Abstract
Peripheral nerve injury afflicts individuals from all walks of life. Despite the peripheral nervous system’s intrinsic ability to regenerate, many patients experience incomplete functional recovery. Surgical repair aims to expedite this recovery process in the most thorough manner possible. However, full recovery is still rarely seen especially when nerve injury is compounded with polytrauma where surgical repair is delayed. Pharmaceutical strategies supplementary to nerve microsurgery have been investigated but surgery remains the only viable option. Brief low-frequency electrical stimulation of the proximal nerve stump after primary repair has been widely investigated. This article aims to review the currently known biological basis for the regenerative effects of acute brief low-frequency electrical stimulation on axonal regeneration and outline the recent clinical applications of the electrical stimulation protocol to demonstrate the significant translational potential of this modality for repairing peripheral nerve injuries. The review concludes with a discussion of emerging new advancements in this exciting area of research. The current literature indicates the imminent clinical applicability of acute brief low-frequency electrical stimulation after surgical repair to effectively promote axonal regeneration as the stimulation has yielded promising evidence to maximize functional recovery in diverse types of peripheral nerve injuries.
Keywords
The Need for Electrical Stimulation: Poor Functional Recovery After Nerve Injury and Surgical Repair
Despite the regenerative capacity of injured peripheral neurons and the permissive growth environment in the peripheral nervous system, functional recovery remains frequently suboptimal after primary surgical repair.1-5 This is especially true in patients with traumatic nerve injuries close to the spinal cord and over long distances between the sites of transection and target reinnervation.6-8
Contrary to the belief of irreversible muscle atrophy being the cause, the major factors limiting functional recovery have been defined as (a) the duration of chronic axotomy (where injured neurons remain without peripheral targets and lose their regenerative capacity) and chronic denervation (referring to the short timeframe in which Schwann cells in the distal nerve stump can support regenerating axons) and (b) the slow growth of regenerating axons across surgical coaptation sites and/or nerve gaps that are coapted via autologous nerve grafts.9-12
To address this clinical issue, the potential of electrically stimulating injured peripheral axons has been investigated resulting in positive outcomes of enhanced nerve regeneration and functional recovery. The technique relies on the application of low-frequency electrical stimulation (ES) just after primary surgical repair of the transected peripheral nerve. 13 The duration of the ES paradigms varies but it is now generally agreed that low-frequency (20 Hz or less) ES paradigms are effective in enhancing functional recovery after peripheral nerve injuries in both animal models and in patients. 14 Furthermore, the technique can be readily combined with nerve reconstruction surgery in the operating room, making it very attractive to nerve surgeons. More recently, a potential use of daily stimulation of denervated muscle has been shown to accelerate reinnervation of the muscle after surgical repair of a transected nerve. 15
We focus our review on the biological basis for the regenerative effects of brief low-frequency ES on axonal regeneration, and we outline the recent clinical applications of the ES protocol to demonstrate the significant translational potential of this modality for repairing peripheral nerve injuries. The review concludes with a discussion of emerging new advancements in this exciting area of research.
Brief Electrical Nerve Stimulation Accelerates the Regeneration of Injured Peripheral Nerves
The basis for applying electrical stimulus to a peripheral nerve to promote nerve regeneration derives first from the studies of Hoffman. 16 These studies demonstrated accelerated axon sprouting in partially denervated muscle in response to ES. Sinusoidal ES at 50 to 100 Hz for 10 to 60 minutes was applied directly to the spinal cord or the nerve roots of the sciatic nerve following section of the L5 spinal root. Interestingly, moderate but not excessive exercise programs were later found to promote axonal sprouting in partially denervated muscles.17,18
The application of ES to nerve regeneration after crush injuries was later pursued in the laboratories of Nix and Hopf 19 and Pockett and Gavin. 20 In a rabbit model of crush injury, the former investigators demonstrated accelerated recovery of twitch and tetanic tensions and muscle action potentials in reinnervated soleus muscle when the soleus nerve proximal to the crush site was electrically stimulated continuously (24 hours/day) for 4 weeks at 4 Hz. 19 Pockett and Gavin 20 reported congruent findings of an earlier recovery of the toe spread reflex after a sciatic nerve crush injury in a rat with proximal ES delivered at 1 Hz for periods of 15 minutes to 1 hour. Similarly, a single 30-minute period of ES at 20 Hz was effective in accelerating recovery of whisker movement after facial nerve crush injury.21,22 However, these studies could not distinguish whether the ES affected more rapid reinnervation of denervated muscle fibers and/or accelerated axon regeneration. Al-Majed et al used a rat femoral nerve transection model to address the question of whether ES accelerated axon regeneration directly by retrogradely labelling the femoral motoneurons that regenerated their axons into the quadriceps motor nerve branch and/or into the saphenous sensory nerve branch. 13 The frequency of the applied ES that was chosen for this study was 20 Hz based on the mean firing frequency of motoneurons in animals and humans. 23 Continuous ES was applied for 1 hour, 24 hours, 1 week, or 2 weeks. The 2-week period was chosen because, up until 2 weeks after using a locally made implantable stimulator nerve transection and repair, preferential nerve regeneration into appropriate motor or sensory pathways is random. Al-Majed et al reported that ES accelerated axonal regeneration and preferential motor nerve regeneration at all durations of application. 13 These and later experiments provided direct evidence that ES of injured peripheral nerve accelerated axon outgrowth from both motor and sensory neurons following injuries.13,24-26 For motor nerve regeneration, ES was as effective when applied for periods of 1 hour to 2 weeks, but only a 1-hour period was effective for sensory nerve regeneration.13,24-26
The key supportive finding to confirm the effect exerted by ES of injured peripheral nerves was via the application of tetrodotoxin (which acts to block retrograde conduction of action potentials), demonstrating total negation of any beneficial effect of ES following injury and repair on nerve regeneration. 13 Moreover, the experiments localized the ES effect to the neuronal cell bodies.
Many studies from several different laboratories have confirmed the efficacy of ES in promoting nerve regeneration in several rodent models of nerve injury, including sciatic, femoral, and facial nerve injuries. These studies include those of from the laboratories of Navarro, 27 English, 28 Rafuse, 29 Jones, 30 and Huang. 31 These are summarized in a recent review. 6 The currently known mechanisms of action are summarized below and are further supported by synchronous studies.
Appropriate Pathway Selection by Regenerating Axons
In addition to timeliness, achieving proper functional recovery also requires accurate reinnervation of the affected motor and sensory pathways. Brushart and Mesulam, using retrograde tracing of the sciatic motoneurons that regenerated their axons into the common peroneal and tibial nerve branches, provided definitive evidence of random reinnervation of the nerve branches after nerve transection. 32 Consistent with these findings was that the random reinnervation of two or more denervated muscle targets by regenerating nerves was demonstrated in synergistic and antagonistic muscle compartments in both animals and human patients.33-35 However, there is a natural preference for regenerating motor axons to grow into motor rather than sensory nerve branches: in a rat femoral nerve injury model, motoneurons did indeed show preference for reinnervating the appropriate quadriceps motor nerve branch. 36 ES did not improve this motor selectivity although the appearance of significant preferential reinnervation was accelerated. 13 In the same femoral nerve injury model in the mouse, there is less preferential motor reinnervation than in rats, which was attributed to anatomical differences in the nerves.37,38 Preferential reinnervation and the efficacy of ES in promoting this reinnervation in mice appear to depend on the capacity of the axotomized motoneurons to express polysialic acid (PSA). Franz and colleagues demonstrated in a mouse femoral nerve injury model that, those axotomized motoneurons that do not express PSA, including the genitofemoral neurons, do not show preferential motor reinnervation while the femoral motoneurons that express PSA do. The ES effect of promoting preferential motor reinnervation was eliminated by removing PSA with endoneuraminidase-N from the regenerating axons 29 (but see Robinson and Madison 39 ). Findings that ES did not improve the accuracy of nerve regeneration after removing PSA in neural-cell adhesion molecule (N-CAM) knockout transgenic mice were equally convincing. 29 While there is preferential reinnervation of motor and sensory nerves to appropriate motor and sensory pathways, 25 regenerating motor nerves do not show preference for their original muscles in rats or in humans.33,34 This is also the case in mice irrespective of whether or not the injured nerve is subjected to ES. 40
ES was very effective in promoting the regeneration of sensory axons into the appropriate sensory nerve branch, a selection not normally made by the femoral sensory neurons.25,41 It is significant that, despite misdirection of regenerating axons to inappropriate muscle targets, the pattern of muscle activity during locomotion was surprisingly good. 42 A possible explanation lies in the improved sensory nerve specificity that may contribute to the neural plasticity underlying the appropriate activation of the muscles.
Molecular Basis: Roles of cAMP and Neurotrophic Factors
Electrical stimulation at 20 Hz for 1 hour of injured peripheral nerve elevates neuronal cyclic adenosine monophosphate (cAMP)43-45 and accelerates and increases the upregulation of neurotrophic factors and their receptors in neurons13,21 and Schwann cells.46-48 Thereafter, ES accelerates and increases the neuronal expression of cytoskeletal proteins, including actin and T-α1-tubulin, and growth-associated proteins that include GAP-43.24,49,50 The ES-mediated release of neurotrophic factors from Schwann cells, nerve growth factor in particular, suggests a possible role of the stimulated Schwann cells in accelerating axon outgrowth from the proximal nerve stump of injured neurons (Figure 1). Indeed, ES using direct current (DC) fields for 8 hours of Schwann cell cultures prior to culturing with neurons enhanced neurite outgrowth more than costimulation of both while nonstimulated cultures produced the least amount of outgrowth.51,52 Yet, ES was effective in accelerating axon regeneration in vivo after delayed nerve repair 53 when the expression of neurotrophic factors has declined to baseline levels in the chronically denervated Schwann cells,54,55 an argument against a necessary role of the Schwann cells. That the ES effect of promoting nerve regeneration after immediate femoral nerve transection and surgical repair was blocked with tetrodotoxin application proximal to the location of the stimulating electrodes on the proximal nerve stump 13 provides further evidence against the requirement of Schwann cells.
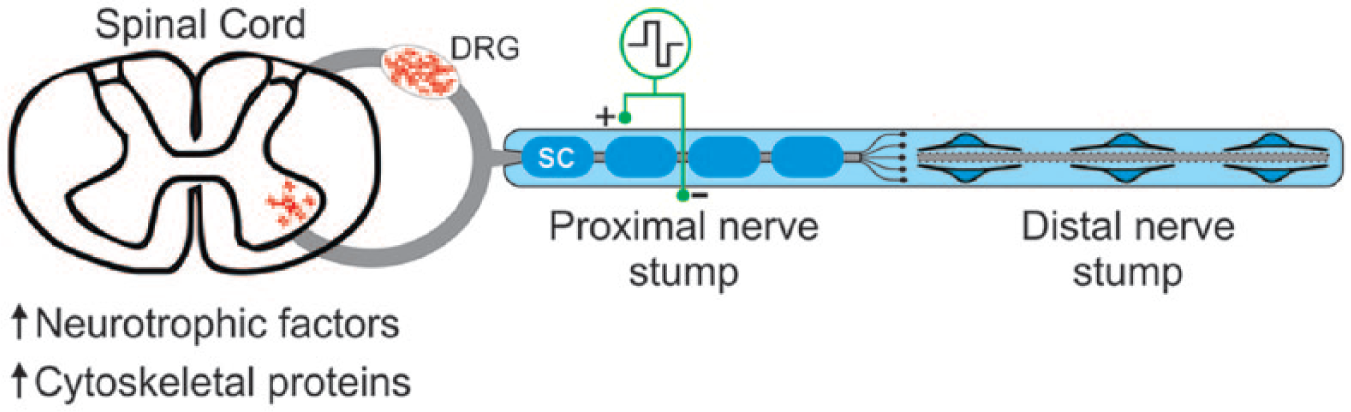
Diagrammatic illustration of the upregulation of neurotrophic factors, including brain-derived neurotrophic factor and neurotrophin 4/5 in both motoneurons in the ventral horn of the spinal cord and the sensory neurons within the dorsal root ganglia (DRG) after 1 hour of electrical stimulation of an injured and surgically repaired peripheral nerve. The upregulated cytoskeletal proteins that include actin and tubulin are important in the accelerated axon outgrowth. The regenerated axons enter into the distal nerve stump whose isolated axons undergo Wallerian degeneration. The degeneration of the latter axons is shown by the dotted lines, and the Schwann cells (SC) that myelinate the axons in the proximal nerve stump undergo transformation from myelinating to growth permissive in the distal nerve stump. These latter SCs lose their myelin and form elongated processes that guide regenerating axons.
The upregulated neurotrophic factors in axotomized neurons include brain-derived neurotrophic factor (BDNF) and neurotrophin-4/5 (NT-4/5) together with their receptor tropomyosin-related kinase B (trkB) receptors.28,50,56 The critical role of BDNF and NT-4/5 in the efficacy of ES in accelerating nerve regeneration after nerve injury and surgical repair was elucidated in experiments using transgenic mouse models in which the neurotrophic factors and/or their trkB receptors were knocked down or out.28,56 The upregulated NT4/5 in injured neurons is essential for the effect of the ES with BDNF also playing a critical role. When a functional blocking antibody against BDNF was administered during the first 3 days after nerve transection, repair, and ES with 20 Hz for 1 hour, the accelerating effects of ES were completely blocked. 57 Conversely, treatments of transected nerves with recombinant BDNF or small-molecule trkB agonists promote axon regeneration.56,58
Recently, Hoke and colleagues and Brushart and colleagues have demonstrated that the Schwann cells in motor and sensory nerve pathways differ in their expression of neurotrophic factors.54,55 Furthermore, the expression of different factors such as BDNF and GDNF within motor Schwann cells and nerve growth factor in sensory Schwann cells reach a peak within 15 days, after which they generally decline to baseline levels by 35 days after their denervation. Gordon, in a commentary on the Hoke and Brushart findings, noted that the attainment of peak expression levels of motor and sensory specific factors by 15 days coincides with the emergence of the preferential reinnervation of motor Schwann cells just distal to the site of surgical coaptation of transected femoral nerve stumps. 6 Furthermore, it was argued that ES-mediated acceleration of motor-specific neurotrophic factor expression in motoneurons as well as in Schwann cells could explain the earlier attainment of preferential motor reinnervation of appropriate motor distal pathways. 50
The expression of receptors for the neurotrophic factors is essential for the efficacy of the ES. The trkB receptors in motoneurons remain receptive to neurotrophic factors whereas they are rapidly downregulated in sensory neurons when ES exceeds 1 hour. 24
Clinical Applicability: The Translational Potential of Electrical Stimulation
Investigators at the University of Alberta recently carried out a randomized controlled clinical trial that assessed the postsurgical outcomes of acute brief low-frequency (20 Hz) ES. 59 The first of its kind to investigate the clinical applicability of the ES protocol on human subjects, the pilot study included patients with severe carpal tunnel syndrome where compression of their median nerve in the carpal tunnel caused marked loss of functional nerve–muscle contacts.59,60 The subjects underwent carpal tunnel release surgery and the randomly allocated stimulation group of patients underwent 1 hour of ES at 20 Hz at the surgical decompression site. 59 By recording electromyographic signals from the target thenar muscles over the course of one year, the findings indeed demonstrated that brief ES immediately following decompression surgery accelerated axon regeneration and resulted in early and complete reinnervation of the target thenar muscles 6 months later. Surgery alone did not yield any significant improvement even one year after the surgery. 59 Behavioral measures were used to assess the functional recovery outcomes while axonal regeneration and target muscle innervation were quantified using conventional sensory/motor nerve conduction studies and motor unit number estimation, respectively. 59 This study constituted a proof of principle study in that the normally innervated flexor muscles in the forearm flex the fingers with the recovery of the innervation of the intrinsic flexor muscles not contributing significantly to the functional recovery. A more recent study of Chan and colleagues demonstrated the efficacy of the same 20 Hz 1 hour ES paradigm in promoting earlier and enhanced recovery of sensory function after surgical repair of the transected digital nerve in patients. 61 In a randomized double blind study, patients with a complete transection of a pure sensory nerve in the hand (digital nerve) underwent nerve repair and either 1 hour of 20 Hz ES or sham stimulation postoperatively. Based on the small distance (2-6 cm) for nerve regeneration, patients were followed for 6 months postoperatively with examination of sensory function being performed on a monthly basis. These sensory tests included evaluating temperature, spatial discrimination, and pressure functions. Patients having 20 Hz ES applied to their injured and repaired nerves postoperatively for 1 hour, had significantly improved sensory function tests compared with sham stimulated patients with complete recovery to baseline levels being achieved by 5 to 6 months.
These findings demonstrate for the first time, that the favorable accelerated nerve regeneration and functional recovery facilitated by low-frequency ES seen in the animal model nerve injuries in rats13,24-26,60 and in mice28,62 can be applied to humans as well.
Importantly, patients did not report any complications in addition to pain due to the surgery or the stimulation, and the ES procedure was demonstrated to be feasible in a clinical setting.48-50 This supported previous work in a rat model investigating neuropathic pain following nerve injury and treatment with 1 hour of ES and treadmill exercise. Cobianchi and colleagues found that ES promoted pain responses due to regeneration of nociceptive fibers but also has a beneficial effect by reducing compensatory adjacent hyperalgesia. 63 Furthermore, the investigators demonstrated that, in fact, acute ES followed by high-intensity training was effective in avoiding the development of neuropathic pain and that both provided synergistic support for functional recovery.
These studies set the stage for future clinical trials of brief ES to promote axonal regeneration and target muscle reinnervation in nerve injuries. Conceivably, any nerve injury at any level could be amenable to ES, provided that the proximal nerve stump is accessible.
Exercise and Electrical Muscle Stimulation
Exercise: Treadmill Training
It has been shown that exercise can also affect an increase in neurotrophins such as BDNF and NT-4/5.64-66 The premise that ES is an artificially induced activity in axotomized neurons whereas exercise is natural led Asensio-Pinilla and colleagues to examine the outcome of combining different patterns of treadmill training with acute ES at 20 Hz for 1 hour in a rat sciatic nerve transection injury model. 27 The experiment elucidated a synergistic effect when ES was combined with moderate-intensity active exercise but only during the early phase of nerve regeneration, which supports previous literature that both treatments share common mechanisms of action.27,67
Over the course of evaluating the extent of activity-mediated axonal regeneration, English and colleagues discovered a sex difference in the effectiveness of treadmill training when comparing 2 treadmill training paradigms: continuous and interval training.68,69 A daily slow training protocol at 10 m/min for 1 hour resulted in marked increase in length of regenerating axons in male mice 2 weeks following nerve transection and repair but the protocol had no effect on the regeneration in female mice.68,69 On the other hand, when the female mice were exposed to a faster 20 m/min training protocol at 2-minute intervals 4 times daily, an impressive enhancement in axonal regeneration was seen but no enhancement was appreciated in the male mice exposed to the same interval training protocol.68,69 Multiple studies postulate that sex steroid hormones, particularly testosterone, are likely to mediate this sex difference in exercised mice. Treating unexercised female mice with an aromatase inhibitor to block the conversion of testosterone or its precursors into estradiol, enhanced axonal regeneration.68,70 Subsequent experiments, demonstrated that treating mice with flutamide, an androgen receptor blocker, inhibits the effect of both exercise and ES in both sexes. 68 As treadmill training can be readily applied to human subjects, the translational potential of this modality in combination with ES in peripheral nerve injuries is promising, while activity-dependent therapies also simultaneously empower patients to assume responsibility for their own recovery.
Electrical Stimulation of Denervated Muscle(s)
Nerve injuries that disconnect axons from the cell body result in denervation of the muscle that the axons innervate. These denervated muscles undergo denervation atrophy that proceeds rapidly.1,71 While treadmill training may provide a systemic affect to enhance nerve regeneration, the denervated muscle will still remain atrophic. Once reinnervated, recovery to full function may take a considerable amount of time. Recently, a protocol of daily 1 hour ES of denervated rat gastrocnemius muscle has been used prior to and during the reinnervation period. 15 Results showed early nerve regeneration and muscle reinnervation, as characterized by motor unit analysis, was enhanced in stimulated muscles. 72 Furthermore, stimulation during the period of reinnervation had no negative impact on sprouting or reinnervation. Functional recovery was also shown to be enhanced with the rats undergoing daily stimulation of their muscles performing significantly better at skilled locomotion tasks than those that did not receive electrical muscle stimulation. 15 Electrical currents used to stimulate denervated muscles are much greater than innervated muscles due to the lower excitability of muscle fibers following denervation. There is evidence that activity-dependent treatments can upregulate intramuscular neurotrophic factors, 73 but unpublished data indicate that ES of muscle does not upregulate these factors in the distal nerve stump (Willand, unpublished data). This effect requires further exploration and the mechanism of this effect remains to be elucidated.
Transvertebral Electrical Stimulation of Spinal Cord
Electrical stimulation has not been constrained to being applied only to proximal nerve stumps to elicit the positive regenerative effect. Recently, ES of the cervicothoracic spinal cord to activate lumbar motoneurons was investigated following transection and primary repair of the femoral nerve in a mouse model. 74 The results demonstrated that the stimulated group led to a significantly higher expression of PSA-NCAM as well as better specificity of motor reinnervation in comparison to no treatment. 74 However, a reduction in the number of retrogradely labeled neurons was found 2 weeks post-repair, which did not support previous proximal nerve stimulation data, and the reason for this discrepancy is not clear. 74 Nonetheless, the study suggests a potential advantage of spinal cord ES over nerve stimulation as a more natural way of activating motoneurons via intrinsic spinal circuitry and further exploration of central nervous system–directed approaches would be beneficial.
Conclusions
Peripheral nerve injuries can have profound functional and psychological effects on patients. Understanding of the biological basis of nerve regeneration at both cellular and molecular levels as well as many therapeutic targets, we are at the turning point of an exciting era to transform the field of peripheral nerve injuries. The current literature indicates the imminent clinical applicability of acute brief low-frequency ES after surgical repair to effectively promote axonal regeneration as it has yielded promising evidence to maximize functional recovery in diverse types of peripheral nerve injuries. We anticipate that this feasible modality will continue to be refined with further advancements to provide more individualized treatment with time.
Footnotes
Authors’ Note
Michael P. Willand and May-Anh Nguyen are co–first authors. Gregory H. Borschel and Tessa Gordon are co–senior authors.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
