Abstract
Background. Previous studies have demonstrated improved neurobehavioral outcomes when prosthesis users learn task-specific behaviors by imitating movements of prosthesis users (matched limb) compared with intact limbs (mismatched limb). Objective. This study is the first to use a unique combination of neurophysiological and task performance methods to investigate prosthetic device training strategies from a cognitive motor control perspective. Intact nonamputated prosthesis users (NAPUs) donned specially adapted prosthetic devices to simulate the wrist and forearm movement that persons with transradial limb loss experience. The hypothesis is that NAPUs trained with matched limb imitation would show greater engagement of parietofrontal regions and reduced movement variability compared with their counterparts trained with a mismatched limb. Methods. Training elapsed over 3 days comprised alternating periods of video demonstration observation followed by action imitation. At the beginning and end of the training protocol, participants performed a cued movement paradigm while electroencephalography and electrogoniometry data were collected to track changes in cortical activity and movement variability, respectively. Results. Matched limb participants showed greater engagement of motor-related areas while mismatched limb participants showed greater engagement of the parietooccipital system. Matched limb participants also showed lower movement variability. Conclusions. These results indicate that the type of limb imitated influences neural and behavioral strategies for novel prosthetic device usage. This finding is important, as customary prosthetic rehabilitation with intact therapists involves mismatched limb imitation that may exacerbate challenges in adapting to new motor patterns demanded by prosthesis use.
Introduction
Action-observation therapy is a recently developed rehabilitation protocol that aims to enhance rehabilitation of motor deficits through the observation of daily actions combined with concomitant physical training of the same observed actions. 1 In action-observation therapy, patients observe demonstrations of actions and then imitate the actions themselves. This form of training has been shown to have positive rehabilitative effects on motor performance in stroke survivors,1-8 adults with Parkinson disease, 9 aphasia, 10 and children with cerebral palsy. 11 The proposed mechanism underlying these beneficial results is a shared action encoding network in the motor-related cortical regions that is activated by both the observation and execution of a motor task. 1 This training strategy provides an opportunity to further stimulate the cortical regions responsible for action encoding through task-specific action observation.
Our previous electroencephalography (EEG) study suggested that prosthesis users who imitated the movements of intact actors (mismatched limb imitation) elicit a unique set of cortical activations. 12 Instead of solely engaging the parietofrontal mechanism typically involved in planning and executing tool-use movements and pantomimes,13,14 activity was also observed in the right parietal and occipital cortical regions. It has been suggested that these right parietooccipital areas are part of a mentalizing system that may be responsible for the visuomotor processing of unfamiliar and motor dissonant actions outside of one’s own movement repertoire. 15 Importantly, the typical pattern of parietofrontal system activity could be observed in prosthesis users, but only when they imitated actions performed by other prosthesis users with a matching prosthetic limb (matched limb imitation). 12
Further behavioral studies sought to investigate the behavioral effects of matched limb action imitation in intact users of a specially designed prosthetic device that fits over an intact limb (Figure 1A). Nonamputated prosthesis users (NAPUs) who performed matched limb imitation showed a decrease in movement variability compared with those who performed mismatched limb imitation. 16 This finding has particular relevance with regard to therapy aimed at prosthesis training. Occupational therapists administering prosthesis training are normally intact and able-bodied individuals themselves. Thus, from the onset of motor training, persons with amputations are tasked with learning to use their device from an individual with 2 sound limbs. 17 The resulting scenario is one where a person with amputation must imitate motor tasks performed by an intact limb and may necessitate atypical action encoding activity. Deviations from normal control strategies may influence the degree to which a patient successfully incorporates their device into activities of daily life.18,19 Knowledge gained regarding the action encoding networks involved in imitating an intact limb with a prosthesis may reveal insights into the process of adapting to the functional constraints of a prosthetic limb.
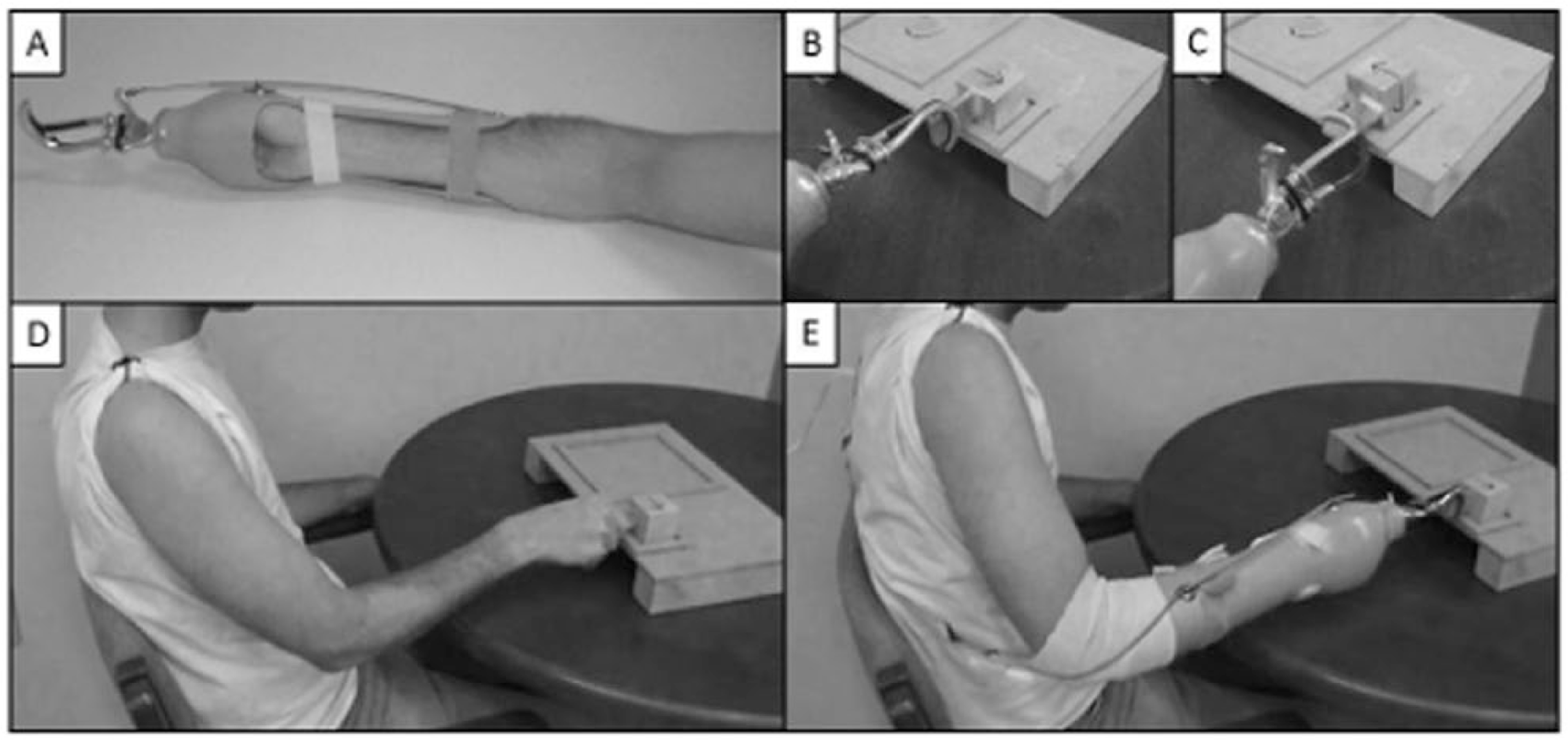
Prosthesis with Hosmer Model 5X Hook terminal device (A), workspace board and block used for motor task in starting position (B) and rotated 90° clockwise (C), screenshots of intact (D) and nonamputated prosthesis user (E) video demonstrations observed by participants.
The aim of the current study is to investigate the longitudinal effects of a matched limb imitation training paradigm on the cortical action encoding activity and motor behavior in NAPUs. The primary hypothesis is that matched limb training would result in greater longitudinal activity in the parietofrontal regions consistent with action encoding. Mismatched limb training would show greater activity in the parietooccipital regions consistent with mentalizing. Secondarily, due to the established importance of the parietofrontal system in planning and executing movements, we hypothesized that matched limb training would result in reduced movement variability compared with mismatched limb training. Together, these results would suggest that matched limb demonstration training may be beneficial in the process of adapting to a novel prosthesis.
Methods and Materials
Participants
Twenty right-handed intact participants were recruited for this study (14 males, mean age = 24.1 ± 4.2 years, range = 18-33 years). The Edinburgh Handedness Inventory 20 was used to confirm handedness. Signed informed consent was acquired from all participants according to the procedures set forth by the institutional review board at The Georgia Institute of Technology. The experimental paradigm required each participant to attend 5 sessions (Figure 2, top panel): an initial data collection (day 1), 3 rounds of prosthesis training (days 2-4), and a final data collection (day 5).
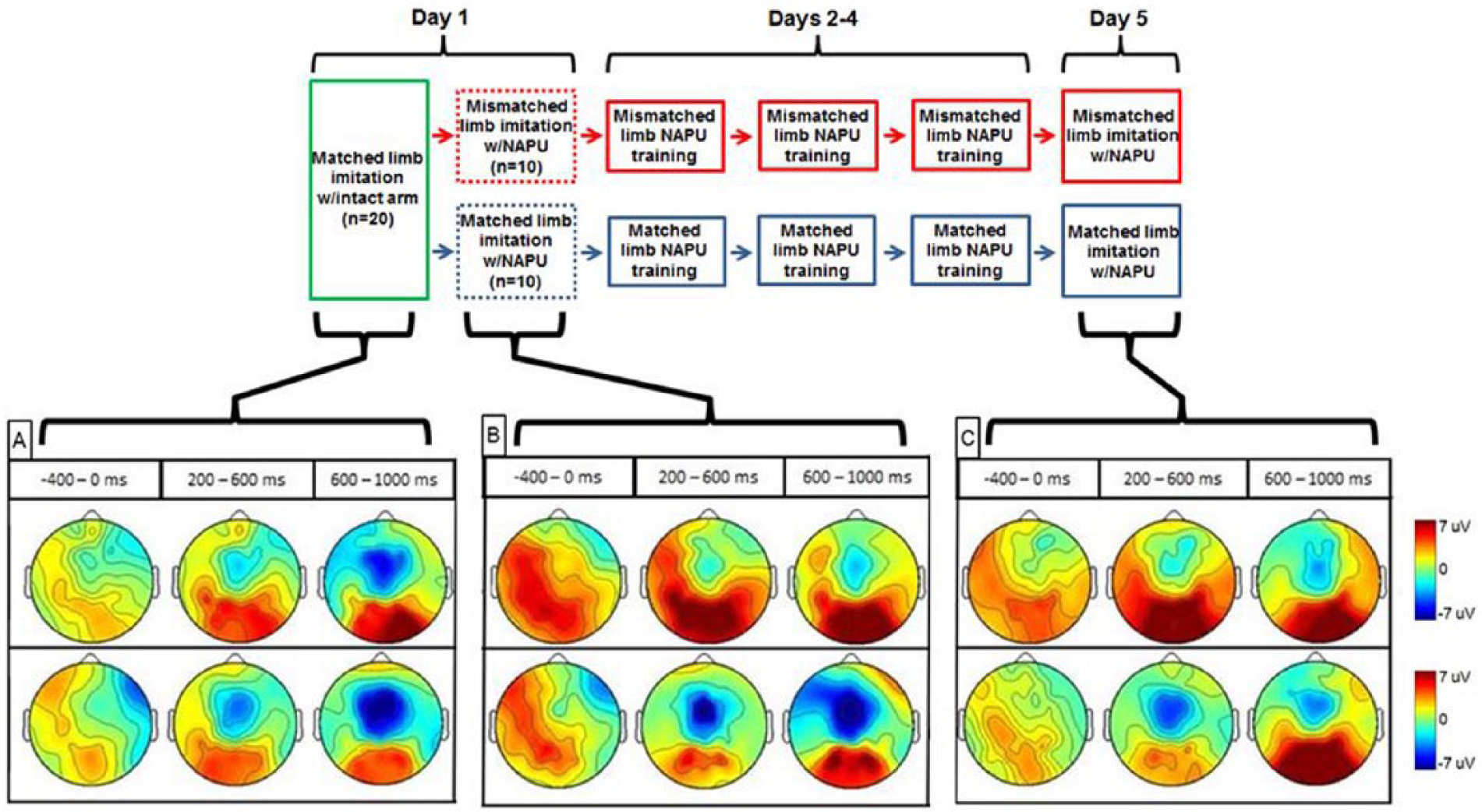
Diagram depicting experimental paradigm for matched and mismatched limb imitation training groups (top panel) and grand-averaged electrode headplots for all experimental conditions (bottom panel). For each condition, data were averaged over three 400 ms time windows corresponding to the phases of movement planning (−400 to 0 ms), early execution (200 to 600 ms), and late execution (600 to 1000 ms).
Nonamputated Prosthesis System
We utilized a specially adapted prosthesis socket that accommodates an intact participant’s forearm and hand. The device is designed to simulate the loss of forearm pronation and supination and wrist flexion and extension that a transradial amputee would experience after the loss of the wrist. No active prehension with the prosthesis was required during the task, as the block was fixed in the terminal device. Subjects were not required to exert force through the cabling harness during the movement. In amputees, the altered kinematics of the limb-prosthesis system necessitates the development of novel motor control strategies that yield unique movement characteristics.21,22 Similar devices have been successfully implemented in intact participants in our own work 16 and in other studies that investigated modes of prosthesis control 23 and the design of prosthesis training protocols.17,24 This approach allows us to evaluate the potential benefits of matched-limb action observation training in a controlled paradigm.
Block Rotation Motor Task
Participants viewed and imitated video demonstrations of a block rotation motor task adapted from the Southhampton Hand Assessment Procedure.16,25 The task involves (a) placement of the wooden cube within the square target area (Position 1, Figure 1B); (b) lifting the cube, rotating 90° clockwise, placement back into target area (Position 2, Figure 1C); and (c) lifting the cube, rotating 90° counterclockwise, placement back into target area (Position 1, Figure 1B).
Data Collection Sessions
During the data collection sessions (days 1 and 5), participants were fitted with a 58-channel EEG cap (Electrocap, Eaton, OH) that recorded scalp potential activity (1 kHz sampling rate, filtered at DC-100 Hz) via the Synamps 2 data acquisition system (Compumedics Neuroscan, Charlotte, NC). Three twin-axis electrogoniometers and one single-axis torsiometer (models SG110/150 and Q110/Q150, respectively, Biometrics Ltd, Newport, UK) were fitted on the right arm of each participant and connected to an 8-channel MyoSystem data collection system (model 1400L, Noraxon, Scottsdale, AZ). ELGON data were sampled with 1 kHz frequency.
There were 7 degrees of freedom in the arm that were of interest in this study: wrist flexion/extension (WFE), wrist abduction/adduction (WAA), elbow flexion/extension (EFE), shoulder abduction/adduction (SAA), horizontal shoulder flexion/extension (SFE), forearm supination/pronation (FRO), and shoulder internal/external rotation (SRO). The placement of the sensors was informed by previous studies in upper extremity kinematics,26-30 the manufacturer’s user manual, 31 and in our previous study that involved the same block rotation task. 16
Participants were seated in the upright position at a workspace board (Figure 1D and E) with a computer screen placed 1.5 m away. Demonstration videos lasted 30 seconds, contained 15 repetitions of the block rotation task, and were observed while subject remained motionless. Each video showed an expert user performing the task 15 consecutive times with either their intact hand or prosthesis, depending on the condition. To minimize performance variability, each demonstration video was identically paced by a metronome. Following video observation, initiation of the movements was controlled using a visual presentation cueing scheme developed in Stim (Compumedics Neuroscan, Charlotte, NC). On conclusion of the video demonstration, participants remained motionless and fixated on a white cross for a randomized baseline period of 4.0 to 6.0 seconds. Participants were then prompted to begin planning for the imitation movement on seeing a “Get Ready!” cue that appeared for 1.0 second. A final cue then appeared cueing participants to “Move!” and remained on the screen for 10.0 seconds. This cued movement sequence was repeated 40 times, for a total of 200 individual repetition movements. On day 1, subjects performed this paradigm first with their intact hand, then with the NAPU. On day 5, subjects only performed the paradigm with the NAPU.
NAPU Training Sessions
NAPU training sessions (days 2-4) commenced with video presentation of either mismatched limb (Figure 1D) or matched limb (Figure 1E) demonstrations (according to their assigned group), which lasted 30 seconds and contained 15 movement repetitions. Participants fitted with the prosthesis were then instructed to imitate the task seen in the video for 2 continuous minutes at a self-selected pace. This pairing of observation and imitation was repeated 4 times, with a break given half-way to mitigate effects of fatigue. During each training session, each participant performed approximately 200 repetitions of the block rotation task.
Data Analysis
Using Scan4.5 (Compumedics Neuroscan, Charlotte, NC), continuous EEG data were band-pass filtered from DC-30 Hz. Data were then segmented into 3000 ms epochs capturing the 1500 ms preceding and following the presentation of the “Move!” cue that serves as the zero point for all event-related potential (ERP) data. Linear detrending of the entire sweep and baseline correction with a baseline interval of −1600 ms to −1100 ms was then performed. Custom MATLAB (version R2012a, The MathWorks Inc, Natick, MA) software was employed for artifact averaging and regression analysis to remove ocular movement artifacts. 32 Data were averaged over three 400 ms time windows corresponding to the phases of movement planning (−400 to 0 ms), early execution (200-600 ms), and late execution (600-1000 ms). The choice of these particular time windows was based on our previous work in which cortical activity differences in prosthesis users were found during a similar observation and imitation paradigm. 12 Data were further grouped into regions of interest that were defined in the left/right premotor areas (LPM/RPM), left/right motor areas (LM/RM), left/right parietal areas (LP/RP), and occipital area (OCC).12,33
Using custom MATLAB software, all ELGON data were low-pass filtered at 6 Hz with a fourth-order Butterworth filter and epoched, averaged, and time normalized using the methods described in our previous study. 16 Each instance of peak shoulder abduction was chosen to mark the beginning of each movement cycle, while the next instance was chosen as the end of that movement cycle. Behaviorally, these time points correspond to the transitions between counterclockwise and clockwise rotations in the block rotation task. Prior work has demonstrated that movements with lower variability are associated with enhanced technique.34,35 The coefficient of variation (CV) for all movement groups was calculated on a participant level according to CV(%) = MEAN(%)/STD(%), where MEAN(%) and STD(%) are the angular displacement standard deviation and mean as functions of percent movement cycle, respectively. All reported displacement and CV values are calculated relative to the reference position at the beginning of each movement.
Statistical Design
On day 1, subjects in the matched and mismatched groups first performed the experimental tasks with their intact hand (INT-MAT-D1 and INT-MIS-D1, respectively). In this same session, subjects in the matched and mismatched groups then executed the tasks with the NAPU (NAPU-MAT-D1 and NAPU-MIS-D1, respectively). After completing the 3 days of training, subjects in matched and mismatched groups performed the experimental tasks on day 5 with the NAPU (NAPU-MAT-D5 and NAPU-MIS-D5, respectively).
A repeated measures multivariate analysis of variance (rmMANOVA) was performed to evaluate the impact of training group type (matched, mismatched) on ERPs in the 7 brain regions of interest (LPM/RPM, LM/RM, LP/RP, OCC) across the 3 data collection sessions (intact hand day 1, NAPU day 1, NAPU day 5). Separate analyses were performed for each movement phase (planning, early execution, late execution). This statistical approach has been used previously to investigate longitudinal changes in EEG activity during stroke rehabilitation. 36 The behavioral data were also similarly examined with an rmMANOVA to investigate the influence of training group type on the angular displacement and CV of the 7 joint-level degrees of freedom (WFE, WAA, EFE, SAA, SFE, FRO, SRO) across the 3 data collection sessions. All statistical analyses were conducted using SPSS Statistics software (version 22, The IBM Corporation, Armonk, NY). Subsequent post hoc t tests were performed with significance set at P < .05 using Bonferroni correction.
Results
EEG
Effects of Matched Limb Imitation
An rmMANOVA revealed main effects of video (F(3, 564) = 32.8, P < .001), electrode (F(6, 566) = 75.8, P < .001), as well as an interaction effect of video and electrode (F(6, 566) = 2.3, P = .037). An important follow-up comparison is that of cortical activity between groups on day 1 after the initial donning of the NAPU (Figure 2B). Post hoc analyses indicated greater negativity for the matched limb imitation group in: RPM, RP, and OCC for the planning phase (all comparisons F(6, 566) ≥ 4.1, P ≤ .043); LPM, RPM, LM, RM, LP, RP, and OCC for the early execution phase (all comparisons F(6, 566) ≥ 12.4, P < .001); LPM, RPM, LM, RM, and RP for the late execution phase (all comparisons F(6, 566) ≥ 5.2, P ≤ .022). Comparison between the 2 training groups on day 5 was made in order to investigate the effects of video type on cortical activity once training was completed (Figure 2C). Post hoc analyses revealed greater negativity for the matched limb imitation group in: RP for the planning phase (F(6, 566) = 8.3, P = .004); LPM, RPM, LM, RM, LP, RP, and OCC for the early execution phase (all comparisons F(6, 566) ≥ 4.6, P ≤ .032); LP for the late execution phase (F(6, 566) = 6.6, P = .01).
Longitudinal Effects of Matched Limb Imitation Training
An rmMANOVA revealed a main effect of session (F(6, 561) = 37.1, P < .001), interaction effects of session and video (F(6, 561) = 34.6, P < .001), session and electrode (F(6, 566) = 5.4, P < .001), and a 3-way interaction between session, video, and electrode (F(6, 566) = 5.2, P < .001). An important follow-up comparison is that of cortical activity within each respective group as the training progressed. There are specific comparisons of interest: intact hand versus NAPU performance on day 1 (referred to as the effect of donning the prosthesis) and NAPU performance on day 1 versus that on day 5 (referred to as the effect of prosthesis training).
Exclusive to the mismatched limb group, post hoc analyses indicated an effect of donning the prosthesis that involved decreased negativity in: LPM, RPM, LM, RM, LP, RP, OCC for the planning phase (all comparisons F(6, 566) ≥ 4.7, P ≤ .01); LPM, LM, LP, RP, OCC for the early execution phase (all comparisons F(6, 566) ≥ 3.3, P ≤ .041); LPM, RPM, LM, LP, and RP for the late execution phase (all comparisons F(6, 566) ≥ 3.50, P ≤ .032; Figure 3). Unique to the matched limb group, post hoc analyses demonstrated an effect of training that included decreased negativity in: LPM, RPM, LM, RM, LP, RP, and OCC for the late execution phase (all comparisons F(6, 566) ≥ 6.0, P ≤ .003; Figure 4).
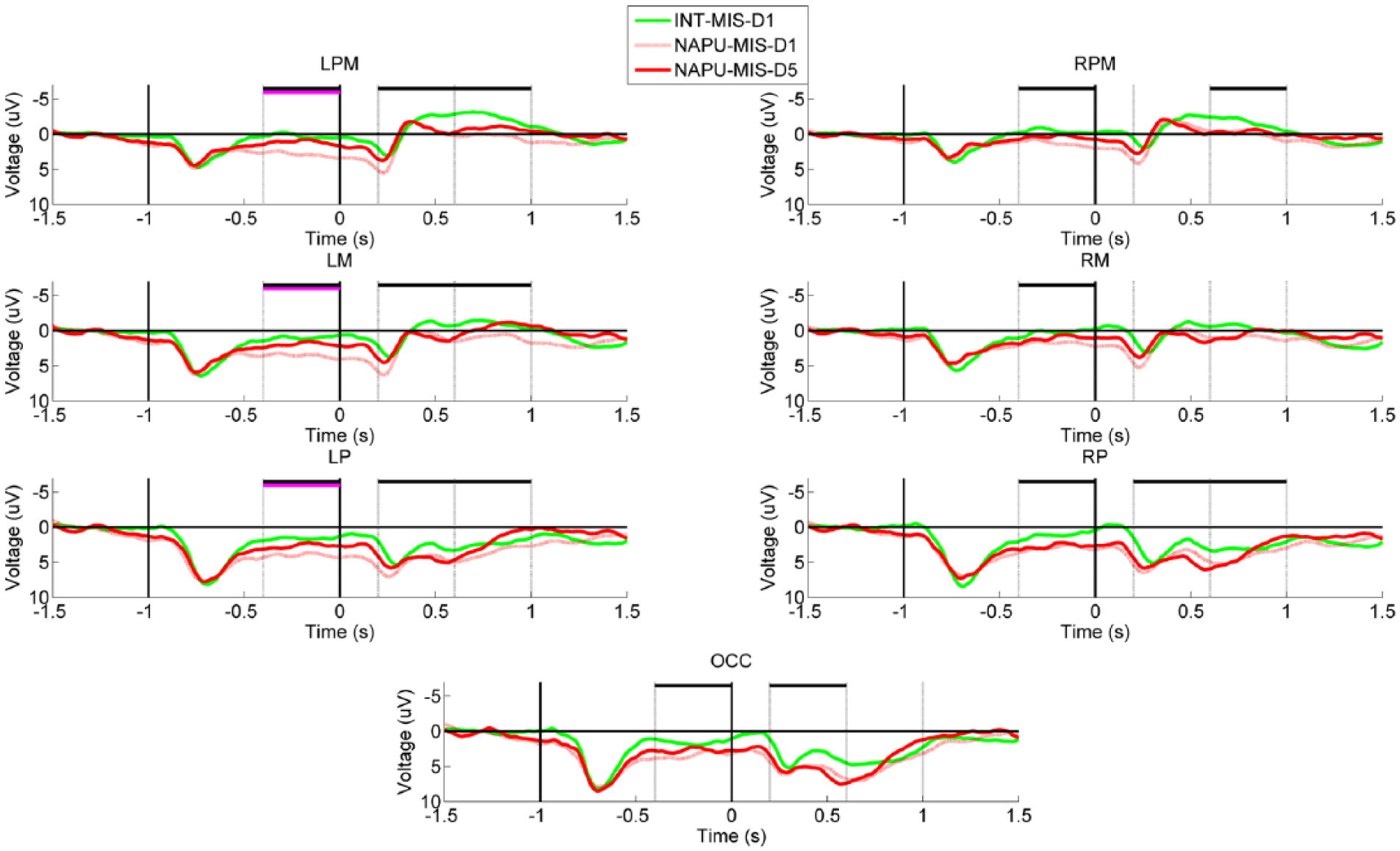
Grand-averaged region-level voltage plots showing the longitudinal effects observed exclusively in the mismatched limb imitation training groups. The presentation of the “Get Ready” and “Move” cues are marked with vertical black lines at −1.0 and 0.0 seconds, respectively. Time windows corresponding to the phases of movement planning (−400 to 0 ms), early execution (200 to 600 ms), and late execution (600 to 1000 ms) are marked with gray vertical lines. There are 2 comparisons of interest: intact hand versus pretrained nonamputated prosthesis user (NAPU; significant differences marked with horizontal black lines, P < .05) and pretrained NAPU versus trained NAPU (significant differences marked with horizontal pink lines, P < .05).
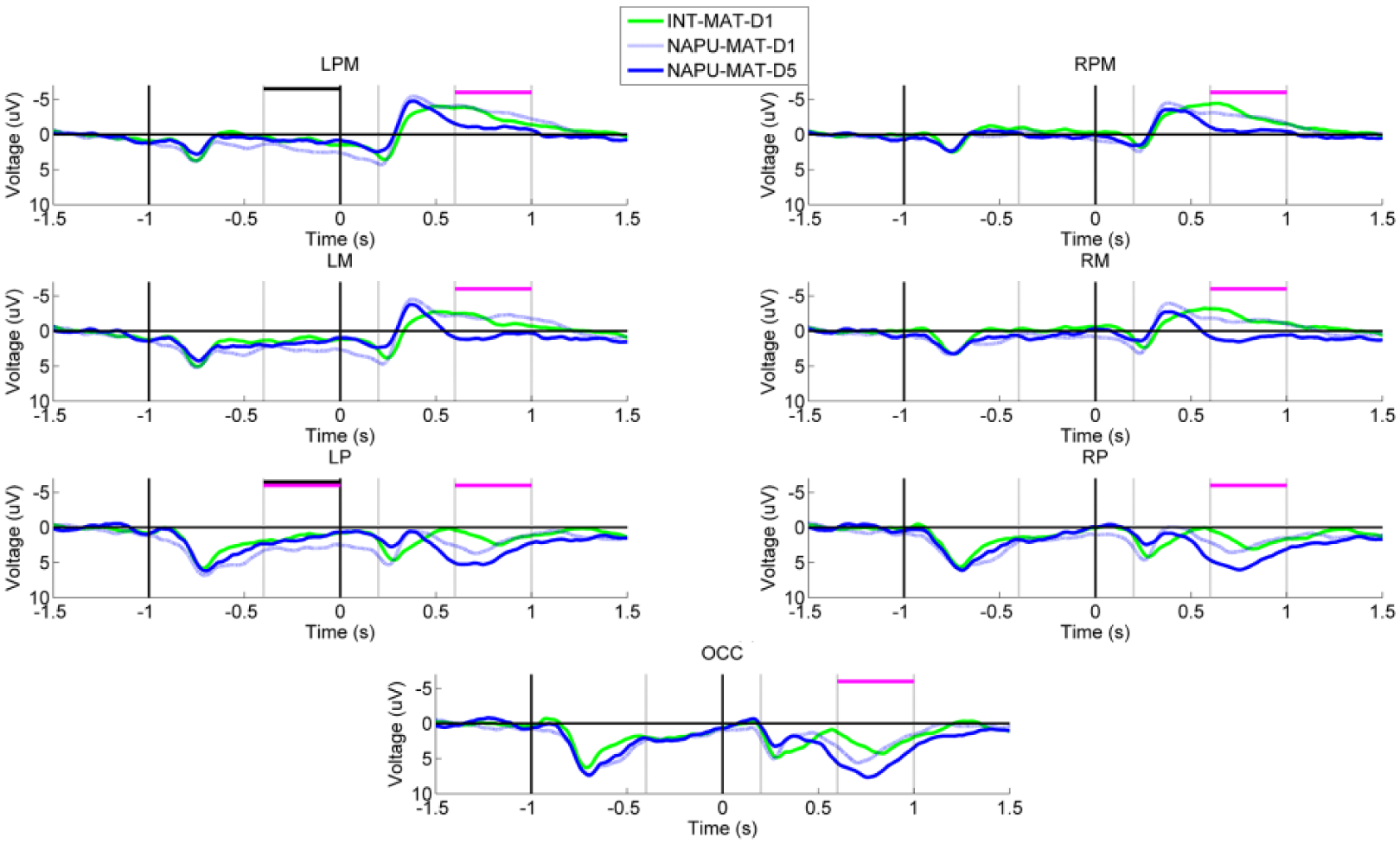
Grand-averaged region-level voltage plots showing the longitudinal effects observed exclusively in the matched limb imitation training groups. The presentation of the “Get Ready” and “Move” cues are marked with vertical black lines at −1.0 and 0.0 seconds, respectively. Time windows corresponding to the phases of movement planning (−400 to 0 ms), early execution (200 to 600 ms), and late execution (600 to 1000 ms) are marked with gray vertical lines. There are 2 comparisons of interest: intact hand versus pretrained nonamputated prosthesis user (NAPU; significant differences marked with horizontal black lines, P < .05) and pretrained NAPU versus trained NAPU (significant differences marked with horizontal pink lines, P < .05).
To summarize, in the mismatched group, donning of the prosthesis uniquely affected the right hemisphere in the planning phase, and affected both hemispheres in the execution phases. Contrastingly, there were minimal effects of donning the prosthesis in either hemisphere during movement planning or execution in the matched group. Matched limb prosthesis training yielded longitudinal effects in both hemispheres, particularly in the late execution phase. Contrastingly, there were effects of mismatched limb prosthesis training in left hemisphere during movement planning.
Movement Angular Displacement
An rmMANOVA was performed on angular displacement data to investigate the effect of donning the prosthesis on intact limb kinematics. The analysis revealed main effects of session for WFE, WAA, SAA, SFE, FRO, and SRO (all comparisons F(2, 450) ≥ 5.2, P ≤ .006). After donning the prosthesis, post hoc comparisons demonstrate decreased angular displacement in WFE, WAA, and FRO, as well as increased angular displacement in SAA (all comparisons F(2, 450) ≥ 6.8, P ≤ .002). Behaviorally, the range of motion was significantly reduced and/or constrained by the device in wrist (WFE, WAA) and forearm (FRO). These kinematic restrictions were compensated for by changes to the range of motion in the remaining unconstrained degrees of freedom in the shoulder (SAA).
Movement Coefficient of Variation
An rmMANOVA was performed on CV data from each degree of freedom to investigate the effect of matched and mismatched NAPU training on movement variability (Figure 5A-D). The analysis revealed a main effect of video for SAA, SFE, FRO, and SRO (all comparisons F(1, 450) ≥ 7.7, P ≤ .006), a main effect of session for EFE and FRO (all comparisons F(2, 450) ≥ 4.1, P ≤ .018), and an interaction effect of video and session for WFE, WAA, and SFE (all comparisons F(2, 450) ≥ 3.5, P ≤ .032).
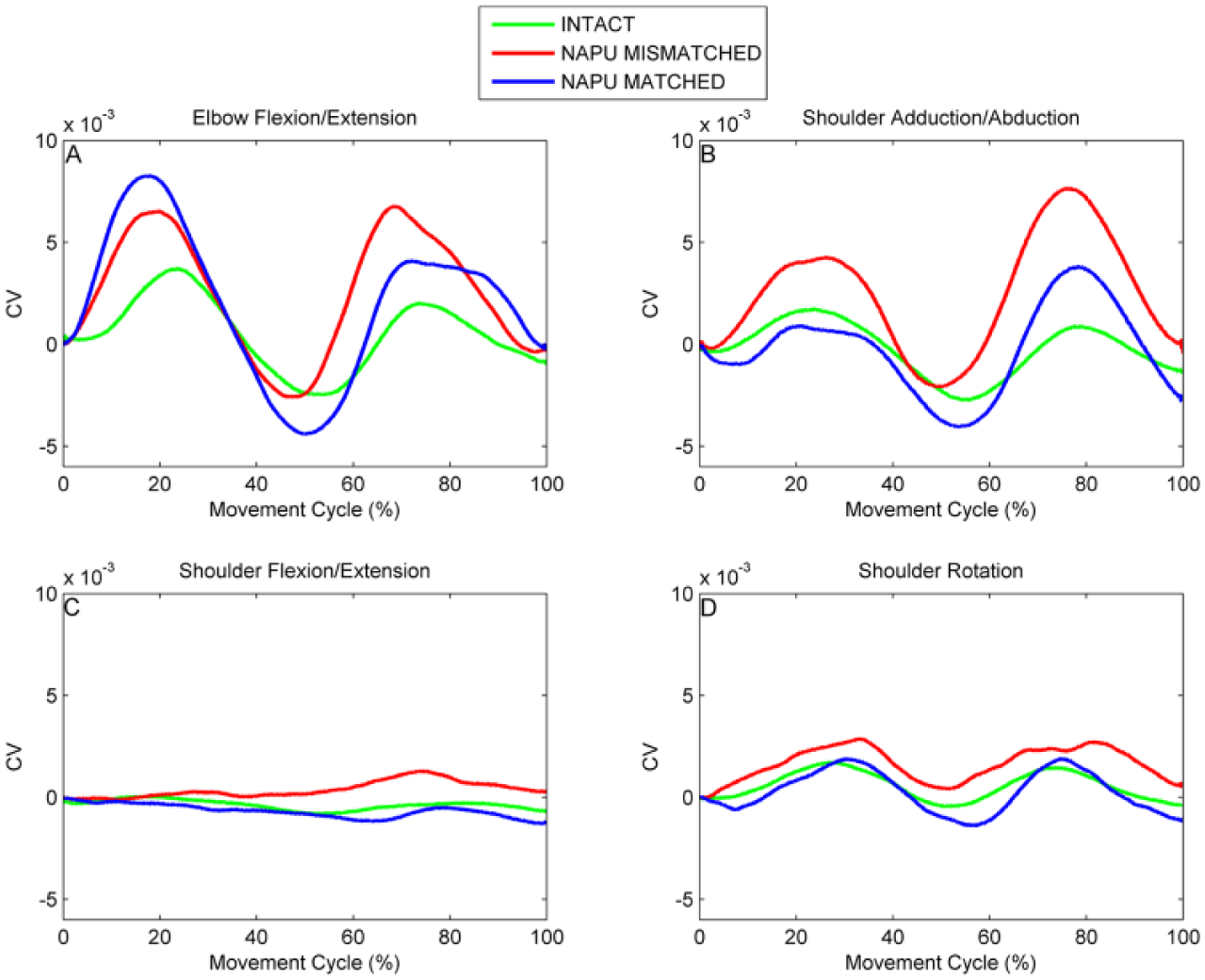
Grand-averaged coefficients of variation (CV) plots showing the effects of matched versus mismatched limb imitation for EFE (elbow flexion/extension; A), SAA (shoulder adduction/abduction; B), SFE (shoulder flexion/extension; C), and SRO (shoulder rotation; D).
Elbow Flexion/Extension (EFE)
A post hoc comparison of EFE CV data (Figure 5A) revealed a main effect of session (F(2, 450) = 5.9, P = .003) and no main effect of video (F(1, 450) = 0.024, P = .88) or interaction effect of session × video (F(2, 450) = 1.5, P = .24). Behaviorally, this result corresponds to a significant increase in CV from the intact hand condition (0.5 × 10−3) to the prosthesis conditions (pretrained: 2.4 × 10−3; trained: 2.1 × 10−3).
Shoulder Abduction/Adduction (SAA)
The results of a post hoc comparison of SAA CV data (Figure 5B) indicated a main effect of video (F(1, 450) = 14.8, P < .001), but no main effect of session (F(2, 450) = 2.4, P = .09) or interaction effect of session × video (F(2, 450) = 1.4, P = .26). Behaviorally, this result indicates a significantly lower movement CV in the matched (−0.5 × 10−3) versus mismatched imitation (1.8 × 10−3) group.
Shoulder Flexion/Extension (SFE)
Post hoc comparison on SFE CV data (Figure 5C) showed no main effect of session (F(2, 450) = 2.0, P = .14), but did show a main effect of video (F(1, 450) = 10.0, P = .002) and interaction effect of session × video (F(2, 450) = 5.5, P = .005). Post hoc analyses showed less movement CV in matched (−0.57 × 10−3) versus mismatched imitation (0.10 × 10−3) during the pretrained prosthesis condition (F(2, 450) = 11.6, P < .001).
Shoulder Internal/External Rotation (SRO)
A post hoc comparison of SRO CV data (Figure 5D) demonstrated no main effect of session (F(2, 450) = 1.2, P = .32) but did reveal a main effect of video (F(1, 450) = 7.8, P = .006), with no interaction effect of session × video (F(2, 450) = 2.8, P = .064). Behaviorally, this result corresponds to less movement CV in the matched (0.30 × 10−3) versus mismatched imitation (1.30 × 10−3) group.
Discussion
The goal of this study was to determine the longitudinal effects of a matched limb imitation training paradigm on the cortical action encoding activity and motor behavior in NAPUs. Matched limb participants showed greater engagement of the motor areas, while mismatched limb participants showed greater engagement of the bilateral parietooccipital areas. Furthermore, the matched limb imitation group showed a longitudinal reduction in motor area negativity, while the mismatched limb imitation group exhibited minimal effects of prosthesis training on cortical activity. Over the course of the paradigm, matched limb imitation participants also showed lower movement variability compared with those trained with mismatched limb imitation. Together, these results suggest that matched limb imitation may play an important neurobehavioral role in prosthetic device training and rehabilitation.
Effects of Matched Limb Imitation
The matched limb participants showed greater engagement of motor areas, while mismatched limb participants showed greater engagement of the bilateral parietooccipital system, an effect that was consistent across training. This aligns with our prior studies, suggesting that activation of the typical motor-area action encoding system is possible in prosthesis users, but only when they are able to imitate a limb state that matches their own. 12 These results also support the proposal that the degree to which the motor areas are engaged is a function of the motor resonance between the observed and the observer. 37 Furthermore, without such a match, the planning phase can no longer occur as normal and relies more heavily on the parietooccipital action encoding system.
As matched limb training seemed to aid in the development of motor representations, there were also notable effects in behavior. Overall, matched limb imitation showed less movement CV in shoulder adduction/abduction, flexion/extension, and internal/external rotation compared with mismatched limb imitation. In alignment with the hypothesis, this result suggests a behavioral advantage to matched imitation, as it yielded more consistent and prototypical movements in the joint principally responsible for adapting to the novel prosthesis kinematics. We are proposing that in the case of the matched limb imitation group, observing the movements with greater motor resonance and functional congruity enhanced the engagement of the motor-area action encoding system and better facilitated the formation of the new movement strategy.
Contrastingly, additional parietooccipital activity in the mismatched limb group may reflect an increased visuospatial demand of the imitation motor task. 15 This visuospatial demand may arise in the mismatched case due to the lack of relevant movement guidance provided by the video demonstration. The influence of prosthesis training type on the relative activity in the motor-area versus visuospatial action encoding systems may explain the differences in movement variability observed in the current study. This result and interpretation is corroborated with our previous study, in which mismatched limb imitation resulted in a greater shoulder adduction/abduction variability compared with matched limb imitation. 16
Longitudinal Effects of Matched Limb Imitation
The matched limb imitation group uniquely showed longitudinal decreases in bilateral premotor and motor activity after training was complete. Previous studies have shown that the learning of new motor tasks is accompanied by an initial increase in activity in the presupplementary motor, premotor, parietal, and prefrontal areas, and is then followed by a decrease in activity in these same areas as the motor task becomes more automatic.38-40 We suggest that matched limb imitation is more effective at facilitating automaticity of a novel motor task. The behavioral data discussed above support this interpretation, as matched limb imitation resulted in lower variability in several dimensions of shoulder movement.
Contrastingly, the mismatched limb imitation group revealed no effects of training on cortical activity. This group showed initial widespread right premotor, motor, parietal, and occipital activity during the planning phase that was sustained between the pretrained and trained prosthesis conditions. As discussed above, this increased activity in right parietooccipital areas may be indicative of a greater degree of visual comprehension required to complete the goal-directed movement.
Clinical Importance
As of 2005, there were over 1.5 million persons with amputation living in the United States, 548 000 of whom have some level of upper limb amputation. The total number of persons with amputation is forecast to rise to at least 2.2 million by 2020.41-45 It has been established that full functional adaptation of a prosthesis is not common; with 75% of amputees considering their devices to be primarily aesthetic while 33% reject the device due to lack of perceived utility. 46 Persons with amputation have cited inadequate training, limited usefulness of devices, and poor initial prosthetic experience as reasons for rejection. 47
The results of the current study suggest that matched limb training may promote positive neural and behavioral outcomes in prosthesis adaptation. Furthermore, the conversion of the therapist’s intact movements into movements that can be completed with a prosthetic device may contribute to the difficulties experienced by novice prosthesis users. While the movements presented here are basic, we propose that improvements in performance at this level may also be beneficial to more complex tasks that require combinations of simple movements. Additional work is needed that will involve more complex tasks. A follow-up regression analysis of coefficient of variation and peak movement voltage over motor cortex revealed no clear correlation as a result of training (NAPU-MAT-D5: r2 = .19; NAPU-MIS-D5: r2 = .03). It is possible that other outcome metrics such as beta band activity (15-30 Hz) in the motor cortex could yield better correlations between behavior and neural activity. 48 Future studies will investigate which neural variables characterize and predict movement quality.
An important distinction to be made for this study is that the experimental paradigm is not intended to replicate the entire rehabilitative process that a person with upper extremity amputation experiences with their occupational therapist. Instead, the experiment is designed to focus on one crucial aspect of this interaction that involves mismatched limb imitation of basic movements. 17 The extension of NAPU results to those of persons with limb deficiency must be carefully considered, and it is a topic of current research.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Institutes of Health 2009 Pre-Doctoral Prosthetics and Orthotics Research Fellowship Program; National Institutes of Health National Center for Medical Rehabilitation Research T32 Award (#5T32HD055180-03).
