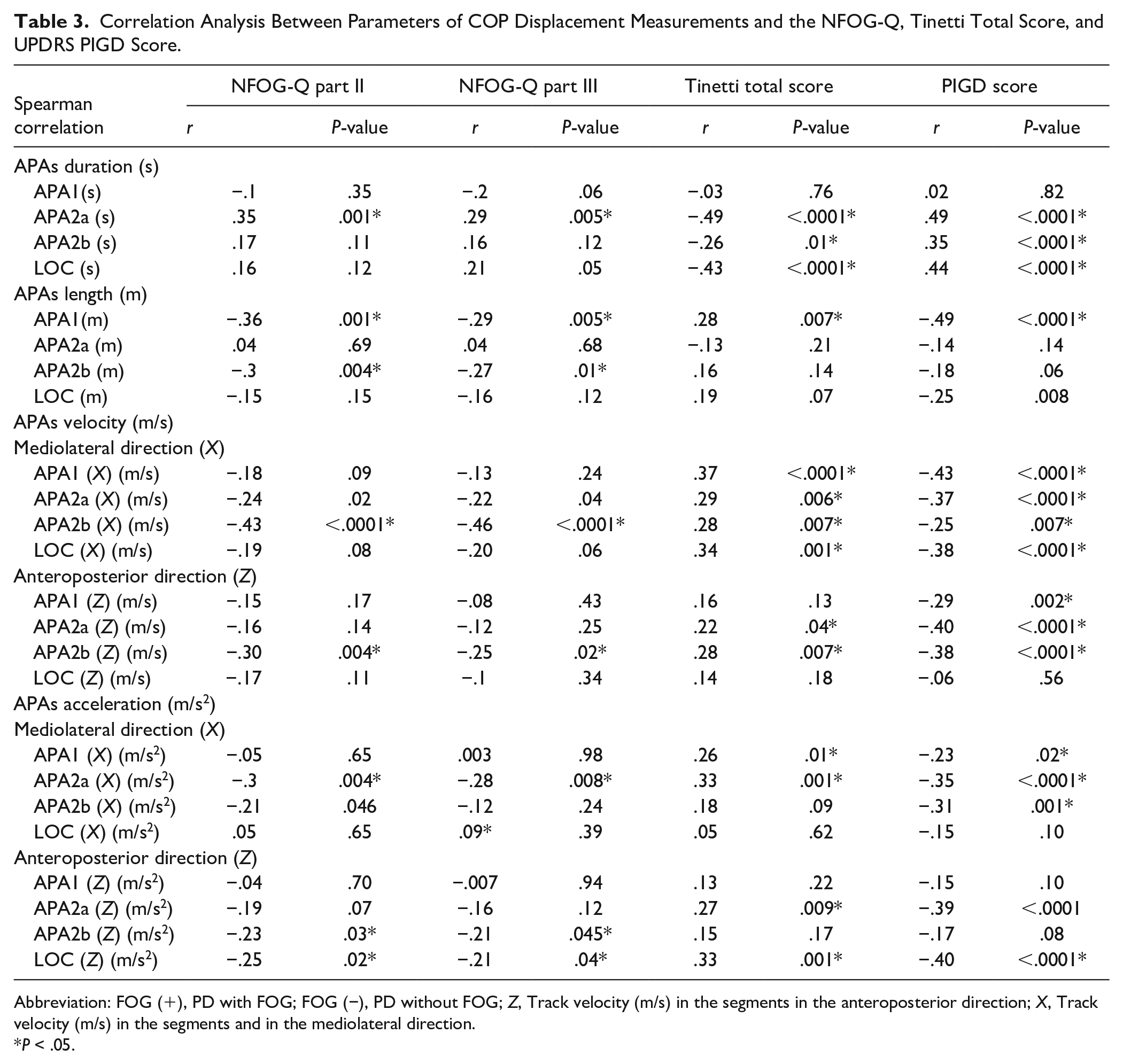
Abbreviation: FOG (+), PD with FOG; FOG (−), PD without FOG; Z, Track velocity (m/s) in the segments in the anteroposterior direction; X, Track velocity (m/s) in the segments and in the mediolateral direction.
P < .05.
Clinical Factors Significantly Associated With NFOG-Q Parts II and III, Tinetti Balance and Gait Score, and PIGD Score
The effects of the variables that were significantly associated with NFOG-Q parts II and III, Tinetti balance and gait score, and PIGD score in the multiple linear regression model are listed in Table 4. We used only the significant variables in the correlation analysis (Table 3) for the multiple linear regression analysis models to identify the crucial factors that influence the augmented NFOG-Q parts II and III, Tinetti balance and gait score, and PIGD scores in patients with PD. In the multiple linear regression model, NFOG-Q parts II and III, Tinetti balance and gait score, and PIGD score were introduced as the dependent variables, respectively. The significant parameters of COP displacement measurements shown in Table 3 served as the independent variables into the analysis. The results revealed that APA2a (seconds) (β = 9.3, P = .03) and APA2b (X) (m/s) (β = −.28, P = .001) were predictors of the NFOG-Q part II (severity of FOG), APA2b (X) (m/s) (β = −.18, P < .0001) as a predictor of the NFOG-Q part III (severity of FOG in daily living), APA2a (seconds) (β = −18.65, P < .0001) as a predictor of the Tinetti balance and gait score (fall risk) and APA2a (seconds) (β = 11.43, P < .0001) as a predictor of the PIGD score (Table 4).
Effects of COP Displacement Parameters on NFOG-Q Part II, NFOG-Q Part III, Tinetti Total Score, and UPDRS PIGD Score: Multiple Linear Regression.
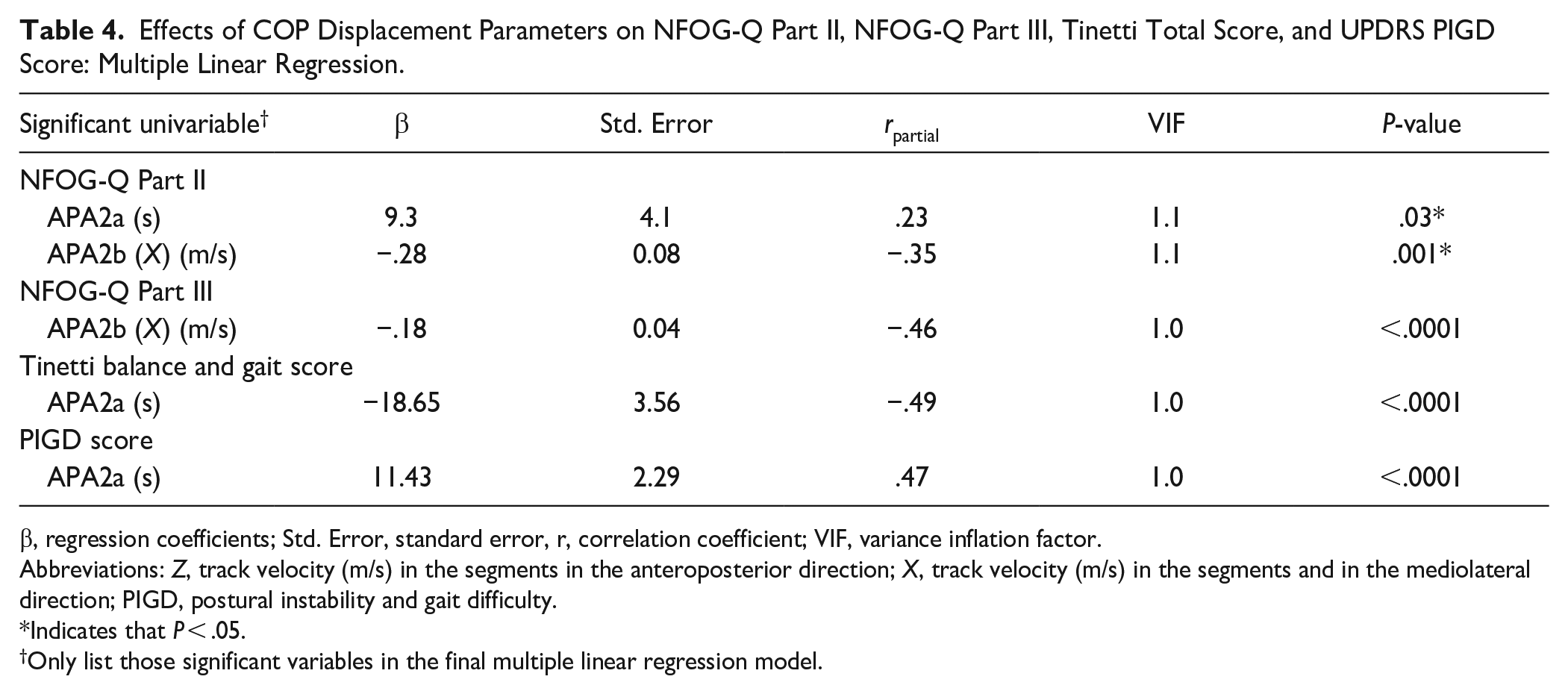
β, regression coefficients; Std. Error, standard error, r, correlation coefficient; VIF, variance inflation factor.
Abbreviations: Z, track velocity (m/s) in the segments in the anteroposterior direction; X, track velocity (m/s) in the segments and in the mediolateral direction; PIGD, postural instability and gait difficulty.
Indicates that P < .05.
Only list those significant variables in the final multiple linear regression model.
Discussion
Major Findings
This study evaluates COP trajectory measurements during GI in patients with PD with and without a history of FOG and produced 3 major findings. First, patients who had a history of FOG had a longer APA duration and higher LEDD, UPDRS total scores, and Tinetti balance and gait scores. Second, PD patients with a history of FOG had a longer APA duration and decreased COP displacement velocity than those patients without a history of FOG. Finally, velocity in the mediolateral direction in different phases of COP trajectory in patients with FOG history showed a significant increase after dopaminergic therapy. It demonstrated the favorable response of APAs to dopaminergic replacement in PD with FOG history.
Proposed Mechanism for APA on the Pathophysiology of FOG
The APA precedes step initiation to shift the COM laterally and forward over the stance limb through activation of hip abductors and ankle dorsiflexors before foot lift. 31 The clinical model demonstrated that the lateral COP shift toward the swing foot is dominated by ankle muscles during the APA sub-phase (APA1) 17 while the lateral COP shift toward the stance foot (APA2a and APA2b) is dominated by the hip abductor/adductor muscles. 17 The lateral gastrocnemius plays an important role in the push-off of the foot during the stance phase, 32 and the medial gastrocnemius muscle is important for forward propulsion of the body and vertical support, to maintain gait speed and postural balance. 32 Patients with PD had reduced muscle activity on ankle plantar flexion, 32 longer APA duration,5,33 and bradykinesia. PD with FOG is associated with more severely impaired preparation and GI execution. The hip muscle forces produced by muscle contractions contribute to the joint contact force and generate moments during straightforward walking. 34 Major abnormalities could explain the inability to create effective forward-stepping movements in patients with FOG. 1
Comparison of APA Parameters Between Patients With and Without FOG
One study showed that the lateral COP shift toward the swing foot was reduced in patients with FOG history, but the backward COP shift between patients with and without FOG history did not show a statistical difference. 12 The lateral COP shift was also reduced in other studies, including early stage 5 and moderately affected PD patients. 35 Our study also showed both the length of COP shift toward the swing foot (APA1 length (m)) and lateral COP shift in medio-lateral direction toward the stance foot (ApA2b (X) length (m)) are significantly reduced in patients with FOG. Reduced APA can serve as an explanation for the difficulty in GI, which may be a possible cause of the freezing phenomenon, though “trembling in place” has also been postulated as any etiology of FOG. 4
Reduction in APA2b Velocity Is Strongly Associated With NFOG-Q
The above-mentioned APA measurements for FOG may provide important clues to its etiology. If a particular parameter is implicated in the pathophysiology of FOG when the same parameter is found consistent with the FOG severity, then there is a plausible mechanistic link between the parameter and disease progression. Our study showed that APAs velocity in the mediolateral (X) and anteroposterior directions (Z) in APA1, APA2a, APA2b, and APAs acceleration in the mediolateral (X) and anteroposterior directions (Z) in APA2a, APA2b, were significantly lower in patients with FOG history. The APA velocity in APA2b in the mediolateral direction was significantly associated with both NFOG-Q parts II and III. In summary, APA2b (X) (m/s) can serve as a surrogate parameter of COP trajectory to assess PD with FOG history for its significant association with both NFOG-Q parts II and III.
The Effects of Dopaminergic Therapy on APA Measurements
Although the effectiveness of dopaminergic therapy on APA measurements remains controversial,8,9 most studies in favor of a reduction in APA measurements can be partially compensated by dopaminergic therapy.8,9 Our study showed that the patients with FOG history had significant improvement in APA measurements, especially APA velocity in the mediolateral direction, after dopaminergic therapy. For patients with FOG, dopamine replacement therapy may improve bradykinesia in the mediolateral direction, especially the APA2b (X) velocity, contributing to increasing hip muscle forces in the hip abductor/adductor muscles, corresponding to an increased hip range of motion, 36 ultimately making the gait more efficient.
Strengths and Limitations
This study had 3 strengths. First, this study confirmed the feasibility that COP trajectory serves as a less time-consuming evaluation method for APA measures to assess those patients with FOG history. Second, PD with FOG history showed a favorable response of APAs to dopaminergic replacement. Third, it highlights that APA parameters, especially the lateral COP shift toward the stance foot (eg, APA2a (seconds) and APA2b (X) (m/s)) are the surrogate markers of APA to assess those patients with FOG history.
Our study has some limitations. First, we used the COP trajectory to evaluate the PD with FOG history, but there was a lack of lower limb joint kinematics and kinetics. However, our study attempted to quantify the GI strategy in PD with a less time-consuming method using 1 force platform than other conventional assessments. Second, patients with advanced PD or mild-to-moderate dementia were excluded. Thus, there is some uncertainty in assessing the role of COP displacement measurements in non-selected patients with FOG history and fall risk. Third, no patient suffered FOG during the study, and our study only provides information about background neurological status rather than during FOG status in PD patients with FOG. Finally, the underlying pathophysiology of FOG in patients with PD remains unclear. Dopaminergic therapy generally reduces the severity of FOG, and long-term dopaminergic therapy may contribute to FOG development. 37 Although our study showed the favorable response of APAs to dopaminergic replacement in those with FOG, dopaminergic treatment is not always successful in managing FOG, and, sometimes, can aggravate FOG (“ON” freezing). Furthermore, the New FOG questionnaire cannot identify ON freezing. We did not record those patients who were responsive FOG, “on” state FOG, and unresponsive FOG at the baseline during enrollment. Therefore, our study did not investigate the differences in APA measurements between different subtypes of FOG (eg, responsive, and unresponsive to levodopa). We also did not investigate the differences in APA measurements between different subtypes of FOG (eg, responsive FOG and unresponsive FOG to levodopa and “on” state FOG). 38
Conclusion
Our study highlights the favorable response of APAs to dopaminergic replacement in PD with FOG history. The APA parameters by COP trajectory, especially the lateral COP shift toward the stance foot (eg, APA2b (X) (m/s) and APA2a (seconds)) are surrogate markers to assess PD with FOG history. The COP trajectory can serve as a less time-consuming evaluation method for APA and could provide scientific evidence for developing exercise programs aimed at specifically improving stability during postural transitions.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683231166934 – Supplemental material for Effectiveness of Center of Pressure Trajectory as Anticipatory Postural Adjustment Measurement in Parkinson’s Disease With Freezing of Gait History
Supplemental material, sj-docx-1-nnr-10.1177_15459683231166934 for Effectiveness of Center of Pressure Trajectory as Anticipatory Postural Adjustment Measurement in Parkinson’s Disease With Freezing of Gait History by Chien-Feng Kung, PhD, Yun-Ru Lai, MD, PhD, Wen-Chan Chiu, MD, Chia-Yi Lien, MD, Chih-Cheng Huang, MD, PhD, Ben-Chung Cheng, MD, PhD, Wei-Che Lin, MD, PhD, Yueh-Sheng Chen, MD, Chiun-Chieh Yu, MD, Yi-Fang Chiang, BS, Yan-Ru Guo, BS, Yin-Hong Chen, BS and Cheng-Hsien Lu, MD, MSc in Neurorehabilitation and Neural Repair
Footnotes
Author Contributions
Conceptualization: CFK and CHL; methodology: CFK and CHL; formal analysis: YRL and CHL; investigation: CFK, YRL, WCC, CYL, CCH, BCC, WCL, YSC, CCY, YFC, YRG, YHC, and CHL; resources: CHL; writing-original draft preparation: CFK; writing-review and editing: YRL and CHL; supervision: CHL; project administration: CHL; funding acquisition: CHL. All the authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the Ministry of Science and Technology (MOST 109-2314-B-182A-079-MY3).
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Institutional Review Board Statement
The study was conducted by the guidelines of the Declaration of Helsinki and was approved by the Institutional Review Board of Chang Gung Medical Foundation (IRB 201901802B0).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
