Abstract
Background. We previously demonstrated that step training leads to reorganization of neuronal networks in the lumbar spinal cord of rodents after a hemisection (HX) injury and step training, including increases excitability of spinally evoked potentials in hindlimb motor neurons. Methods. In this study, we investigated changes in RNA expression and synapse number using RNA-Seq and immunohistochemistry of the lumbar spinal cord 23 days after a mid-thoracic HX in rats with and without post-HX step training. Results. Gene Ontology (GO) term clustering demonstrated that expression levels of 36 synapse-related genes were increased in trained compared with nontrained rats. Many synaptic genes were upregulated in trained rats, but Lrrc4 (coding NGL-2) was the most highly expressed in the lumbar spinal cord caudal to the HX lesion. Trained rats also had a higher number of NGL-2/synaptophysin synaptic puncta in the lumbar ventral horn. Conclusions. Our findings demonstrate clear activity-dependent regulation of synapse-related gene expression post-HX. This effect is consistent with the concept that activity-dependent phenomena can provide a mechanistic drive for epigenetic neuronal group selection in the shaping of the reorganization of synaptic networks to learn the locomotion task being trained after spinal cord injury.
Introduction
Spinal cord injury (SCI) results in severe motor and sensory dysfunction. 1 Although neurorehabilitation, including locomotor training, can improve motor function post-SCI, characterization of neuroplastic changes at the synaptic level remains poorly understood. 2 Many studies have demonstrated the effectiveness of this training in the recovery of motor function, highlighting the importance of activity-dependent mechanisms in long-term prognosis.2-4 It is also recognized that an incomplete SCI is more likely to have positive outcomes than a complete one. 4 Using an incomplete SCI injury model, a lateral hemisection (HX), step training facilitated functional recovery after injury. 5 Using the retrograde neuronal tracer, tetramethyl rhodamine dextran amine, the greater number of labeled neurons above and below the lesion site, observed in trained compared with nontrained rats, suggests that training contributed to increased numbers of synapses on lumbar neurons. There also was evidence that the spinal networks projecting to lower limb motor pools were reorganized differently in the trained versus nontrained rats post-SCI. 5 We reasoned that a better understanding of molecular mechanisms underlying the effects of training on spinal synaptic plasticity could facilitate functional excitatory connectivity among and between spinal and supraspinal networks.
To begin to understand the training-induced molecular changes of synapses, we performed ribonucleic acid (RNA) sequencing (RNA-Seq) on the lumbar spinal cords of rats with and without treadmill training after HX injury. We observed significant upregulation of synapse-related genes and direct evidence for the upregulation of dendritic synapses within the L1 spinal cord using RNA-Seq and immunohistochemistry (IHC), respectively.
Methods
All experimental procedures were performed in compliance with the University of California Los Angeles Chancellor’s Animal Research Committee (ARC) and the guidelines of the National Institutes of Health Guide for the Care and Use of Laboratory Animals (National Research Council, 2011).
Experimental Groups, Procedures, and Timeline
Adult female Sprague-Dawley rats (255-285 g body weight, n = 8) were kept on a 12/12-hour. light-dark cycle with ad libitum access to food and water throughout the duration of the study. The experimental groups and timeline for the study are shown in Figure 1A. All rats received a mid-thoracic (T9 spinal level) HX. Prior to surgery, rats were randomly assigned to the treatment group (step training, Tr, n = 4) or control group (no step training, n = 4), and there was no significant difference between groups in their body weight (P > .05, Wilcoxon rank-sum test). All surgeries were performed under aseptic conditions with the rats deeply anaesthetized with isoflurane gas (1.0%-2.5%). Postsurgical care was performed as described previously. 5 A partial laminectomy was performed from T7 to T10 to expose the spinal cord. The spinal cord was lifted with a curved probe and hemisected using microdissection scissors at the T9 level. All exposed areas were kept moist with 0.9% saline washes. The depth of anesthesia was assessed regularly throughout surgery using toe-pinch. We confirmed that all animals exhibited lack of motor function (BBB [Basso, Beattie, and Bresnahan] score = 0) in the hemisected side of the spinal cord after awakening from anesthesia.
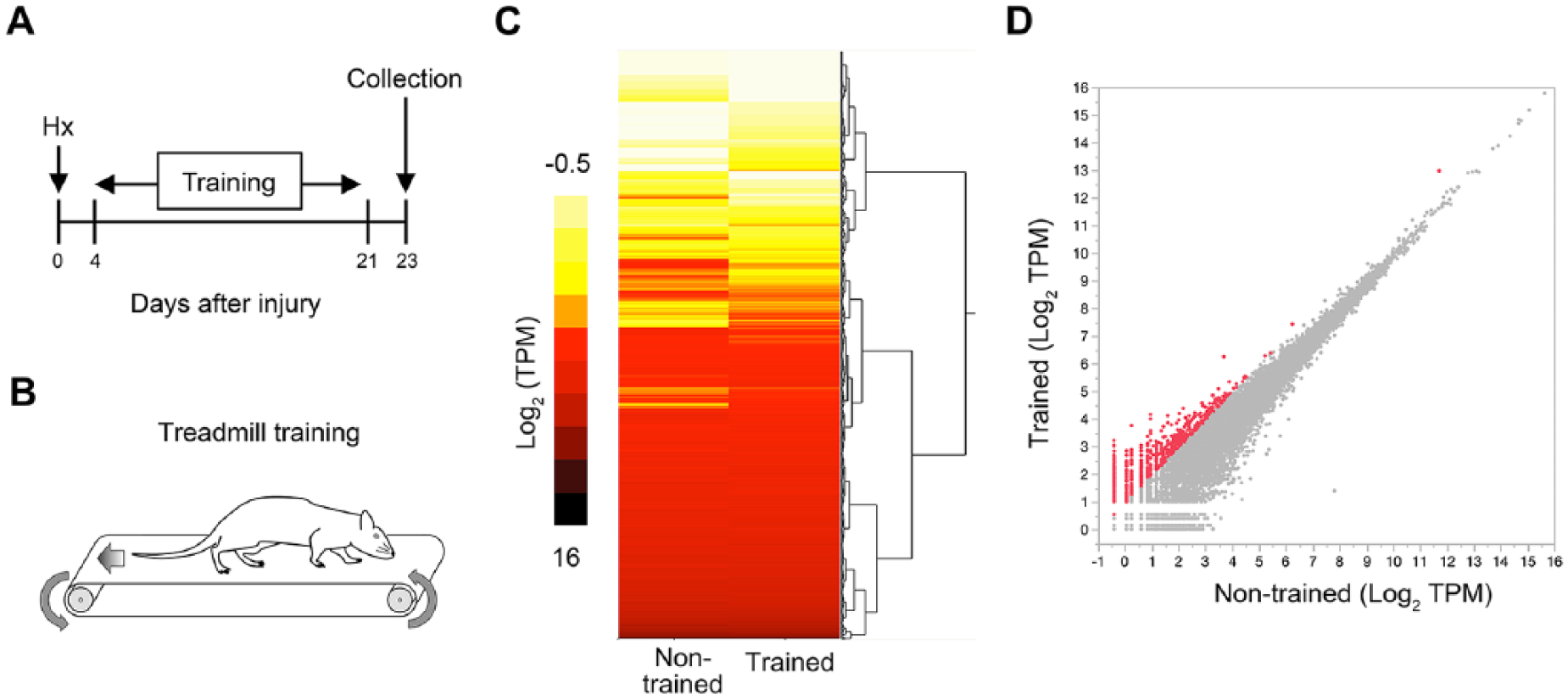
RNA-Seq analysis of lumbar spinal cord with gait rehabilitation after spinal cord injury (SCI). (A and B) Timeline of experimental procedures. Rats underwent a right lateral thoracic hemisection (HX) injury at T9 and were either nontrained or trained to step (35 min/d) for 18 days beginning 4 days after injury. L1 spinal cord was collected for RNA-Seq analysis at 23 days posthemisection (dph). (C) A heat map showing gene expression changes in the L1 spinal cord of nontrained and trained rats at 23 dph. Values are expressed in transcripts per kilobase million (TPM). (D) TPM values of 806 overexpressed (>2-fold change) genes in the RNA-Seq analysis of the trained rats compared with those of the nontrained rats shown in red dots.
Locomotor Training
Rats were placed on a level treadmill surface. Treadmill speed began at 9 cm/s and gradually increased to 21 cm/s based on the rate of functional recovery post-SCI. Rats were trained 7 d/wk in 35 min/session for 18 days starting 4 days after HX surgery.
Cell Expression by Linear Amplification and Sequencing 2 (CEL-Seq2) and Data Analysis
Under deep anesthesia using a glass probe, the L1 root was carefully followed from the L1 intervertebral foramen to the entry point of the spinal cord. A rostrocaudal 3.7-mm length 6 section of the L1 spinal cord was resected. We used a 1 mm length of rostral or caudal L1 spinal cord segment for either RNA-Seq or histological analysis, respectively. The rostral L1 region was removed and immediately frozen in liquid nitrogen. Libraries were constructed from spinal cords following the protocol as previously described. 7 We sequenced the libraries using the Illumina HiSeq 1500 (Illumina) and the reads were mapped by the aligning software HISAT2 (hierarchical indexing for spliced alignment of transcripts 2) (version 2.2.6) 8 to the reference genome (Rnor_6.0). Read counting per gene was performed with HTSeq (version 0.6.1) 9 such that duplicates in unique molecular identifiers were discarded. The data were deposited with the accession number GSE115156. The list of genes of which expression levels increased (>2-fold) in trained compared with nontrained rats was used in an enrichment analysis for the biological processes performed based on the Gene Ontology (GO) database annotations with the Database for Annotation, Visualization and Integrated Discovery (DAVID).10-12 The minimum kappa value of 0.50 was considered to be biologically significant for the similarity threshold gene clustering.
Quantification of NGL-2-Positive Synaptic Structures
Caudal L1 spinal cord segments were collected, fixed overnight with 4% paraformaldehyde as described previously. 5 Spinal cords were placed in small peel-away histomolds, covered with O.C.T embedding medium (Tissue-Tek), frozen over dry ice, and stored at −80°C until cryosectioning. Each tissue block was cut transversely (20-μm thick sections), stained with primary antibodies (1:200) overnight at 4°C, incubated with Alexa Fluor secondary antibodies (1:200) for 2 hours, and cover-slipped with Vectashield Mounting Media. Primary antibodies used in this study were: mouse antineuronal nuclei (NeuN, Millipore), chicken anti-microtubule-associated protein 2 (MAP-2, Novus Biologicals), mouse anti-synaptophysin (SYP, Millipore Sigma) and rabbit anti-leucine-rich repeat-containing protein 4 (Lrrc4, Biorbyt). Secondary antibodies (Jackson ImmunoResearch) used in this study were: donkey anti-mouse Alexa Fluor488, donkey anti-rabbit Alexa Fluor594, and donkey anti-chicken AMCA. The spinal cord sections were imaged on a confocal microscope (Leica TCS SP5, Leica) using Leica LAS AF software (Leica) and a fluorescence microscope (Axio Imager 2, Carl Zeiss Microscopy, LLC) using Image-Pro Plus software (Media Cybernetics Inc). Spinal cord segmental levels were verified by the shape of the spinal cord gray matter and Rexed laminae as a guide. 13 Ipsilateral ventral horns were photographed and SYP/NGL-2-positive synaptic puncta within the motoneuronal area at lamina IX (2,500 μm2) were counted by a tester blind to the experimental design, as described previously. 14
Statistical Analysis
The Wilcoxon rank-sum test was used to compare the medians of the data for relative expression ratio of RNA-Seq. Two-factor nested random effects model was used to compare the cell numbers of NeuN+ neurons as well as the synaptic puncta. All tests were 2-tailed, and the level of significance was set at P <.05. All values of bar graphs for groups are presented as the mean ± standard error of the mean. The data of box-and-whisker plots are presented as median (middle line), upper and lower quartiles (boxes) and minimum-maximum (whiskers). All statistical analyses were carried out using the JMP software program (version 13; SAS Institute).
Results
RNA-Seq Analysis of Injured Spinal Cord
For analysis of the transcriptome, rats were trained from 4 days post-HX (dph) to 21 dph, and spinal cord samples were collected at 23 dph (Figure 1A and B). Using RNA-Seq, the effects of training on gene expression levels caudal to the spinal lesion (L1) were assessed. The heat map indicated substantial differences in gene expression profiles in trained and nontrained rats at 23 dph (Figure 1C). The expression level differences of 806 genes were >2-fold between trained and nontrained rats (Figure 1D).
Biological Interpretation of a Gene List From RNA-Seq Analysis
In a previous study, a retrograde tracer technique demonstrated greater connectivity among neurons in rats engaged in treadmill training after HX; a finding consistent with some level of synaptic plasticity. 5 To make a biological interpretation of the RNA-Seq data focusing on the synapse, we performed a GO term analysis of genes that showed elevated expression levels in trained rats. 10 The gene-term 2D map view developed clusters related to the synapse, demonstrating genes and their corresponding GO terms (Figure 2). Each cluster mainly consists of the terms related to anatomy, assembly, or function of the synapse. This implies that trained rats, compared with nontrained, experienced a greater level of reorganization and functional changes at the synapse.
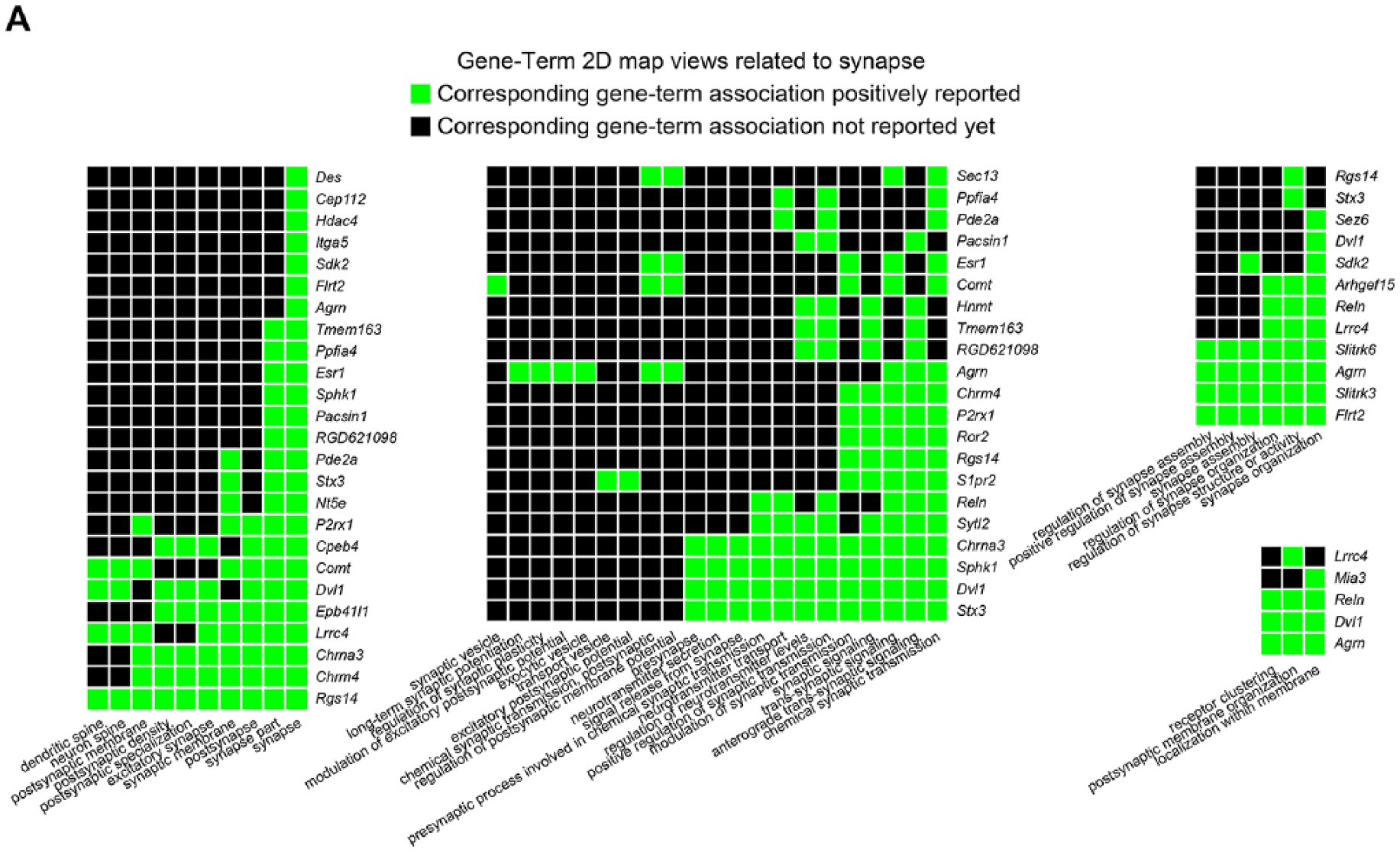
Gene-Term 2D map views. (A) Gene Ontology (GO) term clustering was performed using 806 increased genes in Figure 1D to identify the specific gene clusters related to synapse. Each green square indicates that a gene is annotated to the corresponding GO term. A total of 36 genes were listed in the Gene-Term 2D map views as synapse-related genes.
Lrrc4 Was the Most Highly Expressed Among Training-Induced Synapse-Related Genes
Using the list of genes shown in the gene-term 2D map related to synapse, we analyzed gene expression levels of each of these genes. Among these clustered synapse-related genes, Lrrc4 (coding netrin-G ligand 2, NGL-2), was the most highly expressed in trained rats (Figure 3A). Given that the expression level of Lrrc4 in trained rats was significantly higher than that in non-trained rats (Figure 3B), we focused on NGL-2 for further investigation.
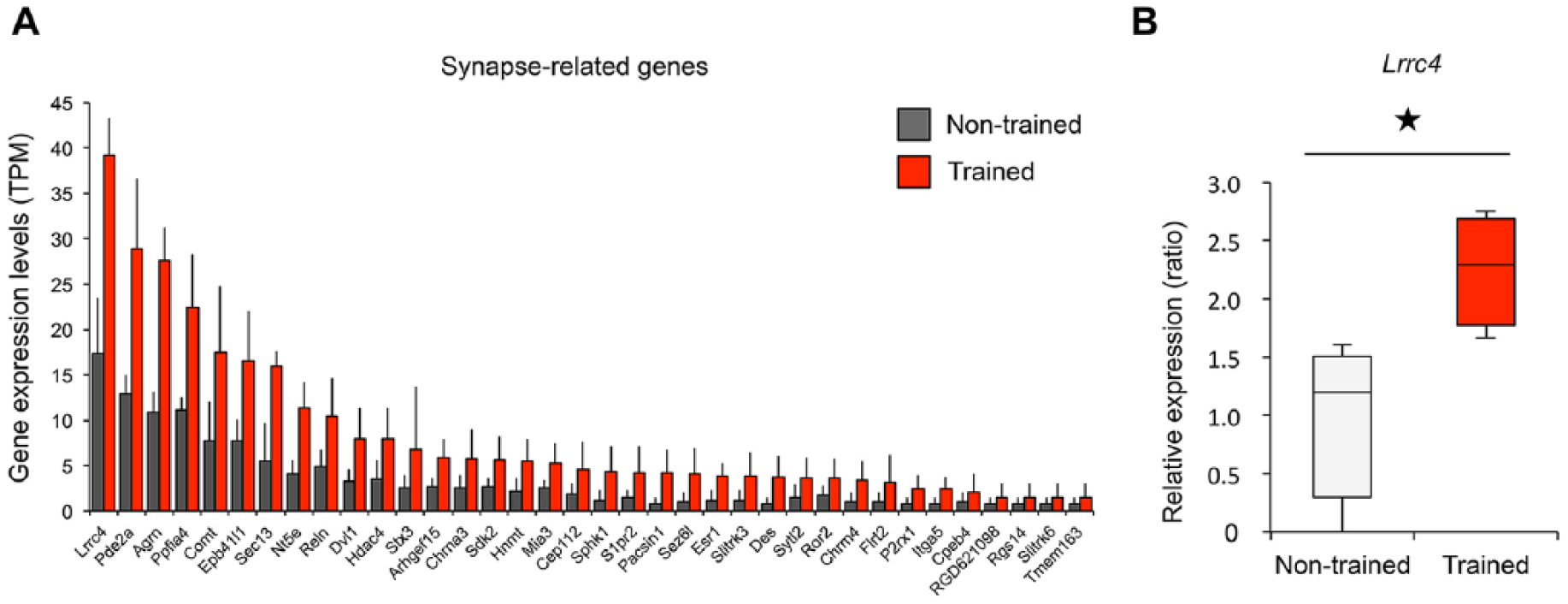
Expression levels of synapse-related genes. (A) The expression levels of 36 synapse-related genes in trained (red) and nontrained (gray) rats (n = 4 per group). (B) The comparison of gene expression levels of Lrrc4 in trained and nontrained animals. P < .05, Wilcoxon rank-sum test. The data are presented as the means ± SEM (standard error of the mean) (A) and median (middle line), upper and lower quartiles (boxes), and minimum-maximum (whiskers) (B).
Training Increased NGL-2-Positive Synaptic Puncta in the L1 Spinal Cord After Hemisection Injury
NGL-2 (postsynaptic) colocalization with synaptophysin (presynaptic) has a significant effect on the number of excitatory synapses. 15 Considering the previous results that training induced greater excitability of motor neurons after spinal cord injury, 5 the training-induced increase of Lrrc4 (NGL-2) may be associated with the number of synapses on motor neurons. Immunohistochemical analysis demonstrated that, while there was no statistical difference in the number of NeuN+ neurons between groups (Figure 4A and B), the lumbar spinal cord of trained rats contained a higher number of NGL-2+/SYP+ synaptic puncta compared with nontrained rats (Figure 4C-E).
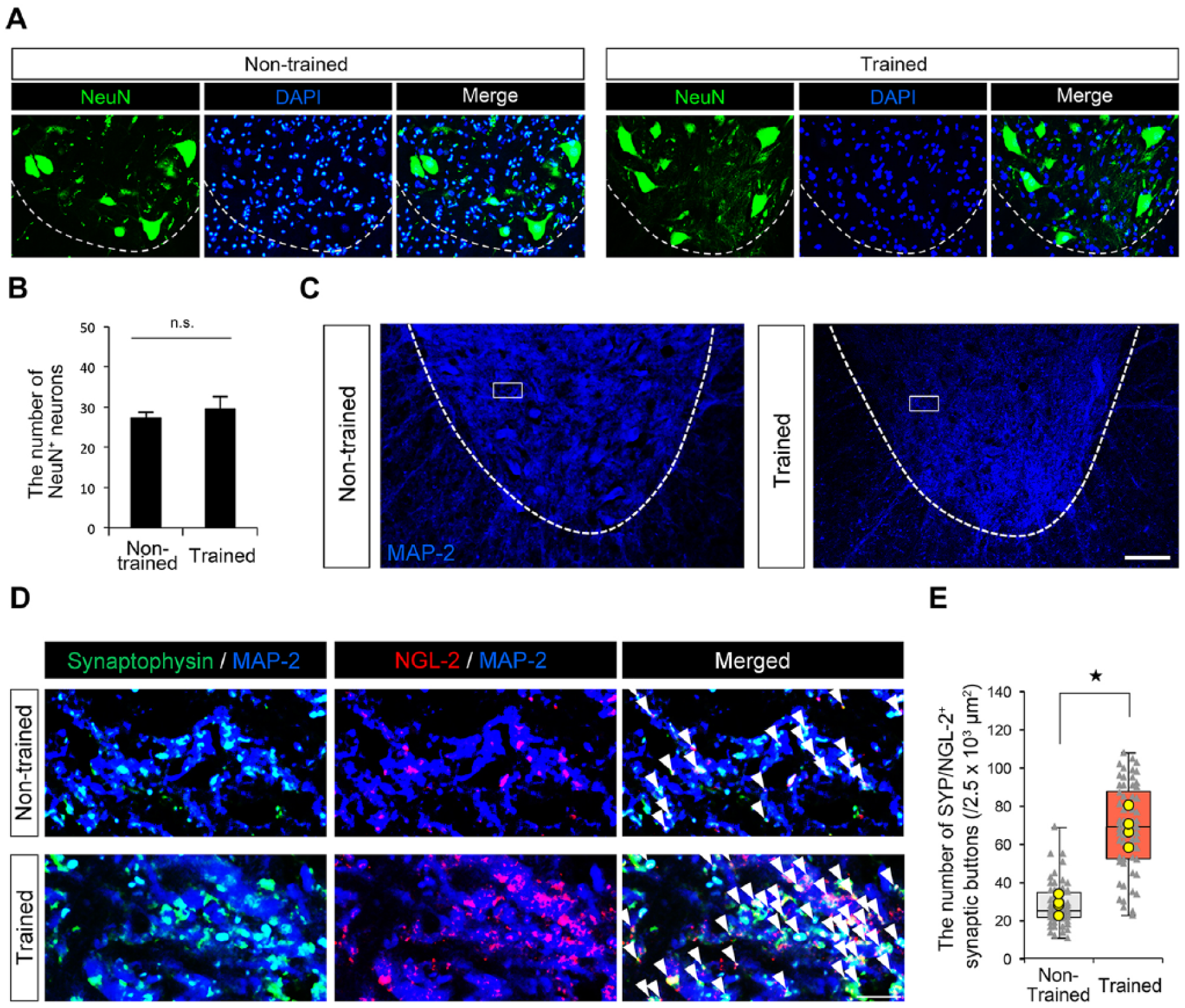
Increased NGL-2-positive synaptic puncta in trained rats. (A) The representative images of immunofluorescent staining of NeuN-positive neurons in ipsilateral ventral horns of nontrained and trained rats. (B) The quantitative analysis of the number of NeuN-positive neurons in ipsilateral ventral horns of nontrained and trained rats (n = 12 sections; 4 rats per group). Scale bar: 100 μm. P > .05, n.s., not significant. The error bars indicate the standard error of the mean (SEM). (C) The representative images of immunofluorescent staining of MAP-2 (a marker of dendrites) in ipsilateral ventral horns of nontrained and trained rats. (D) The representative confocal images of immunofluorescent staining of magnifications of the boxed areas in Figure 4C with a presynaptic marker synaptophysin (SYP; green), postsynaptic NGL-2 (red), and MAP-2 (blue). Arrowheads indicate SYP/NGL-2 colocalized synaptic puncta (yellow). Scale bar: 10 μm. (E) The quantitative analysis of the number of SYP/NGL-2 colocalized synaptic puncta in lamina IX of ipsilateral ventral horns of nontrained and trained rats (n = 60 areas; 4 rats per group). The yellow points indicate the average of 15 areas obtained from same animals. P < .05. The data are presented as median (middle line), upper and lower quartiles (boxes), and minimum-maximum (whiskers).
Discussion
In this study, we performed an RNA-Seq analysis and identified Lrrc4 (coding NGL-2) as the most highly expressed synapse-related gene in trained compared with nontrained rats after HX injury. Immunofluorescent analysis demonstrated that NGL-2+ synaptic puncta markedly increased after training, suggesting that post-HX step training increased expression of Lrrc4 leading to an increase in the number of synapses in the lumbar spinal cord.
Live imaging using 2-photon laser scanning microscopy has shown that training a forelimb reaching task led to rapid and long-lasting changes in postsynaptic dendritic spines on the pyramidal neurons in the contralateral motor cortex, 16 indicating that motor training is closely associated with long-lasting synaptic reorganization. Locomotor training after spinal cord injury was also reported to have increased VAChT+ (vesicular acetylcholine transporter) or PSD-95+ (postsynaptic density protein 95) synapses and GABAergic (γ-aminobutyric acid) or glycinergic axonal terminals in lumber motor neurons.17-19 Previously, we demonstrated that treadmill-trained animals exhibited greater excitability of the lumbar motor neurons. This was reflected in the lower level of excitation thresholds for epidurally evoked potentials in hindlimb muscles, suggesting training-induced changes in interneurons and/or motor neurons. 5 In this study, we detected an elevated expression of Lrrc4 (coding NGL-2). Elevated expression of NGL-2 in rat neurons was linked to more PSD-95+ dendritic protrusions in vitro. 15 An NGL-2 knockdown reduced excitatory synapse numbers and currents, suggesting that NGL-2 can regulate the formation of predominantly excitatory synapses. 15 In the hippocampus, NGL-2 was also reported to be important for synapse development and regulation of excitatory transmission at synapses. 20 These findings suggests that higher expression of NGL-2 in trained rats may increase excitability of spinal neurons so that a lower level of epidural spinal stimulation is required to evoke a motor response. 5 An increase in the number of NGL-2+/SYP+ synaptic puncta in treadmill-trained rats is also consistent with greater numbers of excitatory synapses. This could be an important contributor to the greater excitability of neurons associated with improved motor performance.
The etiology of traumatic human SCI includes crush injuries accompanied with vertebral fractures and, more rarely, gun and knife injury.21-25 Of these, HX injury opens the dura and axotomy is followed by axonal die-back and reduced synaptic connections to motor neurons.26,27 Contusion injuries, however, tend to induce greater recruitment of inflammatory cells and activation of astrocytes, followed by secondary injury leading to demyelination, axonal damage and neuronal cell death without cutting dura.28-31 Following a spinal contusion, a significant increase in the expression of genes related to inflammation of lumbar spinal cord was observed, but no significant changes in expression levels of Lrrc4 were observed (nontrained vs trained: 3.33072 ± 0.02432 vs 3.41630 ± 0.10916, P = .752). 32 In both studies, terms associated with transcriptional activity, metabolism, biosynthesis, cytoskeleton, apoptosis, proliferation, immunity and inflammation, lysosome, ubiquitination, synapse, and angiogenesis were enriched in treadmill-trained rats. Particularly, signal transducers and activators of transcription 5b (Stat5b), DNA damage-binding protein 1 (DDB1) and cullin-4 (CUL4) associated factor 8 (Dcaf8), membrane associated ring-CH-type finger 6 (March6), and phosphodiesterase 2A (Pde2a) were included in terms related to inflammation (Stat5b), ubiquitination (Dcaf8 and March6), and intracellular signaling (Pde2a) in both studies (Supplementary Table 1). However, the majority of genes included in these GO terms are different between both studies. Moreover, GO terms enriched in treadmill-trained rats observed in this study were not found in previous microarray studies. These terms include: brain development, axonogenesis, dendritic spine organization, and generation of neurons.
The difference between the data derived from microarray analyses and our data could be attributed to several differences in experimental design such as treadmill training protocol, body weight of rats, and the tissue studied (the entire lumbar and sacral cord vs only L1 spinal cord). Moreover, RNA-seq is not perfectly comparable to microarray technologies at the gene level. Comprehensive gene expression analysis, such as RNA-Seq and microarray, however, provides the advantage that data can be analyzed without further animal experimentation since the data can be downloaded onto a database for future access. 33
In conclusion, we demonstrated that treadmill training after a thoracic spinal HX increases the expression of multiple synapse-related genes, with one of the most responsive being Lrrc4, and could play a primary role in changing synaptic structures. Our findings provide further insight as to how rehabilitation, after CNS injury, could facilitate activity-dependent synaptic reorganization. This activity-dependence could then play a key role in mediating the underlying activity-dependent mechanisms of training-induced network reorganization.
It is notable, also that these findings are consistent with the concept of neuronal group selection, which leads to a dynamic selective activation of different populations of synapses that transforms networks into different physiological states as has been proposed for networks within the brain. 34 Perhaps these types of changes could lead to the underlying activity-dependent mechanisms of improved coordination among motor pools and, thus, functional recovery after spinal cord injury.
Supplemental Material
Supplementary_table – Supplemental material for Locomotor Training Increases Synaptic Structure With High NGL-2 Expression After Spinal Cord Hemisection
Supplemental material, Supplementary_table for Locomotor Training Increases Synaptic Structure With High NGL-2 Expression After Spinal Cord Hemisection by Kazu Kobayakawa, Kyleigh Alexis DePetro, Hui Zhong, Bau Pham, Masamitsu Hara, Akihito Harada, Jumpei Nogami, Yasuyuki Ohkawa and V. Reggie Edgerton in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors thank Czarina Juan-Sing and Nicole Zoghby for their assistance during surgeries and experiments, and Sharon Zdunowski for her technical support.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of the article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: VRE holds shareholder interest in NeuroRecovery Technologies and holds certain inventorship rights on intellectual property licensed by The Regents of the University of California to NeuroRecovery Technologies and its subsidiaries.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Dana & Albert R. Broccoli Charitable Foundation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
