Abstract
Background. Ischemic stroke carries a high mortality rate and is a leading cause of severe neurological disability. However, the efficacy of current therapeutic options remains limited. Objective. We aimed to investigate the treatment efficacy of transcranial direct current stimulation (tDCS) in motor function rehabilitation after ischemic stroke and explore the underlying mechanisms. Methods. Male Sprague-Dawley rats with epicranial electrodes were used to establish pathogenetic model through temporary right middle cerebral artery occlusion (MCAO). Subsequently, animals were randomly divided into 4 groups: MCAO + tDCS/sham tDCS, Control + tDCS/sham tDCS. Animals in the groups with tDCS underwent 10 days of cathodal tDCS totally (500 µA, 15 minutes, once a day). During and after tDCS treatment, the motor functions of the animals, ischemic damage area, proliferation and differentiation of neural stem cells (NSCs), and distribution, and protein expression of Notch1 signaling molecules were detected. Results. The rehabilitation of MCAO-induced motor function deficits was dramatically accelerated by tDCS treatment. NSC proliferation in the subventricular zone (SVZ) was significantly increased after MCAO surgery, and tDCS treatment promoted this process. Additionally, NSCs probably migrated from the SVZ to the ischemic striatum and then differentiated into neurons and oligodendrocytes after MCAO surgery, both of which processes were accelerated by tDCS treatment. Finally, tDCS treatment inhibited the activation of Notch1 signaling in NSCs in the ischemic striatum, which may be involved in NSC differentiation in the MCAO model. Conclusion. Our results suggest that tDCS may exert therapeutic efficacy after ischemic stroke in a regenerative medical perspective.
Introduction
Stroke is the second leading cause of death in people older than 60 years worldwide and the fifth leading cause of death in people within the age range of 15 to 59 years. 1 Among stroke-related deaths, ischemic events account for up to 80% of the stroke events. 2 Approximately 60% of stroke survivors suffer from motor disability at 6 months after stroke. 3 However, current therapeutic options remain limited, including intravenous administration of recombinant tissue plasminogen activator and endovascular intervention. 4
Recently, stem cell transplantation has been suggested as an effective alternative therapy for neurological disorders, including ischemic stroke.5,6 However, stem cell transplantation presents several outstanding risks, including immunological rejection and the capacity for oncogenicity. Neural stem cells (NSCs) exist in the subventricular zone (SVZ) of the lateral ventricle and the subgranular zone (SGZ) of the dentate gyrus throughout adulthood in mammals and can differentiate into most of the cerebral cell types affected by ischemic insult. 7 Therefore, the activation of endogenous NSCs for proliferation, migration, and differentiation in injured brain regions may exert therapeutic efficacy.
Transcranial direct current stimulation (tDCS) is a noninvasive neural control technology that has been increasingly proved to modulate plasticity in neuropsychiatric disease states.8,9 Importantly, the therapeutic potential of tDCS in stroke sequelae has been reported in clinical trials10-12 and animal studies13-15; however, the underlying mechanism remains unclear. In this study, we investigated the effects of tDCS treatment on motor function recovery and neurogenesis in rats after ischemic stroke and explored the underlying mechanisms.
Materials and Methods
Animals and Surgery
Male Sprague-Dawley rats (230-250 g), purchased from the Laboratory Animal Center of the Fourth Military Medical University (Xi’an, China), were housed in a temperature-controlled room in plastic cages (3 animals per cage), with free access to food and water at 22°C to 25°C on a 12-hour light/dark cycle. All experiments reported in this study were conducted according to an experimental protocol approved by the Animal Use and Care Committee for Research and Education of our institute (No. 20170607).
Four days before the middle cerebral artery occlusion (MCAO) operation, epicranial electrode implantation was performed in rats under 1% pentobarbital sodium anesthesia (45 mg/kg intraperitoneally [ip]). After removal of the scalp and underlying tissues, a homemade epicranial electrode with a defined contact area of 3.5 mm2 was mounted on the intact skull at the coordinates bregma AP + 2.0 mm and ML + 2.0 mm using glass ionomer dental cement (Ketac Cem, ESPE Dental AG, Seefeld, Germany). The electrode remained in place during the whole experiment.
After 24 hours, animals with implanted electrodes were trained on a rotarod for 5 trials per day at a speed of 4 rpm until they could run continuously for a minimum of 300 s. Then, 117 animals underwent a MCAO surgery with a silicone-coated nylon suture (RWD Life Science, Shenzhen, China), and 58 animals underwent a sham operation, as described previously. 16 To correct fluid loss during the surgical intervention, 0.5 mL of sterile isotonic saline (0.9% NaCl ip) was injected in each animal.
Groups and tDCS Treatment
At 1 day post-MCAO operation (POD 1), the severity of neurological deficits was evaluated by a 5-point deficit score, as previously reported. 17 A total of 62 animals with moderate neurological deficits (grade 2 of 3) selected from 117 stroke animals were randomly divided into 2 experimental groups: tDCS group (MCAO + tDCS, n = 32) and sham tDCS group (MCAO + Sham, n = 30). The other 58 sham stroke animals were randomly divided into 2 control groups: Control + tDCS (n = 29) and Control + Sham (n = 29). The animals in the 2 tDCS groups underwent cathodal tDCS (500 µA, 15 minutes, once per day; corresponding charge density is 128 571 C/m2) with a constant current stimulator (PMP18-3TR, Kikusui, Japan) beginning on POD 2 (Figure 1); this parameter has been frequently adopted in tDCS therapeutic studies.14,18 The animals in the 2 sham tDCS groups were only connected to the stimulator, with no current output. To avoid stimulation break effects, the current intensity was automatically ramped up and down over a 10-s period instead of being switched on and off abruptly. 19 Notably, the animals were maintained in an awake state during tDCS treatment to mimic the clinical application and to avoid possible interactions between tDCS and anesthetic drugs.20,21
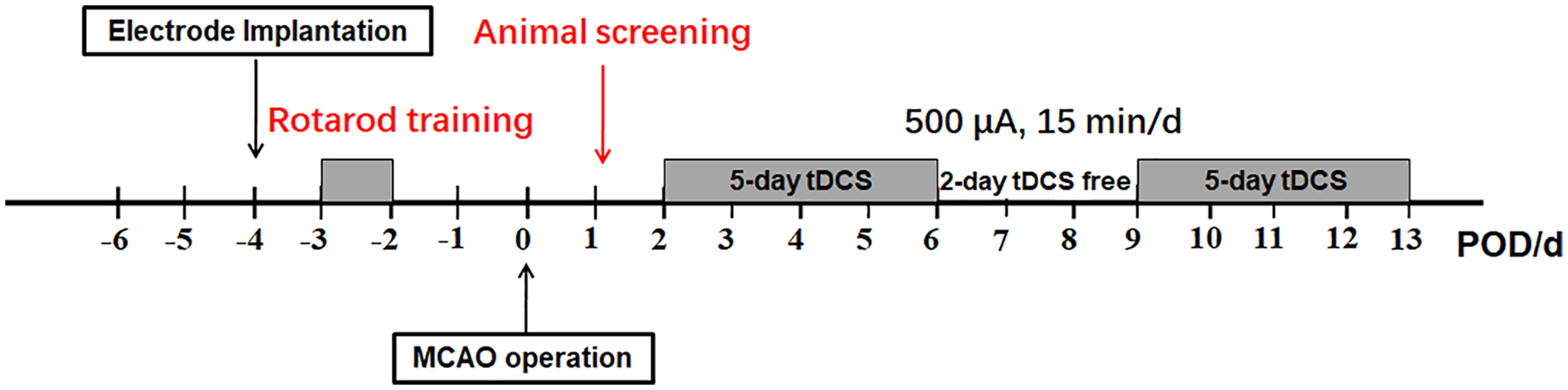
Experimental procedure and tDCS administration.
5′-Bromo-2-Deoxyuridine (BrdU) Administration
BrdU labeling is commonly used to track cell proliferation and differentiation. The animals were injected with 60 mg/kg BrdU ip (Sigma-Aldrich, Missouri) once a day for 7 consecutive days (ie, POD 0 to POD 6), as previously reported. 22
Behavioral Assessments
Open Field Test
The open field test was carried out 2 days before MCAO surgery as a baseline and then recorded every 2 days from POD 2 to POD 14. Immediately after tDCS, animals were habituated to the test environment for at least 30 minutes, and locomotor activity was evaluated by 2 investigators who were blinded to the experimental design. 23 First, rats were gently placed at the center of the open field device (100 cm × 100 cm × 40 cm) with their backs toward the investigator. Then, each rat was allowed to explore freely for 5 minutes, and movement traces were monitored by the Noldus Ethovision XT 5 system.
Rotarod Test
Athletic endurance (ie, duration time) was evaluated by a rotarod treadmill (Ugo Basile, Comerio, Italy) under an accelerating rotor mode (10 speeds from 4 to 40 rpm for 5 minutes), as previously described. 24 The baseline was recorded after 2 consecutive days of training before MCAO surgery, and athletic endurance was evaluated every 2 days from POD 2 to POD 14.
Footprint Analysis
Footprint analysis was performed to evaluate the ischemic stroke–induced motor deficits as previously. 14 Briefly, the animal’s left forepaw (contralateral to MCAO surgery) was dipped in a dye. 25 Then, the animals were allowed to walk across a narrow box (50 cm × 8.5 cm × 10 cm) with a sheet of white paper on the bottom. The footprints were recorded at 1 day before MCAO and every 2 days from POD 2 to POD 14, respectively.
2,3,5-Triphenyltetrazolium Chloride (TTC) Staining
TTC staining is widely used to evaluate the severity of cerebral ischemia. 26 Immediately after tDCS treatment on POD 3, the brains were collected quickly under anesthesia, frozen at −20°C for 15 minutes, and then sliced coronally into 2-mm thick sections and stained with 2% TTC (Sigma-Aldrich) at 37°C for 30 minutes.
Immunofluorescence Confocal Staining (IFC)
At POD 3 and POD 7, the animals were deeply anesthetized by pentobarbital (60 mg/kg ip) and transcardially perfused with 200 mL of 5-mM sodium phosphate-buffered 0.9% (w/v) saline (PBS; pH 7.2-7.4) followed by 500 mL of 4% paraformaldehyde in phosphate buffer. The brains were harvested and immersed in 30% (w/v) sucrose at 4°C until they sank to the bottom. Serial coronal, 25-µm brain sections were prepared using a cryotome (Leica CM1800; Heidelberg, Germany) and stored at −20°C. For IFC staining, the sections were rinsed in PBS 3 times (5 minutes each) and then blocked in primary antibody dilution buffer (2% [w/v] bovine serum albumin [BSA] and 0.3% [v/v] Triton X-100 in 0.1% PBS) for 30 minutes at 37°C. After that, the following primary antibodies were individually added to the sections: monoclonal mouse anti-Nestin (1:250, cat# sc-33677, Santa Cruz Biotechnology, CA), polyclonal rabbit anti-doublecortin (DCX; 1:200, cat# ab18723, Abcam, UK), polyclonal rabbit anti-NG2 (1:400, cat# AB5320, Millipore, Germany), monoclonal rabbit anti-NeuN (1:6000, cat# ab177487, Abcam, UK), monoclonal rabbit anti-Hes1 (1:200, cat# ab108937, Abcam, UK), monoclonal rat anti-BrdU (1:200, cat# ab6326, Abcam, UK), and monoclonal rabbit anti-Ki67 (1:250, cat# ab16667, Abcam, UK). The sections were incubated overnight at 4°C and rewarmed at room temperature for 40 minutes. After being washed in PBS, the appropriate corresponding secondary antibody was added to the sections: Alexa Fluor 594-conjugated donkey anti-rabbit IgG (1:500, cat# 127803, Jackson Immuno Research Labs, West Grove, PA), Alexa Fluor 488-conjugated donkey anti-mouse IgG (1:500, cat# 127820, Jackson Immuno Research Labs, West Grove, PA), or Alexa Fluor 488-conjugated donkey anti-rat IgG (1:800, cat# A21208, Thermo Fisher, USA). The sections were incubated with secondary antibodies for 3 hours at room temperature. Finally, all the sections were mounted onto glass slides and cover-slipped with 50% (v/v) glycerol and 2.5% (w/v) triethylenediamine (antifading agent) in 5-mM PBS. Digital images were obtained using a confocal laser-scanning microscope with Z-stack and projection function (LSM 800, Zeiss, Oberkochen, Germany). For quantitative analysis, the software Image J was adopted, and 3 different fields per section were analyzed in a “blinded” manner. Briefly, the immunofluorescence images were converted to grayscale images; then after subtracting the background and adjusting the threshold, the cell number and fluorescence intensity within the area of interest were calculated.
Transmission Electron Microscopy (TEM)
The brains were removed at POD 14 under anesthesia and rapidly infused with 4% paraformaldehyde for 30 minutes. Then, the striatum ipsilateral to the MCAO was cut into the size of 1 mm3 and fixed in 3% glutaraldehyde for 3 days at 4°C. After that the samples were postfixed with 1% osmium tetroxide in sodium cacodylate trihydrate at room temperature for 2 hours, dehydrated in a graded series of ethanol, and embedded in Epon resin (TAAB Laboratories Equipment, Aldermaston, UK). Thin (100-nm) sections were cut with an ultra-microtome (UC6, Leica, Heidelberg, Germany), stained with uranyl acetate and lead citrate, observed with TEM (JEM-1230 electron microscope, Jeol, Tokyo, Japan), and analyzed as previously. 27
Western Blotting (WB)
Protein expression levels of striatal tissues ipsilateral to the MCAO were detected by WB at POD 3 and POD 14. Briefly, the tissues were weighed and homogenized in radioimmunoprecipitation assay lysis buffer supplemented with the proteinase inhibitor phenylmethylsulfonyl fluoride using a homogenizer device (Leica, Heidelberg, Germany). The supernatant was acquired after centrifugation at 12 000 rpm for 20 minutes at 4°C, and the total protein levels were quantified by a bicinchoninic acid assay kit (Beyotime Biotechnology, Jiangsu, China). Subsequently, the samples were normalized by dilution with loading buffer (Beyotime Biotechnology, Jiangsu, China) and heated in a water bath for 8 minutes. Then, total protein (30-50 µg per lane) was separated on 10% SDS-polyacrylamide gels (Bio-Rad Laboratories, CA) by electrophoresis and transferred onto polyvinylidene difluoride membranes (Millipore, MA). Membranes were blocked in a 5% BSA solution for 2 hours and incubated with the following primary antibodies overnight at 4°C: monoclonal mouse anti-Jagged1 (1:800, cat# ab89663, Abcam, UK), polyclonal goat anti-Delta like-1 (DLL1; 1:1000, cat# ab85346, Abcam, UK), polyclonal rabbit anti-Notch1 (1:600, cat# E-AB-12815, Elabscience, China), polyclonal rabbit anti-NUMB (1:1500, cat# ab14140, Abcam, UK), polyclonal rabbit anti-Notch intracellular domain (NICD; 1:1000, cat# ab52301, Abcam, UK), polyclonal rabbit anti-RBPJK (1:3000, cat# ab180588, Abcam, UK), polyclonal rabbit anti-Hes1 (1:200, cat# ab108937, Abcam, UK), polyclonal rabbit anti–myelin basic protein (anti-MBP; 1:200, cat# ab40390, Abcam, UK), and monoclonal mouse anti-β-actin (1:3000, cat# 7G6, CMCTAG, USA). Finally, the membranes were incubated with horseradish peroxidase–conjugated secondary antibodies at a 1:5000 dilution for 3 hours at room temperature and visualized using an enhanced chemiluminescence reagent (Millipore, MA).
Statistical Analysis
All data were collected and analyzed by researchers blinded to the experimental design. Repeated-measures ANOVA, 1-way ANOVA with post hoc least significant difference t-test (LSD-t), and Breslow test were used for data analysis. All data are presented as the means ± SEM, and all statistical analyses were performed using SPSS version 20.0 (SPSS, Chicago, IL). P values less than .05 were considered as statistically significant.
Results
tDCS Accelerated Motor Function Rehabilitation After MCAO
The total distance traveled in open field in MCAO groups was significantly decreased (t = 7.45; P = .00) compared with that in control surgery groups. After tDCS administration, this index in MCAO + tDCS obviously improved at POD 6 (t = 3.21, P = .00, vs MCAO + Sham), and this priority maintained until POD 14 (F = 32.80, P < .00; Figure 2A). The retention time on rotarod was dramatically reduced after MCAO surgery (t = 4.57, P = .00; Figures 2B, 2B1, and 2B2). With tDCS treatment, this index significantly increased in MCAO + tDCS from POD 6 to POD 14 (F = 32.03, P = .00, vs MCAO + Sham; Figures 2B and 2B3).
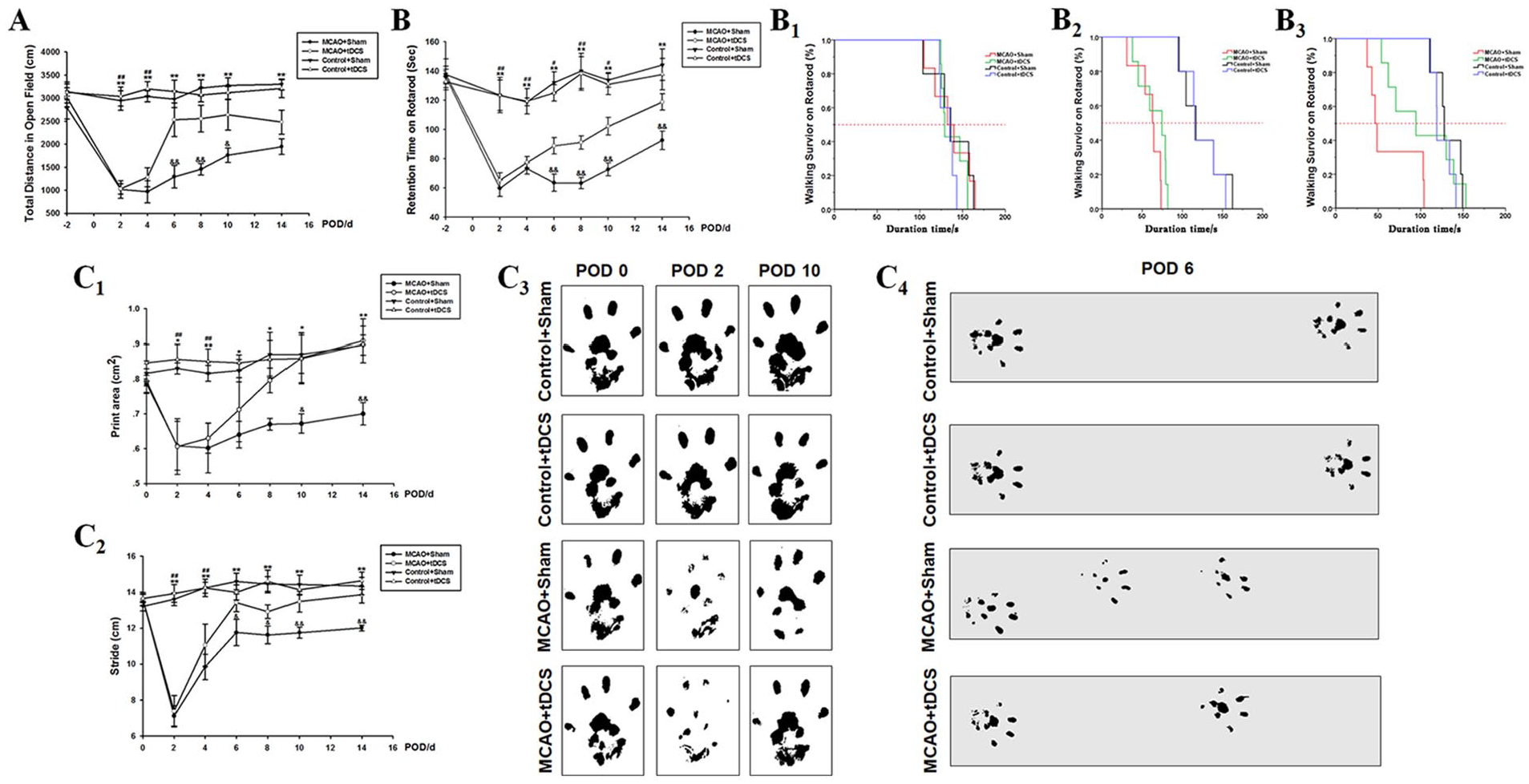
tDCS accelerated the rehabilitation of motor function deficits caused by MCAO. Total distance traveled in the open field (A): n = 6 for Control + Sham, Control + tDCS, and MCAO + Sham groups; n = 7 for the MCAO + tDCS group. Retention time on the rotarod (B): n = 5 for Control + Sham and Control + tDCS groups; n = 6 for the MCAO + Sham group; n = 7 for the MCAO + tDCS group. Survival analysis of retention time on the rotarod before MCAO surgery (B1), at POD 2 (B2), and at POD 6 (B3). Footprint analysis of the MCAO contralateral forepaw: quantitative analysis of footprint area (C1) and stride (C2); representative images of the print area (C3) and stride (C4); n = 5 per group. Data are presented as the means ± SEM.a
MCAO-induced claudication and tDCS efficacy were evaluated by footprint analysis. The print area, a surrogate for limb strength, gradually increased from POD 2 to POD 14 in MCAO rats and was obviously accelerated by tDCS treatment (F = 10.72, P = .00, vs other 3 groups; Figures 2C1 and 2C3). Additionally, the stride length, a surrogate for limping gait, significantly increased in MCAO + tDCS (t = 2.58, P = .20, vs MCAO + Sham; Figure 2C2 and 2C4). No obvious differences were found between the 2 control groups (print area: t = 0.32, P = .75; stride length: t = 0.10, P = .92; Figure 2).
tDCS Promoted NSC Proliferation in SVZ and Accelerated NSC Migration From the SVZ to the Ischemic Striatum of MCAO Model Rats
As shown in Figure 3A, a wide infarct area (shown in white color) including the right cortex and striatum was observed after MCAO surgery, whereas these areas were normal in control groups. The infarct area significantly decreased at POD 3 in MCAO + tDCS (t = 5.412, P = .00, vs MCAO + Sham; Figure 3A3). Also, some alive cells appeared in the ischemic striatum (circled by yellow dotted) of MCAO + tDCS, instead of MCAO + Sham at POD 3. To confirm whether those cells were NSCs, we performed IFC for Nestin. No Nestin-positive (Nestin+) cells were observed in the striatum of control animals, and many more Nestin+ cells appeared in the ischemic striatum of MCAO + tDCS rats (t = 18.96, P = .00, vs MCAO + Sham; Figure 3A). In addition, we found that the percentage of Nestin+/Ki67+ cells in SVZ of MCAO + tDCS rats was significantly higher than that in MCAO + Sham at POD 3 (t = 6.20; P = .00), indicating that tDCS accelerated the proliferation of NSCs (Figures 3A, and 3A1 and 3A2). However, the total number of Nestin+ cells in SVZ of MCAO + tDCS group was significantly lower than that in MCAO + Sham (t = 4.66; P = .00). Considering that Nestin+ cells appeared in the ischemic striatum of the MCAO + tDCS group but not in the other 3 groups at POD 3 (t = 20.22, P = .00, vs control; t = 18.96, P = .00, vs MCAO + Sham; Figures 3A, and 3A1 and 3A2), we speculated that Nestin+ cells existing in the ischemic striatum probably migrated from SVZ.
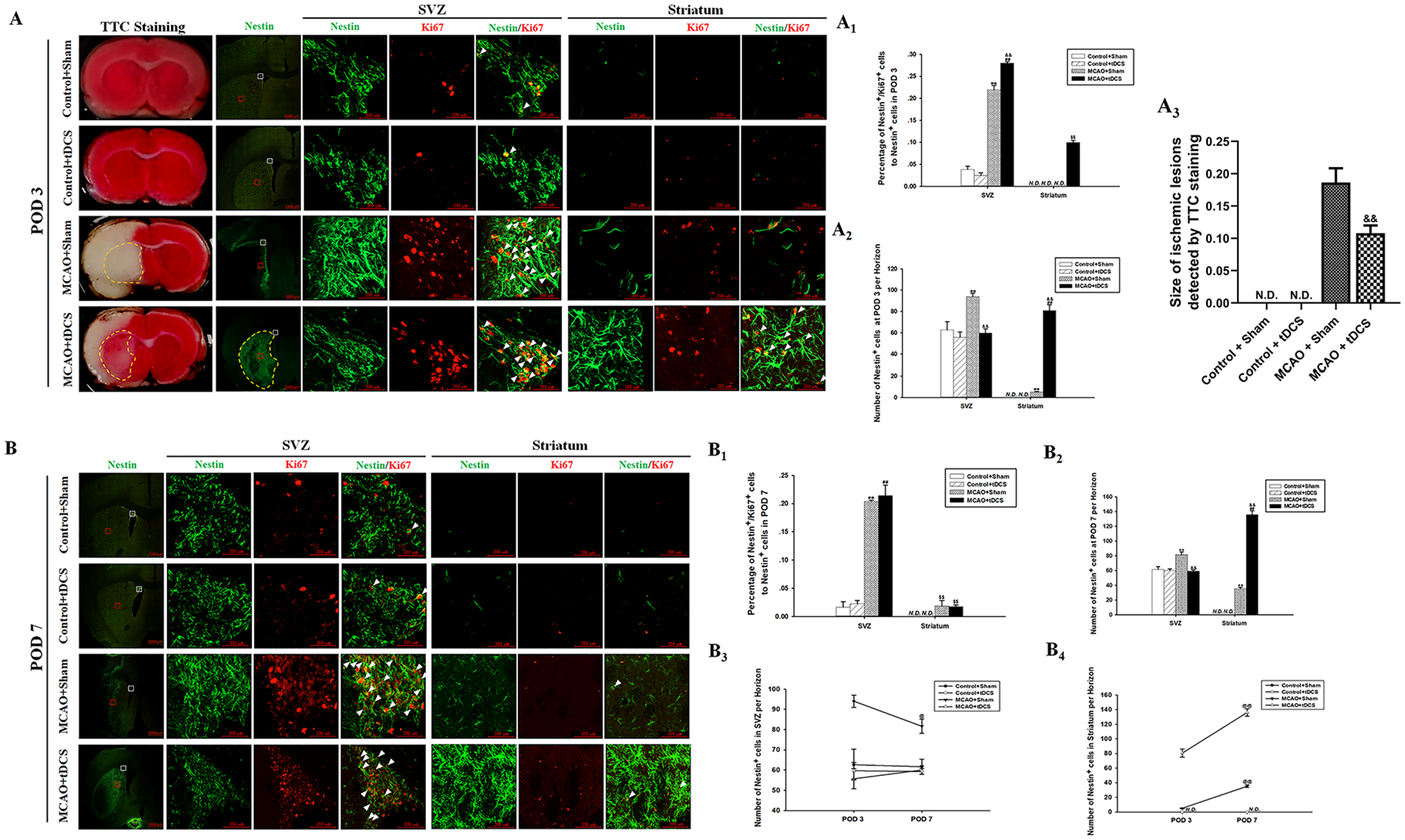
tDCS promoted NSC proliferation in the SVZ and accelerated NSC migration from the SVZ to the ischemic striatum after MCAO. TTC staining and Nestin/Ki67 double staining of brain sections at POD 3 (A); semiquantitative analysis of Nestin+ cell number (A1); semiquantitative analysis of the percentage of Nestin+/Ki67+ cells (A2); size of ischemic lesions detected by TTC staining (A3). Nestin/Ki67 double staining of brain sections at POD 7 (B); semiquantitative analysis of Nestin+ cell number (B1); semiquantitative analysis of the percentage of Nestin+/Ki67+ cells (B2). Comparison of Nestin+ cell number in the SVZ (B3) and striatum (B4) between POD 3 and POD 7. Data are presented as the means ± SEM from 3 independent experiments, and representative images are shown.a
At POD 7, the percentage of Nestin+/Ki67+ cells in the SVZ of MCAO groups dramatically increased (t = 12.12, P = .00, MCAO + Sham vs Control + Sham), and this index showed no significant difference between the 2 MCAO groups (t = 0.67, P = .52; Figures 3B and 3B1 and 3B2). However, Nestin+ cell numbers in the SVZ of MCAO + tDCS was similar to that of Control + tDCS (t = 0.54, P = .60, POD 3; t = 0.17, P = .87, POD 7) and obviously increased in the ischemic striatum (F = 52.99, P = .00, POD 7 vs POD 3; Figures 3B and 3B4). As for the MCAO + Sham group, no difference was found in NSC proliferation between POD 7 and POD 3 (F = 2.44, P = .19; Figures 3B and 3B3), but Nestin+ cell numbers significantly decreased in the SVZ (F = 7.00, P = .05, POD 7 vs POD 3) and dramatically increased in the ischemic striatum (F = 243.56, P = .00, POD 7 vs POD 3; Figures 3B and 3B4). No differences were found between the 2 control groups, regardless of Nestin+ cells in the SVZ or striatum.
tDCS Promoted NSC Differentiation in the Ischemic Striatum of MCAO Model Rats
As shown in Figures 4A1 and 4A2, the number of DCX+ and NG2+ cells dramatically increased in the ischemic striatum of MCAO + tDCS rats at POD 3 (t = 17.95, P = .00, DCX+ comparison; t = 8.13, P = .00, NG2+ comparison; vs MCAO + Sham), and this difference continued to POD 7 (F = 12.10, P = .03, DCX+ comparison; F = 133.01, P = .00, NG2+ comparison; Figures 4B1 to 4B3). Additionally, at POD 7 but not POD 3, some NeuN+/BrdU+ cells were found in the ischemic striatum of MCAO + tDCS rats (Figures 4B1 and 4B4); meanwhile, DCX+ cells in the ischemic striatum of MCAO + tDCS rats significantly decreased at POD7 (F = 37.87, P = .00, vs POD 3; Figure 4B3). Under the overall view, the fluorescence intensity showed a corresponding relation among Nestin+, DCX+, and NG2+ cells (Figures 4A and 4B). No DCX+ or NG2+ cells were observed in the striatum of the 2 control groups. Furthermore, the protein level of MBP obviously decreased in the ischemic striatum of MCAO + Sham rats (t = 8.63, P = .00, vs Control + Sham) and significantly increased in MCAO + tDCS (t = 3.83, P = .01, vs MCAO + Sham; Figure 4C) at POD 14. The results of TEM showed that the number of myelinated nerve fibers significantly increased (t = 11.08; P = .00), and the G-ratio of axons obviously decreased (t = 3.08; P = .02) in the ischemic striatum of MCAO + tDCS rats at POD 14 compared with those in MCAO + Sham.
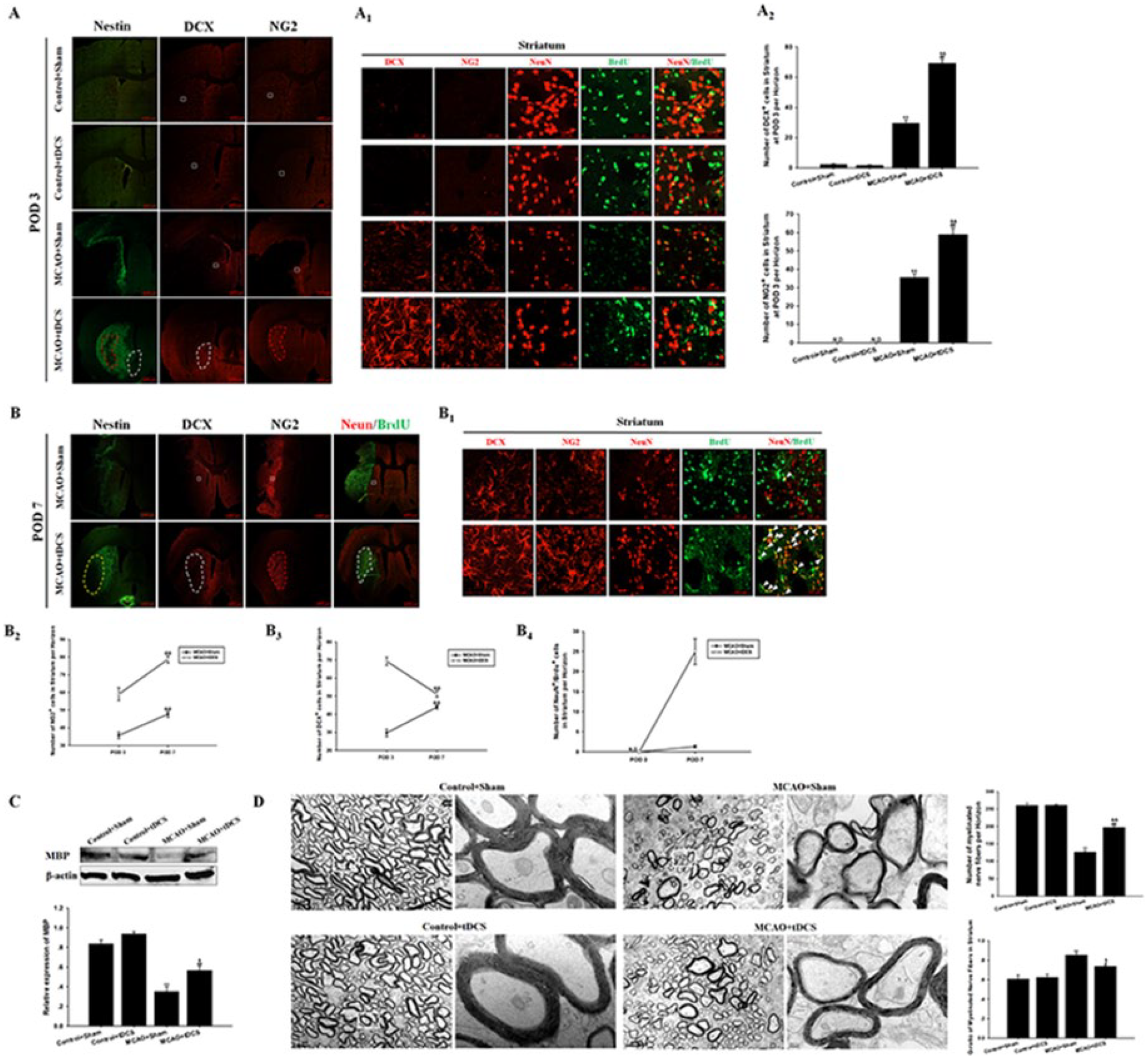
tDCS promoted NSC differentiation to neurons and oligodendrocytes in the ischemic striatum after MCAO. The macrograph (A) and micrograph (A1) of Nestin, DCX, NG2, and NeuN/BrdU staining at POD 3; semiquantitative analysis of the number of DCX+ and NG2+ cells (A2). The macrograph (B) and micrograph (B1) of Nestin, DCX, NG2, and NeuN/BrdU staining at POD 7. Comparison of the number of NG2+ (B2), DCX+ (B3), and NeuN+/BrdU+ cells (B4) in the striatum between POD 3 and POD 7. The protein level of MBP in the striatum at POD 14 (C). The number of myelinated nerve fibers and G-ratio of axons in the striatum under TEM (D). Data are presented as the means ± SEM from 3 independent experiments, and representative images are shown.a
tDCS Inhibited Notch1 Signaling Pathway Activation in NSCs in the Ischemic Striatum
To explore the role of Notch1 pathway in NSC differentiation after MCAO surgery, the protein levels of some critical Notch1 signaling molecules in the striatum were determined at POD 3. The expression of DLL1 obviously increased, and the level of Jagged1 was unchanged after MCAO surgery (t = 6.20, P = .00, DLL1 comparison; t = 1.93, P = .09, Jagged1 comparison; MCAO + Sham vs Control + Sham). Meanwhile, both indexes significantly decreased in MCAO + tDCS rats (t = 8.58, P = .00, Jagged1 comparison; t = 6.73, P = .00, DLL1 comparison; vs MCAO + Sham; Figure 5A). The expression of NUMB was upregulated in MCAO + tDCS rats (t = 5.46, P = .00, vs MCAO + Sham), and no difference was found in Notch1 and RBPJK expression among the 4 groups (F = 3.43, P = .07, Figure 5B; F = 2.23, P = .16, Figure 5C). The protein level of NICD and Hes1 was upregulated in MCAO + Sham rats (t = 3.27, P = .01, NICD comparison; t = 11.67, P = .00, Hes1 comparison; vs Control + Sham) and decreased in MCAO + tDCS rats (t = 7.43, P = .00, NICD comparison; t = 10.58, P = .00, Hes1 comparison; vs MCAO + Sham; Figure 5C).
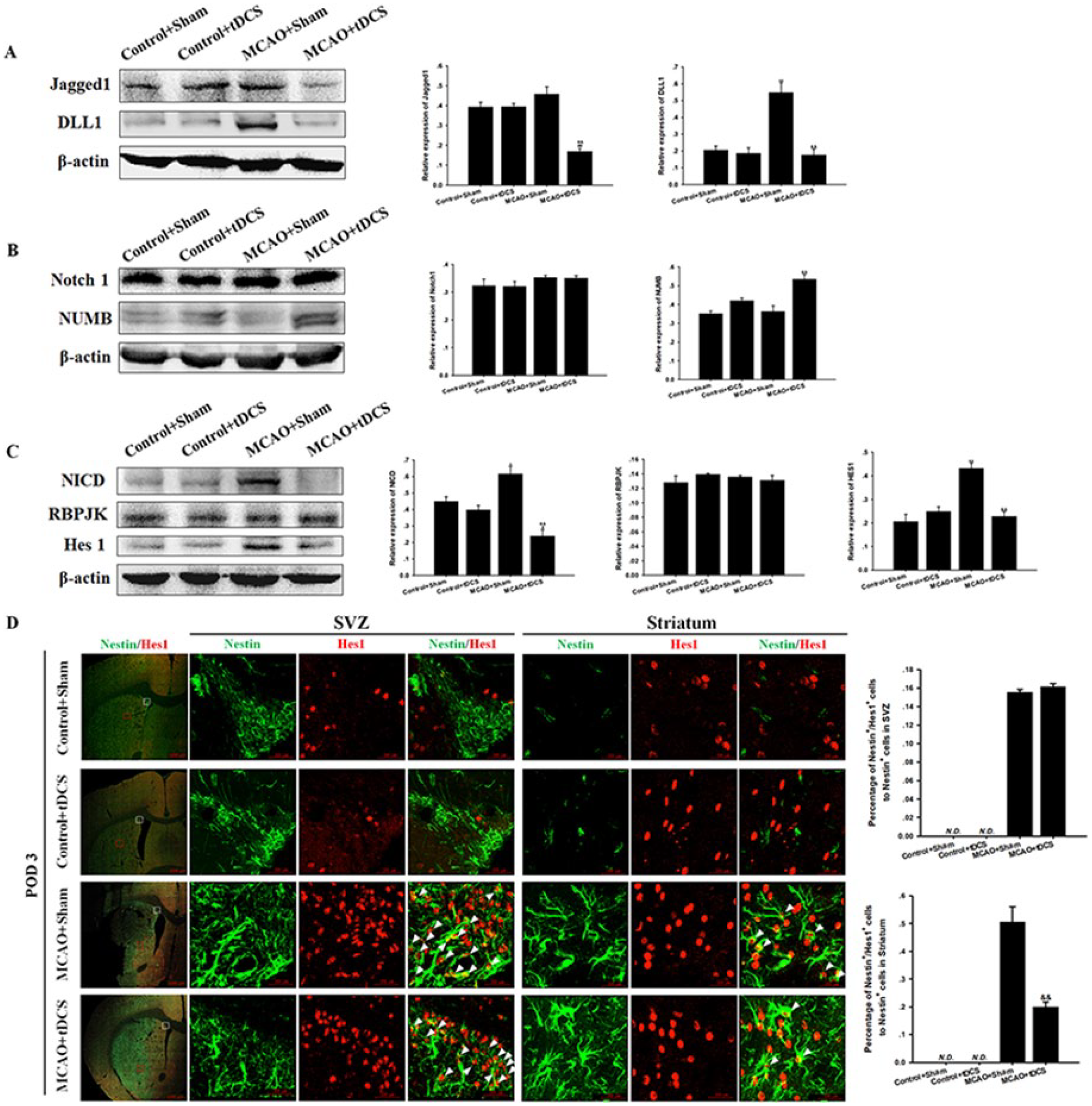
tDCS inhibited the activation of Notch1 signaling pathway in NSCs in the ischemic striatum. The protein levels of Jagged1 and DLL1 (A); Notch1 and NUMB (B); and NICD, RBPJK, and Hes1 (C) are shown. IFC staining results of Nestin/Hes1 at POD 3 (D). Data are presented as the means ± SEM from 3 independent experiments, and representative images are shown.a
Additionally, the percentage of Nestin+/Hes1+ cells significantly increased both in the SVZ and striatum after MCAO surgery (t = 45.34, P = .00, in SVZ; t = 12.19, P = .00, in the striatum; vs Control + Sham), whereas this index dramatically decreased in the striatum but not the SVZ of MCAO + tDCS rats (t = 1.72, P = .13, in the SVZ; t = 7.37, P = .00, in the striatum; vs MCAO + Sham; Figures 5D and 6), which was consistent with WB results. No Nestin+/Hes1+ cells were observed in the SVZ or striatum of the 2 control groups.
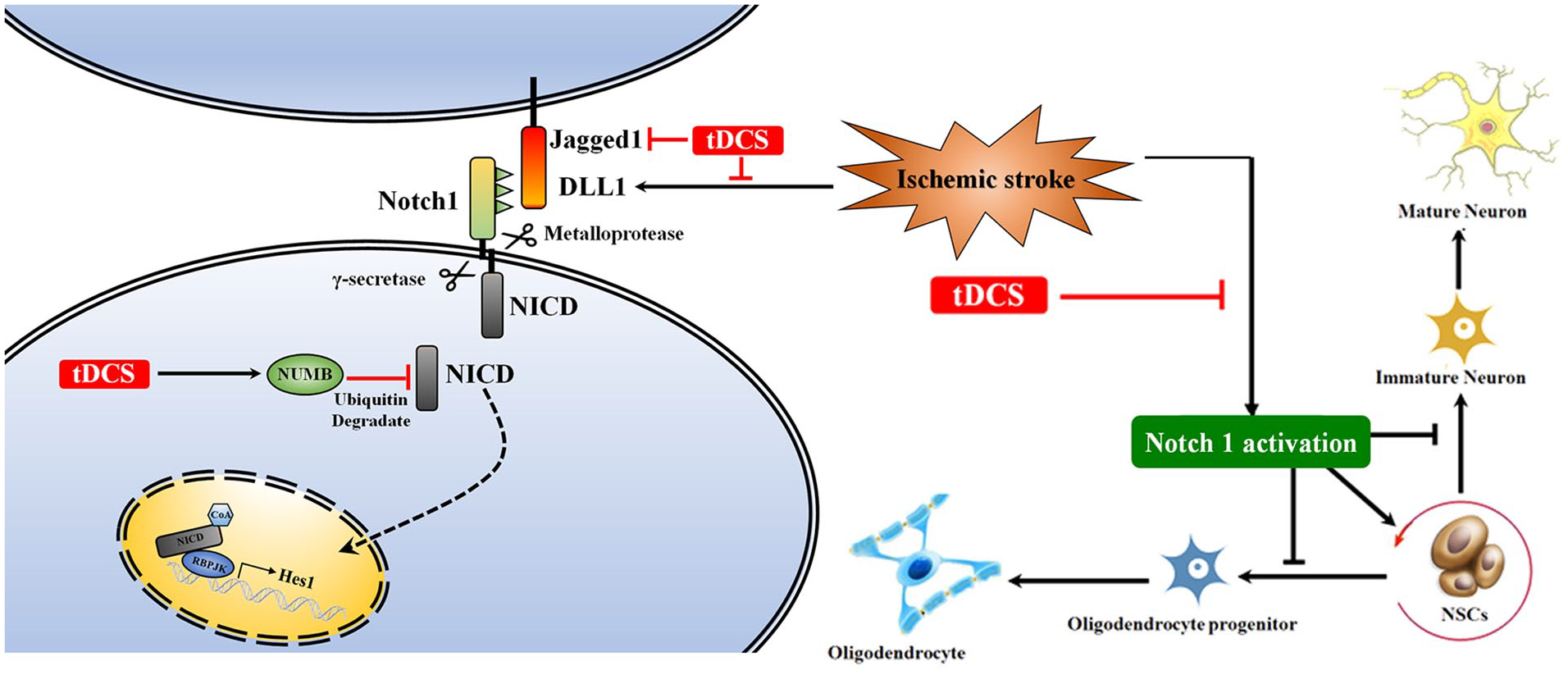
tDCS treatment inhibited the ischemic stroke–induced activation of the Notch1 pathway in NSCs in the ischemic striatum. Ischemic stroke activated the Notch1 pathway through DLL1 upregulation, whereas tDCS treatment inhibited this process through DLL1 and Jagged1 downregulation and NUMB upregulation; ischemic stroke–induced Notch1 activation promoted NSC proliferation, whereas tDCS treatment inhibited this process and promoted NSC differentiation to oligodendrocytes and neurons after MCAO.
Discussion
Stroke is a common cause of adult disability worldwide. More than two-thirds of stroke survivors develop poststroke sequelae, including impaired motor functions and spasticity. 28 The inability to walk severely reduces a patient’s functional independence and quality of life. Therefore, it is vital to accelerate the rehabilitation of motor function deficits for poststroke survivors. In this study, we found that tDCS treatment not only improved locomotor activity and athletic endurance deficits induced by MCAO, but also accelerated the recovery of limping gait, which is consistent with previous clinical research.29,30 However, some inconsistent results were reported, 31 and the tDCS parameters of current intensity, 32 location, and type (anodal or cathodal) of electrical stimulation used 33 as well as the age of subjects 34 may account for this conflict. Currently, we adopted the same parameters (500 µA, 15 minutes, cathodal) as those in a previous study. 14 Although the charge density (128 571 C/m²) in the present study is much higher than that in clinical studies with human patients, the bio-safety of this stimulating parameter and its efficacy for motor function recovery after ischemic stroke has been confirmed in rats. Notably, it is a problematic issue for tDCS clinical translation, and the differences in brain structure between rats and humans may affect the current density distribution as well as the tissue distribution. Thus, simple parameter conversion is not advisable, and the technology of computer simulation would be necessary.
To explore the underlying mechanisms of tDCS-induced rehabilitation, TTC and IFC staining were performed. The size of ischemic lesions significantly decreased after tDCS treatment, and some alive cells appeared in the ischemic striatum of MCAO + tDCS rats but not MCAO + Sham at POD 3; these cells were verified as Nestin+ (a recognized marker of NSCs). Not until POD 7were a few Nestin+ cells observed in the ischemic striatum of MCAO + Sham rats. These data above suggest that tDCS could increase NSC numbers in the ischemic striatum after MCAO. As reported, NSCs are mainly located in 2 neurogenic niches in the adult mammalian brain—the SVZ of the lateral ventricle 35 and the SGZ of the dentate gyrus 36 —and NSCs in the SVZ could be activated after brain injury.37,38 In the present study, we found that Nestin+ cells only appeared in the SVZ but not the striatum of control groups, and the proliferation of NSCs in SVZ significantly increased after ischemic stroke, which could be promoted by tDCS treatment. Additionally, tDCS did not increase the number of NSCs in the SVZ, but rather increased the number in the ischemic striatum where they theoretically did not previously reside. Based on the previous literature, 39 we speculate that NSCs in the ischemic striatum probably migrate from the SVZ. Besides, reactive astrocytes can also overexpress Nestin and exhibit progenitor-like properties. 40 Combined with the neuroprotective effect of tDCS, 18 it was possible for tDCS to salvage astrocytes in the surviving ischemic striatum and further contribute to the increased Nestin+ cells. Therefore, there might be 2 sources of Nestin+ cells. However, the proportion of NSCs that migrated or transformed is unclear, which needs to be explored in the future. Additionally, no differences were found in the number of proliferating NSCs in the 2 control groups, suggesting that tDCS does not affect the proliferation or migration of NSCs under physiological conditions. Furthermore, the number of reserved NSCs in the SVZ was the same between 2 tDCS groups, in spite of some out-migrated NSCs in MCAO + tDCS. It seems that the body has a strict control over the basic level of NSCs in neurogenic niches. 41
To further investigate whether the NSCs appearing at the ischemic striatum after MCAO surgery have a differentiation capacity, IFC staining for DCX and NG2 was performed. IFC results showed that the NSCs in the ischemic striatum could differentiate into immature neurons labeled with DCX and oligodendrocyte progenitor cells labeled with NG2 at POD 3 after MCAO surgery. Moreover, these immature neurons observed in the ischemic striatum have a capability to mature, and tDCS treatment could accelerate this process, because MCAO rats without tDCS treatment only had a few immature cells until POD 7. Besides, the number of myelinated nerve fibers, myelin sheath thickness, and the expression of MBP (a vital protein generated from mature oligodendrocytes) significantly increased in the ischemic striatum of MCAO + tDCS rats, indicating that oligodendrocyte progenitor cells could develop into mature functional cells and further integrate into neural circuits as reported. 42 In terms of control surgery groups, few DCX+ or NG2+ cells were observed in the striatum, suggesting that tDCS has no neurogenesis effects under physiological conditions.
Notch1 signaling pathway plays an important role in the modulation of NSC fate. 42 Once Notch1 is knocked out in mice, although quiescent NSCs still exist in the SVZ, 43 they cannot be activated. 44 Usually, the Notch pathway is initiated by the binding of DLL or Jagged to the extracellular domains of Notch receptors. Then, the NICD is released and complexes with the transcriptional repressor RBPJK and finally activates Notch target genes (Hes1). 45 To explore whether the Notch1 pathway participates in NSC differentiation regulation during tDCS treatment after MCAO surgery, several critical signaling molecules in the ischemic striatum were detected. We found that DLL1 was overexpressed in the ischemic striatum after MCAO surgery. Accordingly, the expression of NICD was upregulated, which resulted in Hes1 overexpression. However, tDCS treatment could inhibit MCAO-induced DLL1 overexpression, downregulate Jagged1 expression, and upregulate NUMB expression (a negative regulator of Notch to mediate the degradation of NICD 46 ). As a result, lower levels of NICD and Hes1 were found in the ischemic striatum of MCAO + tDCS rats, suggesting that tDCS inhibits MCAO-induced Notch1 activation in the striatum. What is more, double staining of Nestin+/Hes1+ verified that it is the Notch1 pathway within NSCs that are inhibited by tDCS treatment in the ischemic striatum. Considering that Notch inhibition promotes NSC differentiation into neurons and oligodendrocytes and inhibits NSC proliferation and differentiation into astrocytes,47-49 we speculate that Notch1 inhibition may play an important role in NSC fate determination in the MCAO-induced ischemic striatum and that tDCS treatment could modulate this pathway for neurogenesis and neural circuit reconstruction. However, this tDCS-Notch1 inhibition pattern was not found in the SVZ of MCAO rats, which might be explained by the microenvironment difference between the ischemic striatum and the SVZ. 50 Additionally, microenvironment difference between control and experimental groups may also explain why tDCS could neither activate quiescent NSCs nor modulate Notch1 expression within quiescent NSCs under physiological conditions. Therefore, microenvironment change induced by brain injury factors such as ischemic stroke may activate quiescent NSCs in the SVZ, and tDCS accelerates and amplifies this process, leading to functional recovery.
However, there are many limitations to the present study that need to be stated. First, the evidence of NSCs migrating from the SVZ to the ischemic striatum is not direct, which may be improved by the cell tracer technique. Also, because we do not have the specific gene knock out rats, we could not find the direct experimental link between behavioral effects and the cellular/molecular results. Next, the issue that improved behavior is attributed to neurogenesis or simply to the result of reduced infarct volume (based on TTC); this must be addressed. Finally, translating this preclinical study performed in rats to clinical application still needs to solve many problems, such as the stimulating parameter translation.
Conclusion
Our results suggest that under these experimental conditions, tDCS treatment can accelerate the recovery of motor function deficits and promote neurogenesis after ischemic stroke. The inhibition of the Notch1 pathway by tDCS treatment may be involved in NSC differentiation in the MCAO model.
Supplemental Material
Supplemental_data – Supplemental material for tDCS Accelerates the Rehabilitation of MCAO-Induced Motor Function Deficits via Neurogenesis Modulated by the Notch1 Signaling Pathway
Supplemental material, Supplemental_data for tDCS Accelerates the Rehabilitation of MCAO-Induced Motor Function Deficits via Neurogenesis Modulated by the Notch1 Signaling Pathway by Keying Zhang, Ling Guo, Junping Zhang, Gang Rui, Guangzhou An, Yan Zhou, Jiajin Lin, Junling Xing, Tao Zhao and Guirong Ding in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We thank Prof Mai Lu (Lan Zhou Jiao Tong University) for his technical assistance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the special fund of the Fourth Military Medical University (2016TSA-004) and the special fund of PLA (16CXZ020).
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website at website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
