Abstract
Background
Motor recovery after stroke in humans and in rodent models is time sensitive. Recovery in patients is a result of biological spontaneous recovery via endogenous repair mechanisms and is likely improved by enhancing the synaptic plasticity required for endogenous repair. Cerebrolysin is a polypeptide preparation known to enhance neuroplasticity and may improve recovery in patients. In mice, we tested the hypothesis that Cerebrolysin can act poststroke to enhance both spontaneous and training-associated motor recovery.
Methods
Mice were trained to perform a skilled prehension task. We then induced a photothrombotic stroke in the caudal forelimb area, after which we retrained animals on the prehension task in the presence or absence of Cerebrolysin after a 2-day or 8-day delay. Mice received daily intraperitoneal Cerebrolysin or saline injections starting poststroke day 1 or poststroke day 7.
Results
Prior studies showed that poststroke recovery of prehension can occur if animals receive rehabilitative training during an early sensitive period but is incomplete if rehabilitative training is delayed. In contrast, we show complete recovery of prehension, despite a delay in rehabilitative training, when mice receive daily Cerebrolysin administration starting on poststroke day 1 or on poststroke day 8. When Cerebrolysin is given on poststroke day 1, recovery occurred even in the absence of training. Stroke volumes were similar across groups.
Conclusions
Poststroke Cerebrolysin administration leads to recovery of motor function independent of rehabilitative training without a protective effect on stroke volume. This is one of the first demonstrations of training-independent motor recovery in rodent stroke models.
Introduction
The majority of rodent models of stroke recovery focus on training-dependent recovery of novel tasks such as the skilled reach-to-grasp (prehension) task.1,2 For example, in prior work, we have shown that there is diminishing responsiveness to motor training over the first week after stroke in mice.2,3 Others have presented similar findings in the rat.4,5 We have previously defined this early, training-responsive time after stroke as the poststroke “sensitive period.” Although human studies also indicate the presence of an early period of recovery,6,7 there is little evidence linking motor training during the sensitive period with human recovery.8,9 That is to say, the majority of true recovery (reduction in impairment) in poststroke patients is largely independent of current rehabilitation efforts.5,10 This should not diminish the importance of rehabilitation 11 or that more intense therapy may be beneficial.12,13 Nevertheless, human recovery is often referred to as “spontaneous.” This is an important distinction: the majority of human recovery is mediated by endogenous repair processes rather than behavioral interventions. To date, we know less about the mechanisms of spontaneous biological recovery (SBR) than we do about recovery associated with behavioral interventions in animals; further, we have no proven interventions that enhance SBR.
Regardless of the mechanism, the hope is that one might improve recovery via some directed intervention. For example, fluoxetine has been shown to improve motor recovery poststroke in both animal models and in humans.2,14 Recent work has shown that Cerebrolysin, a polypeptide mixture derived from porcine brains, led to improved neurological recovery in rats when given early after stroke and when paired with rehabilitative tasks. 15 Furthermore, Cerebrolysin has been shown to enhance electrophysiological and anatomical measures of neuronal plasticity16,17 as well as gene expression changes18,19—mechanisms thought important in motor recovery. 8 Although the early poststroke administration of Cerebrolysin conveyed no reduction in mortality when tested in human patients, Cerebrolysin led to improved poststroke neurological recovery as assessed by National Institutes of Health Stroke Scale, modified Rankin Scale, and Action Research Arm Test,20,21 and these findings have been confirmed in a recent meta-analysis. 21
The idea that poststroke Cerebrolysin administration enhanced human recovery early after stroke raises the possibility that it may influence SBR. Thus, using a mouse model in which a prehension task is affected by a focal motor stroke (photothrombosis), we tested the hypothesis that early Cerebrolysin administration after stroke can enhance training-independent versus training-dependent motor recovery. We also tested whether these effects were a result of a change in stroke volume related to Cerebrolysin treatment. Improved motor recovery in the presence of Cerebrolysin may occur through a number of pathways, but decreased inhibition in the surrounding cortices has been proposed as one mechanism of motor recovery postischemic stroke. To investigate whether Cerebrolysin decreases inhibition, we stained the medial premotor cortex for parvalbumin in Cerebrolysin- and saline-injected animals. Parvalbumin is a marker of inhibitory interneuron identity and activity and has been previously shown to change in the premotor cortex in the setting of stroke.1,22
Methods
Subjects
Adult male C57bl/6 mice 140 to 170 days old were singly housed in custom-made chambers and kept on a 12/12-hour light/dark cycle similar to prior experiments.1-3 We included a total of 60 mice in the study with at least 6 animals per group, based on effect sizes in previous studies.1,4,23 Mice that were unable to attain a baseline prehension success rate of >30% or died perioperatively (and therefore prior to Cerebrolysin/saline administration) were excluded from the study and not considered part of the analysis. Of mice included in the study, 8 mice were excluded from the analysis: 3 mice died prior to completion of the study (2 in the Cerebrolysin arm prior to training and 1 in the saline arm prior to training), and the other 5 mice were excluded because of failure to induce a stroke in the caudal forelimb area (CFA), which was revealed upon pathological analysis (2 in the saline group and 3 in the Cerebrolysin group). Mice were randomized to receive either Cerebrolysin or saline, and investigators were blinded to treatment condition. Specifically, animal identifiers were randomly picked out of a concealed container, and blind was not broken until analysis at the end of experiment was performed. An investigator not involved in the training of the animals injected preselected syringes of randomized drug. All animal handling and use was performed according to the protocols set by the Johns Hopkins University Animal Care and Use Committee.
Skilled Prehension Task
Behavioral tasks1-3 were carried out in the same room and the same modified chambers in which the mice were housed to reduce the stress of new surroundings. Two to 3 days prior to learning the prehension task, mice were placed on a scheduled administration of Bio-serv dustless precision pellet mouse chow per day with water ad libitum. Mice were food restricted to 85% of their starting weight and were trained to reach and grasp (prehension) for 45-mg dustless precision pellets (Bio-Serv). Prehension was scored as successful when the mouse reached its forelimb through the slit, grabbed the pellet, and ate it without knocking it from its resting space, dropping it, or in any other way losing control. The percentage of successful prehension attempts was determined per pellet. A training block consisted of 30 pellets at a distance of 1 cm, with each pellet presented 1 at a time. After familiarization and paw determination, the animals underwent 2 blocks of 30 reaching attempts per training day. The animals had 1 training day off per week (including the day after stroke induction). All animals began poststroke training beginning on day 8 poststroke; one group of animals (Cerebrolysin early assessment) received a poststroke assessment of 20 at 24 hours poststroke to confirm stroke effect on prehension.
Cerebrolysin
Mice were randomized to receive either Cerebrolysin (obtained from Everpharma) 5 mL/kg or normal saline 5 mL/kg15,24 administered via intraperitoneal (IP) injection beginning either 24 hours or 7 days after stroke induction. Investigators were blinded to Cerebrolysin versus saline injection.
Stroke Induction
The location of motor areas was identified based on prior anatomical 25 and functional 26 data. These data also indicate that these areas are geographically consistent within a given strain. We have used these with prior success and followed our previously published protocol. 1 Briefly, under isoflurane anesthesia (4% induction, 1%-2% maintenance in 2L O2), a fiber optic bundle of a cold light source (Zeiss 1500 electronic, Jena, Germany) with a 20-gauge aperture was centered at 2 mm lateral and 0.5 mm anterior from bregma for CFA infarction. The brains were then illuminated through the intact skull for 15 minutes, starting 5 minutes after the IP injection of 150 µL of a 10-mg/mL rose Bengal solution in sterile normal saline.
Tissue Preparation and Histology
On the day when they were killed humanely, the mice were placed under deep anesthesia with 2.5% avertin and transcardially perfused with 4% paraformaldehyde in 0.1 M sodium phosphate, pH 7.4. The brains were dissected out and placed in 4% paraformaldehyde for 24 hours. Brains were coronally sliced at 50 µm on a vibrating microtome. Free-floating sections were washed 3 × 5 minutes in phosphate-buffered saline slices and placed for 4 hours in block solution (10% normal goat serum and 0.04% Triton X-100, in Tris buffered saline) followed by overnight incubation at 4 °C with antiparvalbumin (Sigma; diluted 1:1000 in block solution). Sections were subsequently washed 3 × 5 minutes in Tris buffered saline with 0.04% Triton X-100 and incubated at room temperature for 4 hours with secondary antibodies (Alexa goat-anti-mouse 633 diluted 1:500). Sections were washed 2 × 5 minutes in Tris buffered saline + 0.04% Triton X-100, 1 × 5 minutes Tris buffered saline, and mounted in ProLong Gold reagent (Invitrogen).
Quantification of Stroke Volume
From brain slices prepared as above, the entire anterior-posterior extent of the CFA contralateral to the preferred paw was imaged and reconstructed in 3 dimensions using Zen Blue Imaging software (Zeiss). An investigator blinded to conditions demarcated the stroke pathology, and volumes were calculated using Imaris (Bitplane) imaging software.
Quantification of Parvalbumin-Labeled Cells
From brain slices as prepared above, the ischemic stroke was grossly identified in the CFA. Immediately medial to the ischemic tissue, the medial agranular cortex (AGm) was identified anatomically as previously described.1,25,26 We defined a medial boundary of the AGm from which we extended a 1.2-mm2 area slice from the medial and dorsal pial boundaries. This represents a subarea of the AGm, which prevents us from confounding our counts with cells from neighboring areas. The AGm was imaged at 10× magnification throughout the entire depth of the 50-µm slice at 2-mm intervals using Zeiss Apotome technology to precisely localize cells 27 (total volume imaged 0.06 mm3). Both AGm cortices ipsilateral and contralateral were imaged in the Cerebrolysin- and saline-injected mice, which were killed immediately after training of the last day. The z-slices were reconstructed into a 3D image in ImageJ software “Volume Viewer.” This acquired 3D image was processed with the “Threshold” function, and parvalbumin cells were counted using the automated “3D Objects Counter.” The generated objects’ mask was overlaid on the original image to visually inspect that the automated cell count was appropriate. The cell counts were then averaged for Cerebrolysin-injected or saline-injected animals and analyzed as described below.
Statistics
Behavioral data were analyzed based on prior work with linear mixed-effect models.2,28 Briefly, row means were analyzed with a 2-way ANOVA corrected for multiple comparisons using the Tukey method. Stroke volume data were analyzed using an unpaired, 2-tailed, nonparametric t-test. Immunofluorescent data were analyzed using GraphPad Prism’s 2-way ANOVA with Tukey’s posttest.
Results
Early Cerebrolysin Injections Led to Spontaneous Motor Recovery
We have previously shown that poststroke rehabilitative training on the prehension task beginning after a 7-day delay led to incomplete motor recovery; however, if training began after a 1-day delay, recovery was complete.1-3 The main hypothesis of this study was that Cerebrolysin administration during the sensitive period would improve poststroke motor recovery even when rehabilitative training was delayed by 7 days. To test this hypothesis, we trained mice on the prehension task to an asymptotic level of performance, 1 induced a focal stroke in the CFA, and then administered daily IP injections of either Cerebrolysin or saline beginning 24 hours after the stroke. Mice were free to roam their home cages but received no rehabilitative training for 7 days. To ensure that the animals’ prehension success was affected by the stroke, we performed a limited prehension assessment (20 trials) 24 hours poststroke (Figure 1). Then, on poststroke day 8, mice were again evaluated on the prehension task. A significant difference between the Cerebrolysin and saline groups was observed on day 8 and persisted throughout the remainder of the experiment. Because even limited motor training early poststroke can affect recovery, we injected Cerebrolysin in a separate group of animals that did not receive an early poststroke assessment, which also demonstrated a significant improvement in prehension compared with the saline group. Only the Cerebrolysin groups showed a return to prestroke levels of prehension success, which was apparent even in the absence of training (Figure 1A). Mice who had saline injections during the training delay never returned to their prestroke baseline, consistent with prior studies (Figure 1A). The effect on recovery of Cerebrolysin persisted for the extent of poststroke training, and Cerebrolysin-injected mice continued to outperform saline-injected mice (Figure 1A).
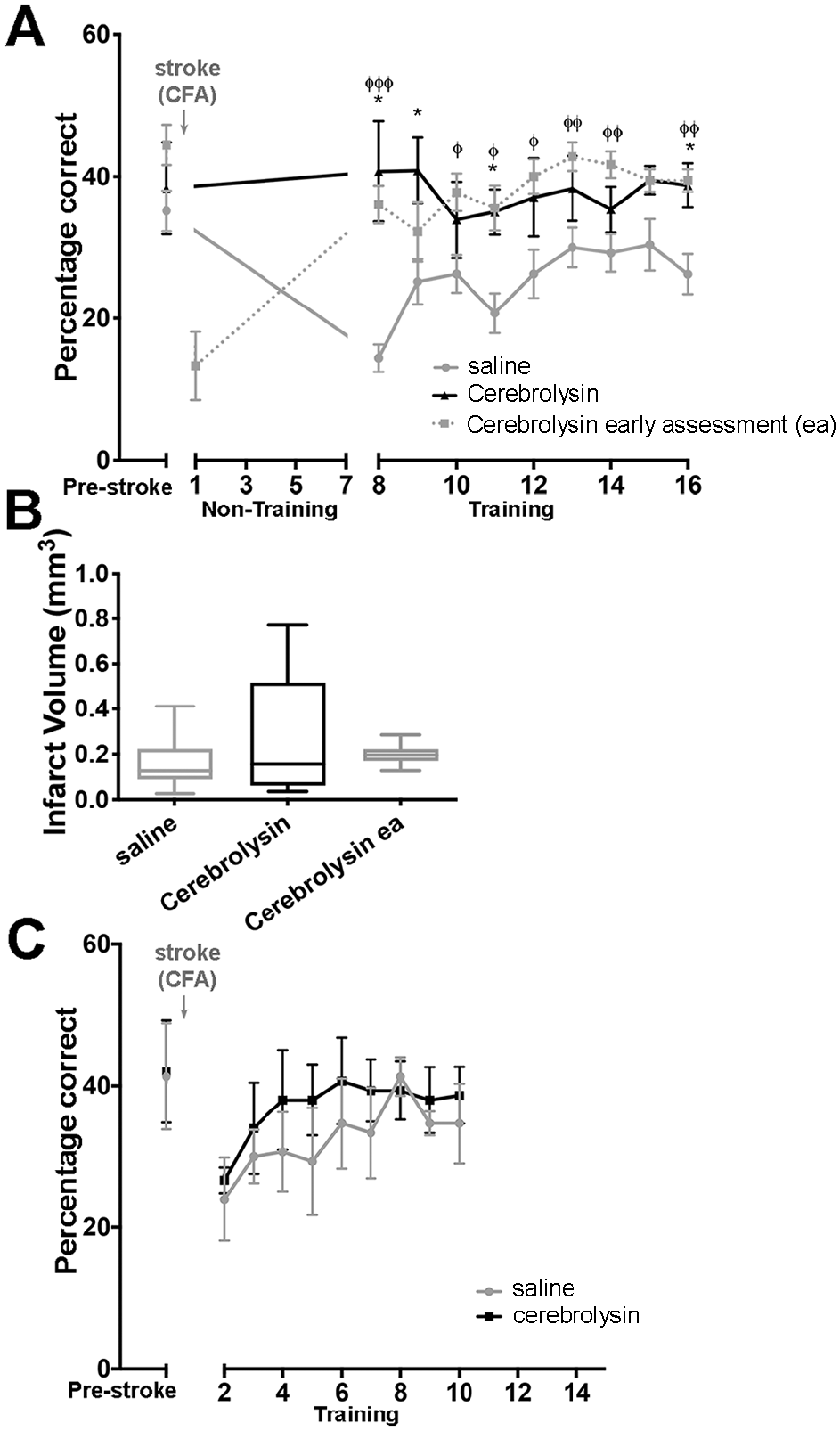
(A) Cerebrolysin administration beginning 1 day after stroke led to recovery of prehension performance independent of rehabilitation. Prehension success (mean ± SEM) for mice undergoing CFA stroke and training beginning after a 7-day poststroke delay. Mice received daily injections of saline (gray n = 9), Cerebrolysin with 1 limited assessment on poststroke day 1 (dotted, n = 7), or Cerebrolysin with training beginning on poststroke day 8 (black, n = 10). Overall, all ANOVA P values <.05. * P < .05; post hoc comparison between Cerebrolysin versus saline. φ P < .05; post hoc comparison between Cerebrolysin with 1 limited assessment on poststroke day 1 versus saline. φφ P < .01. φφφ P < .001. (B) There was no significant difference between CFA infarct volumes in mice receiving saline (n = 9) versus Cerebrolysin (n = 9; training starting on poststroke day 8) versus Cerebrolysin early assessment (n = 7; with 1 limited assessment early poststroke). (C) Cerebrolysin administration beginning 1 day poststroke did not produce a sustained effect on recovery when training began on day 2 poststroke. Prehension success (mean ± SEM) for mice undergoing CFA stroke and training beginning after a 2-day poststroke delay. Mice received daily injections of saline (gray, n = 6) or Cerebrolysin (black, n = 6).
It is possible that Cerebrolysin enhanced poststroke prehension success by reducing infarct severity via a neuroprotective effect. To investigate this, we killed the animals described in Figure 1A after their last training session for volumetric analysis of the CFA strokes. Regardless of condition, there were no significant stroke volume differences between the animals receiving saline compared with either group receiving Cerebrolysin (Figure 1B).
We assessed prehension performance after a 2-day poststroke delay in animals receiving either Cerebrolysin or saline. We trained mice to an asymptotic level of performance on the prehension task, induced a focal stroke in the CFA, and then administered daily IP injections of either Cerebrolysin or saline beginning 24 hours after the stroke. Mice were then trained 24 hours after their first dose of Cerebrolysin (2 days post-stroke). Mice who received either Cerebrolysin or saline showed a similar decline in prehension success early poststroke (Figure 1C). Although there was an initial nonsignificant accelerated recovery in the animals receiving Cerebrolysin, both the saline and Cerebrolysin groups recovered to the same baseline performance.
Late Cerebrolysin Injections Improved Training-Dependent Recovery
In our previous mouse experiments, if retraining or pharmacological treatment with fluoxetine was delayed, poststroke prehension success did not return to prestroke baseline.2,3 To test if delayed Cerebrolysin administration could recover poststroke prehension in animals without post-stroke training for 7 days, we trained mice to an asymptotic level of performance on the prehension task, induced a focal stroke in the CFA, and then administered daily IP injections of either Cerebrolysin or saline beginning 7 days after the stroke. On day 8 poststroke (24 hours after the Cerebrolysin injection), prehension assessment revealed that the Cerebrolysin and saline groups were similar in their prehension performance (Figure 2A). However, over the ensuing days, the animals receiving Cerebrolysin displayed improved motor recovery even though both Cerebrolysin administration and rehabilitative training were delayed by 7 days and 8 days, respectively (Figure 2A). Eventually, mice that received Cerebrolysin starting 7 days poststroke returned to prestroke levels of prehension, in contrast to saline-injected mice (Figure 2A).
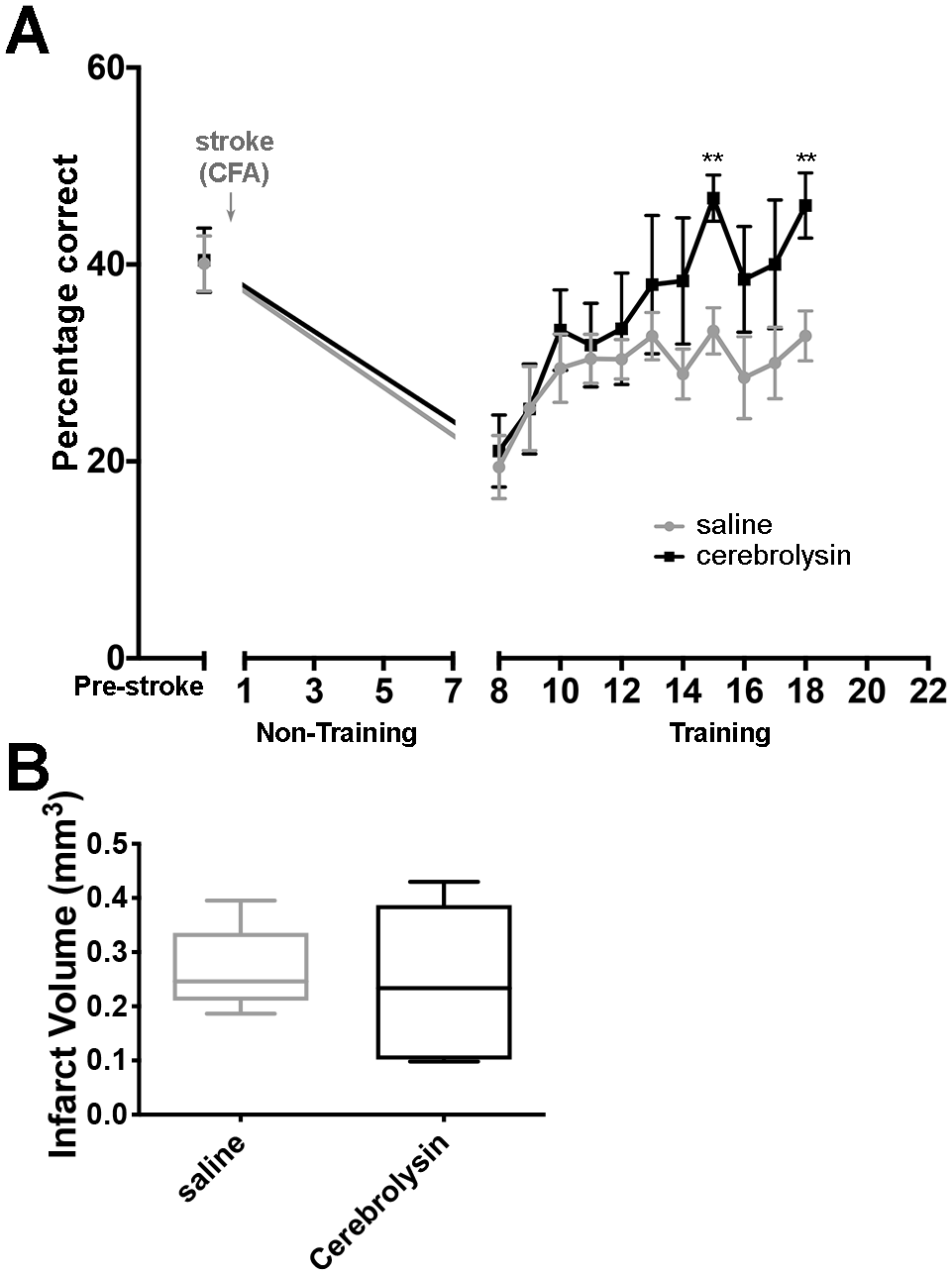
Cerebrolysin administration beginning 7 days after stroke led to recovery of prehension performance. (A) Prehension success (mean ± SEM) for mice undergoing CFA stroke and training beginning after a 7-day poststroke delay. Mice received daily injections of saline (gray, n = 7) or Cerebrolysin (black, n = 7). Overall, all ANOVA P values <.05. ** P < .02. (B) There was no significant difference between CFA infarct volumes in mice receiving saline (n = 7) versus Cerebrolysin (n = 7).
To assess volume of infarct pathology, mice were killed after their last training session and their brains harvested. Regardless of condition, there were no significant stroke volume differences between Cerebrolysin and saline groups (Figure 2B).
Recovery Was Not Associated With Decreased Inhibitory Markers in a Medial Premotor Area
The balance between excitatory and inhibitory cortical inputs has been proposed as a mechanism of plasticity after neuronal injury.1,2,8 To test if Cerebrolysin changes the inhibitory balance poststroke, we assayed parvalbumin expression in the medial premotor cortex as a marker of inhibitory interneuron identity and activity. Animals receiving Cerebrolysin 24 hours after stroke followed by training after a 7-day poststroke delay (animals presented in Figure 1) as well as animals receiving Cerebrolysin 7 days after stroke followed by training after a 7-day poststroke delay (animals presented in Figure 2) were killed the day of their last training session and prepared for immunoreactivity. As shown in Figure 3, there was a decrease in parvalbumin immunoreactivity in the premotor cortex of mice treated early poststroke with Cerebrolysin compared to saline; however, there was no statistical difference in inhibitory interneuron immunoreactivity in mice receiving Cerebrolysin compared with saline injections under any of the tested conditions (Figure 3).
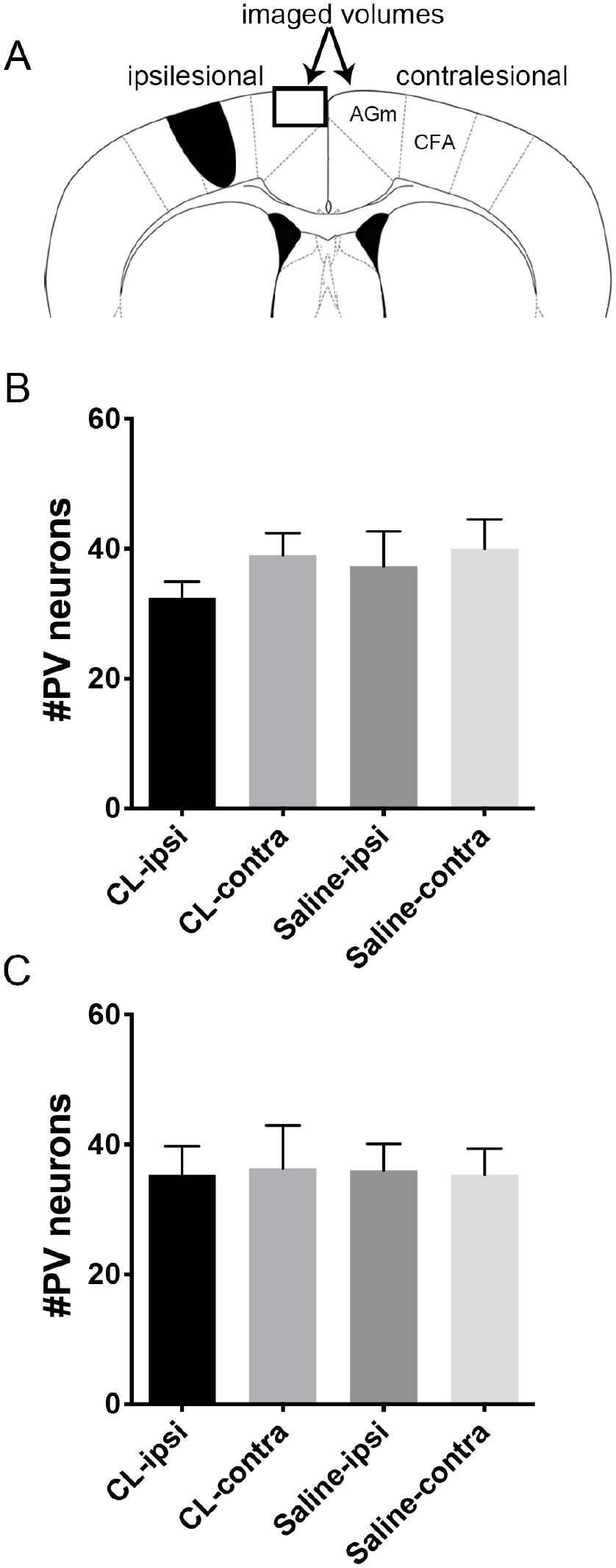
Cerebrolysin administration after stroke is not associated with decreased parvalbumin expression in the medial premotor cortex (AGm). (A) Coronal schematics of mouse brain showing caudal forelimb area (CFA) stroke and medial premotor cortex (AGm; box). (B) Number of parvalbumin positive neurons per 107 µm3 in the AGm of animals receiving Cerebrolysin 24 hours after stroke followed by training after a 7-day delay; n = 6. (C) Number of parvalbumin-positive neurons per 107 µm3 in the AGm of animals receiving Cerebrolysin 7 days after stroke followed by training after a 7-day delay; n = 6.
Discussion
Using a mouse photothrombotic stroke model, we repeated prior data showing that CFA strokes produce contralateral motor deficits. We then showed that early poststroke Cerebrolysin administration was associated with motor recovery despite a lack of prehension retraining, which is a novel result in mouse models. The Cerebrolysin-injected mice had motor recovery that persisted throughout the time course of the experiment and had consistently better performance than the saline-injected mice. Importantly, the enhanced motor recovery was independent of stroke volume modulation.
Spontaneous Recovery in the Poststroke Time-Sensitive Period
Data reveal that humans show substantial spontaneous motor recovery and that this recovery follows a proportional rule in the majority of patients, with the degree of recovery correlated with initial severity of deficit on the Fugl-Meyer Scale.29,30 Consistent results across different patient groups suggests that proportional recovery occurs independent of current rehabilitation efforts and is, instead, dependent on innate biological mechanisms—so-called spontaneous recovery. In contrast, although there may be modest SBR, the majority of poststroke recovery in rodent models is training dependent. For example, Jeffers et al31,32 have demonstrated proportional recovery in the rat similar to that seen in humans (62%-70%); however, in contrast to humans, the intensity of rehabilitation, especially in severely affected animals, was an important predictor of functional recovery. 31 The reasons for this difference between rats and humans is not clear but may be related to the following: (1) inherent differences between rat and human neuroanatomy; (2) rat rehabilitation is better suited to capitalize on SBR with enriched environments, increased time on task, and rehabilitation specifically suited to the outcome measure; and/or (3) a pairing of lesion location and rehabilitation that is better suited to influence recovery in rodents compared with humans. 33
Here, we report recovery of function independent of poststroke training in mice that receive Cerebrolysin (see Figure 1A), similar to the spontaneous recovery seen in stroke patients. Such recovery cannot be because mice receiving Cerebrolysin injections had reduced poststroke deficits immediately after the stroke because our data show that mice tested immediately after stroke (Figures 1A and 1C) had reduced prehension success, similar to prior reports. Although there are a small number of prior reports that have shown training-independent recovery in rodents associated with either pharmacological or genetic manipulations,34-36 our data represent the first poststroke, training-independent recovery using the complicated prehension task.
Importantly, the majority of motor recovery occurs in the first 3 months in humans and in the first month in rodents. We administered Cerebrolysin 24 hours poststroke, which is during a time when we, and others, have previously shown that mice are sensitive to poststroke training. Although we additionally show that Cerebrolysin administration beginning on day 7 is also associated with a return to baseline, the multiple comparisons between the Cerebrolysin and saline groups suggest that Cerebrolysin’s efficacy in this longer delay period may be reduced. Reduced efficacy at later time points may be secondary to temporal distance from the poststroke sensitive period and/or repair of ischemia-induced damage of the blood-brain barrier and, thus, reduced access of Cerebrolysin to the central nervous system. Further experiments are necessary to determine if Cerebrolysin can act outside of the known sensitive period to enhance recovery without training.
Mechanism of Cerebrolysin on Motor Recovery
Although previous studies have suggested that Cerebrolysin may play a neuroprotective role in stroke and traumatic brain injury,37-39 our data reveal no difference in infarct size between Cerebrolysin- or saline-injected mice. In previous studies, reduced lesion volumes were observed under Cerebrolysin treatment, however at higher doses than used in our study. 39 Although nonsignificant, animals that received Cerebrolysin had a trend toward larger stroke volume sizes. This is similar to previous reports in which rehabilitation-induced increases in stroke volume were associated with improved behavioral outcomes,2,40,41 reflecting a possible pruning effect, whereby energy-compromised neurons are eliminated early on as a result of use-dependent activation associated with rehabilitation. 9
Our data suggest that Cerebrolysin does not alter the excitatory-inhibitory balance, at least as measured by changes in parvalbumin immunoreactivity. As such, Cerebrolysin may encourage motor recovery by engaging mechanisms distinct from those that alter the excitatory-inhibitory balance. For example, the plethora of peptide fragments found in Cerebrolysin may act via the sonic-hedgehog signaling pathway, 18 neurotrophin-based pathways,42,43 and/or neurogenesis, 44 all of which have been shown to be important in stroke recovery.45-47 Although the exact mechanisms of SBR are unknown, the formation of new circuits within the brain is a fundamental element of poststroke recovery, and likely SBR, described across several species, including rodents and primates.47-50 As such, it is not difficult to imagine that Cerebrolysin may act on a variety of targets to stimulate new circuit formation. Of particular importance is proposed activity on the cortical spinal tract (see below). It is also possible that Cerebrolysin acts on mechanisms independent of SBR to influence recovery, which may account for why we still see recovery associated with Cerebrolysin well into the third week poststroke (Figure 2). Further studies will be necessary to determine (1) the exact mechanisms of how Cerebrolysin supports spontaneous recovery and (2) if these are distinct from Cerebrolysin’s mechanisms of action that enhanced delayed motor recovery.
Relevance to Human Trials
Cerebrolysin is an attractive pharmacological candidate for use in recovery trials because of the presence of multiple neuropeptides, likely leading to a plethoric effect on the nervous system. 37 In human patients, the CARS-1 trial showed a significant benefit in motor recovery when administered in the acute poststroke period. 51 These data were not replicated in the CARS-2 trial, which overall had a lower stroke severity index and, therefore, less sensitivity to detect improvements in motor recovery 90 days poststroke. 20 A subsequent meta-analysis by Bornstein et al 21 supports previous evidence that Cerebrolysin has a beneficial effect on early global neurological deficits, including motor deficits, in patients with acute ischemic stroke and that such recovery is independent of an effect on mortality. The meta-analysis further suggested that patients with severe strokes benefited from Cerebrolysin more than patients with mild deficits. The mild stroke cases in the analyzed trials might introduce a substantial ceiling effect and, thus, prevent detection of group differences. 52 An alternative explanation is that the severely affected stroke patients who received Cerebrolysin, on average, showed full proportional motor recovery that they otherwise would not have without Cerebrolysin. That is to say, patients with severe stroke-induced motor deficits are less likely to undergo full proportional motor recovery,29,30,53 especially if the corticospinal tract is severely injured. 54 In light of our data showing that Cerebrolysin is able to stimulate spontaneous motor recovery, it is possible that Cerebrolysin may have nudged severe patients into full proportional recovery. This theory is supported by recent data showing that Cerebrolysin may help repair damaged corticospinal connectivity in humans, 55 the residual integrity of which supports spontaneous recovery. 54
However, an interaction between Cerebrolysin and SBR in human patients is still uncertain. First, no Cerebrolysin study formally assessed whether SBR was greater for the treatment group than the control group. Second, no Cerebrolysin study used biomarkers of ipsilesional corticomotor integrity for patient selection or stratification. Thus, it is possible that more patients in the Cerebrolysin arms of the meta-analyzed trials randomly had more residual ipsilesional corticospinal tract integrity. However, we find this to be highly unlikely because similar findings were seen across multiple different well-randomized trials. A larger study with stratification based on the PREP algorithm 56 will be needed to evaluate whether Cerebrolysin effectively promotes motor recovery in human patients.
Conclusion
We show here a training-independent spontaneous motor recovery effect for Cerebrolysin in mice. We also show that delayed Cerebrolysin administration improves recovery when given during retraining. These data support further evaluation of the mechanisms of action of Cerebrolysin in stroke models and support further clinical trials in patients.
Footnotes
Acknowledgements
The authors would like to acknowledge Dr John Krakauer for his time, his ideas, his careful reading, and most of all, his support.
Authors’ Note
Scott R DeBoer now affiliated to Medstar Franklin Square Medical Center, Georgetown School of Medicine. Rob Hubbard now affiliated to University of Pittsburgh, Swanson School of Engineering.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by grant from Ever Pharma to SZ.
