Abstract
Background. In rat models of spinal cord injury, at least 3 different strategies can be used to promote long-term cortical reorganization: (1) active exercise above the level of the lesion; (2) passive exercise below the level of the lesion; and (3) serotonergic pharmacotherapy. Whether and how these potential therapeutic strategies—and their underlying mechanisms of action—interact remains unknown. Methods. In spinally transected adult rats, we compared the effects of active exercise above the level of the lesion (treadmill), passive exercise below the level of the lesion (bike), serotonergic pharmacotherapy (quipazine), and combinations of the above therapies (bike+quipazine, treadmill+quipazine, bike+treadmill+quipazine) on long-term cortical reorganization (9 weeks after the spinal transection). Cortical reorganization was measured as the percentage of cells recorded in the deafferented hindlimb cortex that responded to tactile stimulation of the contralateral forelimb. Results. Bike and quipazine are “competing” therapies for cortical reorganization, in the sense that quipazine limits the cortical reorganization induced by bike, whereas treadmill and quipazine are “collaborative” therapies, in the sense that the reorganization induced by quipazine combined with treadmill is greater than the reorganization induced by either quipazine or treadmill. Conclusions. These results uncover the interactive effects between active/passive exercise and serotonergic pharmacotherapy on cortical reorganization after spinal cord injury, emphasizing the importance of understanding the effects of therapeutic strategies in spinal cord injury (and in other forms of deafferentation) from an integrated system-level approach.
Keywords
Introduction
The somatotopic organization of the primary somatosensory cortex is not a static map unmodifiable after development but rather a variable map that is continuously updated based on the dynamics of ongoing levels of cortical activity1,2: if a specific part of the body is used relatively more, its corresponding cortical area expands, if another part of the body is used relatively less, its corresponding cortical area shrinks. Although this physiological process of cortical reorganization continuously takes place during sensorimotor learning,3,4 its extreme consequences can be observed in pathological conditions that lead to cortical deafferentation such as stroke, amputation, nerve injuries, and spinal cord injuries: when a cortical area is deafferented, the dynamical equilibrium of the somatotopic map is disrupted, often leading to massive cortical reorganization.5-11 From a clinical perspective, the degree of cortical reorganization after somatosensory deafferentation can be critical in the tradeoff between functional recovery12-15 and the appearance of disabling symptoms such as neuropathic pain.16-20 It is, therefore, important to gain a deep understanding of the mechanisms underlying cortical reorganization and develop optimized clinical strategies to modulate it. 21
We previously showed that in rat models of complete spinal cord transection at least 3 different strategies can be used to promote long-term cortical reorganization: (1) active exercise above the level of the lesion, likely through activity-dependent plasticity 22 ; (2) passive exercise delivered below the level of the lesion that upregulates proteins in the cortex associated with plasticity 23 ; (3) serotonergic pharmacotherapy, likely through the direct action of serotonin on sensorimotor systems. 24 From a mechanistic perspective, these findings expose the complexity in the regulation of the dynamical equilibrium underlying cortical reorganization after deafferentation. From a translational perspective, they provide the rationale for developing and optimizing both nonpharmacological (active and passive exercise) and pharmacological (serotonin) therapeutic strategies to promote or control cortical reorganization. Whether and how these potential therapeutic strategies—and their underlying mechanisms of action—interact remains unknown.
To address this issue, in the present study, we compared the effects of active exercise above the level of the lesion (treadmill), passive exercise below the level of the lesion (bike), serotonergic pharmacotherapy (quipazine), and combinations of the above therapies (bike+quipazine, treadmill+quipazine, bike+treadmill+quipazine) on long-term cortical reorganization after thoracic spinal cord transection. Cortical reorganization was measured as the percentage of cells recorded in the deafferented hindlimb cortex that responded to tactile stimulation of the contralateral forelimb.
Methods
A total of 70 adult Sprague-Dawley rats were used in this study (initially 10 per group); 23 animals did not survive the entire study, and the remaining 47 were from the following 7 experimental groups (Figure 1). The first 3 groups were animals that were spinally transected and received individual therapies after the transection: (1) treadmill animals (n = 8), which received active exercise through treadmill locomotion; (2) bike animals (n = 5), which received passive exercise through hindlimb bike training; and (3) quipazine animals (n = 6), which received serotonergic pharmacotherapy. The following 3 groups were animals that were spinally transected and received combinations of therapies after the transection: (4) treadmill+quipazine animals (n = 9); (5) bike+quipazine animals (n = 7); and (6) bike+quipazine+treadmill animals (n = 8). The last group (7) was a control group composed of sham animals (n = 4) that were spinally transected and received sham therapies. Each animal group received its therapy (or sham) for 8 weeks after transection. Within 1 week after the last therapy session (ie, week 9 after the spinal transection), animals were anesthetized to perform an acute, single-neuron mapping study in the deafferented hindlimb cortex to electrophysiologically quantify cortical reorganization, similarly to our previous studies.22-24 The data for group 2 (bike) were previously published as part of another study, 23 but the data were collected at the same time as the other data from the other groups. Animals were entered into the study at a rate of 2 per week, transected, and assigned to 1 of the 7 groups in a serial fashion (animal 1 to group 1, animal 2 to group 2, . . . , animal 8 to group 1, etc). All procedures were performed under the guidelines of the National Institutes of Health and approved by the Institutional Animal Care and Use Committee of Drexel University.
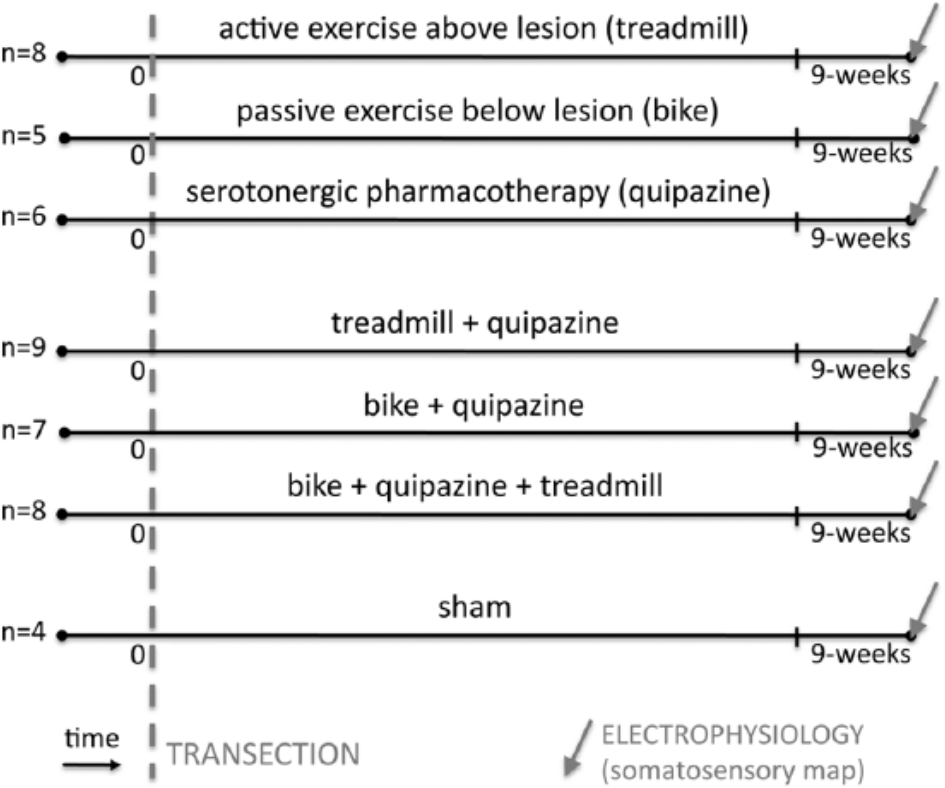
Experimental protocol: We used 70 adult rats, of which 47 survived the entire study; they were divided into 7 experimental groups: (1) treadmill animals (n = 8), which received active exercise through treadmill locomotion; (2) bike animals (n = 5), which received passive exercise through hindlimb bike training; (3) quipazine animals (n = 6), which received serotonergic pharmacotherapy; (4) treadmill+quipazine animals (n = 9); (5) bike+quipazine animals (n = 7); (6) bike+quipazine+treadmill animals (n = 8); and (7) sham animals (n = 4), which were spinally transected and received sham therapies. Each animal group received its therapy (or sham) for 8 weeks after transection. Within 1 week of the last therapy session (ie, 9 weeks after the spinal transection), animals were anesthetized to perform an acute, single-neuron mapping study in the deafferented hindlimb cortex to electrophysiologically quantify cortical reorganization,
Spinal Cord Transection
Animals received a complete thoracic transection of the spinal cord with procedures that are similar to those in our previous studies.23-25 Adult, female Sprague-Dawley (Charles River) rats were anesthetized with isoflurane (2%-3% with oxygen), and the spinal cord was exposed by laminectomy at the T8/T9 level. The cord was transected with iridectomy scissors followed by aspiration of tissue within the cavity. A collagen matrix, Vitrogen, was injected into the site of the transection to fill the cavity. The muscle and skin were sutured in layers with 5-0 silk. Animals were then warmed, and when they became active, returned to their home cages. Bladders were manually expressed until the animals were able to void on their own. Animals were housed under a 12-hour light/dark cycle (lights on at 07:00 hours) with free access to food and water. All behaviors were performed in the light phase of the rats’ light/dark cycle.
Active Exercise Above Lesion: Treadmill Locomotion
Treadmill therapy was chosen to be consistent with our studies in adult rats spinalized as neonates and consisted of placing the animals on a motorized treadmill apparatus for 3 min/d at a speed of 6.5 m/min, 5 d/wk.22,26 This time limit on the exercise was chosen because it is the limit of what the animals are capable of performing—after 4 minutes they become tired and stop trying to locomote. No weight support or other stimulation was applied to the rats during treadmill locomotion, and the rats used their forelimb to move along the treadmill dragging their hindlimbs. Treadmill exercise began 1 week after transection and was delivered for 8 weeks.
Passive Exercise Below Lesion: Hindlimb Bike
Hindlimb bike exercise 27 consisted of two 30-minute sessions with a 10-minute break, 3 d/wk (Monday, Wednesday, and Friday).23,24 This exercise regimen involved suspending the rats on a sling with the hindlimbs hanging down and the hind feet strapped onto the pedals of a bicycle-type device that was driven by a motorized belt. The exercise consisted of a pedaling motion that flexed one limb while extending the other without overstretching the limbs. Cycling speed was 0.5 Hz. This was, therefore, a passive exercise of the hindlimbs only. Passive hindlimb bike exercise or sham bike exercise started the week after the spinal transection and was delivered for 8 weeks.
Serotonergic Pharmacotherapy: Quipazine
Daily quipazine injections (0.075 mg/kg, IP, 5 d/wk) were delivered to the animals starting 2 weeks after transection. Quipazine was chosen because it is a nonspecific 5-HT agonist (ie, it acts at both the 2A and 1 5-HT receptors) that has been shown to induce locomotor-like behaviors in the hindlimbs of spinal injured rats.28,29 The 2-week lag postinjury allowed time for 5-HT receptor upregulation in the spinal cord caudal to the lesion. 30 The dose was selected to be sufficient to elicit hindlimb locomotor-like movement but minimize tremor and/or spasticity side effects known to occur in spinal injured animals after chronic administration of 5-HT agonists (data not shown).
Combined Therapies
Combined therapies were provided in the same way as individual therapies. In animals that received bike and quipazine, quipazine was always delivered 5 minutes before bike. In animals that received quipazine and treadmill, quipazine was always delivered 5 minutes before treadmill. All animals were given an injection first (either quipazine or saline). The peak effect of quipazine in the central nervous system occurs about 60 minutes after injection, 31 so it was active throughout the therapy.
Sham Therapy
Sham therapies were provided to maintain similar experiences across animals and ensure that differences were related to the therapy and not handling or somatosensory contact with the experimental devices. Sham treadmill exercise consisted of placing the animal on the treadmill for 3 min/d, but the treadmill did not move. Sham bike exercise consisted of placing the animals on the bike for 70 minutes, 3 times per week, but the pedals did not move. Sham drug therapy was provided by daily injection of saline. Sham animals received all 3 sham therapies.
Behavioral Testing
Basso, Beattie, Bresnahan (BBB) scoring in the open field was used to test hindlimb behavioral recovery32,33 during week 8. Animals that received drug therapy were tested after a 3-day washout period during which no drug was given. Spontaneous hindlimb motor activity was evaluated for 4 minutes in a 762 mm x 915 mm diameter enclosure and scored by 2 trained observers with an interrater reliability ≥95%. BBB scores of 8 or below describe various degrees of behavioral recovery of locomotor-like movements that do not include weight support. BBB scores of 9 or above (to a maximum of 21) indicate some degree of hindquarter weight support starting in stance and progressing to weight-supported stepping. 32
Electrophysiology
Acute single-neuron mapping of the deafferented hindpaw cortex was performed at the end of the study, with similar techniques as in our previous studies.22-24 Rats were anesthetized by intraperitoneal injection of urethane anesthesia (1.3 g/kg) and placed in a stereotaxic frame. Craniotomies were performed over either the right or left cortex to expose the hindlimb representations in the primary somatosensory cortex. The stereotaxic coordinates for hindlimb craniotomy were from 0 to 3 mm posterior to bregma and from 2 to 3 mm lateral. 34 Electrode penetrations were defined using the stereotaxic coordinates for the hindlimb somatosensory cortex. 35 For all animals, the anesthesia level was maintained at stage III-4.36,37
A tapered, high-impedance (10 MΩ) tungsten microelectrode (FHC, Inc, Bowdoin, ME, part no UEWSGGSE0N1E) was mounted on a stereotaxic electrode manipulator. A ground wire was inserted into the brain adjacent to the craniotomies. The microelectrode was then moved to the anterior-posterior and medial-lateral coordinates that defined a predetermined location above the hindlimb somatosensory cortex and lowered, perpendicular to the surface of the brain, to penetrate the dura and pia. The microelectrode was then slowly inserted into the brain. Signals were amplified (10-15 k), bandpass filtered (154 Hz to 13 kHz), and digitalized (40 kHz) using a Multi-Neuron Acquisition Processor (Plexon Inc, Dallas, TX).
The signals from the microelectrode were continuously monitored on the oscilloscope and audio speakers as the electrode was lowered. When a neuron was encountered, the dorsal/ventral coordinates of the cell were noted. Two experimenters then determined whether the identified cell responded to sensory stimulation. The first experimenter, with knowledge of the electrode placement, used wooden probes to touch the hair/skin on the forelimb and shoulder. The second experimenter, blind to the position of the electrode and treatment group of the animal, determined if the cell responded to the stimulus, predominantly by listening for a change in firing rate. If the cell did not modulate its firing rate in response to the stimulation, the cell was noted as negative. If the cell did modulate its firing rate, the cell was noted as positive. If the cell was noted as positive, then the receptive field of the cell was identified by tapping locations on the body rostral to the level of the injury. Stimulation of any body surface that modulated the cell’s firing rate was considered part of the cell’s receptive field. To ensure that tapping forces between animals and across sites were uniform, the responses elicited by the wooden probe were periodically compared with responses elicited by von Frey filaments to calibrate the stimulus applied by the wooden probe. The stimulation consisted of pressing a filament gently against the skin, perpendicular to its surface until the filament bent 90°. This procedure was done 5 times for each filament and skin site to ensure reproducibility of the results.
After a cell was characterized, the microelectrode was moved at least 50 µm deeper (with respect to the cortical surface) before another cell could be identified in the same penetration to ensure that a new cell was encountered. Every identified cell was assigned to 1 of 3 cortical layers based on the stereotaxical depth of the microelectrode at the time the cell was recorded: supragranular, 50 to 700 µm (layer II/III); granular, 750 to 1000 µm (layer IV); or infragranular, 1050 to 2000 µm (layer V/VI).22-24 To minimize tissue damage and its possible effects on cell responsiveness during later penetrations, no more than 6 penetrations were performed per animal.
Perfusion and Histological Processing of the Spinal Cord
At the end of the mapping sessions, the rats were perfused transcardially with buffered saline, followed by buffered 2% paraformaldehyde, and then by buffered 2% paraformaldehyde containing 10% sucrose. Spinal cords were removed and placed in phosphate buffer containing 30% sucrose for 72 hours. Specimens were frozen in optimum cutting temperature (OCT) and sectioned on a freezing microtome at 20 µm. The transection segments of the spinal cords were sectioned parasagitally, and alternate sections were Nissl-myelin stained. The resulting sections were examined under a microscope to confirm completeness of the transection.
Statistical Analyses
To assess the effects of individual therapies and combined therapies after spinal transection on the electrophysiological reorganization of the deafferented cortex, we performed several types of analyses. First, the number of recorded cells per track, per animal were entered into separate 1-way or 2-way ANOVAs for individual therapies or for combined therapies. Second, the proportion of cells recorded in the deafferented hindlimb cortex responding to forelimb stimuli was analyzed by entering the raw binary neural data (responding/nonresponding neurons, sample size is number of cells) into generalized linear models (GZLM) with binomial distribution and logit link function. GZLMs allow binary data to be rigorously analyzed with ANOVA-like designs. Post hoc comparisons were performed with Tukey Honest Significant Difference Test or less conservative 2-proportion tests. Third, the percentage of cells per track stereotaxically located in the deafferented hindlimb cortex that responded to stimulation of the intact forelimb were entered into independent-measures ANOVAs, considering each track and layer as independent samples. Tukey’s test was used for post hoc comparisons. Using similar statistical designs, we verified that between-group differences in percentage of responsive cells were not a result of between-group biases in recording locations (anteroposterior and medial-lateral coordinates were analyzed separately) or number of tracks per animal (in this case, we used nonparametric statistics). All results were considered significant at P < .05.
Results
Each animal group received its therapy (or sham) for 8 weeks after complete thoracic transection of the spinal cord. Within 1 week of the past therapy session (ie, during week 9 after the spinal transection), animals were anesthetized to perform an acute, single-neuron mapping study in the deafferented hindlimb cortex (2-6 tracks per animal). A total of 2697 single neurons from the supragranular, granular, and infragranular layers were isolated and identified as either responsive or not responsive to cutaneous stimulation of the forelimbs (Table 1). Animals were behaviorally evaluated with BBB scores at the end of therapy (week 8).
Electrophysiological Data and BBB Scores.
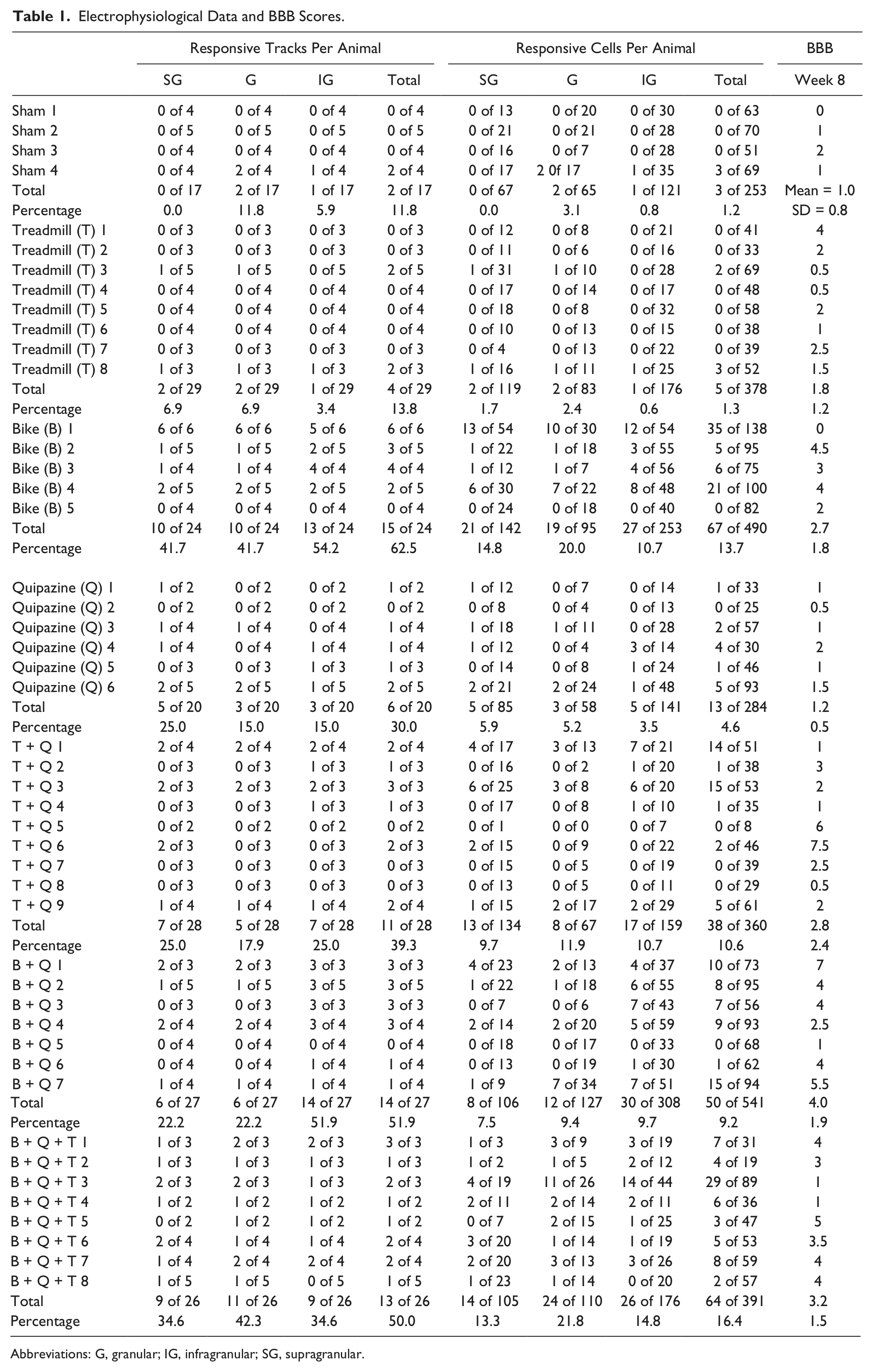
Abbreviations: G, granular; IG, infragranular; SG, supragranular.
Individual Therapies
We first compared the ability of individual therapies—active exercise above the level of the lesion (treadmill), passive exercise below the level of the lesion (bike), or serotonergic pharmacotherapy (quipazine)—to induce neurophysiological reorganization of the deafferented hindpaw cortex after complete thoracic transection of the spinal cord with sham therapy (sham) in transected animals. Even though the scope of the present work was not to optimize individual therapies, these initial comparisons represent an important basis to understand the interactions between therapies, which will be assessed in the next section.
The average number of cells recorded per track, per animal was significantly higher in the bike group (20.3 ± 1.7) compared with other individual therapies (1-way ANOVA, factor therapy [sham, treadmill, bike, quipazine], F(3, 19) = 7.9, P = .0013; Tukey, bike vs quipazine, 14.1 ± 3.8, P = .0090; bike vs treadmill, 13.1 ± 2.4, P = .0025; bike vs sham, 14.9 ± 2.0, P = .0506). The percentage of cells stereotaxically located in the deafferented hindlimb cortex that responded to tactile stimulation of the contralateral forelimb was significantly higher in the bike group (13.7% of 490 cells) compared with other individual therapies—2-way GZLM, factor therapy, Wald χ2(3) = 37.6, P < .0001; Tukey P < .0001; Figure 2A. The difference between bike and other individual therapies was not layer specific (interaction Therapy × Layer: Wald χ2(6) = 1.0; P = .98). A less-conservative post hoc analysis revealed that compared with the low percentage of responding cells in sham animals (1.2% of 253 cells), quipazine induced only a small increase (4.6% of 284 cells; 2-proportion test: P = .0080), whereas treadmill did not have any effect (1.3% of 378 cells; P = .73). Essentially the same results were obtained considering the percentage of responding cells in each track as independent samples—2-way ANOVA, factor therapy: F(3, 258) = 16.9, P < .0001; interaction Therapy × Layer: F(6, 258) = 0.55, P = .76. These results were not a result of possible between-group biases in the recording locations—group average ranges: 1.2 to 1.4 mm posterior, factor therapy, F(3, 86) = 0.7, P = .55; 2.5 to 2.6 mm lateral, F(3, 86) = 1.6, P = .19. From a behavioral perspective, no individual therapy was able to significantly improve the BBB scores of the animals (1-way ANOVA, factor therapy: F(3, 20) = 1.19, P = .40; Table 1).
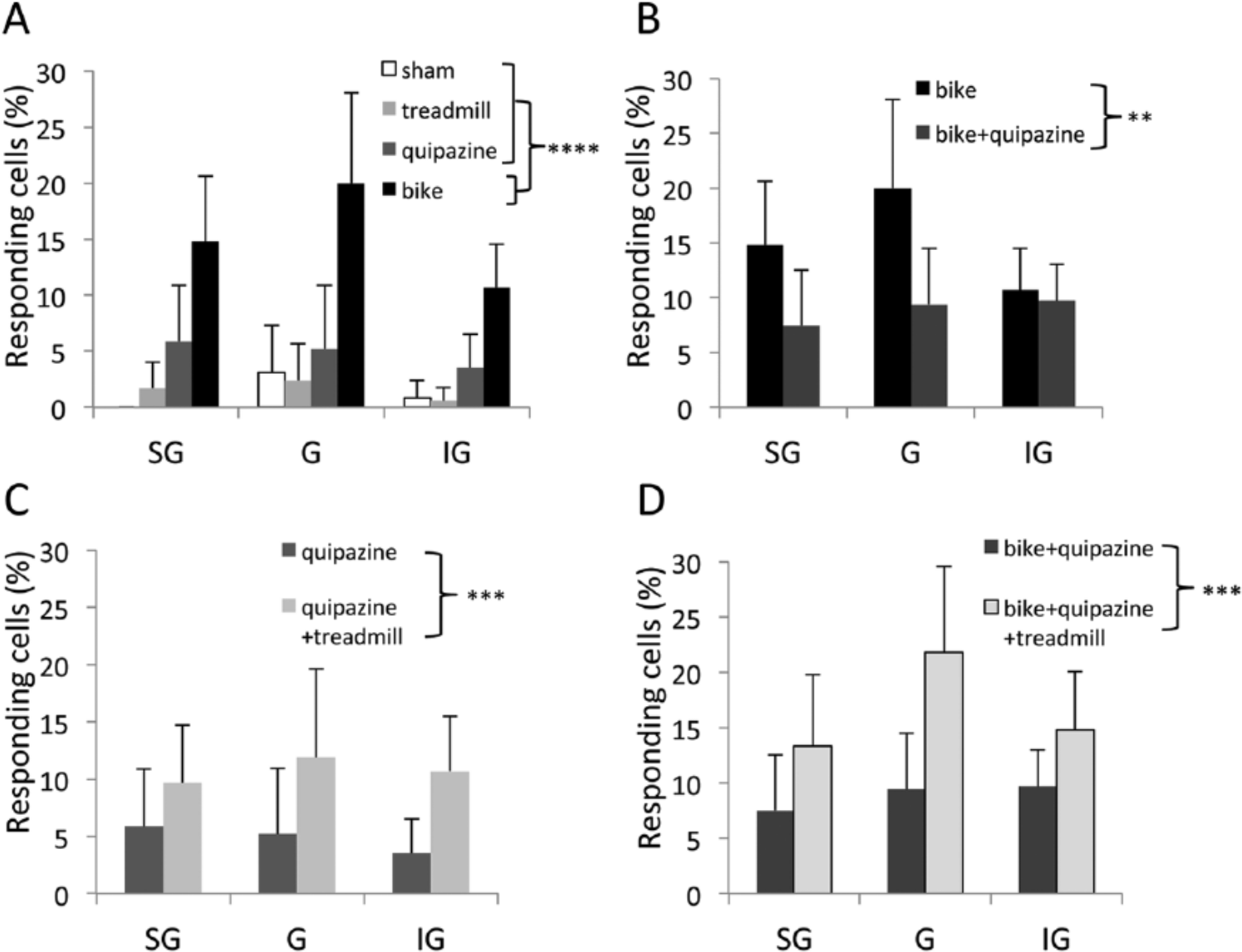
Neurophysiological reorganization of the somatosensory cortex. (A-D) Percentage of responding cells in the different animal groups, separated by cortical layer: supragranular (SG), granular (G), and infragranular (IG). (A) Individual therapies. (B-D) Combined therapies. Error bars indicate 95% confidence intervals. **P < .01; ***P < .001; ****P < .0001.
Combined Therapies
We then investigated how the above therapies interact when combined (ie, when 2 or more different therapies were provided to the same animal). When bike was combined with quipazine (bike+quipazine), the average number of cells recorded per track (20.3 ± 3.5), per animal did not change compared to bike alone: F(1, 10) = 0.01; P = .97. Surprisingly, however, the percentage of responding cells significantly decreased with bike+quipazine compared with bike alone (from 13.7% of 490 cells, reported above, to 9.2% of 541 cells; 2-way GZLM, factor therapy [bike, bike+quipazine], Wald χ2(1) = 7.0, P = .0081; Figure 2B), without a clear layer specificity (Therapy × Layer: Wald χ2(2) = 3.2, P = .20). This result was confirmed when considering the percentage of responding cells in each track as independent samples (2-way ANOVA, factor therapy: F(1, 147) = 9.0, P = .0032; Therapy × Layer: F(2, 147) = 1.2, P = .30). These results were not a result of possible between-group biases in the recording locations—group averages: 1.0-1.2 mm posterior, t(49) = 0.96, P = .34; 2.6-2.6 mm lateral, t(49) = 0.59, P = .55). BBB scores did not change in animals that received bike+quipazine compared with animals that received bike alone (Table 1; t test: P = .26). These results suggest that bike and quipazine are competing therapies for cortical reorganization, in the sense that quipazine interferes with the cortical reorganization induced by bike.
Even though treadmill alone did not induce a measurable increase in cortical responsiveness in our experiments (see above), treadmill combined with quipazine (treadmill+quipazine) produced a higher percentage of responding cells compared with quipazine alone (from 4.6% of 284 cells, reported above, to 10.6% of 360; 2-proportion test: P = .0044). This positive effect of treadmill was also evident in the combination of the 3 therapies (bike+quipazine+treadmill), which again increased the proportion of responding cells compared with bike+quipazine (from 9.2% of 541 cells, reported above, to 16.4% of 391 cells; P = .0012). This result was confirmed by more rigorous statistical analysis on the percentage of responding cells—3-way GZLM, factor treadmill (yes or no), Wald χ2(1) = 14.8, P = .0001; interactions with Layer and Therapy, Wald χ2 < 0.98, P > .61 (Figures 2C and 2D)—as well as when considering the percentage of responding cells in each track as independent samples—3-way ANOVA, factor treadmill (yes or no): F(1, 97) = 4.1, P = .0451; interaction Treadmill × Therapy (quipazine, bike+quipazine): P = .63. The effect of treadmill did not depend on cortical layer (interaction Treadmill × Layer: P = .77; interaction Treadmill × Therapy × Layer: P = .29). These results were not a result of possible between-group biases in the recording locations—group average ranges, 1.0 to 1.4 mm posterior: factor Therapy, F(1, 97) = 1.6, P = .21; factor Treadmill, F(1, 97) = 3.8, P = .054; interaction Treadmill × Therapy, F(1, 97) = 0.11, P = .73; 2.5 to 2.6 mm lateral, P > .19. Note that there was a tendency for recording locations to be slightly more medial in animals that received treadmill therapy as a combined therapy compared with animals that did not. Because more medial implies further away from the forelimb cortex, this tendency is conservative for our results. From a behavioral perspective, BBB scores tended to increase in animals that received quipazine+treadmill compared with animals that received quipazine or treadmill alone (Table 1; t test: P = .0732). BBB scores did not change in animals that receive bike+quipazine+treadmill compared with animals that received bike+quipazine (Table 1; t test: P = .37).
These results suggests that treadmill and quipazine were collaborative therapies for cortical reorganization, in the sense that the percentage of responding cells obtained with treadmill+quipazine (with or without bike) was greater than the percentage of responding cells obtained with either treadmill alone or quipazine alone (with or without bike).
Discussion
The main result of the present work is that exercise and serotonergic pharmacotherapy interact in their effects on cortical reorganization after spinal cord injury: passive exercise below the level of the lesion (bike) and serotonergic pharmacotherapy (quipazine) are competing therapies (ie, quipazine limits the cortical reorganization induced by bike), whereas active exercise above the level of the lesion (treadmill) and serotonergic pharmacotherapy (quipazine) are collaborative therapies (ie, the reorganization induced by quipazine combined with treadmill is greater than the reorganization induced by either quipazine or treadmill). From a mechanistic perspective, these findings expose the complex interactions between different pathways to cortical reorganization after massive deafferentation. From a translational perspective, they emphasize the importance of understanding the neuroplasticity effects of therapeutic strategies in spinal cord injury (or in other forms of deafferentation) from an integrated system-level approach.
Methodological Considerations
The main measure we used to assess cortical reorganization after complete spinal cord transection is the percentage of neurons recorded in the deafferented hindlimb cortex that responded to light tactile stimulation to body areas above the level of the lesion. This binary measure, which consists of empirically categorizing each recorded neuron as either responder or nonresponder is particularly suitable for our purpose because of the following reasons: (1) in our experimental conditions, the great majority of recorded neurons are nonresponders (ie, their response magnitude, if quantified, would be approximately zero at all locations); (2) from a biological perspective, the transition from nonresponder to responder is particularly relevant and is often used as electrophysiological evidence of cortical reorganization after deafferentation22-24; (3) from an experimental perspective, the binary categorization allows a high number of neurons to be sampled per rat, maximizing statistical power. It is important to note that the rigor of the empirical measurement was guaranteed by having 2 neurophysiologists always involved in the experiments, with the one responsible for the binary categorization being blinded to both the location of stimulation and the animal group.
Several points should be mentioned to raise caution about the possible translational value of our findings. First, our experiments were not designed to perform analyses with sample size is number of animals, so any analysis/inference using sample size is number of animals is not reliable. Nonetheless, at a qualitative level, it is interesting to note that some animals appeared to be reorganizers, with other animals being nonreorganizers. This observation will deserve further investigation. Second, all behaviors were performed in the light phase of the rats’ light/dark cycle—that is, during the rat’s inactive phase (note that this is common practice in the field). Third, our behavioral data (BBB scores) are reported for completeness; it is premature to imply any causal relationship between reorganization of the somatosensory cortex and possible functional recovery in this complete transection model. Cortical reorganization could be either adaptive—that is, contributing to functional recovery—or maladaptive—that is, contributing to neuropathic pain. 38 In fact, allodynia was recently reported in the same rat model of spinal cord transaction, 39 which encourages further work to clarify the behavioral impact of therapies and the role of cortical reorganization in this model.
Individual Therapies
We previously showed that passive bike exercise of the hind limbs after complete thoracic transection of the spinal cord promotes reorganization of the deafferented hind limb cortex, at least in part related to the ability of bike exercise to increase cortical levels of adenylate cyclase (ADCY1) and brain-derived neurotrophic factor (BDNF). 23 Increased level of BDNF could explain the intriguing present finding of higher number of recorded cells per track when animals received bike compared with other individual therapies.
Passive bike exercise was the therapy that induced the greatest cortical reorganization when provided alone, but this greater impact is not conclusive, because we did not fully optimize each individual therapy, which is beyond the scope of the present work. Furthermore, we specifically tested cortical reorganization using light tactile stimuli that reach the brain through the dorsal column pathway. Different reorganization profiles might be observed using stimuli of higher intensity that maximize dorsal column inputs and also activate the spinothalamic tract.10,11,40-43 Nonetheless, because passive exercise of the hind limbs is a common rehabilitation practice in patients with spinal cord injury,44-50 spinal transection with passive bike exercise in rats provides a clinically relevant model of cortical reorganization after spinal cord injury. 24
We previously showed that serotonergic pharmacotherapy (quipazine combined with 8-OH-DPAT) promotes cortical reorganization after complete spinal cord transaction. 24 Here we showed that quipazine alone induces a small yet significant reorganization of the deafferented hindlimb cortex. Quipazine is well known to improve functional recovery after spinal cord injury, which has been documented in cats,51,52 rats,28,29 and mice.53-56 The suggested mechanism mediating this functional improvement was the excitation of neurons in the central pattern generator below the lesion that have been deprived of their normal descending 5-HT input from the raphe nucleus. However, this mechanism is unlikely to explain our increase in cortical reorganization, which instead could be related to the ability of 5-HT to promote cortical plasticity after sensory deafferentation, as previously described both in the visual cortex57,58 and in the barrel cortex. 59 Therefore, the results of the present study and of our previous work 24 collectively suggest a supraspinal action of serotonergic therapy that supports cortical reorganization after spinal cord injury.
We previously showed that treadmill therapy promotes cortical reorganization and functional recovery in adult rats spinalized as neonates. 22 In the present study, the same treadmill therapy alone was not sufficient to promote cortical reorganization or to induce functional recovery in adult rats spinalized as adults, which identifies an important comparative difference between spinal cord injury models. Even though no individual therapy was able to induce significant functional recovery as measured by BBB scores, we cannot exclude the fact that more subtle improvements might be detected with finer behavioral measures.
Combined Therapies
The treadmill therapy used here is in line with previous strategies of increasing the activity of the intact cortex to maximize cortical reorganization by actively exercising the nonaffected body or the residual functions of the affected body, investigated both in animals models26,60,61 and in patients with spinal cord injury.12,62-64 Even though treadmill therapy alone was not sufficient to promote cortical reorganization or to induce functional recovery in adult spinalized rats, it did promote cortical reorganization and tended to induce functional recovery when combined with quipazine compared with quipazine alone. This collaborative interaction of treadmill and quipazine could have at least 2 possible nonexclusive explanations: (1) behavioral collaboration—that is, because quipazine was always given before the treadmill session in our experiments, the acute effects of quipazine might have helped the rat perform a more efficient treadmill exercise; (2) mechanistic collaboration—that is, treadmill exercise might ultimately act on the same pathway to cortical reorganization as quipazine. Interestingly, the cortical reorganization induced by quipazine combined with treadmill was actually greater than the sum of the reorganization induced by quipazine plus the reorganization induced by treadmill. Even though it is tempting to speculate a synergic interaction between quipazine and treadmill, this synergy could also be explained by a nonlinearity in the dose response of individual therapies (eg, a threshold effect).
Quipazine combined with passive bike exercise decreased cortical reorganization compared with bike alone. This result is in contrast with our previous study, in which quipazine+8-OH-DPAT combined with bike increased cortical reorganization compared with bike alone. 24 The mechanisms underlying these complex interactions between serotonergic pharmacotherapy and passive exercise remain unclear, but differences among specific 5-HT receptors seem to be critical. At the molecular level, an intriguing possibility is that quipazine (but not quipazine+8-OH-DPAT) might interfere with the upregulation of BDNF and/or ADCY1 induced at the cortical level by passive bike exercise. 23 Future investigations on the effects of 8-OH-DPAT alone (and of 8-OH-DPAT+bike) on cortical reorganization after spinal cord injury will be necessary to obtain a complete mechanistic picture.
Overall, the present results have several important translational implications: (1) different therapies that individually promote cortical reorganization do not necessarily benefit from each other when combined (eg, bike and quipazine); (2) the same serotonergic therapy (eg, quipazine) can either promote brain plasticity or limit brain plasticity depending on the model of sensory deafferentation (eg, spinal transection vs spinal transection+bike); (3) different serotonergic therapies (eg, quipazine vs quipazine+8-OH-DPAT) can induce opposite effects on brain plasticity in the same animal model of sensory deafferentation (eg, spinal transection+bike). In any case, particular caution should be adopted when attempting to translate results and therapies from animal models to patients.
In conclusion, our findings uncover the interactive effects between active/passive exercise and serotonergic pharmacotherapy on cortical reorganization after spinal cord injury, emphasizing the importance of understanding the effects of therapeutic strategies in spinal cord injury (and in other forms of deafferentation) from an integrated system-level approach.
Footnotes
Acknowledgements
We thank Dr Elizabeth Dugan for assistance with the transection surgery, animal care, and bicycle therapy and Jennifer Garcia for performing the spinal surgeries, managing the therapy protocols, and caring for the animals. We would also like to thank Dr Tim Himes and Theresa Connors and the Spinal Cord Research Center at Drexel University for assistance with the therapy protocols, animal care, transection surgery, and histological analysis and Dr Robert Flint and Matt Cozon for their assistance during the electrophysiological recordings.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grant 36206 from the Neilsen Foundation and Grants P01 NS 055976 and R01 NS05741 from the National Institutes of Health. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
