Abstract
Cognitive impairment is an important target for rehabilitation as it is common following stroke, is associated with reduced quality of life and interferes with motor and other types of recovery interventions. Cognitive function following stroke was identified as an important, but relatively neglected area during the first Stroke Recovery and Rehabilitation Roundtable (SRRR I), leading to a Cognition Working Group being convened as part of SRRR II. There is currently insufficient evidence to build consensus on specific approaches to cognitive rehabilitation. However, we present recommendations on the integration of cognitive assessments into stroke recovery studies generally and define priorities for ongoing and future research for stroke recovery and rehabilitation. A number of promising interventions are ready to be taken forward to trials to tackle the gap in evidence for cognitive rehabilitation. However, to accelerate progress requires that we coordinate efforts to tackle multiple gaps along the whole translational pathway.
Background
Epidemiology and Importance
The first Stroke Recovery and Rehabilitation Roundtable (SRRR I) 1 focused primarily on motor recovery, as it was a more developed area in terms of mechanistic understanding and readiness for clinical trials. Cognitive function has since been identified as an area of unmet need requiring discussion. The definition of post-stroke cognitive impairment, adopted by the cognition working group in SRRR II, is a new cognitive deficit that develops in the first three months following stroke and persists for a minimum of six months, which is not explained by any other condition or disease. 2 Such deficits occur in 30–40% of individuals, 3 in one or more cognitive domains, including, language, executive function, visuospatial cognition, episodic and working memory. 4 Furthermore, cognitive, affective and behavioral consequences of stroke are more strongly associated with poor quality of life (QoL) than measures of physical disability. 5 The risk of dementia after stroke is high, with a post-event incidence of 34% one year after severe stroke (NIHSS>10), with lower rates after TIA and minor stroke. 6 International guidelines highlight the lack of evidence on specific approaches for rehabilitation of cognitive function as a significant gap. 7 Therefore, a major goal of SRRR II was to define current consensus and research priorities to advance our understanding and maximize research alignment in post-stroke cognition.
Mechanisms of Impairment and Recovery
Cognitive function emerges from complex interactions between cortical and subcortical sites across distributed brain networks. Lesions, such as focal stroke, may disrupt networks either directly, or indirectly, through secondary mechanisms of injury. A proper account of the consequences of damage to specific domains therefore requires an understanding of the distributed neural networks that underpin these neurocognitive domains and their interactions 8 (Figure 1). Typically following stroke, multiple networks are affected to varying degrees. This heterogeneity, along with a vast range of approaches for testing cognition (replicated in animal behavioral paradigms) creates difficulties in defining consistent measurement approaches for use in rehabilitation trials. Acutely, impairments can be exaggerated by systemic factors, such as infection, metabolic disorders and drugs causing delirium. Moreover, many cognitive impairments can be subtle and often delayed, and manifest when patients return fully to the complex demands of daily life. Another complicating factor is the lack of information on pre-morbid cognitive status. 9 On the positive side, alterations in structural and functional connectivity can now be evaluated non-invasively by imaging and electrophysiological techniques. However, longitudinal studies with clinical outcomes are needed to fully understand how brain connectivity changes underpin the evolution of post-stroke cognitive impairment and impact on QoL.
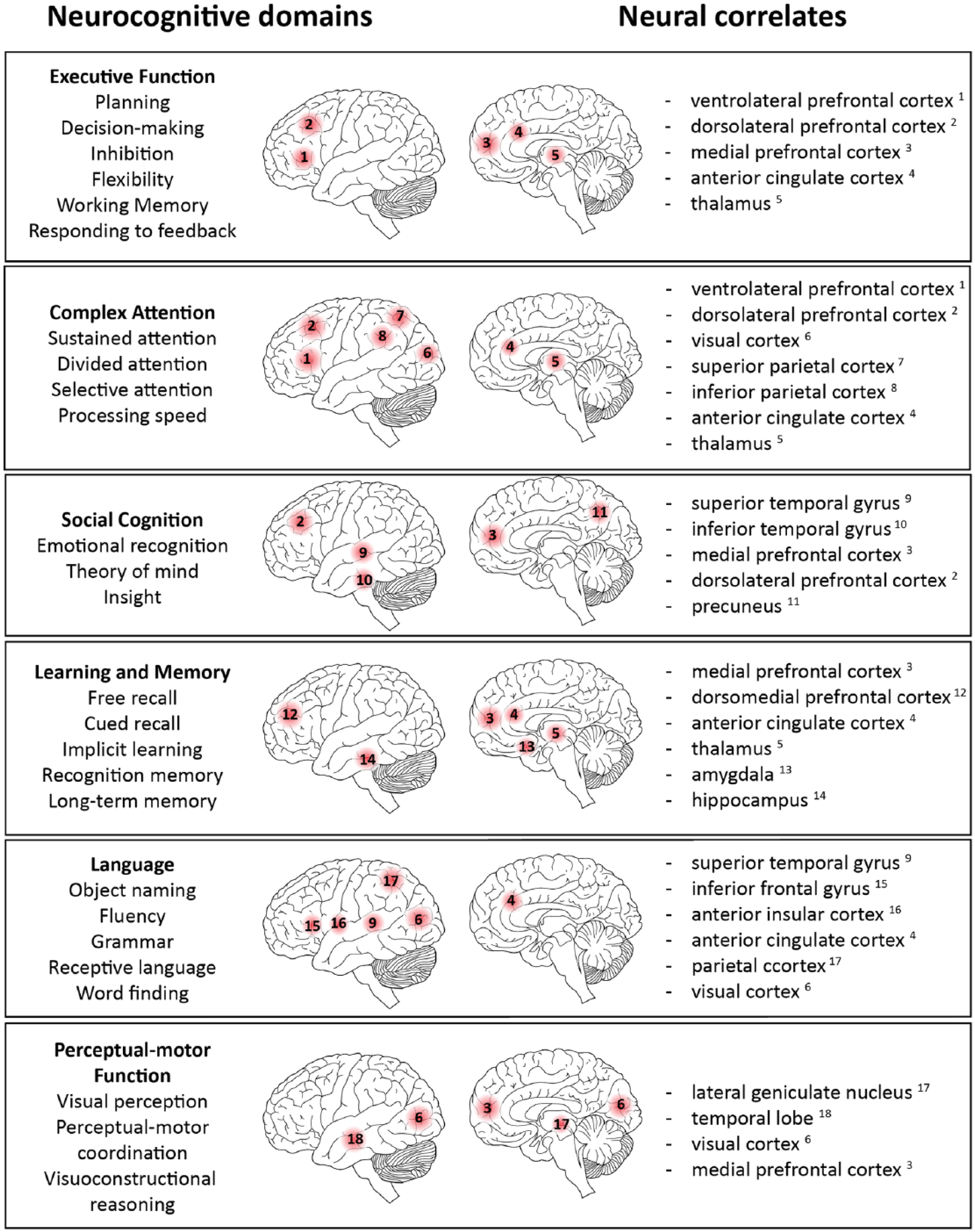
Neural networks that underpin the neurocognitive domains. Each figure sketches the major regions recognized as part of the network supporting each domain. Key points are that all networks are widely distributed across the brain frequently intersecting and overlapping so that multiple networks may be injured by a single stroke.
Context and Scope
There have been a number of initiatives to develop consensus in relation to clinical entities that overlap or can co-exist with acute stroke, such as cerebral small vessel disease and vascular cognitive impairment (VCI).10,11 These include agreed guidelines for diagnosis and standardized approaches to neuropsychological testing and imaging.12,13 A current UK initiative is seeking to build consensus around animal models of VCI. 14 These efforts are relevant, notably in the emphasis on translation and new therapies. However, the SRRR II Cognitive working group remains focused primarily on the setting of stroke rehabilitation (process of care) and recovery trials. Trials to modify the course of VCI, and aspects such as silent infarction or insidious small vessel disease, were not within the group’s scope.
Animal models provide a means to explore basic mechanisms of plasticity and repair and test interventions to promote repair and ameliorate secondary injury. The two-way interaction between preclinical and clinical research was therefore viewed as central to cognitive recovery research and a core component of the working group’s mission.
Methods
The cognition working group gathered experts from a diverse range of fields. A core group met in Saint-Sauveur, Canada in October 2018 and a wider advisory group was established to provide additional expertise. Expertise in clinical stroke, rodent models of stroke, human and animal neuroimaging, neuropsychology, the neurobiology of language and cognitive rehabilitation were represented.
In advance of SRRR II, a structured survey was sent to participants and from this, a list of major challenges was defined, and an agenda formed for the working group meeting. In a number of areas, it was recognized that there was inadequate evidence to support formal consensus. In these areas, the methodology shifted to definition of major priorities for research in post-stroke cognition.
Cognition in Rehabilitation Research: Generic Recommendations
An essential aspect of outcome to be measured in a stroke recovery study might be defined as one that: is likely affected by stroke; is sensitive to therapy; and has importance relative to patients’ overall outcome. Therapeutic approaches are often not specific to motor function (e.g. exercise) so that cognitive improvement may be part of a wider therapeutic effect. Furthermore, because cognitive impairment determines QoL after stroke, the collective view was that cognitive function meets the predefined criteria for evaluation in all trials and observational studies of stroke recovery (see Table 1). No consensus was reached on a single approach to assessment, but the need for tests that are easily implemented, validated in different cultural settings and sensitive to executive function was recognized.
Consensus recommendations.
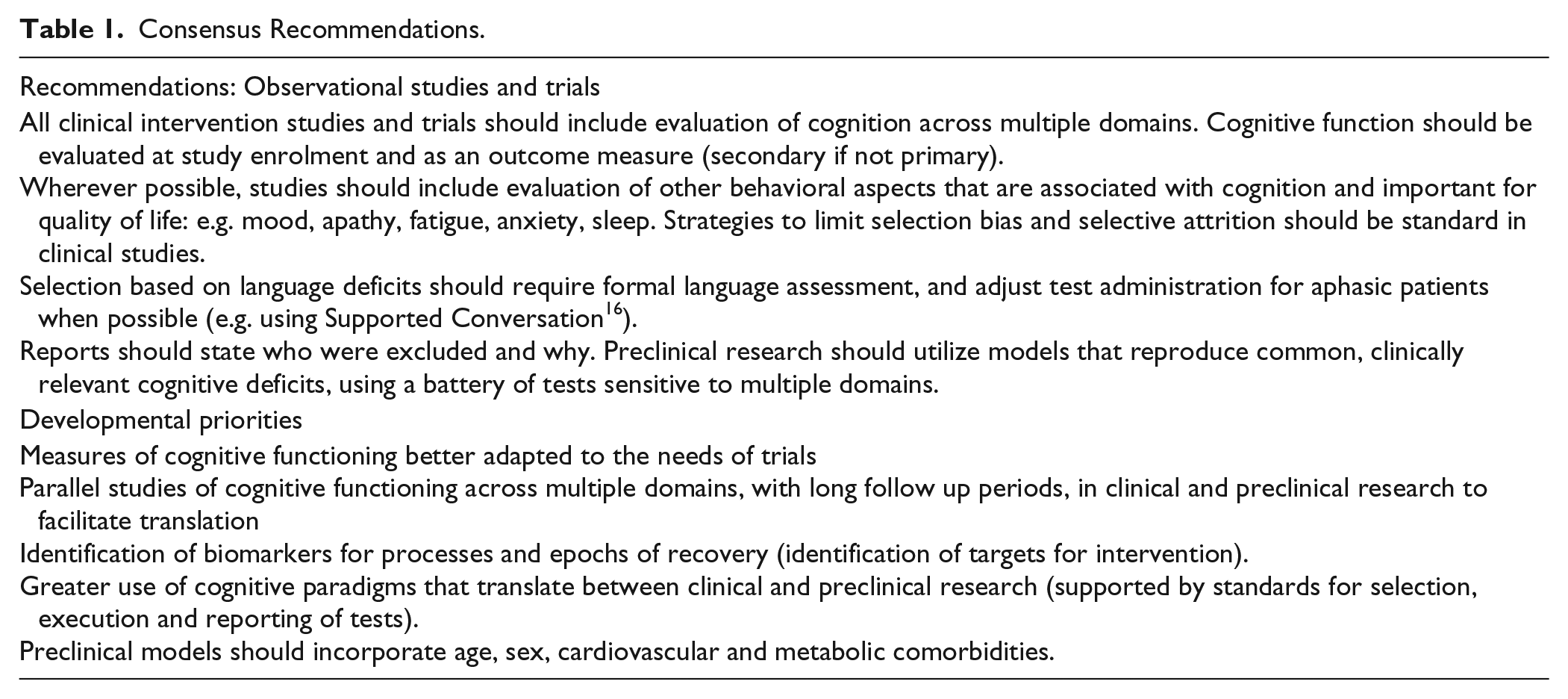
Selection bias and selective attrition—for example, the exclusion of patients with aphasia or other barriers to standard cognitive assessments (e.g. visual impairment) 15 was identified as a major concern. Inclusion of explicit strategies to minimize bias was another area of agreed consensus (Table 1).
Research Priorities: Enhancing the Translational Potential of Preclinical Cognitive Recovery Research
Preclinical stroke recovery research focused on motor systems has identified critical periods of sensorimotor recovery and cellular mechanisms that govern neural repair following stroke.17,18 Previous consensus recommendations for aligning preclinical and clinical stroke recovery research from SRRR I focused on sensorimotor recovery, although many of these guidelines could be applied to cognitive recovery. 19 The preclinical recommendations emphasized the importance of using sensitive outcome measures that are analogous to measures used in humans.
Traditionally, preclinical research has utilized the middle cerebral artery occlusion (MCAo) model. Going forward, greater priority should be given to models that replicate behavioral impairments observed clinically using photothrombosis, endothelin-1, and microvascular emboli models that produce targeted damage to brain regions without major motor deficits. Additionally, models should assess cognitive deficits in a variety of domains, particularly higher-order processes commonly affected in humans. Most of these domains can be examined in rodents (Table 2); however, for some higher order functions non-human primate models may have greater translational potential. While traditional paradigms that assess cognition still have merit (e.g. Morris water maze, etc.), greater emphasis should be placed on cognitive tasks that directly translate across species. This can be achieved using rodent touch-screen tablets that allows testing paradigms that mirror those employed in humans. 20 A significant component of stroke rehabilitation is relearning; therefore, preclinical studies should consider including paradigms that measure both relearning of old information as well as learning new task information. We also know that the severity of cognitive deficits can be modulated by underlying cognitive risk factors. Therefore, preclinical models should incorporate features such as age, sex, cardiovascular and metabolic comorbidities. 19
Cognitive paradigms and translation from models to humans.
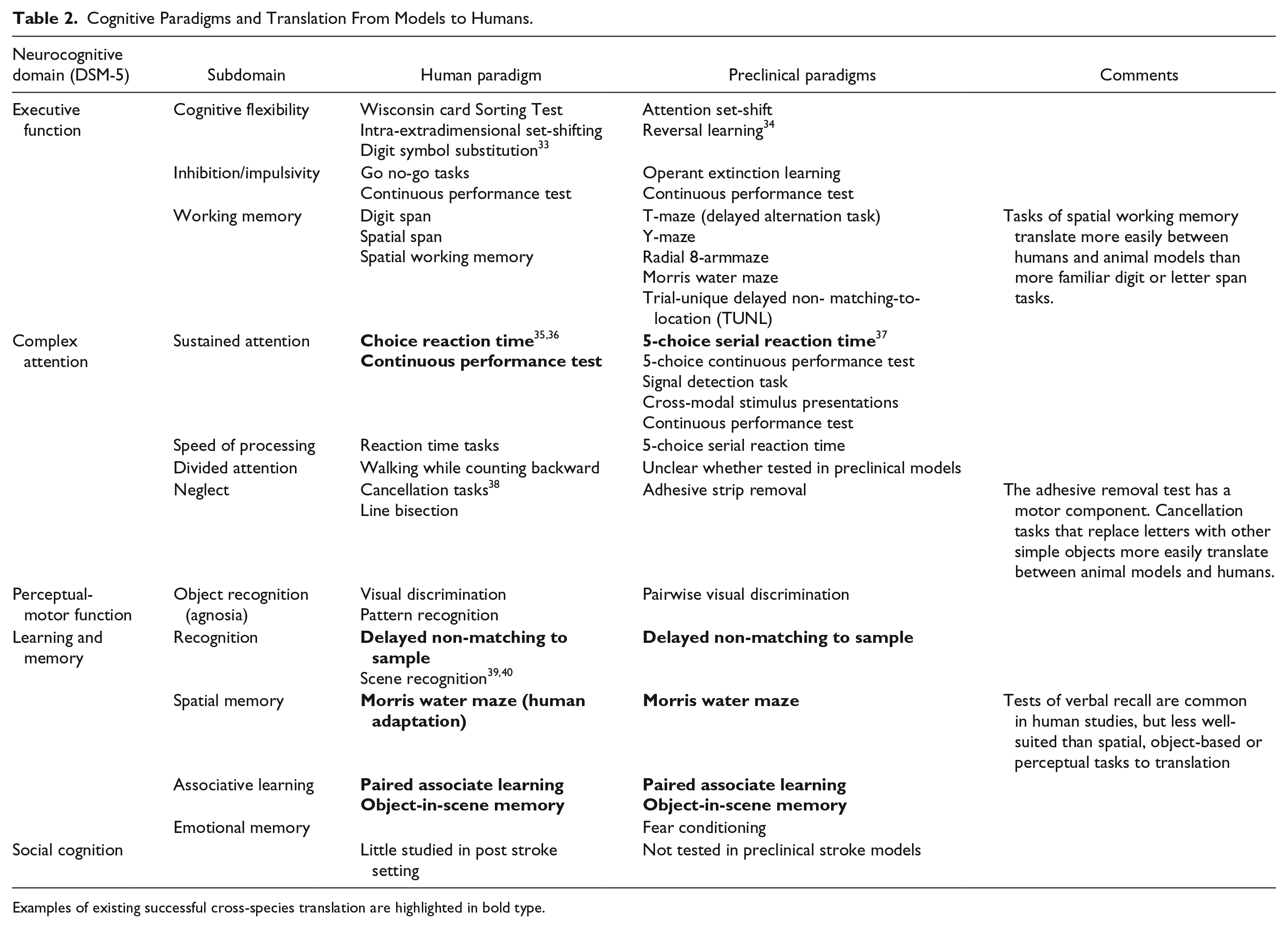
Examples of existing successful cross-species translation are highlighted in bold type.
Effort should be made to monitor long-term behavioral changes to reflect chronicity and progressive decline in cognitive function, in combination with structural and functional changes in neural networks (histology, neuroanatomical tracing, MRI, electrophysiology), to identify important epochs and markers of cognitive recovery. In addition, the preclinical environment can readily validate novel therapeutic strategies. To maximize translation, adoption of a level of rigor equivalent to clinical trials in humans is required, including randomization, blinding, consistent training in outcome evaluation, and standards for reporting of experimental procedures. 20
Research Priorities: Translational and Clinical Research
Recovery Epochs, Therapeutic Windows and Biomarkers
The cellular and biochemical changes triggered by stroke include both early and late events that occur both proximal and distal to the site of injury. The notion of distinct recovery epochs dominated by one or several cellular or biochemical mechanisms emphasizes the challenge of correct timing of interventions in trials. One consensus conclusion was that epochs need to be defined mechanistically because mechanisms define candidate therapies. Major gaps in existing data include a lack of detailed longitudinal studies using imaging (structural and functional) and other biomarkers in humans, and a relative paucity of long-term follow up data in animals. Biomarkers provide promising avenues for the definition of epochs, with the potential to span clinical and preclinical models. For example, positron emission tomography (PET) ligands can track microglial activation to provide insights into inflammatory mechanisms, and label biochemical hallmarks of late neurodegeneration, such as amyloid and tau deposition. Translation is simply one aspect of the role that biomarkers can play in optimizing approaches to rehabilitation and recovery research. 21 The previous consensus position—that no cognitive biomarkers are yet ready for use in phase III trials, with the possible exception of lesion anatomy-based prediction of language outcome—remains intact. However, identifying and evaluating sensitive cognitive biomarkers, to support translational research, is a developmental priority. One promising area to begin biomarker development is chronic reactive astrogliosis within white matter tracts, which is linked to delayed impairment in memory retrieval in humans and rodents. 22
Premorbid Function and Functional Reserve
Pre-stroke cognitive performance is thought to influence cognitive outcome after stroke, including the risk of developing future dementia. However, accurate ascertainment of premorbid ability is challenging. 9 Further, demographic factors (e.g. education) may also contribute to altered cognitive outcomes following stroke. There is no validated method to assess cognitive reserve although multivariate approaches to neuroimaging data are beginning to reveal information about overall brain status, or “brain age.” Application in a stroke rehabilitation setting is, however, complicated by the structural and functional alterations induced by injury, which are known to extend well beyond sites of visible infarction. The potential of multivariate approaches to address the difficult questions in stroke rehabilitation 21 is an exciting area of future research.
Integration of Patient- and Carer-Reported Outcome Measures
Much attention has focused on defining optimal objective testing approaches for post-stroke cognitive impairment. However, more work is required to link this approach to stroke outcome as defined by patients, relatives and carers. Understanding the associations between cognitive function and QoL after stroke is essential both in setting research priorities—across the translational spectrum—and defining the health economics benefits of new interventions. Technology provides new opportunities for integration of objective, patient- and carer-reported outcome measures. For example, touchscreen tablets and smartphones can deliver cognitive tests and prompt reporting by patients and carers. In addition, the development of virtual reality provides a new way to test cognition in a more ecologically valid manner. 23 Wearable devices can provide information on natural behavior (locomotion) and information about factors that modulate cognitive performance, such as sleep. 24 The integration of patient- and carer-reported and technology-derived information with more traditional evaluation of cognition presents an opportunity for recovery research.
Candidate Therapies for Cognitive Rehabilitation
The approaches we are interested in directly target cognitive impairments themselves (e.g. executive functions) and not aids (e.g. pagers) to improve daily real-world functioning without changing cognitive processing. There are several detailed reviews relating to this topic,25,26 so here we confine ourselves to outlining some key issues. Firstly, it is difficult to isolate individual cognitive functions in terms of measuring outcomes (e.g. impairments in working memory and attention can both impact performance on tests of executive function). Secondly, interventions need to be delivered in high enough doses to maximize the likelihood of clinically meaningful gains. A way to do this is to augment therapist-delivered, face-to-face training with digital carer-delivered therapies. This has shown promise in studies designed to improve: working memory 27 ; goal processing and sustained attention. 28 While not all cognitive interventions and tests currently have direct equivalents in animal models (Table 2), there are synergies in cellular and genetic mechanisms that mediate higher cognitive functions across species. For example, stroke induces an elevation in tonic GABA signaling and compounds that dampen this response have shown promise in animal models for motor recovery 29 and are currently being tested in a Phase II trial (ClinicalTrials.gov ID; Servier RESTORE BRAIN Study—NCT02877615). These compounds have also been tested in a preclinical model of VCI and shown to improve working memory. Similarly, Brain Derived Neurotrophic Factor (BDNF), which plays an important role in regulating plasticity, decreases with age and negatively impact on recovery. Aerobic exercise training can elevate BDNF levels and this has been implicated as mediating improved spatial memory in healthy older adults; training increased hippocampal volume, effectively reversing age-related loss by one to two years. 30 A recent phase II clinical trial in patients with post-stroke cognitive impairment showed that exercise paired with cognitive training improved fluid intelligence, but the relationship to BDNF was less clear. 31 It is here that animal models provide a much more fine-grained approach to understanding the intricacies of the cellular and genetic substrates that underpin human cognition, 32 allowing us to be better positioned to test interventions for cognitive recovery studies in patients.
Conclusions
Research on cognitive recovery after stroke is at an earlier stage of evolution than research in motor recovery. Nevertheless, international consensus is possible in a number of areas. All clinical stroke recovery studies should integrate cognitive evaluation and outcome into their design. Preclinical basic neuroscience studies are essential for developing new interventions to enhance recovery. In order to achieve this, greater alignment between preclinical and clinical research—and the development of an agenda of shared priorities—is required to accelerate progress towards novel therapies. This is best achieved using a bedside to bench to bedside approach.
Footnotes
Acknowledgements
The authors disclose receipt of the following financial support to conduct this meeting: Canadian Institutes of Health Research (CIHR) CaSTOR (Canadian Stroke Trials for Optimized Results) Group (note that CaSTOR is a joint initiative of the Canadian Stroke Consortium and the Canadian Partnership for Stroke Recovery), Heart and Stroke Canadian Partnership for Stroke Recovery, and NHMRC Centre of Research Excellence in Stroke Rehabilitation and Brain Recovery. An unrestricted educational grant was provided by Ipsen Pharma. We would also like to acknowledge, Julie Bernhardt for convening the second Stroke Rehabilitation and Recovery Roundtable (SRRR II), Dale Corbett and Karen Borschmann for organizing the meeting and Farrell Leibovitch for moderating discussions.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Note
This contribution, first published in International Journal of Stroke, is being co-published in the following journals: Neurorehabilitation and Neural Repair.
