Abstract
Objective. To systematically review the literature with meta-analysis to determine whether persistence of sensorimotor or physiological impairment exists between 4 weeks to 6 months post mild traumatic brain injury (mTBI), and assign level of evidence to findings. Method. The databases PubMed, pscyINFO, SPORTdiscus, Medline, CINAHL and Embase were searched from inception to November 2016 using a priori inclusion criteria. Critical appraisal was performed, and an evidence matrix established level of evidence. Meta-analysis of pooled results identified standardized mean difference (SMD) and 95% confidence intervals (95% CI) between mTBI and healthy controls for a variety of physiological and sensorimotor indicators. Results. Eighteen eligible articles, with a mean quality score of 15.67 (SD = 2.33) were included in the final review. Meta-analysis of center of motion variable; maximal mediolateral center of motion/center of pressure separation distance SMD [95% CI] approached significance at (−0.42 [−0.84, −0.00], I2 = 0%) for dual task, level walking indicating a potential reduction in maximal mediolateral excursion during gait in the mTBI group compared to healthy controls. Significantly reduced variability in the standard deviation of heart beat intervals was observed in the mTBI group (−0.51 [−0.74, −0.28], I2 = 0%). Overall, significant group differences in 36 sensorimotor and physiological variables (eg, balance, gait velocity and motion analysis outcomes, various oculomotor tasks, as well as heart rate variability frequency domains) were identified. Conclusion. Findings demonstrate that persistence of sensorimotor and physiological changes beyond expected recovery times following subacute mTBI in an adult population is possible. These findings have implications for post-injury assessment and management.
Keywords
Introduction
Recent literature has highlighted that traditional measures for mandating recovery post mild traumatic brain injury (mTBI), also known as concussion, such as neurocognitive assessment and resolution of self-reported symptoms may have limited clinical utility.1-3 Individual studies investigating sensorimotor and physiological outcomes demonstrate some evidence of persisting impairment beyond the expected recovery time for mTBI of 10 to 14 days in adults, regardless of reported symptoms.4-6 Such findings are reflected in changes to current international sports concussion position statements 1 as well as revised mTBI management protocols,2,3 where routine incorporation of physiological and sensorimotor assessment is now recommended.1-3
Nevertheless, synthesis of findings related to sensorimotor and physiological impairment post mTBI is challenging, with few meta-analyses undertaken. Differences in age-related response to injury7-9 indicate that inclusion criteria should be limited to a specific age range or subgrouping. Furthermore recurrent injuries have protracted recovery times10,11 and should be considered separately. Uncertainty related to prognosis following complicated (vs noncomplicated)12,13 as well as blast-related injuries,14,15 suggests that outcomes following these specific injury types also be considered in isolation. Additionally, the need to separate injury definition and diagnostic criteria as either mild traumatic brain injury or concussion remains debatable.1,16 Therefore further synthesis of studies investigating persisting sensorimotor and physiological impairments beyond expected recovery time in adult uncomplicated mTBI would be clinically informative.
The primary aim of this study was to determine if evidence exists for persisting sensorimotor and physiological impairment beyond expected recovery time following uncomplicated mTBI in adults. Where possible we assigned level of evidence for any observed individual sensorimotor and physiological impairments. A secondary aim was to investigate whether observed impairments are accompanied by reported symptoms, or if impairment may exist in the absence of reported symptoms.
Methods
We conducted our review according to a clear a priori criteria (PROSPERO Registration Number: CRD 42016037396) as per PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines.17,18 A single amendment was submitted in June 2017. The research question, and subsequent eligibility criteria were developed within the PICO (problem/population, intervention, comparison, outcome) framework. 19 Assistance with search terms and strategies was provided by a health science research librarian with extensive experience in systematic review design (Supplementary Material e-1, available in the online version of the article). Electronic databases; Pubmed, pscyINFO, SPORTdiscus, Medline, CINAHL, and Embase were searched from database inception to November 2016. Peer reviewed studies reported in English, French, Italian, German, Chinese, and Spanish were included. No date restriction was set for study inclusion.
Study Selection Criteria
Studies were included where diagnosis of mTBI or concussion was (a) based on The World Health Organization Collaborating Centre for Neurotrauma Prevention, Management and Rehabilitation Task Force on Mild Traumatic Brain Injury definition 20 or (b) performed by a health professional with specialist knowledge of mTBI in accord with injury criteria. Studies where diagnosis was based on self-report were not included.
Additionally, given diagnosis of postconcussion syndrome (PCS), based on persistent symptom reporting following a proximate head injury,21,22 most frequently occurs at greater than 4 weeks from injury, 23 time from injury for study inclusion was set at 4 weeks to 6 months. This criterion allowed sufficient time to elapse for what is frequently considered to be aberrant or protracted recovery from mTBI 22 as well as ensuring a sufficiently reduced time since initial injury so as to prevent development of comorbid conditions that could unduly confound results.
Additional inclusion criteria:
Adult participants between the ages of 18 and 60 years (or where the mean age minus standard deviation remains ≥18 or ≤60 years).7-9,24
Published full-text cohort or prospective cohort studies inclusive of a nonhead injured and otherwise healthy control group (including dissertation excerpts).
Outcome assessment using functional sensorimotor and physiological measures (excluding functional imaging).
Exclusion criteria:
Study Identification and Data Extraction
Following database searches and exclusion of duplicate titles, consensus was sought between 2 independent reviewers (OAG and MAC) regarding studies for full text review (full texts were obtained anyhow when required to clarify inclusion eligibility) after which study eligibility was determined. Reference lists of eligible studies were then screened to identify any further studies for inclusion. Data were extracted from eligible studies by the primary author (OAG) using a structured form. A second reviewer (MAC) confirmed accuracy. Any disagreement between authors regarding study inclusion or extracted data was settled by discussion and annotated on hard copies. Where relevant information was considered missing primary authors were contacted via e-mail.
Quality Assessment
Following data extraction independent quality assessment was performed using the Downs and Black checklist. 25 This tool has been peer reviewed by 2 independent national research standard agencies and recommended for use in rating observational studies.26,27 As reflected in previous systematic reviews, 28 modification (removal of items 13, 14, 19, 23, 24) was made to suit design of studies included. Despite this, reported scores represent raw unadjusted values regardless of items removed.
Level of evidence classification, recommended for assessment of risk of bias and formulation of empirically based recommendations,29,30 was determined individually for each study, and overall by both reviewers independently. The American Academy of Neurology’s (AAN’s) classification of evidence matrix for population screening studies was selected. 31 Based on this matrix possible (1 or more level-3 studies and up to 2 level-2 studies), likely (multiple level-2 studies), or highly likely (2 or more level-1 studies) inferences could be made from combined results. 31
Data Synthesis and Meta-Analysis
While the majority of results required narrative synthesis, where sufficient data were reported (via numeric or graphical representation), standardized mean difference (SMD) and 95% confidence intervals (95% CI) were calculated for between-group differences (Supplementary Material e-2, available in the online version of the article). Where data pooling was possible (>2 studies) and heterogeneity between study populations was low (I2 < 0.4) 32 fixed effects models were used to calculate group differences,33,34 otherwise random effects models were used to account for variability. 35 Meta-analysis was conducted using Review Manager (RevMan) (Windows 7). Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Heterogeneity of data reporting meant it was at times necessary to use reported values to calculate group means or standard deviations. Where only standard error 36 or 95% CIs 37 were reported, recommended methodology was used to calculate the standard deviation (SD). 32 Where group numbers were low, denominator values were adjusted.32,36 For median and interquartile range, 38 the median value was used as the mean 32 and SD calculated using suitable published methods.32,39
Results
Database searching identified 18 studies that met inclusion criteria (Figure 1, Table 1). Five studies included data that could be pooled. Between-group differences comparing mTBI to controls were observed for sensorimotor measures including balance, motion analysis of gait, vestibular, and oculomotor outcomes. Physiological outcomes included cardiac autonomic function (via electrocardiography) reported in terms of heart rate variability (HRV) domains, and polysomnography assessment of sleep indices. Overall methodological score achieved on the Downs and Black checklist was 15.67 (SD 2.33) (Figure 2).
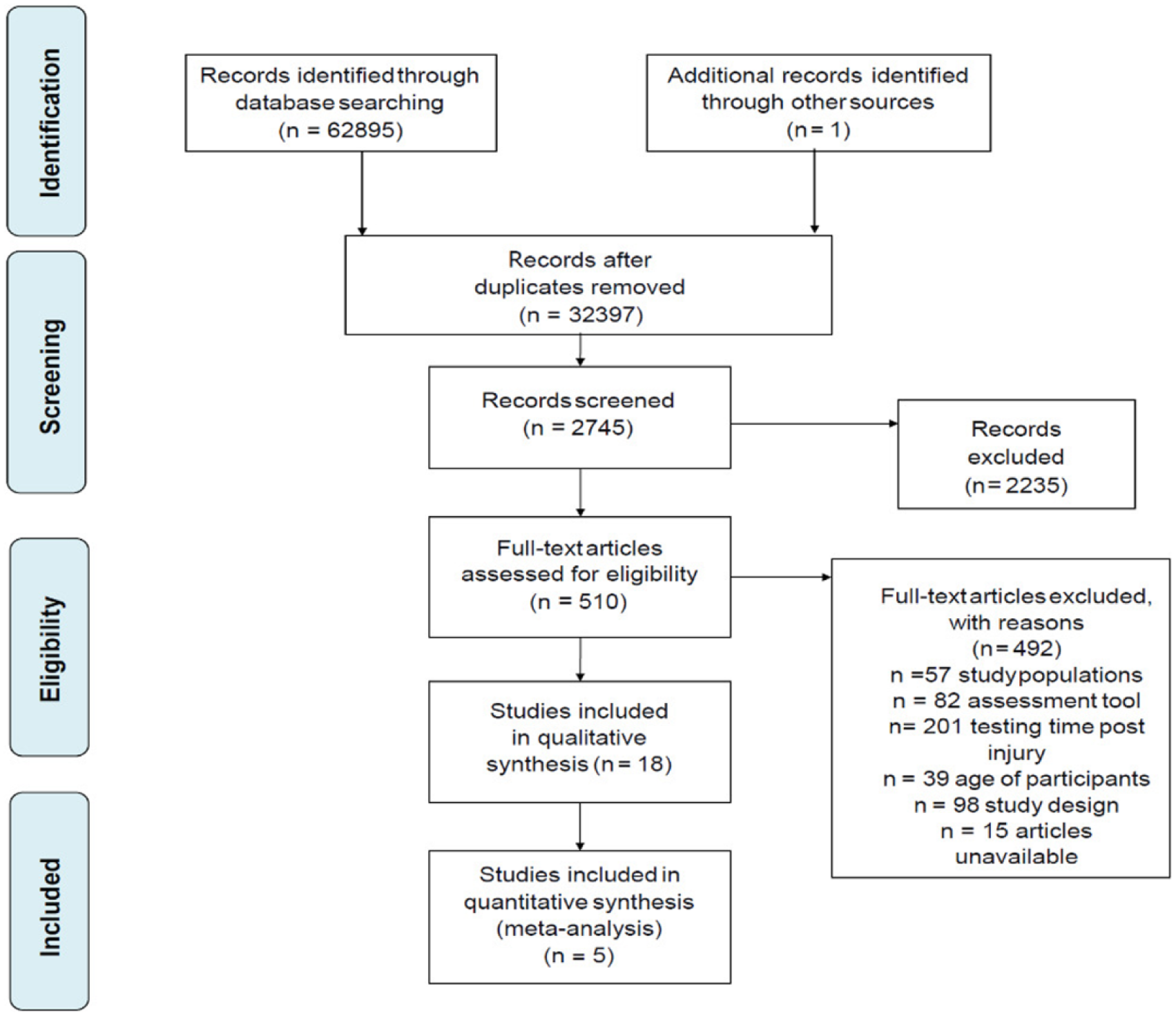
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram of study selection.
Summary of Characteristics of Studies Included in Systematic Review and Meta-Analysis.
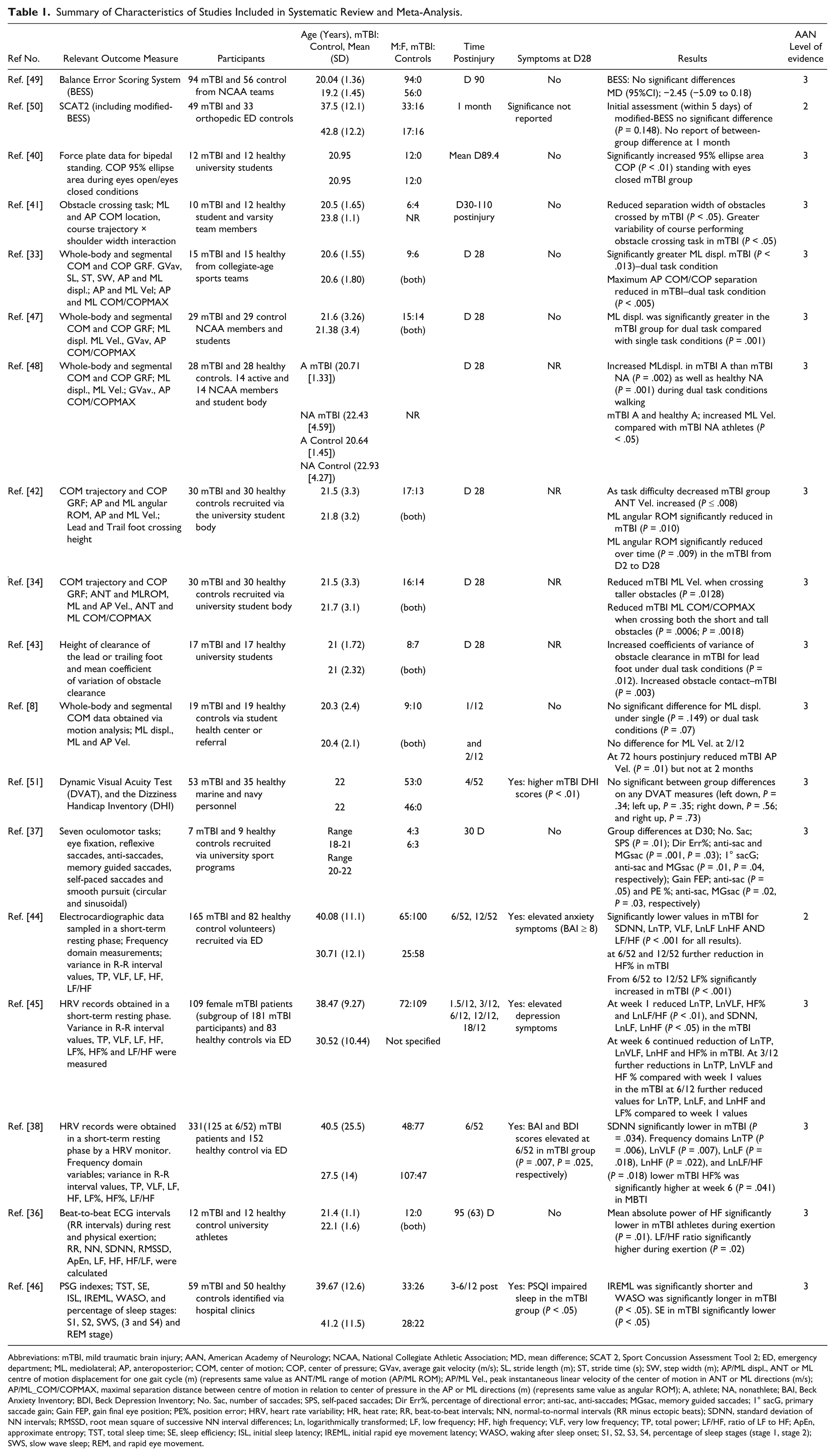
Abbreviations: mTBI, mild traumatic brain injury; AAN, American Academy of Neurology; NCAA, National Collegiate Athletic Association; MD, mean difference; SCAT 2, Sport Concussion Assessment Tool 2; ED, emergency department; ML, mediolateral; AP, anteroposterior; COM, center of motion; COP, center of pressure; GVav, average gait velocity (m/s); SL, stride length (m); ST, stride time (s); SW, step width (m); AP/ML displ., ANT or ML centre of motion displacement for one gait cycle (m) (represents same value as ANT/ML range of motion (AP/ML ROM); AP/ML Vel., peak instantaneous linear velocity of the center of motion in ANT or ML directions (m/s); AP/ML_COM/COPMAX, maximal separation distance between centre of motion in relation to center of pressure in the AP or ML directions (m) (represents same value as angular ROM); A, athlete; NA, nonathlete; BAI, Beck Anxiety Inventory; BDI, Beck Depression Inventory; No. Sac, number of saccades; SPS, self-paced saccades; Dir Err%, percentage of directional error; anti-sac, anti-saccades; MGsac, memory guided saccades; 1° sacG, primary saccade gain; Gain FEP, gain final eye position; PE%, position error; HRV, heart rate variability; HR, heat rate; RR, beat-to-beat intervals; NN, normal-to-normal intervals (RR minus ectopic beats); SDNN, standard deviation of NN intervals; RMSSD, root mean square of successive NN interval differences; Ln, logarithmically transformed; LF, low frequency; HF, high frequency; VLF, very low frequency; TP, total power; LF/HF, ratio of LF to HF; ApEn, approximate entropy; TST, total sleep time; SE, sleep efficiency; ISL, initial sleep latency; IREML, initial rapid eye movement latency; WASO, waking after sleep onset; S1, S2, S3, S4, percentage of sleep stages (stage 1, stage 2); SWS, slow wave sleep; REM, and rapid eye movement.
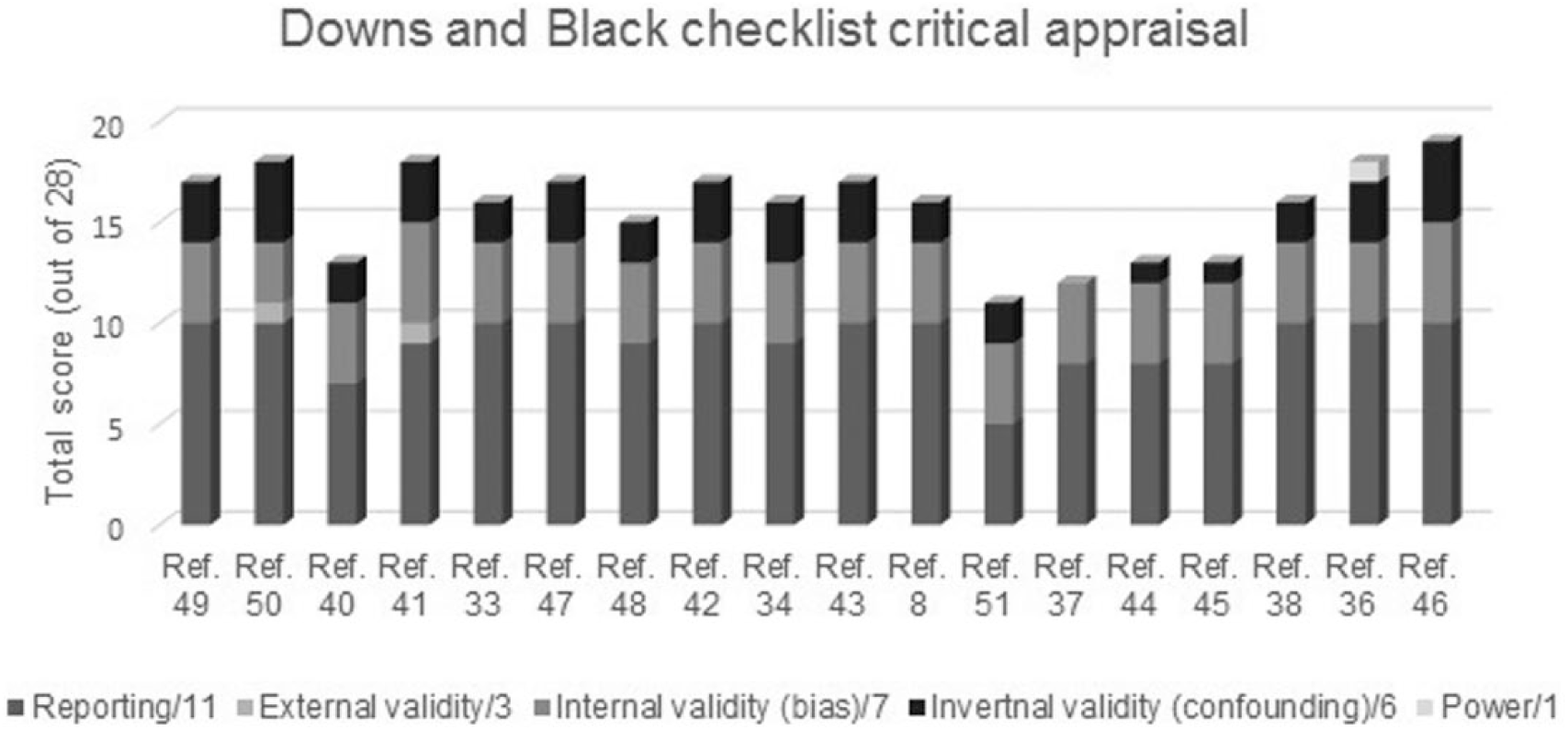
Quality assessment of included studies.
Control groups included healthy (or with strict health-related inclusion criteria),36,38,40-46 normal,33,37,47,48 or matched nonhead-injured individuals.8,34,49 An additional study included orthopaedic (1-2 years post ankle injury) nonhead-injured controls, 50 while 1 study sourced control participants from active duty military personnel without history of substance abuse or psychiatric disorders. 51 No studies identified in preliminary screening required exclusion based on the health of control participants.
Since no studies specifically recruited participants with known sensorimotor or physiological impairment, all studies were rated as AAN evidence level 1 to 3. Only results pertaining to sensorimotor or physiological function were examined and reported.
Sensorimotor Function
Balance
Three studies40,49,50 (AAN evidence level 2, n = 1; level 3, n = 2) used balance assessment tools. Two studies using the Balance Error Scoring System (BESS) 49 or the Modified-BESS 50 identified no between-group differences when comparing mTBI participants with healthy 49 or trauma controls 50 (SMD, −2.45, 95% CI −5.09 to 0.18; P = .148). Using athlete populations, however, Thompson et al 40 found persistence of increased center of pressure (COP) area (m2) in the mTBI group (P < .005) during an eyes closed standing balance task on a force plate.
Gait Assessment
Eight studies (all AAN evidence level 3) using motion analysis technology to assess gait under varying conditions were identified.8,33,34,41-43,47,48 Six studies assessed sagittal and frontal plane center of motion (COM) and pressure (COP) variables.8,33,34,42,47,48 Additional measures included variability of distance of trail and lead foot clearance during obstacle crossing,42,43 as well as obstacle avoidance patterns in relation to shoulder diameter versus obstacle breadth ratios. 41 Dual task conditions were consistent across 6 studies33,34,42,43,47,48 with 1 study using the Stroop auditory test. 8
Despite the range of gait variables assessed, relatively few reached significance. Those identified as significant mostly occurred in a mediolateral (ML) direction, under dual task conditions. ML instantaneous angular range of motion was significantly reduced in the mTBI group in 1 study (P = .01) 42 . Post hoc analysis we performed on variable data identified reduced range in the mTBI group during obstacle crossing (SMD, −0.68, 95% CI −1.20 to −0.16, P = .01) and cognitive task performance (SMD, −0.61, 95% CI −1.13 to −0.09, P = .02) also, evident more so when conditions were combined (SMD, −0.72, 95% CI −1.25 to −0.20, P = .007). 42 During tall and short obstacle crossing tasks, significantly reduced ML COM-COP separation distance (P = .0006, P = .0018), ML peak velocity (P = .0128) 34 and lead foot clearance height variability in the mTBI group was observed (P = .012). 43
Increased overall ML displacement for a single gait cycle (P < .013) as well as reduced anterior COM-COP separation distance (P < .005) was demonstrated by the mTBI group during cognitive dual task conditions. 33 The same authors identified that ML displacement under dual versus single task conditions was significantly increased within the mTBI group (P < .05), whereas in the control group it was not. 47 Increased ML displacement (P < .001) was also found in concussed athletes compared with healthy nonathletes. 48 One study found no evidence of group differences for COM displacement or velocity in any direction. 8
Gait impairments were also observed during easier tasks suggestive of less cautious gait strategies in the mTBI group. This included increased sagittal plane average velocity during single task walking (P = .003), 48 and reduced obstacle separation distance between which mTBI individuals walked compared with controls (P < .05). 41
Oculomotor Function
Only 1 study (AAN evidence level 3) was eligible for inclusion. 37 Significant differences in complex oculomotor function (latency, gain, directional and positional error indices of anti-saccade, self-paced and memory guided saccades) assessed by monocular eye-tracking devices, was evident in the mTBI compared with healthy control group (P ≤ .05) (see Table 1).
Vestibular Function
One (AAN evidence level 3) study using the computerised dynamic visual acuity test (DVAT), a rapid screen of the visual-vestibulo ocular reflex (VVOR), in conjunction with the Dizziness Handicap Inventory (DHI) was identified. 51 No persistent vestibular impairment was evident in the mTBI group compared with healthy controls (P > .34) despite ongoing symptoms (P < .01).
Physiological Outcomes
Cardiovascular Autonomic Function
Four studies36,38,44,45 (AAN evidence level 2, n = 1; level 3, n = 3) assessing HRV via electrocardiography were identified. Time domains measured included heart rate (HR), and beat-to-beat (RR) intervals. Normal-to-normal (NN) intervals (RR minus ectopic beats), standard deviation of NN (SDNN) intervals, and root mean square of successive NN interval differences (RMSSD) were then calculated. Power spectral analysis calculated frequency domains: low frequency (LF), high frequency (HF), LF%, HF%, very low frequency (VLF), total power (TP), and LF/HF ratio. Data was collected over a 5-minute resting period for all 4 studies. Abaji et al 36 also collected measures during a 3-minute submaximal (30% × 1 RM) isometric grip task.
Comparison of domain outcomes between groups (including 1 female only subgroup) 45 demonstrated persistent differences in the subacute time frame following mTBI. Most differences were identified under resting conditions.38,44,45 One study 38 reported significant differences for SDNN (P = .034), TP (P = .006), VLF (P = .007), LF (P = .018), HF (P = .022), HF% (P = .041), and LF/HF (P = .018) and another study 36 for HF (P = .01) and LF/HF (P = .02) with exertion only. Interestingly, participants in this study were reported to be symptom free and had returned to sport.
For 2 further studies,44,45 significant group differences (P < .05 and P < .001) at week 1 and subsequent significant trends for increasing between-group disparities for HF, VLF,44,45 as well as TP 45 at later time points (6 weeks to 6 months later) were observed. Thus significant differences at these time points was inferred.
Sleep
One study (AAN evidence level 3), using polysomnography to measure sleep cycle variables, was identified. 46 Compared with healthy controls, a faster onset of initial rapid eye movement latency, increased waking after sleep onset, and poorer sleep efficiency were shown in the mTBI group (P < .05), indicating persistently poorer sleep quality.
Symptoms
Of the 18 studies included, 4 did not indicate whether symptom reporting was evaluated34,42,43,48 although all 4 studies identified persistent sensorimotor impairment. Of the remaining studies identifying persistent sensorimotor or physiological impairment (n = 9), 5 identified these impairments in the absence of symptoms33,36,37,40,41 and 4 reported impairment in the presence of ongoing symptomology.38,44-46 Two studies, which also included correlation analyses, identified positive relationships between significant HRV outcomes and anxiety (mixed gender: P < .05 and P < .01) 44 or depression (in a female-only subgroup: P < .05) 45 symptom scores at 6 weeks 44 and 3 months44,45 postinjury.
Meta-Analysis
Heterogeneity of outcome measures and methods of reporting results permitted pooling of data for only a small number of variables. Pooled gait variable results included; maximal ML COM-COP separation (m), anterior peak velocity (m/s), and ML peak velocity (m/s) under both single and dual task conditions for all variables. Of these 6 variables only 1 approached significance (SMD, −0.42, 95% CI −0.84 to −0.00, I2 = 0%) (Figure 3) indicating reduced maximal ML displacement with gait under dual task conditions may persist following mTBI.
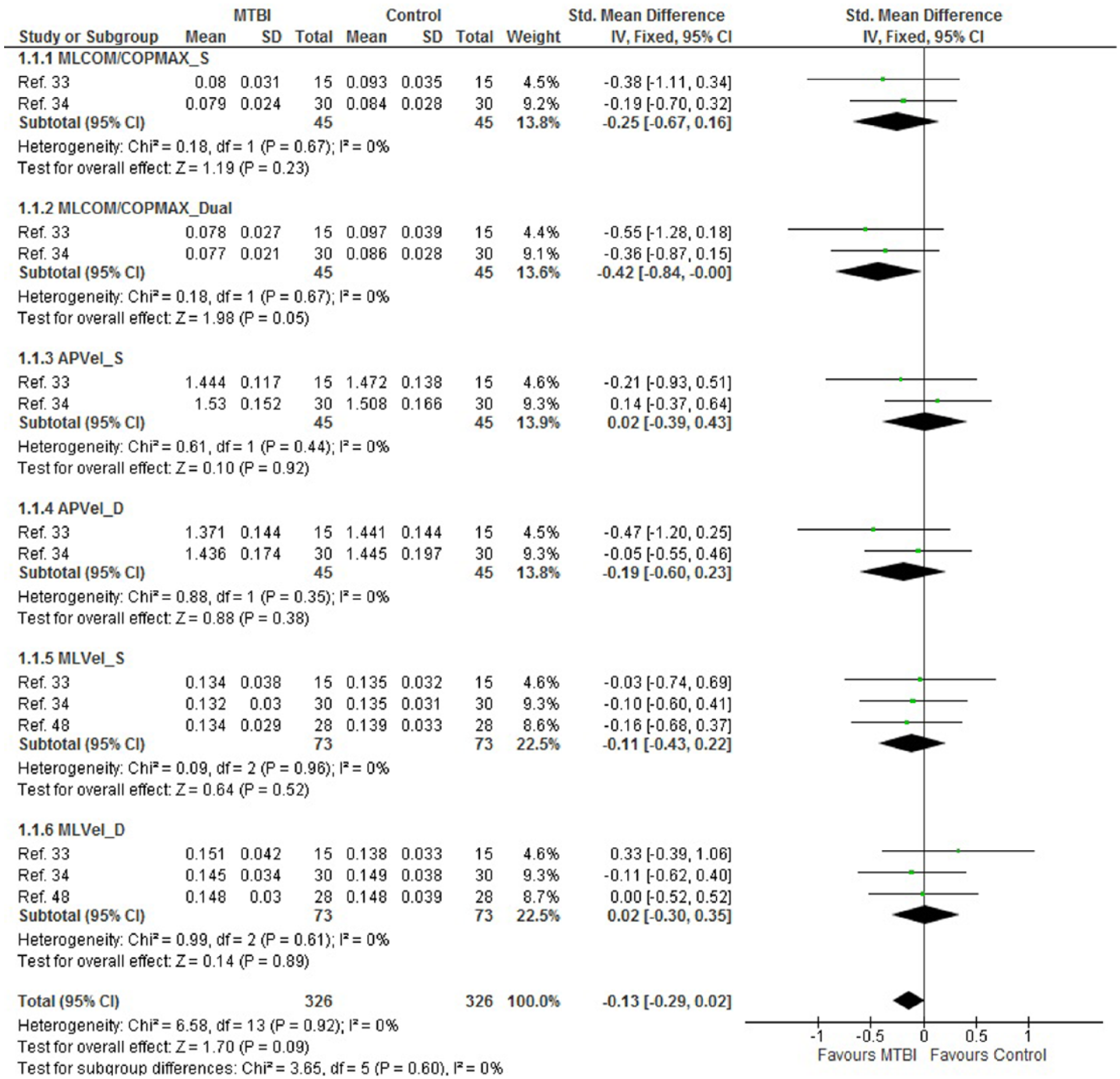
Meta-analysis: motion analysis variables—gait. MLCOM/COPMax_S, maximal mediolateral displacement of center of motion in relation to center of pressure–single task (m); MLCOM/COPMax_D, maximal mediolateral displacement of center of motion in relation to center of pressure—dual task (m); APVel_S, anterior peak velocity–single task (m/s); APVel_D, anterior peak velocity–dual task (m/s); MLVel_S, mediolateral peak velocity–single task (m/s); MLVel_D, mediolateral peak velocity–dual task (m/s).
HRV measures were pooled for HR, SDNN, and LF/HF. Pooling for frequency domains LF and HF could not be performed since they were reported in absolute 36 or logarithmically transformed values. 38 Of the 3 pooled outcomes, only SDNN remained significant (SMD, −0.51, 95% CI −0.74 to −0.28, I2 = 0%) (Figure 4) indicating reduced interval variability between heart beats and persistent autonomic cardiovascular impairment.
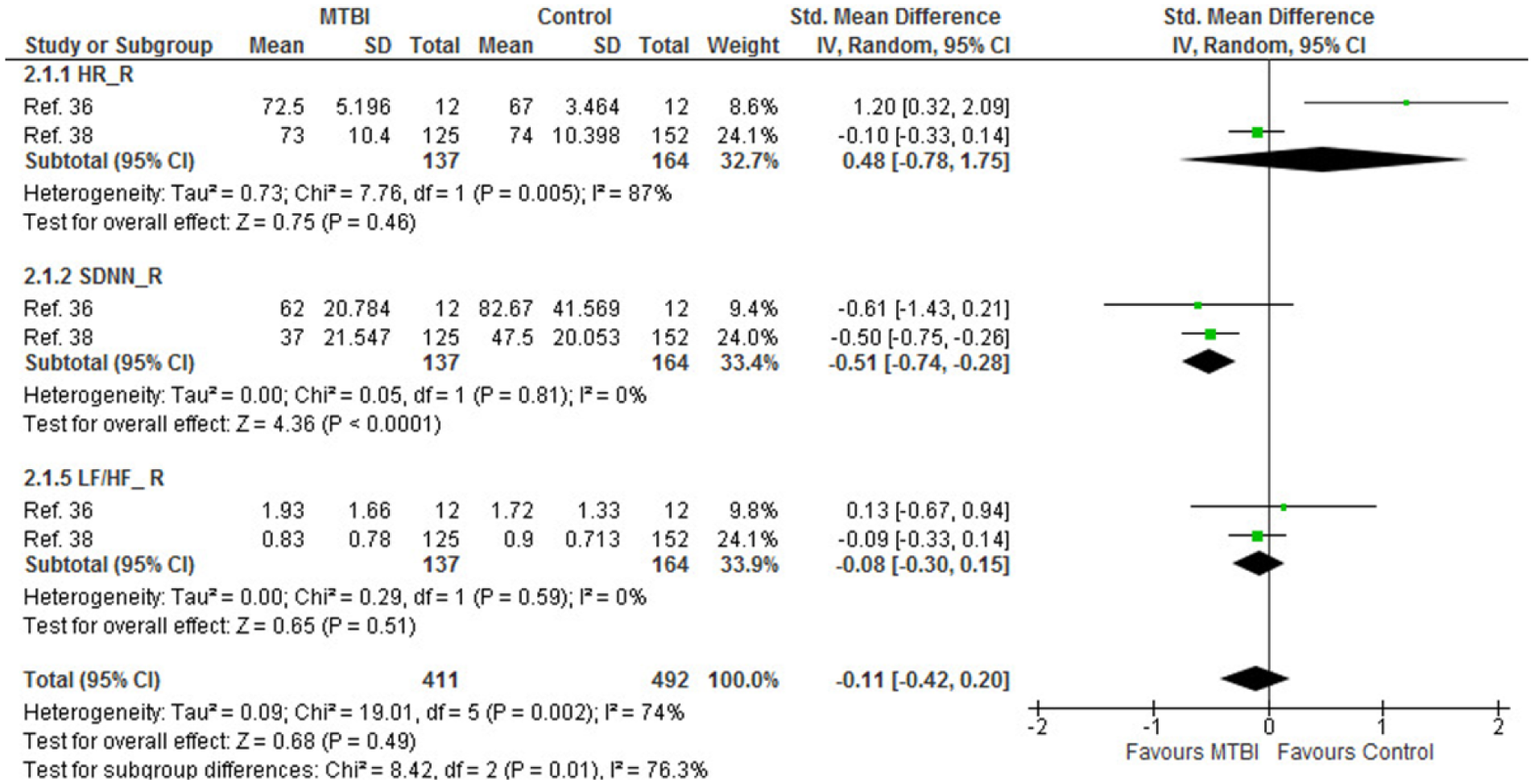
Meta-analysis: heart rate variability. HR_R, resting heart rate (beats per minute); SNDD_R, standard deviation of normal to normal interval at rest (milliseconds); LF/HF_R, low frequency/high frequency power ratio.
Discussion
Overall, using the AAN classification of evidence matrix, 31 level-3 evidence exists for the persistence of gait as well as autonomic cardiac function impairments, indicating that persistence of these impairments beyond expected recovery time following adult noncomplicated mTBI is possible.
Sensorimotor Impairments
The persistence of frontal plane gait impairments following mTBI, characterized by reduced maximal ML excursion and velocity, was evident under dual task conditions in 4 level-3 studies.33,34,42,48 Maximal ML COM-COP separation approached significance when data were pooled (P = .05). These findings combined indicate that persistent dynamic instability in the mTBI group may exist sub acutely, and that to counteract this a more conservative gait strategy, may be adopted by the mTBI group, especially under conditions with greater falls risk. Furthermore, the more complex the task, the more conservative the gait strategy adopted.34,42 Additional findings of reduced sagittal COM-COP separation in conjunction with increased overall ML displacement, 33 reflect those previously identified in studies investigating gait in mixed severity TBI and also provide evidence for potential dynamic instability.52,53
During tasks posing less threat to dynamic stability (ie, single task walking on a flat surface directly or around obstacles), less conservative behavior and strategies may be adopted by the mTBI group.41,48 While suggestion was made that conservative gait patterns may reflect increased awareness of the importance for a safe obstacle crossing strategy, 34 it may also be true that such “awareness,” as exhibited by behavior with greater injury risk, is absent during less complex gait conditions.
Clinically these results highlight the potential for persistence of impaired dynamic stability during gait, 53 as well as the possible exhibition of behaviors with greater likelihood for injury during more simple tasks. 41 Such findings are significant in light of the high rate of mTBI recurrence 11 and elevated risk of sustaining additional injuries following mTBI. 54 Furthermore, they highlight the need for intervention to address these specific deficits postinjury. Additional studies investigating persistent gait impairment following mTBI are therefore required to qualify these gait changes and any relationship they may have with reinjury or maintenance of dynamic stability.
Insufficient evidence was identified to conclude whether impairment persists post-injury for other sensorimotor functions such as static balance, oculomotor, and vestibular function, and these factors will require further study in the mTBI population.
Physiological Impairments
Individual study outcomes of physiological function combined to indicate that persistent impairment of cardiac autonomic function is possible beyond usual recovery. Two level-3 studies36,38 identified significantly reduced HF, indicative of impaired vagal nerve parasympathetic modulation of cardiac function, and increased LF/HF ratios, indicating a possible alteration of sympathovagal balance. 55 Persistent impairment of overall HRV as indicated by SDNN was supported by meta-analysis. These findings reflect those of other systematic reviews investigating the effects of concussion on physiological function in more acute 56 and mixed populations. 57
However, HRV as an objective assessment tool is subject to variability based on various testing parameters, including duration, body position, 58 as well as pattern of breathing 59 and independent variables such as gender, age, medication use, and psychological status.60,61 Noteworthy in this meta-analysis is that assessment was conducted for all pooled studies at rest, over 3 to 5 minutes of analysis. Furthermore, no statistically significant difference was identified between controls and mTBI participants in either study with regard to gender, or psychotropic medication use although between-study differences for these variables did exist.
Nevertheless, HRV remains the measure of choice for assessment of autonomic nervous system pathophysiology. Furthermore, HRV indices reflective of results of this study have also been identified as independent prognostic indicators for critical health conditions, including severe TBI62,63 and cardiac mortality following myocardial infarction. 64 Therefore, while exact mechanisms of the relationship between autonomic regulation and poor prognosis following critical health conditions are predominantly hypothetical,62,64-66 further investigation into the value of HRV indices as indicators of ongoing autonomic nervous system impairment is warranted in mTBI.
Further studies investigating outcomes during the subacute period are required to confirm findings reported in this review since an individual study, but not pooled results, demonstrated evidence for persistent change to HF and LF/HF. Research using standardized assessment criteria to account for the influence of testing parameters would also allow for greater between-study analyses. Additionally, more extensive investigation of the effect of physical exertion on HRV in the subacute phase postinjury is needed.
Although a single study regarding sleep outcomes as measured by polysomnography demonstrated persistent change of multiple sleep indices in the mTBI group, overall evidence at this point is inconclusive. Given the important role sleep plays in restoration of neurological function post TBI, 67 a greater need for enhanced understanding of the impact of mTBI on subacute to chronic sleep disturbance is indicated.
Clinical Implications
Of relevance to clinical practice (including those in the sporting and industrial setting) is that of the 13 studies demonstrating persistent sensorimotor33,34,37,40-43,48 or physiological36,38,44-46 impairment, 5 studies33,36,37,40,41 reported these findings in the absence of symptoms, 4 studies34,42,43,48 did not investigate symptoms, while only 4 reported persistence of symptoms.38,44-46 Of note, in studies where physiological impairments and ongoing symptoms were identified, specific symptom assessment tools rather than post concussion scales were used. For example, validated tools to assess anxiety,38,44 depression,38,45 or sleep quality 46 were administered. These findings challenge the usefulness of symptom resolution as an indicator of recovery, and also suggest that resolution of impairment specific symptoms may not always occur within 14 days postinjury. 1 Investigation of a broader array of symptoms may yield more useful indicators of recovery as well as more precisely identifying the relationship between symptom resolution and recovery.
Another factor highlighted by this systematic review was the lack of studies investigating the potential impact of mTBI on other systems, such as the cervical afferent sensorimotor system. Known cervical sensorimotor deficits relating to balance, gait, and oculomotor function have been identified in those with neck disorders, especially those with trauma. 68 This is noteworthy since shared symptom profile and injury mechanisms between traumatic neck pain and mTBI are recognized. 69
Additionally, despite the decades of research investigating the consequences of mTBI when quality assurance measures were imposed, few studies were eligible for assessment of the various sensorimotor and physiological indicators. This outcome underscores both the imminent need for further research investigating these systems and or measures, as well as the potential these measures may have to be important for prognosis and establishing recovery post mTBI.
Limitations and Strengths
There are some limitations and strengths of this study. The study design accommodated published material only. Hence results presented in this study are subject to inherent publication bias. Because of the low number of articles for each sensorimotor or physiological system analyzed, as well as the heterogeneity of outcome measures used, potential for meta-analysis was limited. Where meta-analysis could be performed, the number of studies included for each variable was at best inclusive of three unique cohorts. Power of meta-analysis results was therefore limited both by lower numbers of studies as well as reduced size of cohorts included in some studies.
Additionally, because of low number of pooled studies, it was not useful to investigate the effect of heterogeneity via meta-regression analysis. Guidelines relating to performance of meta-regression have previously and consistently stated that where aggregate data for a low number of studies only can be pooled, individual or multivariate meta-regression is unlikely to yield scientifically useful results.70,71 Therefore it is possible that influence of independent variables may to some degree account for meta-analysis results reported.
The AAN evidence levels assigned to studies were at best identified as level 2. The consistent reason for this was that included studies rarely accessed all mTBI participants within the available cohort, or did not report if they had done so, or else experienced high numbers of dropouts when assessment was repeated longitudinally. Both methodological concerns represent common study limitations 72 ; however, if higher levels of evidence are to be assigned, consideration as to how to overcome these issues is needed.
This review was very specific regarding inclusion criteria for the mTBI population under study, as well as design of studies eligible for inclusion. Presence of a control group was considered essential and has also been recommended for research investigating mTBI prognosis. 73 In order to minimise information and confounding bias, retrospective studies were also ineligible.72,74 We also attempted to control for certain confounding variables by ensuring that age, and injury diagnosis (including relative severity and causation) were consistent. This aided potential for results of this study to be applied specifically to adults (≥18 and ≤60 years old) sustaining an mTBI. However, the subsequent number of ineligible studies underpins the need for further research with greater homogeneity of design and improved methodological quality.
Finally, although rigorous selection criteria were employed to ensure methodological quality and consistency of diagnosis, we were unable to confirm that all mTBI study cohorts included only participants where individuals reporting recurrent injury or complicated injuries were excluded. Hence potential for more severe injuries to influence reported findings is possible.
Conclusion
This review indicates that impairments in gait and heart rate variability may persist following mTBI beyond the expected recovery time. Furthermore, these impairments may persist in the absence of symptoms. Clinically, the findings also suggest that these sensorimotor and physiological factors may need to be assessed in individuals who have suffered a mTBI. Optimal care in those with subacute mTBI is still uncertain due to insufficient evidence pertaining to balance, oculomotor, vestibular, and sleep dysfunction. Further research with greater homogeneity of design and reporting methods is required in order to strengthen quantitative synthesis and facilitate robust clinical guideline development.
Supplemental Material
Supplementary Material – Supplemental material for Sensorimotor and Physiological Indicators of Impairment in Mild Traumatic Brain Injury: A Meta-Analysis
Supplemental material, Supplementary Material for Sensorimotor and Physiological Indicators of Impairment in Mild Traumatic Brain Injury: A Meta-Analysis by Olivia A. Galea, Michelle A. Cottrell, Julia M. Treleaven and Shaun P. O’Leary in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
Dr Michael David assisted with provision of appropriate statistical methodologies for data conversion. Mrs Christine Dalais for her assistance with data base terms and searches. Miss Xiaoqi Chen, Ms Nathalia Costa, and Mr Marcus Kiel for their assistance with translation.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website, along with the online version of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
