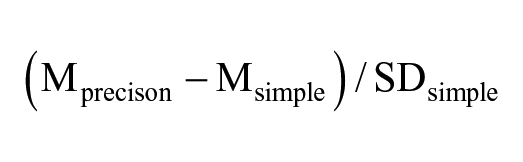Abstract
The corticospinal tract has been shown to be involved in normal walking in humans. However, its contribution during more challenging locomotor tasks is still unclear. As the corticospinal tract can be a potential target to promote gait recovery after neurological injury, it is of primary importance to quantify its use during human walking. The aims of the current study were to: (1) quantify the effects of precision walking on corticospinal excitability as compared to normal walking; (2) assess if corticospinal modulation is related to task difficulty or participants’ performance. Sixteen healthy participants walked on a treadmill during 2 tasks: regular walking (simple task) and stepping onto virtual targets (precision task). Virtual targets appeared randomly at 3 different step lengths: preferred, and ±20%. To assess corticospinal excitability, 25 motor evoked potentials (MEPs) were recorded from the tibialis anterior muscle in each task during walking. Performance for each participant (global success score; % of target hit) and task difficulty related to step length adjustments (success score for each step length) were also calculated. MEP size was larger during the precision task in all participants (mean increase of 93% ± 72%; P < .05) compared to the simple task. There was a correlation between MEP facilitation and individual performance (r = −.64; P < .05), but no difference in MEP size associated with task difficulty (P > .05). In conclusion, corticospinal excitability exhibits a large increase during the precision task. This effect needs to be confirmed in neurological populations to potentially provide a simple and non-invasive approach to increase corticospinal drive during gait rehabilitation.
Introduction
Gait is a complex sensorimotor task involving interactions between 3 different levels of neural control. First, central pattern generators in the spinal cord produce a basic rhythmic muscle activation pattern that is modulated by sensory feedback and supraspinal centers to adapt to the environment. 1 Second, sensory feedback, provided by visual, vestibular, and somatosensory inputs, allows for reactive control to environmental stimuli. 2 Finally, supraspinal pathways such as the corticospinal tract modify the basic locomotor rhythm using anticipatory and volitional control, more particularly during visually guided gait modifications and fine movement adjustments.3,4 Moreover, considering the plastic adaptive capacity of the central nervous system, the corticospinal tract has also been suggested as a potential target to restore gait control after neurological injury such as spinal cord injury 5 or stroke. 6 A precise quantification of the corticospinal tract use during human walking is therefore necessary to provide enhanced rehabilitation strategies for people with neurological conditions. 7
Most evidence regarding the corticospinal tract contribution to voluntary gait control comes from animal models. Studies in cats and monkeys have shown that after a corticospinal tract lesion, while only small changes in basic walking ability are reported,8 -10 larger deficits are present when challenging walking environments need to be negotiated, such as stepping over obstacles. 8 Similar results were also observed with motor cortex inactivation.11,12 Single cell recordings in behaving animals have corroborated these findings and shown that cell discharge activity in the primary motor cortex increases when locomotion requires precise step placement, for example when cats walk on a ladder 13 or over an obstacle.14,15 More specifically, the discharge activity is increased just prior to task execution, suggesting that the motor cortex contributes to the voluntary modifications in the muscle activation pattern required to produce the changes in limb trajectory during the precision task.
In contrast to animal models, studies in human using neuroimaging (reviewed in Barthelemy et al 16 ), subthreshold transcranial magnetic stimulation (TMS), 17 electroencephalography – electromyography (EEG–EMG) coherence,18,19 and metabolic tracers in the primary motor cortex20,21 suggest that the corticospinal tract contributes substantially to simple level walking. This is supported by clinical observations of people with an incomplete spinal cord injury presenting important walking deficits after injury that are related to the extent of corticospinal damage.22,23 TMS studies have shown that corticospinal excitability is dynamically modulated during walking (ie, in a phase-dependent manner),24,25 reinforcing the idea of a substantial contribution of the corticospinal tract in humans.
Knowing this corticospinal contribution to normal gait, what happens when humans need to negotiate more complex walking situations? In a precision stepping task, Schubert et al 26 have shown significant but small changes in MEP size in the Tibial Anterior muscle compared to normal gait. The limited amplitude of these changes is somewhat surprising considering the animal model literature. It could potentially be due to the fact that the required gait modifications in that study were also very small, but it remains to be determined whether larger gait modifications require a larger change in corticospinal drive. This question is relevant considering that the corticospinal tract represents a potential target to enhance neuroplasticity to improve gait recovery after neurological impairments. It is therefore important to better understand how and how much it can be recruited/further engaged for rehabilitation purposes.
The aim of the current study was therefore to determine, using a paradigm easily implementable in the clinical setting, the effects of a visually guided precision walking task requiring step length adjustments on corticospinal excitability in healthy adults. Secondary aims were to assess the relationship between changes in corticospinal response and: (1) task difficulty; and (2) global motor performance.
Materials and Methods
Participants
Nineteen healthy participants (27 ± 3.4 years old, 9 females, 174.5 ± 8.4 cm, 68.1 ± 10.4 kg) were recruited to participate in 1 session at the Center for Interdisciplinary Research in Rehabilitation and Social Integration (Cirris, Quebec City, Canada). All participants were right foot dominant according to the Waterloo Footedness questionnaire. They were excluded if they presented any contraindications to TMS, pain or other neurological/musculoskeletal conditions that could affect their performance in the walking tasks. The study was approved by the local ethics review board (CIUSSS-CN #2016-511) and all participants provided written informed consent.
General Protocol
The general protocol was adapted from Choi et al. 27 Participants came to 1 assessment session of 1 hour. Corticospinal excitability was measured while participants performed 2 different locomotor tasks on an instrumented treadmill (instrumented split-belt treadmill, Bertec, Ohio, USA): a simple task (regular walking) and a precision task (walking onto virtual targets retro-projected on a large screen in front of them). These 2 tasks were performed in counterbalanced order. The session started with a familiarization to treadmill walking (3-4 minutes). Then, participants were asked to walk normally on the treadmill at 1 m/s while the virtual reality system (8 Vicon cameras + Vizard VR software; see below) measured their step length (mean of anterior-posterior distance between left and right foot for 24 consecutive steps). Each contact was detected using both the VR system and the treadmill force plates. Finally, a short familiarization with a simplified version of the actual precision task was performed, where participants only had to hit 1 left and 1 on right target while walking.
In the simple task, participants walked on the treadmill at 1.0 m/s with the real-time position of their feet projected on the screen. No targets were presented.
In the precision task, participants walked also at 1.0 m/s and had to adjust their locomotor pattern to walk specifically on virtual targets (squares of 30 cm; actual screen size) retro-projected on a large screen facing them (Figure 1A). During this task, the distance between targets (sagittal plane) varied from one step to the next. The right and left step lengths were modified in a variable manner between legs (asymmetrically) and across strides. Three different distances were presented to the participants (block randomization): (1) a distance equal to the normal step length of each participant (100%); (2) a shorter distance (80% of the step length), and (3) a longer distance (120% of the step length) (Figure 1B). A total of 360 targets were presented to the participants during the task (180 to each foot). Targets were initially shown in red and turned green when hit. A success score (%), only visible to the experimenter was calculated in real time.
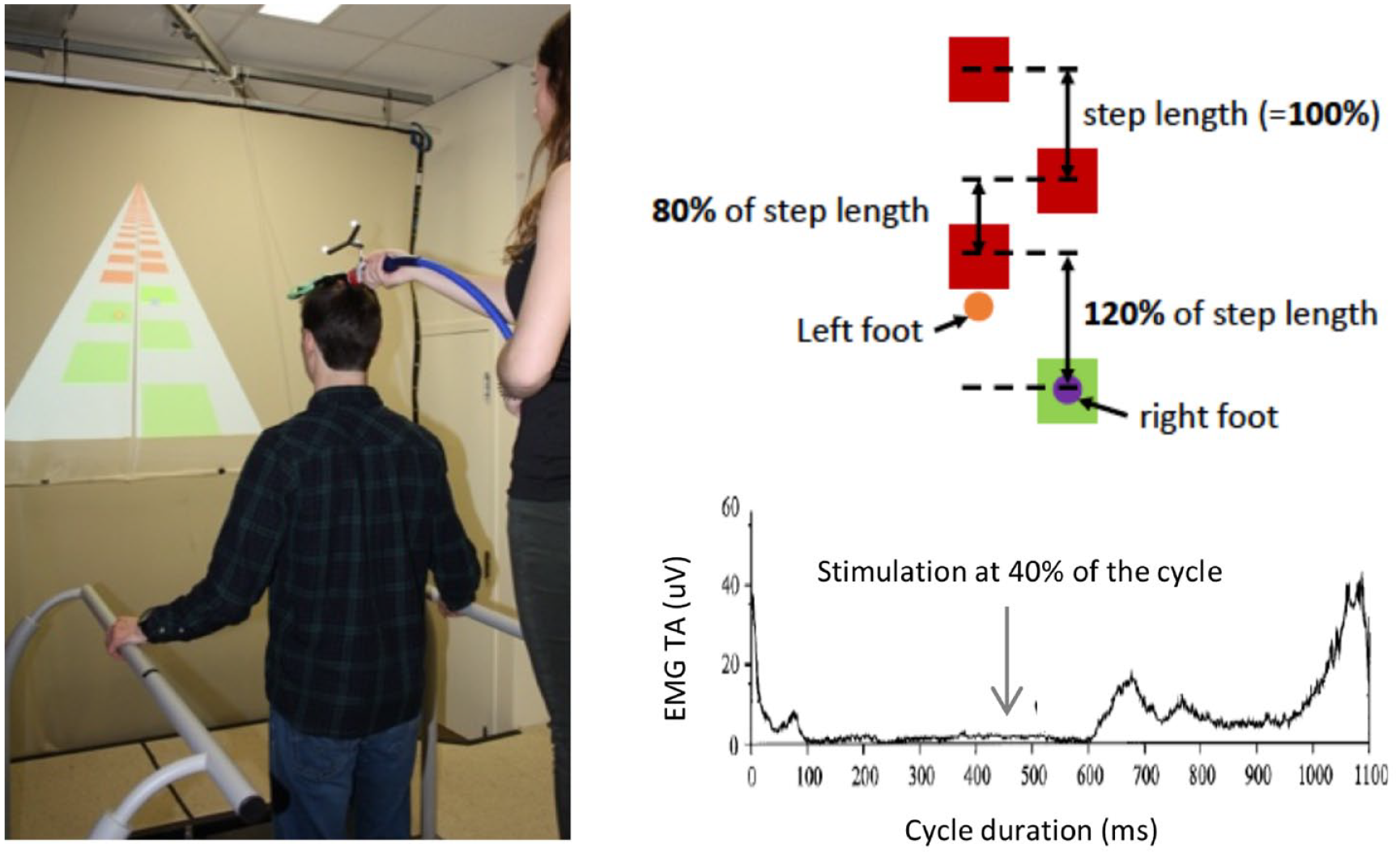
(A) Experimental set-up. Participants had to walk on virtual targets projected on a screen. During walking, transcranial magnetic stimulation (TMS) was applied by an experimenter standing on a platform behind the participant. (B) Precision task protocol. The targets were separated by different distances (80%, 100%, or 120% of participant’s step length) in a pseudo random order. The targets were initially red and turned green when hit. (C) TMS stimulation in the gait cycle. Motor evoked potentials were induced by single pulse TMS in the late stance of the gait cycle (40%).
Virtual Environment and Real-Time Feedback of Foot Position
During the 2 tasks, all participants walked on a treadmill at a speed of 1.0 m/s (imposed gait speed), with their hands lightly touching lateral support bars to limit fore-aft movement and postural changes. A custom-made virtual environment was created using Vizard 4.0 (Worldviz, Santa Barbara, USA) and projected on a large screen (150 × 250 cm; located 1.65 m in front of the participants) using a video projector (SANYO, Osaka, Japan).
Real-time position of the feet was also projected on the screen, where each foot was represented by a small sphere (4.5 cm diameter; actual screen size) centered at heel position as measured with a motion capture system (8 camera-VICON mocap system, CA, USA).
Corticospinal Excitability Assessment
Corticospinal excitability was measured using transcranial magnetic stimulation (Magstim rapid 2 ; figure-of-eight shaped bat-wing coil, 17 Whitland, UK) during the 2 walking tasks. This coil model is optimized to stimulate deeper cortical structures, such as leg area of the motor cortex. 28 Single-pulse stimulations were delivered over the right motor cortex and motor evoked potentials (MEP) were recorded in the left tibialis anterior muscle (TA). As this study was designed in order to be adapted to neurological populations, all TMS settings were elicited during sitting to minimize standing time. Hotspot and resting motor threshold (rMT) were then determined when participants were sitting with knees flexed at 90° and ankles in neutral position (no TA activity). The hotspot was identified as the optimal zone to evoke the largest MEPs amplitude in the TA at the lowest intensity. The rMT was determined as the lowest stimulation intensity required to evoke a MEP in 50% of trials. During walking, stimulation intensity was adjusted at 110% of rMT. Coil position was maintained during walking by an experimenter standing on a platform behind the treadmill. A neuronavigation system (Brainsight, Rogue research, Montreal, QC, Canada) provided coil position feedback on a computer screen, allowing the experimenter to make fine real-time adjustments as needed. For each of the 2 tasks, 25 MEPs were evoked during late stance (40% of the gait cycle; based on the average of the last 3 left heel contacts detected by the treadmill force plates, see Figure 1C). To avoid anticipation by participants, stimulations occurred on random gait cycles, on average every 5 strides (range 3-7). Note that only 1 period (late stance) of the gait cycle was targeted for 3 main reasons. First, as MEP size is only slightly modulated by the phase of the gait cycle when stimulation is above rMT,24,25 late stance was chosen because it corresponds to a moment where TA activity is minimal and similar across the 2 tasks, thereby limiting the influence of background EMG changes on MEP modulation and simplifying data interpretation. Second, late stance represents an important timing for the preparation of the swing movement, and could therefore be a key phase for promoting clinically-relevant plasticity in patients. 29 Finally, this paradigm was designed to be eventually performed by neurological populations, making it necessary to minimize test duration.
Regarding the reliability of the technique, it was previously showed that TMS measurements elicited in the TA muscle were reliable in a healthy population. 30 In this study, all stimulations were delivered during the same laboratory session for each participant, under the same conditions with the same electrodes’ placement between the 2 walking tasks. In addition, the tasks duration was designed to limit potential effects of fatigue. Together, these elements further reduced potential variations between the 2 tasks.
EMG Recordings
EMG activity of the left Tibialis Anterior (ankle dorsiflexor; TA) was measured using a pair of surface adhesive Ag/AgCl electrodes (Kendall Medi-trace 200, Covidien) placed on the skin over the belly of the muscle according to the SENIAM recommendations. 31 The ground electrode was positioned on the left patella. EMG signals were amplified (×1000) and band-pass filtered (10-500 Hz) using a custom-made EMG amplifier (NTI, Quebec City) prior to acquisition using custom-written software. All signals were digitized at a sampling frequency of 1000 Hz.
Analysis
Custom-made software written in MATLAB (The MathWorks Inc., Natick, USA) was used for data analysis. To quantify corticospinal excitability, mean area of MEPs measured as the area under the rectified EMG; 30-ms window starting at 30 ms post stimulation artefact was compared across tasks. 32 In order to compare across participants, MEPs latencies were measured as the time between stimulation artefact and beginning of the MEP and normalized to each participant’s height ((ms/cm × 100) 33 ). They were also compared across tasks. Mean background EMG amplitude (window of 50 ms preceding every TMS stimulation) was also compared across tasks. A global success score, representing participant’s performance, was calculated for each participant as the percentage of successfully hit targets during the precision task. Specific success scores were also calculated for each step length (80%, 100%, and 120%) to assess the relative difficulty across the 3 conditions. Coil position error, as measured by the neuronavigation software at the time of each stimulation, was compared across tasks. It was defined as the average difference between coil position at the time of stimulation and the hotspot (in millimeters). Finally, considering that the corticospinal pathway is likely already engaged during simple walking (see Introduction), an analysis was performed to verify that participants with larger MEPs amplitude during simple walking could still increase corticospinal excitability during precision walking, by plotting percentage of corticospinal excitability increase during precision walking as a function of MEPs size during simple walking (see Results).
Statistical Analysis
Statistical analyses were performed with SPSS version 24 (SPSS Inc., Chicago, IL, USA). As data did not follow a normal distribution (Shapiro–Wilk test) nonparametric tests were used. Coil position, MEP timing within the gait cycle, background EMG and MEPs size (mean area) and latency were compared between locomotor tasks (simple and precision) using Wilcoxon matched-pairs signed rank test. Intra-subject analyses were also performed for the difference in background EMG, MEPs size, and latency during the simple and precision tasks using Mann–Whitney test. The effect size of the change in MEPs size was calculated with delta of Glass as follow:
where M represents the mean MEPs size, and SD represents the standard deviation. Effect sizes were interpreted as follow: no effect (0-0.19); small effect (0.2-0.49); intermediate effect (0.5-0.79) and large effect (>0.8). 34 Relationships between change in MEPs size and TA background EMG, success score and MEPs size during simple task were determined with Spearman correlation coefficients. Correlation results were interpreted as follow: negligible (0-.3); low (.31-.5); moderate (.51-.7); high (.71-.9); or very high (.9-1.0). 35 Friedman test with Dunn’s multiple comparisons analyses were used to compare success scores and MEP increase relative to step length adjustments. A significant level of .05 was used.
Results
Nineteen participants were recruited for this study. Two were excluded from the analysis as it was not possible to induce MEPs in TA. Another participant was excluded due to large lateral body sway during walking that made TMS coil position too difficult to maintain stable. Finally, due to technical issues, it was not possible to calculate the success score for 1 participant.
Methodological Consideration: MEP Timing Within the Gait Cycle, Coil Displacement, Background EMG, MEP Modulation Capacity According to Raw MEP Size
Analysis showed that MEP timing within the gait cycle were very precise (41% ± 2%, and 40% ± 2% for the simple and precision tasks, respectively) and not significantly different (P = .1).
No difference was found in coil position error between the simple and precision tasks (simple task: 2.28 ± 0.77 mm; precision task: 2.32 ± 0.67 mm; P = .77) (Figure 2A).
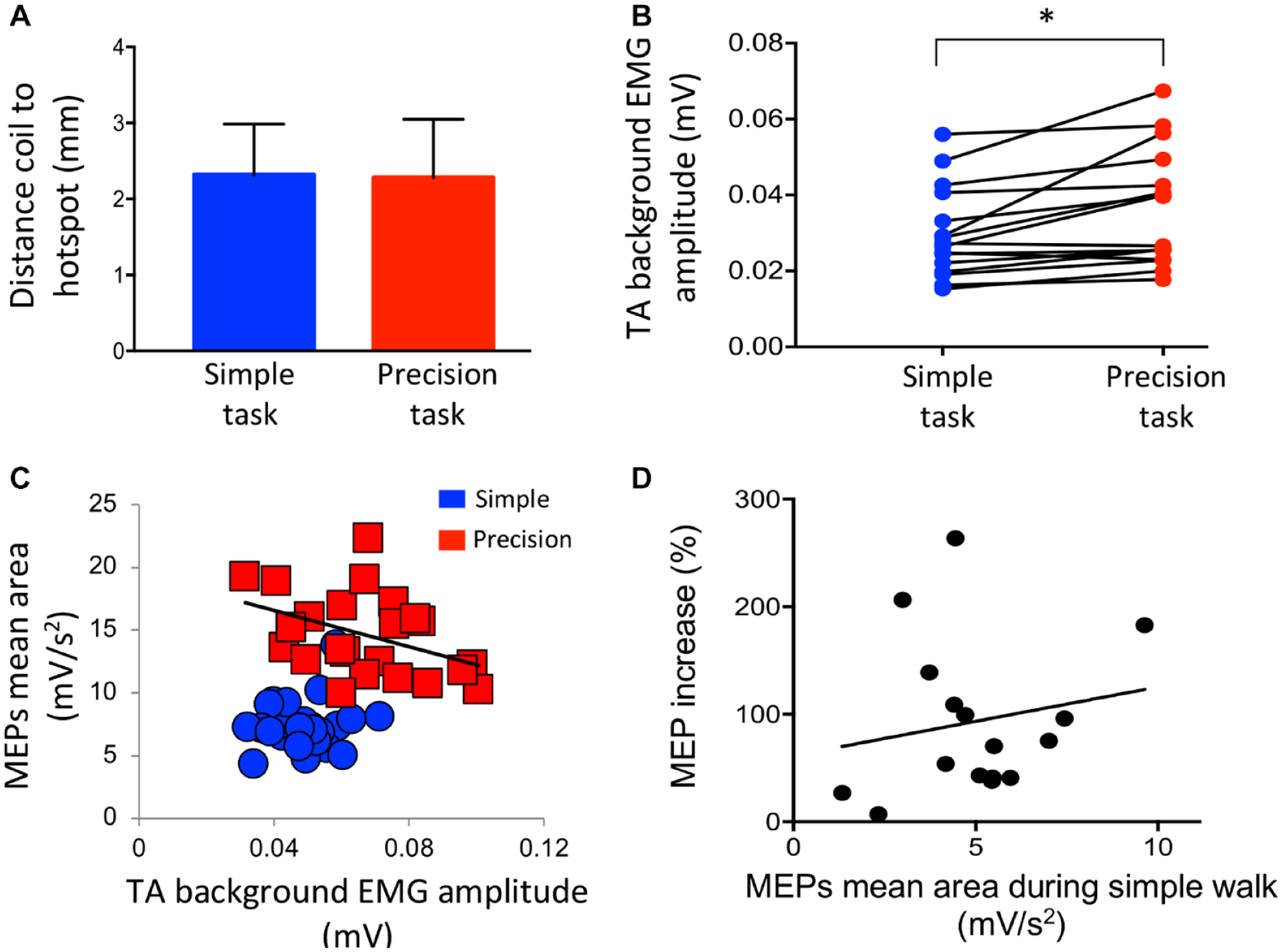
Methodological considerations. (A) Distance between the transcranial magnetic stimulation coil and the hotspot during the simple (blue) and precision (red) tasks. (B) Difference between the simple (blue) and precision (red) tasks for the tibialis anterior (TA) background EMG amplitude for the group. (C) Motor evoked potentials (MEPs) mean area during simple (blue) and precision (red) tasks as a function of their respective background EMG for 1 participant. (D) Correlation between MEPs mean areas (mV/s2) during the simple walk and the percentage of MEPs increase (%) during the precision task. Each dot represents 1 participant.*P < .05.
A statistically significant difference was found in TA background EMG amplitude between the simple and precision tasks (simple task: 30 ± 12 µV; precision task: 36 ± 15 µV; P = .0003) (Figure 2B). As background EMG is an important aspect to control in TMS studies, intra-subject analyses were performed, and they showed a statistically significant difference between precision versus simple task for 7 of the 16 participants (P < .05).
To document the potential effect of background EMG change on MEPs size in these 7 participants, a correlation between MEPs size during precision task and background EMG was performed. Only 1 significant correlation was measured in 1 participant and it was small and negative (r2 = .17; r = −.41; P = .02). These results suggest that background EMG changes have little effect on TA MEP size (no change in 6/7 participants and a small reduction in 1 participants). In addition, a graphical analysis of the MEP versus background EMG relationship was performed. 36 Figure 2C shows an example for 1 participant where a significant difference was found. It can be seen that, at similar EMG background levels (here between 30 and 70 µV), MEPs during the precision task were significantly higher than during the simple task (P < .0001).
Figure 2D shows that there was no correlation between MEPs size during the simple task and the percentage of MEP increase during the precision task for the group (r = .11; P = .34). Therefore, even the participants with the larger MEPs during simple walking could still increase corticospinal excitability during precision walking.
Comparisons of MEPs Between Locomotor Tasks
There was a large facilitation of MEPs during the precision task (9.9 ± 6.0 mV/s2) compared to simple gait (5.0 ± 2.0 mV/s2); (P < .0001; effect size Glass Δ = 2.43), with a mean increase of 93.4% ± 71.9% (Figure 3). Intra-subject analyses revealed significant difference for 14/16 participants between the 2 gait tasks (all P < .05). No significant difference was observed for the normalized MEPs latency between the precision (16.4 ± 0.82 ms/m) and simple tasks (16.1 ± 0.75 ms/m; P > .05). Intra-subject analyses revealed no significant difference in MEPs latency for the 16 participants between the 2 gait tasks (P > .05).
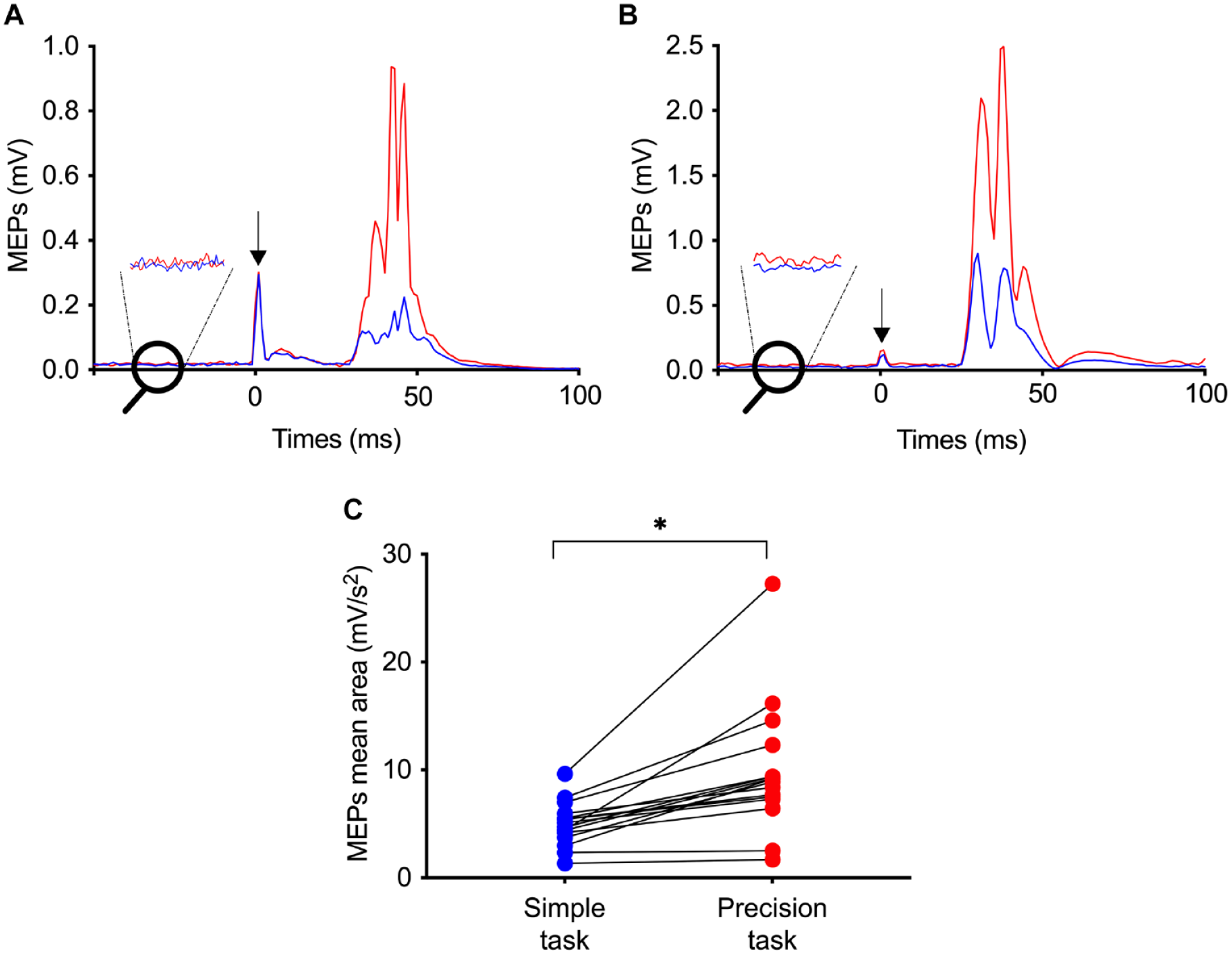
Corticospinal excitability modulation during the simple task (blue) and the precision task (red). (A) Rectified motor evoked potentials (MEP) amplitude (mean of the 25 MEPs during the entire precision task) modulation in the 2 tasks for 1 representative participant with no background EMG amplitude, and (B) for 1 representative participant with a different background EMG amplitude. Black arrows indicate the timing of transcranial magnetic stimulation pulse. (C) MEPs mean area during the 2 tasks for the 16 participants. Each dot represents 1 participant.*P < .05.
Global Performance
Performance Evolution Over Time
A gradual performance improvement (mean success score) was observed during the precision task execution, starting at 79.7% (first target) and reaching 91.9% by the end (last target) (Figure 4A; group results). A significant difference was also found between the first and last 10 MEPs during the precision task for the group (reduction of 18.88%; P = .0004). Importantly, the last 10 MEPs remained significantly increased compared to the 10 last MEPs measured during the simple task (33.64% higher; P < .0001) (Figure 4B).
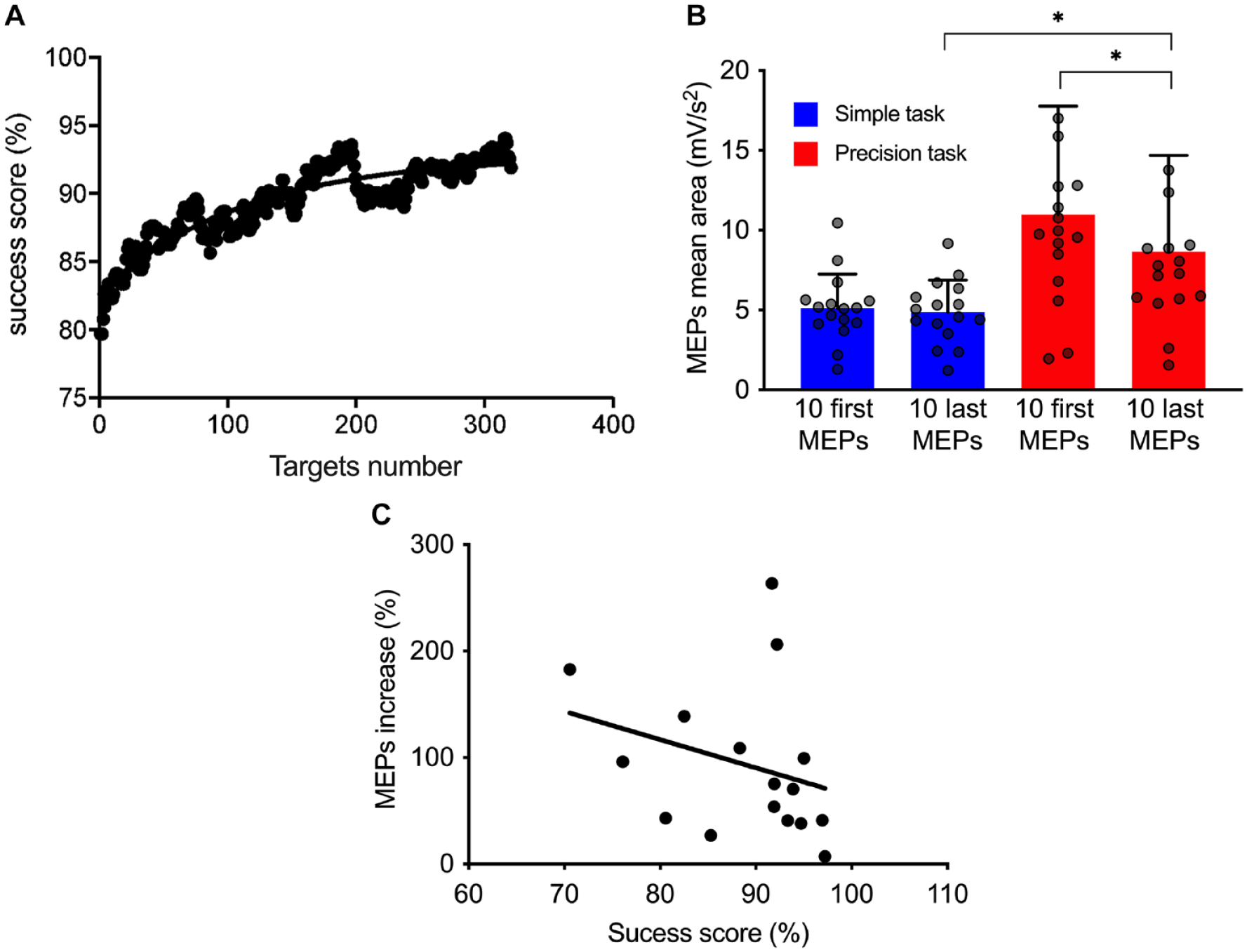
(A) Evolution of the success score (mobile average of 40 data) as a function of target number during the precision task (mean of 15 participants). A trend curve was plotted and the time constant (τ) was identified at the target 111. (B) Comparisons between the first 10 motor evoked potentials (MEPs) mean area and the last 10 for the simple (blue) and precision (red) tasks. (C) Correlation between the MEPs size increase and global performance (success score). Each dot represents 1 participant.*P < .05.
Relationship Between MEPs Increase and Global Performance
As showed in Figure 4C, there was a negative correlation between MEP percent increase and global performance (r = −.64; P = .006).
Step Length Adjustments
During the precision task, the average success scores were 74.9% ± 15.94%, 93.3% ± 7.72%, and 96.6% ± 3.31% for the 80%, 100%, and 120% of step length targets, respectively. Significant differences in performance were observed only between 80% and 100% (P = .003) and between 80% and 120% (P < .0001) (Figure 5A), but not in MEPs amplitude (80%: 10.8 ± 9.16 mV/s2; 100%: 10.46 ± 8.81 mV/s2 and 120%: 9.1 ± 6.15 mV/s2; P = .47) (Figure 5B).
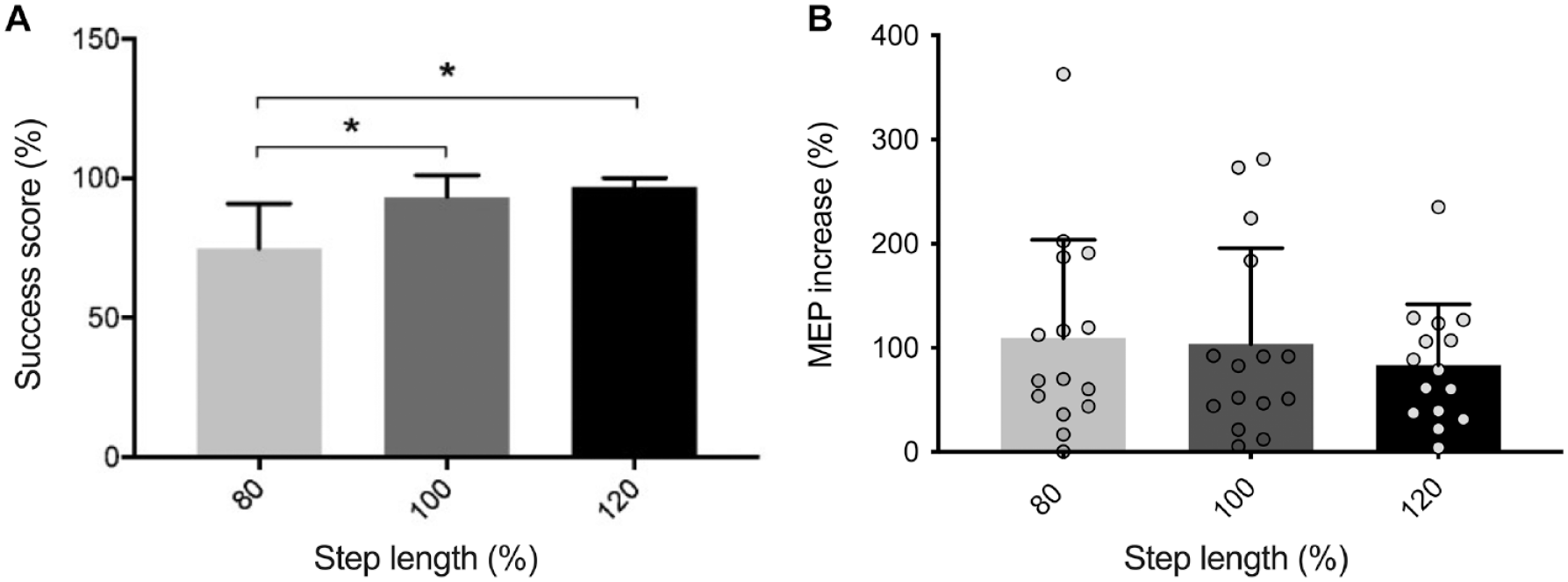
(A) Group success score during the precision task for each step length adjustments. (B) Motor evoked potentials (MEPs) increase as a function of required step length. Each dot represents 1 participant.*P < .05.
Discussion
This study is one of the first to quantify corticospinal excitability during the execution of a complex walking task in humans. The results suggest that corticospinal excitability is enhanced and maintained during precision walking compared to simple walking. This increase seems to be related to each participant’s performance while the relative difficulty between the 3 step length adjustments seems to have no effect. This increase is not simply due to motion of the coil relative to the hotspot or changes in background EMG.
Corticospinal Excitability is Increased During Precision Walking
In humans, it is well known that the corticospinal tract is involved during simple, level treadmill walking. 16 Here, we showed that it is further involved in a task that required precise foot placement, suggesting an additional contribution of this pathway in voluntary gait control, and more particularly in fine movement control as previously reported in animal studies. 4 To our knowledge, only 1 prior study measured TMS during visually guided locomotor pattern modifications in humans (step on virtual targets projected on a treadmill). The effects reported were small, probably because this task only required small adjustments in the locomotor pattern (5 cm of lateral targets displacements) and did not require precision gait modifications, but was likely more a visual task, thereby not unraveling the full ability of the corticospinal system to adapt ongoing gait movements. 26
Previous studies have measured corticospinal excitability during another type of challenging gait, that is, walking adaptation to a resistive force.36,37 Similarly, the authors reported an increase in corticospinal excitability when participants walked with a resistive force. The current study reports a much larger increase in MEPs size, however. This could be due to the fact that our visually-guided task further involves the corticospinal tract, more particularly the motor cortex and the posterior parietal cortex (PCC),4,38 compared to adaptive tasks that may preferentially recruit other structures such as the cerebellum.39,40
Corticospinal Excitability Increase is Maintained Over Time During the Task
During the precision task, the mean success score increased over time within a single session. This time course likely reflects a fast-learning mechanism.41,42 MEP size was initially very large and then stabilized at a lower level that remained above simple walking, however. As described previously in brain imaging studies, fast learning in motor tasks initially increases motor cortex and dorsolateral prefrontal cortex activity, followed by a decrease over time.43,44 In TMS studies, it was shown that corticospinal excitability is increased after a fast-learning effect in a manual task.45 -48 In this study, it is possible that the global execution of the precision task increased the corticospinal excitability compared to the simple task, and that this excitability was enhanced by a fast-learning effect at the beginning of the precision task. This could explain why MEPs were higher at the beginning compared to the end of the precision task, and also explain why MEPs at the end remained higher than MEPs during the simple task. An alternate possibility is that initial walking in the precision task required more attentional demand, 49 but our experimental paradigm did not allow to distinguish between the 2 potential explanations (see study limitations).
Influence of Performance and Task Difficulty on Corticospinal Excitability
A global success score was calculated for each participant, in order to investigate if performance could explain inter-subject differences in MEPs size changes. The results showed a moderate significant correlation between the 2. Therefore, it seems that corticospinal excitability can be modulated depending on participant’s success score, and more precisely results showed that participants with lower success scores (ie, likely more difficulty to execute the task) had a larger corticospinal (CS) excitability increase. In this case, it seems that the complexity of the task could further increase corticospinal excitability as previously reported for finger tasks with different complex movements and different hand force-matching difficulties,50,51 and therefore increasing task difficulty could be important for maximizing rehabilitation training. Alternatively, previous studies also reported that a progressive increase of corticospinal excitability sustains the early stage of learning. 52 This peak of increased corticospinal excitability has been associated with a change in motor strategy (less reactive and more anticipatory).52,53 It is thus possible that a learning effect induced by the complex task is at the origin of this significant negative correlation, with an increase of corticospinal excitability reflecting a progressive learning effect even more important for participants facing difficulties to walk on the targets. In addition, it is possible that participants for whom the precision task was considered as particularly challenging (ie, with the lowest success scores) have experienced a higher level of stress than participants who performed well on the task. Consequently, a real-time feedback of foot position was given to participants, with targets turning green only when hit. Thus, even if the participants were not informed of their success score, they could see in real time during the task the number of missed targets. Previous studies provide evidence that negative emotional state, pressure, and anxiety increase MEP amplitude measured at rest.54,55 Therefore, it is possible that in addition to the increase of corticospinal excitability due to the precision task (vs the simple task), being confronted with a large number of missed targets induces for these participants a form of stress that further increased the MEP amplitude, thus leading to the negative correlation observed between the final score and the MEP amplitude increase.
We also calculated a success score specific to each step length adjustment during the precision task to know if within the task, it was possible to modulate corticospinal excitability according to step length. The results showed that it was more difficult to execute shorter steps (80% of step length; success score of 75.5%) as also reported by previous studies.56,57 However, no statistical differences were reported in MEPs increase for the 3 distances. Hoogkamer et al 57 have shown that the strategy used to make longer steps is similar to that used for normal steps whereas shorter steps require quick movement changes that challenge postural stability. The absence of a difference in MEPs size between 80% to 100% and 80% to 120% is therefore quite surprising and could be due to the fact that longer and shorter steps were continuously performed in a pseudo-randomized order, thereby limiting our ability to discriminate specific step-size related changes in CS excitability.
Together, these results suggest that the main factor for cortical excitability increase is going from normal walking to precision stepping more than modulating each step length.
Potential Influence of Other Neural Structures
In this study, we measured corticospinal excitability using TMS over the motor cortex. Animal studies have shown that the motor cortex is one of the central nervous system (CNS) areas involved in voluntary gait modifications, among others, such as the PCC, the somatosensory cortex, and the cerebellum.4,58 The PPC is known to contribute to the planning of locomotion during visually-guided tasks, 38 while the cerebellum and somatosensory cortex contribute in global motor planning. 58 In addition, a fNIRS study showed that the prefrontal cortex could also be involved during a precision walking task in human, 59 suggesting that precision walking requires complex integration from different structures in order to make the appropriate movement.
Moreover, as TMS over the motor cortex can also activate sub-cortical and spinal structures,17,60 and considering the importance of these structures in neural control of walking, 61 it is therefore possible that the corticospinal excitability facilitation observed during our precision task involved these regions. 62 However, the results obtained in the present study cannot conclude to the relative contribution of these structures.
Potential Clinical Relevance
Results of the present study show that it is possible to increase corticospinal excitability during a precision walking task. Indeed, we demonstrated that corticospinal excitability is enhanced for all participants and that the response is on average twice as large as during normal walking. These observations suggest that humans have the capacity to increase their corticospinal excitability to allow adequate movement control in complex walking tasks requiring precise foot placement. The corticospinal tract is a neural structure often targeted in gait rehabilitation after neurological injuries to improve gait recovery. It is therefore important to measure in future studies if individuals with such lesions have also the capacity to increase corticospinal excitability depending on the executed task, and if it the case, to measure the long-term effects of this capacity on gait circuitry plasticity.
Strengths and Limitations
The originality of this work was to quantify corticospinal excitability during a visually-guided precision walking task in humans. Another contribution of this study was to evaluate if changes in performance or task complexity affected MEP size, to better understand the origin of the reported increase in corticospinal excitability.
Some limitations were also present. Indeed, even if we demonstrate a large effect in corticospinal excitability changes, the number of participants was small and age range narrow, potentially limiting generalizability of the findings to other age groups. Moreover, in order to reduce inter-individual variability in corticospinal excitability, MEPs could be normalized using maximal M-wave (Mmax) in future studies.63,64 Another limitation that should be noted is that the level of attention during the 2 gait tasks was not controlled and could have an impact on corticospinal excitability, especially at the beginning of the precision task when participants walked on the targets for the first few times. Finally, it was not possible to induce MEPs in 2 participants, probably due to methodological challenge associated with stimulation of a lower limb muscle, as shown in other studies. 65 To overcome this limitation, future studies should probably define TMS settings in a standing position, since corticospinal excitability in the TA muscle has been shown to be higher during standing as compared with that during sitting. 66 Furthermore, previous studies using TMS in neurological populations have shown a similar rate of success at eliciting MEPs as in healthy controls. We therefore anticipate a similar success rate at eliciting MEP during walking in neurological populations.
Another participant was excluded because of a large body sway during walking and his discomfort to walk on a treadmill at 3.6 km/h. Considering that this study was designed to later be performed in neurological populations, these limitations need to be considered for future studies. However, as patients walk on average more slowly than healthy controls, if treadmill gait speed is adjusted to patients’ capacity, this will help to limit the effect of body sway on the stable support of the TMS coil over the head while walking.
Finally, corticospinal excitability was only measured in the TA muscle, and we did not consider the soleus or rectus femoris muscles that are also involved when walking. At this stage, the results obtained cannot be generalized to all muscles and deserve further investigations.
Conclusion
This study showed an important increase in corticospinal excitability during a visually-guided precision walking task compared to normal walking in humans. We demonstrated that it is possible to increase the involvement of the corticospinal tract with an easy to use, non-invasive virtual reality task. This effect needs now to be confirmed in neurological populations using the same paradigm. If confirmed, visually-guided precision walking may have a valuable potential in gait rehabilitation, and provide a new tool to enhance corticospinal drive to optimize gait recovery after CNS lesions such as spinal cord injury or stroke.
Footnotes
Acknowledgements
We would like to thank Nicolas Robitaille and Steve Forest for their technical support throughout this project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Traumatology Research Consortium from the Fonds de Recherche du Québec-Santé (FRQ-S) and the ERA-Net Neuron. Charline Dambreville’s doctoral studies was supported by studentships from Universite Laval, Neuroscience Thematic Research Center, Centre for Interdisciplinary Research in Rehabilitation and Social Integration (Cirris), and from Fonds de recherche du Québec Santé (FRQ-S). Cécilia Neige’s graduate studies were supported by a studentship from Cirris. Catherine Mercier is supported by an Emeritus Research Scholar Award from Fonds de recherche Québec – Santé (FRQS).