Abstract
Despite an increase in the amount of published stroke recovery research, interventions have failed to markedly affect the trajectory of recovery poststroke. We argue that early-phase research to systematically investigate dose is an important contributor to advance the science underpinning stroke recovery. In this article, we aim to (a) define the problem of insufficient use of a systematic approach to early-phase, multidimensional dose articulation research and (b) propose a solution that applies this approach to design a multidimensional phase I trial to identify the maximum tolerated dose (MTD). We put forward a design template as a decision support tool to increase knowledge of how to develop a phase I dose-ranging trial for nonpharmaceutical stroke recovery interventions. This solution has the potential to advance the development of efficacious stroke recovery interventions, which include activity-based rehabilitation interventions.
Introduction
Effective nonpharmaceutical stroke recovery interventions (including activity-based rehabilitation interventions) contain multiple interrelated components that collectively address a specific poststroke impairment. 1 Dose is one of these components.2,3 Early-phase trials can systematically investigate dose during intervention development—that is, preclinical experiments through to phase II clinical trials. These trials can be unidimensional, manipulating only 1 dose dimension while keeping others fixed, or they can be multidimensional, manipulating more than 1 dose dimension within the same dose articulation trial. The purpose of early-phase dose articulation research is to establish a clear justification and mechanistic rationale for dose selection in a phase III trial.4,5 This research approach would provide confidence that the dose is safe and has potential to be clinically relevant. In stroke recovery, the completion of early-phase trials to articulate the dose of a nonpharmaceutical intervention is limited. 6 Increasing the uptake of early-phase trials may help address the neutral findings in stroke recovery.7-9
There are 2 important concepts that guide the adoption of early-phase dose articulation research: a systematic discovery pipeline5,10 that informs the dose articulation process and a multidimensional dose framework 11 that conceptualizes the dimensions of dose. Both are briefly outlined below.
Systematic Discovery Pipeline
For a new pharmaceutical intervention to be approved by regulatory bodies11,12 it needs to be safe and have a clinically relevant effect. 12 Establishing a tolerable dose range is required—selecting a dose too high could have safety implications, whereas selecting a dose too low may not produce a clinically relevant effect. Early-phase dose articulation trials are routinely implemented to investigate the tolerable dose range. These trials identify the maximum tolerated dose (MTD) via safety (eg, toxicity levels) and tolerability outcomes before investigating for efficacy,10,13 with the outcome of prior studies contributing to the rationale for future studies. Developing a systematic approach to nonpharmaceutical dose articulation research, adapted from the one routinely implemented in pharmaceutical trials,10,13 is challenging. We assert that this will be a critical step in the development of a safe and clinically effective stroke recovery intervention.
An opportunity exists to learn from the success of early-phase pharmaceutical trials to systematically determine a safe and tolerable dose of a nonpharmaceutical stroke recovery intervention, which cannot be assumed to be safe. 14 Dobkin 15 highlights that recovery trials have tended to “move forward in random increments compared to the Food and Drug Administration process for drug approval.” The justification for dose selection is often based on clinical expertise or false pragmatism.16,17 Several neutral phase III randomized control trials7,8 also recommend the completion of high-quality, early-phase dose articulation trials in their discussion. To guide the uptake of nonpharmaceutical early-phase trials, we can adapt a previously defined systematic discovery pipeline 5 (Figure 1). This pipeline approach is based on the principles and clinical trial phases set out by regulatory bodies10,13 for clinical research and is routinely implemented in pharmaceutical development.18,19 The pipeline aims to complement existing nonpharmaceutical intervention development tools20,21 and address an international consensus recommendation that testing dose needs to be systematic. 3
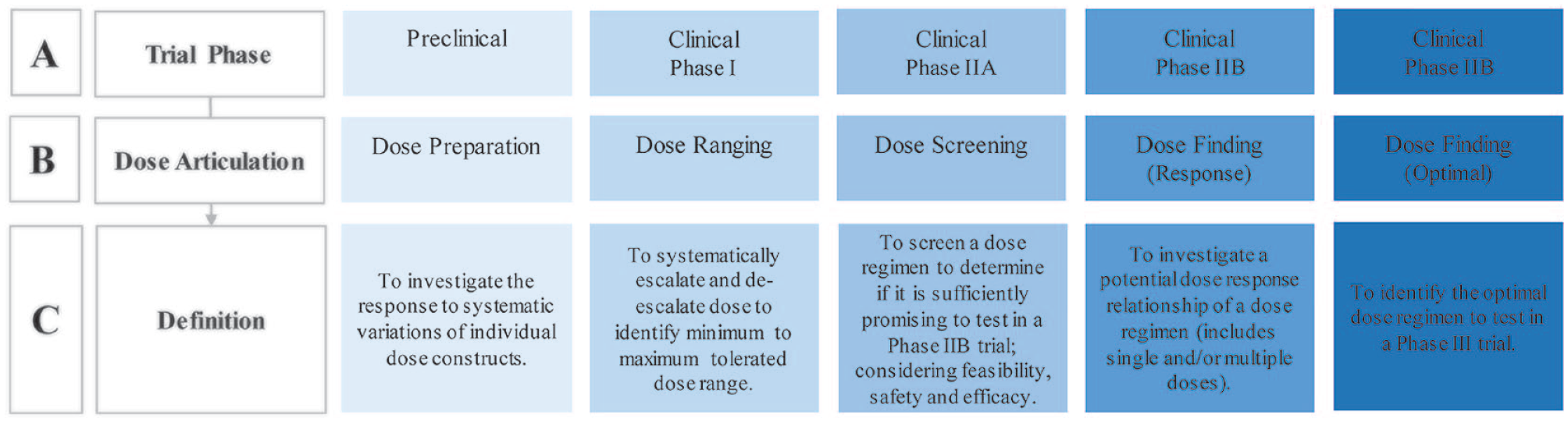
Systematic discovery pipeline 5 for nonpharmaceutical dose articulation trials. Panel A: Trial phases included in early-phase research, from preclinical through to clinical phase IIB as per regulatory bodies.10,13 Panel B: The type of dose articulation that occurs at each phase. Panel C: Definition of the dose articulation purpose at each phase.
Multidimensional Dose Framework
There are similarities between pharmaceutical and nonpharmaceutical doses, including the ability to vary dimensions (eg, schedule, intensity) within a prescribed dose regimen. Pharmaceutical interventions use the unidimensional measure of amount of a drug (eg, in milligrams) to determine a safe and tolerable dose. 18 Nonpharmaceutical interventions do not have an equivalent measure and, therefore, need to articulate all dimensions to establish a safe dose range. To make this possible, we can apply a previously defined multidimensional dose framework 11 (Figure 2). This framework needs to be carefully contextualized to the intervention being investigated in a specific early-phase trial, which will in turn inform the selection of dose dimensions to articulate. 11 For example, repetitions may best reflect episode intensity for an upper limb trial, whereas heart rate reserve may be most appropriate for an exercise trial. In this way, the framework enables transparent description, implementation, monitoring, and reporting of dose in early-phase nonpharmaceutical stroke recovery trials. 11
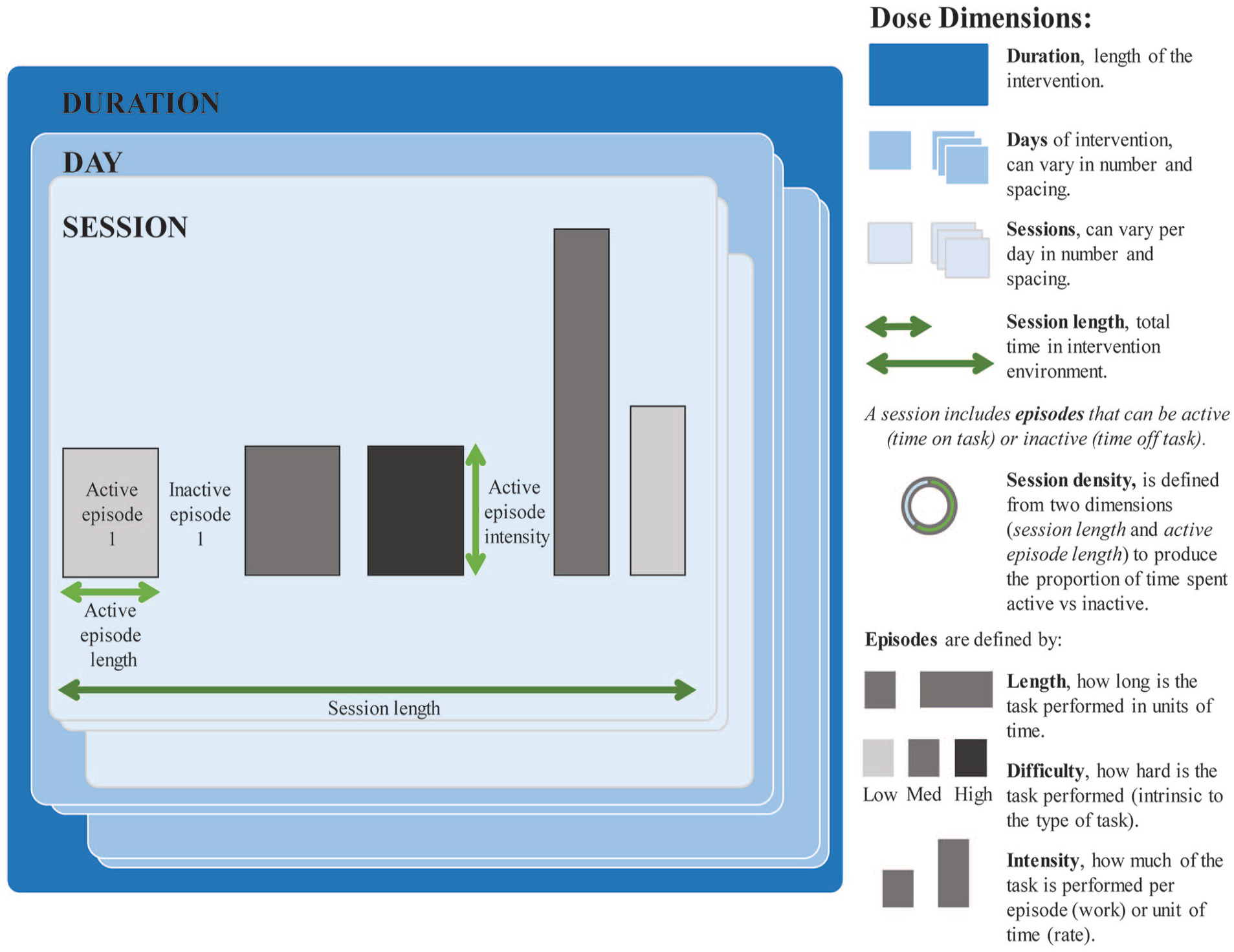
Multidimensional dose framework 11 for application to nonpharmaceutical stroke recovery interventions.
Objective
The scope of this point-of-view article is to investigate early-phase dose articulation trials, with a specific focus on phase I dose ranging trials for nonpharmaceutical stroke recovery interventions. The purpose of a phase I trial is to determine the safe and feasible dose range defined by the MTD. Correct identification of such a range provides the most informed foundation for subsequent selection of the optimal dose, based on considerations of efficacy or effectiveness. Our objectives are to:
Define the problem of insufficient use of a systematic approach to early-phase, multidimensional dose articulation research, and
Propose a solution that applies this approach to design a multidimensional phase I trial to identify the MTD.
We use upper limb motor research poststroke to illustrate the main points because there is a sufficient body of preclinical and clinical stroke recovery research 3 to inform our discussion. We acknowledge that there are other elements integral to intervention development, such as careful selection of the study population (eg, participants), intervention quality (eg, prescription of feedback, fidelity), and pragmatic considerations (eg, cost). These elements are not the focus of this article and are addressed by other intervention development tools (eg, Template for Intervention Description and Replication checklist 21 ). As such, the information presented in this article specifically targets the notion of dose and should be used in conjunction with previously defined tools.20,21 Common key terms are listed in a glossary in Supplement File 1.
Problem
Systematic Discovery Pipeline
Limited Adherence to the Principles of the Systematic Discovery Pipeline for Early-Phase Stroke Recovery Trials
The systematic articulation of dose as a key component to investigate during intervention development is an international consensus recommendation for stroke recovery.1,3 A scoping review 6 of early-phase dose articulation trials for stroke recovery, specifically targeting motor interventions, demonstrates that a systematic discovery pipeline 5 approach is not consistently adopted. The majority of studies from the review progress straight to a phase IIA/B trial without building on appropriate preclinical evidence or completing a phase I trial. 6
The best example of a program of upper limb research that adopts some principles from the discovery pipeline 5 will inform the following discussion. We will explore the design and dose justification process for the 4 trials within the program of research (Figure 3). We acknowledge that these trials are not published as a sequential program of research by the authors, and at the time of their design, the systematic discovery pipeline 5 addressing dose articulation studies had not been presented. The function of this example is to highlight the importance of completing adequate early-phase dose articulation trials, especially phase I, to inform the development of later-phase trials, thus providing the best foundation for success while concurrently minimizing research waste.

Upper limb program of research mapped to systematic discovery pipeline. 5 The vertical axis denotes discovery pipeline phase (preclinical through to clinical phase IIB), and the horizontal axis denotes the upper limb trials by publication date. Preclinical dose justification denotes the preclinical experiments referenced by the trial to justify their dose selection. Clinical dose justification denotes the clinical studies referenced to by the trial to justify their dose selection.
A phase IIA dose screening trial published in 2010 by Birkenmeier et al 22 is the first trial outlined in Figure 3. This trial tests the feasibility of completing ≥300 repetitions in a 60-minute outpatient session. The authors’ dose justification is based on preclinical experiments,23-25 which indicate that hundreds of daily repetitions are required for neural adaptations in animal models. Alongside their preclinical justification, clinical studies are referenced, which highlight a correlation between higher repetitions and positive brain changes.26-28 The dose justification within this trial is important because this rarely occurs in nonpharmaceutical stroke recovery trials. 4 The missing piece informing this trial is a phase I dose ranging trial that would allow the dose tested to be selected from a safe and tolerable range.
A subsequent phase IIA dose screening trial by Waddell et al 29 in 2014 is the second trial outlined in Figure 3, drawing justification from the Birkenmeier et al 22 protocol. This trial aims to test the same dose (≥300 repetitions) with stroke patients in the early subacute phase 29 as compared with the chronic phase. 22 Purposefully sampling participants from similar recovery epochs when conducting early-phase trials is important. 30 The translation of the Birkenmeier et al 22 protocol to the Waddell et al 29 trial is a systematic step within the same phase of the discovery pipeline 5 (Figure 3).
Birkenmeier et al 22 phase IIA trial also informs the dose justification for a phase IIB trial with chronic stroke patients completed by Lang et al 31 in 2016. This dose finding response trial (see Figure 1) evaluates 4 different doses and their impact on upper limb outcomes: 3200 versus 6400 versus 9600 versus individual maximum at greater than 9600 repetitions. The lowest dose (3200) is justified from usual care observational studies that count upper limb repetitions32,33 and the Birkenmeier et al 22 trial outcome. The translation of findings from phase IIA to phase IIB (to inform the lowest dose) is appropriate as per the discovery pipeline principles. 5 No justification, however, is provided for the decision to select the remaining doses, which are achieved by doubling and tripling the lowest dose. Conducting a phase I dose ranging trial could have provided a safe and tolerable dose range in which to systematically select the remaining doses to test. The phase IIB trial reports a moderate change in motor function overall but a neutral dose response outcome, with no difference in efficacy outcomes between dose groups. 31
The final study is a phase I dose ranging trial by Colucci et al 34 published in 2017. This trial steps back to the first clinical trial phase of the discovery pipeline 5 (Figure 1). The primary objective is to develop a rule-based dose-finding design, with a secondary objective to determine if a dose-finding design can identify the MTD of exercise-based therapy for chronic stroke patients. The justification for the starting dose (50 repetitions) is drawn from preclinical experiments35-40 and the 2 phase IIA trials discussed.22,29 The results suggest that a phase I design is feasible and able to identify the MTD; however, the authors strongly caution against the application of results to clinical practice given the primary objective of a phase I trial. The MTD is 209 repetitions; therefore, the safe and tolerable dose range is between 50 repetitions (starting dose) and 209 repetitions (MTD) per day. 34 It is difficult to compare these results to the average 322 repetitions tolerated by chronic stroke patients in the Birkenmeier et al 22 trial because the type of intervention and how it is delivered differ. A unidimensional approach (repetition goal) to dose articulation is also present in both studies, which may challenge the capacity to reach tolerability. Colucci et al 34 identify difficulties with monitoring intervention adherence and trial stopping rules that provide important learnings to develop future phase I stroke recovery trials. This trial also highlights the importance of keeping items (eg, what type of intervention or who delivers the intervention) from the Template for Intervention Description and Replication (TIDieR) 21 consistent as you move along the systematic discovery pipeline, 5 so evidence from one phase can optimally inform another phase.
The main gap from this program of research is the lack of an initial phase I trial, which would have ensured the dose(s) tested in subsequent trials were based on a safe and tolerable range (minimum through to MTD). A lack of funding opportunities as well as a lack of understanding of how to design and conduct a phase I trial for a nonpharmaceutical stroke recovery intervention may contribute to this outcome.
Few Phase I Stroke Trials Are Funded
We have established that there is a lack of published phase I dose ranging trials in early-phase nonpharmaceutical stroke recovery research. 6 To understand whether phase I trials are consistently neglected across all stroke research (pharmaceutical and nonpharmaceutical), a search of peak national government funding agencies (between calendar years 2017-2019) in Australia 41 and the United States 42 can demonstrate the number of funded phase I trials. Supplement File 2 outlines the search process for each funding body and the results. In summary, a total of 6487 projects have received funding across Australia and the United Sates, 9% of which is stroke related. Of these projects, only 2 phase I trials, both targeting a pharmaceutical intervention, have received funding. The Australian call for funding states that the intended outcomes should “provide reliable evidence of the effects of health-related interventions.” 43 The US funding call is similar with projects needing to “evaluate the effects of interventions on health-related biomedical outcomes.” 44 The purpose of a phase I trial is to identify the MTD to inform later-phase efficacy trials. The focus of funding calls is for trials of intervention efficacy. This could be contributing to the small number of phase I trials being funded and thus completed. Increasing funding opportunities for early-phase research is a critical step to progress the adoption of phase I trials in practice.
Limited Phase I Stroke Trials Are Registered
A search of the Australian and New Zealand Clinical Trial Registry 45 and ClinicalTrials.Gov 46 up until June 2020 provides the number of unpublished, ongoing, and completed phase I stroke trials. Supplement File 2 provides details of each registry search. Broadly, we sought to include trials of any recruitment status listed under the condition of “stroke” and phase of “phase I.” We have extracted the number of registered phase I stroke trials compared to registered stroke trials overall and classify phase I trials as either pharmaceutical or nonpharmaceutical (Table 1). The intervention type and stated purpose of the nonpharmaceutical phase I trials have also been extracted (Table 1). The search result accuracy relies on the individual study investigator assigning a trial phase during the registration process, which is not mandatory and may not be correctly applied.
Registered Phase I Stroke Trials Until June 2020.
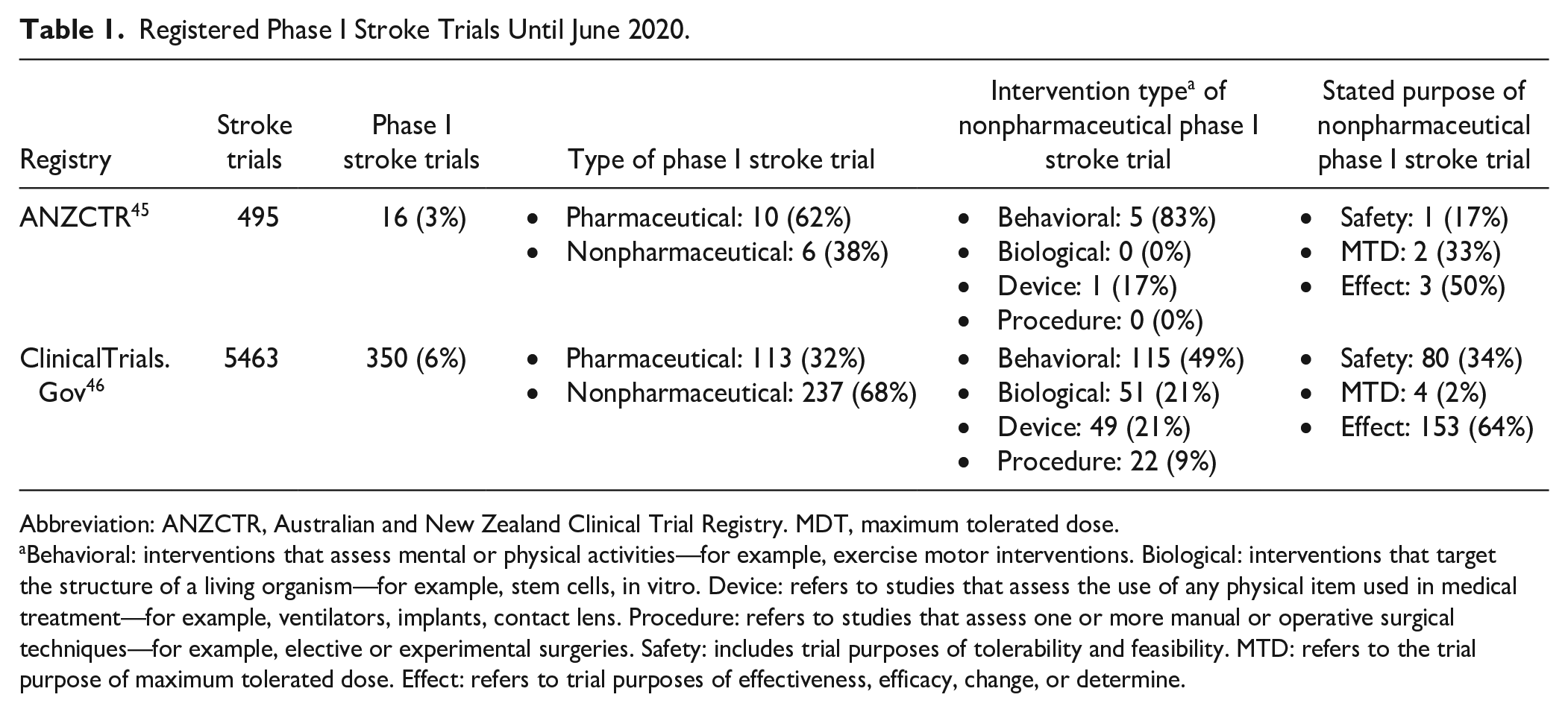
Abbreviation: ANZCTR, Australian and New Zealand Clinical Trial Registry. MDT, maximum tolerated dose.
Behavioral: interventions that assess mental or physical activities—for example, exercise motor interventions. Biological: interventions that target the structure of a living organism—for example, stem cells, in vitro. Device: refers to studies that assess the use of any physical item used in medical treatment—for example, ventilators, implants, contact lens. Procedure: refers to studies that assess one or more manual or operative surgical techniques—for example, elective or experimental surgeries. Safety: includes trial purposes of tolerability and feasibility. MTD: refers to the trial purpose of maximum tolerated dose. Effect: refers to trial purposes of effectiveness, efficacy, change, or determine.
We expect to see more early-phase trials in contrast to later-phase trials because the very purpose of early-phase trials is to reduce potential interventions to the most promising options to take forward to phase III. Phase I trials (pharmaceutical and nonpharmaceutical), however, account for only 6% of all registered stroke trials (Table 1), suggesting that this phase is either poorly registered or not completed. A review of registered nonpharmaceutical phase I stroke trials (66% vs 34% pharmaceutical) indicates that the majority do not follow the principles outlined in the systematic discovery pipeline 5 (Figure 1). A phase I dose ranging trial should aim to determine the tolerated minimum to maximum dose range; this is stated as the trial purpose for only 3% of nonpharmaceutical phase I stroke trials in our evaluation. Nearly two-thirds (64%) stated a primary aim to determine efficacy (effect or change or improve), which should not be the focus of a phase I trial. These results confirm that, against natural expectations, phase I trials make up a small proportion of registered stroke trials and an even smaller proportion of trials that aim to identify the MTD. 5 To advance the field, phase I trials need to become a critical and necessary step to complete during intervention development.
Multidimensional Dose Articulation Has Not Been Adequately Considered in Stroke Recovery Interventions
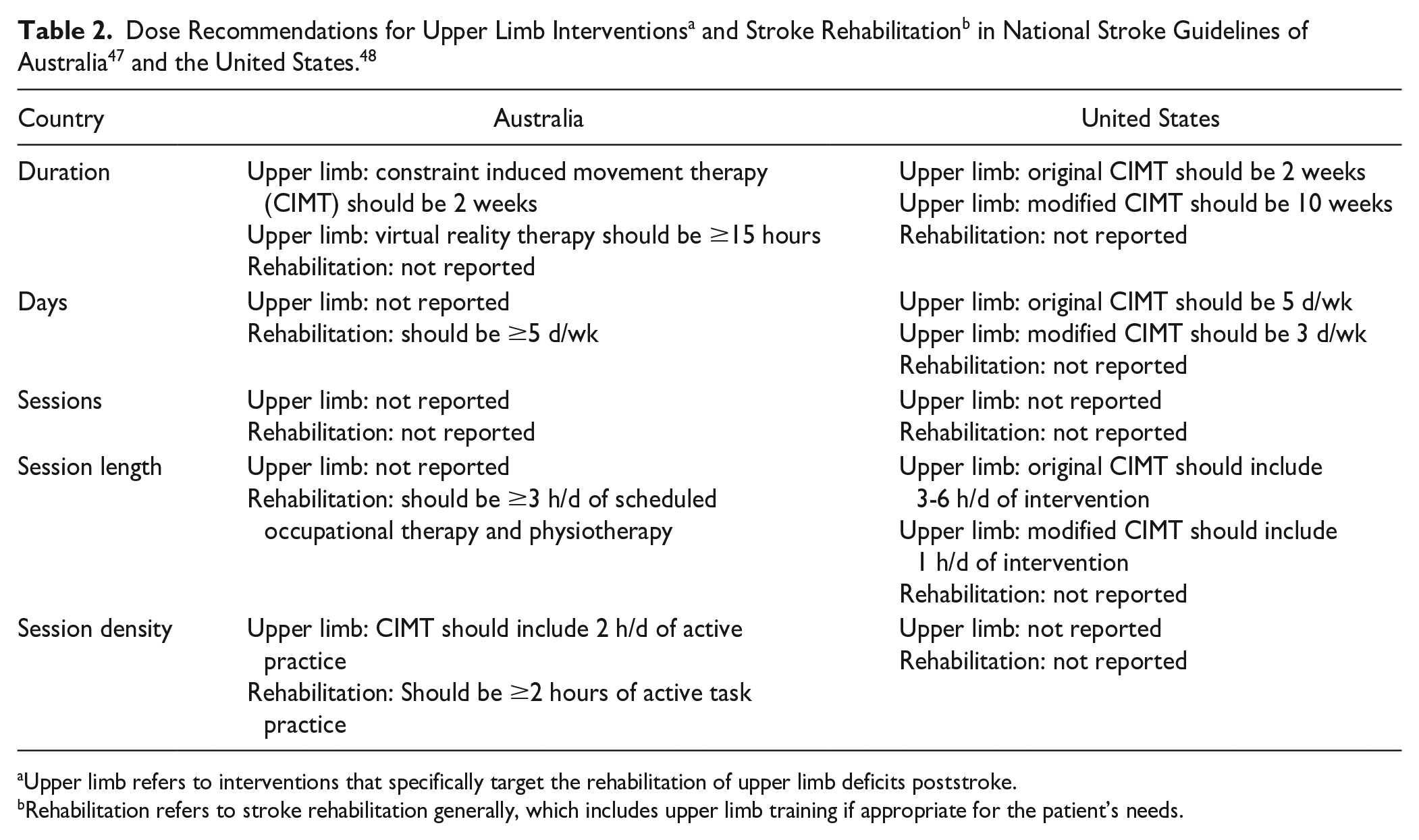
Multidimensional phase I trials receive minimal funding and contribute to a small proportion of stroke recovery research, affecting national clinical practice guidelines. To explore the impact of this gap, we investigate the dose recommendations (amount of practice) for upper limb interventions and rehabilitation programs for the first 5 dose dimensions of the multidimensional dose framework 11 (Figure 2) using the Australian 47 and US 48 stroke guidelines (Table 2).
Upper limb refers to interventions that specifically target the rehabilitation of upper limb deficits poststroke.
Rehabilitation refers to stroke rehabilitation generally, which includes upper limb training if appropriate for the patient’s needs.
Guidelines in both countries report at least 1 recommendation for the dose dimensions of duration, days, and session length. The Australian guidelines 47 report a recommendation consistent with session density. Neither guideline reports a recommendation for sessions. Despite several upper limb interventions commonly featuring in both guidelines, there is only specific dose information recommended for constraint induced movement therapy and virtual reality. Given that national guidelines are based on best available evidence, the lack of dose recommendations confirms that there is limited high-quality, dose articulation research of upper limb interventions for stroke recovery. The guidelines also highlight a lack of consideration for dose as multidimensional, with recommendations commonly describing a time-based dose dimension (eg, duration, days), rarely reporting information on intensity or difficulty.
Overall, the lack of systematic early-phase trials is a gap for nonpharmaceutical stroke recovery interventions. Whereas the field identifies the need for a systematic discovery pipeline approach5,10,15,49,50 and a multidimensional dose framework,2,11,51 there remains limited evidence of application in research practice. Systemic changes (eg, funding opportunities) will take time; however, increasing our knowledge of how to conduct a multidimensional phase I trial per the systematic discovery pipeline 5 can be addressed now. There is a lack of guidance in the literature concerning how to design a multidimensional phase I trial for nonpharmaceutical interventions. This is not surprising given that phase I trial dose ranging designs have been traditionally designed for pharmaceutical interventions with the amount of a drug successfully articulated. Nonpharmaceutical stroke recovery interventions lack an equivalent, unidimensional measure. Early-phase research, particularly the appropriate design and implementation of multidimensional phase I nonpharmaceutical stroke recovery trials, is in its infancy. 6 The proposed solution serves to increase knowledge and, thus, application in this field.
Solution
To operationalize the systematic discovery pipeline 5 for nonpharmaceutical stroke recovery research, we need to understand the desirable properties that shape the design of early-phase trials and adapt them for multidimensional dose articulation. Here, we address phase I trial design only as it forms the first clinical phase in the discovery pipeline and is the least common early-phase design approach completed. 6 Acquired knowledge from the pharmaceutical field and the 2 published phase I stroke recovery trials34,52 will be used to guide the solution. We present the desirable properties, each followed by a hypothetical worked example, to guide the design of multidimensional phase I dose ranging trials for stroke recovery interventions. We then formulate the solution that satisfies these desirable properties in the form of a design template (Supplement File 1). This design template serves as a decision support tool for trial design and protocol development of future phase I trials in the field.
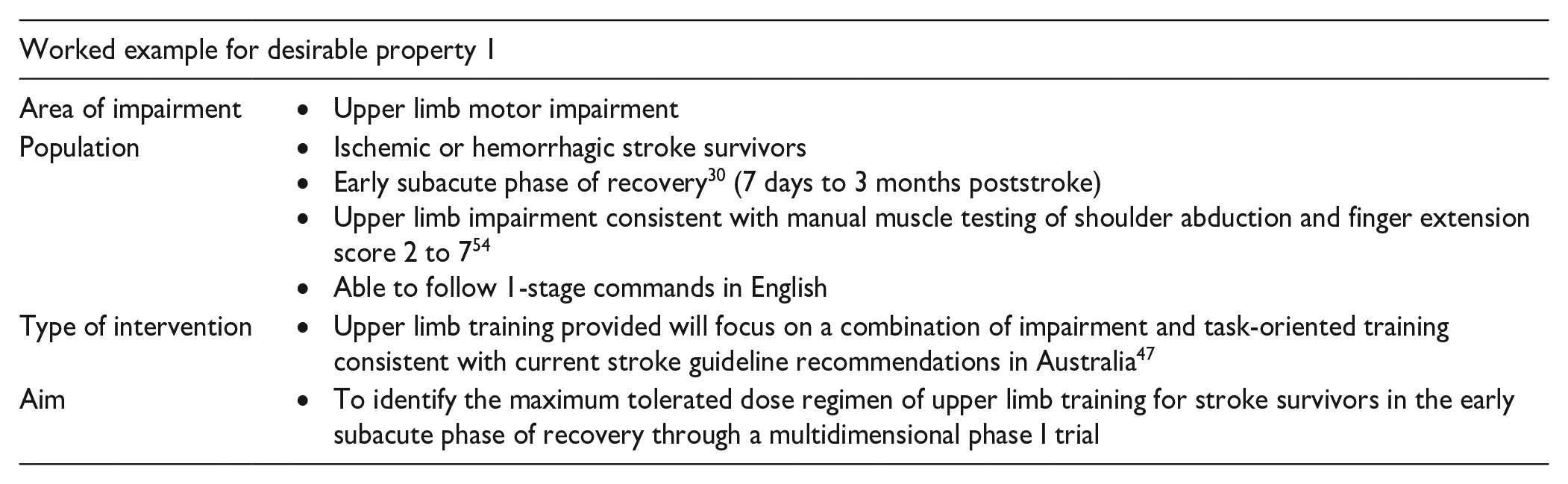
Desirable Property 1: Clearly Established Trial Scope, Which Includes Area Impairment, Population, Type of Intervention, and Research Aim
Dose ranging is the first clinical trial phase (phase I) outlined in the discovery pipeline 5 (Figure 1). Such trials systematically escalate and de-escalate dose to identify a minimum to maximum dose range.5,10,13 Safety and feasibility outcomes help determine the MTD for a given trial scope. Efficacy outcomes may (or may not) be used to inform future early-phase trials, but they are not the focus of phase I trials. Consistent with clinical trial development, the area of impairment (eg, upper limb) and type of intervention (eg, task-specific training) needs to be established to guide trial design. The trial scope chosen for phase I should reflect what you hope to test in later-phase trials. The population of participants to recruit also needs to be determined. Phase I trials should purposefully sample participants, based on markers such as recovery phase, 30 impairment level, 30 or biology. 53 These should be reflected in the research aim, along with the phase I trial purpose.
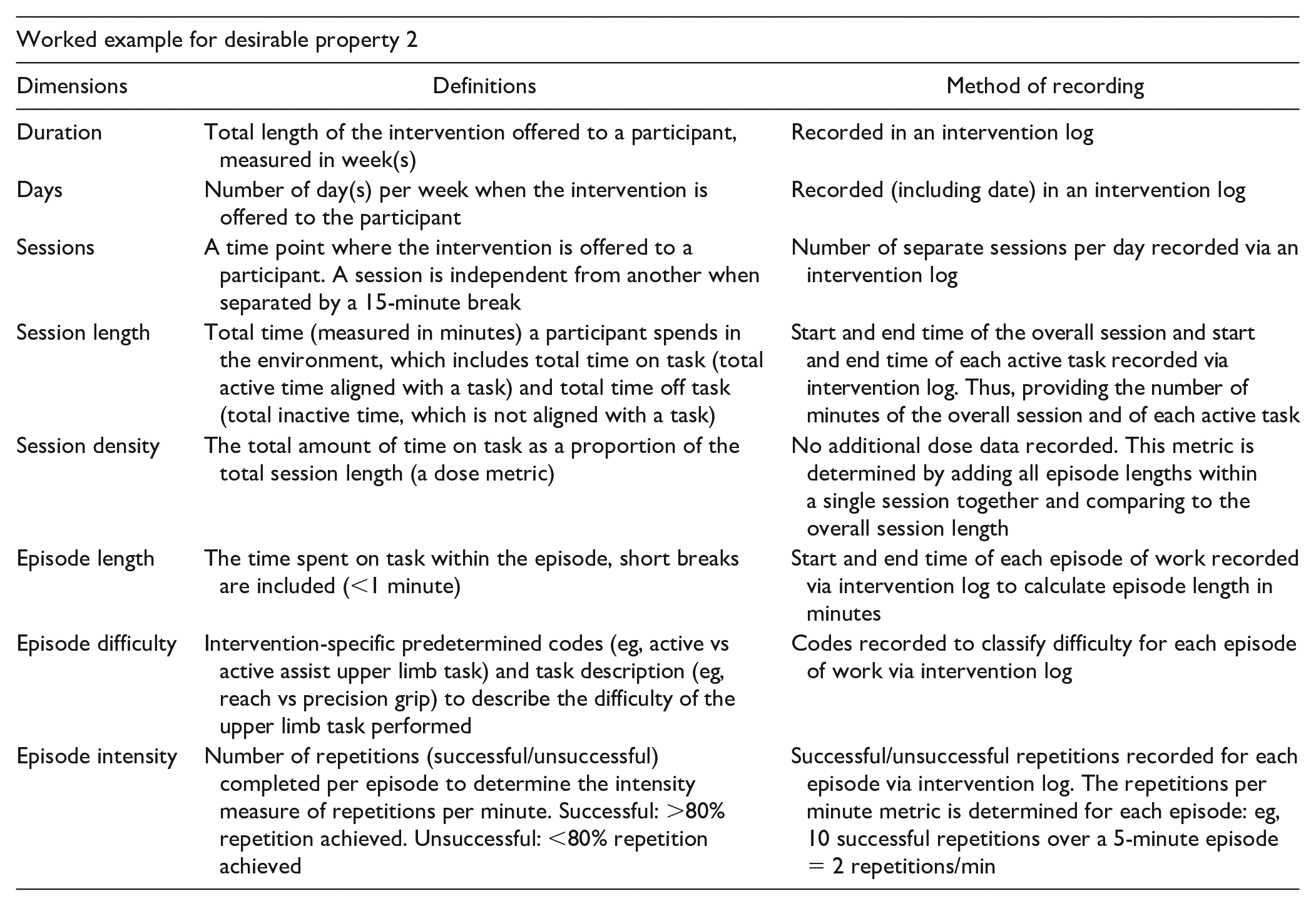
Desirable Property 2: Clearly Established Dose Dimensions for the Chosen Intervention Via the Multidimensional Dose Framework 11
The multidimensional dose framework 11 (Figure 2) conceptualizes dose as consisting of 8 dimensions. Consideration of all dimensions is required to establish a maximum tolerable dose range (includes all doses up to the MTD). Real-time documentation of delivered dose for a nonpharmaceutical trial is challenging, and development of methods to reduce burden of data collection is required. 11 The collection of dose information requires objective and subjective measures reported by the therapist and/or participant. This may evolve in the future with the adoption of point-of-care technology, which can facilitate real-time data collection. 11 To achieve multidimensional dose articulation within a phase I trial, each individual dose dimension and corresponding measurement needs to be defined for the trial scope established in desirable property 1.
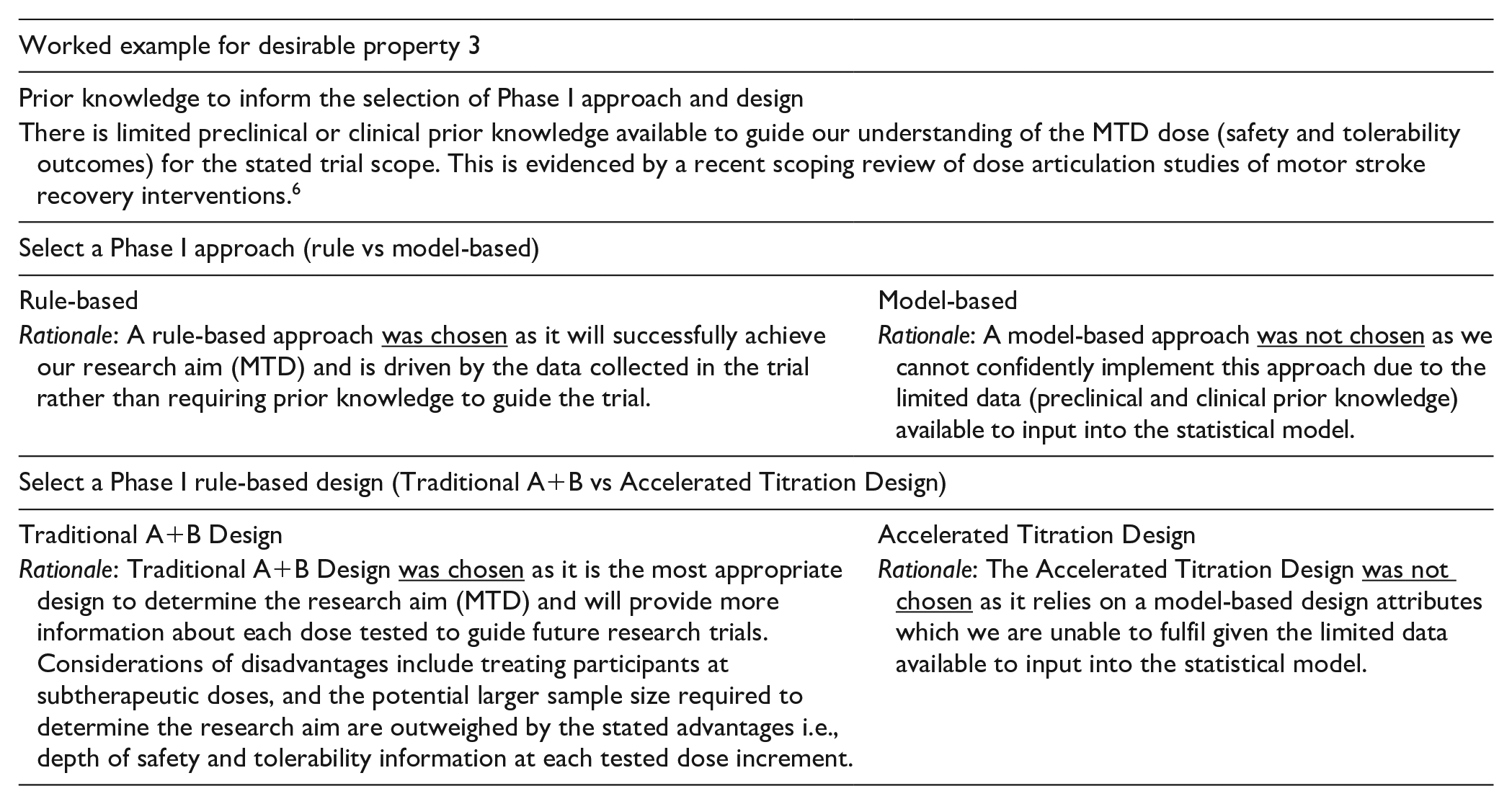
Desirable Property 3: Appropriate Choice of Phase I Approach and Design
Two broad phase I approaches (rule based and model based) provide a guiding philosophy for the selection of specific attributes for phase I trial designs. A rule-based approach stipulates that the MTD is identifiable exclusively through the data without relying on prior assumptions about the relationship between the dose and the probability of experiencing dose-limiting criteria.18,55 A model-based approach relies on prior assumptions (eg, opinions of experts who have experience with the intervention) to support the estimation of the potential relationship between the dose and the probability of experiencing dose-limiting criteria, often using Bayesian modeling. 18 Dose-limiting criteria are markers that are presumably related to the intervention and are considered unacceptable, therefore limiting further dose escalation. 18 Phase I trial methodology is rapidly developing, with discussions regarding the circumstances that should guide the selection of a rule- or model-based approach continually evolving. 56
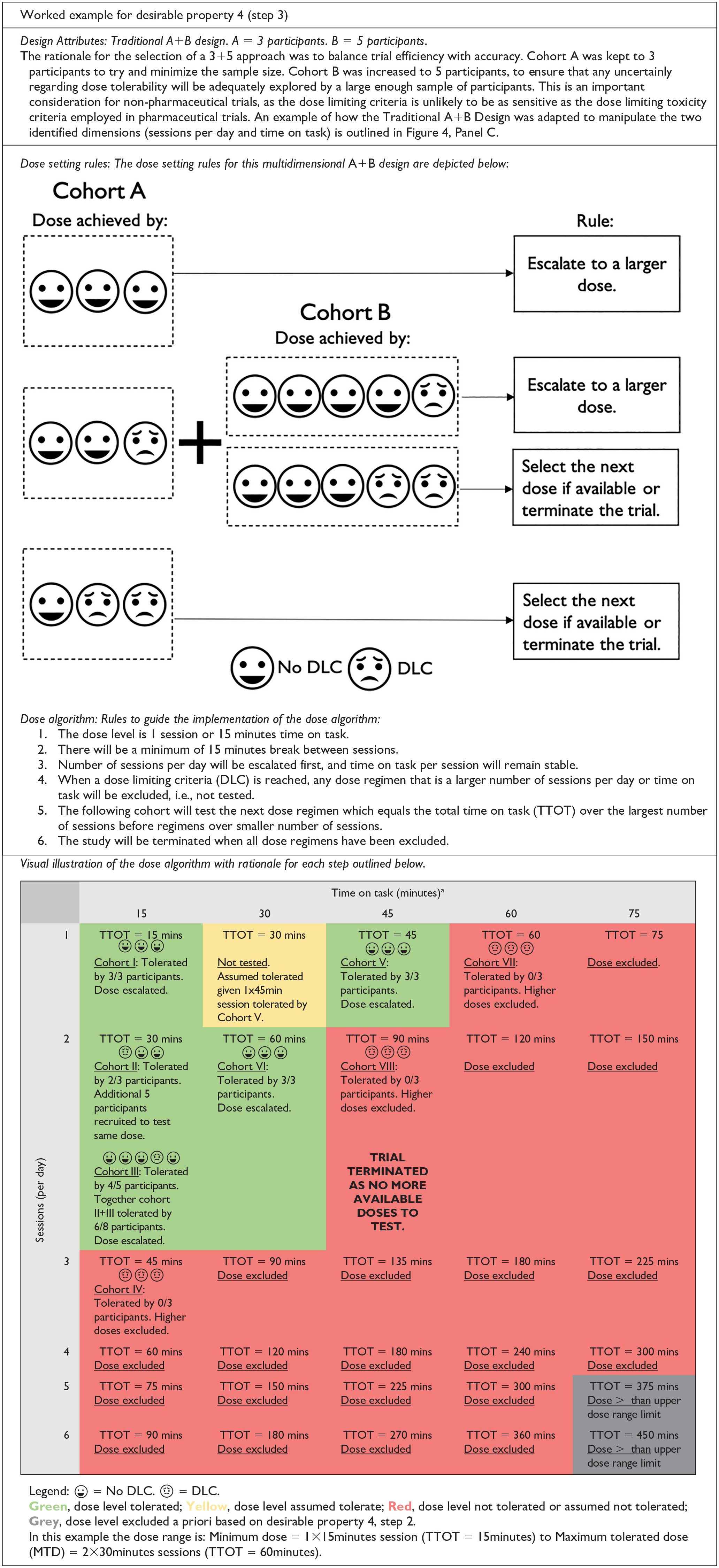
A rule-based approach includes several phase I trial designs. In this article, 2 common designs are discussed and represented in Figure 4, Panel A: traditional A + B design and accelerated titration design. For the traditional A + B design, dose is escalated in cohorts defined by the attributes of A and B, until the prespecified dose limiting criteria are reached. The value of A is the initial number of participants recruited to a cohort to test the dose. Based on their response, the prespecified dose setting rules determine if an additional number of participants B is required to test the same dose. 18 Typically, in pharmaceutical trials, A would equal 1 or 3 participants, and B would equal 3 participants. The values of A and B can be tailored based on the trial scope—for example, A = 3 participants and B = 5 participants might be more appropriate for a nonpharmaceutical stroke recovery trial. An example of a dose setting rule is if 1 participant out of 3 in cohort A experiences dose limiting criteria, an additional B cohort of 3 participants is recruited to test the same dose. The advantage of this design is that it provides extra information regarding interparticipant variability by potentially recruiting up to A + B participants for a given dose; however, this also means that it can require quite a large number of escalations. 18
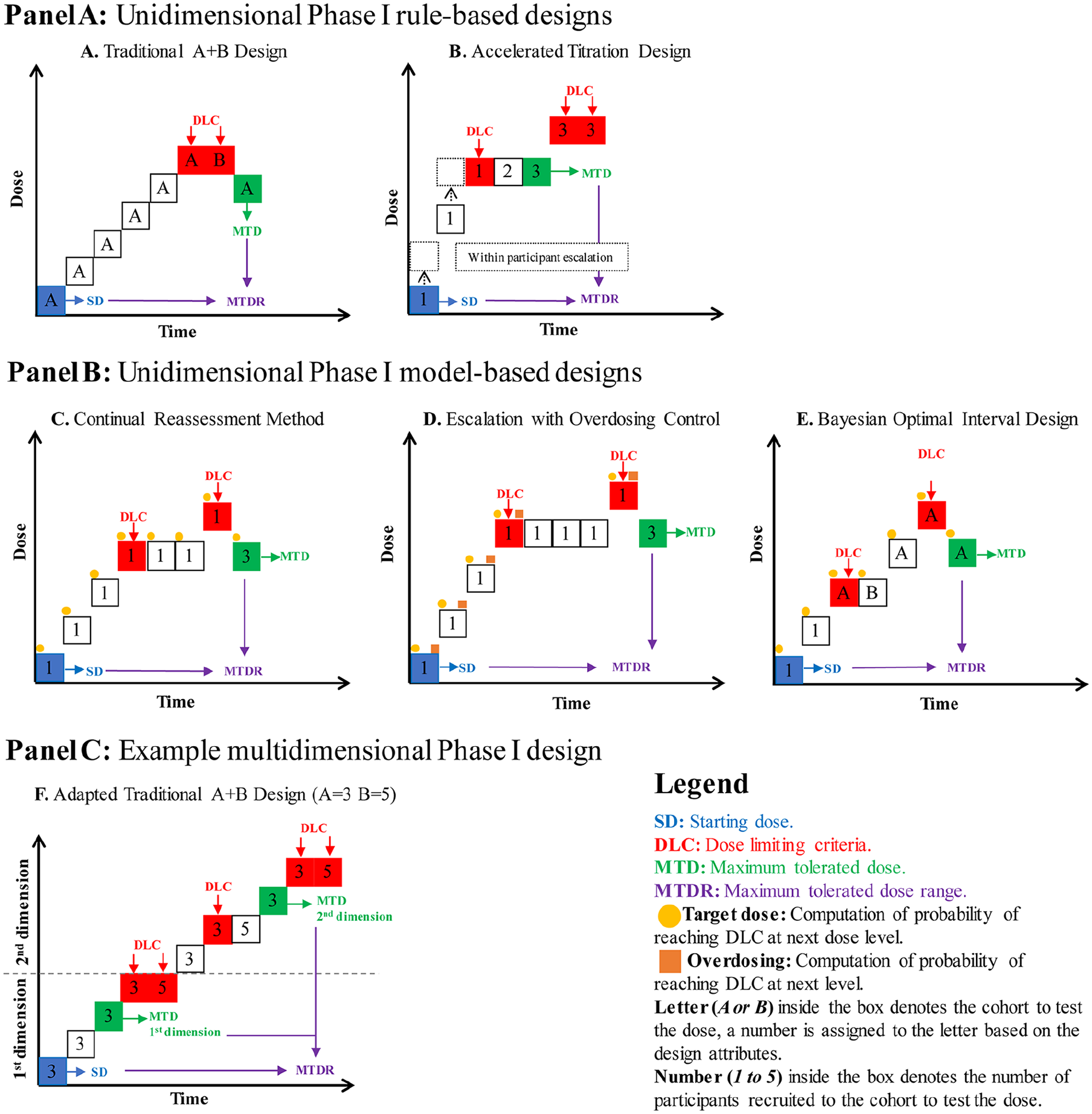
Visual representation of phase I dose ranging trial designs. Panel A: Unidimensional rule-based phase I trial designs (discussed in Solution, Desirable Property 2). Panel B: Unidimensional model-based phase I designs (discussed in Solution, Desirable Property 2). Panel C: Example of adapted multidimensional rule-based design (discussed in Solution, Desirable Property 4).
The accelerated titration design also sets out prespecified criteria to guide participant allocation; however, it uses some model-based features and, therefore, may be regarded as a model-based design by some. 18 This design usually includes an initial acceleration phase, which implements intraparticipant dose escalation. This means that 1 participant is recruited to test escalating dose levels, until the prespecified criteria are met. At this point, an additional cohort of participants will be recruited to explore the dose level further. The accelerated titration design uses a model to estimate the MTD from the data collected in the trial. The advantage of this design is that it can reduce the number of participants treated at subtherapeutic doses but can equally mask cumulative or delayed presentation of dose-limiting criteria. 57
A model-based approach includes several trial designs, 3 of which are shown in Figure 4, Panel B: Continual Reassessment Method, Escalation With Overdose Control, and Bayesian optimal interval design. The continual reassessment method is based on the principle that all participants are treated at the dose closest to the MTD. 18 Each participant who enters the trial provides more information to update the estimation process. 58 The trial ends when a prespecified criterion is met—for example, X number of participants are assigned to the same dose, and all reach a dose-limiting criterion. The uptake of this design in pharmaceutical trials has been limited because of concerns that it can expose participants to an unacceptably high dose if the prespecified model is not accurate. 18 To overcome these limitations, escalation with overdosing control was developed.59,60 It has been described as a modified continual reassessment method, with additional safety precautions to avoid exposing participants to a dose that has too high a risk of reaching dose-limiting criteria. 18 These measures include assessing the probability of administering a dose higher than the MTD after each participant as well as providing a clear prespecified value to determine if the probability is too high. 59
The Bayesian optimal interval design61,62 aims to combine the favorable design attributes from rule- and model-based approaches. 61 It uses the robust algorithm attributes of a rule-based approach combined with the efficient Bayesian model attributes to efficiently guide dose allocation. 62 The rule-based algorithm attributes include the prespecified decision-making steps to determine the dose allocation. The Bayesian model attributes focus on establishing a dose-limiting rate (ie, the number of participants who experience dose-limiting criteria divided by the total number of participants treated) to efficiently guide dose allocation. 62 The combination of these attributes within the Bayesian Optimal Interval Design could minimize the number of participants treated at subtherapeutic doses while also safeguarding participants from being treated at harmful dose levels.61,62 Like other model-based designs, this design relies on prior knowledge to establish the model.
A rule-based approach is the most commonly implemented phase I design for pharmaceutical cancer trials. 18 This trend is reflected for nonpharmaceutical stroke recovery research, with 2 published phase I trials implementing a rule-based approach.34,52 The tradeoff between accuracy and efficiency is the key difference. A rule-based approach leans toward higher accuracy. It has been established as a successful approach to determine the MTD; however, it can be inefficient, taking more time and potentially resulting in several participants being treated at subtherapeutic doses. A model-based approach leans toward higher efficiency because it uses preexisting knowledge to guide estimations of the MTD to test. This results in more participants being treated at a therapeutic dose. The accuracy of preexisting knowledge and the ability to adequately present this knowledge through the Bayesian modeling are 2 factors that can help increase the likelihood that MTD is accurately estimated for model-based designs. Once the approach is decided, the type of design needs to be selected. Each design determines the MTD differently, and consultation with an experienced biostatistician is highly recommended.
Desirable Property 4: Successful Integration of the Multidimensional Dose Framework 11 Into Phase I Trial Designs
Phase I trial designs do not routinely accommodate the manipulation of more than 1 dose dimension within the same trial. To successfully integrate the multidimensional dose framework 11 into existing phase I designs, we need to maintain the systematic processes of phase I designs while allowing multiple dose dimensions to be tested. There are 3 main steps to guide this process for both rule- and model-based approaches.
Step 1: Define the Dose-Limiting Criteria
The primary outcome (MTD) is established when a set of prespecified criteria are met. In pharmaceutical trials, the criteria are commonly labeled dose-limiting toxicity because safety outcomes are usually monitored using toxicity markers in the blood. In nonpharmaceutical trials, dose-limiting criteria are based on measure(s) that capture what could be indicative of dose intolerability (eg, fatigue level that would prevent continuing with the intervention). Both the measure and the criteria need to be specific to the intervention and sensitive enough to determine the MTD. They also need to allow for small inherent differences in dose tolerability between participants. Thoughtful consideration of the measure used (eg, Visual Analog Scale) and how it will be assessed (eg, routinely asked by therapist vs voluntarily reported by the participant) are required. Environmental and pragmatic limitations may also be appropriate to include in the criteria based on the setting chosen for the trial.

Step 2: Operationalize Dose Dimensions
The total number of dose combinations may become prohibitively large to test in 1 trial because each dose dimension can have multiple potential values. To manage possible combinations, investigators can systematically “manipulate” some dimensions through a range (lower and upper dose limit). Other dimension(s) might be considered “fixed” within a predefined boundary (eg, rest time is allowed up to a maximum of 15 minutes). A fixed dose dimension is selected at the beginning of the trial and remains within the assigned boundary dose during the trial. A manipulated dose dimension is assigned a lower and upper value at the beginning of the trial, which determines the starting dose and the maximum feasible dose to test, respectively. The doses between the lower and upper values form the range that will be tested to determine the MTD. The decision about which dimensions should be fixed or manipulated requires a combination of intervention-specific knowledge and consideration of how the choices made may affect later-phase trials. This process should result in a rank-ordered list of dimensions in terms of their ability to present information of sufficient granularity for the stated trial aim (MTD).
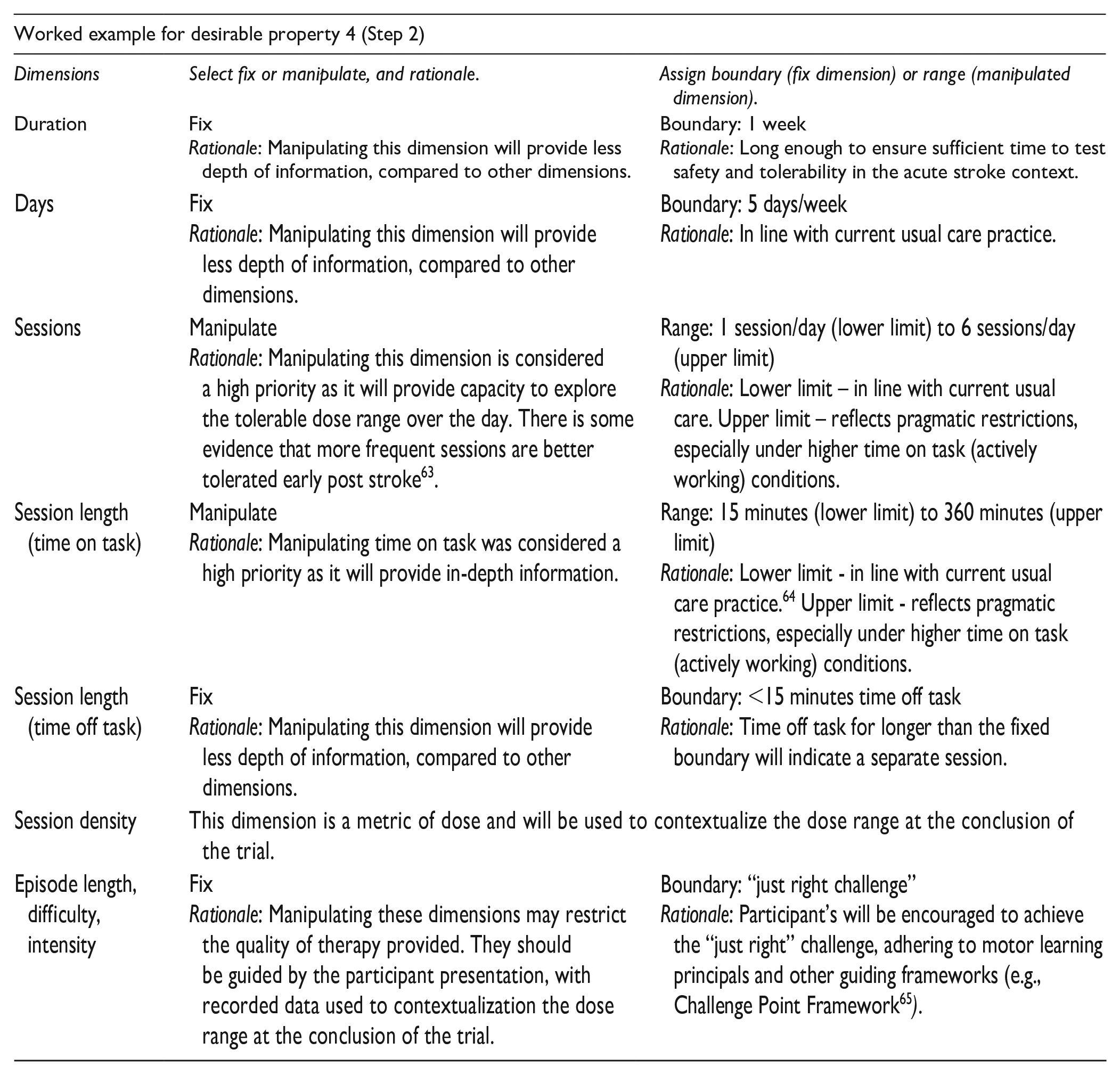
Step 3: Determine the Sequential Dose Allocation Process
This step defines the sequential process to allocate the dose to be tested in the next cohort—that is, the next combination of manipulated dimensions. There are different considerations for trial designs based on whether they fall under a rule- or model-based approach. For a rule-based approach, the dose allocation is guided by a rule-based algorithm that determines the next dose and the size of the cohort to be tested at that dose. The execution of the algorithm is guided by a set of rules that determine decisions such as the dimension to be manipulated first, the dose level increments, and how the increments will be escalated or de-escalated. An example of the Traditional A + B design adapted for multidimensional dose articulation is demonstrated in Figure 4, Panel C. For a model-based approach, the dose allocation is guided by a model-based algorithm, which is supported by the chosen statistical model. Establishing the model requires the combination of prior knowledge, exploration of the risks to participants if overdosing occurs, and discussion regarding how the model will be managed and updated during the trial—for example, what computer software will be used, who will update the model after each participant. The execution of the algorithm is guided by the model that determines decisions such as the dimension to be manipulated first, the dose level increments, and how the increments will be escalated or de-escalated.
Design Template to Guide the Development of a Multidimensional Phase I Dose-Ranging Stroke Recovery Trials
The solution that satisfies these desirable properties is formulated in a design template to serve as a decision support tool for the development of future multidimensional phase I dose ranging trials of stroke recovery interventions. This template incorporates the desirable properties into steps to guide the decision process. The goal of the design template is to support the uptake of good quality early-phase research in the field of nonpharmaceutical stroke recovery. The full template (Supplement File 1) provides space to apply the properties to develop your own multidimensional phase I trial.
Recommendations
Early-phase designs are underutilized, resulting in the limited investigation of dose ranging during nonpharmaceutical stroke recovery intervention development. By providing the knowledge of how to systematically investigate the multiple dimensions of dose during intervention development, we hope to increase the uptake and quality of early-phase dose articulation research for nonpharmaceutical stroke recovery interventions. The expected outcome of this is a series of trials that systematically investigate dose—for example, phase I through to phase IIB. This would provide a clearly justified dose for use in a phase III efficacy trial that is underpinned by a whole of pipeline approach. The systematic investigation of dose is one piece that can move the field closer to delivering a game-changing intervention.
Moving forward we suggest that the stroke recovery field adopts the following recommendations:
Dose should be systematically investigated during the intervention development stage.
Early-phase trials should be developed based on the systematic discovery pipeline (which should include collaboration between preclinical and clinical researchers where relevant) and establish the MTD via safety and tolerability outcomes prior to efficacy outcomes.
Sustained knowledge dissemination efforts are required to ensure that funding bodies and journal editors support early-phase research as a priority area.
Phase I trials adapted to the context of stroke recovery interventions should be implemented to support multidimensional dose articulation.
The design template for multidimensional phase I dose ranging stroke recovery trials should be used as a decision support tool to inform trial design.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683211019362 – Supplemental material for Multidimensional Phase I Dose Ranging Trials for Stroke Recovery Interventions: Key Challenges and How to Address Them
Supplemental material, sj-docx-1-nnr-10.1177_15459683211019362 for Multidimensional Phase I Dose Ranging Trials for Stroke Recovery Interventions: Key Challenges and How to Address Them by Emily J. Dalton, Leonid Churilov, Natasha A. Lannin, Dale Corbett, Bruce C. V. Campbell and Kathryn S. Hayward in Neurorehabilitation and Neural Repair
Supplemental Material
sj-pdf-1-nnr-10.1177_154596832110193621 – Supplemental material for Multidimensional Phase I Dose Ranging Trials for Stroke Recovery Interventions: Key Challenges and How to Address Them
Supplemental material, sj-pdf-1-nnr-10.1177_154596832110193621 for Multidimensional Phase I Dose Ranging Trials for Stroke Recovery Interventions: Key Challenges and How to Address Them by Emily J. Dalton, Leonid Churilov, Natasha A. Lannin, Dale Corbett, Bruce C. V. Campbell and Kathryn S. Hayward in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The Florey Institute of Neuroscience and Mental Health acknowledges the support of the Victorian Government’s Operational Infrastructure Support Grant.
Author Contributions
ED, KH, and LC conceived the idea for this article and were involved in all aspects of the study, with manuscript writing led by ED. NL, BC, and DC were involved in manuscript editing. All authors made intellectual contributions to the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by an Australian Government Research Training Program Scholarship (ED); National Health and Medical Research Council (KH: 1088449; BC: 1174514), and Heart Foundation Future Leader Fellowship (NL: GNT102055).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.