Abstract
Background. Vagus nerve stimulation (VNS) paired with rehabilitation may improve upper-limb impairment and function after ischemic stroke. Objective. To report 1-year safety, feasibility, adherence, and outcome data from a home exercise program paired with VNS using long-term follow-up data from a randomized double-blind study of rehabilitation therapy paired with Active VNS (n = 8) or Control VNS (n = 9). Methods. All people were implanted with a VNS device and underwent 6 weeks in clinic therapy with Control or Active VNS followed by home exercises through day 90. Thereafter, participants and investigators were unblinded. The Control VNS group then received 6 weeks in-clinic Active VNS (Cross-VNS group). All participants then performed an individualized home exercise program with self-administered Active VNS. Data from this phase are reported here. Outcome measures were Fugl-Meyer Assessment—Upper Extremity (FMA-UE), Wolf Motor Function Test (Functional and Time), Box and Block Test, Nine-Hole Peg Test, Stroke Impact Scale, and Motor Activity Log. Results. There were no VNS treatment–related serious adverse events during the long-term therapy. Two participants discontinued prior to receiving the full crossover VNS. On average, participants performed 200 ± 63 home therapy sessions, representing device use on 57.4% of home exercise days available for each participant. Pooled analysis revealed that 1 year after randomization, the FMA-UE score increased by 9.2 points (95% CI = 4.7 to 13.7; P = .001; n = 15). Other functional measures were also improved at 1 year. Conclusions. VNS combined with rehabilitation is feasible, with good long-term adherence, and may improve arm function after ischemic stroke.
Introduction
Long-term problems with arm function affect many people after stroke and are associated with poor quality of life and disability.1,2 Intense, high-dose, repetitive, and goal-directed upper-limb rehabilitation improves motor function after chronic stroke.3,4 However, in many cases, motor function gains achieved in rehabilitation deteriorate over time after the in-clinic rehabilitation stops. 5 Adults with stroke still have the capacity to improve motor function several months to years after stroke. 6 The recruitment of residual motor networks to subserve lost function could be enhanced by capitalizing on the brain’s ability to reorganize long after the injury.7-9
One such approach is vagus nerve stimulation (VNS) paired with upper-limb rehabilitation training. Stimulation of the vagus nerve activates the ascending neuromodulatory network that releases plasticity-promoting neuromodulators, including acetylcholine and norepinephrine, throughout the cortex.10,11 VNS-driven activation of this system, concurrent with motor training, drives robust and specific plasticity in rat primary motor cortex. 12 In animal models of stroke, VNS combined with task-specific motor training enhanced motor recovery compared with motor training without VNS or VNS alone.10,13 In another study, increased synaptic connectivity in corticospinal tracts controlling the impaired forelimb was observed after rehabilitation with VNS compared with rehabilitation alone. 14
In humans, VNS paired with rehabilitation was assessed in 2 pilot clinical trials in individuals with long-term moderate to severe arm weakness after ischemic stroke.15,16 Both studies included 6 weeks of in-clinic rehabilitation and blinded assessment of outcomes at 90 days. However, in the second study, in-clinic rehabilitation was followed by an individualized daily home exercise program paired with self-administered VNS, 16 and only in this study were further improvements seen after in-clinic therapy. In this study, all participants then received Active VNS paired with home exercises during unblinded long-term follow-up. In this article, we report the 1-year data from this second clinical pilot study. 16 Our aim was to assess the long-term safety and feasibility of and adherence to home-based self-administered VNS paired with rehabilitation in people with chronic stroke. We also assessed whether continuation of the self-directed, home-based VNS program improved upper-limb impairment and function over 1 year.
Methods
Participants were enrolled at 3 centers in the United States (University of Texas Southwestern Medical Center, TX; TIRR Memorial Herman, Houston, TX; and University of Minnesota, MN) and 1 in the United Kingdom (NHS Greater Glasgow and Clyde/University of Glasgow). The study was approved by the institutional review board or ethics committee at each site, and the appropriate regulatory approvals were obtained (FDA Investigational Device Exemption [IDE, No. 130287] and UK MHRA No. CI/2015/0011). Written informed consent from each participant was obtained in compliance with the requirements set forth in US FDA, Code of Federal Regulations Title 21. The study was registered on ClinicalTrials.gov (NCT02243020) and conducted according to the Declaration of Helsinki.
Participants with a history of unilateral supratentorial ischemic stroke that occurred 4 months to 5 years prior to randomization, aged between 30 and 80 years, and with a Fugl-Meyer Assessment—Upper Extremity (FMA-UE) score between 20 and 50 out of a maximum score of 66 were eligible for inclusion. Full inclusion and exclusion criteria have been published previously. 16
Methods for the randomized portion of the therapy (in-clinic therapy) have been previously reported. 16 Briefly, all participants were implanted with the VNS device (Implantable Pulse Generator, Model 1001, MicroTransponder Inc, Austin, TX) and randomized to either Active VNS (VNS stimulation paired with an intense rehabilitation program) or Control VNS (intense rehabilitation with sham VNS). Therapists, participants, and assessors were blinded to the group assignment. After participants in both groups underwent 6 weeks of in-clinic rehabilitation therapy, the treating therapist designed and initiated an individualized home exercise program to address each individual’s hand and arm impairments. Each session lasted approximately 2 hours. The home exercise program paired with VNS was designed to not only be an extension of in-clinic therapy, but was also customized by the treating physical or occupational therapist to support and advance the participants’ functional movement goals. The therapist gave each participant detailed instructions on how to perform the home exercise program and how to self-administer VNS via a magnet swipe at the start of each session. The magnet swipe triggered the implanted device to deliver cyclical VNS every 10 s for 30 minutes. For the first 90 days, the rehabilitation exercises were paired with either Active VNS and/or Control VNS, according to the group assignment. 16 Thereafter, the Control group (henceforth referred to as Cross-VNS group) crossed over to receive 6 weeks of in-clinic Active VNS paired with rehabilitation, followed by the home exercise program, but now with the Active VNS paired with the rehabilitation tasks. The group initially randomized to Active VNS continued to perform home exercises paired with Active VNS (Supplemental Figure 1). Participants were regularly followed up about their home programs during phone calls with the treating therapist (at least once per month) and when they were seen at scheduled assessment visits.
A device log file was downloaded from each participant’s VNS device at assessment visits to gather device and usage data. The log file included the number of magnet swipes performed by the participant, thus, giving an indication of the number of home exercise sessions performed. We report the number of magnet swipes as a measure of the number of home exercise sessions. Adherence was expressed as a percentage of device use per number of days for the entire 1 year follow-up period and quarterly adherence (for each 3-month period prior to assessment date). Quantifying quarterly adherence in addition to overall adherence to the home therapy program has the added benefit of detecting epochs of high or low device use. We also report adherence and the total number of hours each participant spent doing home exercises over the 1-year follow-up period.
Outcome assessments for this report were collected at 6 months, 9 months, and 1 year post–in-clinic Active VNS therapy (and at the same time after cross-over for the Control VNS group). Shorter-term outcomes have been previously reported. 16 Outcome measures included FMA-UE, 17 Wolf Motor Function Test (WMFT; Functional and Time), 18 Box and Block Test, 19 Nine-Hole Peg Test, 20 Stroke Impact Scale (SIS), 21 and Motor Activity Log (MAL). 22 These long-term outcome measures were not assessed in a blinded fashion. We defined a ≥6-point improvement on the FMA-UE score as a clinically meaningful improvement. 23 All adverse events and serious adverse events occurring between day 90 and 1 year post–in-clinic therapy completion are reported here. Adverse events occurring prior to day 90 were previously reported. 16
Statistical Analysis
Safety, adherence to the home exercise program, and clinical outcome measures are reported descriptively. For adherence and clinical outcome measures, we report change from baseline as mean and SD, unless noted otherwise. Although differences between groups were not anticipated at 1 year, we assessed this by comparing the time spent doing home exercises and change in FMA-UE score at 6, 9, and 12 months between the Active and Cross-VNS groups, using an independent 2-sample t-test. Because no differences were observed (Supplemental Tables 1 and 2), data from both groups (n = 15) were combined into a pooled analysis of the long-term changes using a paired-sample t-test (2-tailed level of significance was set at P < .05). For all participants, change was calculated as the difference between the original baseline measure (ie, prior to starting the first in-clinic session for both groups) and each of the follow-up visits. We also report data separately for Active and Cross-VNS participants.
Results
All participant demographics have been previously reported. 16 The 6-, 9-, and 12-month outcomes were available for 15 of 17 participants (Active VNS: 8/8; Cross-VNS: 7/9). Mean age was 61 ± 9.6 years (Active VNS: 59.5 ± 7.3 years, 4 male participants; Cross-VNS: 63 ± 12.0 years, 4 male participants; P = .50). The time from stroke was 20.4 months ranging from 10.3 to 53 months (Active VNS: 18.43 ± 8.1; Cross-VNS: 22.7 ± 15.6; P = .50). Baseline FMA-UE was 31 ± 6.8 (Active VNS: 29.3 ± 7.5; Cross-VNS: 32.8 ± 6.0; P = .34). One participant felt that they had improved motor function sufficiently during the randomized portion of the study and, therefore, withdrew from the study after 4.5 months. Another participant withdrew after learning that they had been randomized to the Control VNS group. The participant did not wish to attend further visits because of extended travel time, and the device was explanted. Home therapy adherence data were not available from 1 additional participant on the Cross-VNS group who had improved to such a degree that they did not want to attend further sessions or complete home exercises. No other participant was explanted during the first year of the VNS home program.
Safety
A serious adverse event unrelated to VNS therapy occurred in 1 participant (spinal pathology). This participant also had shortness of breath resulting from a previously diagnosed paradoxical vocal cord motion, unrelated to VNS, which resolved with speech therapy. There were no VNS therapy–related serious adverse events post–90 days through 1 year of treatment. Adverse events probably related to stimulation included hiccups in 1 participant. Hoarseness, one of the most common adverse effect of VNS, 24 was not reported by any participant during the home program. All adverse events are listed in Supplementary Table 3.
Adherence to Home Exercise Program
On average, participants performed 200 ± 63 home therapy sessions, which represent device use on 57.4% of home exercise days available for each participant (typically 1 year, 349.9 ± 10.8 days; Supplemental Table 4). Participants spent 2 ± 0.6 h/wk doing the home therapy program. The time spent doing the home therapy program for individual participants is reported in Supplemental Tables 4 and 5.
Efficacy
In the 1-year follow-up of VNS paired with rehabilitation, participants demonstrated significant improvement from baseline in multiple functional assessment measures (Table 1). The pooled FMA-UE had improved by 9.2 ± 8.2 points at 1 year (CI = 4.7 to 13.7, P = .001; Active VNS 10.8 ± 7.6 and Cross-VNS 7.2 ± 8.9 points; Figure 1 and Table 1). In total, 73% of participants (11/15) demonstrated a clinically meaningful improvement (≥6 points) in FMA-UE at 1 year. Participants also demonstrated significant improvement from baseline at 1 year (Table 1) on the WMFT (Functional: mean, 0.3; CI: 0.1-0.6; P = .003), SIS Hand Function (mean: 17.7; CI: 6.5-28.8; P = .004), and MAL (mean: 0.8; 95% CI: 0.3-1.3; P = .005). The Box and Block and Nine-Hole Peg Tests did not show improvements from baseline at 1 year. Outcome measures by initial randomization group are shown in Supplemental Table 6 and Supplemental Figure 2. There was no suggestion of a difference in outcomes measures between 6, 9, and 12 months (Supplemental Table 7).
Long-Term Change in Outcome Measures.
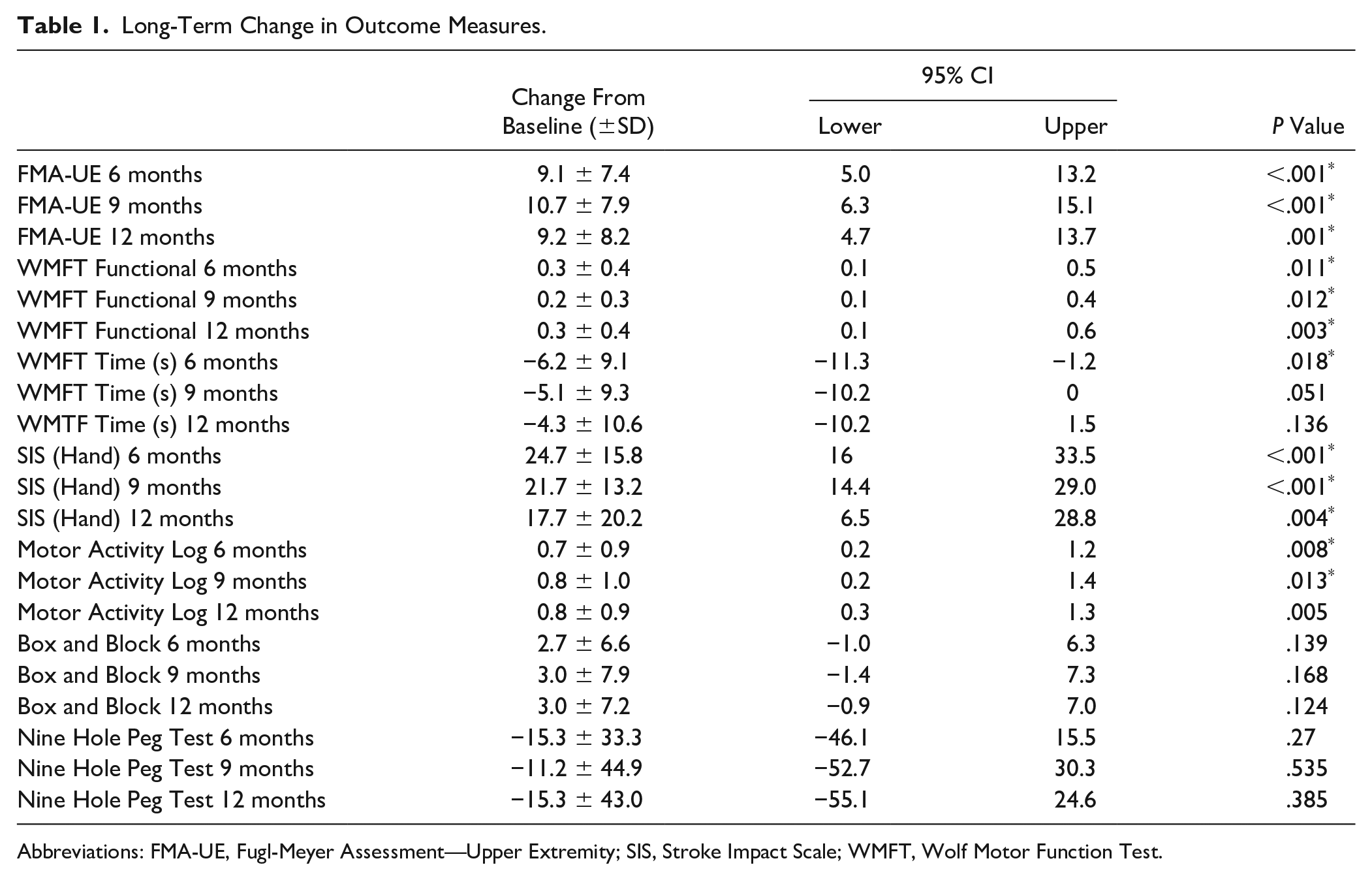
Abbreviations: FMA-UE, Fugl-Meyer Assessment—Upper Extremity; SIS, Stroke Impact Scale; WMFT, Wolf Motor Function Test.
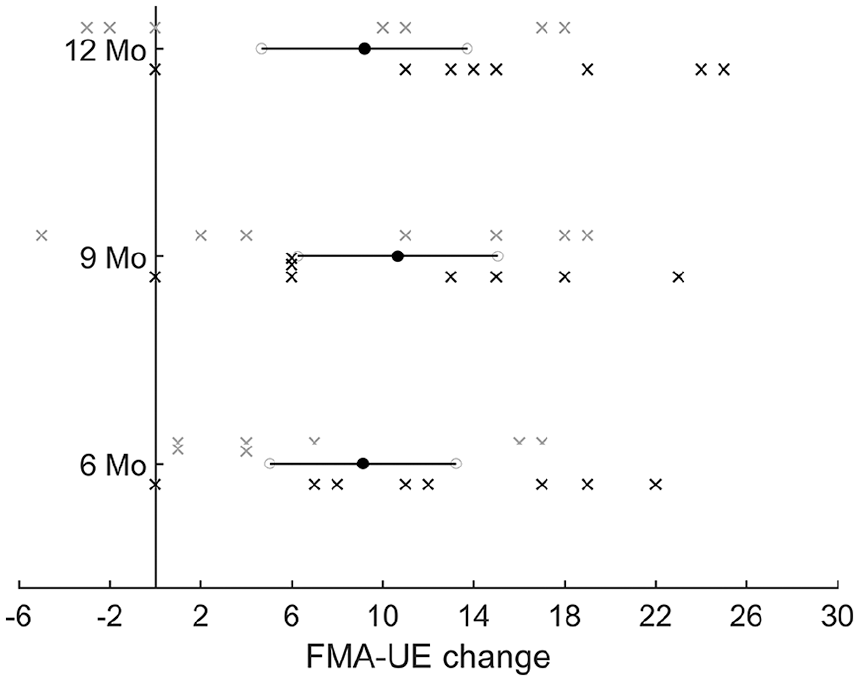
Long-term changes in FMA-UE on the home VNS program. Mean FMA-UE improvement (black circles) at 6 (9.1 ± 7.4 points), 9 (10.7 ± 7.9 points), and 12 (9.2 ± 8.2 points) months for participants in both groups (n = 15) doing the home VNS program. Horizontal lines denote confidence intervals. Black and gray crosses denote individual participants in the Active and Cross-VNS group, respectively. All participants did the VNS home exercise program during this long-term phase, and differences between groups was not significant at any of the time points.
Discussion
We found that people with moderate to severe chronic ischemic stroke treated with rehabilitation paired with VNS had a sustained improvement in upper-limb impairment and functional measures at 1 year. The improvement following a period of intense, in-clinic, paired VNS rehabilitation 16 was largely maintained or improved by participation in an individualized home exercise program with self-administered VNS. Given these significant and clinically meaningful changes, we conclude that self-administered VNS coupled with a long-term home exercise program may be beneficial for individuals with moderate to severe weakness after chronic ischemic stroke and warrants further study. These findings are based on unblinded follow-up and a small sample, so definitive conclusions cannot be drawn.
The home-based activities were safe, with only mild adverse events related to their performance. For most participants who regularly performed the home therapy program, motor function generally continued to improve over time. An earlier study investigating VNS paired with rehabilitation but without a continued structured home exercise program demonstrated that the initial gains obtained after 6 weeks of in-clinic therapy were not maintained long-term (data not reported). In our study, we did not see changes in upper-limb measures between 6, 9, and 12 months. However, this result may be influenced by a small number of participants who developed comorbidities and could not continue therapy in the longer term. Thus, overall, our data support long-term use of the device for at least 1 year, but this needs to be confirmed in a larger study.
There are limitations to our analysis. It is important to note that this analysis is based on unblinded data; because of the control group cross-over, we lack a comparator group for longer-term therapy, and control participants received additional therapy ahead of their Active VNS. This and the small sample size limit the conclusions we can draw. Since both groups continued to receive Active VNS in the long-term, it was not possible to differentiate the potential benefit of a home exercise program without VNS versus with VNS. Additionally, we cannot distinguish the relative impact of regular phone call monitoring by the therapist, but it is unlikely that this alone will be effective. The small decline in some measures from 9 to 12 months suggests an opportunity to reinforce home therapy assignments in the late phase of a 12-month, home-based rehabilitation program.
Participants in our study were enrolled many months after stroke, and evidence for effectiveness of interventions for upper-limb recovery in this period is limited. For example, in a randomized controlled trial of robotic therapy, FMA scores increased by 3.9 points with robotic therapy and by 4.01 points in the intensive comparator groups. 25 Other studies report similar or smaller gains,26-28 and this is consistent with the improvement we saw in participants receiving control therapy in our pilot studies.15,16 Furthermore, in the recent RATULS trial, subgroup analysis showed no suggestion of benefit of either robotic therapy or an enhanced upper-limb therapy intervention in participants who were enrolled after 12 months. 29 Thus, although indirect comparisons between trials are flawed, we are encouraged that we see sustained responses of a greater magnitude than those reported in other trials.
These results also reinforce the fact that it is feasible to do Active VNS therapy at home. Each participant chose to do the exercises at a time that was convenient to them and without direct supervision of a trained physical or occupational therapist. Participants readily and consistently were able to swipe a magnet once over the device to initiate VNS and then perform the prescribed exercises during a 30-minute session. Home exercises included tasks that were targeted to the participants’ impairment and relevant to the participant (eg, grasp and release practice of household objects, washing dishes, doing laundry). Thus, the participant could do therapeutic tasks at home, avoiding the time demands, transportation complexities, and added expense of multiple in-clinic visits. The high adherence to the intervention seen in the study is similar to that reported in recent studies of home telebased rehabilitation 30 and supports the notion that high adherence to engaging and goal-orientated upper-limb rehabilitation at home is possible. It is difficult to compare our adherence results with that in other types of interventions because patient self-report overestimates the actual performance. 31
For the in-clinic portion of the study, participants received between 300 and 500 discrete stimulations to the vagus nerve per visit paired with a task-specific movement. 16 This intensity of repetition is thought to be associated with the upregulation of biomarkers associated with neuroplastic changes32,33 and important for translation, and was similar to the number of repetitions in the animal VNS studies. 34 For the home-based program, the study did not utilize a motion tracking system (eg, an accelerometer or home-based motion sensing system) to analyze arm movements during home exercises. Therefore, we were unable to determine the precise amount of repetition or intensity with which the home program was performed. As reported, we were able to determine the amount of time each participant spent doing the home program based on the magnet swipe and self-reporting. Because the duration of each self-administered VNS session of the home program was 30 minutes, versus approximately 90 minutes in the in-clinic sessions, the number of repetitions per session is assumed to be far fewer. Although participants were encouraged to do the home program daily, our realistic expectation was that participants would do the exercises about 50% of the time, which is what was observed. This level of compliance was sufficient for the majority of participants to maintain or continue to show improvements.
The relatively shorter sessions of the home program, in general, were associated with retention of gains from in-clinic therapy. One possibility is that the in-clinic portion of the intervention may have caused neuroplastic changes in the brain upon which less practice was required to maintain or improve function. This finding is consistent with the previous report in which motor improvements after paired VNS were greater at 3 months posttherapy compared with improvements immediately after in-clinic VNS therapy. 16 Another possibility is that the extended period of the home program (several months) facilitated motor improvement, despite its shorter session duration. Participants did 18 sessions (27 hours) of in-clinic VNS therapy compared with more than 200 sessions during the home program. It is well known that the amount, intensity, and duration of neurorehabilitation after brain injury are important contributors to effective motor learning.30,35 It is clear, however, from recent research findings that optimizing long-term recovery is complex, and simply increasing the volume of therapy is unlikely to maximize recovery. 29 A paired intervention of rehabilitation with a device that facilitates neuroplastic change may be needed to optimize recovery. In future studies, it will be important to consider patient selection to maximize the chances of success and response to therapy. We recently completed an analysis of predictors of response to VNS treatment. Baseline FMA score, volume of normally appearing white matter, and systolic blood pressure were independently associated with response to VNS. 36 Whether it is possible to select people for VNS-based rehabilitation therapy using clinical and imaging variables will be evaluated in the ongoing phase 3 study.
In summary, this study demonstrated that home-based VNS combined with rehabilitation therapy is feasible and safe. High-intensity in-clinic rehabilitation paired with VNS may improve upper-limb motor outcomes in individuals with chronic ischemic stroke that are maintained by long-term home exercises. It warrants further investigation. An ongoing pivotal clinical trial (MT-St-03) with 108 implanted participants is expected to be completed this year with results expected in 2020. 37
Supplemental Material
Supplementary_File1_NE – Supplemental material for Vagus Nerve Stimulation Paired With Upper-Limb Rehabilitation After Stroke: One-Year Follow-up
Supplemental material, Supplementary_File1_NE for Vagus Nerve Stimulation Paired With Upper-Limb Rehabilitation After Stroke: One-Year Follow-up by Jesse Dawson, Navzer D. Engineer, Cecília N. Prudente, David Pierce, Gerard Francisco, Nuray Yozbatiran, W. Brent Tarver, Reema Casavant, Danielle K. Kline, Steven C. Cramer, Ann Van de Winckel and Teresa J. Kimberley in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors sincerely thank the participants, research nurses, and coordinators who contributed to the study. The authors sincerely thank the following people for their vital role in this project: Teresa Bisson DPT, oversight and assessor; Kate Frost, PhD, assessor; Charlotte Quinton, RN, BSN, study coordinator and research nurse; Kathryn Nedley, OT, assessor; Pamela MacKenzie and Ozzy Dincarsalan, trials managers; Jen Alexander, physiotherapist; Elizabeth Colquhoun, RN, research nurse; and Diana Hansen, data validation.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website at website along with the online version of this article.
Authors’ Note
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jesse Dawson has received reimbursement for expenses from MicroTransponder, Inc, for conference attendance to present results from this study. Steven C. Cramer has served as a consultant for Abbvie, Constant Therapeutics, MicroTransponder, Neurolutions, Regenera, SanBio, Stemedica, Fujifilm Toyama Chemical Co, Biogen, and TRCare. Cecília N. Prudente, David Pierce, Navzer Engineer, Reema Casavant, and W. Brent Tarver are employees of MicroTransponder, Inc.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The trial was funded by MicroTransponder Inc. The funders had no responsibility in the acquisition and analysis of the data, nor in the decision to submit the results for publication.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
