Abstract
Background
Moderate-to-vigorous physical activity (MVPA) may confer benefits for axonal and/or neuronal integrity in adults with multiple sclerosis (MS).
Purpose
Examine the association between device-measured MVPA with optical coherence tomography (OCT) metrics of retinal nerve fiber layer (RNFL) thickness and total macular volume (TMV) in persons with and without MS.
Methods
Adults with MS (N = 41), along with sex-matched healthy control (HC) participants (N = 79), underwent measurements of retinal morphology via OCT and wore an accelerometer for a period of 7 days as a measure of MVPA.
Results
Persons with MS had significantly lower MVPA, RNFL thickness, and TMV compared with HCs. MVPA was correlated with RNFL (r = .38, P < .01) thickness and TMV (r = .49, P < .01). Hierarchical linear regression analyses indicated that addition of MVPA attenuated the Group effect on RNFL and TMV. MVPA accounted for 8% and 3% of the variance in TMV (β = .343, P < .01) and RNFL thickness (β = .217, P = .03), respectively.
Conclusion
MVPA was positively associated with axonal and neuronal integrity assessed by OCT and partially explained group differences in those metrics. These results present possible future targets for MS management by increasing MVPA.
Keywords
Introduction
Multiple sclerosis (MS) is one of the most common demyelinating diseases in the world with prevalence rates exceeding 1000/1,000,000 persons in some countries. 1 Clinically, MS is a chronic neurological disease in which inflammation triggers the demyelination and axonal lesions located throughout the central nervous system (CNS) including the brain, spinal cord, and optic nerve. 2 Eventually, this degradation will result in the delay or obstruction of action potentials along neuronal pathways causing neurological impairments. 3 Common symptoms of MS include cognitive impairments, loss of vision, tremors, spasticity, pain, and fatigue. 4
There is considerable interest in physical activity (PA) participation as an outcome of MS and a behavior with secondary benefits for managing the consequences of the disease. There is consistent evidence demonstrating that individuals diagnosed with MS engage in less PA than healthy controls (HCs). 5 Further, there is a growing body of literature examining the potential therapeutic effects of PA in persons with MS. PA is beneficial in managing MS symptoms 6 and provides protection against visual field loss. 7 There is less known about PA and its association with axonal and neuronal integrity, particularly with relevance for protection against vision loss in MS.
One previous study has reported a positive association between device-measured steps/day and both total macular volume (TMV) and retinal nerve fiber layer (RNFL) thickness measured through an optical coherence tomography (OCT) device in a sample of 84 people with MS. 8 However, that initial study had key limitations including the lack of a HC group, and the use of an outdated OCT device. The OCT device (Zeiss Stratus OCT 3, Carl Zeiss Meditec, Dublin, CA) used by Sandroff et al has been utilized in previous studies 9 and shown to be reliable. 10 However, as a time-domain OCT, the depth of the retina is collected as a function of time through a moving reference mirror. 11 In contrast, many of the newer OCT device models utilize a spectral domain which instead utilizes a fixed mirror and a Fourier transformation to further decompose the light spectrum based on spatial and temporal frequency thereby allowing higher OCT scan rates, greater depth penetration, and improved sensitivity. 12
The present study builds on the work by Sandroff et al 8 by utilizing improved techniques to measure retinal morphology through OCT and PA through accelerometers. The present work primarily focuses on the average thickness of the RNFL and TMV as these measurements have been suggested to reflect axonal and neuronal integrity, respectively.13,14 Similarly, whereas the previous study examined device-measured steps per day, the present study focuses on moderate-to-vigorous physical activity (MVPA) through previously reported cut-points in individuals with MS. 15 Although steps-per-day 16 is a validated metric in adults with MS, it does not quantify the intensity of the PA. Further, much of the current literature has reported a dose-dependent response with greater benefits at MVPA 17 and reported a quality of evidence sufficient enough to recommend PA as minutes of MVPA in the Physical Activity Guidelines for Americans. 18 Thus, examining objectively measured MVPA provides the benefit of quantifying PA at a specific intensity that is more commonly utilized.
Accordingly, the present study assessed differences in MVPA and retinal morphology among persons with MS and age and sex-matched HCs, and then examined the association between MVPA and retinal health. The hypotheses of this study were as follows: (1) persons with MS will engage in less MVPA and have reduced RNFL thickness and TMV compared with HCs; (2) MVPA will be positively associated with RNFL thickness and TMV; and (3) MVPA will account for the group differences in RNFL thickness and TMV between MS and HCs.
Methods
Participants
Secondary data collected from 82 adults with MS (age: 59.6 ± 9.2 years) and 82 HCs (age: 59.5 ± 9.1 years) from 2 published studies were utilized.19,20 Of the original sample, participants were excluded if either accelerometer (N = 39) or OCT (N = 5) data were invalid or missing. Both studies and associated experimental procedures were approved by the University of Illinois at Urbana-Champaign Institutional Review Board and were conducted in accordance with the use of human participants in research as outlined in the Declaration of Helsinki. Briefly, participants were recruited through emails from a list of individuals diagnosed with MS in Illinois gathered from MS-related events, a database of previous research participants, and advertisements on the Greater Illinois chapter of the National Multiple Sclerosis Society either at the University of Illinois or the East and Central Illinois area. Both individuals with MS and HCs were excluded if they had been diagnosed with any neurodegenerative disease other than MS, age-related macular degeneration and other macular pathologies, glaucoma, were currently smoking or were unable to walk with or without assistance.
Moderate-Vigorous Physical Activity
MVPA (minutes per day) was measured through waist-worn accelerometers (ActiGraph, model GT3X+; ActiGraph LLC, Pensacola, FL). Participants were instructed to wear accelerometers over the non-dominant hip secured with an elastic band for a 7-day period. Raw data were reduced using ActiLife6 software through a low-frequency extension and divided into 60-second epochs. MVPA cut-points were derived from previous literature 15 in which the minute increments were designated as MVPA if the counts per minute exceeded 1584 and 1950 for individuals with MS and HCs, respectively. All participants with 2 or more valid days of accelerometer data were included in the analyses.21 -23
Retinal Morphometry
Macular RNFL thickness and TMV were assessed via data extracted from retinal images collected with the Heidelberg Engineering Spectralis, Spectral-Domain OCT (SD-OCT; Heidelberg Engineering). As previously described, this study utilized a low-coherence 870 nm laser through a super luminescence diode to capture the layers of the retina. 20 Retinal outcomes were obtained from central, inner, and outer rings with respective diameters of 1, 3, and 6 mm around the fovea and combined. Volume and thickness were quantified using Heidelberg software (version: 6.0.110). Layers were partitioned by trained researchers through auto-generated segmentation lines followed by manual corrections referencing a standardized image to account for incorrectly generated lines and/or blood vessels. Variables were quantified using the average between the 2 eyes. Values were taken from single eye measurements if the value for 1 eye was either missing or exceeded 2 standard deviations from the mean (N = 8).
Statistical Analyses
Data were analyzed using SPSS Statistics version 26 (IBM, Somers, NY) and alpha levels of P = .05 were specified to indicate statistical significance. Normality was assessed using the Shapiro–Wilk test, skewness, and kurtosis with visual confirmation through Q-Q plots, histograms, and boxplot analysis. MVPA was transformed using a cube root transformation. The differences between MS and HCs in age, sex, ethnicity, and BMI were examined using independent samples t-tests or chi-square tests when appropriate. TMV, RNFL thickness, and MVPA comparisons were conducted through independent samples t-tests with the magnitude of differences between groups expressed through Cohen’s d. Values of 0.2, 0.5, and 0.8 were designated as small, moderate, and large group differences, respectively. 24 Due to the difference in age between groups and the literature supporting age-related macular morphology changes,25,26 age was included in all subsequent analyses as a possible confounding variable. The correlations between age, MVPA, TMV, and RNFL thickness in the overall sample were examined through bivariate Pearson correlation analysis. Finally, linear regression analysis in which variables were directly entered in steps were conducted to assess the contribution of MVPA to explaining variance between groups in retinal morphology. Briefly, TMV and RNFL thickness were regressed on Group (MS and Control) in Step 1 followed by the addition of age in Step 2 and MVPA in Step 3. Model fit was determined through R2 and the relative strength of each variable in the model was determined through standardized β-coefficients. Both change in R2 (ΔR2) with β-coefficients were examined to identify the independent contributions of Group, age, and MVPA to the model.
Results
Participant Characteristics
Table 1 presents the characteristics of the participants with complete PA and retinal data. Upon excluding individuals with missing or invalid data, the MS (n = 41) and HC (n = 79) groups did not significantly differ by sex (χ2 = 1.49, P = .22), race (χ2 = .08, P = .78), or BMI (t = 0.08, P = .94), but differed in age (t = −2.53, P = .01).
Demographic and Clinical Characteristics of Individuals With MS and HCs.
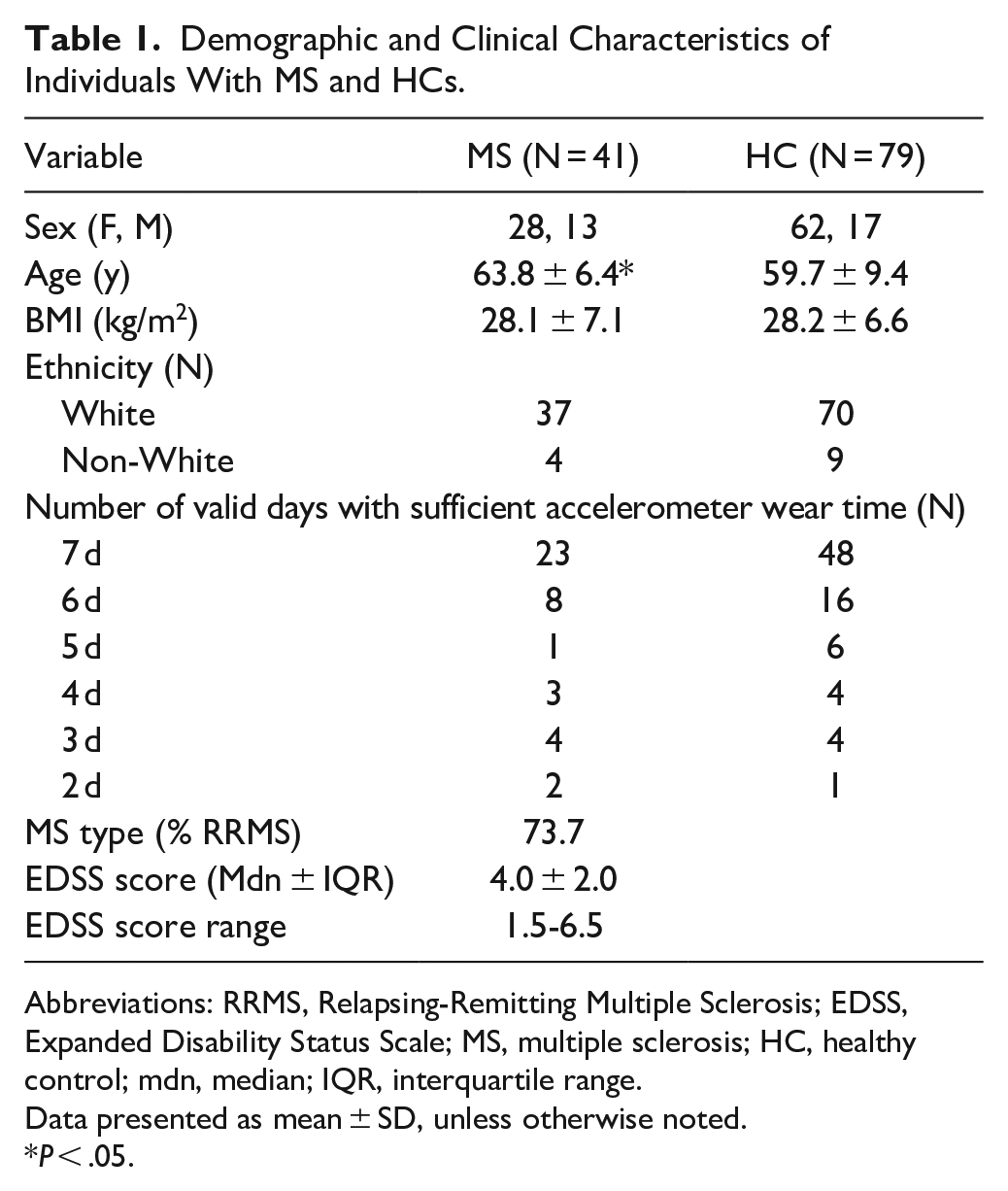
Abbreviations: RRMS, Relapsing-Remitting Multiple Sclerosis; EDSS, Expanded Disability Status Scale; MS, multiple sclerosis; HC, healthy control; mdn, median; IQR, interquartile range.
Data presented as mean ± SD, unless otherwise noted.
P < .05.
Group Differences
Table 2 presents the descriptive statistics for MVPA, RNFL thickness, and TMV for the MS and HC groups. The MS group had significantly lower MVPA (t = 6.6, P < .01), RNFL thickness (t = 4.69, P < .01), and TMV (t = 4.82, P < .01) than HCs. Group differences were large in magnitude for MVPA (d = 1.17), RNFL thickness (d = 0.98), and TMV (d = 0.99).
Descriptive Characteristics and Statistical Analyses of Retinal Morphology and PA in Individuals With MS and HCs.
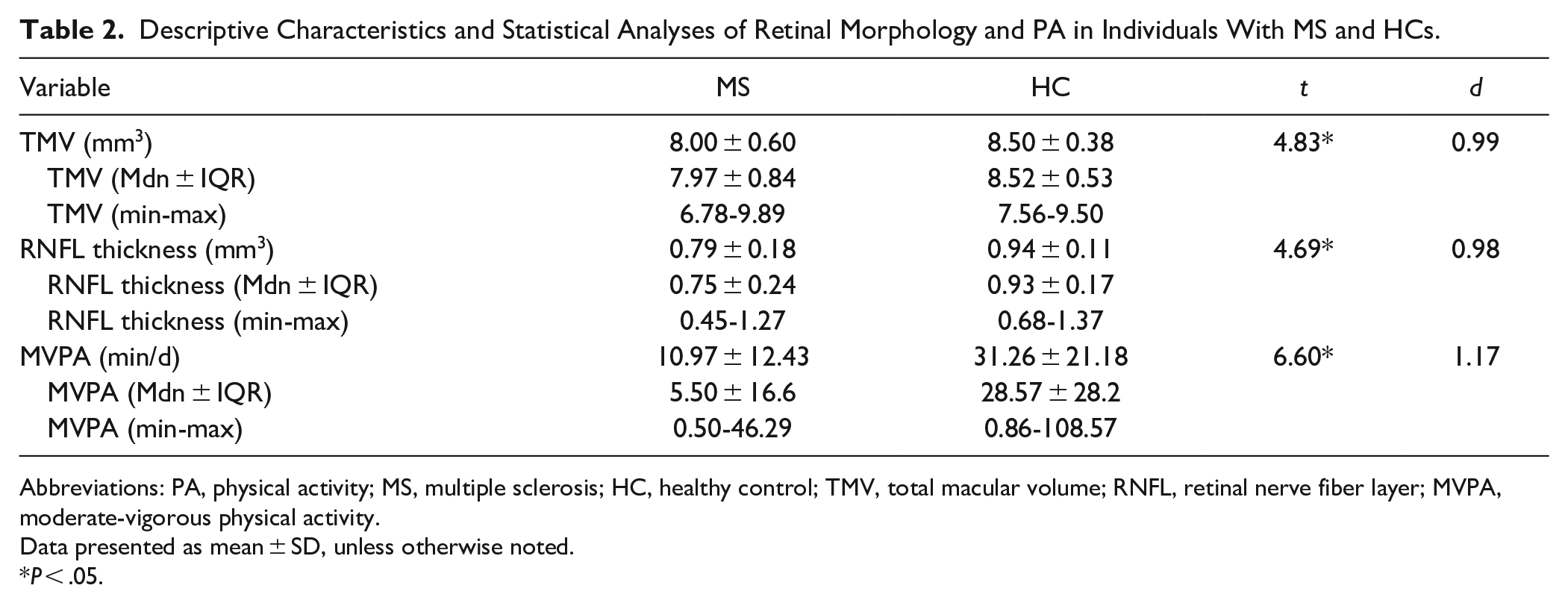
Abbreviations: PA, physical activity; MS, multiple sclerosis; HC, healthy control; TMV, total macular volume; RNFL, retinal nerve fiber layer; MVPA, moderate-vigorous physical activity.
Data presented as mean ± SD, unless otherwise noted.
P < .05.
Bivariate Correlations
MVPA was skewed (Shapiro–Wilk = 0.90, Skewness = 1.25, Kurtosis = 2.19), whereas TMV (Shapiro–Wilk = 0.98, Skewness = −0.34, Kurtosis = 0.72) and RNFL (Shapiro–Wilk = 0.98, Skewness = −0.10, Kurtosis = 0.80) were normally distributed. Upon cube root transformation, MVPA was normally distributed (Shapiro–Wilk = 0.97, Skewness = −0.26, Kurtosis = −0.63). Age was associated with MVPA (r = −.22, P = .02) and TMV (r = −.20, P = .03), but not RNFL thickness (r = .01, P = .90), and MVPA was positively associated with both TMV (r = .49, P < .01) and RNFL thickness (r = .38, P < .01) when collapsed across groups.
Regression Analyses
Table 3 presents a summary of the results for the linear regression analyses examining MVPA as a mediator for group differences in retinal morphology. Group (coded as 1 = MS and 0 = HC) explained 21% of the variance in TMV (β = −.45; P < .01). The inclusion of age (β = −.10; P = .23) in Step 2 of the model did not improve the model and accounted for 0.1% of the variance. The addition of MVPA (β = .34; P < .01) in Step 3 of the model accounted for an additional 8% of TMV variance. The effect of Group was attenuated with the addition of MVPA in Step 3 (Group β Step 1 = −.45; P < .01, Group β Step 3 = −.25; P < .01), and both Group and MVPA were significant correlates of TMV.
Summary of Stepwise Linear Regression Analyses of PA and Retinal Morphology in Individuals With MS and HCs.
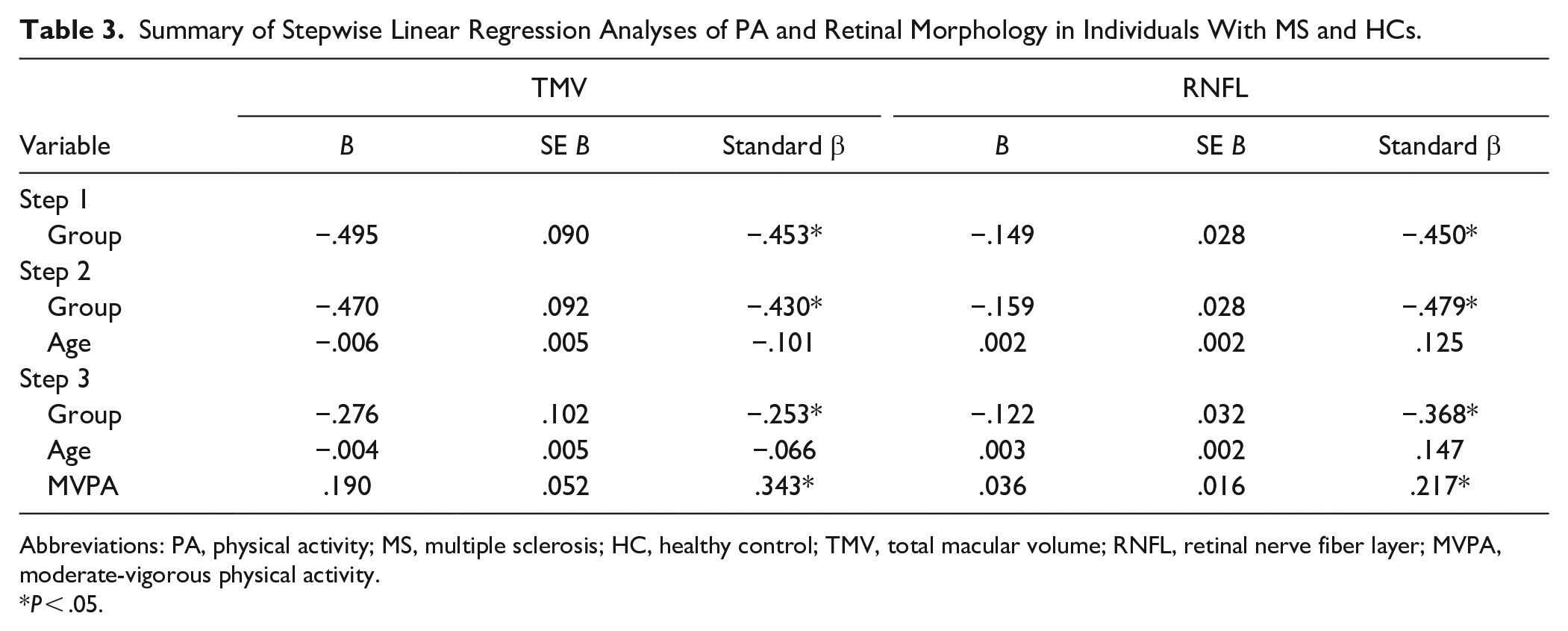
Abbreviations: PA, physical activity; MS, multiple sclerosis; HC, healthy control; TMV, total macular volume; RNFL, retinal nerve fiber layer; MVPA, moderate-vigorous physical activity.
P < .05.
Group explained 20% of the variance in RNFL thickness (β = −.45; P < .01) with the inclusion of age (β = .13; P = .15) in Step 2 accounting for 1% of the variance and the addition of MVPA (β = .22; P = .03) in Step 3 of the model explaining 3% of the variance in RNFL thickness. The effect of Group was attenuated with the addition of MVPA in Step 3 (Group β Step 1 = −.45; P < .01, Group β Step 3 = −.37; P < .01) and both MVPA and Group were significant correlates of RNFL thickness.
Discussion
The present study examined the association between MVPA and retinal health for adults with MS and HCs. This study reported significant differences in MVPA, RNFL thickness, and TMV between groups, and further observed that MPVA was associated with both RNFL thickness and TMV and partially accounted for group differences in both RNFL thickness and TMV. These results provide additional support for future research examining lifestyle interventions that target PA for improving integrity of the CNS (ie, neuroprotection) in persons with MS.
Consistent with the previous literature, individuals with MS had lower MVPA, 27 RNFL thickness, and TMV 28 compared with HCs. Furthermore, MVPA was positively associated with both TMV and RNFL. This is consistent with the current body of work as previous studies have reported a positive association between PA and retinal morphology in persons with MS 8 and HCs. 29 Importantly, this study provides evidence that the association between MVPA with TMV and RNFL are sustained when controlling for MS diagnosis. Specifically, higher MVPA was associated with larger values of 0.19 and 0.04 mm3 for TMV and RNFL thickness, respectively, upon controlling for MS diagnosis and age. This suggests that MVPA may provide retinal benefits for both individuals with and without MS.
The possibility that MVPA may benefit retinal integrity has important implications as retinal degeneration has been associated with a lower quality of life and greater emotional distress. 30 Future research using longitudinal or intervention designs is necessary to understand the exact relationship between MVPA and retinal health, yet these findings provide promising preliminary results. Examining the effects of increased MVPA in the MS population particularly important as individuals with MS to understand full effects. As individuals with MS often experience greater retinal degeneration, 2 it is a distinct possibility that these individuals may experience greater emotion distress which may lead to greater systemic inflammation 31 that may further exacerbate the symptoms of MS. 32
Future studies should consider examining potential mechanisms through which MVPA may benefit neuronal and axonal integrity in individuals with MS. Previous studies have suggested brain-derived neurotrophic factor (BDNF) and/or ocular pressure to be associated with retinal health. 33 Indeed, a growing body of literature supports the association between PA and BDNF levels, 34 and BDNF has been suggested to aid in retinal recovery. 35 Additionally, PA has been associated with decreased intraocular and increased ocular perfusion pressures which may increase blood flow to maintain healthy retinal morphology and function. 36
The findings of this work extend our knowledge on the association between PA and retinal health for healthy individuals and individuals with MS, but there are several limitations that should be considered. First, as this study represents a cross-sectional analysis, it cannot be presumed that habitual PA causally influences retinal morphology. In order to address this limitation, randomized control trials and longitudinal studies examining the changes in retinal morphology as a result of changes in PA are required. Finally, dietary patterns or nutritional intake were not accounted for in the present sample. As dietary patterns 37 have been suggested to ameliorate the symptoms and disabilities associated with MS and specific molecules, such as lutein and zeaxanthin, have been reported to protect again age-related macular degeneration by ameliorating oxidative stress, 38 the retinal results may have been partially influenced by participant’s diet. These limitations notwithstanding, this study is the first to report that MVPA is associated with axonal and neuronal integrity among individuals with and without MS through objective and high-resolution techniques.
A growing body of literature supports the benefits of PA on the onset and severity of the symptoms of MS. However, the dose-dependent benefits remain unknown. The work presented in this study provides important evidence demonstrating a potential link between MVPA on neuronal and axonal integrity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the University of Illinois Paul D. Doolen Graduate Scholarship for the Study of Aging and the National Multiple Sclerosis Society.
