Abstract
Lesion load of the corticospinal tract (CST-LL), a measure of overlap between a stroke lesion and the CST, is one of the strongest predictors of motor outcomes following stroke. CST-LL is typically calculated by using a probabilistic map of the CST originating from the primary motor cortex (M1). However, higher order motor areas also have projections that contribute to the CST and motor control. In this retrospective study, we examined whether evaluating CST-LL from additional motor origins is more strongly associated with post-stroke motor severity than using CST-LL originating from M1 only. We found that lesion load to both the ventral premotor (PMv) cortex and M1 were more strongly related to stroke motor severity indexed by Fugl-Meyer Assessment cut-off scores than CST-LL of M1 alone, suggesting that higher order motor regions add clinical relevance to motor impairment.
Introduction
The corticospinal tract (CST) is the primary descending white matter pathway that supports voluntary motor function. Measures of CST damage have been related to motor outcomes both cross-sectionally and longitudinally following stroke, where greater damage to the CST results in worse motor performance. 1
CST lesion load (CST-LL), the amount of overlap between the lesion and the CST, is calculated using probabilistic maps of CST fibers originating from the primary motor cortex (M1). However, roughly fifty percent of CST inputs are from other higher order motor areas. 2 Injury to tracts originating from these areas may contribute to post-stroke motor deficits. Measuring injury to these additional tracts has the potential to provide a better understanding of stroke, a more accurate prediction of treatment gains, and improve stratification of patients for clinical trials.
One notable exception to the traditional approach to M1-CST studies is Riley et al., 2011. 3 The authors examined how tract-specific injury predicted behavioral gains made in therapy measured by changes in Fugl-Meyer Assessment (FMA) scores before and after therapy. They found that damage to tracts originating from M1, dorsal premotor (PMd), and supplementary motor area (SMA) was correlated with changes in FMA, a measure widely used to capture sensorimotor impairment after stroke. However, it remains unclear if damage to CST tracts originating from different regions is a stronger indicator of motor scores than damage to the traditional M1-CST only.
Here, we assessed whether CST-LL from various motor origins can better explain motor performance than M1-CST alone. Because FMA cut-off scores are routinely used to stratify research subjects and define clinical outcomes, 4 we used a three-class classification scheme from Woytowicz and colleagues 5 to examine the relationship between CST-LL to various motor tracts and post-stroke motor severity.
Methods
We used 138 T1-weighted anatomical MRIs of individuals with stroke and corresponding lesion masks from the open-source ATLAS database, for which we obtained FMA scores from five cohorts retrospectively.
6
MRIs and corresponding lesion masks were registered to MNI152 space with ANTs (http://stnava.github.io/ANTs/). Ipsilesional lesion load was calculated using the PALS
7
toolbox as the percentage of overlap between each registered lesion mask and each region of interest (ROI). Six CST ROIs were obtained from SMATT
8
templates, with origins from M1, S1, SMA, preSMA, PMv, and PMd (Figure 1A). (A) Six CST templates from the SMATT templates were used to calculate lesion load. (B) Correlogram of correlations between each pair of ROI and Fugl-Meyer scores (FMA_UE), where blue indicates negative correlations and red indicates positive correlations, and smaller circles indicate smaller correlations.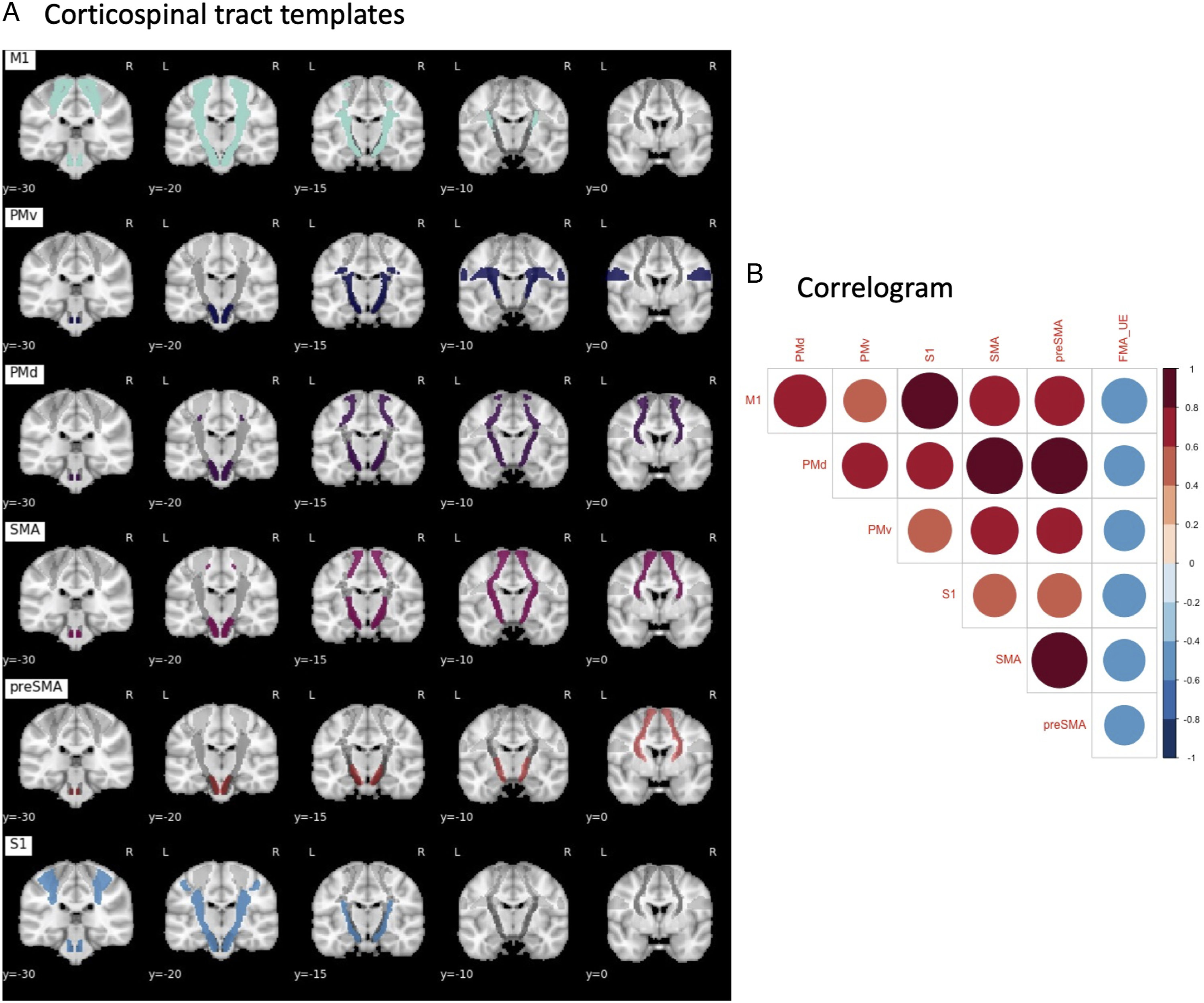
We first calculated pairwise correlations between and among tract origins and FMA scores (corrected p < .0083). FMA scores were categorized according to cut-offs from Woytowicz and colleagues 5 (mild: 42-60, medium: 28-41, and severe: 0-27). Because we found a high degree of multicollinearity as indicated by the variance inflation factor (VIF) on a linear model with all six ROIs, we used a lasso-regularized multinomial classifier with cross-validation to perform variable selection (Supplementary Materials). The resulting sparse model was refit to assess the significance of the retained tracts. To examine whether the effects of lesion load were driven by lesion volume, total lesion volume was added as a covariate to the sparse model containing only significant tracts. Finally, we compared nested models (M1 CST-LL only, the selected sparse model, M1 CST-LL with each selected tract in the sparse model, and the model including lesion volume) using likelihood ratio tests (LRT).
Results
Percentage Overlap Between Corticospinal Tract Templates.
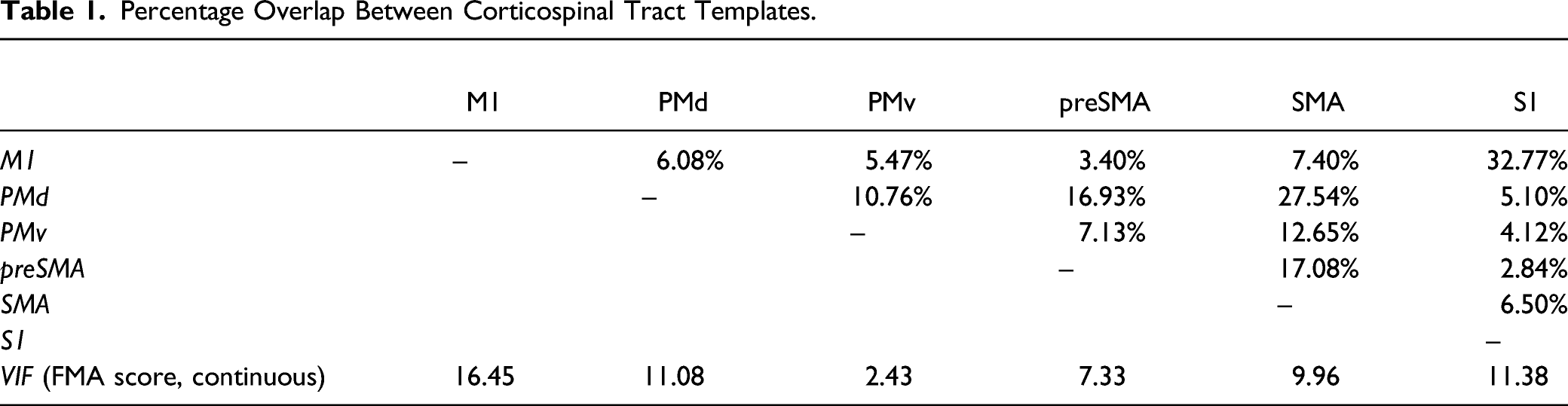
FMA classification groups were imbalanced and accounted for in the tuning of the model (Supplementary Materials; mild = 77, moderate = 21, and severe = 40). Multinomial lasso retained CST-LL originating from M1, PMv, and SMA. VIF for the sparse model was as follows: VIFM1 = 2.11, VIFSMA = 2.64, and VIFPMv = 1.78.
Sparse Model Selected from Multinomial Logistic Lasso.

Values show regression coefficients. AIC: 235.54, Residual deviance = 219.54.
LRT comparing the M1 only and the sparse model was significant (χ2(2) = 10.94, p = .03), as was LRT comparing the M1 only and M1 + PMv models (χ2(2) = 9.59, p = .008; VIFM1, PMv =1.39), indicating that the models that contained both M1 and PMv were a better fit than the models with M1 only. LRT comparing M1 only to a model with M1 + SMA was not significant (χ2(2) = 5.07, p = .08; VIFM1, SMA = 2.08).
Total lesion volume was not significantly associated with the FMA group (p > 0.5) and did not result in a better fit compared to the final M1 + PMv model (χ2(2) = 1.40, p = .50).
Discussion
Here, we found that CST-LL to PMv and M1 classified FMA groups better than M1 alone and that these results were not driven by total lesion volume. Riley and colleagues 3 previously reported that injury to PMv played a smaller role in supporting treatment gains as compared to other tracts (M1, PMd, and SMA). This discrepancy could be due to demographic differences between the two samples, such as FMA scores, therapy received, and time since stroke. Additionally, the findings in Riley et al. were based on correlations between CST-tracts and motor scores, and we also found that damage to the PMv was least correlated to FMA compared to other tracts. This is likely because the PMv had the lowest multicollinearity with other tracts. That is, while other tracts shared redundant information, PMv provided additional information about the FMA. Although speculative, this makes sense in light of the roles of these areas: M1, SMA, preSMA, PMd, and S1 are all involved in the preparation and execution of movements.2,9 PMv alone is involved in positioning and sequencing of hand movements for precision grasping, and has unique frontal and parietal anatomical connections. 10 Limitations in this study include the cross-sectional, multi-site retrospective study design, which could be susceptible to cohort effects and have limited generalizability. However, this study provides preliminary evidence that inclusion of CST-LL from the PMv in addition to M1 may be clinically relevant for explaining post-stroke motor outcomes.
Supplemental Material
sj-pdf-1-nnr-10.1177_15459683211068441 – Supplemental Material for Corticospinal Tract Lesion Load Originating From Both Ventral Premotor and Primary Motor Cortices Are Associated With Post-stroke Motor Severity
Supplemental Material, sj-pdf-1-nnr-10.1177_15459683211068441 for Corticospinal Tract Lesion Load Originating From Both Ventral Premotor and Primary Motor Cortices Are Associated With Post-stroke Motor Severity by Kaori L. Ito, Bokkyu Kim, Jingchun Liu, Surjo R. Soekadar, Carolee Winstein, Chunshui Yu, Steven C. Cramer, Nicolas Schweighofer, and Sook-Lei Liew in Neurorehabilitation and Neural Repair
Footnotes
Declaration of Conflicts of Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SC serves as a consultant for Abbvie, Constant Therapeutics, MicroTransponder, Neurolutions, SanBio, Panaxium, NeuExcell, Elevian, Medtronic, and TRCare. There are no other conflicts of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH R01HD059783-06 to CW and NIH R01NS115845, K01HD091283 to S-LL.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
