Abstract
Background
The rhythm of music can entrain neurons in motor cortex by way of direct connections between auditory and motor brain regions.
Objective
We sought to automate an individualized and progressive music-based, walking rehabilitation program using real-time sensor data in combination with decision algorithms.
Methods
A music-based digital therapeutic was developed to maintain high sound quality while modulating, in real-time, the tempo (ie, beats per minute, or bpm) of music based on a user’s ability to entrain to the tempo and progress to faster walking cadences in-sync with the progression of the tempo. Eleven individuals with chronic hemiparesis completed one automated 30-minute training visit. Seven returned for 2 additional visits. Safety, feasibility, and rehabilitative potential (ie, changes in walking speed relative to clinically meaningful change scores) were evaluated.
Results
A single, fully automated training visit resulted in increased usual (∆ 0.085 ± 0.027 m/s, P = .011) and fast (∆ 0.093 ± 0.032 m/s, P = .016) walking speeds. The 7 participants who completed additional training visits increased their usual walking speed by 0.12 ± 0.03 m/s after only 3 days of training. Changes in walking speed were highly related to changes in walking cadence (R2 > 0.70). No trips or falls were noted during training, all users reported that the device helped them walk faster, and 70% indicated that they would use it most or all of the time at home.
Conclusions
In this proof-of-concept study, we show that a sensor-automated, progressive, and individualized rhythmic locomotor training program can be implemented safely and effectively to train walking speed after stroke. Music-based digital therapeutics have the potential to facilitate salient, community-based rehabilitation.
Stroke is a leading cause of long-term disability worldwide. 1 The most common site for a stroke to occur is the middle cerebral artery, which supplies both the primary motor cortex and the auditory cortex. Motor and auditory brain regions are not only connected by a vascular supply, but are also linked in neural networks that include both cortical and subcortical structures.2-8 Stroke-induced impairments in neuromotor control contribute to the characteristically slow and metabolically expensive gait of people with poststroke hemiparesis. 9 Indeed, whereas healthy individuals naturally self-select a walking cadence that is their most stable and economical, 10 people with poststroke hemiparesis walk with generally slower11,12 and irregular 13 cadences that result in an overall slower, 12 more unstable, 13 and metabolically expensive gait. 14 Neurophysiological studies show that rhythmic auditory stimulation can entrain and prime neurons in motor cortex by way of direct connections between auditory and motor brain regions.15-17 A substantial evidence base that spans more than 2 decades has emerged in support of combining gait training with rhythmic auditory stimulation, revealing a potent intervention that can, among other benefits, increase walking symmetry, improve neuromotor control, promote stability, 18 and increase walking speed12,18,19 (see review articles5,20-23).
Rhythmic auditory stimulation can be provided using a variety of approaches. The use of music as the vehicle for delivering rhythmic stimuli benefits from the salience of music serving to make exercise more engaging and sustainable5,24—a critical consideration for users who are characteristically inactive. 25 Moreover, music may facilitate global activation of neural networks, 26 including those involved in motor control,27-29 that may otherwise not be accessed during gait training without music. 30 Furthermore, encoding a musical beat may better engage similar neural structures as the locomotor actions themselves,16,17 and may lead to improved goal-action coupling (eg, by linking the goal of increasing walking speed to the action of stepping faster),31,32 ultimately improving outcomes.
Motivated by the strong evidence for rhythm-based gait training after stroke,5,19,21,33 we sought to integrate music, adaptive rhythm modulation, and wearable movement sensors in a digital therapeutic designed to automate a rhythm-based rehabilitation protocol centered on training principles of individualization and progression. Digital rehabilitation technologies have the potential to transform long-term outcomes after stroke by providing patients with independent access to targeted interventions, either between physical therapy sessions or after being discharged from rehabilitation. The objective of this foundational study was thus to determine the feasibility and rehabilitative potential of an automated, individualized, and progressive music-based rhythmic locomotor training program. We hypothesized that (a) individuals poststroke would effectively synchronize their gait to the music-based auditory cues, (b) training with the platform would facilitate increased walking speed, (c) multiple visits would produce cumulative gains in walking speed, and (d) patients would indicate an interest in using the platform independently in the community.
Methods
Participants Overview
Eleven individuals with chronic poststroke hemiparesis participated in this study. Study participants were recruited through a research participant registry generated from the clinical programs at Boston University, referrals from local clinics and hospitals, and flyers distributed in and around Boston. Inclusion criteria included >6 months poststroke, age 18 to 80 years, the ability to walk independently with or without an assistive device for at least 2 minutes, and the ability to provide informed consent. Exclusion criteria included pain that impairs walking ability, confusion or an inability to communicate (score >1 on NIHSS [National Institutes of Health Stroke Scale] question 1b and >0 on question 1c), neglect, hemianopia, ataxic hemiparesis, unexplained dizziness in the past 6 months, resting heart rate <50 or >100 bpm, and resting blood pressure <90/60 or >190/110 mm Hg. All study procedures were approved by the Boston University Institutional Review Board and written informed consent was secured for all participants. A physical therapist assessed all inclusion and exclusion criteria to determine safe participation. Trained investigators performed all additional outcome measurements.
Training Overview
All 11 study participants completed 1 session of music-based, rhythmic locomotor training. A subset of 7 study participants were available for 2 additional training visits and a fourth visit consisting of a walking evaluation. Each training visit consisted of 30 minutes of continuous walking practice guided by automated decision algorithms (see section Digital Therapeutic Platform and Algorithms). Study participants were allowed to rest at any time, but to more closely replicate typical intervention durations, participants were instructed that resting would take away from their 30 minutes of training time. Walking assessments were conducted immediately before and after the 30 minutes of training and at the start of each subsequent visit (see Supplementary Material 1. Standardized assessment and training instructions). If indicated for safety, a fall-prevention harness was worn during walking activities. Heart rate and blood pressure were monitored during each visit.
Digital Therapeutic Platform and Algorithms
A digital therapeutic platform was developed to leverage real-time movement data measured using inertial sensors to deliver personalized rhythmic gait training by way of the automatic modulation of the tempo (ie, beats per minute, or bpm) of any song based on (a) a user’s baseline walking cadence and (b) ability to synchronize their cadence to progressively faster musical tempos. The digital therapeutic platform (Figure 1) consists of an inertial sensor (3D gyroscope, 2000 deg/sec) that monitors a user’s real-time walking cadence, a Bluetooth low-energy radio chip with near field communication, and an ARM Cortex M4 floating-point-on-board processor. A smartphone houses the music playlist and delivers the acoustic signals through bone-conducting headphones (AfterShockz). Automated decision processes adapt the auditory stimuli to each user’s rhythm synchronization performance across the 30 minutes of training, with the objective of facilitating an individualized and progressive therapy. Moreover, novel audio algorithms ensure that modulation of the musical tempo does not distort sound quality.
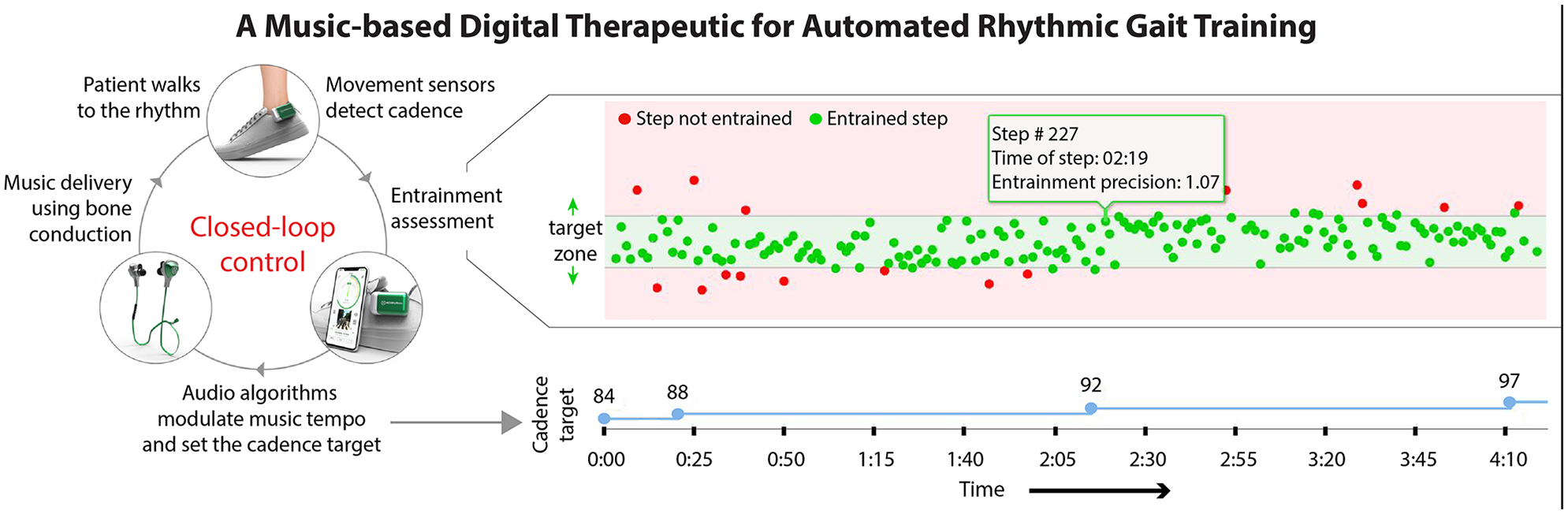
Depiction of the sensor-driven closed-loop control of the rhythm of musical stimuli. Personalized and progressive rhythmic gait training is provided by a music-based digital therapeutic platform consisting of wearable sensors and a smartphone application preloaded with a user’s playlist. Decision algorithms automate training individualization and progression by modulating the music rhythm in response to the user’s abilities. On the right is a 4-minute and 10-second sample of training session data showing entrained (green) and not entrained (red) steps and how the decision algorithm increases the target walking cadence (blue) after a 2-minute period of entrainment, defined as >60% of steps within the target zone.
Music Selection and Progression and Assist Decisions
Each training period begins with an assessment of the user’s baseline walking cadence during a brief walking period without auditory cues. Music from familiar genres (ie, classic rock, Motown, or pop), with 4/4 meter, and that was manually screened for salient beat strength is then introduced. The music is time-shifted to begin playing at the user’s baseline walking cadence and is modulated during the training period according to entrainment outcomes. More specifically, differences between the user’s walking cadence and the musical tempo are assessed in real-time on a stride to stride basis to determine each stride’s entrainment precision relative to a target entrainment zone. Entrainment precision is calculated as the ratio of the time interval between successive musical beats divided by the time interval between successive heel strikes by the same foot, with a ratio of 1.0 indicating perfect alignment of stride to beat. An entrainment zone of 0.9 to 1.1 (ie, ±10% of the musical beat) was used in this study to allow for the natural step-to-step variability inherent to periodic changes in the musical tempo and variable real-world walking conditions (eg, slowing down during a turn). Study participants are considered to have entrained when at least 60% of their steps during a 2-minute period are in the entrainment zone.
When study participants are considered to be entrained (ie, at least 60% of steps during a 2-minute period are within the entrainment zone), the musical tempo is increased by 5% of the baseline tempo. This process of assessment and progression runs continuously throughout the training session, allowing for up to 14 tempo changes in a 30-minute training period if the user entrains 100% of the time. If less than 60% of the user’s steps during a 2-minute period are within the entrainment zone, a metronome beat is overlaid to accentuate the target tempo. If the number of entrained steps consistently fall under the 60% threshold, the volume of the music is gradually decreased and the volume of the metronome beat is gradually increased. If less than 60% of the user’s steps are determined to be within the entrainment zone for an additional 1.5 minutes, the tempo of the music is decreased by 5%.
Outcomes
Safety and Feasibility
Safety was assessed as the incidence of adverse events, including trips and falls, during training. To assess feasibility, we assessed users’ rhythm synchronization ability during a no-music rhythm synchronization test and during training, and used customized questionnaires to survey users’ perceptions of the digital therapeutic. More specifically, to determine each study participant’s baseline rhythm synchronization ability, before each training visit we assessed their entrainment precision while walking to a nonmusical auditory cue. The test consisted of three stages: (a) 30 seconds of walking at a comfortable speed during which baseline cadence was determined, (b) 30 seconds of walking to a metronome beat synchronized to the baseline cadence, and (c) 30 seconds of walking to a metronome beat that was 5% faster than the baseline cadence. Study participants’ baseline rhythm synchronization ability was defined as the proportion of total steps that were within ±10% of the 5% faster metronome beat prescribed during the third and final 30 second stage of the test. Study participants’ entrainment precision was similarly assessed during the training sessions. More specifically, each study participant’s ability to entrain to the music-based rhythmic cues provided during the 30-min training period was assessed as the percentage of total steps that were within ±10% of the target tempos set across the session. Finally, questionnaires were administered after each training session to record qualitative factors such as study participants’ perception of their ability to entrain to the changing musical tempo, how challenging the training was, and their likelihood to use the device at home (see Supplementary Material 2. User Questionnaires).
Rehabilitative Potential
To assess the rehabilitative potential of the automated therapy sessions, walking speed was our primary clinical outcome of interest and was measured by the 10-m walk test at both comfortable and fast walking speeds (m/s, mean of 3 trials). Changes in walking speed were compared to clinically meaningful change scores. Walking cadence (steps/min) was concurrently assessed during each walking speed measurement using an optical motion capture system (Qualisys AB).
Analyses
Data are presented at the level of individual study participants, as well as at the group level using the average ± standard error (SE). All analyses were conducted using commercial statistical software (SPSS version 24, IBM Corp, Armonk, New York) with alpha set to 0.05. To determine how well study participants were able to synchronize their gait to the rhythmic auditory stimuli, we report the percentage of entrained steps across study visits. Differences across study visits were not observed; thus, all entrainment data were averaged across days and reported as average ± SE. To test our hypothesis that training with the digitial therapeutic would facilitate increased walking speed, paired t tests were used to compare pre- to posttraining walking speeds (n = 11). To test our hypothesis that multiple training visits would produce therapeutic gains in walking speed, paired t tests compared the walking speeds and cadences from visit 4 to visit 1 (n = 7). Moreover, individual subject changes in walking speed were compared to previously reported meaningful change scores, which have included a small meaningful change of 0.05 m/s, a large meaningful change of 0.10 m/s, 34 and up to 0.16 m/s. 35 With only 3 training sessions provided, the objective of this analysis was not to test the efficacy of this intervention, rather provide insight into its rehabilitative potential. In addition, to test if the rhythmic gait training had the targeted effect of facilitating faster walking speeds by way of facilitating faster walking cadences, the relationship between changes in walking cadence and changes in walking speed was assessed using linear regression. As an exploratory post hoc analysis, we also evaluated the relationship between baseline rhythm synchronization ability and changes in walking speed, hypothesizing that users with better rhythm synchronization ability would have larger training-induced changes in walking speed. Finally, we characterized the time-course of walking speed changes across sessions by reporting users’ walking speeds for each visit relative to their visit 1 baseline speed.
Results
Safety and Feasibility
Safety
Eleven individuals in the chronic phase after stroke completed a single training visit consisting of 30 minutes of walking practice guided by the music-based digital therapeutic. A subset of 7 individuals completed an additional 2 training visits (Table 1), for a total of 750 minutes of device usage during the study. Two of the 11 study participants were assessed to require contact guard assistance by a physical therapist during training activities. The remaining 9 study participants completed all training activities without the direct supervision of a physical therapist. No serious adverse events, including incidences of trips or falls, were observed during training activities across all study participants. One participant reported the onset of knee pain while training that resolved after the number of turns required during training was reduced.
Study Participants’ Baseline Characteristics.
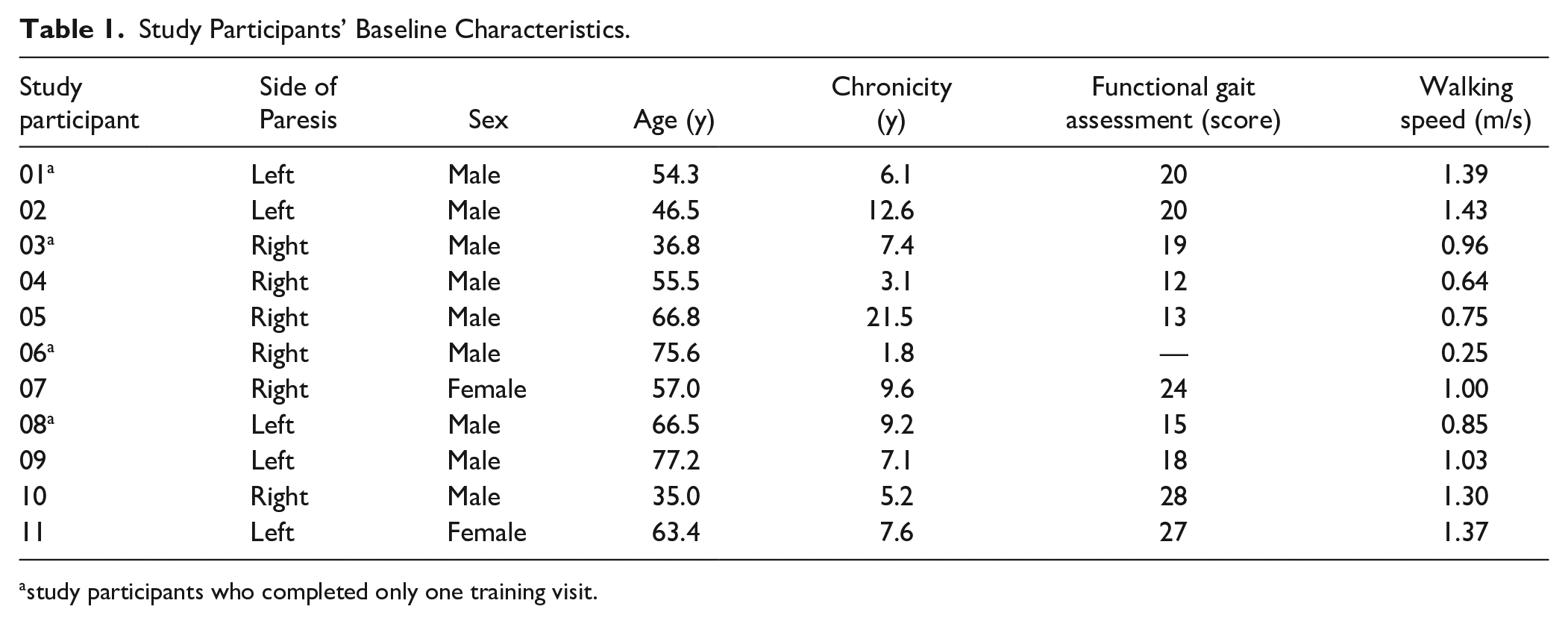
study participants who completed only one training visit.
Rhythm Synchronization Feasibility
Entrainment to both nonmusical and musical auditory stimuli varied across study participants (Table 2). On average, performance on the walking rhythm synchronization test, which measured entrainment to a no-music metronome beat that was 5% faster than baseline walking cadence, consisted of 73.7% ± 3.1% of total steps being within ±10% of the target tempo. In contrast, for the music-based rhythmic stimuli delivered during the 30-minute training visits, 61.9% ± 2.9% of total steps were within ±10% of the target tempo, highlighting the progressive and challenging nature of the device’s decision algorithms. Users’ baseline rhythm synchronization ability was not related to the gait speed changes that occurred following the training visits (Ps > .05).
Entrainment to Nonmusical and Musical Rhythms. a
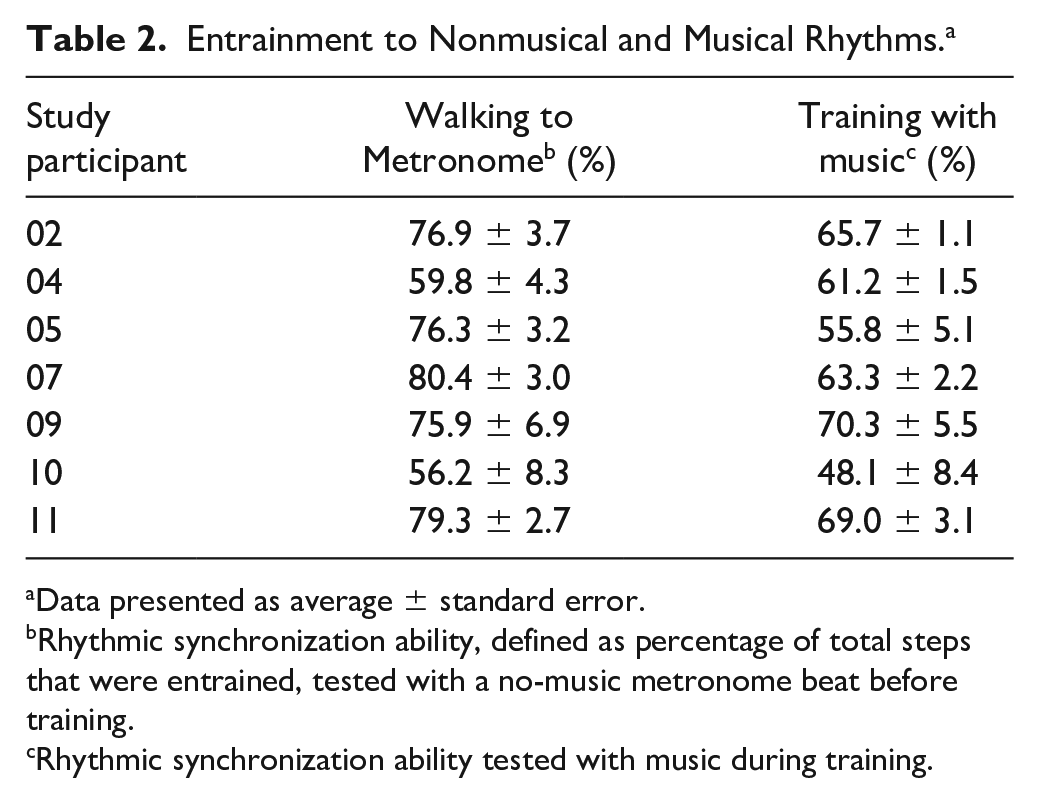
Data presented as average ± standard error.
Rhythmic synchronization ability, defined as percentage of total steps that were entrained, tested with a no-music metronome beat before training.
Rhythmic synchronization ability tested with music during training.
User Questionnaires
After the first training session, 4 out of 7 study participants reported that it was easy or somewhat easy to “get on the beat.” One of the remaining 3 participants reported that it was neither hard nor easy, and the remaining 2 stated that it was hard or very hard to get on the beat. Subsequently, only 3 out of 7 participants said that it was easy or somewhat easy to “stay on the beat,” while the remaining participants stated it was hard (n = 3) or very hard (n = 1). After the final training session, 4 out of 7 participants said it was easy or somewhat easy to “get on the beat,” whereas all participants indicated that once they got on the beat, it was easy to “stay on the beat.”
After the first training session, 5 out of 7 study participants reported that the walking practice facilitated by the digital therapeutic was at a “challenging pace” more than half of the training session (ie, 50%-100% of the time), with an additional study participant reporting the same after the final training session. Concurrently, after the first training session, only 2 out of the 7 study participants said that they were able to walk faster than they normally do with the musical cues. In contrast, after the final training session, all participants said that they were able to walk faster with the musical cues than they normally do.
And finally, following both the first and final training sessions, 5 out of 7 study participants noted that they would use the platform to practice walking at home “most or all of the time.” The remaining 2 respondents replied similarly on each occasion with one indicating that they would use it sometimes and the other indicating that they would never use it. Taken together, these responses reflect the overall positive experience over repeated trials and a general willingness to incorporate the platform into home-based walking training.
Rehabilitative Potential
Within-Session Speed Changes
An average 0.085 ± 0.027 m/s (P = 0.011) posttraining increase in usual walking speed was observed immediately after visit 1, with 8 of 11 users (72%) surpassing the 0.05 m/s small meaningful change score, 4 of 11 users (36%) increasing by more than the 0.10 m/s large meaningful change score, 34 and 2 of 11 users (18%) achieving an increase larger than 0.16 m/s 35 (Figure 2A). Participants’ fast walking speed similarly increased by an average 0.093 ± 0.032 m/s (P = 0.016), with 8 of 11 users (72%) increasing their speed by more than the 0.05 m/s threshold, 5 of 11 surpassing 0.10 m/s (45%), and 2 of 11 surpassing 0.16 m/s (18%) (Figure 3A).
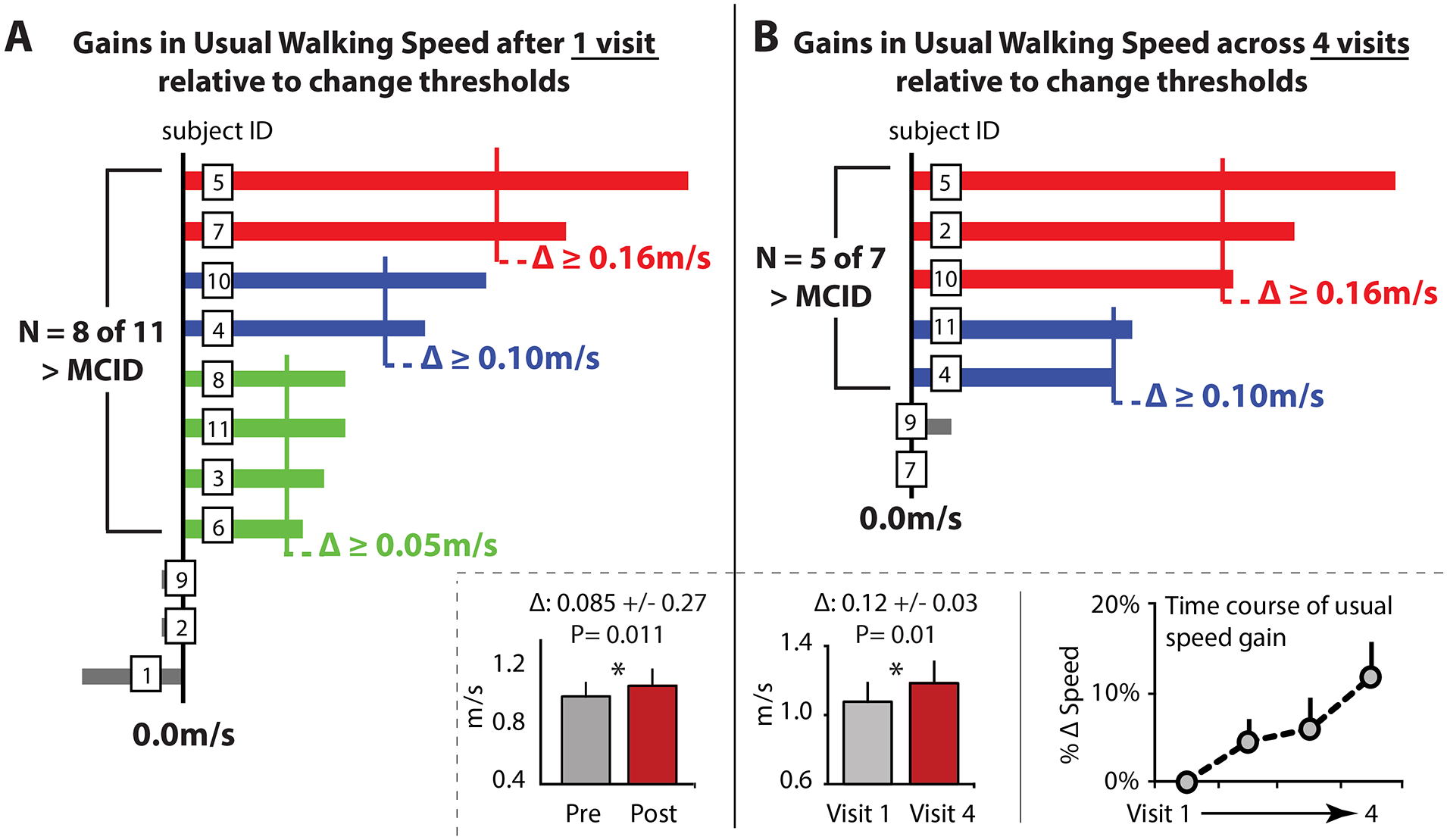
(A) Within-session changes in usual walking speed, as measured using the 10-m walk test, for 11 individuals poststroke who completed 30 minutes of walking practice guided entirely by a music-based digital therapeutic. Individual and group data are shown, with individual subject changes compared to meaningful change scores.34,35 (B) Across-session changes in usual walking speed shown for a subset of 7 individuals who completed an additional 2 training visits (3 in total) and completed a reevaluation on visit 4. Error bars are standard error.
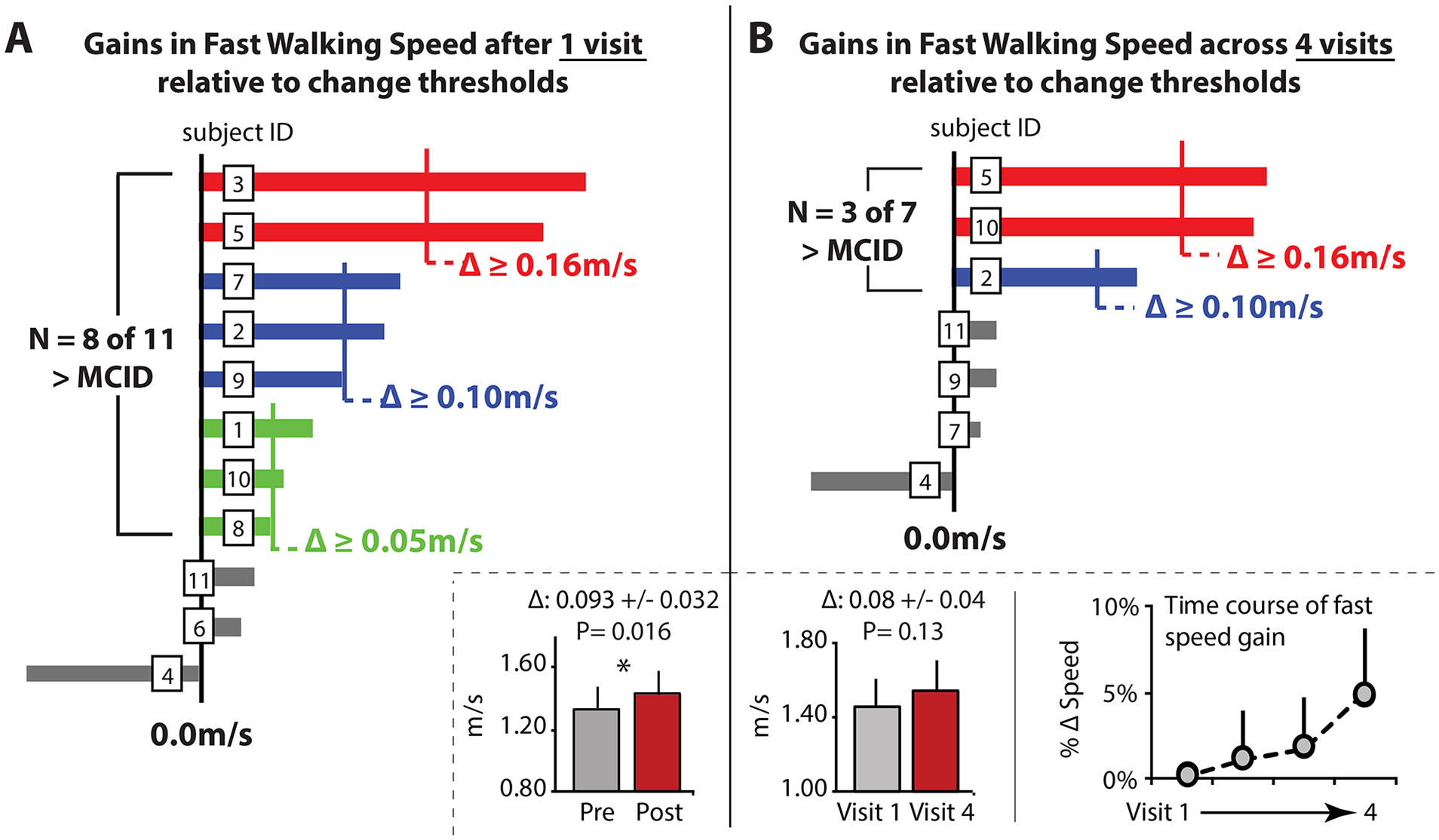
(A) Within-session changes in fast walking speed, as measured using the 10-m walk test, for 11 individuals poststroke who completed 30 minutes of walking practice guided entirely by a music-based digital therapeutic. Individual and group data are shown, with individual subject changes compared to meaningful change scores.34,35 (B) Across-session changes in fast walking speed shown for a subset of seven individuals who completed an additional 2 training visits (3 in total) and completed a reevaluation on visit 4. Error bars are standard error.
Across-Session Speed Changes
A subset of 7 individuals completed an additional 2 training visits. After only 3 training visits, these 7 individuals walked an average 0.12 ± 0.03 m/s faster (P = 0.012), with 5 of the 7 users (71%) increasing their usual walking speed by more than the large meaningful change score of 0.10 m/s and 3 of the 7 (43%) increasing by more than 0.16 m/s (Figure 2B). An examination of the time course of changes in walking speed across days revealed that users had a progressive increase in their usual walking speed over time, presenting with a faster speed to start each training visit (Figure 2B). Participants’ fast walking speed similarly increased across sessions, but to a lesser degree, with only 3 of the 7 users (43%) increasing by more than the large meaningful change score of 0.10 m/s (Figure 3B).
Across the training visits, all users concurrently increased their walking cadence, with an average increase of 5.40 ± 1.26 steps/min (P = 0.01) when walking at their usual walking speed and an average increase of 4.84 ± 2.03 steps/min (P = 0.05) when walking at their fast walking speed. Changes in walking cadence were highly related to changes in usual walking speed (R2 = 0.71, P < 0.05) and fast walking speed (R2 = 0.79, P < 0.05) (Figure 4).
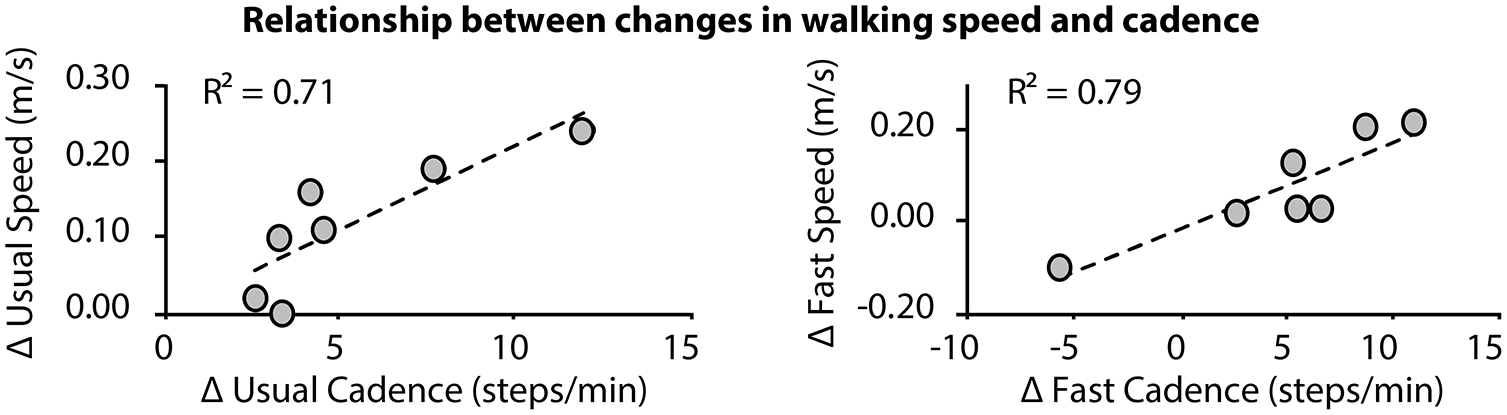
Across-session changes in usual (left) and fast (right) walking speeds were highly associated with changes in usual and fast walking cadences.
Discussion
We assessed the safety, feasibility, and rehabilitative potential of providing individualized and progressive music-based, rhythmic gait training using an automated digital rehabilitation platform. The platform consisted of a smartphone application preloaded with a music playlist, wearable movement sensors, and decision algorithms that modulate musical tempo in real-time based on the sensor data. We show that community-dwelling individuals poststroke can entrain to the individualized and progressive musical tempo, increase their walking speed with training, and are sufficiently motivated by the music-based gait training experience to express interest in using the platform for gait training at home.
Entrainment and Cadence Improvements
Faster walking is the result of increased cadence, stride length, or a combination of the two. Prior studies of how individuals poststroke increase walking speed have shown that while cadence and stride length may both increase, 36 people poststroke tend to walk faster by increasing stride length.37,38 An explanation for this is that a reduced ability to increase plantarflexor moments requires people with poststroke hemiparesis to rely primarily on increasing their trailing limb angle to meet the higher propulsive demands of walking faster.39,40 In contrast, the systematic progression of the musical tempo delivered by our platform was designed to leverage auditory-motor coupling to produce faster walking speeds by increasing walking cadence. Consistent with this hypothesis, we observed that posttraining improvements in walking speed were highly correlated with improvements in walking cadence (Figure 4). It is also noteworthy that whereas only 5 of the 7 study participants increased their walking speed across the 3 training sessions, all 7 study participants increased their cadence. Mechanistically, it is possible that the rhythm-based gait training facilitated more symmetrical corticomotor input to the plantarflexor muscles during walking—a neuromotor change associated with more symmetrical plantarflexor moments during walking. 41 Indeed, studying the effects of this training approach on poststroke propulsion function is warranted given the dearth of therapies available to treat poststroke propulsion impairments. 42 Taken together, these findings support the targeted nature of the rhythm-based training protocol automated by this digital therapeutic platform.
It is worth noting that faster walking cadences do not always lead to faster walking speeds. Indeed, patients with multiple sclerosis were reported to increase their walking cadence in response to either rhythmic music or a metronome but concomitantly decreased their overall walking speed. 43 Whereas this contrasts with other studies that have shown increased walking speed with rhythmic auditory stimulation in the multiple sclerosis population, 44 certain factors within and across diagnostic groups may cause individuals to adopt shorter stride lengths with faster cadences, or slower cadences with larger stride lengths, to preserve walking speed. For people poststroke, given (a) the higher propulsive demand required of taking faster steps and (b) the propulsive deficit characteristic of poststroke hemiparesis, it may be advantageous to train faster stepping even if overall speed remains unchanged (eg, fixing speed using a treadmill or pacing individuals while they walk overground). Examination of the effects that different speed- and cadence-based treatment paradigms have on the mechanics and energetics of poststroke walking, and the potential role that sensor-driven technologies may have in facilitating their delivery, is warranted.
Individuals poststroke can entrain to both nonmusical and musical rhythmic stimuli (Table 2). Our study participants appeared to entrain better to the nonmusical metronome than to the musical rhythmic stimuli; however, differences in measurement conditions likely account for this. That is, entrainment to the musical stimuli was measured during the training period when the platform’s decision algorithms were actively progressing the musical tempo, and thus the user’s target cadence, to more challenging levels. In contrast, entrainment to the nonmusical metronome beat was measured during a short pretraining test of users’ rhythm synchronization ability. Nonetheless, given the evidence that individuals with neurological disorders may entrain better to nonmusical rhythmic stimuli,43,45 a key aspect of the platform’s decision algorithms is the automated overlay of a metronome beat when users struggle to entrain to the musical stimuli alone (see section Digital Therapeutic Platform and Algorithms). This overlay reinforces, not replaces, the music’s rhythm, and thus preserves the salience of the music-based gait training experience while facilitating better entrainment.
Speed Improvements
Consistent with prior studies across multiple patient populations, including older adults, 46 Parkinson’s disease,30,47 multiple sclerosis, 44 and stroke,19,20,23,48 the rhythmic gait training delivered by the digital therapeutic resulted in meaningful increases in walking speed in the majority of study participants (Figures 2 and 3). Notably, these marked improvements were observed following only three training visits, with many study participants improving by meaningful magnitudes after the first training visit (Figures 2A and 3A). The short training duration required to produce these speed gains is noteworthy given that 16 training sessions provided over a 4-week period has been recommended as an effective “dose” for gait training with rhythmic auditory stimulation (see Nascimento et al 22 and Ghai and Ghai 23 ). Future studies are required to examine the durability of this music-based digital therapeutic’s promising effects on walking speed and the cumulative effects of a longer training duration. Additionally, more work is needed to determine optimal training parameters (eg, training time and frequency), and how these may need to vary across individuals with different baseline abilities.
This foundational study demonstrates the feasibility of using wearable sensors to measure walking cadence in real-time, compute the degree of motor entrainment to the rhythmic auditory stimuli, and use these data to automate the treatment progression. Indeed, a unique aspect of the gait training provided by the platform is the use of real-time wearable sensor data to modulate target cadences to levels (either faster or slower) that may be more appropriate for the individual patient. This data-driven approach to training progression within a session contrasts with other paradigms that depend on predetermined progressions (see Suh et al, 18 Bella et al, 30 and Moumdjian et al 43 ). Whereas we posit that data-driven individualized progressions are likely to be superior, a comparison of these alternative approaches is warranted.
Potential Neural Predictors of Response
The rhythmic delivery of auditory stimuli provides a stable temporal template that simplifies motor planning and the calibration of movement parameters. 15 Indeed, when the movement duration is known (eg, the time between beats in a musical composition), it is thought that a listener can leverage anticipatory responses to synchronize their movements to the auditory stimulus. 32 Being able to perceive external rhythmic stimuli and produce rhythmic movements has been posited to be important for rhythm-based gait training 49 (see Damm et al 50 for review). Baseline measures of neuromotor ability and rhythmic motor control may thus influence treatment responses.45,51 Inconsistent with this hypothesis, as well as with findings in patients with Parkinson’s disease, 30 our study participants’ baseline rhythm synchronization ability was not related to the improvements resulting from the training. However, given that rhythm synchronization requires both rhythm perception and production, 49 it is possible that one of these components may have better prognostic value, warranting investigation.
Beyond measures of neuromotor behavior, lesion location—which was not measured in the present study—may also influence the treatment response; however, this is not yet well understood. For example, an intact basal ganglia–thalamo-cortical (BGTC) pathway may be crucial for the self-initiated rhythm6-8 needed to facilitate the transfer of gains made while gait training with external rhythmic stimuli to activities without external rhythmic stimuli; however, patients with BGTC disruption may not present with impaired rhythm synchronization abilities during rhythm-based gait training. Indeed, in compensation, they may engage the cerebellar-thalamo-cortical pathway, which facilitates movement synchronization to external stimuli 5 and is often exploited during treatment for BGTC disorders.30,47,52-54 Moreover, injury to the arcuate fasciculus—which directly couples auditory and motor cortical structures2-5 and plays a crucial role in rhythm perception and auditory-motor entrainment 5 —may lead to reduced outcomes even in patients with an intact BGTC pathway. Prognostic markers of the therapeutic response to rhythm-based gait training are required to prospectively identify likely responders and nonresponders to this targeted therapy. Given the heterogeneity of poststroke impairments, these markers may ultimately necessitate the integration of neuroimaging metrics with measurements of neuromotor behavior.
Limitations
This preliminary, proof-of-concept study of a novel, music-based digital therapeutic has several limitations. These include the lack of a control group, small sample size, and limited participant characteristics. Our future research testing more refined versions of the technology and algorithms will address these limitations by including an active control group, a larger study sample, and the additional baseline assessments necessary to fully characterize study participants and elucidate the generalizability of the results.
Interventions that aim to promote faster walking may need to account for individual characteristics that may make faster walking unsafe (eg, gait asymmetry). Thus, future versions of this digital rehabilitation technology may benefit from real-time measurements of gait quality to inform the modulation of the target walking cadence. However, it should be noted that despite our objective to advance the automation of a progressive and individualized rhythm-based walking training program for people poststroke, it is beyond the scope of the technology—at least in its current form—to identify appropriate candidates for this automated intervention or to define the conditions for its implementation. As with all interventions for the heterogenous poststroke population, individual patient characteristics and use conditions should be considered and may warrant modification of various factors, including the level of supervision required, the need to monitor certain physiological parameters, or to define appropriate treatment settings. That is to say, the automation of rhythm individualization and progression that is provided by this digital therapeutic does not replace the need for clinical oversight, which may range from an initial evaluation and prescription to a period of supervised use in the clinic before unsupervised use in the home and community is recommended.
Conclusions
Wearable movement sensors have rapidly gained popularity for rehabilitation applications.55-65 In this proof-of-concept study, we describe a novel digital rehabilitation platform that leverages real-time movement data collected by wearable sensors to automate an individualized and progressive locomotor rehabilitation program centered on a salient musical experience. Our findings demonstrate the targeted nature of the intervention and provide qualitative data that confirm the value that patients see in a music-based digital therapeutic. The intervention is readily accepted by individuals poststroke and has the potential to facilitate community-based rehabilitation. These findings, taken together with the growing body of work supporting rhythmic auditory stimulation for poststroke gait training,12,18-20,22,23 motivate further study and translation of this promising rehabilitation technology and intervention.
Supplemental Material
sj-docx-1-nnr-10.1177_1545968320961114 – Supplemental material for A Music-Based Digital Therapeutic: Proof-of-Concept Automation of a Progressive and Individualized Rhythm-Based Walking Training Program After Stroke
Supplemental material, sj-docx-1-nnr-10.1177_1545968320961114 for A Music-Based Digital Therapeutic: Proof-of-Concept Automation of a Progressive and Individualized Rhythm-Based Walking Training Program After Stroke by Karen Hutchinson, Regina Sloutsky, Ashley Collimore, Benjamin Adams, Brian Harris, Terry D. Ellis and Louis N. Awad in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-2-nnr-10.1177_1545968320961114 – Supplemental material for A Music-Based Digital Therapeutic: Proof-of-Concept Automation of a Progressive and Individualized Rhythm-Based Walking Training Program After Stroke
Supplemental material, sj-docx-2-nnr-10.1177_1545968320961114 for A Music-Based Digital Therapeutic: Proof-of-Concept Automation of a Progressive and Individualized Rhythm-Based Walking Training Program After Stroke by Karen Hutchinson, Regina Sloutsky, Ashley Collimore, Benjamin Adams, Brian Harris, Terry D. Ellis and Louis N. Awad in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We thank Stian Hanson, Eric Richardson, and members of the Neuromotor Recovery Laboratory at Boston University—especially Ge Chen, Ashlyn Aiello, and Andy Alvarez—for assistance with data collection.
Author Contributions
LNA, TE, KJH, and BH designed the study. RS, AC, BA collected and analyzed the data. LNA and KJH produced the manuscript drafts. LNA produced the figures. RS produced the tables. All authors reviewed and approved drafts of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LNA is a paid advisor to MedRhythms Inc. BH is cofounder and CEO of MedRhythms Inc. This study was completed under a master management plan instituted by the Boston University Financial Conflicts of Interest Committee.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Institutes of Health (Grant KL2TR001411) and MedRhythms, Inc.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
