Abstract
Background
Health-related quality of life (HRQL) in stroke survivors is related to numerous factors, but more research is needed to delineate factors related to HRQL in people with aphasia.
Objective
To examine the relationship between HRQL and demographic factors, impairment-based measures, and lesion characteristics in chronic aphasia.
Methods
A total of 41 left-hemisphere stroke survivors with aphasia underwent cognitive testing and magnetic resonance imaging. To address relationships with demographic and impairment-based measures, test scores were entered into a principal component analysis (PCA) and multiple linear regression was performed for overall and domain (physical, communication, psychosocial) scores of the Stroke and Aphasia Quality of Life Scale (SAQOL-39g). Independent variables included factor scores from the PCA, motricity, lesion volume, depressed mood, and demographic variables. To address relationships with lesion location, multivariate support vector regression lesion-symptom mapping (SVR-LSM) was used to localize lesions associated with SAQOL-39g scores.
Results
The PCA yielded 3 factors, which were labeled Language Production, Nonlinguistic Cognition, and Language Comprehension. Multiple linear regression revealed that depression symptoms predicted lower SAQOL-39g average and domain scores. Lower motricity scores predicted lower SAQOL-39g average and physical scores, and lower Language Production factor scores predicted lower communication scores. SVR-LSM demonstrated that basal ganglia lesions were associated with lower physical scores, and inferior frontal lesions were associated with lower psychosocial scores.
Conclusions
HRQL in chronic left-hemisphere stroke survivors with aphasia relates to lesion location, depression symptoms, and impairment-based measures. This information may help identify individuals at risk for specific aspects of low HRQL and facilitate targeted interventions to improve well-being.
Introduction
Health-related quality of life (HRQL) describes how a person’s specific health condition impacts their perceived ability to lead a fulfilling life. 1 A priority in stroke rehabilitation research is to identify intervention strategies that will improve patients’ quality of life. 2 In order to develop targeted treatment approaches, it is important to understand how objective factors relate to subjective quality of life measures. HRQL is multifactorial, and may include domains related to physical, social, cognitive, and emotional functioning, although there is no consensus on a single definition. 1 Previous findings collectively suggest that HRQL in stroke survivors is related to numerous factors, including physical impairment, depressive symptoms, sex and gender, race, 3 and social support. 4 Currently, there is a need for further HRQL research in stroke survivors with aphasia; additionally, the relationship between lesion location and HRQL has yet to be thoroughly examined.
Roughly one-third of all stroke survivors are affected by aphasia, an acquired language disorder that typically occurs following a left-hemisphere stroke. 5 Multiple studies report positive associations between language ability and various aspects of HRQL in stroke survivors (Supplemental Table 1).6-16 In general, most of these studies have employed metrics (Aphasia Severity Rating Scale,6,7 American Speech and Hearing Association–Functional Assessment of Communication Skills for adults,8,9 Frenchay Aphasia Screening Test,9,14 Norsk Grunntest for Afasi, 10 Western Aphasia Battery Aphasia Quotient12,13) that broadly characterize aphasia severity, without distinguishing between different language processes (such as receptive language, expressive language, repetition, reading, writing, and motor speech). Yet current aphasia assessments and rehabilitation plans are designed to identify and improve impairments in specific language processes. 17 This means that there is currently limited evidence available to help identify language treatment approaches that would additionally target the patients’ HRQL, despite the consensus in stroke rehabilitation that improving quality of life is a priority. 2 A deeper understanding of which language domains correlate with reduced HRQL may be beneficial, in order to identify language rehabilitation plans that can in parallel improve HRQL outcomes of importance to the individual patients. 18
Because HRQL is multifactorial, is it important to consider demographic and social factors alongside aphasia characteristics. 19 Depression,6-8,12,14 fatigue, 6 functional status,6-8,12,14,20 and cognition8,21 have been associated with measures of HRQL in people with aphasia. The relationship between demographic variables and HRQL is unclear in people with aphasia. Some findings demonstrate that HRQL is associated with age,8,16,20 sex 12 and gender, 20 education,12,20 time since stroke, 20 and socioeconomic status,12,14 while others show that age,12,15,21 gender,8,10,21 education,15,16 time since stroke,8,15,16,21 and socioeconomic status 8 are not related to HRQL in people with aphasia.
Patient-reported outcome measures (PROM) capture HRQL information directly from the patient without practitioner or caregiver input and are critical for the development of patient-centered approaches to intervention. 22 The Stroke and Aphasia Quality of Life Scale (SAQOL) is a well-established 39-item PROM for assessing HRQL in stroke survivors with and without aphasia. 23 It was designed to accommodate the challenges of measuring HRQL in people with communication impairments by using simple verbiage and content layout; however, it may not be suitable for patients with severe comprehension deficits. The SAQOL-39g has been validated in a generic stroke sample of survivors without aphasia and measures 3 domains that are commonly affected by stroke: physical, communication, and psychosocial. 24
The role of lesion characteristics in HRQL has not been thoroughly examined; a few studies have suggested that subcortical and brainstem lesions may relate to poor HRQL.25,26 Relationships with overall lesion volume have been inconsistent, with studies finding lesion volume to be moderately related, 27 unrelated, 25 or related only to cognitive domains 28 of HRQL. Previous studies investigating lesion location and HRQL have used dichotomous classifications for lesion location 29 or quality of life,25,26 or excluded people with moderate to severe aphasia. 30 Further research is needed to understand how biological factors such as lesion location relate to HRQL outcomes. If present, relationships between lesion location and HRQL are likely indirect, in that lesions of particular brain structures may cause behavioral effects that could relate to HRQL. However, in some cases lesion location could theoretically relate more strongly to subjective HRQL than individual objective standardized assessment scores (such as the Western Aphasia Battery subscores) because a lesion to one brain structure may cause multiple behavioral consequences that all contribute to HRQL. Given that lesion location is identified early after stroke onset, if there is a relationship between lesion location and HRQL, then this information may allow for early identification of individuals at risk for low HRQL, providing an opportunity for early interventions to improve HRQL.
The goal of the current study was to examine the relationship between HRQL, behavioral measures, and lesion characteristics in left-hemisphere stroke survivors with aphasia. This study focuses on the physical, communication, and psychosocial domains of HRQL, demographic factors (age, gender, race, education), linguistic and nonlinguistic impairment-based measures, and motricity. Findings may help to identify left-hemisphere stroke survivors with aphasia at risk for low physical, communication, and psychosocial aspects of HRQL, in order to develop targeted rehabilitation plans that address these domains and improve patient quality of life.
Methods
Participants
This study included 41 participants that were recruited as part of a clinical trial of transcranial direct current stimulation for aphasia (Clinicaltrials.gov NCT01709383). All participants were left-hemisphere stroke survivors (36 ischemic, 5 hemorrhagic) with a history of aphasia, at least 6 months poststroke, native English speakers, with no history of major psychiatric disorder or other significant central nervous system disorder. See Table 1 for demographic information. This study was approved by the Georgetown University Institutional Review Board, and all participants provided written informed consent.
Participant Demographic and Clinical Information.
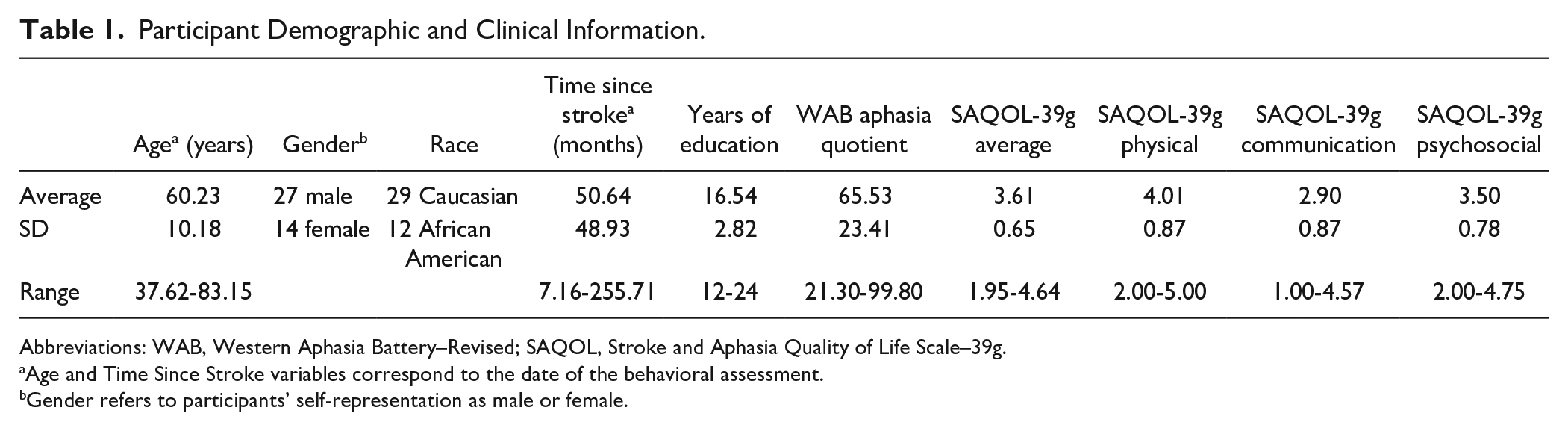
Abbreviations: WAB, Western Aphasia Battery–Revised; SAQOL, Stroke and Aphasia Quality of Life Scale–39g.
Age and Time Since Stroke variables correspond to the date of the behavioral assessment.
Gender refers to participants’ self-representation as male or female.
Behavioral Assessment
The behavioral assessment was designed to evaluate language and nonlinguistic cognition thoroughly and through different modalities, and was conducted at a single timepoint. A brief description of each questionnaire or test follows.
Stroke and Aphasia Quality of Life Scale (SAQOL-39g) 24 : Participants, with facilitation by a study partner or caregiver if needed (in alignment with the user’s manual), filled out the SAQOL-39g as a subjective evaluation of their functioning in the physical, communication, and psychosocial domains. Questions were rated on a 1 to 5 scale, with lower scores indicating a poorer quality of life.
Stroke Aphasic Depression Questionnaire (SADQ) 31 : The SADQ was used to assess severity of depression symptoms, with higher values on a 0 to 3 scale indicating greater severity. The questionnaire was designed to be completed by caregivers, although 4 participants lived independently without caregivers and therefore completed the SADQ themselves.
Motricity Index 32 : Upper-limb motor impairment was measured with the pinch grip, elbow flexion, and shoulder abduction scores on the Motricity Index. Scores on the right and left sides were averaged together, and lower scores indicated greater impairment on a 0 to 33 scale.
Philadelphia Naming Test (PNT) 33 : A reduced 60-item version of the PNT was used to assess confrontation naming. Participants were instructed to name aloud the item depicted in each black and white drawing.
Category and Letter Fluency34,35: To assess expressive language and verbal fluency, participants were instructed to generate as many items as possible within certain categories (animals, items in a supermarket, things you wear) or that began with certain letters (M, F, A, S) in 60 seconds.
Pseudoword Repetition: This in-house test was developed to measure repetition and postlexical phonological output processing. Pseudowords increased in length from 1 to 5 syllables as the 30-item test progressed.
Reading Real Words: Participants were presented with single words, 3 to 9 letters long, and asked to read them aloud as a measure of single word reading.
Reading Pseudowords: To assess sublexical reading, participants were asked to read aloud 20 pseudowords, 3 to 4 letters in length, all 1 syllable.
Western Aphasia Battery–Revised (WAB) 36 : The WAB includes subtests of spontaneous speech (content and fluency), auditory verbal comprehension (yes/no questions, auditory word recognition, and sequential commands), repetition, and naming and word finding (object naming, category fluency, sentence completion, and responsive speech). The test calculates an aphasia quotient (AQ) for each participant (Table 1), which conveys an overall measure of aphasic deficits.
Apraxia of Speech Rating Scale (ASRS 2.0) 37 : The ASRS 2.0 was used to assess motor speech on a 1 to 5 scale, with higher scores indicating more severe speech deficits.
Boston Diagnostic Aphasia Examination (BDAE) 38 : The semantic probe subtest of the BDAE was used, which is comprised of a series of yes/no questions about the category, physical features, and function of common objects.
Cognitive Linguistic Quick Test (CLQT) 34 : The CLQT includes subtests that assess nonlinguistic cognition. Here, individual scores were used from the symbol trails, design generation, and symbol cancellation subtests, as these do not rely on verbal expression.
Spatial Span 39 : To assess visuospatial working memory, participants were instructed to tap a sequence of blocks demonstrated by the test administrator, first forward then backward, with sequence length increasing from 2 to 9. The spatial span task did not rely on verbal expression and was thus more suitable than a traditional verbal digit span task for people with aphasia.
Factor Analysis Methods
In order to reduce multicollinearity, 21 cognitive impairment-based measures were entered into a principal component analysis (PCA) of the correlation matrix in SPSS 26. Tests entered into the PCA included the PNT, category and letter fluency, repetition, reading tests, WAB subtests, ASRS, BDAE, CLQT subtests, and spatial span. In order to produce clean factor scores, the WAB word recognition subtest was excluded, as it produced a complex factor loading. With 41 participants and 21 tests, the subject to variable ratio was 1.95. Although this was a relatively low ratio, lower ratios 40 and smaller sample sizes 41 than this have previously produced good factor stability. A standard eigenvalue >1 cutoff was used to determine the number of factors extracted. Varimax rotation with Kaiser normalization was used to create orthogonal factors and the rotated component matrix was used to examine the loading of each test on the extracted factors. Factor scores were calculated for all participants using the regression method to weight each test proportionally to its loading score on each factor. Thus, tests were not assigned to particular factors. After reviewing the tests that loaded onto each factor, factors were named according to the primary construct measured by the tests.
Regression Analysis Methods
A stepwise multiple linear regression was performed to determine significant predictors of the average SAQOL-39g score and each of the three domain scores (physical, communication, and psychosocial) using SPSS 26. The independent variables entered into each regression included the factor scores from the PCA described above, SADQ scores, motricity index scores, participant age, gender, race, years of education, time since stroke, and lesion volume. Assumptions of homoscedasticity, linearity, normally distributed residuals, independent errors, multicollinearity, and influential outliers were checked for all four regressions. During the iterative stepwise selection process, independent variables were entered into the model if P ≤ .05 and removed from the model if P ≥ .10.
Imaging Methods
High-resolution 3-dimensional T1-weighted MPRAGE images were acquired on a 3.0-T Siemens TIM Trio scanner within 1 week of behavioral testing, with the following parameters: repetition time TR = 1900 ms; echo time TE = 2.56 ms; flip angle = 9°; 160 contiguous 1 mm sagittal slices; field of view = 250 × 250 mm; matrix size = 246 × 256, voxel size = 1 mm3. Additionally, a T2-weighted sampling perfection with application optimized contrasts using different flip angle evolution (SPACE) sequence was acquired with the following parameters: 176 sagittal slices; slice thickness = 1.25 mm, FOV = 240 × 240 mm; matrix = 384 × 384; TR = 3200 ms; echo train length = 145, variable TE; variable flip angle, voxel size = 0.625 × 0.625 × 1.25 mm.
Lesion Tracing Methods
All participants with lesions that could be segmented and without significant damage outside the left-hemisphere were included in the neuroimaging analyses (38 participants total). One participant was excluded for lack of magnetic resonance imaging data, one with intracerebral hemorrhage was excluded because tissue shifts made the lesion impossible to accurately segment, and one was excluded because the lesion included a part of the right anterior cerebral artery territory. These participants were not excluded from the factor and regression analyses, because these analyses were separate from the neuroimaging analysis. Lesion masks were manually segmented on each participant’s MPRAGE using ITK-SNAP software 42 (http://www.itksnap.org/) by P.E.T., a board-certified neurologist. T2-weighted structural images were referenced for additional sensitivity to white matter lesions within the vascular distribution of the stroke. Lesion tracings were warped to the Clinical Toolbox Older Adult Template 43 using a custom pipeline in Advanced Normalization Tools software 44 (ANTs; http://stnava.github.io/ANTs/). Full details are provided elsewhere. 45
Lesion-Symptom Mapping Methods
A multivariate lesion-symptom mapping (LSM) technique called support vector regression LSM (SVR-LSM) 46 was used to localize brain areas that are related to subjective reports of quality of life. Given the nonrandom nature of lesion distributions and the resultant autocorrelation of voxel lesion status, univariate methods such as voxel-based LSM (VLSM) 47 are vulnerable to mislocalization, especially when multiple brain areas support the process of interest. 48 SVR-LSM considers the lesion status of all voxels in a single regression model and is less vulnerable to lesion mislocalization and more sensitive to nonlinear relationships. 46 SVR-LSM was applied in MATLAB R2019a via a graphical user interface implementation developed in our laboratory 49 (https://github.com/atdemarco/svrlsmgui/). Only voxels lesioned in at least 10% of participants were included in each analysis to avoid areas with sparse lesion coverage reducing statistical power. Effects of lesion volume on SAQOL-39g were controlled for by regressing lesion volume on both the lesion data and the SAQOL-39g scores, a method that eliminates lesion volume biases while maintaining sensitivity to lesion-behavior relationships. 49 Four one-tailed (negative) SVR-LSM analyses were run to identify lesions associated with lower average, physical, communication, and psychosocial scores of the SAQOL-39g. Significance was determined using a permutation-based approach in which the domain scores were randomly reassigned to participants and SVR-β-value maps were generated for each of 10 000 permutations. SVR-β values were catalogued on a voxelwise basis and thresholded at P < .005. Because the permutation method determines P values by ranking the voxelwise empirical SVR-β-values among the permuted SVR-β-values at the same voxels, a large number of permutations is needed to reliably measure very small P values. To correct the P-maps for multiple comparisons, a cluster size threshold was applied to achieve a familywise error rate of 0.05 based on the largest cluster in each of the voxelwise thresholded permutation maps.
Results
Factor Analysis
The principal component factor analysis of 21 behavioral tests generated 3 factors that accounted for 72% of the variance in participant test scores (Table 2). See Supplemental Figure 1 for a scree plot. Tests of language production primarily loaded onto factor 1, tests of nonlinguistic cognition primarily loaded onto factor 2, and tests of auditory comprehension primarily loaded onto factor 3.
Factor Loadings for All Behavioral Tests. a
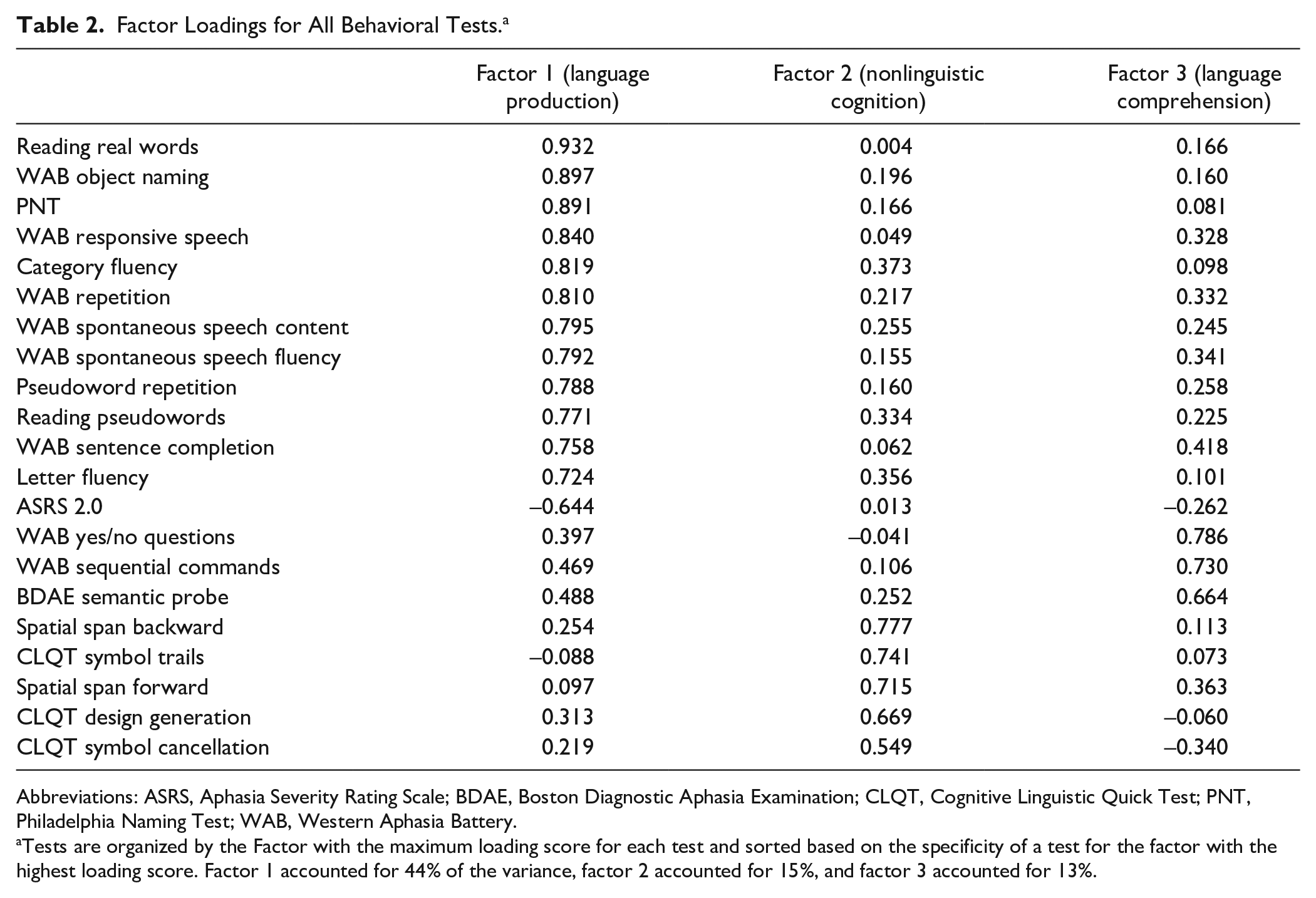
Abbreviations: ASRS, Aphasia Severity Rating Scale; BDAE, Boston Diagnostic Aphasia Examination; CLQT, Cognitive Linguistic Quick Test; PNT, Philadelphia Naming Test; WAB, Western Aphasia Battery.
Tests are organized by the Factor with the maximum loading score for each test and sorted based on the specificity of a test for the factor with the highest loading score. Factor 1 accounted for 44% of the variance, factor 2 accounted for 15%, and factor 3 accounted for 13%.
Regression Analysis
Bivariate correlations for all continuous variables are presented in Supplemental Table 2. These correlations did not inform the regression analyses but have been included in the supplemental material for readers that may be interested. For all regressions, assumptions of homoscedasticity, linearity, and normally distributed residuals were met. Errors were independent as indicated by Durbin-Watson values greater than 1 and less than 3. Multicollinearity assessment showed that all variance inflation factors were less than 2. There were no influential outliers as indicated by maximum Cook’s distance less than 1 for all regressions. The stepwise multiple linear regression analysis examining SAQOL-39g average score identified motricity index, t(34) = 4.12, P < .001, and the SADQ, t(34) = −2.94, P = .006, as significant predictors, R2 = 0.469, F(2, 32) = 14.14, P < .001). The regression examining the physical domain also identified motricity index, t(34) = 7.09, P < .001, and the SADQ, t(34) = −2.09, P = .044, as significant predictors, R2 = 0.645, F(2, 32) = 29.10, P < .001. The regression examining the communication domain identified factor 1 (language production), t(34) = 5.92, P < .001, and the SADQ, t(34) = −2.71, P = .010, as significant predictors, R2 = 0.569, F(2, 32) = 21.11, P < .001. The regression examining the psychosocial domain identified the SADQ, t(34) = −2.52, P = .017, as the only significant predictor, R2 = 0.161, F(1, 33) = 6.34, P = .017. In order to allow for a clean interpretation by readers that may be interested, these regressions were reanalyzed using the same 38 participants from the neuroimaging analyses. These regression analyses yielded the same pattern of results as the analyses using 41 participants, with the exception that the SADQ was not a significant predictor of the physical domain.
SVR-LSM
A lesion overlap map (Figure 1; n = 38) demonstrated broad coverage of left frontal, parietal, and temporal areas, with maximal overlap in the insula and frontoparietal white matter. SVR-LSM identified a significant association between lower average scores on the SAQOL-39g and lesions to inferior frontal cortex, anterior insula, putamen, caudate, and deep white matter that was independent of overall lesion volume (Figure 2; volume = 27 640 mm3; peak MNI [Montreal Neurological Institute] coordinates = −25, 5, −6; center of mass = −34, 15, 8). Lower scores on the physical domain were significantly associated with lesions to the putamen, caudate, and deep white matter, independent of overall lesion volume (Figure 3; volume = 13 859 mm3; peak MNI coordinates = −30, 0, −9; center of mass = −26, 7, 7). Because hemorrhagic strokes tend to affect the basal ganglia, and could potentially bias these results, the SVR-LSM analysis was rerun without the four hemorrhagic participants. This analysis yielded highly overlapping results, confirming that the basal ganglia findings were not driven by hemorrhagic strokes. Lower scores on the psychosocial domain were significantly associated with lesions to the inferior frontal gyrus (IFG) and anterior insula, independent of overall lesion volume (Figure 3; volume = 17 750mm3; peak MNI coordinates = −27.5, 25, −6; center of mass = −40.9, 22.7, 9.8). There were no areas in which lesions significantly related to lower scores on the communication domain.
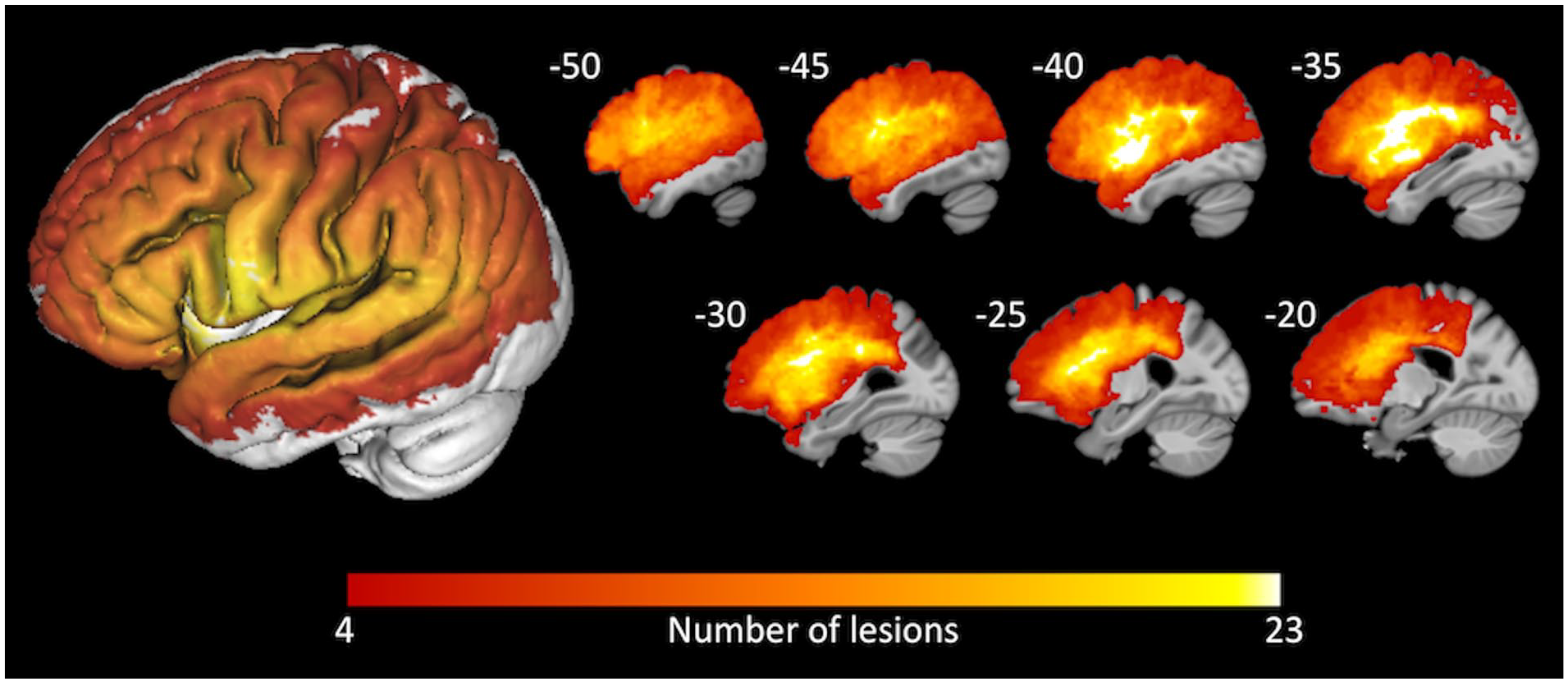
An overlay of the 38 participants’ lesion masks demonstrates the distribution of left-hemisphere areas included in the support vector regression lesion-symptom mapping (SVR-LSM) analysis. A three-dimensional rendering of the overlay is shown on the left. Sagittal slices are shown on the right, at the Montreal Neurological Institute (MNI) x-coordinates shown.
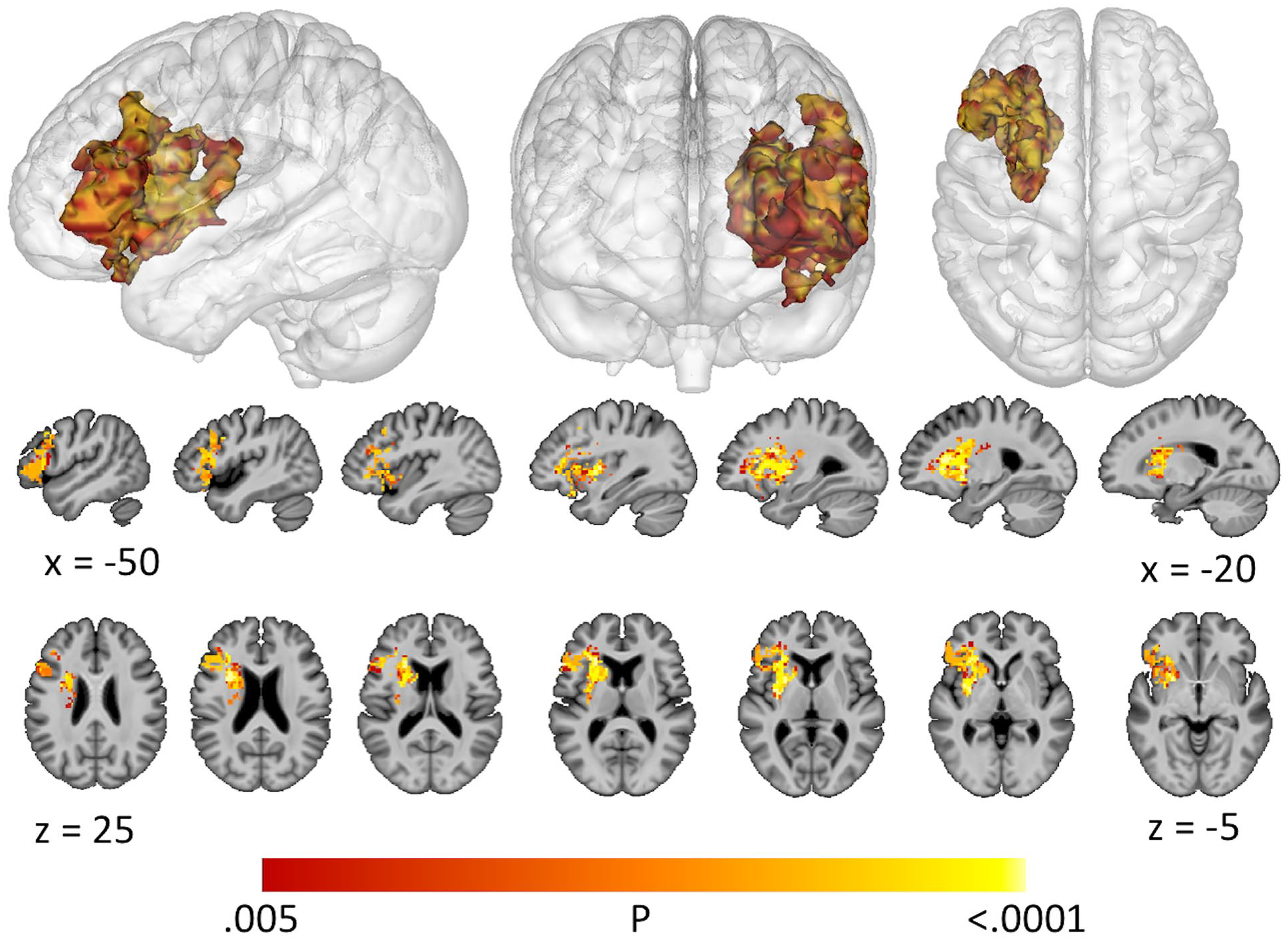
Support vector regression lesion-symptom mapping (SVR-LSM) results for the Stroke and Aphasia Quality of Life Scale (SAQOL-39g) average score, shown at voxelwise P < .005 and clusterwise P < .05.
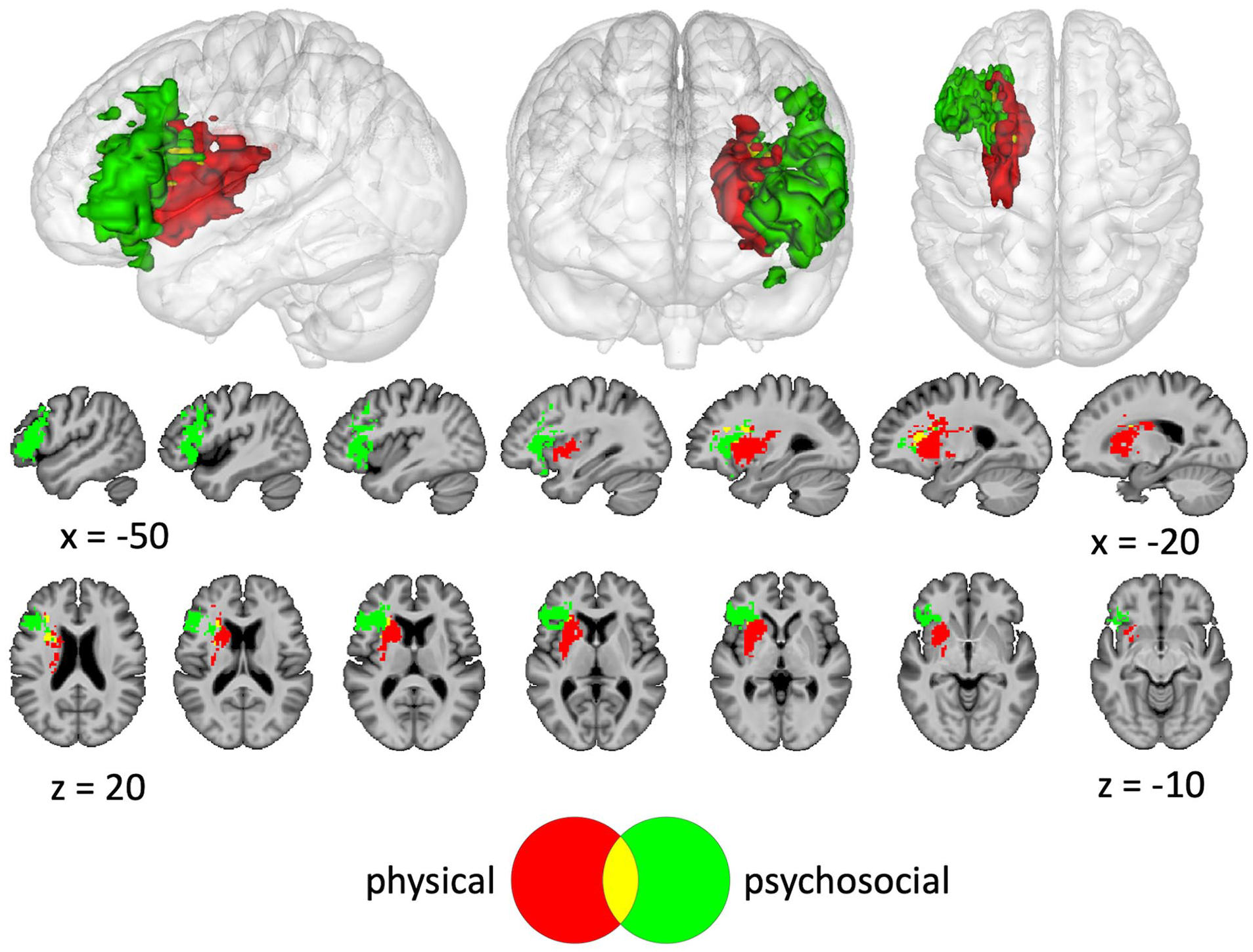
Support vector regression lesion-symptom mapping (SVR-LSM) results for the physical and psychosocial domains of the Stroke and Aphasia Quality of Life Scale (SAQOL-39g), shown at voxelwise P < .005 and clusterwise P < .05.
Discussion
In order to identify targeted rehabilitation plans that may benefit individual stroke survivors with aphasia, it is important to understand factors that relate to the subjective perception of one’s ability to lead a fulfilling life. The current study first explored behavioral variables related to the SAQOL-39g in left-hemisphere stroke survivors with aphasia and demonstrated that depression symptoms predicted lower average and domain scores, lower motricity scores predicted lower average and physical domain scores, and lower Language Production factor scores predicted lower communication domain scores. Second, the current study applied SVR-LSM to examine areas of brain damage related to the SAQOL-39g and showed that basal ganglia lesions were associated with lower physical domain scores, and inferior frontal lesions were associated with lower psychosocial domain scores.
Symptoms of depression related to the average SAQOL-39g score, as well as all domain scores. This result is in line with prior research showing significant associations between depression and HRQL in stroke survivors.6-8,12,14 The current study lends additional support to existing guidelines highlighting the need for ongoing assessment and treatment of depression during stroke rehabilitation. 2 Future studies of poststroke depression should include people with aphasia, as this group is often excluded despite being at high risk for depression. Although there is some evidence for effective treatments of mild depression in people with aphasia, there is currently a lack of evidence for treatment of people with moderate to severe depression, and limited evidence regarding preventative methods. 50 Future work addressing these issues is essential for developing effective strategies to improve HRQL in stroke survivors with aphasia.
The finding that lower scores on the motricity index predicted lower SAQOL-39g average scores is consistent with prior research demonstrating associations between HRQL and measures of physical function.4,6-8,12,14,20 It also aligns with a previous analysis of SAQOL-39g psychometric properties, which revealed that the physical and psychosocial domains contribute more to the overall score than the communication domain. 24 Additionally, this result is compatible with findings that the physical domain of the SAQOL-39 is rated as significantly more important than all other domains by people with aphasia, their significant others, and their speech-language pathologists. 51 The motricity index was also a significant predictor of physical domain scores, corresponding with findings that physical ability is related to physical components of HRQL measures.14,20 Collectively, these findings provide compelling evidence that arm motor impairment is related not only to the physical aspect of well-being but also to overall HRQL. These relationships may reflect the necessary role of arm use for basic activities of daily living and independence and provide support for an emphasis on physical rehabilitation during recovery.
The finding that lower factor 1 (Language Production) scores predicted lower communication domain scores is consistent with prior studies that reported associations between communication components of HRQL and objective language measures.13-15 The current study adds to the literature by distinguishing between language comprehension and language production, and suggests that production, rather than comprehension, is related to the communication domain of the SAQOL-39g. Items in the communication domain are oriented toward production, providing evidence that self-reports on the SAQOL-39g communication domain align with objective measures of impairment. Given that no items are oriented toward comprehension, it is not possible to determine from these results whether language production is more important to HRQL than language comprehension, or whether this finding simply reflects the nature of the SAQOL-39g. To address this question more fully in the future, it may be beneficial to utilize measures of HRQL that include spoken and written language comprehension. 52
The finding that lesions to the basal ganglia were associated with lower scores in the physical domain aligns with the well-known role of the basal ganglia in motor control. 53 The overlap in lesions associated with both psychosocial and physical domains may reflect items on the psychosocial domain that could require mobility (eg, “during the past week did you do your hobbies and recreation less often than you would like” and “during the past week did you feel that your physical condition interfered with your social life”). The relationship between the psychosocial domain and lesions to the anterior insula is consistent with the involvement of the anterior insula in subjective feelings and experiences. 54 Lower scores in the psychosocial domain additionally localized to lesions in the IFG, a region canonically linked with expressive language functions. 55 However, research has also demonstrated that the left IFG is involved in psychological resilience 56 and emotion regulation, 57 which are related to psychosocial well-being.58,59 Although it is possible that some items in the psychosocial domain may require language expression thereby explaining the left IFG localization, this seems unlikely, as items explicitly related to language expression fall under the communication domain. This is in contrast to the previously mentioned psychosocial items that may require mobility, for example, “during the past week did you feel that your physical condition interfered with your social life” is in the psychosocial domain, while “during the past week did you feel that your language problems interfered with your family life” is in the communication domain. Notably, no lesions were identified that related to the communication domain. This is possibly because regions throughout the left hemisphere are associated with speech and language, 60 and communication impairments in this participant sample were likely the result of varied lesion locations. Overall, these results add to the literature by demonstrating that lesions to discrete left-hemisphere regions are related to reduced physical and psychosocial HRQL as measured by the SAQOL-39g. These findings may provide support for early initiation of targeted interventions to improve HRQL based on lesion location. For example, future studies could explore whether it is beneficial to initiate depression prevention methods in stroke survivors with left inferior frontal lesions. However, further research is needed with larger sample sizes and broader distributions of lesion coverage to completely characterize relationships between HRQL and lesion location.
Some limitations of this study are worth noting. First, the sample size may not have provided sensitivity to detect all possible lesion-symptom or behavioral relationships. The analysis is only sensitive to effects in a restricted part of the brain, as shown in Figure 1. As mentioned previously, the subject to variable ratio for the PCA was relatively low. Depression was assessed with the SADQ, which is designed to be administered to caregivers. Therefore, scores on the SADQ may reflect the mood of the caregiver rather than the participant. 31 The use of the motricity index only captured upper-limb impairment, whereas the physical domain of the SAQOL-39g is likely affected by both upper and lower limb impairment. Some factors shown to relate to HRQL in prior studies, such as socioeconomic status, fatigue, and social support, were not measured.
Conclusions
A better understanding of the relationship between objective variables and HRQL in stroke survivors could lead to more tailored treatments to improve individual outcomes. The current study has demonstrated that objective measures of impairment and specific lesion locations relate to subjective self-reports of specific aspects of HRQL in left-hemisphere stroke survivors with aphasia. These results help to validate domain measures of HRQL and highlight areas where additional research on and refinement of HRQL measures are needed.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683211017507 – Supplemental material for Domains of Health-Related Quality of Life Are Associated With Specific Deficits and Lesion Locations in Chronic Aphasia
Supplemental material, sj-docx-1-nnr-10.1177_15459683211017507 for Domains of Health-Related Quality of Life Are Associated With Specific Deficits and Lesion Locations in Chronic Aphasia by Elizabeth L. Dvorak, Davetrina S. Gadson, Elizabeth H. Lacey, Andrew T. DeMarco and Peter E. Turkeltaub in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We would like to thank Mackenzie Fama for contributing to data collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Doris Duke Charitable Foundation (2012062), NIH/NCATS KL2TR000102 to P.E.T., NIH/NIDCD R01DC014960 and R01DC014960-03S1 to P.E.T., and NCATS TL1TR001431 to A.T.D.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
