Abstract
Background. Autologous nerve graft is the most common clinical intervention for repairing a nerve gap. However, its regenerative capacity is decreased in part because, unlike a primary repair, the regenerating axons must traverse 2 repair sites. Means to promote nerve regeneration across a graft are needed. Postoperative electrical stimulation (PES) improves nerve growth by reducing staggered regeneration at the coaptation site whereas conditioning electrical stimulation (CES) accelerates axon extension. In this study, we directly compared these electrical stimulation paradigms in a model of nerve autograft repair. Methods. To lay the foundation for clinical translation, regeneration and reinnervation outcomes of CES and PES in a 5-mm nerve autograft model were compared. Sprague-Dawley rats were divided into: (a) CES, (b) PES, and (c) no stimulation cohorts. CES was delivered 1 week prior to nerve cut/coaptation, and PES was delivered immediately following coaptation. Length of nerve regeneration (n = 6/cohort), and behavioral testing (n = 16/cohort) were performed at 14 days and 6 to 14 weeks post-coaptation, respectively. Results. CES treated axons extended 5.9 ± 0.2 mm, significantly longer than PES (3.8 ± 0.2 mm), or no stimulation (2.5 ± 0.2 mm) (P < .01). Compared with PES animals, the CES animals had significantly improved sensory recovery (von Frey filament testing, intraepidermal nerve fiber reinnervation) (P < .001) and motor reinnervation (horizontal ladder, gait analysis, nerve conduction studies, neuromuscular junction analysis) (P < .01). Conclusion. CES resulted in faster regeneration through the nerve graft and improved sensorimotor recovery compared to all other cohorts. It is a promising treatment to improve outcomes in patients undergoing nerve autograft repair.
Introduction
Outcomes following peripheral nerve injury are strongly influenced by the time required for regenerating motor and sensory axons to reach their target tissue. Functional outcomes can be improved by accelerating the intrinsic rate of nerve regeneration. Although a conditioning crush lesion (CCL) delivered to a nerve 1 week prior to transection and nerve repair has been shown to be capable of markedly accelerating nerve regrowth, it cannot be translated to the clinic due to its injurious nature. 1 Recently, however, we demonstrated that 1 hour of conditioning electrical stimulation (CES) delivered to a nerve 7 days prior to injury and repair induces a conditioning effect comparable to a CCL, but in a noninjurious, thus clinically feasible manner.2-4
While the use of CES is novel, postoperative electrical stimulation (PES) is well described to improve outcomes in animal and human models of nerve injury, and it is the only adjunct to nerve repair that has been successfully translated to the clinic.5,6 However, unlike a conditioning lesion (CCL or CES), PES does not accelerate axon extension, but rather enhances regeneration of axons as they cross the site of surgical coaptation, a process called “staggered regeneration.” 7 As both PES and CES are clinically feasible modalities, a direct comparison of outcomes is of clinical importance.
To inform future clinical trials, the goal of this study was to compare the lengths of axon extension and sensorimotor behavioral outcomes of CES and PES in an animal model of nerve autograft which is the most commonly used method to repair a nerve gap.8,9 Nerve grafts are commonly used following oncological resection, brachial plexus repair, facial reanimation, and chronic injury repair. 10 Since these surgeries are elective, it is feasible to condition one week prior. Graft repair requires the regenerating nerves to extend past 2 coaptation sites, which is more challenging than a primary nerve repair (cut/coaptation) surgery.
We hypothesize that CES is more efficacious than PES in enhancing the rate of nerve regeneration and return of motor and sensory function after autograft nerve repair.
Methods
Animals
Adult male Sprague-Dawley rats weighing 200 g (Charles River Laboratory) were placed under the care of Health Sciences Laboratory Animal Services (HSLAS) at the University of Alberta. We divided 64 animals into 3 separate cohorts: CES prior to an autograft repair, PES immediately following an autograft repair, and the negative control in which there was no electrical stimulation (no-ES) intervention before or after the autograft repair (Figure 1A). Length of nerve regeneration through a graft was measured 14 days following coaptation in 18 animals (n = 6/cohort). Behavioral testing of 48 animals was performed between 6 and 14 weeks and animals were culled 10 or 14 weeks after the autograft repair surgery (n = 8/cohort for each timepoint). All surgical procedures were approved by the University of Alberta Animal Research Ethics Board.
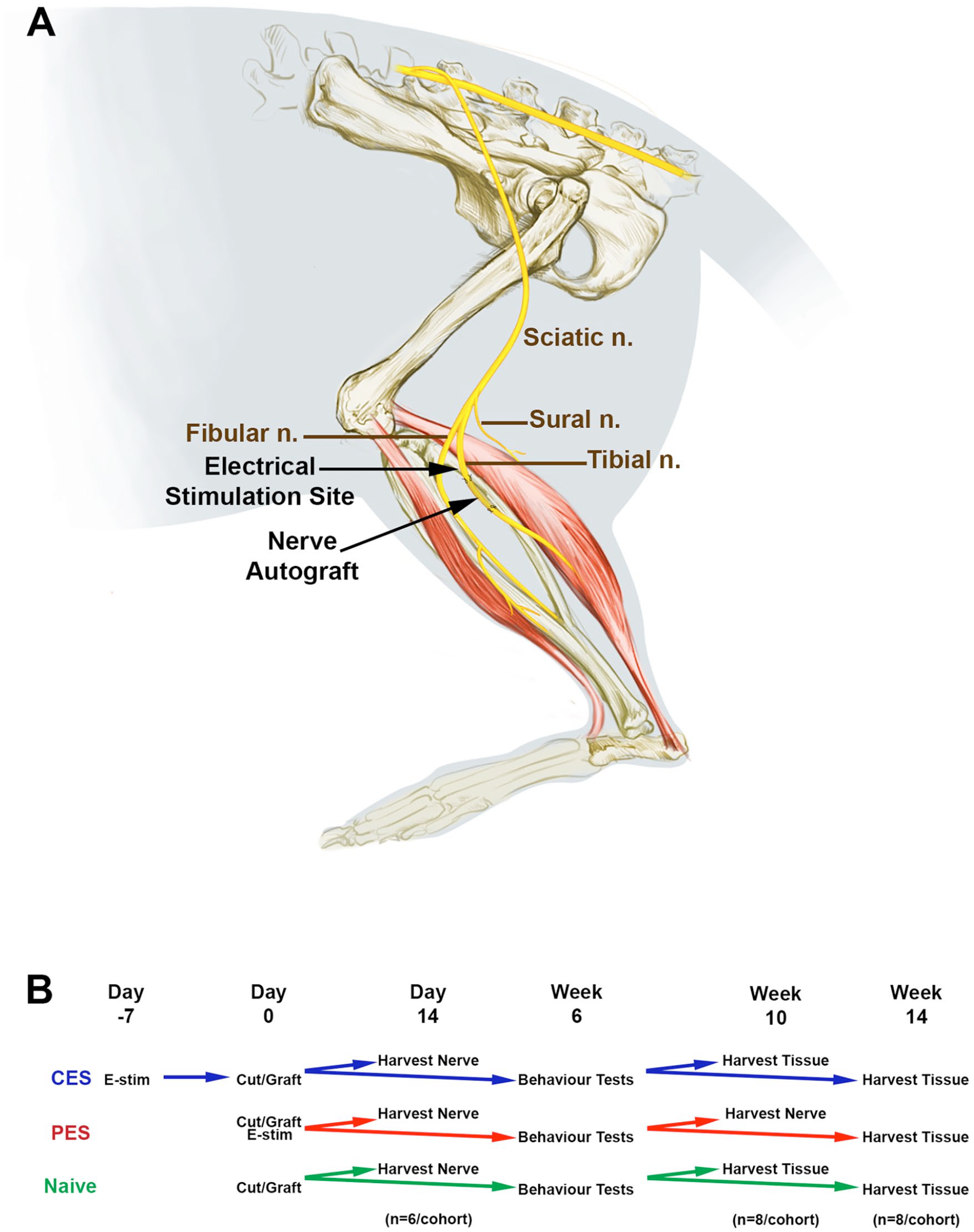
Experimental design of surgical treatment and experimental paradigms. (A) Artistic illustration of the investigation to describe the nerve autograft repair surgeries. Illustration of the sciatic nerve and its trifurcation into the fibular (common peroneal), sural and tibial (used in autograft repair) nerves. The site of electrical stimulation (conditioning electrical stimulation [CES] or postoperative electrical stimulation [PES]) is shown. (B) Animals were divided into three cohorts, CES, PES, and no-ES. Two weeks following nerve repair of animals +/- CES, PES treatment, 24 animals were harvested to determine the extent of nerve regeneration (n = 6/cohort). The remaining animals underwent behavioral tests and were euthanized at 10 and 14 weeks (n = 8/cohort).
Prior to surgery, animals were anesthetized with inhaled isoflurane (2% titrated at 1-2 L/min for maintenance of a surgical anesthetic plane) and received 0.01 mg/kg of buprenorphine subcutaneously. All surgical procedures were performed under 3.5× loupe magnification. At the completion of all procedures, the hamstring muscles were resuspended and the skin was closed with 4-0 Vicryl (Ethicon Inc, Somerville, NJ).
Conditioning Surgery
CES was performed as previously described.2,3 Briefly, a longitudinal incision was made over the lateral aspect of the right lower limb. Within the posterior compartment of the leg, the tibial nerve was identified as it emerged between the 2 heads of the gastrocnemius muscle. The bared end of a stainless-steel wire (cathode, Cooner Wire AS 632) was wrapped around the nerve, and another wire (anode) was placed into the tibialis anterior muscle; these were connected to a SD-9 stimulator (Grass Instruments Co, Quincy, MA). Continuous electrical stimulation was delivered at 20 Hz with a 0.1-ms duration over a 1-hour time span, with voltage titrated to maintain a visible foot twitch (approximately 100 μs; 3 V).
Nerve Autograft Repair
An incision was made along the lateral femur and the hamstring muscles were dissected to identify the sciatic nerve, which was traced distally to isolate the tibial nerve. One cm distal to the site of trifurcation, a 5-mm segment of the tibial nerve was excised. The nerve segment was reversed 180°, and the proximal and distal cut sites were coapted with 9-0 silk suture. The hamstring muscles were resuspended with 4-0 Vicryl and the skin was repaired with a 2-layer closure.
Postoperative Electrical Stimulation
Immediately following nerve grafting, a stainless-steel wire (cathode) bared of insulation was placed proximal to the site of repair, and the anodal wire was buried in the quadriceps muscle. PES was performed using the same parameters as the conditioning surgery (1 hour of continuous stimulation at 20 Hz).
Analyses
The evaluators of all histological, electrophysiological, and behavioral outcomes were blinded to treatment assignments.
Tissue Analysis
Animals treated with an autograft repair were euthanized either 14 days following nerve repair (nerve regeneration cohort) or on completion of behavioral testing at 10 or 14 weeks postgrafting (functional outcome cohorts). Euthanization was accomplished by carbon dioxide asphyxiation followed by exsanguination through a puncture to the left cardiac ventricle.
Tibial nerves were harvested as previously described, 3 with fixation in Zamboni’s fixative (paraformaldehyde, picric acid, NaOH; American MasterTech Scientific, Lodi, CA) for 4 hours, rinsed with 0.01 M phosphate buffered saline (PBS) (ThermoFisher Scientific, Waltham, MA) 5 times, postfixed in 30% sucrose solution overnight at 4°C, and frozen in Optimum Cutting Temperature (OCT) (Sakura Finetek, Torrance, CA) with indirect exposure to liquid nitrogen. Tissue chucks were cut into 12-μm sections, thaw-mounted on Superfrost Plus microscope slides, and stored at −80°C until processing.
Fourteen weeks post nerve repair, the gastrocnemius muscles were harvested from both legs in their entirety and weighed for comparative purposes. The weight of the injured muscle, a function of muscle atrophy, was reported as a percentage of the uninjured contralateral muscle to control for discrepancies in animal size. Ipsilateral gastrocnemius muscle was fixed in Zamboni’s solution overnight, rinsed 5 times in PBS, sunk in 30% sucrose, and flash-frozen in OCT.
Plantar footpads were collected 14 weeks post nerve repair using a 3-mm biopsy punch (Acuderm Inc, Fort Lauderdale, FL) and fixed in 2% paraformaldehyde-lysine-periodate (PLP) fixative overnight.3,11 Tissue sections were rinsed 5 times in Sorenson’s phosphate butter, cryoprotected overnight in 20% glycerol/0.1 M Sorensen’s phosphate buffer, frozen in OCT, and cut into 20-μm sections.
Immunofluorescence
After slides were warmed to room temperature, antigen retrieval was performed for 20 minutes in 60°C citrate buffer (10 mM sodium citrate, 0.05% Tween-20, pH 6.0; ThermoFisher Scientific, Waltham, MA). After the slides cooled, they were washed 3 times in 0.01 M PBS for 5 minutes/wash, and permeabilized for 10 minutes with 0.1% TritonX-100 (ThermoFisher Scientific, Waltham, MA). Tissue sections were blocked for 90 minutes in 10% normal goat serum (MP Biomedicals, Santa Ana, CA) and 3% bovine serum albumin (BSA) (Sigma-Aldrich, St Louis, MO) in 0.01 M PBS. Primary antibodies diluted in 0.01 M PBS with 3% BSA were left on tissue overnight at 4°C. Nerve sections were stained with mouse anti-neurofilament-200 (NF200; 1:500) (Sigma-Aldrich, St Louis, MO). Muscle sections fixed in Zamboni’s were labeled with mouse anti-NF200 (1:500) to stain the innervating nerve, then with conjugated anti-α-bungarotoxin (1:1000, 20 minutes). Footpads were stained with anti-protein gene product 9.5 (PGP9.5, 1:1000) for identification of intraepidermal nerve fibers overnight at 4°C. Secondary antibodies included Cy3-conjugated goat anti-mouse (Sigma-Aldrich), Alexa Fluor 488-conjugated goat anti-rabbit IgG (Invitrogen, Carlsbad, CA), and donkey-anti-chicken 594 (ThermoFisher Scientific). Dorsal root ganglion tissues were stained with nuclear stain NucBlue (ThermoFisher Scientific). All slides were mounted with a coverslip using 50% glycerol in 0.01 M PBS.
Morphological identification of regenerating NF-200 axons was examined to measure (a) the length of axonal extension and (b) the number of regenerating axons at 250-μm intervals from the site of repair to the most distal point of regeneration (minimum of 10 axons must be observed at that distance in order to continue measuring the next distance interval). The operator was blinded to the treatment condition for all tissue analyses and quantification.
Muscle sections and footpads were evaluated using confocal microscopy. The number of innervated neuromuscular junctions in sections of the gastrocnemius muscle was determined, with NF-200 labeling the axons and α-bungarotoxin the acetylcholine receptors. The number of neuromuscular junctions present in each standardized size of tissue was quantified; 6 sections were analyzed per animal. The tibial-nerve innervated footpads were evaluated to identify PGP9.5-labeled axons crossing the dermal-epidermal junction (3 sections were evaluated per animal). A z-stack of 1-µm steps was obtained for each field and intraepidermal nerve fiber (IENF) density was determined by counting the number of axons crossing per 1 mm of dermal-epidermal junction.
Behavioral Outcomes
Sensory and motor behavior testing was performed weekly between 8 and 14 weeks following nerve grafting, by the same examiner blinded to the test group of the animals. Calibrated von Frey monofilaments (1.4-15 g) were used to evaluate sensory reinnervation to the plantar footpad. 9 Three consecutive paw withdrawals to an individual monofilament constituted a positive response.
Motor outcomes included gait analysis for toe spread, horizontal ladder test for motor dexterity, and nerve conduction studies for compound muscle action potentials (CMAPs). 9 Gait analysis to evaluate intrinsic foot muscle reinnervation was performed by placing the animal into a specially designed walkway constructed from transparent Plexiglas with an adjustable mirror underneath to visualize the plantar paw. Animals walked the length of the track 3-times/session while being filmed. The video was analyzed and individual screenshots were taken of each weightbearing hind paw where all toes were visible. Ten images of both the affected and contralateral control footpads were analyzed per testing session; the distance between the first and last toes was measured. Results were reported as the percentage of toe spread of the injured side compared to the uninjured contralateral control.
The horizontal ladder test consisted of a 48 cm elevated horizontal ladder composed of clear plexiglass with metal ladder rungs arranged in a different pattern each testing session to prevent learning. The animals were filmed over three attempts to cross the ladder at each testing session, and videos were analyzed on a frame-by-frame basis. Using the 0-6 scoring system developed by Metz and Whishaw, 12 a score was given for each individual foot placement on the ladder rung, with 0 being a complete slip, 1 included a deep slip, 2 included a slight slip, 3 included foot replacement after a slip, 4 included a foot correction, 5 included a partially successful foot placement, and 6 included correct placement. The average score of the injured limb was calculated for each attempt.
Nerve conduction studies were performed at 10, 12, and 14 weeks following nerve autograft repair surgery. Animals were anesthetized with inhaled isoflurane and the lower limbs were shaved for lead placement. Recording leads were placed in the plantar footpad, and supramaximal electrical stimulation was delivered at the knee. Maximal CMAP with the sharpest rise time was recorded. Results were reported as the CMAP percentage of the injured side compared to the uninjured contralateral control.
Statistical Analysis
Results are presented as the mean ± standard error of the mean (SEM). Groups were compared using a 1-way analysis of variance (ANOVA) to identify differences in the mean between groups, followed by Bonferroni post hoc analysis when a significant difference was found. Statistical significance was accepted with a level of P < .05. All statistical analyses were performed using STATA 14 (StataCorp LP, College Station, TX).
Results
CES Improves Regeneration Outcomes in a Nerve Autograft Model
Length of regeneration 2 weeks following tibial nerve autograft repair surgery was compared between animals treated with CES, PES, and a no-ES negative control. Each tibial nerve and its graft were harvested, and following cryostat sectioning, the tissues were stained with NF-200 immunofluorescence to assess the extent of axon regeneration. Animals treated with CES prior to grafting had a significantly longer length of axon extension at 2 weeks of regeneration (5.9 ± 0.2 mm) when compared with animals that received PES after grafting (3.8 ± 0.2 mm, P < .01) or animals that received no-ES (2.5 ± 0.2 mm, P < .001). Length of axon extension in animals treated with PES was significantly longer than negative control (P < .001) (Figure 2). There were significantly more axons in CES animals compared with PES or no-ES at all 1-mm intervals measured (P < .001) distal to the first coaptation site, whereas the PES cohort had more axons than no-ES at each interval (P < .001). Our previous studies showed sham-ES and no-ES were equivalent in all analyses and therefore a sham-ES treatment group was not included in our study.2,3
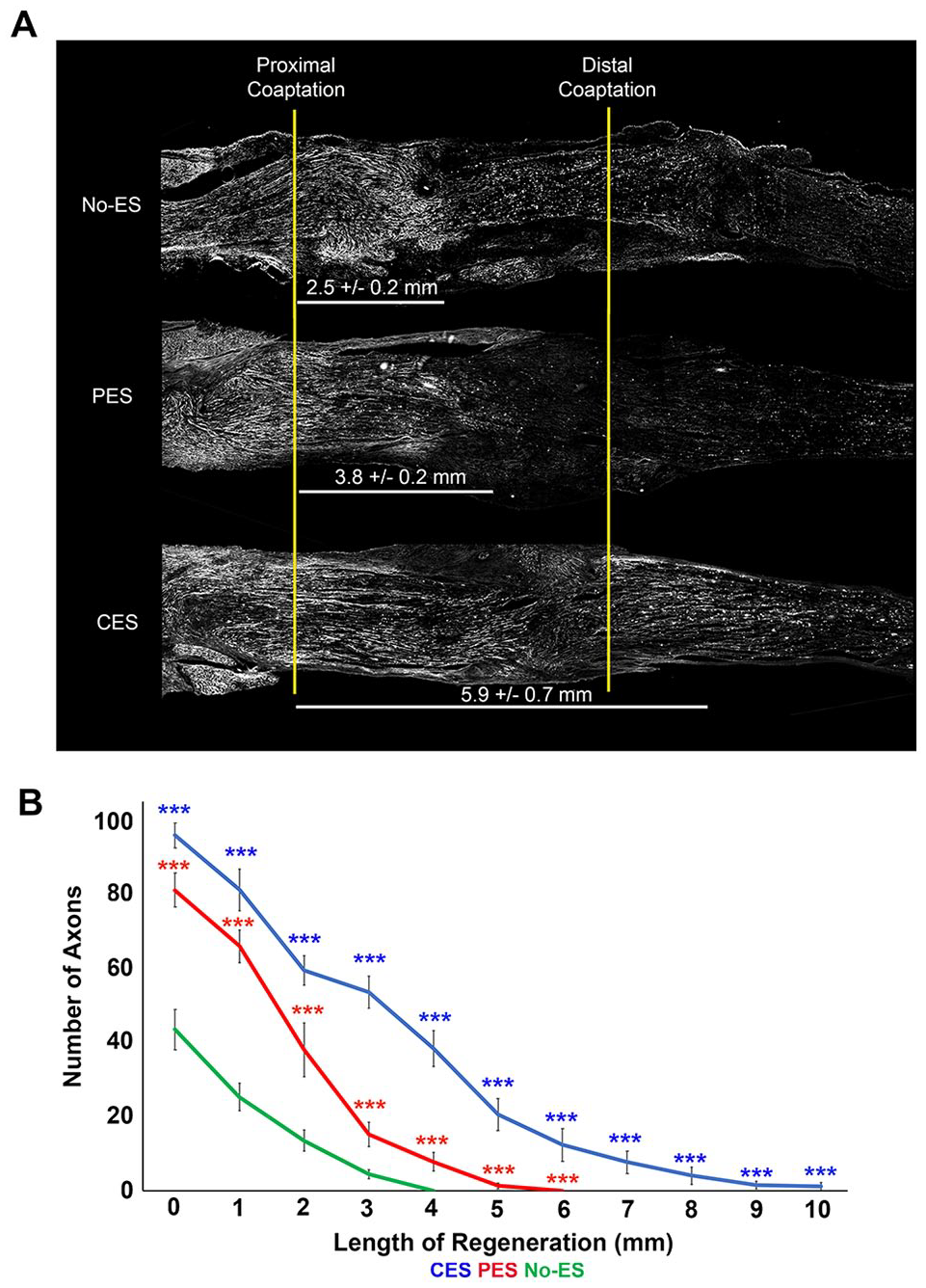
Conditioning electrical stimulation (CES) improves regeneration beyond postoperative electrical stimulation (PES) in a 0.5-cm nerve autograft reconstruction. (A) Representative photomicrographs of NF-200 labeled longitudinal sections of grafted tibial nerve at 14 days of regeneration. Yellow lines depict the proximal and distal coaptation sites abutting the interposed 0.5 cm autograft. Although PES-treated nerves regenerated farther than the negative controls (**P < .01), nerves treated with CES 1 week prior to grafting had significantly longer axon extension compared with animals treated with either PES (*P < .05) or the no-ES controls (**P < .01). (B) At each 1 mm distance distal to the first coaptation site shows there were significantly more axons in the CES (blue line) than PES (red line) or no-ES (green line) (***P < .001). The PES cohort had significantly more axons at each interval counted compared with the no-ES animals (***P < .001).
CES Improves Sensory Reinnervation Outcomes in a Nerve Autograft Model
Sensory outcomes included von Frey filament testing and IENF density counts. Weekly von Frey monofilament testing between 6 and 14 weeks of regeneration revealed significantly greater sensory recovery at all time points in animals treated with CES when compared with the PES and the no-ES cohorts. At 6 weeks of regeneration, animals treated with CES responded to 7.8 ± 0.5 g of mechanical stimulation, which was significantly less than PES (19.3 ± 2.6 g, P < .001) or no-ES (22.1 ± 2.5 g, P < .001). This trend persisted, and animals treated with CES had significantly improved sensory recovery compared to PES and no-ES at 7 (5.5 ± 0.6, 10.6 ± 1.1, 12.4 ± 2.4; P < .001), 8 (5.8 ± 0.4, 11.3 ± 0.7, 13.1 ± 0.4; P < .001), 9 (5.25 ± 0.3, 9.3 ± 0.4, 10.1 ± 0.9; P < .001), 10 (4.25 ± 1.0, 9.3 ± 0.6, 9.6 ± 1.1; P < .001), 11 (5.25 ± 0.5, 11.9 ± 0.9, 12.1 ± 1.0; P < .001), 12 (4.9 ± 0.5, 8.0 ± 0.2; 12.5 ± 1.1, P < .001), and 13 (4.75 ± 0.7, 8.2 ± 0.4, 9.7 ± 0.3; P < .001) weeks of regeneration (Figure 3A). Although PES was significantly improved from the no-ES controls at 12 and 13 weeks of regeneration (P < .05), they did not compare to the increased regenerative potential of the CES treated animals.
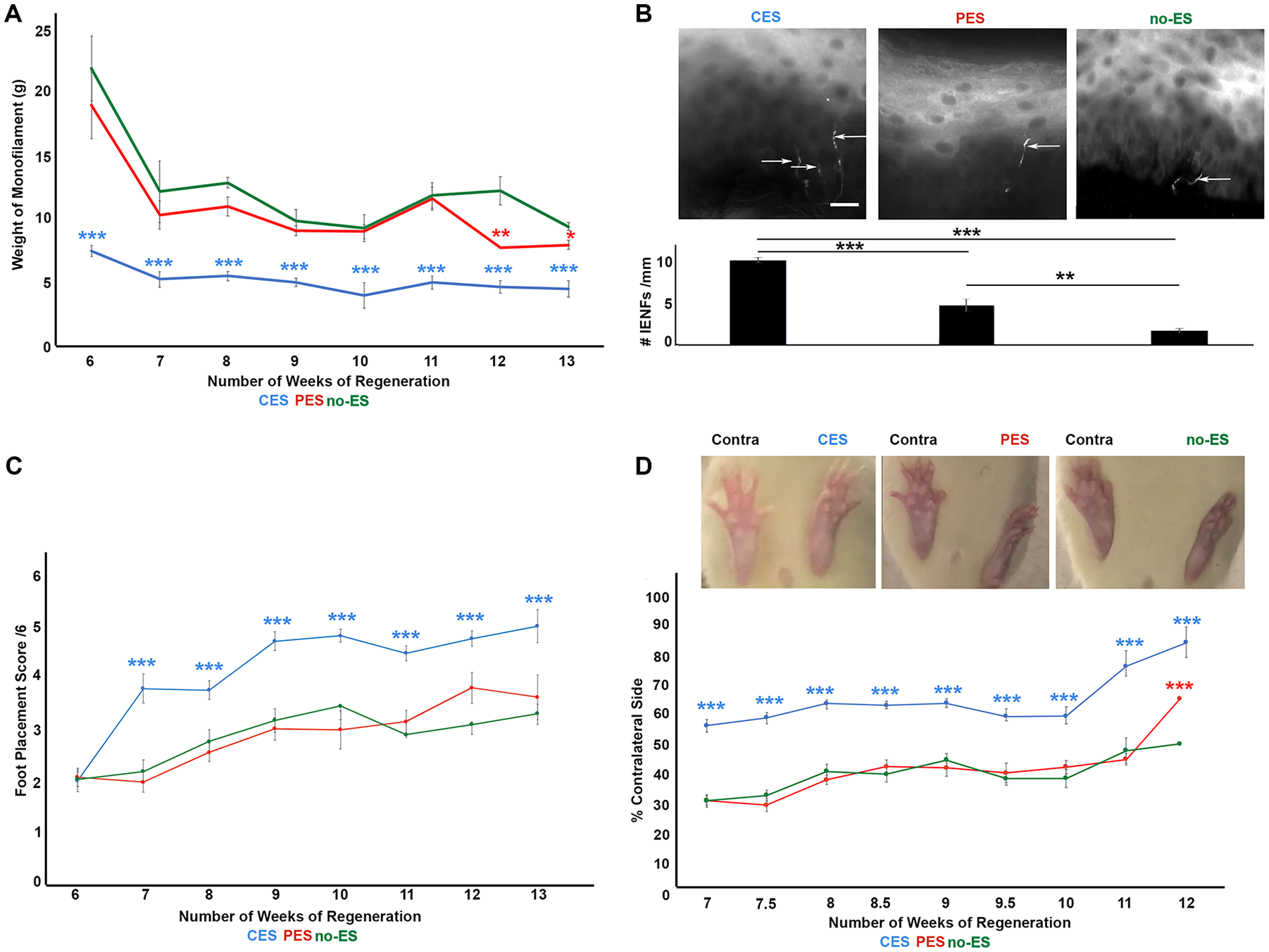
Conditioning electrical stimulation (CES) improves functional recovery greater than postoperative electrical stimulation (PES). (A) Line graph depicts results of von Frey filament testing at 6 to 13 weeks of regeneration. At all time points, animals treated with CES (blue) had significantly improved sensory recovery when compared to animals treated with PES (red), or no-stimulation negative controls (green) (***P < .001). (B) Footpad biopsy specimens were collected at 14 weeks of regeneration and processed for intraepidermal nerve fiber innervation. Significantly more PGP9.5 labeled axons were counted crossing the derma-epidermal junction in specimens obtained from CES-treated animals when compared with PES (**P < .01), or negative controls (***P < .001). (C) Line graph depicts average foot placement scores at 6 to 13 weeks of regeneration. Animals treated with CES (blue) had significantly higher foot placement scores at all timepoints beyond week 6 compared with PES (red, ***P < .001), ***P < .001), or negative controls (green, ***P < .001). (D) Representative photographs of toe-spread width (in % of contralateral side) of animals treated with CES, PES, or negative controls. Line graph of toe-spread at 6 to 13 weeks of regeneration. Wider toe-spread, indicating improved reinnervation of the intrinsic muscles of the foot, in animals treated with CES (blue) was significantly improved compared to animals treated with PES (red ***P < .001), or no stimulation (green, ***P < .001).
Von Frey behavioral outcomes were corroborated by quantification of footpad reinnervation at 14 weeks post nerve transection and autograft repair. IENF density was determined by counting the number of nerves crossing the dermal-epidermal junction. Significantly more nerves crossing the dermal-epidermal junction were identified in the CES cohort (10.1 ± 0.3 nerves/mm) compared with PES alone (4.8 ± 0.8 nerves/mm; P < .001), or the no-ES cohort (1.6 ± 0.8 nerves/mm; P < .001) (Figure 3B). PES animals had improved regeneration into the skin compared with their no-ES counterparts (P < .001).
CES Improves Motor Reinnervation Outcomes in a Nerve Autograft Model
Motor outcomes included performance on the horizontal ladder test, toe-spread gait analysis, nerve conduction testing, and gastrocnemius muscle weight. Performance on the horizontal ladder test is a measure of the motor reinnervation and dexterity of the injured limb. A score of 0 to 6 was awarded for each foot-placement on the ladder rungs, with a higher score indicating a more accurate placement. Performance on the horizontal ladder was similar between all 3 cohorts at 6 weeks of nerve regeneration (CES, 2.0 ± 0.2; PES, 2.1 ± 0.2; no-ES, 2.1 ± 0.1). By 7 weeks, however, foot placement scores for animals treated with CES improved to 3.8 ± 0.3, significantly higher than PES (2.0 ± 0.2, P < .001) or no-ES (2.2 ± 0.2, P < .001). Foot placement scores remained significantly higher in animals treated with CES when compared with PES or no-ES at 8 (3.8 ± 0.2, 2.6 ± 0.2, 2.8 ± 0.2; P < .001), 9 (4.8 ± 0.1, 3.0 ± 0.4, 3.2 ± 0.3; P < .001), 10 (4.9 ± 0.2, 3.0 ± 0.3, 3.5 ± 0.2; P < .001), 11 (4.5 ± 0.3, 3.2 ± 0.4, 2.9 ± 0.2; P < .001), 12 (4.8 ± 0.4, 3.8 ± 0.4, 3.1 ± 0.1; P < .001), and 13 (5.0 ± 0.1, 3.7 ± 0.7, 3.3 ± 0.2; P < .001) weeks of nerve regeneration (Figure 3C).
Gait analysis to evaluate toe-spread as a measure of the reinnervation to intrinsic muscles of the foot was performed at 8 to 14 weeks following nerve injury and autograft repair surgery. At 6 weeks of nerve regeneration, a wider toe-spread, indicating greater reinnervation of the intrinsic muscles of the foot, was identified in animals treated with CES (54.7% ± 2.1% of contralateral uninjured control toe-spread) when compared to PES (29.9% ± 1.5% of contralateral, P < .001) or no-ES negative control animals (29.8% ± 2.1% of contralateral, P < .001). Animals in the CES cohort had a consistently wider toe-spread, which was represented as a percentage of their contralateral paw, when compared with PES or no-ES at 8 (62.1% ± 1.1%, 36.7% ± 1.6%, 39.5% ± 2.4%; P < .001), 9 (62.1% ± 1.5%, 40.6% ± 2.6%, 43.3% ± 2.1%; P < .001), 10 (57.8% ± 2.5%, 41.0% ± 3.2%, 37.2% ± 2.2%; P < .001), 11 (74.4% ± 5.0%, 43.5% ± 2.2%, 46.5% ± 3.0%, P < .001), 12 (82.2% ± 3.1%, 65.7% ± 1.8%, 48.9% ± 4.2%, P < .001), and 13 (79.7% ± 5.5%, 48.9% ± 4.6%, 45.0% ± 3.8%; P < .001) weeks of nerve regeneration (Figure 3D).
Nerve conduction studies were performed on the intrinsic foot muscles at 10, 12, and 14 weeks of nerve regeneration. At 10 weeks of regeneration, CMAP amplitude, expressed as a percentage of the uninjured contralateral limb, was poor in all cohorts: CES (9.7% ± 1.6%), PES (6.1% ± 1.5%), and no-ES (6.2% ± 1.1%). By 12 weeks of regeneration, however, animals treated with CES had a significantly improved CMAP amplitude (58.6% ± 8.7%) when compared with animals treated with PES (13.2% ± 3.4%, P < .001) or without ES (7.6% ± 1.8%, P < .001). Similarly, at 14 weeks of regeneration, CES-treated animals had a significantly greater CMAP amplitude (66.3% ± 16.8%) than PES (16.0% ± 6.9%, P < .001) or no-ES (18.3% ± 3.3%, P < .001) treated animals (Figure 4A and B).
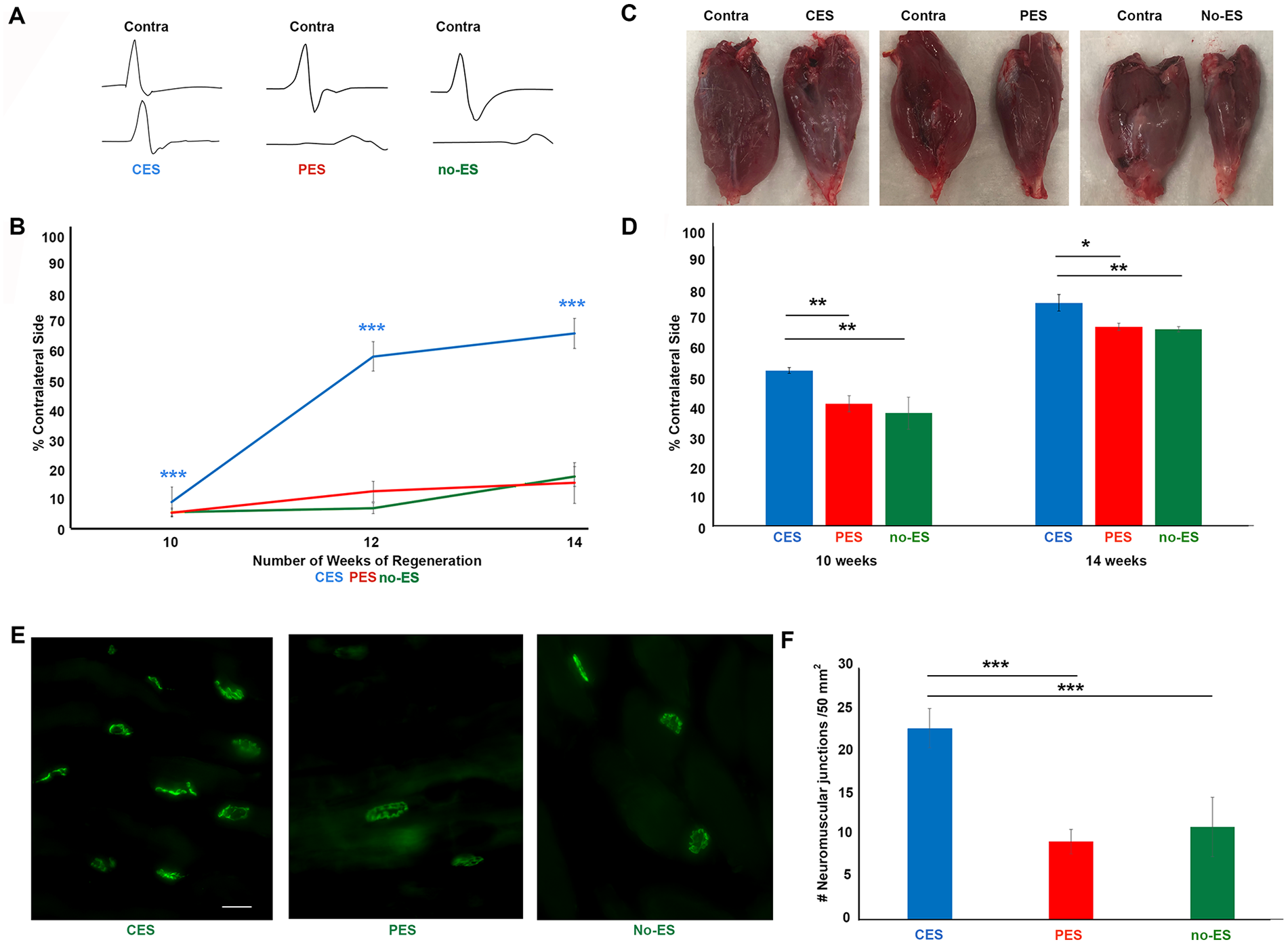
Conditioning electrical stimulation (CES) prior to autograft repair surgery promotes motor functional recovery beyond that of postoperative electrical stimulation (PES). (A) Representative compound muscle action potential (CMAP) amplitudes identified on nerve conduction studies at 14 weeks of regeneration (B). Line graph representing CMAPs (in % of contralateral side) of each animal were recorded at 10, 12, and 14 weeks post autograft repair surgery. CMAP amplitudes of the injured limb were normalized to the contralateral control. At 12 and 14 weeks of regeneration, animals treated with CES (blue) had significantly greater CMAP amplitude recovery compared with the PES (red, ***P < .001) or no electrical stimulation (green, ***P < .001) cohorts. (C) Representative photographs of injured (right) and contralateral control gastrocnemius muscle (left) are shown above a histogram displaying muscle weight. (D) Gastrocnemius muscles were harvested at 10 and 14 weeks of regeneration, and weight of the injured muscle was normalized to the contralateral control side. Animals treated with CES had significantly greater muscle weight recovery in the CES (blue) animals compared with the PES (red, *P < .01) and negative controls (green, P < .01) at 10 weeks and 14 weeks. (E) Representative photomicrographs of neuromuscular junctions labeled with α-bungarotoxin obtained from the gastrocnemius muscle of animals treated with CES, PES, or no electrical stimulation. All neuromuscular junctions were confirmed to be innervated by NF200 (data not shown). (F) Animals treated with CES had significantly more innervated acetylcholine receptor compared with the other cohorts (***P < .001).
The gastrocnemius muscles of the injured and contralateral uninjured lower limbs were weighed to assess loss of muscle bulk. Results were analyzed as the injured weight as a percentage of the uninjured limb to account for differences in animal size. At 14 weeks of regeneration, the weight of the gastrocnemius muscle on the injured side, represented as a percentage of the contralateral uninjured control, was significantly greater in the CES cohort (22.8% ± 2.3%) than in animals treated with PES (9.3% ± 1.5%, P < .01) or no-ES (11.0% ± 3.6%, P < .05). Similarly, at 14 weeks of regeneration, CES-treated animals had significantly greater muscle weight (79.8% ± 3.0%) than PES (71.2% ± 1.3%, P < .05) or no-ES controls (70.3% ± 1.0%, P < .01) (Figure 4C and D). It was observed that there were significantly more innervated neuromuscular junctions (AchRs) in the CES animal at 14 weeks compared with the PES and no-ES cohorts (Figure 4E and F). Collectively, CES had superior decreased gastrocnemius atrophy, increased NMJ innervation and improved CMAPs compared with the PES and no-ES groups.
Discussion
CES is a clinically feasible means of effectively mimicking the traditional crush conditioning lesion, causing upregulation of regeneration associated genes and conferring an increased length of axon regeneration when compared with negative controls. 2 We further demonstrated CES significantly improves sensory and motor recovery as assessed with behavioral, electrophysiological, and immunohistochemical analysis. 3 Clinical translation of CES necessitates a direct comparison with the current “best practice management” strategies. PES remains the only perioperative technique to improve nerve regeneration that has been successfully translated to, and integrated into clinical practice.
CES Has a Greater Proregenerative Effect Than PES
Successful functional recovery following peripheral nerve injury is directly related to the time required for regenerating motor or sensory axons to reach their target muscle or sensory receptor. After nerve injury, axons regenerate across the site of surgical coaptation in a “staggered” distribution rather than as a unified front. This process is termed staggered regeneration and results in a temporary stall in axon extension, delaying end target reinnervation. Delivery of 1 hour of PES immediately following surgical nerve repair improves the efficiency of staggered regeneration, decreasing this delay and thereby accelerating time to reinnervation. 7 PES has long been established as an effective and safe technique to enhance peripheral nerve regeneration. Pioneered by Nix and Hopf in the 1980s, PES has since been thoroughly investigated and shown to improve regeneration and reinnervation in animal14-17 and human models of peripheral nerve injury.5,6,18
The regenerative effects of PES are limited, as this technique enhances regeneration only at the site of surgical coaptation and does not affect the rate of axon extension along the distal stump. 7 A conditioning lesion, by contrast accelerates nerve regeneration. These differences in mechanism underline our results suggesting that nerves treated with CES prior to transection and repair have significantly greater regeneration and functional recovery compared with nerves treated with PES. The differences between these 2 techniques may be particularly important in a human model of peripheral nerve injury rather than a rodent, given differences in size. If PES accelerates regeneration only at the size of coaptation for a proximal nerve injury, regenerating axons have a great distance to regenerate which will proceed at the baseline rate of 1 mm/d, as PES has no effects distal to the site of injury. In contrast, CES accelerates nerve regeneration and therefore has the potential to significantly decrease the amount of time necessary for end target reinnervation, thereby improving functional recovery.
CES Enhances Regeneration and Reinnervation in a Nerve Autograft Model
The nerve gap is a common challenge for the peripheral nerve surgeon. Primary repair under tension precludes regeneration and worsens outcomes, therefore reconstruction alternatives are required to bridge the gap. Nerve autografting is the “gold standard” technique for overcoming a nerve gap due to the relative technical simplicity and availability of donor nerves. In addition to traumatic injuries of the upper and lower limbs, nerve grafts are an important tool in the treatment of facial paralysis and for oncologic reconstruction. 8 Though PES is well studied in animal and human models of primary coaptation, the effects of electrical stimulation on promoting regeneration through a graft and 2 sites of coaptation are poorly understood. Our findings suggest that while PES may be beneficial in early regeneration, it has no effect on reinnervation outcomes in autologous nerve grafts. These findings are in concordance with Witzel et al, 13 who delivered electrical stimulation (1 hour, 20 Hz) to the sciatic nerve at the time of nerve grafting (1 cm graft). 13 When compared with nonstimulated control grafts, they found that animals receiving PES had a significant increase in regenerating axons within the grafted nerve with greater arborization at 5 and 7 days postgrafting, but no significant difference in length or speed of regeneration. In keeping with our findings, Huang et al, 8 found that 1 hour of electrical stimulation delivered at the time of nerve grafting (1 cm) accelerated regeneration across the nerve gap; while PES improved initial motor recovery based on gait analysis and nerve conduction studies, final outcomes did not differ between animals who received PES and those that did not. PES was, however, shown to improve outcomes when a long nerve gap was reconstructed using a longitudinal scaffold. 19 Regeneration outcomes with allograft or nerve conduits have poorer outcomes than autograft repair; therefore, the effects of PES on enhancing outcomes in these models may be more apparent.
Unlike PES, animals treated with CES prior to autograft nerve reconstruction had significantly improved regeneration and sensorimotor reinnervation outcomes at all timepoints investigated. CES may therefore be a promising technique to improve outcomes for patients undergoing elective autograft repair where a large nerve gap is anticipated, such as in oncologic resections, cross-face nerve grafting for facial reanimation, or in the presence of significant soft tissue damage. The decision to perform nerve autograft reconstruction, however, is often not made until intraoperatively when the size of the nerve gap can be adequately assessed. Given the atraumatic nature of electrical stimulation, delivering CES prior to surgery is not anticipated to confer substantial risk to the patient. If it is shown that CES is as well-tolerated and safe as PES, patients with a nerve injury suspected to require grafting could still be treated with CES prior to exploration without increasing the potential risk of injury or discomfort to the patient. Human trials are therefore critical to determining the safety profile and patient acceptance of this technique.
Conclusions
CES significantly improves outcomes following peripheral nerve regeneration. Guiding best practice patient management requires a comparison of outcomes of CES with PES, the only perioperative intervention currently used routinely in clinical practice. Our results suggest that CES enhances regeneration and reinnervation outcomes greater than PES. Further research to determine the molecular mechanism of CES, and how PES affects these processes is required to explain this relationship. Finally, assuming that CES is capable of inducing sustained acceleration of regeneration over long distances in humans, it may be a clinically preferred adjunct treatment to significantly improve regeneration and reinnervation outcomes in elective autograft nerve repair surgeries.
Footnotes
Acknowledgements
We acknowledge the excellent technical assistance of Susanne Lingrell and Haecy Macandili.
Contributions
CAW, J-LBS, JO, and KMC contributed to the concept of the article. J-LBS, AWMC, and TK-W contributed to the experimental science, CAW and J-LBS wrote the manuscript, and KMC and JO edited the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Canadian Institute of Health Research project grant (CAW, KMC), the Canadian Institute of Health Research PhD Scholarship (J-LBS), the University of Alberta Hospital Foundation (CAW, JO, KMC), the Glenrose Research Fund (KMC), and the Edmonton Civic Employees grant (CAW, KMC).
