Abstract
Background. Cognitive impairment is common in long-term survivors of out-of-hospital cardiac arrest (OHCA) but corresponding neuroimaging data are lacking. Objectives. This study explored the relationship among the cortical brain structure, cognitive performance, and clinical variables after OHCA. Methods. Three months after resuscitation, 13 OHCA survivors who had recovered from a coma to living independently and 19 healthy controls were assessed by cerebral magnetic resonance imaging and neuropsychological tests quantifying memory, fine-motor coordination, and attention/executive functions. Cortical thickness (Cth) and surface area (SA) were compared between groups and analyzed for relationships with cognitive performance as well as the clinical variables of coma duration and the time to return of spontaneous circulation (ROSC). All analyses were controlled for age and sex. Results. Analyses of SA revealed no significant differences. Compared with controls, survivors had significantly reduced memory and fine-motor coordination and significantly thinner cortex in large clusters in the frontal, parietal, and inferior temporal cortices, with additional regions in the left occipital lobe and the left temporal lobe. Widespread thinner cortical regions were significantly associated with decreased memory performance in survivors when compared with those in controls and were significantly associated with an increased time to ROSC and increased coma duration in the OHCA group. Increased coma duration, but not increased time to ROSC, was significantly correlated with cognitive test performance. Conclusions. The results suggest that widespread Cth reductions correspond to the cognitive impairments observed after OHCA. Neuroimaging studies of long-term OHCA survivors are warranted to guide the development of diagnostics and treatment options.
Introduction
Out-of-hospital cardiac arrest (OHCA) can cause hypoxic-ischemic brain injury. 1 OHCA can occur for a variety of reasons, but the most common cause is coronary artery disease.2,3 The vast majority of long-term survivors achieve good gross neurological outcomes and can return to independent living.3,4 However, residual cognitive impairments are common among these survivors.5-13 Cognitive impairments are associated with a reduction in both health-related quality of life and participation in society.10,11,14 Memory, attention, and executive functions, as well as fine-motor coordination are most frequently impaired. Most impairments are mild or moderate.3-11 An increased duration of coma after initial resuscitation5,6,11,14,15 and an increased duration of time to return of spontaneous circulation (ROSC) 16 have each been correlated with a reduced cognitive outcome; however, other studies do not support these conclusions.6,13,17
Few previous studies have addressed cognitive outcomes in combination with structural cerebral magnetic resonance imaging (MRI) analytic methods in long-term survivors of cardiac arrest.18-21 Therefore, little is known about the relationship between brain structure and cognition in this patient population. A possible reason is that many survivors have internal cardiac defibrillators (ICDs), which are not MRI safe. Another reason could be that OHCA survivors without overt neurological impairments are predominantly seen as cardiac patients in follow-up care, with less attention being paid to possible cognitive deficiencies.4,5
Recently, we published results from a cerebral MRI volumetric study showing that both reduced hippocampal volume and reduced global cortical volume were associated with decreased memory functioning in 13 OHCA survivors who had recovered from inpatient coma and returned to independent living within 3 months after resuscitation. 20 In another study, Horstmann et al 19 aimed to identify the precise regional pattern of gray matter atrophy post cardiac arrest using MRI and voxel-based morphometry. The 12 survivors they included were assessed with several memory tests and a clinical rating scale for apathy, 4 to 62 months after a cardiac arrest. When compared with healthy controls, these survivors had extensive reductions of gray matter volumes in the anterior, medial, and posterior cingulate cortex, the precuneus, the insular cortex, the posterior hippocampus, and the dorsomedial thalamus. The reductions in volume of these areas were significantly correlated with amnestic deficits and apathy.
Cerebral volumes are composite measures of cortical thickness (Cth) and surface area (SA). 22 To our knowledge, the relative contribution of Cth and SA to the observed reductions in cortical volumes after OHCA has not been previously investigated. In other groups of neurological patients, the use of cerebral MRI and surface-based analyses of the cortex have provided information on the spatial context of Cth and SA alterations, as well as precise estimations of the regional cortical anatomy related to specific indices of cognitive performances.23-27
Our aims in the present study were to examine the differences in both Cth and SA between the 13 survivors and the controls from our previous MRI volumetric study. 20 Furthermore, we explore the relationships among cortical parameters, cognitive performance, and clinical variables. The clinical variables assessed were the time to ROSC and coma duration. Additionally, we expanded the repertoire of cognitive measures from our previous volumetric study, to include tests measuring aspects of attention, executive functions, and fine-motor coordination, in addition to memory tests. We hypothesized that we would observe significant differences in the cortical anatomy of OHCA survivors and controls. Specifically, we hypothesized that compared with the controls, the OHCA group would show widespread regions with a thinner cortex and smaller SA. Furthermore, we hypothesized that those cortical regions with significantly reduced SA and/or Cth would be significantly related to poorer cognitive outcome and increased coma duration.
Methods
This was a nested case-control study from a single-center longitudinal prospective cohort study of cognitive outcomes 3 and 12 months after OHCA with a cardiac cause. Patients were recruited from the cardiac ward at the University Hospital of North Norway between October 2010 and September 2013 (Figure 1). The region has a well-developed public emergency medical system, and survival rates are estimated to be comparable to other regions in Norway and Europe.28,29 Every year, approximately 45 OHCA survivors were discharged from this cardiac ward. The hospital had protocols for the care of resuscitation patients in line with international guidelines and performed induced hypothermia and pacemaker implantations. 3 The present study did not involve any changes to the medical care of OHCA patients. The OHCA survivors were recruited by mail after discharge from the cardiac ward and invited to the follow-up study. Survivors were included in this substudy if they had recovered from a period of inpatient coma; were discharged from the hospital to their regular home environment and were eligible for an outpatient assessment, including cerebral MRI and formal cognitive assessment approximately 3 months after OHCA; were between 18 and 85 years of age; and were fluent in Norwegian language. The exclusion criteria were nonindependent living prior to or after OHCA, brain injury or neurologic disease prior to OHCA, learning disabilities during formal education, prior or ongoing psychiatric diagnosis, or serious comorbid medical condition (eg, cancer). MRI assessment was prohibited if survivors had an ICD or other body implants not compatible with the magnetic environment. Survivors were excluded if they had been conscious on admission to the emergency department, which was a criterion established to keep our results applicable to most previous research trials that examined cognitive outcome after OHCA. Survivors who regained consciousness rapidly after initial hospital admission were usually excluded from these trials.14,20 For the purpose of this substudy, a control group of age-matched healthy volunteers was recruited through advertising in the local newspaper. The controls reported no history of neurologic, cardiovascular, or psychiatric disorder. All participants signed an informed consent form. MRI assessment and cognitive testing were performed on the same day. This study complied with the Declaration of Helsinki and was approved by the Regional Committee for Medical Research Ethics in Northern Norway (REK 2009/1395 and REK 2012/1588).
Standardized neuropsychological tests with published normative data were used to measure aspects of verbal learning and memory, attention, executive functioning, and fine-motor coordination. Fine-motor functioning and coordination were assessed with the Grooved Pegboard. 30 Attention and executive functioning were assessed with Trials 3 and 4 from the Trail Making Test (TMT) from the Delis Kaplan Executive Functioning System (D-KEFS). 31 Verbal learning and memory were measured with the California Verbal Learning Test II (CVLT-II) standard version. 32 In this study, we used the scores from the learning trial, the immediate and delayed recall trial, and the recognition trial. All tests are published in Norwegian. The raw scores from the cognitive tests were transformed into standardized scores using a computer-scoring software from the publishers of the respective tests, which corrects for the individual’s sex and age using published normative data. One exception is the Trail Making Test’s normative data, which were corrected solely for the influence of age.
MRI Acquisition and Preprocessing
Subjects were scanned in a 1.5-T Phillips Intera MR scanner using an 8-channel head coil. The T1-weighted structural scans were performed with a 3D turbo field echo scan with TR = 1825 ms, TI = 855 ms, TE = 4.0 ms, flip angle = 8°, and voxel resolution = 0.94 × 0.94 × 1.25 mm3.
All T1-weighted structural images were analyzed with FreeSurfer (FS) version 6.0 (www.surfer.nmr.mgh.harvard.edu) to provide estimates of detailed anatomical information. Briefly, the FS analysis stream includes the removal of nonbrain regions, transformation into Talairach space, volumetric segmentation of subcortical white matter and deep gray matter structures, normalization of intensity, tessellation of gray and white matter boundaries, automatic topology correction, and labeling of neuroanatomical structures. 33 The cortical thickness is defined as the closest distance from the gray/white boundary to the gray/CSF boundary at each vertex on the tessellated surface. 34
Statistical Analyses
SPSS version 25 was used to analyze the descriptive, demographic, and neuropsychological variables. Continuous data are presented as the mean, SD, and range. Discrete data are given in numbers and percentages. Assumptions of normality and homogeneity of variance were assessed with the Shapiro-Wilks test and Levene’s test, respectively, in addition to inspection of Q-Q plots. Group differences in neuropsychological, demographic, and resuscitation characteristics were analyzed with t-tests. The following data were missing in the neuropsychological tests: 2 survivors had not completed the Grooved Pegboard Test, and 2 survivors had not completed the CVLT recognition subtest. There were no substitutions of missing data. An α level of .05 was used to determine significance unless otherwise specified.
The statistical analyses of cortical thickness were performed with the freely available software Permutation Analysis of Linear Models (PALM). 35 Permutation testing has been recommended over other methods due to better control for the false positive rate. 36 The preprocessing of the cortical surfaces was performed in the mris_preproc module in FS 6.0. The design matrixes for the permutation analyses consisted of group (OHCA survivors vs healthy controls) and sex as factors, whereas age, CVLT-II performance, coma duration, and time to ROSC were treated as continuous covariates. All analyses of cortical thickness and surface area, controlled for the effects of age and sex. All covariates were mean-centered before the analyses. The permutation analyses were performed with threshold-free cluster enhancement, 36 1000 iterations with tail approximations, 37 and FWER (family-wise error rate) corrections for multiple contrasts with a significance level of P < .05. All images were annotated using the Desikan-Killiany atlas 38 incorporated in FS 6.0.
Results
Participants
Figure 1 shows the flow chart of inclusion and exclusion of OHCA survivors for the present study. One survivor was female. The included survivors had medical characteristics associated with good outcomes. 39 All survivors had a first time, normothermic, witnessed OHCA with bystander resuscitation initiated at the scene. The cause of OHCA was infarctions in 12 (92%) survivors, with all having an initial shockable rhythm presented. Nine (69%) survivors were treated with therapeutic hypothermia; 11 (84.6%) were treated with percutaneous coronary intervention (PCI). Documented cardiovascular pathology prior to cardiac arrest was evident from hospital records in 6 (42%) survivors. Nine (69%) survivors were smokers prior to OHCA.
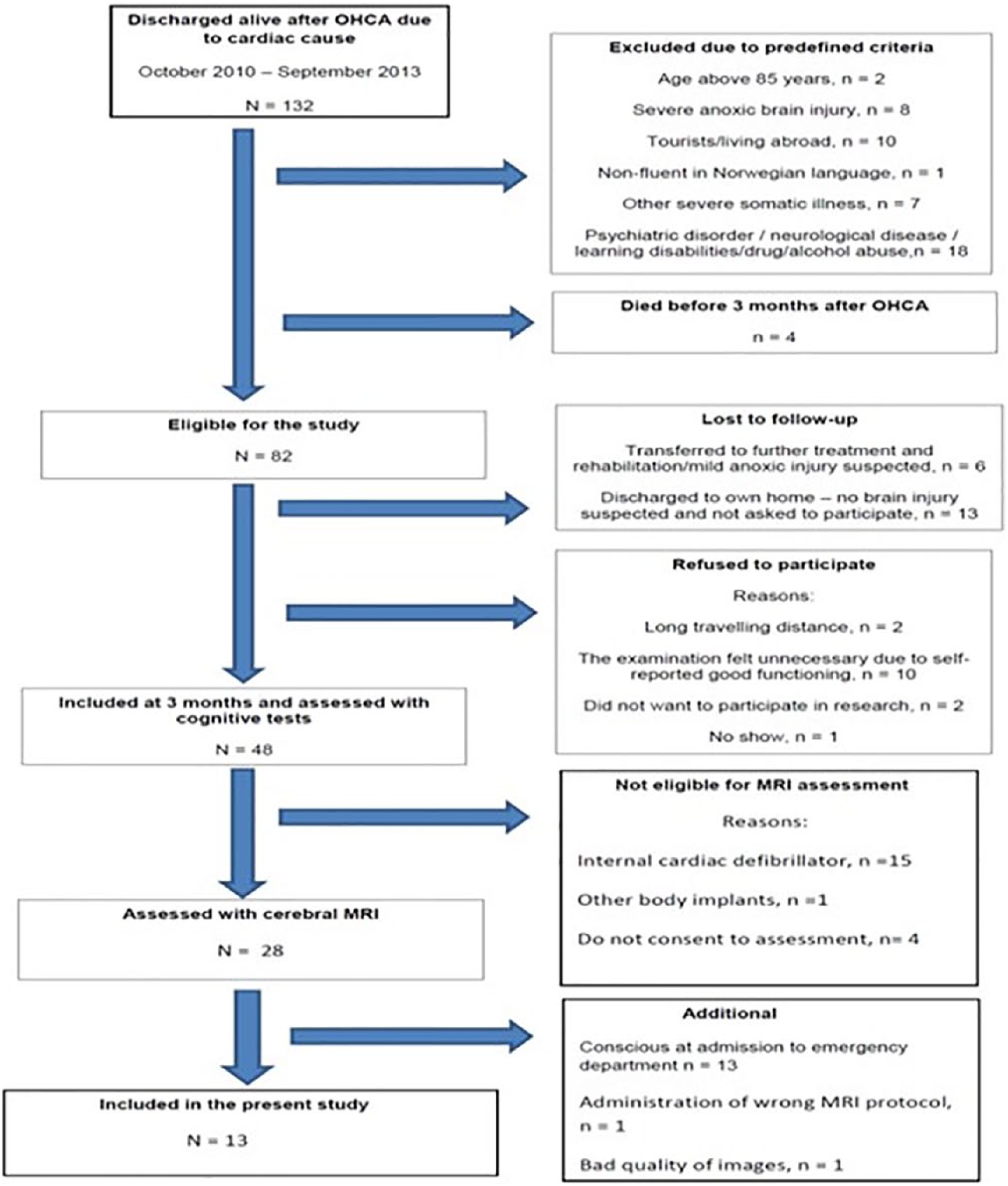
Flow chart of the inclusion and exclusion of the OHCA survivors.
The control group comprised 8 men and 11 women. There were significant differences in sex between patients and controls, with more women in the control group, but there were no significant group differences in age or length of education. There were no sex differences in terms of the mean cortical thickness in the healthy controls (t[17] = 0.63, P = .54) when adjusting for age. All controls and survivors were right-handed, except for one survivor that was left-handed.
Group Comparisons
As presented in Table 1, the survivors showed significantly poorer performance than the controls on all neuropsychological tests (all t > 2.44, P < .02), except for the TMT subtests assessing attention and executive functions and the recognition trial from the CVLT-II test.
Sample Characteristics and Group Comparisons a .
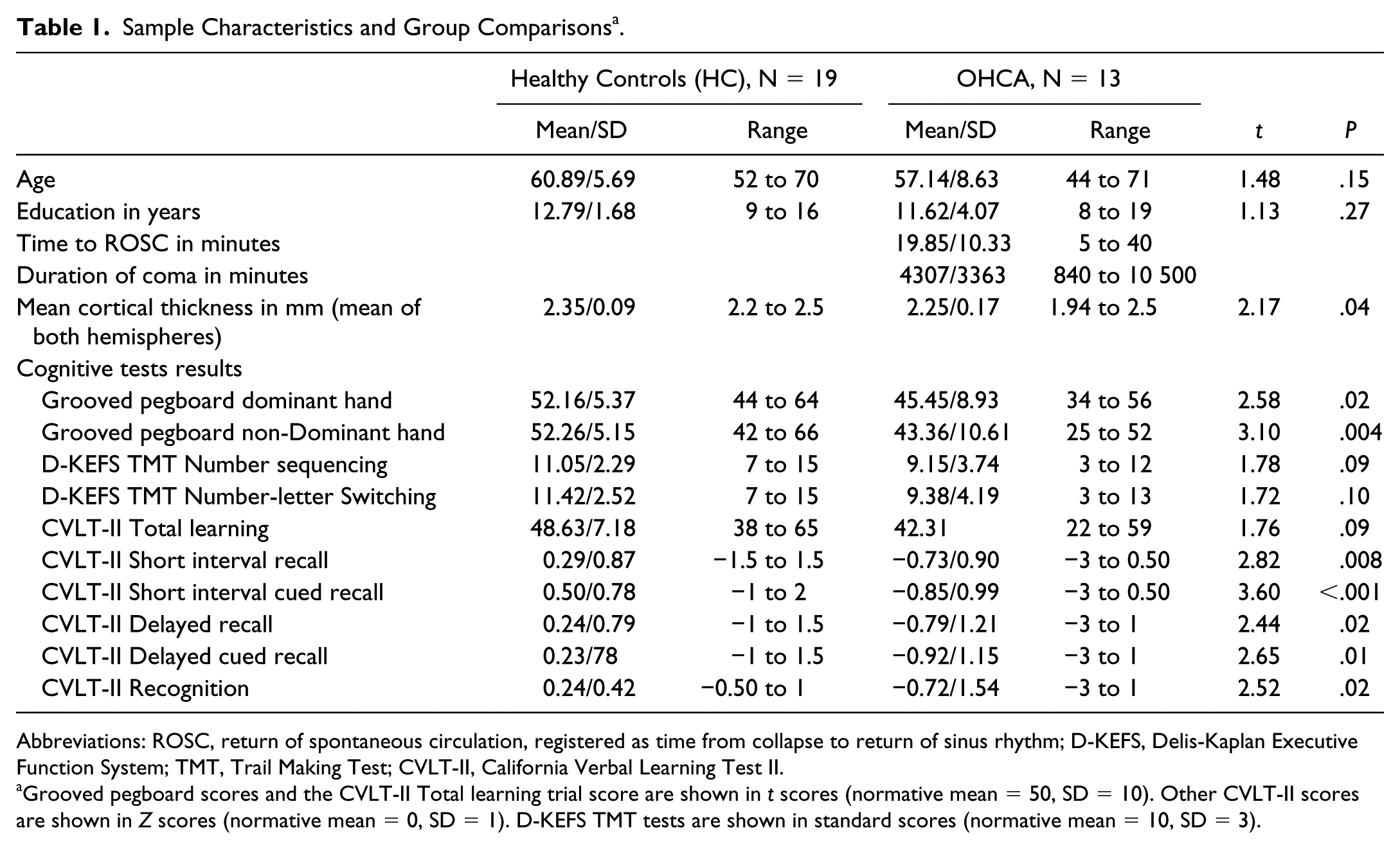
Abbreviations: ROSC, return of spontaneous circulation, registered as time from collapse to return of sinus rhythm; D-KEFS, Delis-Kaplan Executive Function System; TMT, Trail Making Test; CVLT-II, California Verbal Learning Test II.
Grooved pegboard scores and the CVLT-II Total learning trial score are shown in t scores (normative mean = 50, SD = 10). Other CVLT-II scores are shown in Z scores (normative mean = 0, SD = 1). D-KEFS TMT tests are shown in standard scores (normative mean = 10, SD = 3).
The analyses of SA revealed no significant differences between the OHCA survivors and controls. Survivors had a significantly reduced Cth in bilateral clusters located in the frontal cortices, including the caudal middle, superior, and rostral middle frontal areas, compared with the controls. Furthermore, there were significant bilateral differences between the survivors and controls in the clusters covering parts of the parietal lobe including the precuneus, the cuneus, the isthmus cingulate cortex, and the paracentral, postcentral, inferior parietal, and superior parietal areas. The difference in thickness between the groups was also observed in the unilateral parts of the left lateral orbitofrontal, left rostral anterior cingulate, left medial orbitofrontal, left lateral occipital, left middle temporal, and the right supramarginal cortices. The significantly impacted clusters are shown in Figure 2.
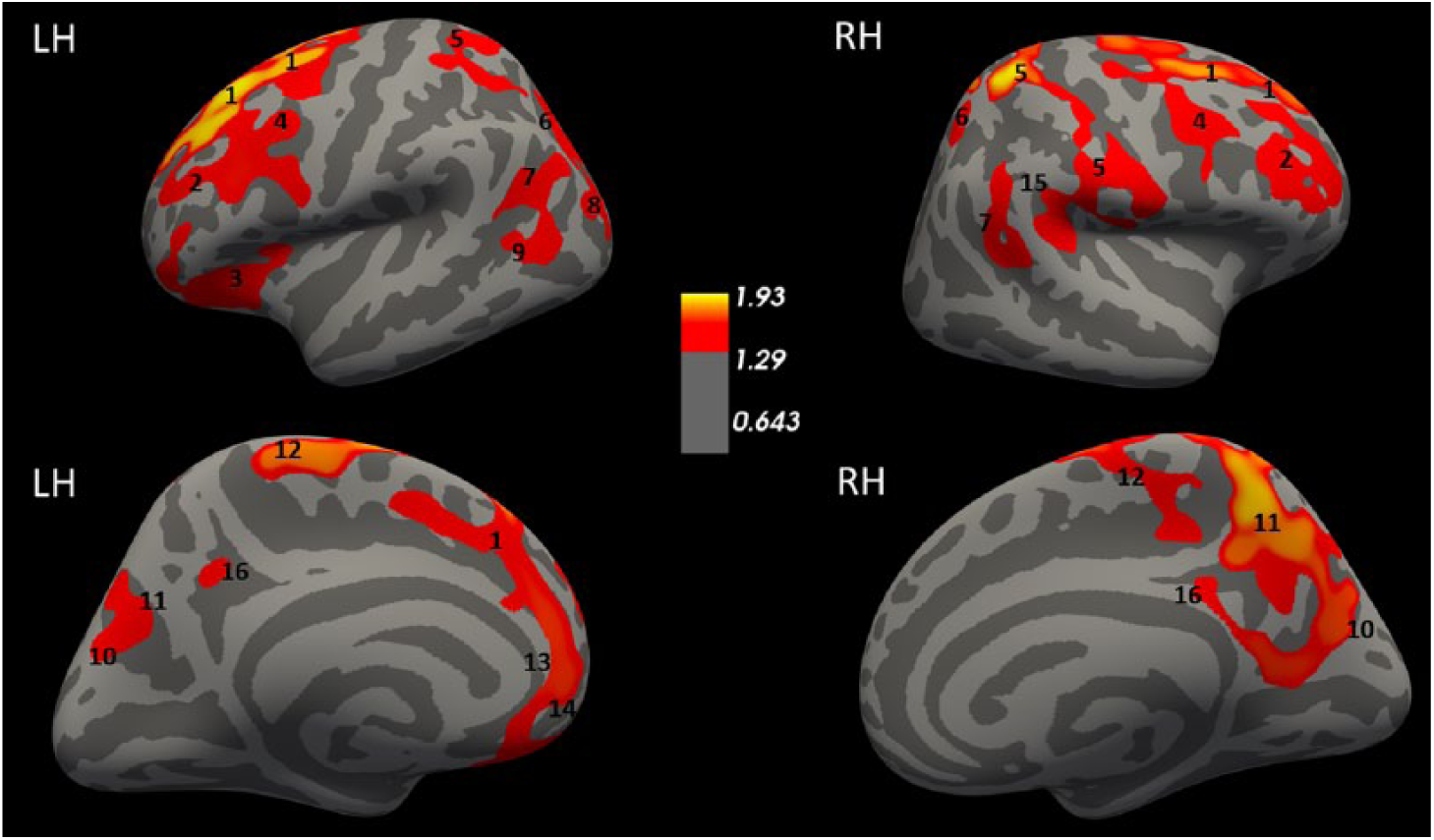
Differences in the Cth of OHCA survivors and healthy controls. Colored areas represent significant clusters where controls had higher Cth than OHCA survivors, adjusted for age and sex. The color coding for significant P values (P < .05) is on a logarithmic scale, and the thresholds were set at −log10(.05) = 1.3. LH, left hemisphere; RH, right hemisphere. The images are annotated according to the Desikan-Killiany atlas: 1 = superior frontal, 2 = rostral middlefrontal, 3 = lateral orbitofrontal, 4 = caudal middlefrontal, 5 = postcentral, 6 = superior parietal, 7 = inferior parietal, 8 = lateral occipital, 9 = middle temporal, 10 = cuneus, 11 = precuneus, 12 = paracentral, 13 = rostral anteriorcingulate, 14 = medial orbitofrontal, 15 = supramarginal, 16 = isthmus cingulate.
To test whether differences in Cth could explain the difference in memory performance between the controls and the survivors, the interactions between group and CVLT-II performance were tested. As shown in Figure 3, there was a significant interaction between group and CVLT-II short-delay recall memory performance on Cth. Improved CVLT-II performance of controls compared with that of patients was associated with increased Cth in bilateral clusters involving the temporal, frontal, parietal, posterior-cingulate, and precentral cortices. Furthermore, increased Cth values in the left fusiform gyrus, lingual gyrus, left lateral occipital cortex, and the left inferior parietal cortex were associated with improved short-delay performance on the CVLT-II. Almost identical findings were found for the CVLT-II long-delay recall. No other interactions between neuropsychological performance and group on Cth were significant after permutation testing.
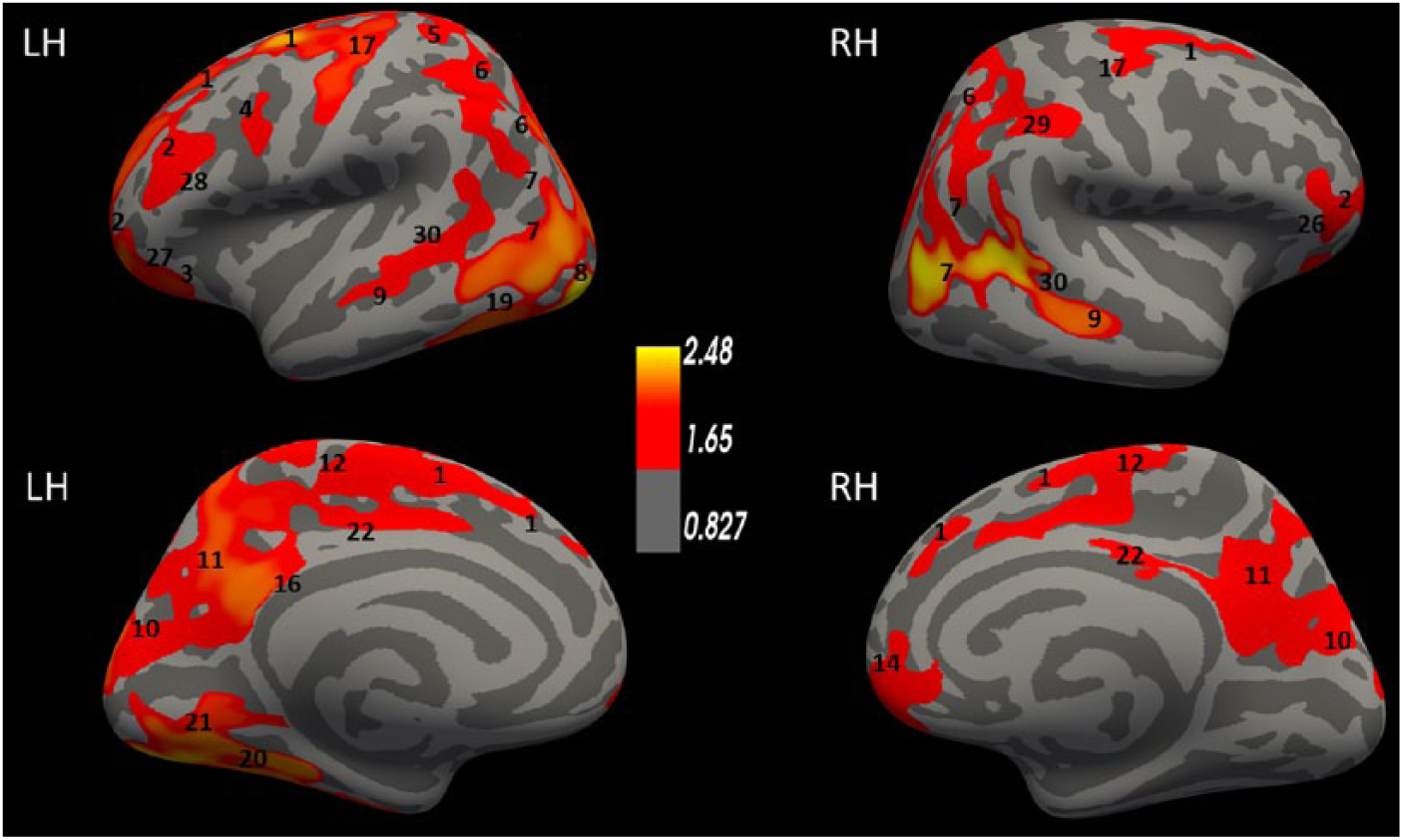
Interaction between group and CVLT-II performance on Cth. Colored areas represent significant clusters where the controls had a higher Cth that was significantly associated with better CVLT-II recall performance than the OHCA survivors, adjusted for age and sex. The color coding for significant P values (P < .05) is on a logarithmic scale, and the thresholds were set at −log10(0.05) = 1.3. L, left; R, right. The images are annotated according to the Desikan-Killiany atlas: 1 = superior frontal, 2 = rostral middle frontal, 3 = lateral orbitofrontal, 4 = caudal middle frontal, 5 = postcentral, 6 = superior parietal, 7 = inferior parietal, 8 = lateral occipital, 9 = middle temporal, 10 = cuneus, 11 = precuneus, 12 = paracentral, 14 = medial orbitofrontal, 16 = isthmus cingulate, 17 = precentral, 19 = inferior temporal, 20 = fusiform, 21 = lingual, 22 = posterior cingulate, 26 = parstriangularis, 27 = pars orbitalis, 28 = pars opercularis, 29 = supramarginal, 30 = banks sts.
Correlational Results in the OHCA Group
Time to ROSC was significantly associated with reduced Cth in several coherent clusters involving the following regions: temporal, frontal, occipital and parietal cortex, inferior parts of the lateral pre- and postcentral gyri, and the cingulate cortices (see details in Figure 4). The duration of coma had significant associations with reduced Cth in large bilateral cortical regions, including clusters in the frontal cortices, the parietal lobes, the temporal lobes, the cunei, and the occipital lobes. The clusters of reduced Cth associated with coma duration shown in Figure 5 largely overlapped with the cluster of reduced Cth associated with time to ROSC. In comparison to the clusters with significant cortical thinning associated with time to ROSC, the clusters associated with coma duration covered larger regions and included additional regions in the parietal, the anterior cingulate, and the superior central and occipital cortices that were not found for time to ROSC. There were no significant correlations between the time to ROSC and the cognitive tests. A longer coma duration was significantly associated with poorer fine-motor functioning as measured by the Grooved Pegboard Test using the non-dominant hand (r = −0.82, P < .01), poorer attention as measured by the TMT-3 (r = −0.61, P = .03), and poorer short-delay recall on the CVLT-II (r = −0.59, P = .03).
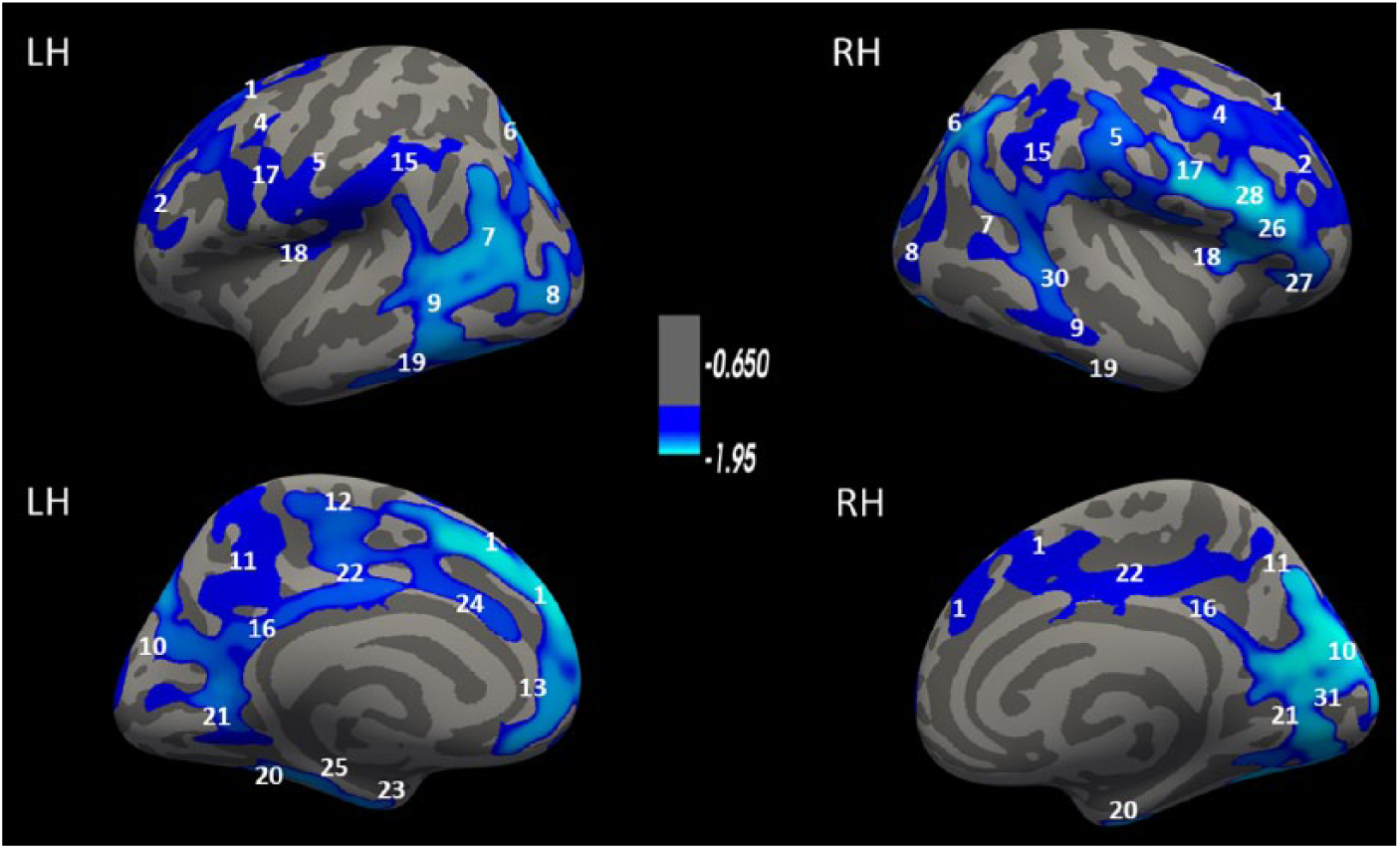
Correlation in OHCA survivors of time to ROSC and Cth. Colored areas represent clusters where time to ROSC had a significant negative correlations with Cth, adjusted for age and sex. The color coding for significant P values (P < .05) is on a logarithmic scale, and the thresholds were set at −log10(0.05) = 1.3. L, left; R, right. The images are annotated according to the Desikan-Killiany atlas: 1 = superior frontal, 2 = rostral middle frontal, 3 = lateral orbitofrontal, 4 = caudal middle frontal, 5 = postcentral, 6 = superior parietal, 7 = inferior parietal, 8 = lateral occipital, 9 = middle temporal, 10 = cuneus, 11 = precuneus, 12 = paracentral, 13 = rostral anterior cingulate, 14 = medial orbitofrontal, 15 = supramarginal, 16 = isthmus cingulate, 17 = precentral, 18 = insula, 19 = inferior temporal, 20 = fusiform, 21 = lingual, 22 = posterior cingulate, 23 = entorhinal, 24 = caudal anterior cingulate, 25 = parahippocampal, 26 = pars triangularis, 27 = pars orbitalis, 28 = pars opercularis, 30 = banks sts, 31 = pericalcarine.
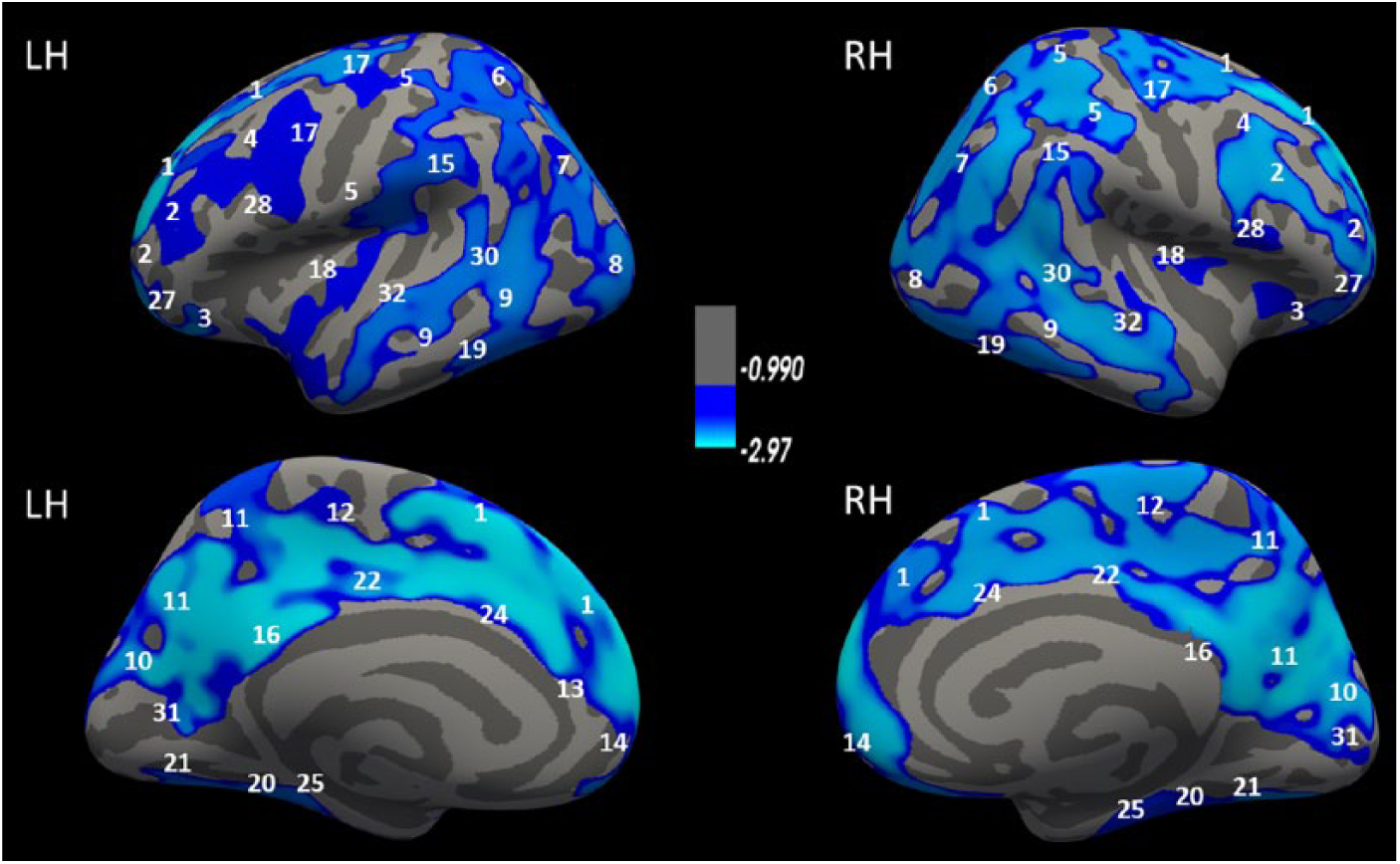
Correlation in OHCA survivors of coma duration and Cth. Colored areas represent clusters where coma duration had a significant negative correlation with Cth, adjusted for age and sex. The color coding for significant P values (P < .05) is on a logarithmic scale, and the thresholds were set at −log10(0.05) = 1.3. L, left; R, right. The images are annotated according to the Desikan-Killiany atlas: 1 = superior frontal, 2 = rostral middle frontal, 3 = lateral orbitofrontal, 4 = caudal middle frontal, 5 = postcentral, 6 = superior parietal, 7 = inferior parietal, 8 = lateral occipital, 9 = middle temporal, 10 = cuneus, 11 = precuneus, 12 = paracentral, 13 = rostral anterior cingulate, 14 = medial orbitofrontal, 15 = supramarginal, 16 = isthmus cingulate, 17 = precentral, 18 = insula, 19 = inferior temporal, 20 = fusiform, 21 = lingual, 22 = posterior cingulate, 24 = caudal anterior cingulate, 25 = parahippocampal, 27 = pars orbitalis, 28 = pars opercularis, 30 = banks sts, 31 = pericalcarine, 32 = superior temporal.
Discussion
Compared with controls, OHCA survivors showed significantly reduced performance in tests for verbal memory and fine-motor coordination, but not in the tests for attention or executive functions. The OHCA survivors showed a significantly thinner cortex than controls in large bilateral clusters in the frontal, parietal, and inferior temporal cortices. Additional unilateral clusters with a thinner cortex than controls were observed in the left occipital lobe and the left temporal lobe of survivors. A thinner cortex in these same areas was related to the reduced memory performance in survivors compared with controls. Cortical thinning in these areas was also significantly related to a longer time to ROSC and a longer coma duration in the OHCA group. A longer coma duration, but not a longer duration to ROSC, was significantly correlated with cognitive test performance. A longer coma duration was significantly associated with both poorer cognitive test performance and a thinner cortex covering large parts of the cortex. Our results suggest that the cognitive problems observed in long-term OHCA survivors involve significant and widespread changes in cortical anatomy and, more specifically, reduced Cth. Our results suggest that Cth is a more relevant cortical morphometric feature than SA in this patient population. Cth was associated with both resuscitation characteristics and memory impairment, whereas the SA analyses revealed no significant results.
The strengths of this study include the attention given to maintain similar etiology of cardiac arrests across all included OHCA participants and to the overall functional outcome of the survivors. This study did not include OHCA survivors from a broad range of possible outcome severity or from various etiologies of hypoxic-ischemic events. The etiology of hypoxic-ischemia may influence the manifestation of symptoms and prognosis. 5 Our survivors were at the same time point in recovery from OHCA, but previous studies have documented both individual improvement and decline in cognitive functions beyond 3 months after resuscitation.10,14,40-42 In our study, age and length of education did not differ between the survivors and the controls, and the influence of age and sex on Cth and SA were controlled for in all statistical analyses.
The limitations to the present study are the small sample sizes and the related lack of power to detect possibly significant group differences in cognition and cortical anatomy beyond the significant results presented here. The results are merely observational and associative, implying that the heterogeneity in cognitive results and cortical thickness values can possibly be attributed to other individual differences among OHCA survivors and not solely to the structural alterations caused by an OHCA. Nevertheless, the included survivors were carefully selected according to stringent inclusion and exclusion criteria. These criteria were designed to avoid poor or suboptimal premorbid brain functioning and to increase the likelihood that any observed group differences in cortical anatomy and cognition were attributable to the OHCA with adjunct treatments and few other factors. Our study has a selection bias toward survivors with favorable functional outcomes. Additional selection bias comes from including only survivors without ICD implants. All our survivors received bystander CPR, and the majority underwent early PCI treatment because of cardiac infarctions and had no prior history of cardiac disease. The reasons for not receiving an ICD in our study are likely due to early and complete reversal of what caused cardiac arrest, relatively good cardiac functioning, and potential for optimal cardiac recovery without the need for secondary prevention. Consequently, our results do not apply to all OHCA survivors. However, our results apply to long-term survivors with good gross neurological outcomes and favorable prognosis for long-term survival.
The results from a recent study of 8 functionally independent OHCA survivors somewhat disagree with our findings. Using similar MRI surface-based analytic software to the present study, the authors concluded that what they defined as brief episodes of hypoxic-ischemia due to OHCA resulted in reduction of hippocampal volume and no significant cortical thinning. 21 In their study, the long-term survivors of OHCA were compared to cardiac controls who had undergone cardiac infarctions but not cardiac arrest. Cardiovascular disease is correlated with cortical thinning 43 and cognitive impairment. 44 It has been emphasized that studies related to neurological and cognitive outcomes after OHCA should control for the impact of both ICU-related risk factors and cardiovascular risk factors.7,45 However, in small samples, one runs the risk that a control group with a heterogeneous cardiovascular burden underestimates the impact of cardiac arrest on the brain as much as a healthy control group may exaggerate group differences attributed to the OHCA. The characteristics of an optimal control group for neuropsychological outcome studies of OHCA survivors have yet to be determined.7,45
The relative vulnerability of the cortical architecture to the hypoxic-ischemia caused by cardiac arrest is supported by previous cerebral MRI studies using the clinical radiological inspection of images or quantitative volumetric analyses.18-20,46 Additionally, these studies indicate cortical involvement in OHCA-related memory problems, suggesting that these memory problems are not well explained by isolated atrophy in the hippocampus.18,19 We previously reported a significant relationship between reduced hippocampal volume and decreased memory performance in the same survivors that were included in the present study. 20 In that study, hippocampus was the only subcortical structure that had significantly smaller volume in survivors than in the controls. With the present results, we suggest that additional, topographically widespread, cortical thinning contributes to memory problems in long-term survivors of OHCA. The general implication of widespread cortical thinning is that several neuropsychological domains, in addition to memory functioning, should be evaluated in clinical follow-up assessments and in the research on long-term functional outcomes after OHCA. 18
Our finding of memory impairments with combined fine-motor impairment is in accordance with the results from a previous study describing the cognitive profile of survivors with comparable OHCA characteristics and functional outcomes. 12 However, others found executive functions to be the most vulnerable cognitive domain after OHCA.8-10 In the present study, we used well-established cognitive tests that have the sensitivity to capture even subtle cognitive limitations. 12 Nevertheless, the selection of different neuropsychological tests could have provided different results, and this last point may especially apply to the selection of tests for attention and executive functioning, which were assessed to a very limited extent by the tests in the present study. 47 Our results need replication and extension in future studies, preferably with larger samples sizes.
A significant correlation between increased time to ROSC and decreased cognitive performance has been found in only one previous study, 16 indicating that there is currently not enough empirical evidence to advocate that long-term cognitive functioning can be accurately interfered from the time to ROSC.6,13 In the present study, a longer time to ROSC correlated significantly with widespread reduced Cth but not with any cognitive tests. The possible influence of the coma duration on Cth and on cognition is complicated to interpret, as the majority of our survivors were treated with therapeutic hypothermia and adjunct therapies, which keep the patient sedated for a possibly longer time than in the natural course of recovery from unconsciousness. A general lack of clinical predictors for long-term neuropsychological functioning in OHCA survivors and the recognition of multiple risk factors for long-term functional impairments in this population have resulted in international clinical recommendations for individualized follow-up assessment of the neuropsychological outcomes in all survivors during the first months after resuscitation. 4 Future research combining various cerebral MRI modalities with the assessment of functional outcomes can result in a better understanding of diagnostics and treatment options for the brain-related problems that prevent a subset of long-term OHCA survivors from returning to the level of prearrest functioning. 45
Conclusions
The results support the hypothesis of topographically widespread cortical involvement in the cognitive impairments observed in long-term OHCA survivors. In our OHCA group, widespread Cth reductions showed significant associations with both clinical and cognitive measures. The cerebral basis of the cognitive problems are poorly understood and understudied. Future studies incorporating neuroimaging methods may advance the understanding of cognition and other functional impairments after OHCA and may guide prediction models, diagnostics, and treatment options.
Footnotes
Acknowledgements
The investigators thank all participants for their generous contributions.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research project was financially funded by the Norwegian Extra Foundation for Health and Rehabilitation, the University Hospital of North Norway, the Faculty of Health Sciences at the UIT, the Arctic University of Norway, and the Norwegian Health Authorities Northern Region. The sponsors did not have any role in the design, methods, subject recruitment, data collection, analysis, or preparation of the article.
