Abstract
Background. Stroke patients present restriction of mobility in the acute phase, and the use of a simple and specific scale can be useful to guide rehabilitation. Objective. To validate and propose a Hospital Mobility Scale (HMS) for ischemic stroke patients as well as to evaluate the HMS as a prognostic indicator. Methods. This study was performed in 2 phases: in the first, we developed the HMS content, and in the second, we defined its score and evaluated its psychometric properties. We performed a longitudinal prospective study consisting of 2 cohorts (derivation and validation cohorts). The data were collected in a stroke unit, and the following scales were applied during hospitalization: National Institutes of Health Stroke Scale to quantify stroke severity and the HMS to verify the degree of mobility. The primary outcome was the proportion of unfavorable functional outcomes, defined as a modified Barthel Index of <95. Results. We defined 3 tasks for HMS: sitting, standing, and gait. In the derivation cohort, the HMS presented an accuracy of 84.5% measured using the area under the receiver operating characteristic curve (95% CI = 78.3-90.7; P < .001), whereas in the validation cohort the accuracy was 87.8% (95% CI = 81.9%-93.7%; P < .001). The HMS presented a large standardized effect size (1.41) and excellent interexaminer agreement (intraclass correlation coefficient = 0.962; 95% CI = 0.917-0.983; P < .001). Conclusion. The HMS was able to predict accurately the functional outcome of poststroke patients, presented excellent interexaminer agreement, and was sensitive in detecting changes.
Introduction
Mobility restriction during the acute phase of a stroke can cause complications such as respiratory infections, deep venous thrombosis, and pressure ulcers. 1 These complications increase mortality, length of hospitalization, and treatment cost, 2 besides affecting the functional outcome after hospital discharge. 3
Studies show that early rehabilitation prevents complications and improves the patients’ functional recovery and their quality of life after stroke,4,5 and the use of an adequate assessment instrument can help direct physical therapy procedures aimed toward a higher mobility level. During the past decades, scales were created specifically to evaluate mobility, such as the Mobility Scale for Acute Stroke for poststroke patients and the Elderly Mobility Scale, already validated for this population.6,7
The Mobility Scale for Acute Stroke is a simple instrument, but its tasks require a standardized environment, which may be logistically challenging in low-resource settings. In this scale, the assessment of sitting balance should be done with legs flexed at right angles to thighs, and feet supported on a stool/floor at right angles to the legs. The bed height should be adjustable or a footstool can be used. This scale also needs a standardized chair (height 43 cm) to evaluate the transfer from sitting to standing. 6 This standardization seems quite simple, but in a low-resource service, such as the hospital where the present study was conducted, it would be difficult to include this scale in the patient’s care routine because such equipment is not available for all patients. The Elderly Mobility Scale presents a greater number of tasks that include balance evaluations such as functional reach, which increases the complexity of its application. 7
The mobility scales specially targeting patients in intensive care units (ICUs), such as ICU Mobility Scale, Functional Status Score for the ICU, and Johns Hopkins Highest Level of Mobility Scale8-10 are also simple but were not specifically designed for stroke patients and do not consider their particularities. These scales do not specify the need for assistance of 1 or 2 people to perform the tasks, an important aspect to be considered when assessing stroke patients.
A simple and practical instrument, which takes into consideration the peculiarities of the hospital environment and the specificities of poststroke patients, could offer a positive impact on rehabilitation, 11 besides helping to define the prognosis, and allow both patients and family members to understand the challenges they will face after hospital discharge.11,12 The Mobility Scale for Acute Stroke was able to predict the length of stay in hospitals and the destination after discharge, but we found no studies that evaluated this or other mobility scales as functional capacity predictors in stroke patients.11,13
Aims
This study aims to validate and propose a new Hospital Mobility Scale (HMS) that identifies the level of mobility of poststroke patients specifically in a hospital environment as well as to evaluate the HMS as a prognostic indicator.
Methods
This trial was performed in 2 phases: in the first phase, we developed the HMS content, and in the second phase, we defined its score and evaluated its psychometric properties.
Phase 1: Development of the Content of HMS
The choice of the initial items for this scale was made by searching instruments used to assess the mobility of poststroke patients in the literature. We excluded tasks that are difficult to evaluate in a hospital environment, such as climbing stairs or ramps, or that cannot be executed by aphasic patients or those with other major cognitive deficits. Tasks that demand a standardized environment or equipment that may not be found in low-resource hospitals were also excluded. Physical therapists used the first version of the scale for 6 months in a stroke unit, in the city of Salvador, Bahia. After this period, a panel of 10 physical therapists, including the physical therapists who used the scale during this period of 6 months, university professors, and experts in neurology, performed the qualitative analysis of the scale, modifying terms and excluding items considered inadequate.
Phase 2: Definition of the Score and Assessment of the HMS Psychometric Properties
In the second phase of this research, we performed a prospective longitudinal study, including patients admitted in a stroke unit, in the city of Salvador, Bahia. We included patients older than 18 years diagnosed with ischemic stroke. Patients with a modified Barthel Index (mBI) <95 before stroke were excluded as well as patients who were previously blind, were amputees, or had a diagnosis of dementia or other neurological diseases that could affect mobility.
We applied the following scales: National Institutes of Health Stroke Scale (NIHSS) 14 to quantify stroke severity and the HMS to verify the degree of mobility. The NIHSS was scored on admission by certified researchers and the HMS on days 1 through 5. For analyses of the HMS predictive validity, we considered the best score within the first 5 days. We then extracted main clinical variables such as the presence of comorbidities, the location of the lesion, and etiological diagnosis into a standardized questionnaire with data collected from medical records. After 90 days, we applied the mBI 15 by telephone interviews. The primary outcome of the study was the proportion of unfavorable functional outcomes, defined as a mBI score <95. 16 Patients who died within the 90-day period were scored as achieving unfavorable outcomes. The mBI was chosen over the original Barthel Index for being more detailed in the functional capacity assessment. Shah et al 15 demonstrated that the mBI was more sensitive and reliable than the original version.
The participants were temporally divided into 2 cohorts with a 3-month follow-up: one cohort for HMS derivation (160 patients consecutively admitted between January and September 2015) and another cohort for HMS validation (160 patients consecutively admitted between October 2015 and April 2016).
Definition of the HMS Score
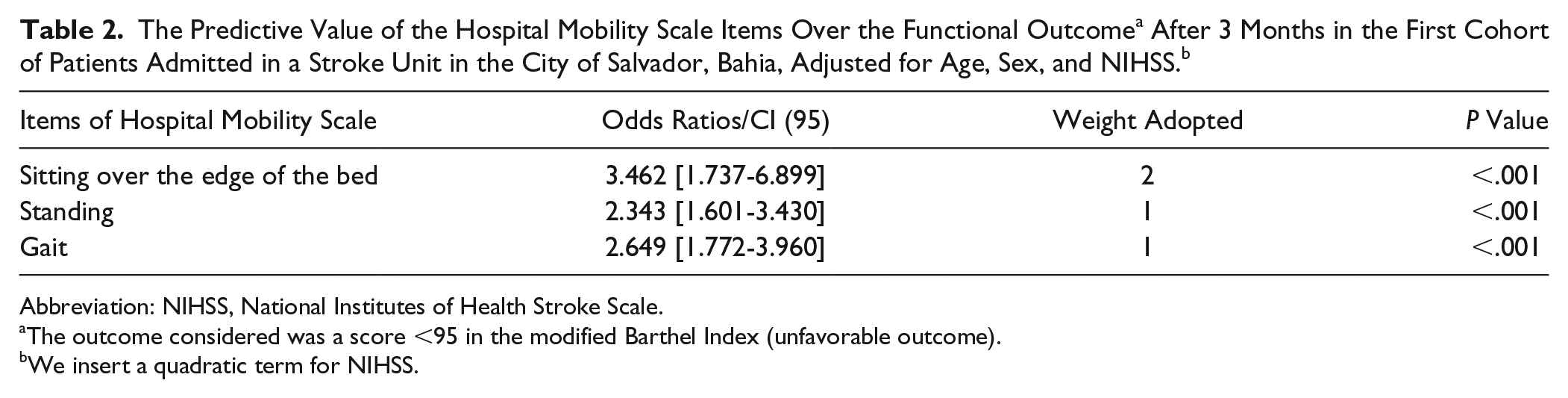
In the first cohort, we calculated the scores of the HMS items considering the predictive value of each item regarding the proportion of unfavorable functional outcomes. Using one model of binary logistic regression for each item of HMS, we found the odds ratios (ORs) and calculated the weight of each item considering the proportion between them. The values were rounded to facilitate the use of the score.
Assessment of the HMS Psychometric Properties
In the first and second cohorts, we analyzed the performance of this scale in predicting functional capacity after 3 months by constructing receiver operating characteristic (ROC) curves to assess accuracy, sensitivity, and specificity for each cutoff point of the HMS. We also evaluated whether the HMS is an independent predictor of functional capacity through multivariate logistic regression models. We evaluated the responsiveness of HMS using the standardized effect size, considering the changes observed between the first and fifth days of hospitalization. To check the interexaminer agreement regarding the HMS, a subgroup of patients was independently evaluated by 2 physical therapists on the same day, in opposite shifts, during the first 5 days of hospitalization.
Sample Calculation
We analyzed articles in the literature that study functional outcome predictors and show, in their results, up to 5 variables as independent predictors of functional capacity in stroke patients.12,17-19 Based on this information, we estimated that 5 variables would be considered as possible outcome predictors to be included in the logistic regression model along with the HMS. We calculated 10 patients for each variable, totaling 60 participants. Considering a previous study with the Brazilian population, in which approximately 40% of patients presented an unfavorable outcome 3 months after the stroke, 20 approximately 160 participants would be required to perform the binary logistic regression. We used 160 participants for the derivation cohort and 160 participants for a validation cohort, resulting in a total number of 320 patients.
Statistical Analysis
In multivariate logistic regression models, we chose covariates that were closely associated to the outcome (P < .2). Considering that HMS was evaluated as the main predictor variable in the multivariate model, we only included variables in the model that were actually confounding variables. The criterion used to consider the variable as confounder was to change the HMS ORs in more than 15%. We also included covariates that were biologically expected to affect the functional outcome (NIHSS, age, and sex).17,18,21 To verify the linearity assumption of the multivariate model, we tested a quadratic term for the continuous variables (age and NIHSS). Age showed a linear association, and therefore, there was no need to maintain the quadratic term for this variable. The NIHSS did not present a linear association with the outcome; thus, a quadratic term was maintained for the NIHSS in the final model. The squared NIHSS score was linearly related to the outcome. The results for the logistic regression model were presented as ORs and CIs.
To evaluate the HMS accuracy, sensitivity, and specificity, we analyzed the ROC curve using only HMS as a predictor, not considering other variables. The interexaminer agreement was checked using the intraclass correlation coefficient. To check the HMS responsiveness, we used the standardized effect size calculated as follows: the difference between the instrument’s average score in the first assessment (day 1) and the final assessment (day 5) divided by the baseline score standard deviation. This measure indicates the sensitivity in detecting changes in the patient over time. The standardized effect size was considered small (<0.50), moderate (0.50-0.79), and large (>0.8). Moderate and large effect sizes indicated sufficient responsiveness. 22
Ethical Aspects
This project was approved by the Research Ethics Committee of Bahiana School of Medicine and Public Health (Approval number CAAE: 27383014.9.0000.54), and all patients or legally authorized representatives provided written informed consent.
Results
Results From Phase I
After the HMS qualitative evaluation by a panel of 10 physical therapists, 3 basic mobility tasks frequently performed by hospitalized patients involving posture maintenance, transfers, and locomotion were included in the HMS. The items defined for the HMS were the following: sitting on the edge of the bed with the feet off the ground, standing, and gait. Each task was classified according to the level of dependence (performs independently, needs help from 1 person, needs help from 2 people, or fails to perform the task; Figure 1).
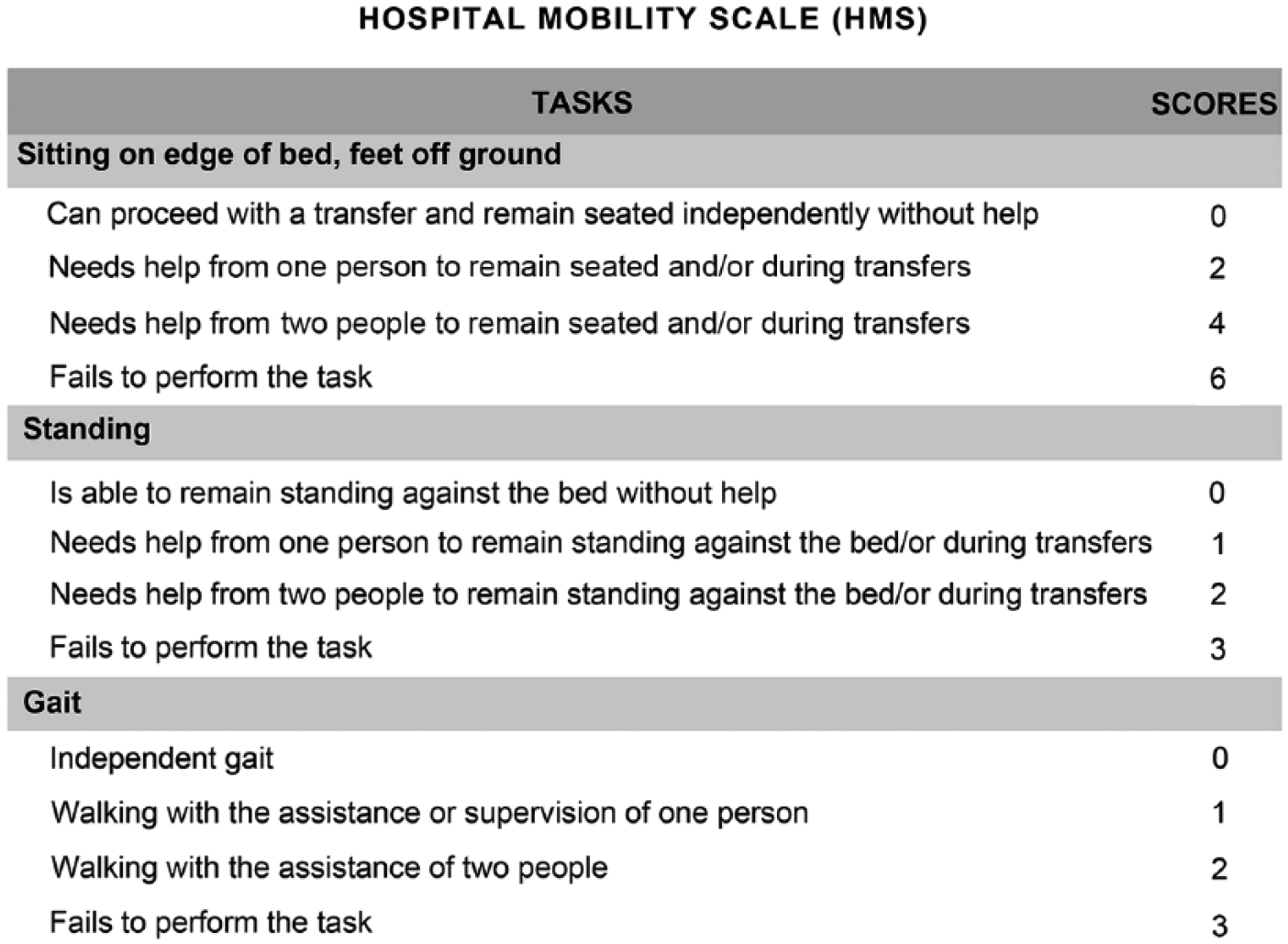
Hospital Mobility Scale.
Results From Phase II
Between January 2015 and April 2016, 447 patients were admitted in the stroke unit with ischemic stroke. Of these patients, 104 were excluded, and during follow-up, 23 patients who could not be reached by phone were lost to follow-up (Figure 2). It is noteworthy that no statistically significant differences were found between most clinical and sociodemographic characteristics of the patients who completed the follow-up and those considered lost to follow-up (Supplementary Table 1).
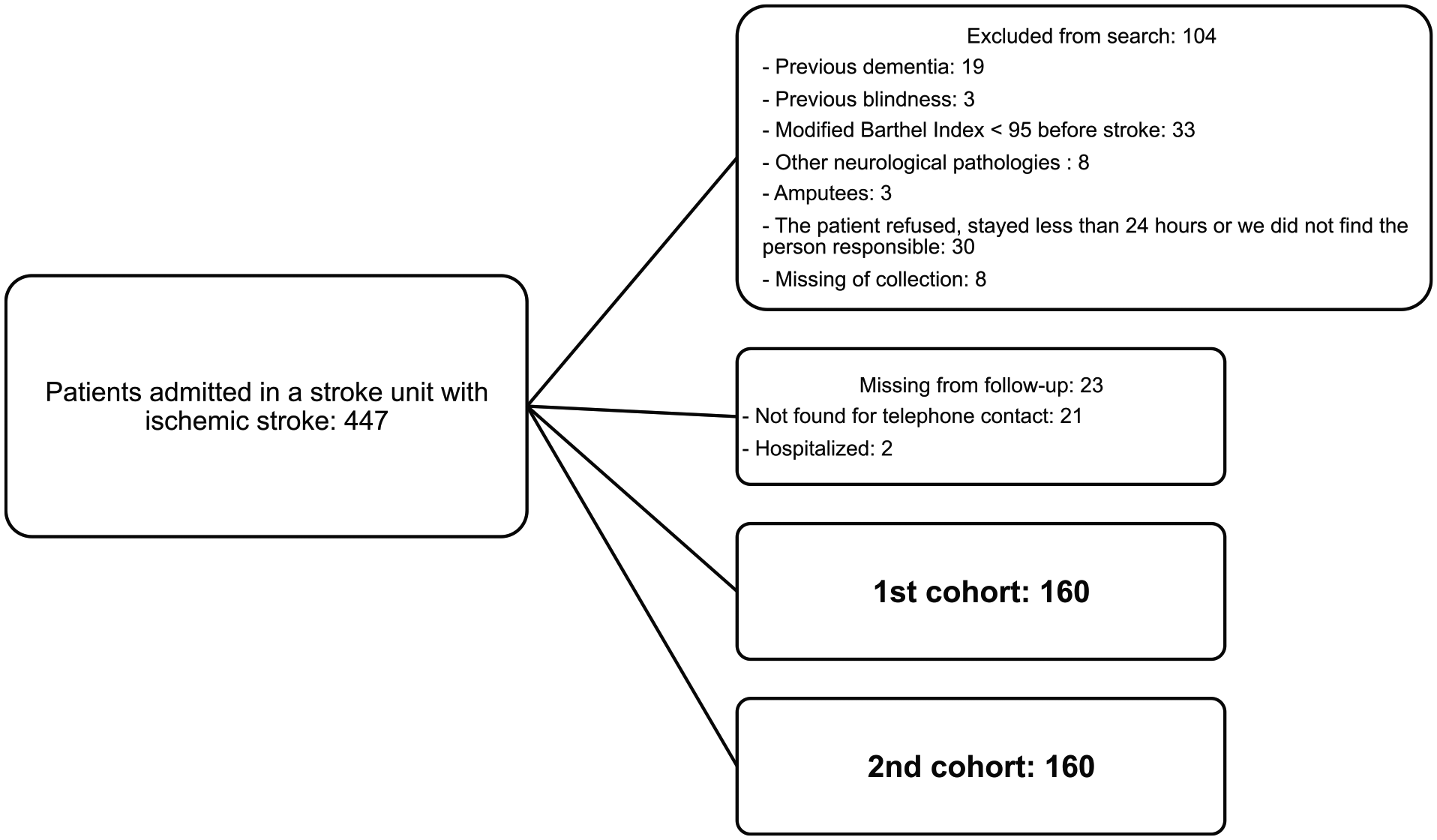
Flowchart of patients admitted between January 2015 and April 2016 at a stroke unit in the city of Salvador, Bahia.
Table 1 presents the clinical, demographic, and functional characteristics of the derivation and validation cohorts. We can observe that the validation cohort was younger, had more years of schooling, and a lower proportion of hypertension and diabetes.
Demographic, Clinical, and Functional Characteristics of 320 Stroke Patients Admitted in a Stroke Unit, in the City of Salvador, Bahia.
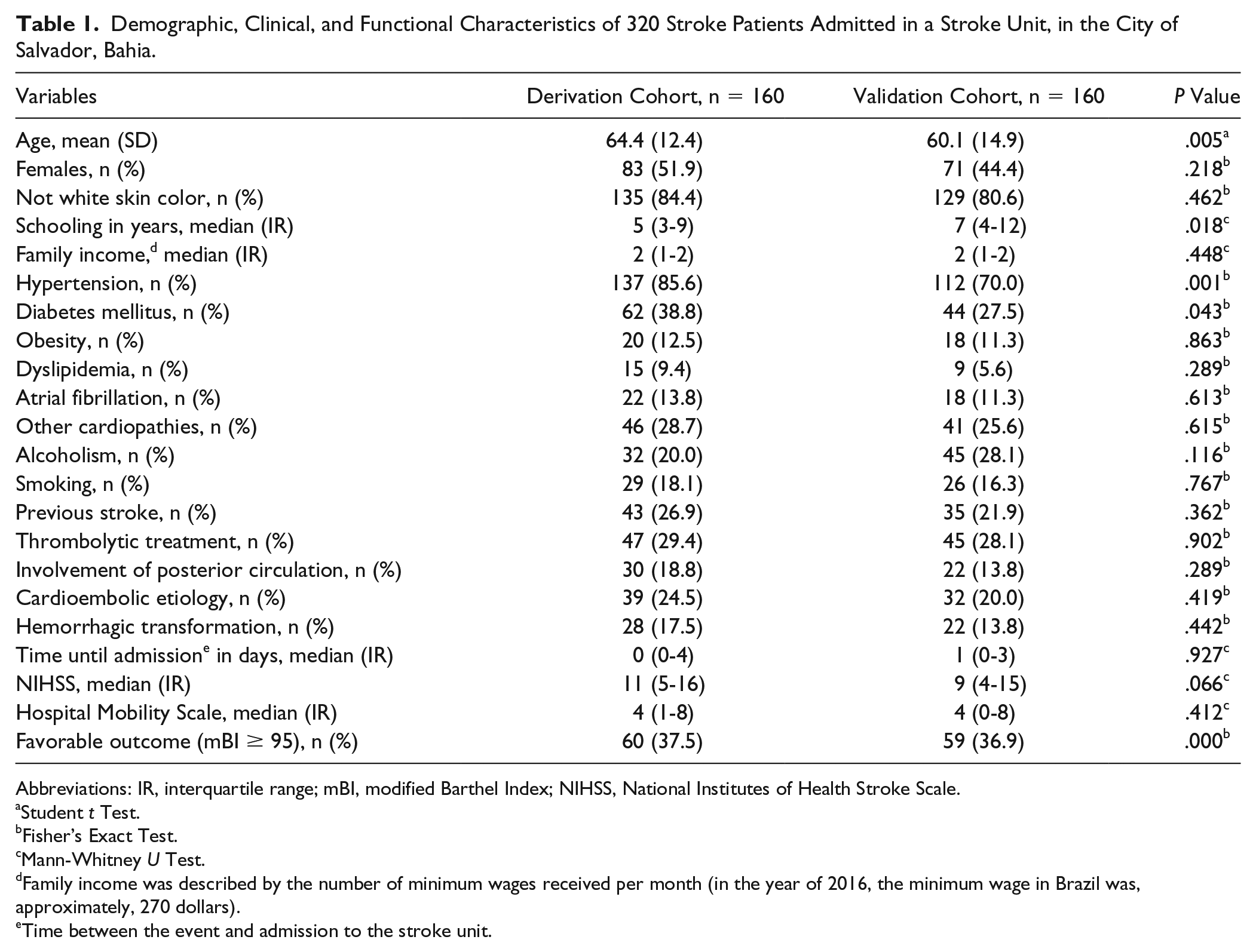
Abbreviations: IR, interquartile range; mBI, modified Barthel Index; NIHSS, National Institutes of Health Stroke Scale.
Student t Test.
Fisher’s Exact Test.
Mann-Whitney U Test.
Family income was described by the number of minimum wages received per month (in the year of 2016, the minimum wage in Brazil was, approximately, 270 dollars).
Time between the event and admission to the stroke unit.
Table 2 presents the results of the univariate logistic regression analysis used to calculate the HMS score. Considering that the OR for the “sitting” task was nearly 2-fold the OR for the “standing” and “gait” tasks, we adopted weight 2 for the sitting task and weight 1 for the standing and gait tasks (Table 2). Figure 1 shows the score developed for the HMS considering the weights found in the logistic regression analysis. The score for the sitting task was —0, 2, 4, and 6—, twice the scores for the standing and gait tasks, which were —0, 1, 2, and 3—. The total HMS score ranged from 0 to 12 and the higher the score the worse the mobility level (Figure 1).
Abbreviation: NIHSS, National Institutes of Health Stroke Scale.
The outcome considered was a score <95 in the modified Barthel Index (unfavorable outcome).
We insert a quadratic term for NIHSS.
After defining the HMS score, we performed a univariate analysis aiming to identify the variables associated with the derivation cohort outcomes and selected those variables for the multivariate model (Supplementary Table 2). The variables that presented statistical associations (P < .2) were included separately in a logistic regression model along with HMS to check if they modified the OR of this scale. However, only NIHSS modified the HMS OR by more than 15%.
In the multivariate logistic regression model, the HMS was an independent predictor of functional capacity (Table 3) when adjusted for age, sex, and NIHSS in the first cohort (OR = 1.379; 95% CI = 1.204-1.581; P < .001) and also in the second cohort (OR = 1.372; 95% CI = 1.168-1.611; P < .001). The ORs of 1.379 in the first cohort and 1.372 in the second cohort indicate that the 1-point increase in the HMS increases the odds of an unfavorable functional outcome for the patients by, respectively, 37.9% and 37.2%, after 3 months. Supplementary Figure 1 shows an increase in the odds of an unfavorable functional outcome associated with increasing HMS scores.
The Performance of Hospital Mobility Scale as a Functional Outcome a Predictor in the Derivation and Validation Cohort of Patients Admitted in a Stroke Unit in the City of Salvador, Bahia.
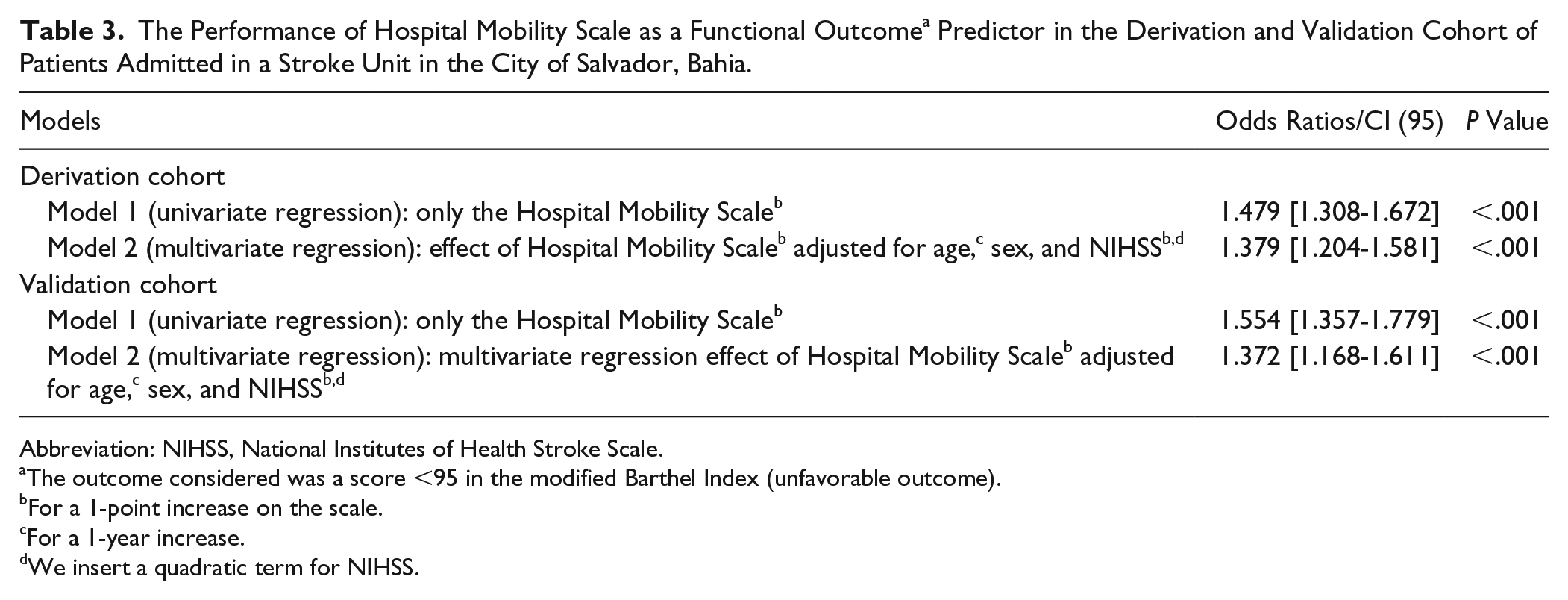
Abbreviation: NIHSS, National Institutes of Health Stroke Scale.
The outcome considered was a score <95 in the modified Barthel Index (unfavorable outcome).
For a 1-point increase on the scale.
For a 1-year increase.
We insert a quadratic term for NIHSS.
In the first cohort, there was no significant association between age (OR = 1.034; 95% CI = 0.997-1.073; P = .073), NIHSS (OR = 1.145; 95% CI = 0.873-1.502; P = .327), and sex (OR = 0.722; 95% CI = 0.316-1.645; P = .438), and the outcome in the multivariate analysis. In the second cohort, there was a significant association between age (OR = 1.073; 95% CI = 1.032-1.115; P < .001) and NIHSS (OR = 1.877; 95% CI = 1.338-2.633; P < .001), and the outcome in the multivariate analysis, which was not observed with the variable sex (OR = 0.541; 95% CI = 0.195-1.497; P = .237).
When analyzing the accuracy of HMS as a functional outcome predictor (Figure 3), considering the outcome using mBI <95 as unfavorable, in the derivation cohort, we found an accuracy of 84.5% measured using the area under the ROC curve (95% CI = 78.3-90.7; P < .001). In the validation cohort, we found an accuracy of 87.8% (95% CI = 81.9-93.7; P < .001). We analyzed the sensitivity, specificity, and likelihood ratios for each cutoff point of the HMS in the derivation and validation cohorts (Supplementary Table 3).
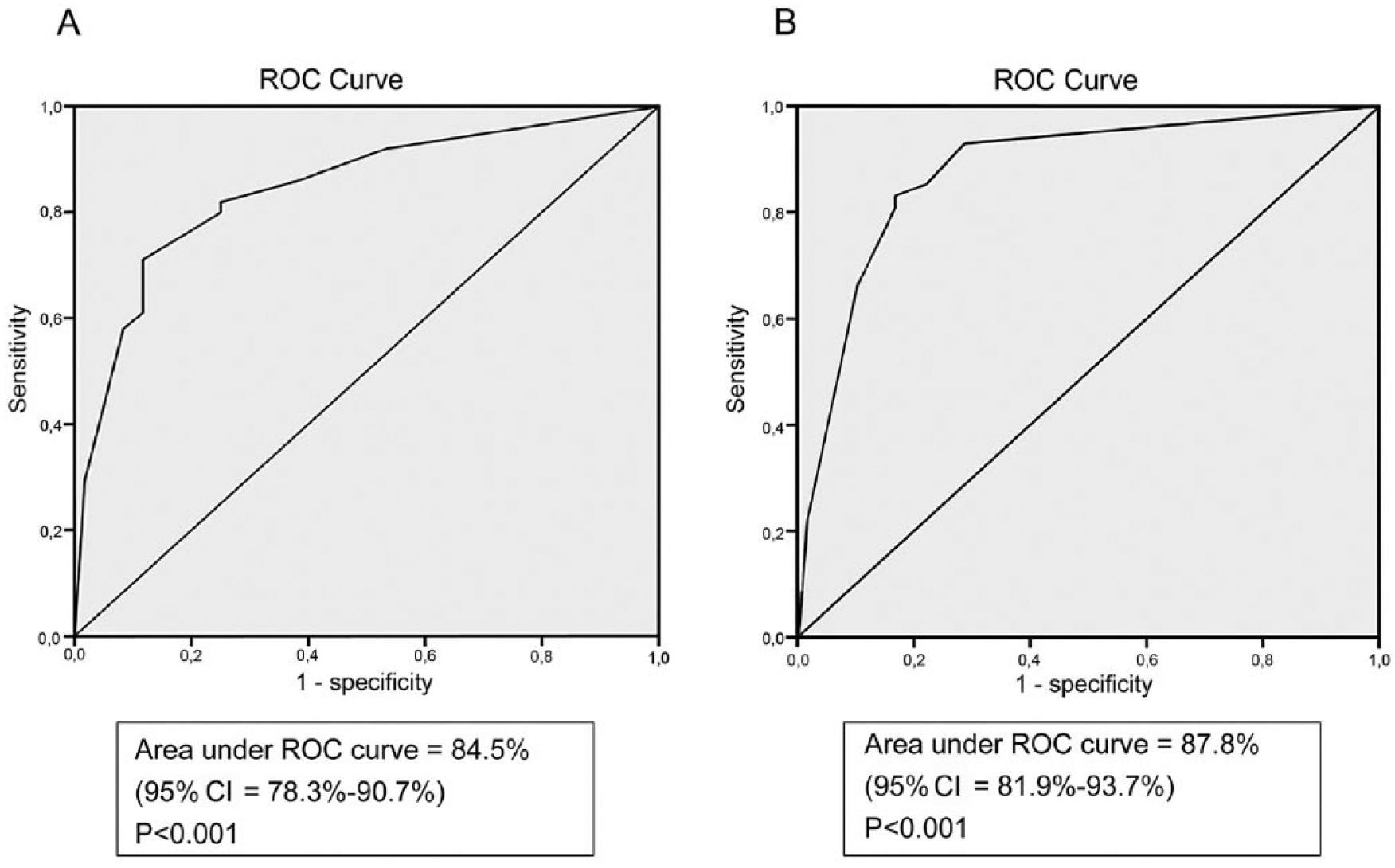
ROC curve for Hospital Mobility Scale as a functional outcome predictor after 3 months. The outcome considered was mBI <95 (unfavorable outcome). (A) Derivation cohort, (B) Validation cohort.
We checked the HMS accuracy through the ROC curve analysis when it was used on the first, second, third, fourth, and fifth days of hospital stay and when the best score found during the first 5 days was used. We observed better levels of accuracy when using the best score during the first 5 days (Supplementary Table 4).
When assessing the responsiveness, we observed that HMS presented a large standardized effect size of 1.41. HMS presented excellent interexaminer agreement, with an intraclass correlation coefficient of 0.962 (95% CI = 0.917-0.983; P < .001). To evaluate the responsiveness of HMS, we used data from 283 patients who remained hospitalized for at least 5 days in the stroke unit, and to evaluate the interexaminer HMS agreement, we used a subgroup of 25 patients.
Discussion
The results of this study demonstrate that HMS has an excellent interexaminer agreement, is responsive to changes during the acute phase, and is able to predict the functional outcome 3 months after the stroke. This new instrument is an option for the specific assessment of mobility, assisting in the definition of rehabilitation management and identifying the prognosis of hospitalized patients after a stroke.
Although there are already scales in the literature designed for the evaluation of mobility, the HMS stands out for being a more practical instrument: there is no need for equipment; understanding each item is simpler and objective; and it can be easily inserted into routine care. The daily record of the mobility level can be used as an indicator of the quality of physical therapy assistance in hospitals, indicating the change in function of the patients during hospitalization. 23
The HMS only evaluates 3 tasks, whereas the Mobility Scale for Acute Stroke evaluates 6 and the Elderly Mobility Scale evaluates 7 tasks.6,7 The smaller number of tasks makes the application more practical, facilitating its inclusion in the patient’s evaluation routine, especially in places where there are fewer human resources available and the time allocated for the care of each patient is shorter.
The scales that assess functional capacity as mBI include tasks that are difficult to evaluate in the hospital environment, such as walking up stairs and using the bathroom, because they are often restricted as a result of the use of probes, monitoring, venous access, or medical advice, especially in ICUs. 24 These scales are usually used after hospital discharge in studies with subacute and chronic patients. 16
Researchers have discussed functional outcome predictors in poststroke patients,12,17,18,21 presenting models that help plan the discharge and allow patients and family members to understand the challenges they will face. 12 We found no studies that evaluated mobility scales as functional capacity predictors in poststroke patients, but mobility scales have been shown to predict discharge destination and hospitalization time.11,13 When comparing HMS with some of the predictive models for functional capacity proposed in the literature that include clinical, demographic, and functional variables,12,17-19,21,25 we can observe that only 1 model, described by Ntaios et al, 25 presented a higher accuracy. This study used the ASTRAL score and found an accuracy of 0.937 in the derivation cohort, higher than that for HMS. However, in the validation cohort, the accuracy was 0.771 and lower than that for HMS. 25 It is worth noting that the ASTRAL score is composed of different variables (NIHSS, clinical, and sociodemographic variables), whereas the HMS reached high accuracy when used alone.
The strong association found between acute-phase mobility assessed by HMS and the functional outcome after hospital discharge can be justified by the presence of tasks involving postural control, such as the patient being able to remain seated, standing, and walking. Thus, mobility is an essential requirement for the proper performance of day-to-day activities, such as preparing food, dressing, and personal hygiene. 11
The responsiveness to change and interexaminer agreement are important features of assessment tools, especially when used in large clinical trials. 16 We observed that HMS presented a large standardized effect size between the first and fifth day, demonstrating the sensitivity to changes in this instrument. No studies were found to assess the standardized effect size of mobility scales in acute stroke patients, to allow a more objective comparison with HMS. Yu et al 26 evaluated the responsiveness of 2 scales that assess postural control in stroke patients after the acute phase—the Hierarchical Balance Short Forms and the Postural Assessment Scale for Stroke—finding a standardized effect size of 0.99 and 0.86, respectively, 26 lower than what we found for HMS. Because these scales for postural control evaluation involve more complex tasks than the HMS, 26 the patients may present less significant changes in their score over time. Similar to other mobility scales described in the literature,10,24,27 the HMS presented excellent interexaminer agreement.
The routine use of HMS in physical therapy care allows the professional to understand the patient’s mobility level before starting the procedure, which facilitates the planning of care procedures considering the level of mobility. The physical therapist can organize the environment to make mobilization easier and more appropriate, in addition to identifying the number of therapists appropriate to conduct the intervention. It is also possible to plan the sequence and moment for each procedure by directing care toward the highest level of mobility.
The use of HMS during hospitalization as a functional capacity predictor for after the patient’s hospital discharge helps the patients, their family, and health professionals to better understand what the patient’s reality will be when he or she goes back home. 12 This helps plan and organize the environment where the patient will live and the accessibility needed, and to know if the patient will be dependent or if he or she will need help in carrying out activities of daily living. 12
When we started to collect data, the mobility scales found in the literature were not yet translated and validated for the Brazilian population after stroke, and for that reason, it was not possible to include them in the study and compare them objectively with HMS. It would be interesting to compare HMS with other mobility scales, such as Mobility Scale for Acute Stroke 6 or Functional Status Score for the ICU, 10 in future studies.
This study included only conscious patients with ischemic stroke, so our results cannot be generalized to hemorrhagic stroke patients or those who present with more severe deficits. Both cohorts were independent, yet all patients were selected in the same unit. This means that an external validation of this scale, using other poststroke populations, is important.
Conclusion
The results of this study showed that the new HMS developed specifically for poststroke patients was valid and reliable for the evaluation of these patients during hospitalization. This instrument presented an excellent interexaminer agreement, was able to predict the functional outcome accurately, was sensitive in detecting changes in the acute phase, and could be useful in both clinical practice and scientific research.
Supplemental Material
Revised_supplementary_file_clean_copy – Supplemental material for A Simple Hospital Mobility Scale for Acute Ischemic Stroke Patients Predicts Long-term Functional Outcome
Supplemental material, Revised_supplementary_file_clean_copy for A Simple Hospital Mobility Scale for Acute Ischemic Stroke Patients Predicts Long-term Functional Outcome by Iara Maso, Elen Beatriz Pinto, Maiana Monteiro, Marina Makhoul, Tassiana Mendel, Pedro A. P. Jesus and Jamary Oliveira-Filho in Neurorehabilitation and Neural Repair
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.