Abstract
Background. Subcortical capsular stroke has a poor prognosis, and it is not yet fully understood how and under what circumstances reach training contributes to motor recovery. Objective. This study was performed to investigate changes in neuronal circuits and motor recovery in a chronic capsular stroke model in the presence or absence of reach training. Method. We generated photothrombotic capsular lesions in 42 Sprague-Dawley rats and evaluated motor recovery with or without daily training in a single-pellet reaching task (SPRT). We used 2-deoxy-2-[18F]-fluoro-D-glucose-microPET (positron emission tomography) to assess remodeling of neuronal circuits. Results. SPRT training was selectively beneficial only for the group with incomplete capsular destruction (P < .05), suggesting the relevance of plasticity in the remaining capsular fibers for motor recovery. Groups that did not receive SPRT training showed no motor recovery at all. The microPET analysis demonstrated that motor recovery was correlated with a reduction in cortical diaschisis in ipsilesional motor and sensory cortices and in the contralesional sensory cortex (Pearson’s correlation, P < .05). We also observed training-dependent subcortical activation in the contralesional red nucleus, the internal capsule, and the ventral hippocampus (P < .0025; false discovery rate q < 0.05). The groups without reach training did not show the same degree of reduction in diaschisis or activation of the red nucleus. Conclusions. Our results suggest that motor recovery and remodeling of neuronal circuits after capsular stroke depend on the magnitude of the capsular lesion and on the presence or absence of reach training. Task-specific training is strongly indicated only when there is incomplete destruction of the capsular fibers.
Introduction
Intensive rehabilitation is known to significantly improve functional outcomes after stroke. In particular, task-specific training improves motor function through repetition and goal-oriented practice, which may induce use-dependent cortical reorganization and neuroplasticity.1,2 Repeated training in a single-pellet reaching task (SPRT) in rodents has been shown to be associated with an increase in hippocampal neurogenesis, 3 modification of synaptic proteins in the ischemic sensorimotor cortex, 4 and a significant increase in axonal sprouting in the area of functional cortical reorganization. 5 However, the responses of different neural structures to reach training after subcortical capsular stroke have not been conclusively demonstrated.
Capsular infarcts, in particular, are associated with a poor prognosis, despite vigorous rehabilitative efforts.6,7 It is plausible that the mechanism underlying functional recovery from capsular stroke may be different from that for cortical stroke. In cortical stroke, injury to the cerebral cortex initiates structural and functional changes in the remaining cortices, leading to functional recovery. However, cortical structures are anatomically intact in capsular stroke, suggesting an alternative remodeling of neuronal circuitry for functional recovery after capsular stroke (hereafter referred to simply as a recovery circuit). Functional imaging techniques, such as positron emission tomography (PET) and functional magnetic resonance imaging, have demonstrated that functional dynamic changes occur throughout the entire course of poststroke recovery and that many neural structures are involved in remodeling the neural circuits that support poststroke recovery.8-10 However, the specific neural structures activated and the dynamic interactions between them during reacquisition of motor skills have rarely been studied in rodent models of capsular infarct.
We have previously demonstrated that microPET is a useful tool for assessing the longitudinal changes in resting-state brain activity (RSA) in a capsular infarct model. 11 In particular, microPET showed that SPRT training appeared to reduce cortical diaschisis and activate several subcortical structures, which presumably contributed to motor recovery. However, all animals received SPRT training in this previous study, so it was not clear whether these changes were the result of SPRT training, or the natural healing process, or a combination of both. Accordingly, the neural mechanisms that underlie recovery from capsular stroke via natural healing and reach training remain an open question. The aims of this study were to determine explicitly the effect of SPRT training on motor recovery in a chronic capsular infarct model and to assess the pattern of neural activation during recovery. To this end, we explored the differences in behavioral recovery patterns between an SPRT-trained group and a control group that received no poststroke training. Using longitudinal microPET scanning, we delineated the remodeling of neural circuits during natural healing and SPRT training. Our results may help us understand the neural mechanisms underlying motor recovery and provide a guide for optimal rehabilitation strategies in capsular stroke.
Materials and Methods
Experimental Animals
Animal experiments were performed in compliance with the institutional guidelines of the Gwangju Institute of Science and Technology (GIST), and all experimental procedures were approved by the Institutional Animal Care and Use Committee at GIST. A total of 42 male Sprague-Dawley rats (9 weeks old, ~300 g) were used for this experiment. Animals were housed in a standard plastic cage (2 animals/cage) with water available ad libitum. The animal care room was maintained on a 12-hour light-dark cycle (07:00-19:00) with controlled temperature (21°C ± 1°C) and humidity (50%).
All experimental animals underwent circumscribed photothrombotic infarct lesioning in the posterior limb of the internal capsule (PLIC) in the hemisphere contralateral to the preferred forelimb. 12 Animals were divided into 2 groups: the SPRT training group (STG: n = 16), which underwent daily postlesional reach training for 3 weeks, and the no SPRT training group (NoSTG: n = 18), which underwent no postlesional SPRT training except for testing sessions held once per week (Figure 1). After histological examination of the extent of infarct lesioning, animals were subdivided into a complete destruction group (CDG: n = 18), with infarct lesioning across the entire breadth of the PLIC, and an incomplete destruction group (IDG: n = 16), with only partial destruction in the PLIC. An additional 8 rats served as a sham-operated control group (SOG). Therefore, there were 5 groups in total: ST-CDG (n = 8), ST-IDG (n = 8), NoST-CDG (n = 10), NoST-IDG (n = 8), and SOG (n = 8).
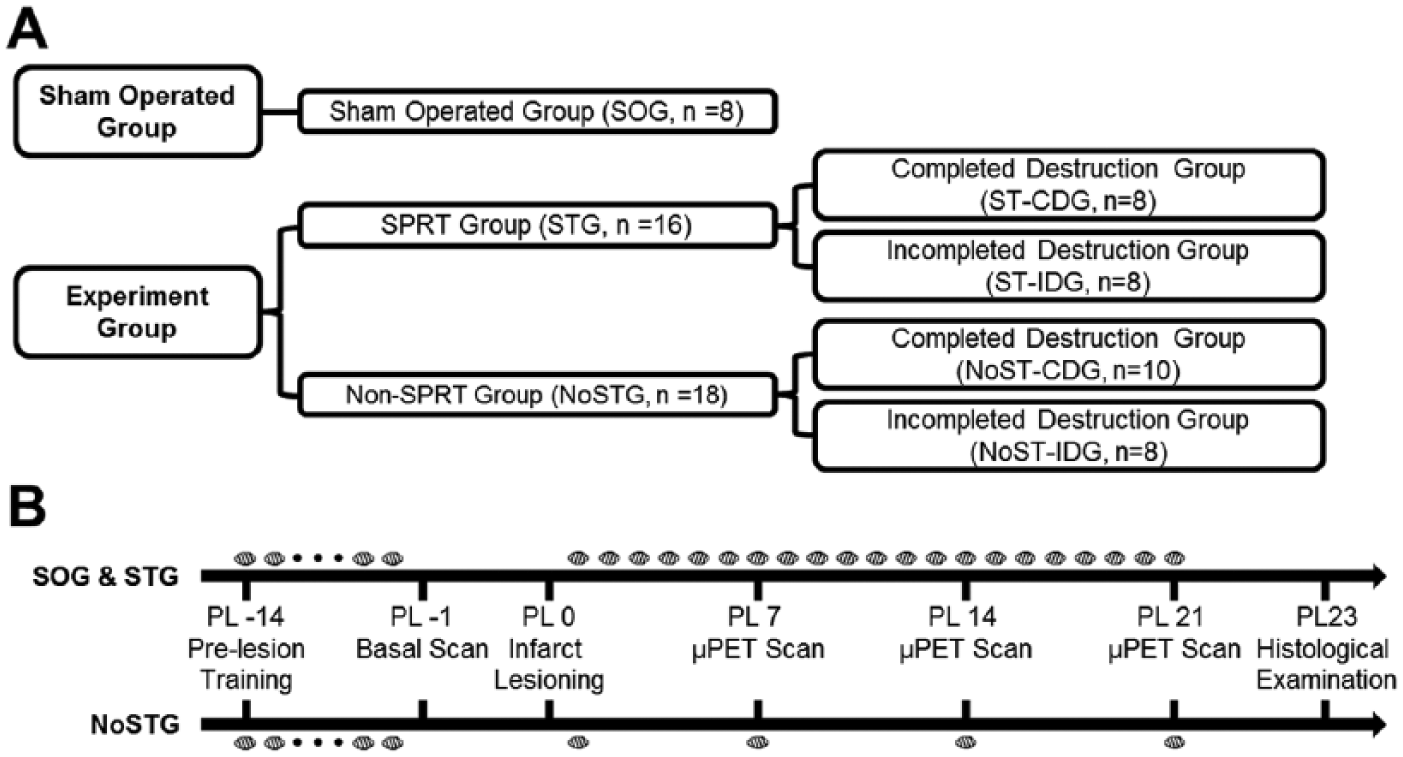
Experimental design: A. Experimental groups. Groups were first divided according to the presence or absence of postlesional training in the single pellet reaching task (SPRT) and then further subdivided according to the magnitude of destruction (complete or incomplete) in the internal capsule after histological examination. B. Timeline of behavioral training and longitudinal microPET (positron emission tomography) scans. Small circles indicate SPRT training days. All animals underwent microPET scanning 4 times: a baseline scan (PL −1) before infarct lesioning on PL 0, and postlesion scans on days 7, 14, and 21 (PL 7, PL 14, and PL 21).
Induction of Photothrombotic Capsular Infarction
The experimental design is shown in Figure 1. Photothrombotic infarct lesions were induced in the PLIC as previously described. 12 Briefly, the head was shaved and fixed in a rodent stereotactic frame after anesthesia. Rectal temperature was maintained at 37°C ± 0.5°C with a thermocouple blanket. A midline scalp incision (approximately 2 cm) was made to expose bregma and lambda. After confirming that bregma and lambda were on the same horizontal plane, a small craniotomy was made 2.0 mm posterior and 3.1 mm lateral to bregma. An optical fiber (core diameter: 62.5 µm; outer diameter: 125 µm) was inserted to reach the PLIC (7.2 mm from the dura). Rose Bengal dye (20 mg/kg) was injected through the tail vein and then the PLIC target was irradiated for 1.5 minutes with a green laser (3.7 ± 0.2 mW at the tip of the fiber). After irradiation, the optical fiber was removed, and the scalp wound was secured. Analgesia (Ketoprofen 2 mg/kg, Uni Biotech, Korea) and antibiotics (Ceftazol 25 mg/kg, Guju Phama Co, Korea) were administered. Animals in the SOG underwent the same procedure for photothrombotic infarct lesioning, except that they received an injection of 0.9% saline (0.2 mL/100 g) instead of Rose Bengal dye.
Single-Pellet Reaching Task and Evaluation
The SPRT was chosen as the behavioral task because it can be used both as a rehabilitative intervention and as a measure of training-induced motor improvement. 13 Furthermore, the SPRT entails similar reaching behavior in humans and trained rodents. 14 The behavioral task was performed as described in Gharbawie et al 15 at a fixed time during the light phase of the diurnal cycle. Animals were food restricted to 90% of their body weight during the entire experimental period. Prior to training, each rat was exposed to a banana-flavored sucrose pellet (Bio-Serve, Frenchtown, NJ) on the shelf and allowed to take the pellet freely. Rats were administered up to 20 pellets for 20 minutes in a single session each day. The preferred handedness of each rat was determined during the SPRT pretraining by evaluating how successful each paw was in retrieving pellets that had been placed obliquely on the shelf. A reach was classed as successful if the rat extended a forelimb through the slit, grasped the pellet, and brought it into the mouth without dropping it. The percentage of successful reaches was calculated as follows: (Number of successful reaches × 100)/20.
After handedness had been determined, rats were not allowed to make reaches using the nonpreferred paw at any subsequent stage of training or testing. All animals were trained every day for 2 weeks before surgery. The preinfarct reaching performance was taken to be the average percentage of successful reaches over the last 3 days of preoperative training. Animals in the STG and the SOG underwent daily postoperative training for 3 weeks using the same procedure. Animals in the NoSTG performed the reaching task once a week postoperatively and on the day they were killed humanely (ie, a total of 4 postoperative training sessions).
MicroPET Image Acquisition and Processing
Longitudinal microPET scans were performed to measure RSA before and after lesioning and behavioral training. All animals were scanned a total of 4 times: a baseline scan after preoperative training but before infarct lesioning (PL-1) and scans on postlesion days 7, 14, and 21 (PL 7, PL 14, and PL 21). The microPET/computed tomography (CT) scanner (Inveon, Siemens Medical Solution, Knoxville, TN) allowed a transaxial resolution of 1.4 mm full width at half maximum and a 12.7-mm field of view. Rats were deprived of food for 12 hours before scanning, and 2-deoxy-2-[18F]-fluoro-D-glucose (0.1 mCi/100 g) was injected into the tail vein under brief anesthesia with isoflurane. After a 30-minute uptake period during which the animals were awake, animals were anesthetized with 2% isoflurane and placed in the microPET scanner. The head was fixed with a customized head holder (Hyosung Inc, Gwangju, Korea). During scanning, vital signs, including rectal temperature (37.0°C ± 1°C), respiration (50 ± 5 respirations/min), and heart rate (280 ± 20 beats/min), were maintained and recorded with a monitoring system (BioVet, m2m Imaging Corp, Cleveland, OH). After a 25-minute static acquisition period, a 5-minute attenuation-correction CT scan was performed. The acquired images were reconstructed with a 3D OSEM/MAP iterative algorithm. Image analysis was performed with the Analysis of Functional NeuroImages (AFNI) package. 16 To ensure that all lesions appeared on the right side during analysis, images from animals with left-hemisphere lesions were flipped. The images were then coregistered to a standard histological rat brain template and manually corrected to the Schweinhardt atlas. 17 Images were spatially normalized to the mean value of the whole brain. Finally, spatial smoothing was performed using a 3D isotropic Gaussian kernel with 1.2 mm full width at half maximum. We chose several single anterior-posterior coordinates to represent the anatomical location of brain activation or deactivation across 4 longitudinal scans.
Statistical Analysis
A group-level, linear mixed-effect model was performed with the 3DLME program in AFNI to assess differences between baseline (PL-1) and PL 7, PL 14, and PL 21 images for each group of animals (STG, NoSTG, and SOG). Statistical maps were corrected and thresholded at the significance level (P < .0025; false discovery rate [FDR] q < 0.05) and then overlaid on the histological template to show areas with significant changes in brain activity. Artifacts presumed to be generated by the ear channel were masked out. We manually defined region-of-interest (ROI) masks over anatomical brain regions that were activated or deactivated to quantify the changes in regional glucose metabolism following capsular infarct.
To investigate the correlation between changes in metabolic activity and SPRT performance, we selected 4 cortical ROIs in the ipsilesional motor cortex, sensory cortex, auditory cortex, and contralesional sensory cortex that showed marked changes in activity following capsular infarct lesioning and 4 additional subcortical ROIs in the ipsilesional internal capsule, contralesional red nucleus, striatum, and ventral hippocampus that were likely to be related to motor recovery. We examined correlations between metabolic activity in these 8 ROIs and SPRT performance across the 3 postlesional sessions using Pearson’s correlation (P < .05).
Data analyses were performed with statistical analysis software (OriginPro version 9.1, OriginLab, Northampton, MA). Skilled reaching performance was analyzed using a 1-way analysis of variance on the effect of groups and times (P < .05, Bonferroni corrected for multiple comparisons). To confirm the tendency in reduction of cortical diaschisis over time in each group, a 1-tailed t-test was used (P < .05).
Histological Examination
After completing the last microPET scan 3 weeks after infarct lesioning (PL 21), rats were anesthetized with ketamine (100 mg/kg body weight) and perfused with 0.9% saline solution followed by 4% paraformaldehyde solution. Brains were carefully removed from the skull cavity, postfixed overnight in 4% paraformaldehyde, and cryoprotected in 30% sucrose in phosphate buffered saline. Brains were serially sectioned with a 40-µm thickness at 200-µm intervals. Nissl staining was performed on the brain sections. Infarct volumes were measured in ImageJ software (NIH, Bethesda, MD). The completeness of the PLIC lesions was assessed by 3 of the authors who were blinded to the groups.
Results
Histological Findings
Figure 2 shows the histological features of the capsular infarct model. Infarct lesions had a loss of axons and myelin, surrounded by reactive gliosis and infiltrating macrophages (Supplementary Figure 1). For animals in the IDG, the extent of the lesion did not cover the whole breadth of the PLIC across serial sections, leaving some portions of the capsular fibers intact. In the CDG, the lesion extended over the whole breadth of the PLIC across multiple serial sections (at least 2 sections), resulting in complete destruction of the capsular fibers (Supplementary Figure 1). The infarct volume was 0.59 ± 0.36 mm3 in the IDG and 0.63 ± 0.37 mm3 in the CDG. Infarct volumes were not significantly different between the STG and the NoSTG.
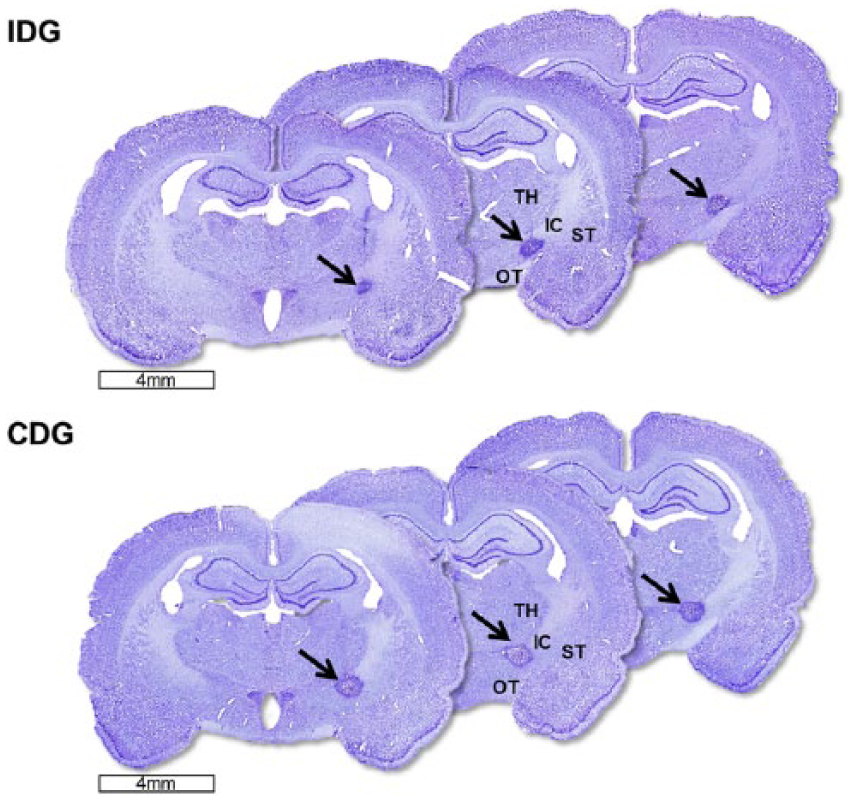
Serial histological comparison of brain sections from 2 representative animals, one from the complete destruction group (CDG) and one from the incomplete destruction group (IDG). Note that the CDG sections show infarct lesioning (arrows) across the entire breadth of the posterior limb of the internal capsule (PLIC), whereas the IDG sections show only partial destruction of the PLIC with some intact capsular fibers remaining above the lesion.
Motor Recovery
Performance in the SPRT following capsular infarct lesioning followed different recovery patterns depending on the group (Figure 3). The SOG showed no decline in motor performance. Both lesion groups (CDG and IDG) showed an immediate decline in reaching performance. Animals in the ST-IDG exhibited progressive improvements in motor recovery from 1 week after the lesion, eventually reaching prelesion levels of 78% correct 3 weeks after the lesion. In contrast, the ST-CDG did not exhibit any motor recovery despite receiving the same degree of daily SPRT training. Importantly, the groups that received no training (NoST-IDG and NoST-CDG) also showed no signs of motor recovery.
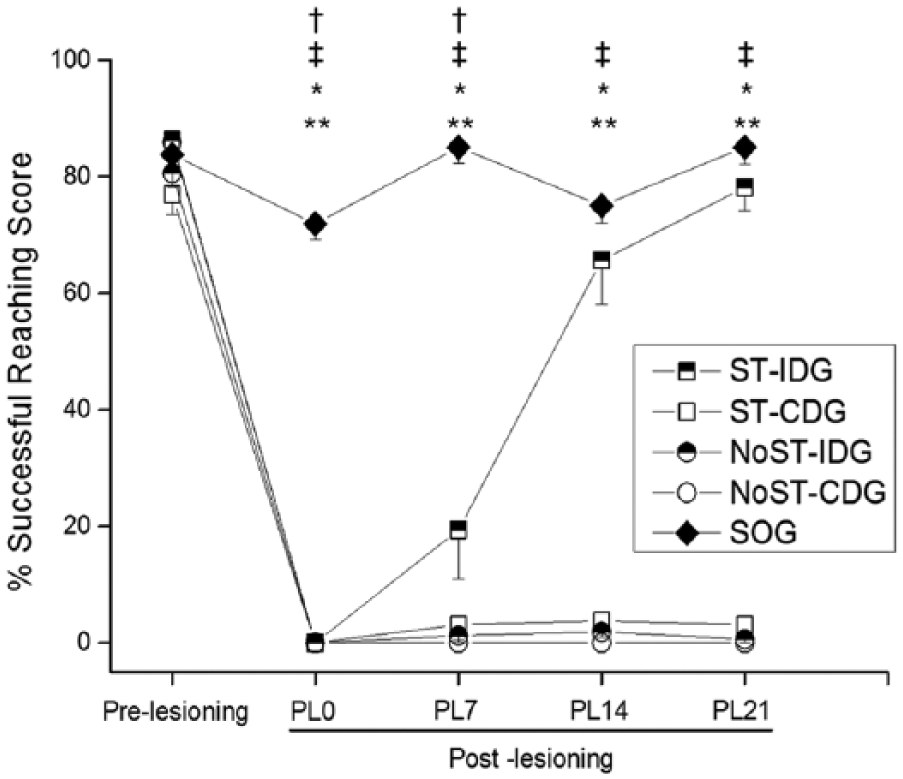
Prelesional and postlesional reaching performance for the 5 different groups (P < .05).a
MicroPET Imaging of RSA
Figures 4 and 5 show the longitudinal changes in RSA from baseline for all groups at PL 7, PL 14, and PL 21 after capsular infarct lesioning. Metabolic differences between the baseline (PL-1) and postlesion scans were used to determine significant changes in RSA.
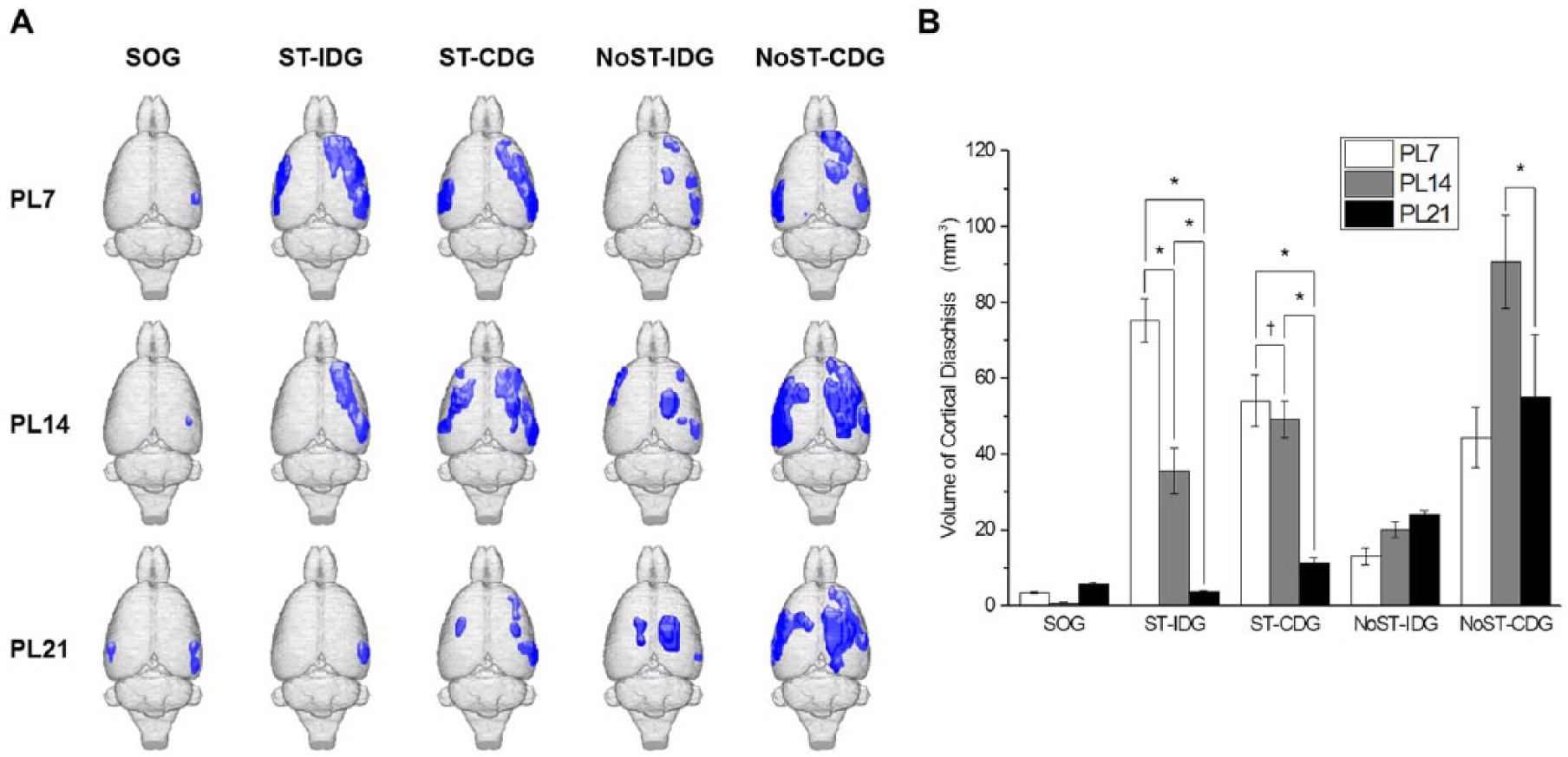
(A) Longitudinal changes in cortical diaschisis in the 5 different groups. Blue regions indicate a significant reduction in metabolic activity (P < .0025, false discovery rate q < 0.05). Note the tendency toward a reduction in cortical diaschisis over time in the ST-IDG and the ST-CDG, whereas there was either no change or an increase in cortical diaschisis in the NoST-IDG and the NoST-CDG. Lesioning was made in the right hemisphere. (B) Bar graph showing the same tendency in the ST-IDG and the ST-CDG.a
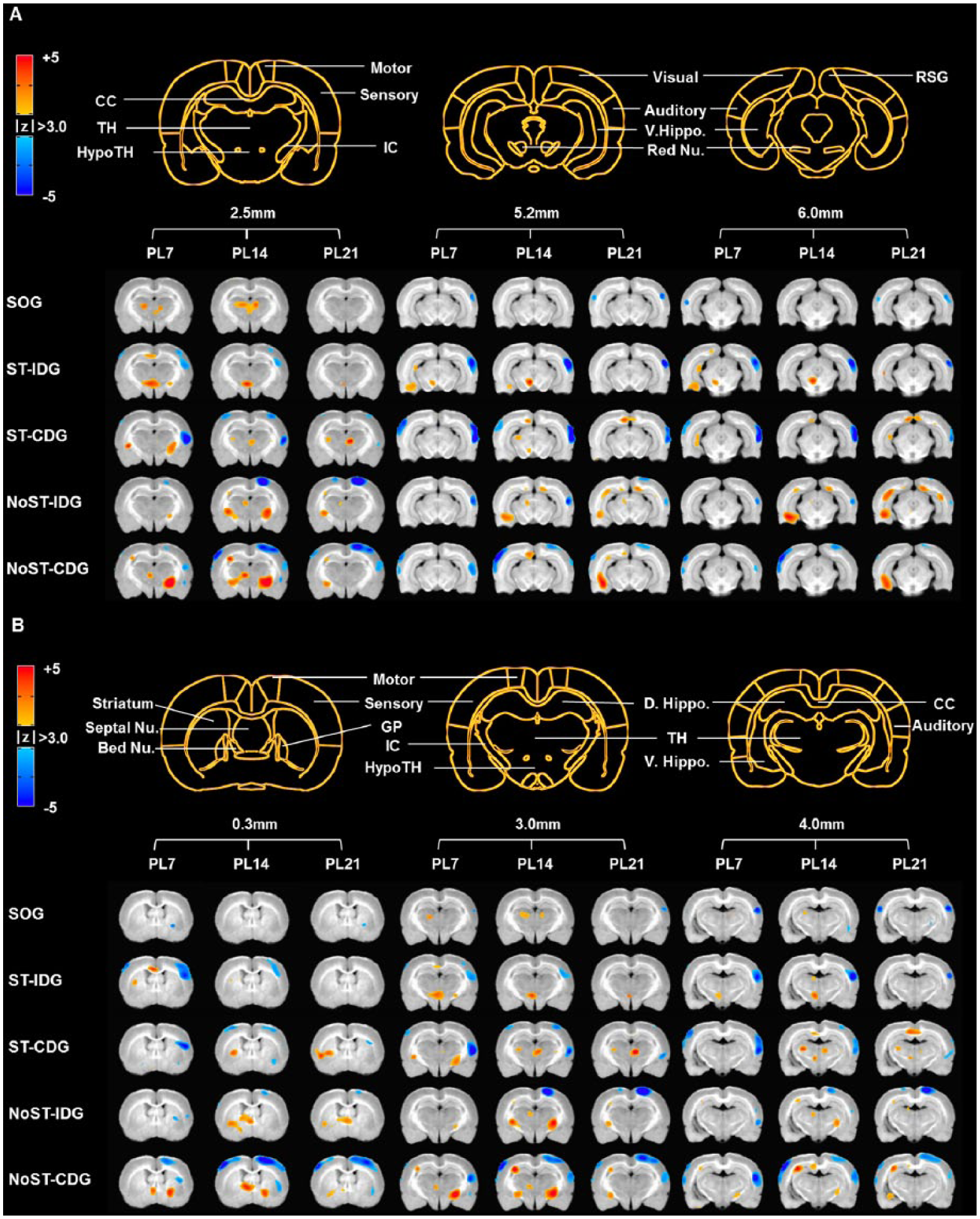
Longitudinal changes in resting-state brain activity in the 5 different groups. (A) Training-dependent subcortical changes include contralateral activation in the red nucleus, ventral hippocampus, and hypothalamus (P < .0025, false discovery rate q < 0.05). (B) Training-independent subcortical changes include bilateral activations in the internal capsule and bed nucleus of the stria terminalis and contralateral activation in the striatum, thalamus, and dorsal hippocampus (P < .0025, false discovery rate q < 0.05).
At the cortical level, decreased brain activity was more prominent in the ipsilesional hemisphere than the contralesional hemisphere (P < .0025; FDR q < 0.05). Decreases in brain activity occurred in bilateral sensory and auditory cortices and the ipsilesional motor cortex (P < .0025; FDR q < 0.05). The areas showing decreased activity (diaschisis) were not initially correlated with any particular group. However, there was a remarkable time-dependent reduction in diaschisis in the groups receiving daily behavioral training (ST groups) compared with the NoST groups, which suggests that reduction of diaschisis may be critical for remodeling of neuronal circuits and motor recovery (Figure 4). The volumes of cortical diaschisis are shown in Supplementary Table 1. At the subcortical level, decreased brain activity was only observed in the ipsilesional striatum in the CDG.
Increases in subcortical RSA were observed in all experimental groups (Supplementary Table 2). Subcortical activations were categorized as training dependent (Figure 5A) or training independent (Figure 5B) on the basis of the presence or absence of daily reach training: training-independent activations were observed in all experimental groups, whereas training-dependent activations were observed only in groups receiving SPRT training (ST groups). Contralesional training-dependent increases were observed in the red nuclei, ventral hippocampus, and the hypothalamus, whereas bilateral training-independent increases were observed in the internal capsule and bed nucleus of the stria terminalis, and contralesional training-independent increases were observed in the striatum, thalamus, and dorsal hippocampus (P < .0025; FDR q < 0.05).
Some subcortical structures showed time-dependent changes. Initial bilateral activation of the internal capsule and contralesional activation of the ventral hippocampus in the ST-IDG and ST-CDG disappeared after PL 7 but persisted in the NoST-CDG and NoST-IDG. Initial activation of the contralesional striatum in the ST-IDG disappeared after PL 7 but persisted in other groups. Activation of the contralesional red nucleus in the ST-IDG and ST-CDG disappeared after PL 14.
Correlations Between Behavioral Recovery and RSA
Figure 6 shows the correlations between changes in metabolic rate and individual behavioral scores for animals in the ST-IDG. At the cortical level, changes in metabolic activity in the ipsilesional sensory cortex (P < .05; R = 0.45), ipsilesional motor cortex (P < .05; R = 0.63), and contralesional sensory cortex (P < .05; R = 0.50) were significantly correlated with increased reaching scores, indicating that the reduced diaschisis in these areas may have contributed to the motor recovery. At the subcortical level, changes in metabolic activity in the contralesional striatum (P < .05; R = −0.42), ventral hippocampus (P < .05; R = −0.43), and red nucleus (P < .05; R = −0.45) and in the ipsilesional internal capsule (P < .05; R = −0.56) were correlated with increased reaching scores, indicating that neuronal remodeling in these structures may have contributed to the motor recovery.
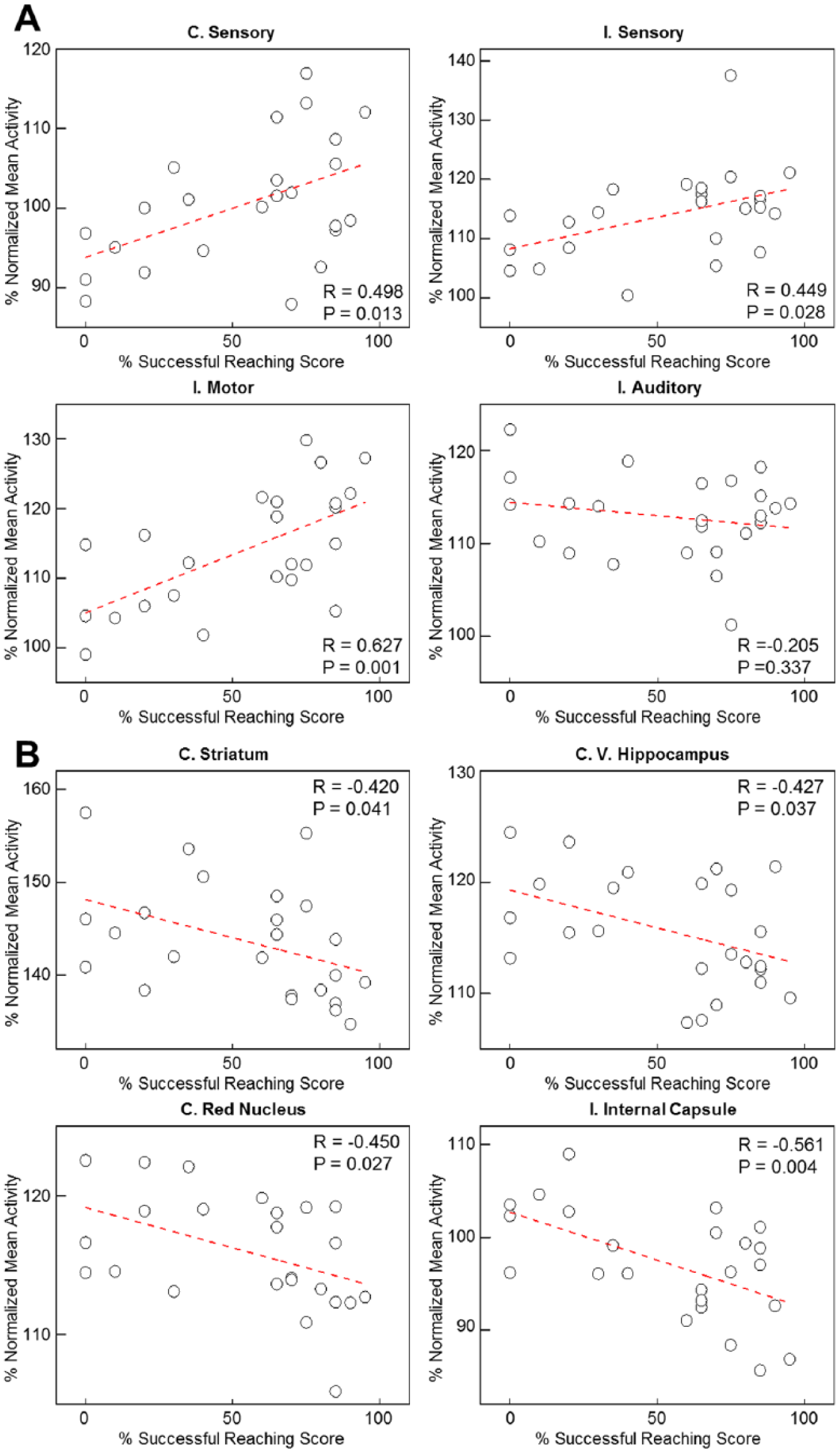
Pearson correlation analysis between resting-state brain activity and reaching performance for SPRT-trained animals in the incomplete destruction group (n = 8, 3 time points per animal). (A) Positive correlations between metabolic activity and reaching scores in the single-pellet reaching task for 4 different cortical regions: the contralesional sensory cortex, and the ipsilesional sensory, motor, and auditory cortices. (B) Negative correlations between metabolic activity and reaching scores for 4 different subcortical regions: the contralesional striatum, ventral hippocampus, and red nucleus, and the ipsilesional internal capsule (P < .05).
Discussion
We compared behavioral recovery and longitudinal changes in RSA in the presence or absence of reach training in an animal model of capsular stroke. We found that daily postlesional reach training enhanced motor recovery only in the group with partial destruction of the PLIC (ST-IDG). The microPET study revealed a greater reduction in cortical diaschisis in this group than in groups in which the PLIC was completely destroyed (CDG). In contrast, there was no reduction in diaschisis or motor recovery in the groups that did not receive daily postlesional reach training (NoST-IDG and NoST-CDG). Furthermore, reach training induced enhanced activation of subcortical structures, such as the red nucleus. These findings suggest that different neuronal remodeling may occur, depending on the degree of integrity of the capsular fibers and the presence or absence of reach training. The hypothesized “recovery circuit” that is activated by training after capsular stroke may, therefore, require the presence of some intact internal capsule fibers.
Task-specific rehabilitative exercises are thought to specifically enhance motor recovery in stroke patients.1,18,19 In our study, it is important to note that, although animals in both the ST-IDG and the NoST-IDG had only partial destruction of the capsular fibers, the group that did not receive training (NoST-IDG) did not show any motor recovery. This suggests that task-specific training is mandatory for enhancement of motor recovery in the capsular infarct model.
The microPET results showed prominent bilateral deactivation over a wide area of the cortex. This pattern of deactivation is compatible with diaschisis at rest because the deactivated areas correspond to a reduced level of metabolism distant to the lesion site (capsular infarct). 20 Cortical diaschisis after subcortical lesions (eg, thalamic or basal ganglia lesions) is distinctive: it is observed more commonly than cortical diaschisis after cortical lesions.21,22 Unilateral thalamic lesions are frequently accompanied by cortical diaschisis and are correlated with neuropsychological impairments. 21 Although it is not completely clear why subcortical lesions are prone to generating wide areas of cortical diaschisis, for capsular infarcts, it may be because the PLIC is the most condensed section of the pyramidal tract and has extensive connections with motor-related structures. Thus, destruction of the PLIC is likely to result in a collapse of related networks, including bilateral motor and sensory cortices. In addition, it is interesting to note that destruction of the PLIC also influences metabolic activity in cross-modal structures such as the auditory cortex.
In this study, capsular-infarct-induced diaschisis was related to training-sensitive recovery in animals with a partial capsular infarct (the ST-IDG). The reductions in diaschisis in ipsilesional motor and sensory cortices and the contralesional sensory cortex were correlated with increased reaching scores, indicating that the reach training may have contributed to the emergence of a recovery circuit involving these areas. However, motor recovery was only observed in the group that maintained partial integrity of the capsular fibers and also received daily reach training. Although reduced diaschisis was also observed in the group with complete destruction of the capsular fibers, the magnitude of reduction was smaller, and it was not correlated with motor recovery, indicating that reduced diaschisis alone is not sufficient for behavioral recovery. However, if an area of diaschisis persists despite reach training, this may be a biomarker to indicate that the recovery process is not progressing well. The reduced diaschisis presented in this study is unlikely to be the only neural signature correlated with recovery; interactions with other neural substrates are also likely to be required. Further investigation will be required for a more refined understanding of how the area of diaschisis relates to the mechanisms underlying motor recovery.
The relationship between lesion-related and recovery-related networks after cortical stroke is controversial because the 2 can share the same physical regions. Lesion-related areas, including the cortical infarct itself, may display widespread functional changes responsible for the observed neurological deficits. Recovery-related cortical areas surrounding the infarct lesion and in unaffected regions may display hyperexcitability related to functional recovery. 23 This hyperexcitability has been linked to a reduction in GABAA (γ-aminobutyric acid) receptors, possibly leading to unmasking of preexisting silent connections. 24 In contrast, differentiation and transhemispheric spreading have been proposed to underlie the genesis of diaschisis.25,26 Given that functional changes that exacerbate the neurological deficits and aid recovery can occur simultaneously and in the same location, both phenomena may be the result of physiological cascades after the stroke.
Our study shows that some subcortical neural structures show similar patterns of activation regardless of the presence or absence of reach training following infarct lesioning. Because natural recovery and training-dependent compensation can occur simultaneously after stroke, it is difficult to distinguish between the recovery patterns. 27 However, in this study, we were able to categorize subcortical activations into those that were training dependent and those that were training independent (natural recovery). In other words, training-independent activations were observed in all experimental groups, whereas training-dependent activations were observed only in the groups receiving training. It was noticeable that a greater number of neural structures in the noninjured hemisphere showed natural-recovery-related activation in our study than observed in other studies. Our results showed that activation of the bilateral internal capsule and bed nucleus of the stria terminalis and the contralesional striatum, thalamus, and dorsal hippocampus was training independent, whereas activation of the contralesional red nucleus, ventral hippocampus, and hypothalamus was training dependent. It should be noted, however, that activation of the hypothalamus was likely related to the rewarding effects of sucrose pellet consumption during the reaching task. In addition, activation of some subcortical structures was time dependent: the group that showed behavioral recovery with reach training (ST-IDG) tended to exhibit early disappearance of activation, whereas the groups that did not show behavioral recovery had persistent or late emergence of subcortical activations. This is compatible with previous studies in humans and animals reporting that areas of activation that are expanded during stroke recovery progressively return to normal in association with recovery.10,18,28
Previous animal and human studies have suggested that stroke recovery occurs via cerebral plasticity, including functional changes (eg, synaptic efficiency) as well as structural changes (eg, synaptic sprouting). 18 However, our results show that such plasticity has a limited ability to produce recovery after complete destruction of the PLIC, suggesting that remaining intact capsular fibers play an important role in any recovery of motor function after capsular infarct. There is recent evidence demonstrating that poststroke recovery is partly associated with white-matter reorganization. 29 MRI studies have also shown modification of white matter, such as the corpus callosum and internal capsule, in a focal stroke model.30,31 Furthermore, a recent study showed that learning a motor skill requires the formation of myelin to strengthen the connectivity within the relevant neural circuit. 32 Therefore, reach training in this experiment likely contributed to reorganization of the white matter in the remaining capsular fibers. We hypothesize that the remaining capsular fibers, which contain oligodendrocytes capable of producing myelin, are actively involved in the white-matter reorganization. Taken together, motor skill learning and the presence of spared capsular fibers may conjunctively contribute to white-matter plasticity, leading to better motor recovery in our capsular stroke model.
Implications and Conclusions
Task-specific training is routinely used to strengthen compensatory mechanisms in stroke recovery. In this experiment, we showed that 2 conditions are mandatory for enhancing motor recovery in capsular stroke: the partial preservation of capsular fibers and rehabilitative reach training. The group that met these conditions showed motor recovery, whereas the groups that did not meet these conditions showed no recovery. Therefore, rehabilitation strategies may differ depending on the magnitude of the capsular lesion. If there is major destruction of the capsular fibers, simple task-specific training may not be sufficient to promote recovery from the neurological deficits. We suggest that alternative means of neuromodulation, such as electrical and pharmacological interventions, should be combined with task training in such circumstances to elicit the most efficient recovery.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Institute of Medical System Engineering (iMSE; K04804) and the “GIST-Caltech Research Collaboration” Project through a grant provided by GIST in 2016, and by the Basic Science Research Program through NRF of Korea, funded by the Ministry of Science, ICT and Future Planning (NRF-2013R1A2A2A01067890).
