Abstract
Background and purpose. A single nucleotide polymorphism at nucleotide 196 (G/A) in the human brain-derived neurotrophic factor (BDNF) gene produces an amino acid substitution (valine to methionine) at codon 66(Val66Met). It is unclear whether carriers of this substitution may have worse functional outcomes after stroke. We aimed to explore the distribution of Val66Met polymorphism and evaluate the effect of different genotypes on stroke functional recovery. Methods. Several databases were searched using the keywords BDNF or brain-derived neurotrophic factor, codon66, G196A, rs6265, or Val66Met, and stroke. Results. A total of 25 articles were relevant to estimate the distribution of alleles; 5 reports were applied in the meta-analysis to assess genetic differences on recovery outcomes. The genetic model analysis showed that the recessive model should be used; we combined data for AA versus GA+GG (GG—Val/Val, GA—Val/Met, AA—Met/Met). The results showed that stroke patients with AA might have worse recovery outcomes than those with GA+GG (odds ratio = 1.90; 95% CI: 1.17-3.10; P = .010; I2 = 69.2%). Overall, the A allele may be more common in Asian patients (48.6%; 95% CI: 45.8%-51.4%, I2 = 54.2%) than Caucasian patients (29.8%; 95% CI: 7.5%-52.1%; I2 = 99.1%). However, in Caucasian patients, the frequency of the A allele in Iranians (87.9%; 95% CI: 83.4%-92.3%) was quite higher than that in other Caucasians (18.7%; 95% CI: 16.6%-20.9%; I2 = 0.00%). Conclusion. Val66Met AA carriers may have worse rehabilitation outcomes than GA+GG carriers. Further studies are needed to determine the effect of Val66Met polymorphism on stroke recovery and to evaluate this relationship with ethnicity, sex, age, stroke type, observe duration, stroke severity, injury location, and therapies.
Introduction
Stroke is the third leading cause of death worldwide. Annually, more than 10 million individuals experience a major stroke. 1 In developed countries, stroke is the second leading cause of death; however, in some countries, such as Japan and China, stroke has become the leading cause of death since 2013. 2 Stroke can lead to permanent disability and is followed by a difficult, long recovery. 3 Moreover, the prognosis of patients with stroke is poor, and neurological degeneration is the major cause of early mortality (within 1 month). 4 Rehabilitation after stroke is critical and can be affected by multiple factors; thus, improved accuracy of recovery prognosis may allow realistic therapeutic goal-setting and medical resource allocation. 5 Such improvements may lead to effective and individualized rehabilitation strategies, consistent with the notion that rehabilitation strategies should provide specific, tailored treatments for individuals. 6
Brain-derived neurotrophic factor (BDNF) modulates axon growth, cell fate, cell shape, neuroplasticity, differentiation, and angiogenesis in normal and ischemic tissues.7-9 Neuroplasticity is considered to be the mechanism of neurological rehabilitation. 10 BDNF is secreted in the central nervous system (CNS) through a constitutive and activity-dependent pathway, and activity-dependent secretion is crucial to the function of BDNF in promoting neuroplasticity. 11 BDNF and its receptor are actively produced and trafficked in multiple regions in the adult brain, 12 and its expression is different between brain structures. 13 Genetic mutation may affect BDNF expression. The presence of the Met allele (an amino acid change from valine[Val] to methionine[Met] at codon66 [Val66Met]) may result in a reduction in the activity-dependent secretion of BDNF in the CNS,14,15 which may downgrade the function of BDNF in neuroplasticity and influence the recovery of the neurological damage in patients with stroke, leading to poorer outcomes in these patients compared with the outcomes in patients without amino acid changes (ValVal carriers).16,17 Moreover, because BDNF affects stroke recovery and the nervous system, it has attracted increasing interest in scientists investigating stroke recovery.
Recently, some studies stated that Met carrier patients are at a disadvantage on stroke recovery,18,19 whereas other studies did not.20,21 Zhou et al 22 found that being an AA carrier was related to the occurrence of stroke in patients with large atherosclerosis, but results showed no correlation between the polymorphism and prognosis of stroke. Besides, the reported distributions of BDNF Val66Met polymorphism were different among stroke patients with different ethnicities, 23 which may influence the association of this polymorphism and stroke recovery. Moreover, exploring the distribution of Val66Met polymorphism in stroke patients with different ethnicities can partly explain the difference in the results of published studies and provide clues for future research. Therefore, in this study, we summarized current original studies and conducted a meta-analysis to provide more precise and comprehensive evidence of the association between Met homozygote carriers (individuals with the AA genotype) and Val carriers (individuals with the GA or GG genotype) in terms of stroke functional recovery. Additionally, we summarized the current original articles to explore the distribution of Val66Met polymorphism of stroke patients in ethnicity.
Material and Methods
Article Retrieval
The current research strategy and meta-analysis protocol followed the Meta-analysis Of Observational Studies in Epidemiology (MOOSE) guidelines, 24 which are specific for observational studies. Articles were retrieved by searching the English databases PubMed, EMBASE, Ovid, and Wiley Online Library as well as the Chinese databases of China National Knowledge Infrastructure (CNKI), WANFANG DATA, and China Science and Technology Journal Database (VIP), using the keywords BDNF or brain-derived neurotrophic factor; codon66, G196A, rs6265 or Val66Met; and stroke. All articles were published before May 4, 2020. We also checked the reference lists of the identified articles to find more studies that are pertinent.
The PICO strategy 25 was used to set criteria to identify relevant studies for this review: P (patient/population): stroke patients; I (intervention): Val66Met polymorphism in BDNF; C (comparison): different genotypes were compared with each other; O (outcomes): functional recovery after stroke.
First, we described and combined data for the distribution of BDNF Val66Met polymorphisms in patients with stroke. The inclusion criteria were as follows: (1) studies focused on investigating the relationships between BDNF Val66Met and stroke and (2) articles providing the distribution of BDNF Val66Met genotypes in patients with stroke. The exclusion criteria were as follows: (1) reviews and animal studies; (2) articles focused on the BDNF serum level, not on BDNF gene polymorphisms; and (3) articles providing no distribution information of BDNF Val66Met polymorphisms.
Second, we included studies to compare the differences in stroke recovery associated with different genotypes meeting the following criteria: (1) original case-control studies of the effects of BDNF Val66Met polymorphisms on the influence of stroke recovery; (2) studies containing available and sufficient data to allow estimation of the pooled odds ratio (OR) and 95% CI; and (3) studies providing certain evaluation scales (National Institutes of Health Stroke Scale [NIHSS], Modified Rankin Scale [mRS], and Barthel Index [BI]) to reflect functional outcomes. When several publications were reported on the same or overlapping data, we chose the most recent article or the article that included the most massive study population. The exclusion criteria were as follows: (1) reviews, repeated and overlapping articles, and animal experiments; (2) studies without sufficient data to estimate the pooled OR and 95% CI; (3) studies providing one-sided information on rehabilitation, such as aphasia only; and (4) studies of the relationships of BDNF serum levels and stroke recovery.
Outcome Measurement
According to the World Health Organization’s International Classification of Functioning, Disability and Health, 26 stroke functional outcomes can be classified into 4 aspects: pathology (disease or diagnosis), impairment (symptoms and signs), activity limitations (disability), and participation restriction (handicap). In the present study, we were mainly concerned with neurological impairments and activity limitations, which can be assessed by several evaluation scales. (1) NIHSS: standardizes and quantifies the basic neurological examination; it is scored from 0 (no impairment) to 42. Those who score 21 or greater are generally considered as having “severe” impairment. (2) mRS: a scale scored from 0 to 6 and evaluates “global disability” in terms of mobility; the cutoff for the good and poor functional recovery groups is often set between 1 and 2. Patients who score 0 to 1 are considered as having minimal or no disability. (3) BI: this evaluates 10 functional tasks that focus on daily living (activities of daily living); scores range from 0 to 100. Participants with scores greater than 80 are usually considered as independent, whereas participants with scores less than 40 are very dependent.27,28
Quality Assessment
Two independent researchers performed the literature search, data extraction, and article identification, and disagreements were overcome by consulting a third expert. For missing data and uncertain information, we contacted the original author.
Quality assessment was conducted for each article using the Strengthening the Reporting of Genetic Association studies (STREGA) for original studies. Higher STREGA scores indicated better article quality, and the article quality was considered as poor when STREGA scores were lower than 7. Poor quality articles were moved out of the assessment.
Statistical Analysis
We extracted data regarding first author, publication year, country, inclusion criteria and exclusion criteria, time of outcome assessment, gender ratio, sample size, type of stroke, and genotypes for differences between genotypes from articles as the primary article information. We then used the STREGA scale to evaluate article quality. In the meta-analysis, the pooled allele frequency was used for distribution analysis of BDNF Val66Met polymorphisms in patients with stroke. The pooled OR, with 95% CI, was the primary metric of interest to calculate the association between BDNF Val66Met and stroke recovery outcome. (OR greater than 1 was interpreted as a negative relationship; OR between 0 and 1 was interpreted as a positive relationship; and OR equal to 1 was interpreted as an irrelevant relationship). The fixed-effects model was used for meta-analysis when the I2 was <50% and P was >.1; otherwise, the random-effects model was used; I2 is a standard statistic used to assess heterogeneity, generated through a specific formula, I2 = {[Q − (K − 1)]/Q} × 100%. Here, K is the number of combined studies and Q is the statistic of Q tests, which was performed to evaluate the heterogeneity. 29 Sensitivity analysis was performed to determine how robust the results are and to exclude decisive articles, and the Egger test was used to evaluate publication bias. Results with P values <.05 were considered statistically significant, indicating the presence of publication bias. 29 The genetic model analysis was performed, and the outline of the procedure (Figure 1) was followed to determine the most suitable model (reference group is considered the AA genotype and risk allele the allele B). 30 The parameters θ2 and θ3 in Figure 1 can be viewed as the typical coefficients in logistic regression, which when exponentiated yield the overall estimate for the odds ratio associated with each genotype: ORAB/AA = exp(θ2); ORBB/AA = exp(θ3). 30 The genetic model is determined by testing the difference of θ2 and θ3. For example, the A dominant genetic model would be used when there is a nonsignificant P value (>.05) that indicates the equality of the log ORs. All the above analyses were performed using STATA 12.0 software with the meta-analysis module.
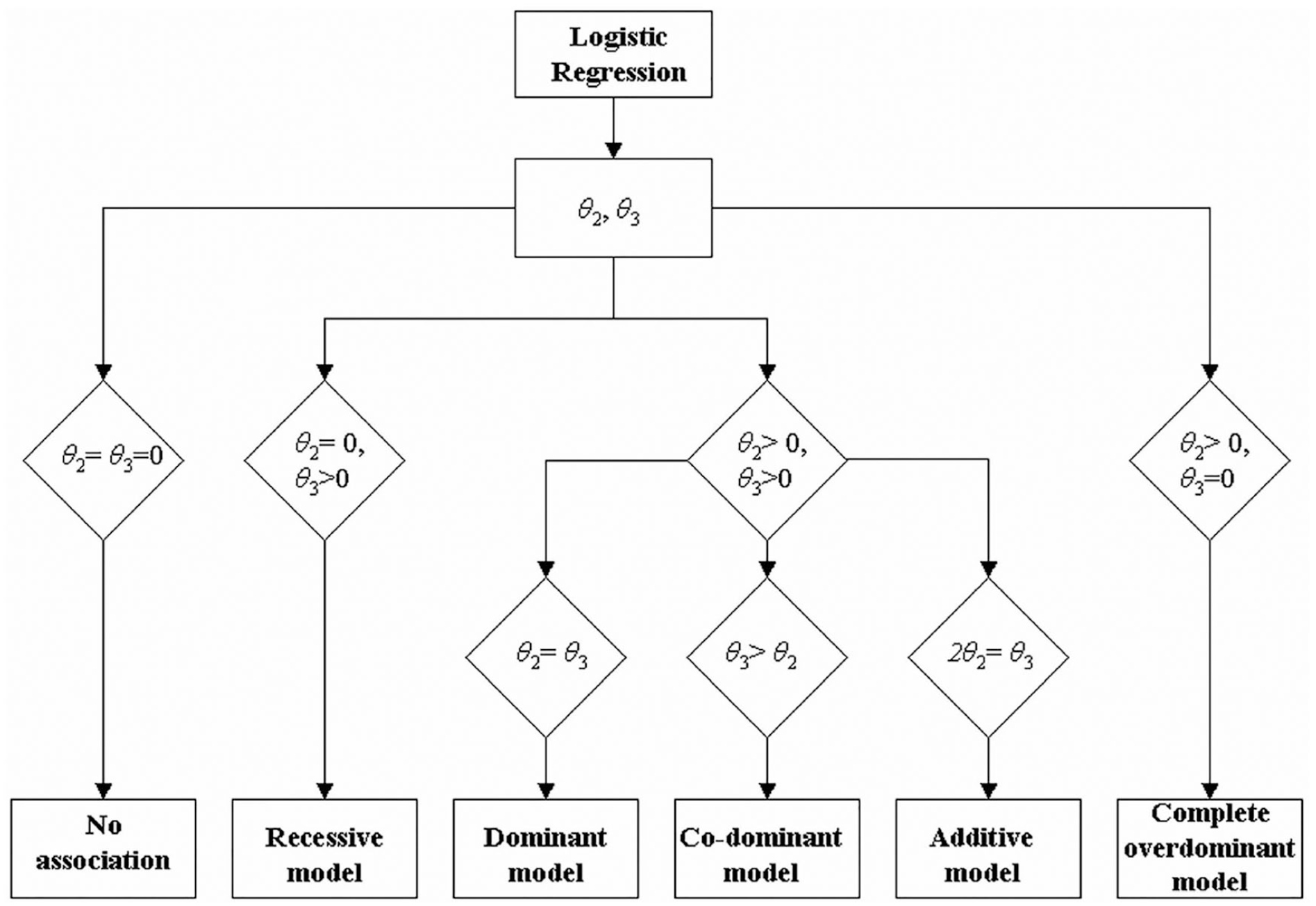
Outline of the procedure followed in order to decipher the most plausible genetic model.
Results
From our initial database search, 646 records were identified (311 from PubMed, EMBASE, Ovid, and Wiley Online Library and 320 from CNKI, WANFANG DATA, and VIP; 15 more studies were retrieved from related citations). After excluding articles according to the inclusion and exclusion criteria, 25 reports18-22,31-50 were included in the qualitative description and the analysis of the distributions of alleles of BDNF Val66Met in patients with stroke. Five reports19,20,22,38,45 including 2089 participants were applied in the meta-analysis to assess the differences in recovery outcomes in patients with stroke having different genotypes (Figure 2).
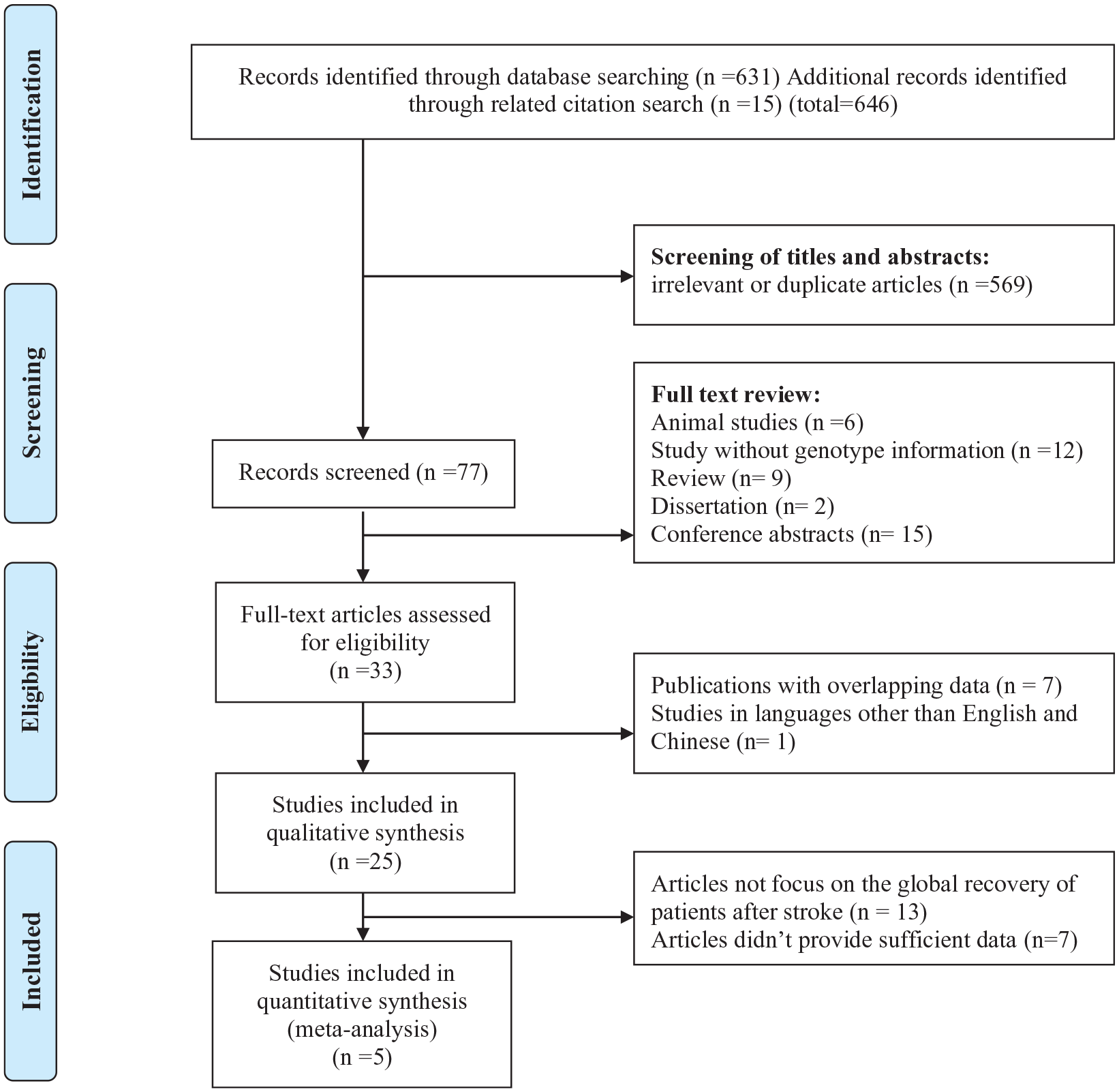
Flowchart of the selection process in the meta-analysis.
Qualitative descriptions of the distributions of genotypes and alleles for BDNF Val66Met on stroke recovery by article are listed in Table 1. Overall, most studies supported that Met carriers probably have poorer recovery after stroke; some studies showed that there were no significant differences or Val carriers might have poorer recovery after stroke. Regarding different aspects of poststroke recovery, we found that most studies indicated Met carriers showed poorer motor recovery, but we lacked enough articles to describe other aspects of poststroke recovery. The distributions of genotypes varied according to ethnicity. Thus, we performed a meta-analysis to explore the differences in the distribution of allele G and A for different ethnicities by subgroup analysis (see Table 2).
Distribution of Patients’ Genotype and Allele and a Qualitative Description of Articles Related to BDNF Val66Met Polymorphism on Stroke Recovery.
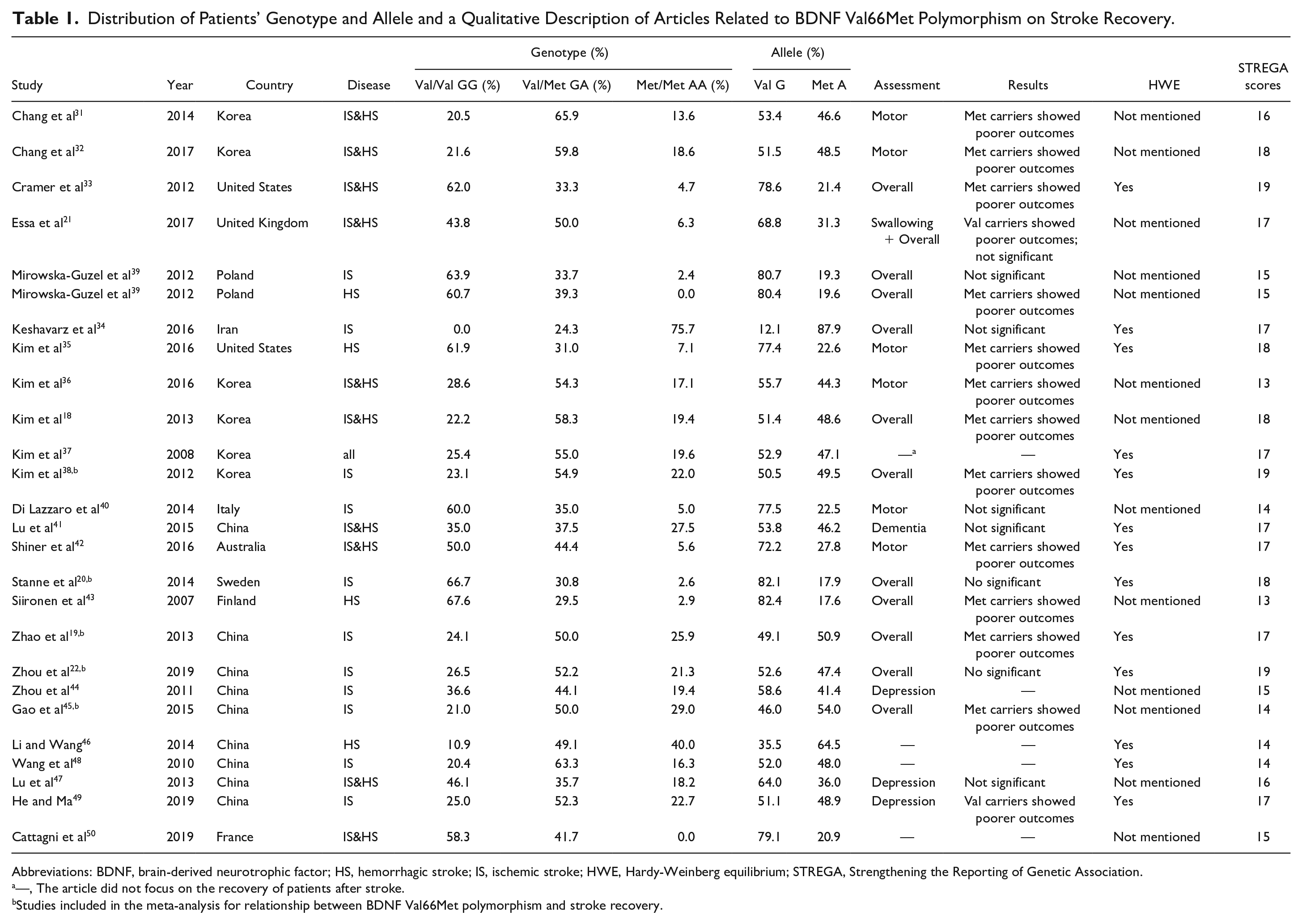
Abbreviations: BDNF, brain-derived neurotrophic factor; HS, hemorrhagic stroke; IS, ischemic stroke; HWE, Hardy-Weinberg equilibrium; STREGA, Strengthening the Reporting of Genetic Association.
—, The article did not focus on the recovery of patients after stroke.
Studies included in the meta-analysis for relationship between BDNF Val66Met polymorphism and stroke recovery.
Summary of Combined Distribution of Allele G and A Through Meta-analysis in Subgroups and Sensitivity Analysis. a

Abbreviation: ES, Effect Size.
Blank cells mean that results were steady.
We listed the 1 Iranian article separately because it was the most influential study; I2 cannot be calculated because there was only 1 article.
We obtained 15 articles reporting Asian patients, 7 articles reporting Caucasian patients, and 3 articles reporting patients with different ethnicity; we took the ethnicity of patients from these 3 articles as “Mixed.” Totally, a combined A allele frequency (39.8%; 95% CI: 31.7%-47.9%; I2 = 97.5; P < .001) was lower than combined G allele frequency (60.2%; 95% CI: 52.1%-68.3%; I2 = 97.5; P < .001). This comparison was more obvious in Caucasian and Mixed patients (Table 2), which means that this substitution may be more common in Asian patients than in Caucasian patients. We noticed that heterogeneity of Caucasian articles was large, so we performed sensitivity analysis, which showed that 1 Iranian article (Keshavarz et al 34 ) influenced the results most. After removing this article, the heterogeneity disappeared, the combined A allele frequency of Caucasian patients (18.7%; 95% CI: 16.6%-20.9%; I2 = 0.00; P = .934) reduced, and the combined G allele frequency of Caucasian patients (81.3%; 95% CI: 79.1%-83.4%; I2 = 0.00; P = .934) rose.
The detailed characteristics of articles and data combined into the meta-analysis of stroke recovery are shown in Table 3. Genetic model analysis was performed; the pooled ORGA/GG and ORAA/GG were 1.21 (95% CI: 0.95-1.55) and 2.45 (95% CI: 1.74-3.45), respectively, which was an indication for a recessive model, so we combined data for AA versus GA+GG. As is shown in Figure 3, we used a random-effects model to combine all data. There was a significant difference in stroke functional recovery between the AA and GA+GG genotypes (OR = 1.90, 95% CI: 1.17-3.10, P = .010, I2 = 69.2%; Figure 3). Specifically, AA carriers may be 1.90 times more likely to have a poor prognosis than GA+GG carriers. Heterogeneity between studies was observed in the recovery comparison; sensitivity analysis (Figure 4) showed that 1 article (Gao et al 45 ) influenced the results most. After removing this article, the association still existed (OR = 1.422; 95% CI: 1.07-1.90; P = .017) and the heterogeneity disappeared (I2 = 0.00%, P = .454; Figure 5). We found that this article’s STREGA score was the lowest among the 5 articles, and individuals from the control group departed from Hardy-Weinberg equilibrium (P = .049), which was considered to contribute to the heterogeneity. The Egger test showed no publication bias in genotype comparative analysis (P = .586).
Studies Characteristics Combined Into Meta-analysis.
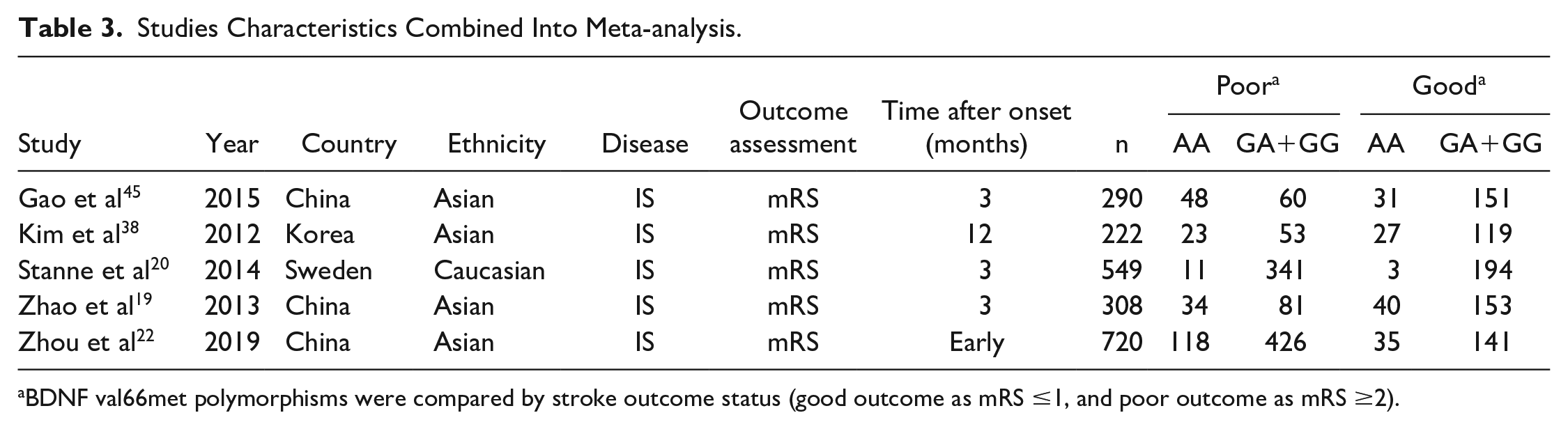
BDNF val66met polymorphisms were compared by stroke outcome status (good outcome as mRS ≤1, and poor outcome as mRS ≥2).
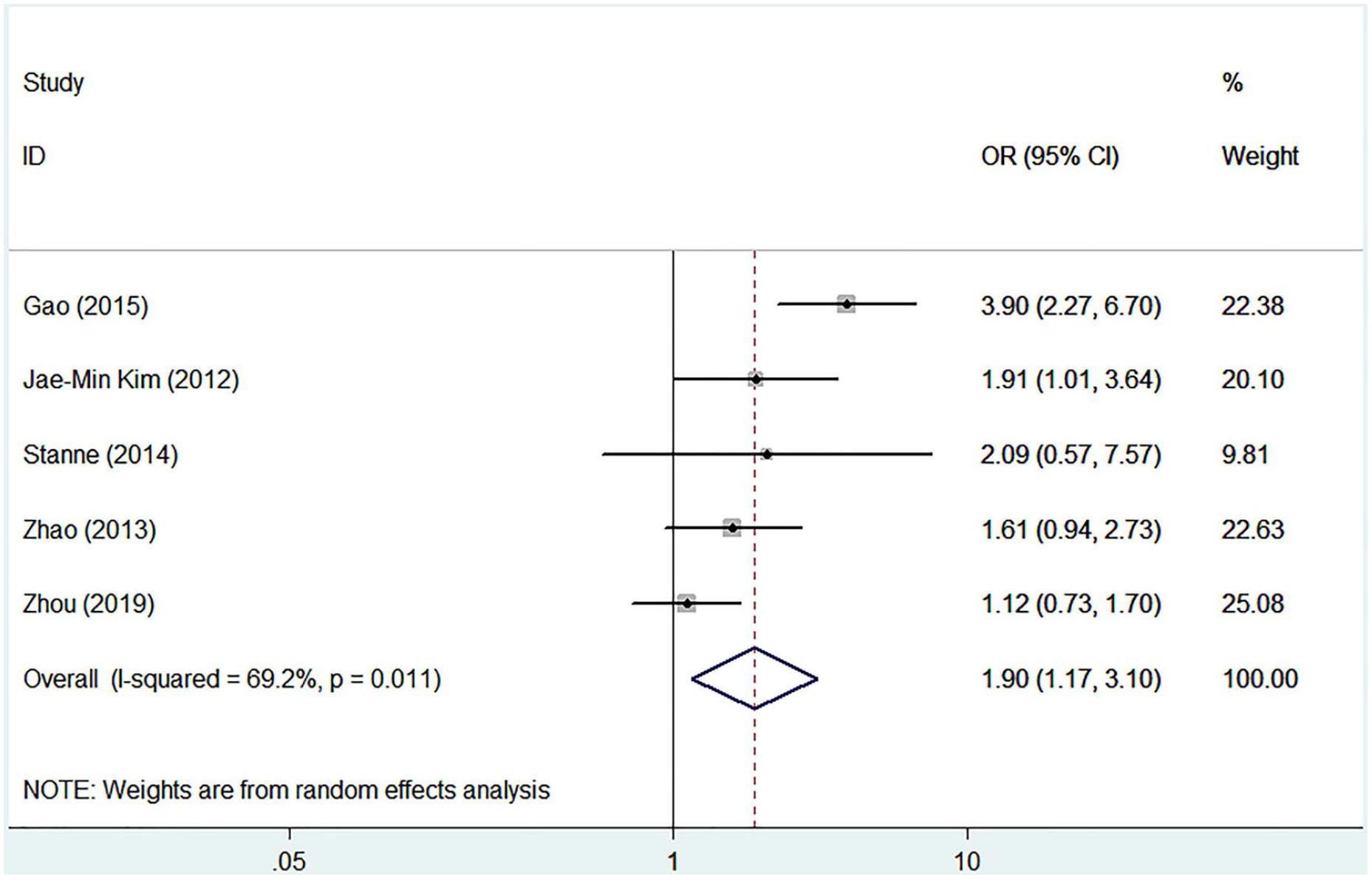
Forest plot of the combined results of the comparisons of genotypes.
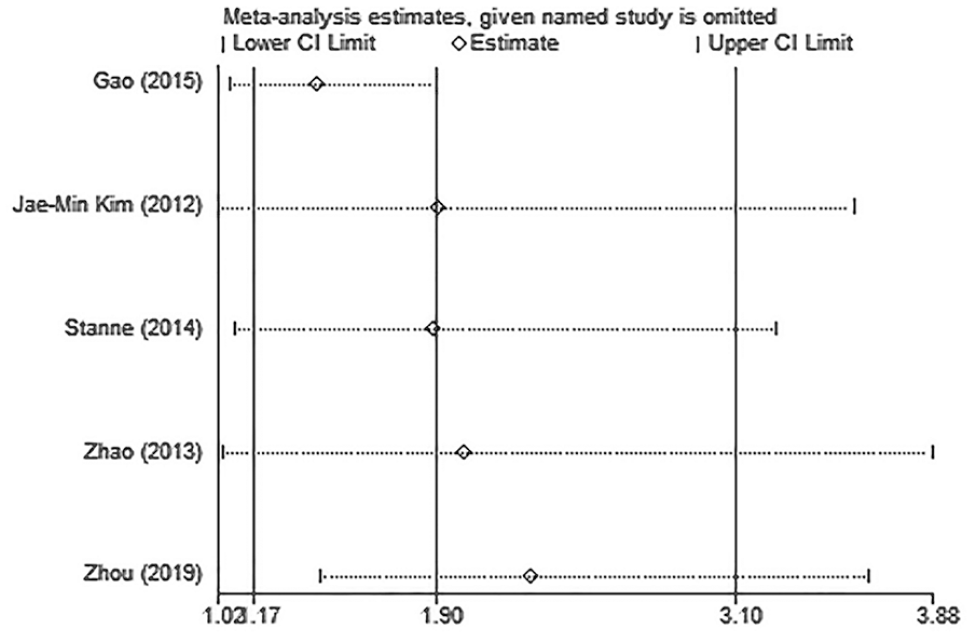
Sensitivity analysis of the influence of each study on pooled odds ratios and 95% CIs in AA versus GA+GG model for associations of brain-derived neurotrophic factor Val66Met polymorphism and stroke recovery.
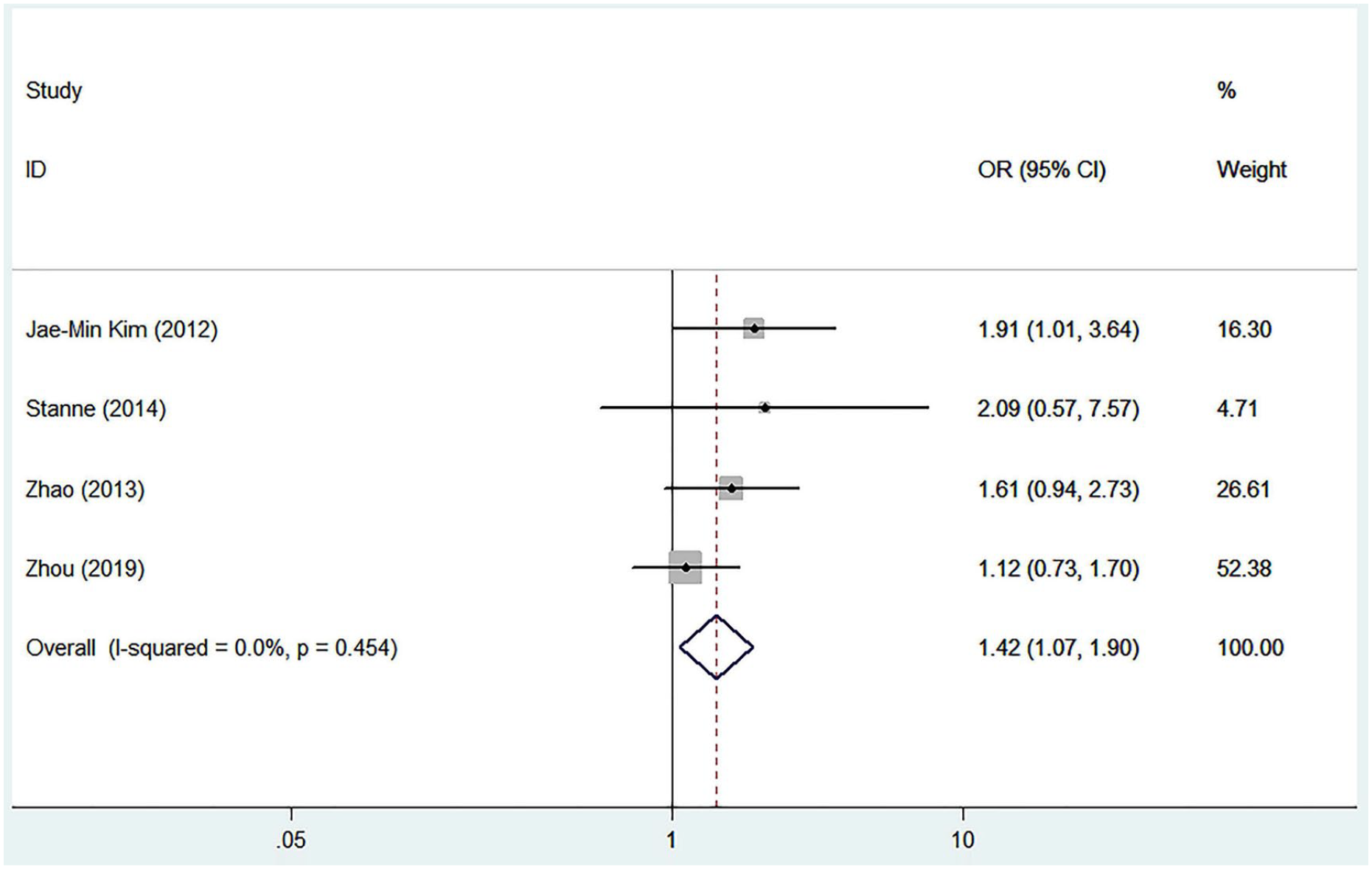
Forest plot of the combined results after removing the most influential study (Gao et al 45 ).
Discussion
The present systematic review and meta-analysis suggests that the presence of the Val66Met polymorphism may be associated with worse functional outcome after stroke. In addition, we summarized current original articles about different ethnicity and found that this polymorphism may be more common in Asian patients than in Caucasian patients generally.
Stroke rehabilitation is crucial because stroke is one of the leading causes of long-term disability. Increasing evidence suggests that genetic variation may influence stroke recovery.51,52 BDNF val66met polymorphism is the most studied single nucleotide polymorphism among the identified genetic variations. Accordingly, multiple studies have reported the relationship between Val66Met and stroke recovery. Some reviews discussed this topic,52,53 and Bao et al 23 performed a meta-analysis to evaluate the associations between rs6265 and poststroke depression, but no meta-analysis has focused on the functional outcomes. In contrast to the findings21,39 that BDNF val66met polymorphism had no impact on outcome in stroke, the current meta-analysis contains a larger sample size (2089 patients) and provides evidence for the adverse association between this polymorphism and stroke functional recovery by combining all studies with considerably different findings.
There is a debate regarding the role of Val66Met polymorphism in functional recovery after stroke. The generally accepted mechanism is that the BDNF val66met polymorphism impairs the intracellular trafficking of BDNF to the activity-dependent pathway, which leads to impaired plasticity in the brain. However, there are some other points of view. Di Pino et al 54 suggested that Met carriers and ValVal do not differ in terms of absolute ability to recover from stroke, but they do differ on the way they recover: ValVal patients may rely more on intracortical plastic processes, whereas Met carriers may rely more on subcortical plasticity. Additionally, Met carriers showed enhanced structural and functional improvement in the striatum. 7 These findings provide some plausible mechanisms about Val66Met polymorphism on stroke recovery, and further studies in humans are warranted to confirm the plausible mechanisms. Additionally, because BDNF is crucial to stroke recovery, some factors affecting BDNF may interact with the polymorphism, and exercise has been studied most. BDNF is secreted in the CNS through both a constitutive and an activity-dependent pathway. In particular, the activity-dependent pathway plays an important role in improving neuroplasticity. 14 Exercise may increase the amount of BDNF secreted by its activity-dependent pathway, resulting in an increase in motor rehabilitation and recovery, 14 whereas this effect may be attenuated in Met carriers. Hopkins et al 55 found that patients with the val66met polymorphism showed less improvement in object recognition memory than those without the polymorphism after 4 weeks of aerobic exercise. Therefore, interactions may exist between Val66Met polymorphism and other factors in stroke recovery, and more studies are needed to clarify the mechanism in the future. Considering the above evidence, doctors may adopt different rehabilitation planning for different Val66Met genotypes in the future in order to foster optimal recovery.
We also explored the allele distribution of BDNF Val66Met. In the current data, we found that this polymorphism may be more common in Asian patients than in Caucasian patients generally. We noticed that the heterogeneity of Caucasian articles was high, and the Iranian article (Keshavarz et al 34 ) was the source of heterogeneity. Notably, in this Iranian article, about 87.9% of patients were A carriers; however, the distribution of the A allele in Caucasian patients excluding Iranians was only 18.7%. Increased heterozygosity and presence of zero G allele in this study were thought to contribute to the heterogeneity. Unfortunately, only 1 article was obtained from Iran, which cannot represent the whole Iranian population; thus, further studies are needed. Additionally, the 3 studies with different ethnic patients also showed a low distribution rate of allele A, probably because most of the individuals in these studies were Caucasian. Notably, the reported distributions were similar to the results described by Shimizu et al. 56 They combined the rate of the BDNF Val66Met genotype and allele in healthy individuals from Japan, Italy, and the United States and found that the rate of the A allele was higher in Japan (41.1% for the A allele and 15.9% for the AA genotype) compared with that in Italy (29.7% for the A allele and 8.1% for the AA genotype) and in the United States (18.8% for the A allele and 4.5% for the AA genotype). The above evidence points out the critical issue of public health relevance among East Asian populations in terms of the increased genetic vulnerability to poor functional outcomes.
Our findings indicate that genetic factors may partly account for the variability in stroke functional recovery and could be added to the prognosis prediction of stroke patients in the future. Although stroke functional recovery outcome of AA patients may be worse than in patients with GA and GG in disability and handicap, it does not suggest that treatment planning for these patients is ineffective but, rather, that different treatment methods may be needed to provide individualized rehabilitation strategies, and further studies are needed to focus on the therapeutic effects of various therapies on patients with or without Val66Met polymorphism.
Like all meta-analyses, methodologies between studies often vary. Rehabilitation methods, outcome measurement, observe duration, stroke severity, and injury location varied between articles and may influence the degree of association between the polymorphism and stroke recovery; to minimize the variability in outcome measures between studies, we have categorized them as consistently as possible. In our meta-analysis, all 5 articles studied ischemic stroke and assessed the functional recovery by mRS; 3 of the 5 articles followed up patients for 3 months to investigate outcome differences, whereas the other 2 articles followed up patients for an early period and 12 months, respectively. A review 33 demonstrated that the poorer outcomes of Met carriers only occurred within the first 1 month after stroke, but Kim et al 36 and Mirowska-Guzel 16 both found that differences in outcomes were not highly significant within the first 1 month after the occurrence of stroke. Because of the insufficient number of articles included in the meta-analysis, we were unable to perform subgroup analysis of the time after stroke onset; in 4 of 5 articles, no significant association was found between severity of stroke and BDNF variants (except Gao et al 45 ), and all 5 articles mentioned that the stroke patients received standard treatment, but more detailed information was not available. Further studies focusing on these aspects are needed.
This study had some limitations. First, although we selected multiple English databases and Chinese databases, we did not include studies in languages other than English and Chinese. Additionally, some studies based on measurement data were not included because the existing data did not allow us to calculate the pooled OR and 95% CI. Moreover, we cannot conduct further analysis such as subgroup analysis because of the small number of included studies and insufficient information. This is our main limitation, and the results should be interpreted with caution. Regional diversity and ethnic differences may be associated with specific outcomes. However, few studies have evaluated this polymorphism in patients other than Asian and Caucasian. Considering the difference of Iranians from other people and that only 1 Iranian article was included, the result of the combined distribution of allele G and A cannot represent all Iranians; more studies are needed in the future to provide more comprehensive evidence on ethnicities. Finally, there may be a small bias because the current study is not registered, but we conducted this work in strict accordance with the process of the systematic review. The present research strategy and meta-analysis protocol followed the Meta-analysis of Observational Studies in Epidemiology (MOOSE) guidelines. Literature search, data extraction, and article identification were performed by 2 independent researchers, and disagreements were overcome by consulting a third expert. So in general, the study was less likely to be biased by subjective tendencies.
Notably, this meta-analysis had several strengths. Primarily, this is the first meta-analysis on the relationship between Val66Met polymorphism and stroke functional recovery. Thus, this analysis allowed for increased sample size by combining the outcomes of original articles to reduce bias and verify existing hypotheses and assumptions qualitatively and quantitatively. Second, we investigated the distribution of BDNF Val66Met in stroke patients and found that the incidence rates of the mutation (Val to Met) in Asian patients and Iranian patients were higher than in other Caucasian patients (except Iranians). Finally, we provide evidence that AA carriers may have more risk of suffering from poorer functional outcomes after stroke than GA+GG carriers.
Conclusion
In this meta-analysis, we found that Val66Met AA carriers may have poorer functional recovery outcomes than GA+GG carriers. The conclusions are limited because of the limited number of articles that can be included. Further studies are needed to determine the effect of the Val66Met polymorphism in the BDNF gene on poststroke recovery and to evaluate this relationship with ethnicity, sex, age, stroke type, observe duration, stroke severity, injury location, and therapies.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683211014119 – Supplemental material for The Influence of Val66Met Polymorphism in Brain-Derived Neurotrophic Factor on Stroke Recovery Outcome: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-1-nnr-10.1177_15459683211014119 for The Influence of Val66Met Polymorphism in Brain-Derived Neurotrophic Factor on Stroke Recovery Outcome: A Systematic Review and Meta-analysis by Xuan Liu, Jun-Chao Fang, Xin-Yue Zhi, Qiu-Yu Yan, Hong Zhu and Juan Xie in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-2-nnr-10.1177_15459683211014119 – Supplemental material for The Influence of Val66Met Polymorphism in Brain-Derived Neurotrophic Factor on Stroke Recovery Outcome: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-2-nnr-10.1177_15459683211014119 for The Influence of Val66Met Polymorphism in Brain-Derived Neurotrophic Factor on Stroke Recovery Outcome: A Systematic Review and Meta-analysis by Xuan Liu, Jun-Chao Fang, Xin-Yue Zhi, Qiu-Yu Yan, Hong Zhu and Juan Xie in Neurorehabilitation and Neural Repair
Footnotes
Authors’ Note
All data sets generated during and/or analyzed during the current study are included in this published article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Natural Science Foundation of China (Grant Number: 81602922).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
