Abstract
Background. Severe poststroke arm impairment is associated with greater activation of the nonlesioned hemisphere during movement of the affected arm. The circumstances under which this activation may be adaptive or maladaptive remain unclear. Objective. To identify the functional relevance of key lesioned and nonlesioned hemisphere motor areas to reaching performance in patients with mild versus severe arm impairment. Methods. A total of 20 participants with chronic stroke performed a reaching response time task with their affected arm. During the reaction time period, a transient magnetic stimulus was applied over the primary (M1) or dorsal premotor cortex (PMd) of either hemisphere, and the effect of the perturbation on movement time (MT) was calculated. Results. For perturbation of the nonlesioned hemisphere, there was a significant interaction effect of Site of perturbation (PMd vs M1) by Group (mild vs severe; P < .001). Perturbation of PMd had a greater effect on MT in the severe versus the mild group. This effect was not observed with perturbation of M1. For perturbation of the lesioned hemisphere, there was a main effect of site of perturbation (P < .05), with perturbation of M1 having a greater effect on MT than PMd. Conclusions. These results demonstrate that, in the context of reaching movements, the role of the nonlesioned hemisphere depends on both impairment severity and the specific site that is targeted. A deeper understanding of these individual-, task-, and site-specific factors is essential for advancing the potential usefulness of neuromodulation to enhance poststroke motor recovery.
Keywords
Introduction
Stroke exacts extensive monetary and personal costs. Its high incidence (nearly 800 000 per year in the United States) and increasing survival rates have made it a leading cause of long-term disability.1,2 Upper-extremity hemiparesis is a major contributor to poststroke disability, and the majority of stroke survivors demonstrate some degree of hemiparesis on initial assessment. 3 Following a 3- to 6-month period of rapid improvement in motor function, recovery slows, and many are left with permanent impairment of the affected arm.3-6
The severity of chronic motor impairment depends to a large degree on the amount of corticospinal tract (CST) damage.7,8 The fibers forming the CST converge and pass through the posterior limb of the internal capsule (PLIC). The PLIC is particularly vulnerable to middle cerebral artery infarction, which is the most common type of stroke. Because the fibers of the CST are so tightly bundled as they descend through the PLIC, even a small infarct in this area can have devastating effects on motor function. Any potential benefit of reorganization within cortical regions “upstream” from the infarct are limited without this critical conduit. Indeed, there is increasing evidence that motor recovery can be best predicted when markers of CST integrity are taken into account.7,9-11
Functional imaging studies have long shown that, during movement of the affected hand, well-recovered patients have lateralized activation centering on the motor areas of the lesioned hemisphere, whereas those with more severe residual impairment have more bilateral activation of motor areas.12-14 Several studies have demonstrated a functional role for certain types of nonlesioned hemisphere activity, particularly in patients with more severe CST damage.15-17 The factors that lead to an adaptive contribution of the nonlesioned hemisphere, however, need to be more clearly delineated. 18 It has yet to be affirmatively identified, for example, if the adaptive contribution is site specific, although evidence suggests that the nonlesioned dorsal premotor cortex (PMd) may be a particularly promising site for more severely impaired patients.17,19,20
Here, we examined the role of nonlesioned hemisphere PMd versus the primary motor cortex (M1) in subcortical stroke patients with no voluntary finger or wrist movement (“severe impairment”) versus those with partial recovery of voluntary wrist and finger movement (“mild impairment”), as they performed a motor task that used more proximal muscle groups: goal-directed reaching. Because the ability to perform voluntary finger movements is directly related to the integrity of the CST, 21 these 2 groups were considered to have partial sparing versus near-complete disruption of the CST.
We used transcranial magnetic stimulation (TMS) to transiently perturb cortical activation while patients performed a reaching task and measured the effect on reaching performance. We studied forward reaching movements instead of hand movement because severely impaired patients often retain (or regain) some amount of shoulder and elbow movement and, thus, have the potential to perform the task. In addition, reaching is functionally important, and a deeper understanding of potential contributors to reaching recovery could have a major impact on functional independence and quality of life, even in patients who lack hand function.
We hypothesized that the nonlesioned hemisphere would have a greater functional role in patients with severe impairment. More important, we hypothesized that within the nonlesioned hemisphere, PMd would have a greater role than M1 in patients with severe impairment but not in patients with mild impairment. We also hypothesized that motor areas of the lesioned hemisphere would have a more prominent role in patients with mild versus more severe impairment.
Methods
Participants
The study sample was recruited from MedStar National Rehabilitation Hospital and the surrounding community. MedStar Health Research Institute’s Institutional Review Board approved the study protocol and recruitment procedures, and written informed consent was obtained from each patient. Patients were assigned to either the mild or severely impaired group based on the presence or absence of ≥10° of voluntary wrist and finger extension. Inclusion criteria for both groups included having had a single, subcortical stroke (with or without cortical involvement) >6 months prior to study enrollment and being between the ages of 18 and 85 years, with no history of other neurological disorders or orthopedic injury affecting the upper extremity. Patients were excluded if they were unable to perform the reaching task or had contraindications to magnetic resonance imaging (MRI) or TMS procedures (pregnancy, history of craniotomy or epilepsy, metallic objects implanted in the body, or implanted medical devices). Patients were also excluded if they had lesions involving the brainstem, cerebellum, or a large area of cortex extending to or near the surface of the brain.
Clinical Measures
Clinical measures included the Upper Extremity Fugl-Meyer Assessment (UEFM),22-24 the Modified Ashworth Scale, 25 the National Institutes of Health Stroke Scale, NINDS 2011,26,27 and the Mini Mental Status Exam. 28 Licensed and trained clinicians administered all the clinical assessments.
Experimental Setup
Reaching Task
Patients were seated in a high-backed, adjustable-height chair with their torso restrained by a harness attached to the seat back (for a schematic of the experimental setup, 29 see Figure 1). The chair faced a table with a half-circle cut out and was adjusted so that the table surface was just above the patient’s thighs and the patient’s torso was in contact with the innermost aspect of the cut out half-circle. This arrangement minimized compensatory movements of the trunk during reaching movements. Two 7.6 × 7.6 cm2 response pads, their inside edges separated by 2 cm and centered on the individual’s sternum, were placed at 80% of the maximum distance the patient could reach with his/her affected arm. The position of the chair relative to the table and the position of the response pads on the table were recorded and carefully reproduced for each subsequent test session.
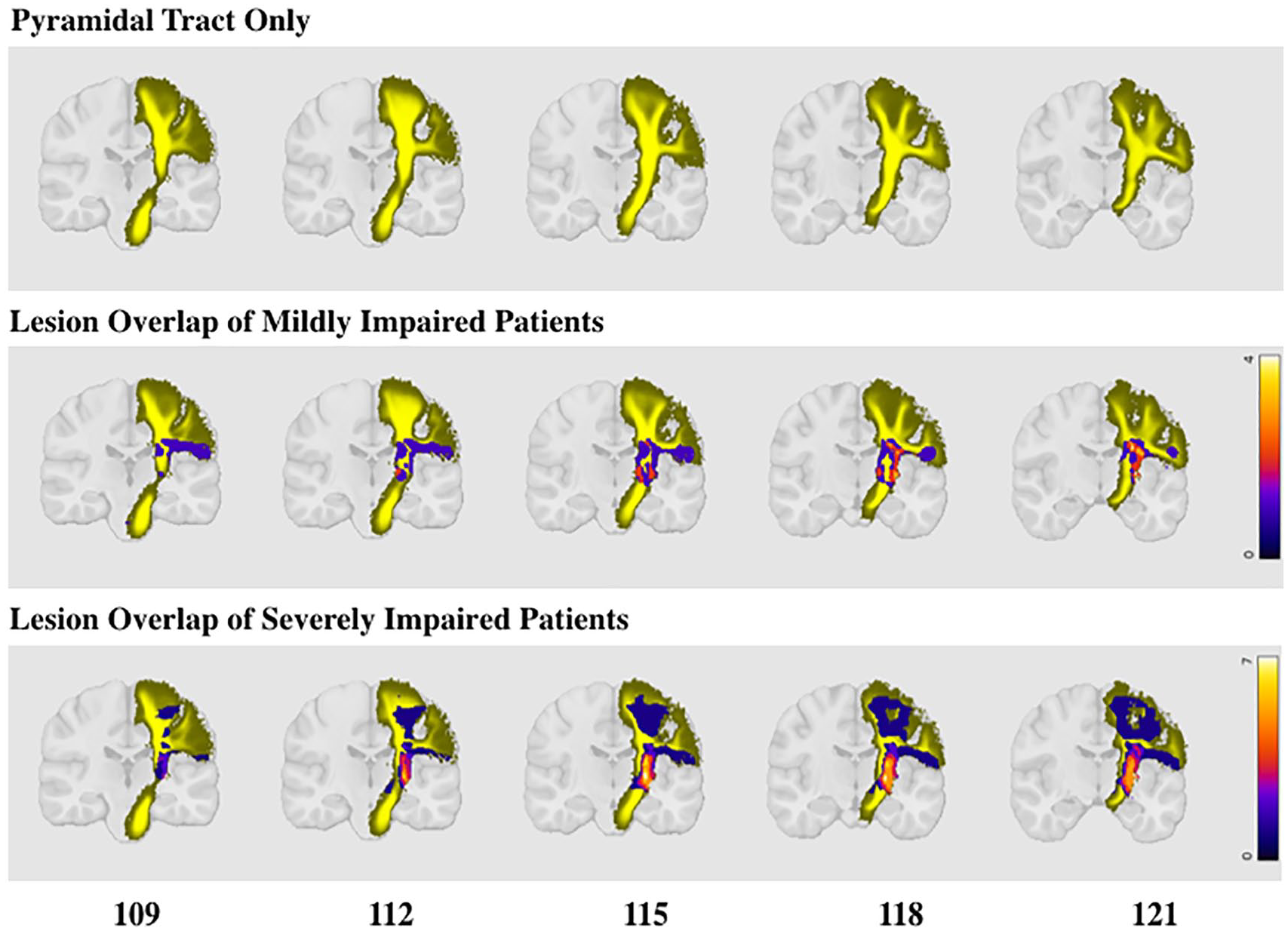
Lesion overlap maps for each group (mild and severe), overlaid on whole-fanning pyramidal tract atlas. Top: Population-based whole-fanning pyramidal tract atlas from Chenot et al. 43 Middle: Lesion overlap of mild group with pyramidal tract. Bottom: Lesion overlap of severely impaired group with pyramidal tract. Whereas the between-group difference in the amount of overlap with the whole pyramidal tract was not significant [2.6% vs 4.3% overlap; t(18) = −0.8, P = .5], the amount of lesion overlap specifically with the PLIC was significantly greater in the severe (16.3% overlap) than in the mild (3.8% overlap) group [t(18) = −2.3; P = .03].
The reaching task was performed within a choice reaction time paradigm. A visual cue indicated which response pad to press (left or right) and also served as the “Go” cue. Instructions to the patient were as follows: “When the ‘Go’ signal appears, quickly reach out to touch the indicated target.” A choice reaction time task was used to prevent the motor plan from being fully assembled prior to the appearance of the “Go” cue. To maintain consistency between patients and groups, all reaching movements were performed with the forearm pronated and fingers flexed. Time between successive trials and order of response pad selection were randomized across trials to prevent anticipation or learning effects. The timing and target of each “Go” signal and the time of target contact were programmed and recorded using E-Prime 3 (Psychology Software Tools, Sharpsburg, PA). An infrared marker, placed on the dorsum of the wrist at the midpoint between the radial and ulnar styloids, and 2 wall-mounted camera systems (containing 3 cameras each) were used to record the 3D endpoint trajectory of the movement at a sampling frequency of 300 Hz (Northern Digital Inc, Optotrak Certus movement capture system, Waterloo, Canada). The event timing, delivery of TMS stimuli, and recording of kinematic data were synchronized using E-Prime.
TMS Perturbation
Prior to the first test session, T1-weighted MRI brain scans were recorded without contrast at 1-mm slice thickness on either a Phillips CV Intera 1.5T MRI (TE = 4.76 ms; TR = 1400 ms; 25° flip angle; NEX = 1; FOV = 250 × 250 cm2) or a Siemens 3T Tim Trio (TE = 2.52 ms; TR = 1900 ms; 9° flip angle; NEX = 1; FOV = 250 × 250 cm2) scanner. The DICOM images were reconstructed using the Brainsight Neuronavigation System (Rogue Research, Inc, Montréal, Canada), which provided real-time visualization and recording of stimulation location relative to the underlying surface anatomy.
TMS was delivered via a figure-of-eight TMS coil with inner and outer diameters of 35 and 75 mm, respectively (MagProX100 with MagOption, MagVenture Inc, Atlanta, GA). The TMS coil was oriented on the scalp over the target site with the handle pointed posteriorly, 45° between the coronal and sagittal planes. 30 Coil position for targeting M1 was identified anatomically as the “hand knob” area of the precentral gyrus 31 and then adjusted to the TMS position and orientation that most readily elicited motor-evoked potentials (MEPs) from biceps or triceps brachii of the contralateral arm. This site was used to determine resting motor threshold (RMT) prior to each data collection session. RMT was defined as the minimum stimulation intensity required to elicit MEPs >50 µV in 5 of 10 trials. For the lesioned hemisphere, when no MEPs could be elicited even with high-intensity stimulation, the M1 site was defined as the mirror image of the nonlesioned hemisphere M1 site, and nonlesioned hemisphere RMT was used to set subsequent stimulation intensities for both hemispheres (average RMTs are presented in Table 1; data for only mildly impaired patients are presented for the lesioned hemisphere because data for almost all severely impaired patients were greater than maximum stimulator output). The PMd was identified anatomically as the posterior third of the region between the middle and superior frontal gyri. 32 A test of stimulus spread (10 repetitions of TMS at 120% RMT) was applied to PMd to ensure that no MEPs were elicited from the contralateral arm, confirming that stimulation applied to the PMd site did not directly affect M1 output via generalized spreading.
Demographic Data, Clinical Similarities, and Differences in Movement Abilities Between Mildly and Severely Impaired Patients. a
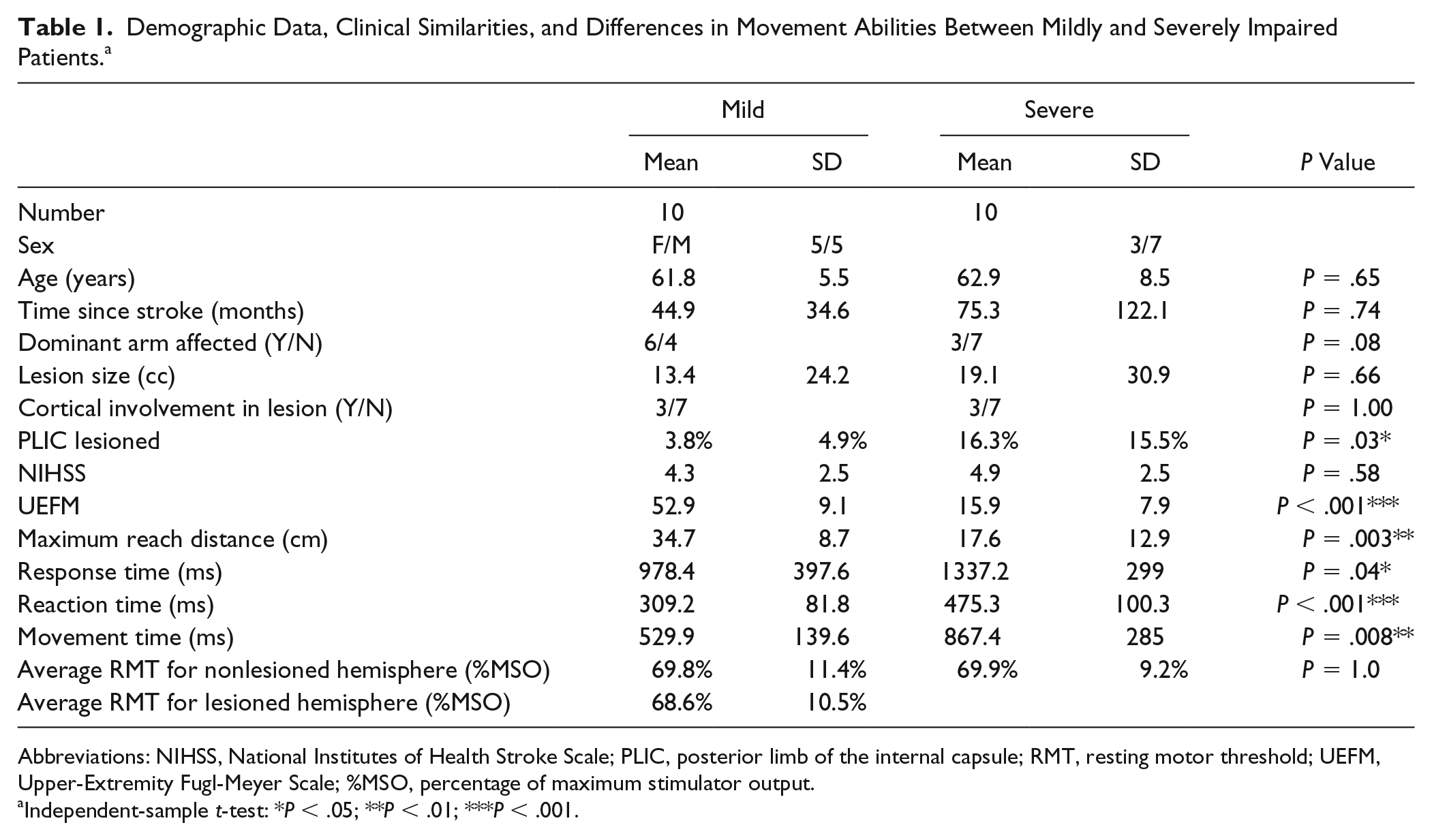
Abbreviations: NIHSS, National Institutes of Health Stroke Scale; PLIC, posterior limb of the internal capsule; RMT, resting motor threshold; UEFM, Upper-Extremity Fugl-Meyer Scale; %MSO, percentage of maximum stimulator output.
Independent-sample t-test: *P < .05; **P < .01; ***P < .001.
The order in which the cortical sites were targeted was counterbalanced across individuals, with each cortical site targeted in a separate test session. For each session, the reaching task was performed in 10 blocks of 10 trials each. Each block of trials included the following randomly presented conditions: reaching with no TMS delivered, reaching with TMS perturbation, and “catch” trials. TMS perturbation consisted of double-pulse TMS (DP-TMS; 25 ms interstimulus interval) delivered 150, 200, or 250 ms after the “Go” signal, at an intensity of 120% RMT. 33 This stimulation paradigm introduces a momentary disruption of neural processing in the underlying area. If that processing is relevant to task performance, a change in performance (eg, slowing) should be observed when compared with the unperturbed (no TMS) trials. The 25-ms interstimulus interval has been shown to perturb choice reaction time tasks when applied to PMd in previous studies. 34 In “catch” trials, no “Go” cue appeared, but TMS was delivered. This served as a performance control to ensure that patients attended to and reacted to the “Go” cue and not to the sound or sensation of the TMS. Each block of 10 trials included 6 trials with TMS perturbation (2 at each timepoint), 3 trials with no TMS perturbation, and 1 “catch” trial. Average response time was calculated at the end of each block of trials to monitor patient fatigue. Rest breaks were given per patient request or if average response time worsened by more than 25% between blocks.
Data Analysis
The timing of the TMS perturbation (150, 200, or 250 ms after the “Go” cue) that was closest to 50% of the individual’s reaction time (time point chosen) was used for the subsequent analysis for each participant and each session. Not surprisingly, patients in the mild group had shorter reaction times (Table 1). Therefore, the perturbation time points occurred at a higher percentage of total reaction time (on average, 150, 200, and 250 ms after the “Go” cue corresponded to approximately 46%, 61%, and 76% of reaction time, respectively, in the mild group). Patients in the severe group had longer reaction times and, thus, the perturbation time points occurred at a lower percentage of their total reaction time (150-, 200-, and 250-ms time points corresponded to approximately 32%, 42%, and 52% of reaction time, respectively, in the severe group). The time point closest to 50% of individual reaction time represented the point of maximal overlap between the 2 groups, and previous studies have demonstrated important shifts in motor-related physiology at this time point. 35
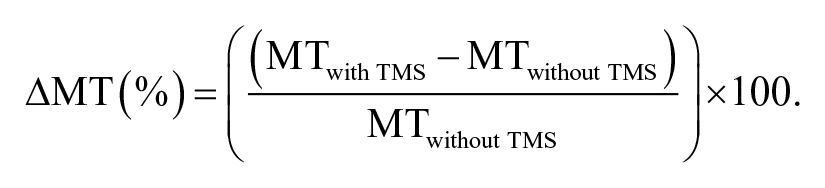
Response time was calculated as the time elapsed between the appearance of the “Go” cue and the first recorded contact with the response pad. Response time is subdivided into reaction time and movement time (MT) based on the time of movement initiation. Movement initiation was identified as the time point at which the 3D wrist position value exceeded the baseline value by >3 SDs and >3 mm for at least 100 ms. Reaction time was defined as the time between the “Go” cue and movement initiation, and MT as the time between movement initiation and target contact. The primary outcome measure was the difference in MT (ΔMT) between trials with and without TMS perturbation, as defined by the following equation:
Dependent measures were computed using a custom program written in MATLAB (MathWorks, Natick, MA).
Lesion Mapping
Stroke lesion masks were drawn on each MRI slice 36 using the ROI toolbox in MRIcro (MRIcron; Columbia, SC). Lesion masks and whole-brain images were oriented and normalized into Montreal Neurological Institute (MNI) standard stereotaxic space using FSL (FMRIB Software Laboratory). 37 FSL procedures were adapted from the procedure developed by Riley et al. 8 The BET brain extraction tool was used to remove the skull and other nonbrain tissue from the images (fractional intensity threshold set to 0.035). 38 The brain-only images were aligned with the MNI152 T1 1mm brain and the FLIRT transformation was applied to the lesion map (FMRIB’s Linear Image Registration Tool).39,40 Lesion masks were all flipped to the left hemisphere and overlaid on the Johns Hopkins University White Matter template (1 mm slice thickness)41,42 to estimate the amount of PLIC involvement and on the left hemisphere population-based atlas of the human pyramidal tract to determine the amount of pyramidal tract involvement. 43 Lesion overlap images for the mild and severely impaired groups overlaid on the pyramidal tract atlas are shown in Figure 1.
Statistics
For hypothesis testing, after confirming homogeneity of variance, we used separate 2 × 2 mixed-model ANOVAs [within-subjects factor: perturbation Site (M1, PMd); between-subjects factor: Group (mild, severe)] for perturbation of the lesioned and nonlesioned hemispheres. Planned pairwise comparisons included a within-group comparison (using Student’s paired t-test) of M1 versus PMd perturbation, and between-group comparison (using independent t-tests) of perturbation effects at M1 and PMd. For descriptive purposes and future hypothesis development, Student’s 1-sample t-tests were used to identify which of the perturbation conditions induced a measurable effect on reaching (the test value for ΔMT was set to zero to identify whether the test condition induced a change in MT that was significantly different from zero). All statistical analyses were performed using IBM SPSS Statistics Version 24 (Chicago, IL) and the α-level was set to .05.
Results
A total of 20 patients, aged 51 to 78 years (62 ± 7 years), were included in this analysis: 8 female and 12 male, who were an average of 60.1 (6-401) months poststroke. Motor impairment groups (“mild” and “severe”) were matched in age, sex, time since stroke, lesion size, amount of pyramidal tract involvement, and overall stroke-induced impairment. Groups differed only in their level of arm impairment and amount of descending tract damaged, as reflected by the significant between-group difference in upper-extremity Fugl-Meyer score and percentage of PLIC lesioned. As expected, the groups also differed in reaching performance of the affected arm, including maximum voluntary reaching distance, total response time, reaction time, and MT (Table 1).
When TMS perturbation induces a difference in the motor behavior as compared with trials without TMS perturbation, it can be inferred that the perturbation site (and/or the network to which it projects) has a functionally relevant role in the reaching movement. Under some conditions, ΔMT in trials with versus without TMS perturbation was positive, indicating that the TMS perturbation induced longer (ie, slower) MTs when compared with trials without perturbation. On average, the largest effects on reaching performance were observed with perturbation of nonlesioned-hemisphere PMd in the severe group and lesioned-hemisphere M1 in the mild group.
TMS Perturbation of the Nonlesioned Hemisphere
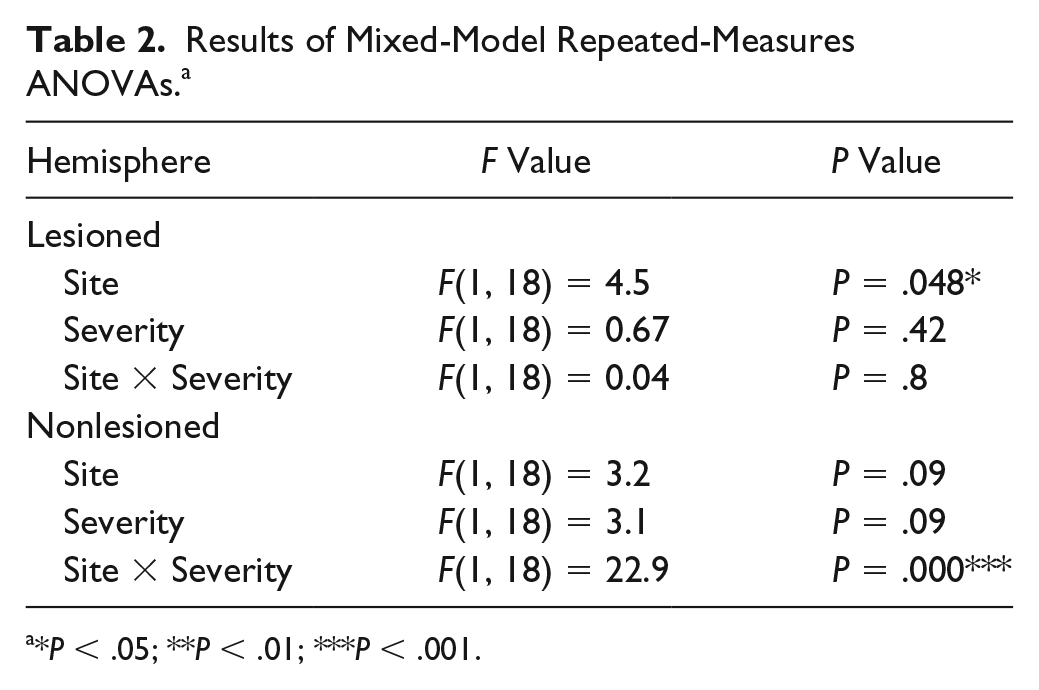
For the nonlesioned hemisphere, there was a significant interaction effect of Site × Group (P < .001) as well as a trend toward a main effect of Site and toward a main effect of Group that approached statistical significance (P = .09; Table 2, Figure 2). The significant interaction effect indicates a difference between how patients with mild versus severe arm impairment were affected by perturbation of the 2 different nonlesioned hemisphere sites. Pairwise comparisons revealed a significantly greater effect on MT with perturbation of PMd than M1 in the severe group [t(9) = 3.8; P < .005] and a significantly greater effect of M1 than PMd in the mild group [t(9) = −2.9; P > .05]. In addition, the effect of PMd perturbation was significantly greater in the severe group than in the mild group [t(18) = −3.5; P < .005], but the effect of M1 perturbation was not significantly different between the 2 groups [t(18) = 1.7, P = .1; Table 3; Figure 2].
Results of Mixed-Model Repeated-Measures ANOVAs. a
*P
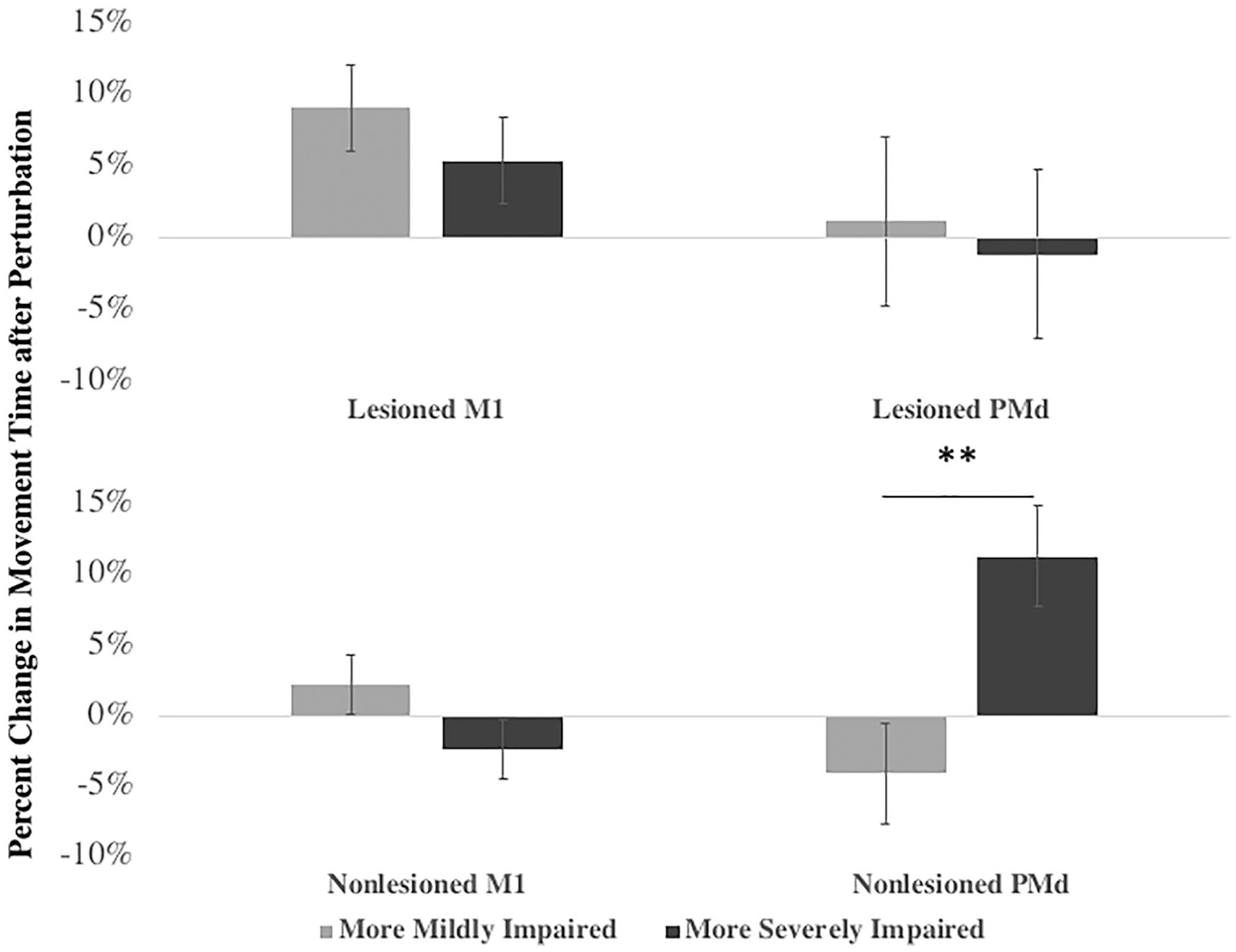
Difference in movement time induced by TMS perturbation of different sites (M1 vs PMd) within the nonlesioned and lesioned hemispheres for stroke patients with mild versus severe upper-extremity motor impairment. Percentage change in movement time of the affected arm with perturbation of lesioned (ie, contralateral) hemisphere M1 and PMd (top row) and nonlesioned (ie, ipsilateral) hemisphere M1 and PMd (bottom row). Only nonlesioned PMd differed between the 2 groups. Lesioned M1: t(18) = 0.6, P = .6; lesioned PMd: t(18) = 0.9, P = .4; nonlesioned M1: t(18) = 1.7, P = .1; nonlesioned PMd: t(18) = −3.5, P = .003**. Results of RMANOVA are given in Table 2 and Student t-test in Table 3.a
Change in Movement Time (%) After Perturbation of All Sites for Each Group. a
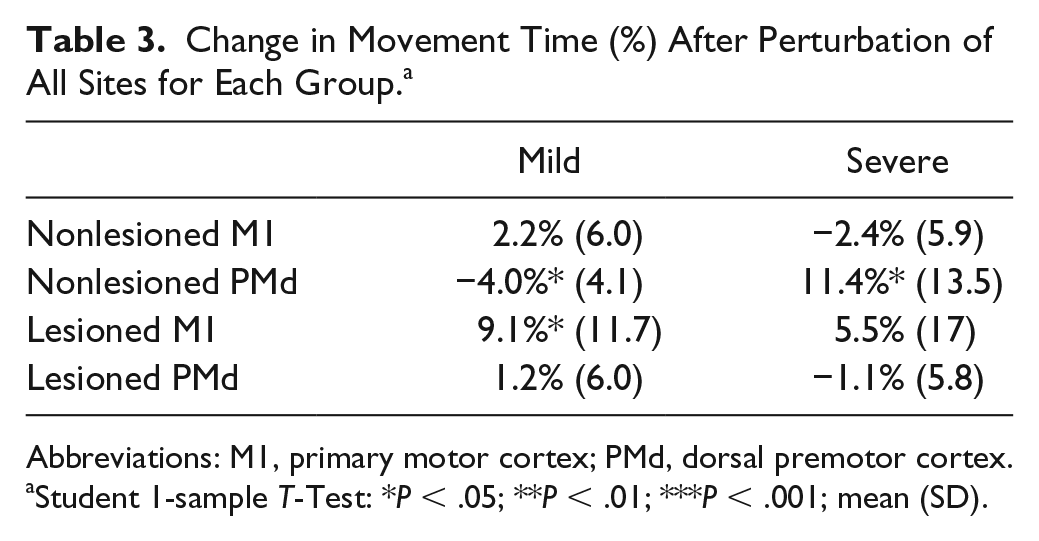
Abbreviations: M1, primary motor cortex; PMd, dorsal premotor cortex.
Student 1-sample T-Test: *P < .05; **P < .01; ***P < .001; mean (SD).
TMS Perturbation of the Lesioned Hemisphere
For TMS perturbation of the lesioned hemisphere, there was no significant interaction effect of Site × Group and no significant main effect of group, though there is a significant effect of Site (Table 2, Figure 2). Results of the planned comparisons showed that, within the mild group, perturbation of M1 had a greater effect on MT than PMd [t(9) = 2.5; P = .03]; however, no such trend was evident in the severe group [t(9) = 1.1; P = .3]. There were no significant differences between the mild and severe groups for perturbation of either M1 or PMd of the lesioned hemisphere.
Secondary Analysis
The exploratory analysis of the individual perturbation conditions (Table 3) revealed which perturbation conditions induced a significant effect on movement. In the severe group, nonlesioned hemisphere perturbation induced a change in MT (ie, positive ΔMT) when PMd was targeted [t(9) = 2.7; P = .03] but not when M1 was targeted. In the mild group, perturbation of the nonlesioned hemisphere PMd induced a small but significant negative ΔMT [t(9) = −3.1; P = .01]. Perturbation of lesioned-hemisphere sites in the severe group had no significant effect on reaching performance. In the mild group, however, perturbation of the lesioned hemisphere M1 induced positive ΔMT [t(9) = 2.5; P = .04].
Discussion
The purpose of this investigation was to identify differences between the contributions of primary and secondary motor regions of the lesioned and nonlesioned hemispheres to reaching performance in patients with mild and severe impairment. Importantly, in this study, the task paradigm did not require hand function, the severe group included patients who completely lacked hand function, and different sites within the same hemisphere were directly compared. In support of the hypothesis, the results indicated that the nonlesioned hemisphere has a site-specific role for reaching movements in patients with severe impairment, but not in patients with mild impairment. Specifically, we observed a significantly larger role of nonlesioned hemisphere PMd than M1 in patients with severe, but not mild, impairment.
These results demonstrate that the role of nonlesioned hemisphere PMd in ipsilateral arm movements is both compensatory (ie, adaptive) and site specific. For decades, functional brain imaging studies have shown that, during movement of the affected hand, lateralization of cortical activity to the lesioned hemisphere is positively correlated with motor recovery and function.12-14 In patients with more activation of nonlesioned hemisphere and poorer motor function, the nonlesioned hemisphere activation is often strongest in the area of PMd, leading some to suggest that this activity may serve a compensatory function.14,19 Some NIBS studies have provided additional evidence for this possibility.45,46 In the present study, we directly perturbed activity in this area and compared the effect to that of perturbing the neighboring M1 area.
Interestingly, in the mild group, there was actually a small decrease in MT (ie, faster movement) with perturbation of the nonlesioned hemisphere PMd. This was not an expected finding but may represent the disruption of a system that normally inhibits the area(s) that support reaching in this group (perhaps located primarily in the lesioned hemisphere). The interhemispheric interactions that occur in the context of reaching movements, and possible differences between patients with different levels of CST disruption, is an important area for future study. The largest effect on reaching in the mild group, however, occurred with perturbation of the lesioned hemisphere M1, as expected.
Nonlesioned Hemisphere PMd Versus M1
Based on differences in connectivity and task-related activation patterns, nonlesioned hemisphere PMd may indeed be better suited than M1 to partially compensate for severe damage of the contralateral CST by providing cortical input to the affected arm. Single-unit recordings in nonhuman primates have shown that there is considerably more ipsilateral (and bilateral) activation in PMd than M1 during unilateral reaching. 47 Direct projections from PMd to brainstem regions associated with arm movements are one plausible means by which PMd could influence the ipsilateral arm.48-50 In contrast, M1 has strong and relatively direct corticospinal projections to the distal aspect of the contralateral (unaffected) arm but considerably weaker projections to the proximal aspect of the ipsilateral (affected) arm. With its bilateral activation during reaching and bilateral projections to the upper extremity, PMd may be biologically better suited to control of the ipsilateral arm than M1.
There is also behavioral evidence that PMd can contribute to ipsilateral upper-extremity movements in a compensatory manner. For example, in healthy volunteers performing an action selection task, suppressing activity in contralateral PMd had no measurable effect on task performance. However, ipsilateral PMd activity was shown to have increased. When this ipsilateral PMd activity was momentarily perturbed with DP-TMS, errors in task performance were induced, demonstrating the functional relevance of the newly emerged activity. 20 Thus, PMd ipsilateral to the moving hand flexibly compensated when activity in its contralateral counterpart was suppressed. There is evidence that nonlesioned PMd could contribute in a compensatory manner to hand and finger movement tasks in patients with stroke as well.15,19 The present results extend this possibility to reaching movements in patients who are unable to perform hand and finger movements.
Implications for Noninvasive Brain Stimulation (NIBS)
Initial work in the area of NIBS to enhance stroke rehabilitation has been guided by the principle of interhemispheric competition or the idea that a loss of inhibitory input from the lesioned hemisphere results in overactivation of the nonlesioned hemisphere, which further suppresses activity in the lesioned hemisphere. 51 Many NIBS studies were, therefore, designed to increase excitation of the lesioned hemisphere and/or decrease activation of the nonlesioned hemisphere. The results of these studies have been mixed and often dependent on impairment severity and/or ipsilesional CST integrity.52,53 This observation led Di Pino et al 54 to suggest a Bimodal Balance Recovery Model in which the role of the nonlesioned hemisphere varies in a bimodal distribution along a spectrum of “structural reserve.” According to this model, patients with the most structural reserve are subject to the effects of interhemispheric inhibition, and the classical stimulation paradigms would likely be most effective. But in patients with low structural reserve, facilitation of nonlesioned hemisphere areas may be the most helpful in recovering function. CST integrity has been suggested as a measure of structural reserve as well as a biomarker for stroke outcome.8,55-58
In addition to structural reserve, we have suggested that selection of targets for neuromodulation should be both site specific (vs whole-hemisphere facilitation or inhibition) and dependent on the type of motor task that is the target for rehabilitation. 59 In the present study, we demonstrated how different brain areas (“sites”) within the same hemisphere can contribute to movement differently depending on the structural integrity of the CST from the lesioned hemisphere. The results suggest, therefore, that it is less a question of which hemisphere should be facilitated or inhibited than which specific cortical region should be targeted.
The other important consideration is the specific motor task being studied. Much of what we currently understand about mechanisms of poststroke motor recovery is limited to recovery of tasks that require finger movements. Yet finger movements are controlled differently from proximal-arm reaching and aiming movements. Control of finger movement is dependent on relatively direct CST projections from the contralateral M1, 21 whereas projections to the more proximal muscles of the shoulder and elbow are more oligosynaptic and diverse in their origins and include inputs from both hemispheres. 60 The reaching task itself is controlled by a unique network that is overlapping with but distinct from the network that controls grasp61-63 and includes cortical areas involved in visually guided aiming movements and the coordination of the shoulder and elbow joints.64,65 Therefore, the best sites to target with neuromodulation to improve reaching movements may differ from those to improve finger movements.
The issue of task specificity connects back to the issue of structural reserve. Following stroke, if there is partial sparing of projections from the lesioned hemisphere, reorganization within the lesioned hemisphere could improve control of the affected hand and arm. When the damage is more severe, however, with little or no sparing of projections from the lesioned hemisphere, there may be limited substrate to support distal arm recovery. In the present study, for example, there was significantly greater lesion overlap with the PLIC in the severe than the mild group. But, as we and others have shown, proximal arm recovery can still occur even in this more severely impaired population.66-68 The current results suggest that this may occur via enhanced input from pathways originating in the nonlesioned hemisphere that can influence the ipsilateral arm.
Study Limitations
The present findings were observed in the context of a reaching task. In finger movement tasks, previous studies have demonstrated a correlation between nonlesioned hemisphere activation and impairment severity that suggests an adaptive role for the nonlesioned hemisphere.15,19 Such observations, by definition, were made in a group of patients who had recovered some finger function (similar to the “mild” group in the present study). In contrast, we did not show a role of nonlesioned hemisphere PMd in reaching movements of patients with partially recovered finger function. This does not, however, preclude a possible role in hand and finger tasks, a distinction that bears further investigation.
Because of the technical challenges of performing functional brain imaging during reaching movements, we were not able to use recorded task-related activation to individually identify the target sites. Instead, we used evoked potentials (for M1) and previously published anatomical guidelines to identify the sites of interest. This approach was also limited, however, when it was not possible to induce MEPs from the lesioned hemisphere M1 in patients with more severe impairment. For these patients, we identified the lesioned M1 site by using the mirror location of the nonlesioned M1 site. Also, although “on-line” DP-TMS disruption is temporally precise, it must be remembered that it is perturbing a region within a network. An effect of DP-TMS on the subsequent movement is not only indicative of the role of the targeted region, but also reflects the roles of areas to which it is closely connected. Network effects were partially controlled by perturbing 2 sites within each hemisphere that exist within the same motor network for reaching. We also controlled for the nonspecific effects of TMS by targeting multiple sites and including “catch” trials to encourage attention to the “Go” cue and discourage responsivity to the TMS alone. Additionally, stroke patients, particularly those with more severe impairment, can have highly variable motor performance. We attempted to address this by offering frequent rest periods and randomly intermixing the experimental conditions within each block of trials such that perturbation trials could be compared with randomly interspersed no-perturbation trials.
Conclusions
In patients who lack voluntary finger movement because of the severity of CST damage, nonlesioned hemisphere PMd can have a functionally relevant role in the performance of reaching movements with the affected arm. This effect is not observed in the nonlesioned hemisphere M1 or in patients who have at least partial function of the stroke-affected corticospinal system. Thus, the neural networks that can support a particular motor function poststroke can differ depending on the severity of motor impairment and likely also the type of motor task in question.
Footnotes
Acknowledgements
We wish to thank all the stroke survivors who volunteered for and participated in this study. We also wish to thank Dijo Abraham, Dr Sambit Mohapatra, Dr Seth Kiser, Dr Shashwati Geed, Khadija Zaidi, Amanda Brouillard, Linda Azab, Sophia Brumme, Pallavi Samudrala, and Kansas Essington for assistance with data collection. We would like to thank Khadija Zaidi and Juby Mathews for help with data organization and analysis. We would like to thank Rahsaan Holley, Kathaleen Brady, Harsh Thakkar, Dr Elizabeth Lacey, Inger Ljungberg, and the MedStar National Rehabilitation Hospital research, inpatient therapy, and outpatient therapy departments for help with recruitment and support of our project. We also wish to thank Drs Michael Harris-Love, Elissa Newport, George Wittenberg, Guinevere Eden, and the Georgetown Center for Brain Plasticity and Recovery for their help in shaping the project and analysis. We thank Dr C. Nikki Arrington for her helpful edits. We acknowledge the following: NIH K01 HD060886 (PI: MLH-L); NIH T32 NS041231 (PI: K Gale); AHA 14GRNT20460001 (PI: MLH-L); AHA 15PRE24920006 (PI: RMH); NIH R21 5R21HD090516; SCANR: NINDS StrokeNet 1U10 NS086513 (PI: AWD).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NIH K01 HD060886 (PI: MLH-L); NIH T32 NS041231 (RMH, PI: K Gale); AHA 14GRNT20460001 (PI: MLH-L); AHA 15PRE24920006 (PI: RMH); NIH R21 5R21HD090516 (PI: MLH-L); SCANR: NINDS StrokeNet 1U10 NS086513 (PI: AWD).
