Abstract
Background. Spinal cord injury (SCI) induces alterations in cardio-autonomic control of which autonomic dysreflexia (AD), a condition characterized by life-threatening hypertension, is arguably the most insidious. Passive hind-limb cycling represents a low-cost therapeutic intervention with demonstrable cardiovascular, sensory, and motor benefits. Objective. To investigate the effect of passive hind-limb cycling on AD in rodents with T3 SCI. Methods. Forty-five male Wistar rats were evenly assigned to either uninjured control (CON), SCI, or SCI plus hind-limb cycling exercise (SCI-EX). At the end of the experimental period (day 32), rats were randomly assigned to stream 1 (n = 24) or stream 2 (n = 21). Stream 1 rats were assessed for AD severity (pressor response to colorectal distension) and were then perfused for tissue dissection and immunohistochemistry. Stream 2 rats underwent excision of the superior mesenteric artery for in vitro myography assessments. Results. From 2 weeks post-SCI onwards, SCI-EX rats exhibited a significant reduction in the pressor response to colorectal distension versus SCI (P < .001). Reduced AD severity in SCI-EX rats was accompanied by a prevention of the SCI-induced increase in density of CGRP+ afferents in the dorsal horn (P = .001). Conversely, both SCI and SCI-EX rats exhibited a similar degree of mesenteric endothelial dysfunction and α-adrenoceptor hypersensitivity versus CON. Conclusion. Passive hind-limb cycling reduces the severity of AD in SCI, and is correlated with changes in primary afferent morphology, but has limited effects on the peripheral vasculature.
Introduction
Spinal cord injury (SCI) disrupts autonomic pathways and perturbs cardiovascular homeostasis. 1 When injury occurs at or above the major splanchnic sympathetic outflow (sixth thoracic segment, T6), loss of tonic inhibitory and excitatory drive to the sympathetic preganglionic neurons causes a sequence of life-threatening cardiovascular complications. 2 Arguably the most dangerous is autonomic dysreflexia (AD), a condition characterized by reflex-evoked episodic hypertension. 3 AD is triggered by both noxious and nonnoxious visceral or somatic stimuli, of which distension of the bowel and bladder is the most common. 4 Left untreated, AD becomes a medical emergency that can cause seizures, myocardial infarction, intracranial bleeding, and even death. 5 Although poorly understood, the mechanisms underlying AD are likely to be multifactorial. In the periphery, the development of adrenoceptor hypersensitivity has been reported across numerous vascular beds after SCI and has been directly linked to AD. 6 In the spinal cord, sprouting of the central branches of calcitonin-gene related peptide (CGRP+) fibers into laminae III/IV of the lumbar dorsal horn and alterations in intraspinal circuitry are both thought to play an integral role in the AD reflex.7-9
Exercise is a viable intervention to reduce cardiovascular disease risk post-SCI, 10 and specific evidence-informed physical activity guidelines now exist for the adult SCI population. 11 To our knowledge, no clinical study has specifically examined whether chronic exercise training reduces AD severity. During acute exercise, body weight–supported treadmill training and functional electrical stimulation cycling have been associated with the presence12,13 or absence of AD,14,15 whereas lower-limb passive cycling appears to elicit only moderate increases in blood pressure that do not resemble AD, 16 suggesting perhaps that exercise modality is causatively related to the presence/absence of AD. In pre-clinical animal models of SCI, a single bout of treadmill exercise reduces arterial blood pressure immediately postexercise. 17 Although the authors attributed this finding to reduced AD severity, it is also possible that the rats exhibited postexercise hypotension that masked any AD events from being detected. In the only study investigating the effect of chronic exercise training on AD severity, it was reported that 6 weeks of treadmill training (10 min/day) exacerbates AD severity, increases sprouting of CGRP+ afferents into deeper laminae (III/IV) of the lumbar spinal cord, and potentiates responses to phenylephrine in the renal artery. 18 Of note is that treadmill training in both rodents and humans with SCI requires the experimenter to manually move (walk) the lower limbs. It is plausible that such repetitive somatic stimuli explains the exacerbation of SCI-induced plasticity within peptidergic afferents that was noted by Laird et al 18 and likely explains the negative effects of chronic treadmill exercise on AD severity.
Passive hind-limb cycling exercise, achieved via a motor driven hind-limb cycler, circumvents the requirement of the experimenter to manually move the hind-limbs and may therefore provide a more elegant method for exercising the lower limbs after SCI. Passive cycling has been previously shown to maintain hind-limb muscle mass, 19 stabilize rhythmic firing patterns in lumbar motoneurons, 20 and increase production of anti-inflammatory cytokines. 21 Moreover, we recently reported that passive cycling normalizes resting blood pressure and prevents maladaptive cardiac remodeling in rodents with T3 SCI. 10 The associated hyperemia during cycling may offset known SCI-induced maladaptive changes in the peripheral vasculature that have been previously implicated in AD. 22 From a sensory standpoint, hind-limb cycling may be advantageous over treadmill training since cycling would be expected to provide continuous (rather than repetitive) sensory input to the spinal cord from the foot remaining in contact with the pedal. Such sensory input may stabilize specific peripheral-central circuitry and prevent the SCI-induced maladaptive plasticity of peptidergic afferents within the cord from developing.
Thus, the aim of the present study was to investigate the effect of a 1-month passive hind-limb cycling intervention on AD severity, vascular function, and spinal cord plasticity. We hypothesized that hind-limb cycling reduces AD severity through both peripheral and central mechanisms.
Materials and Methods
Experimental Design
All procedures were conducted in strict accordance with the Canadian Council for Animal Care. Ethics approval was granted by the University of British Columbia. Experiments were conducted on 45 male Wistar rats (250-300 g; Harlan Laboratories, Indianapolis, IN). Animals were randomly assigned to the following groups: uninjured control (CON; n = 15), T3 complete SCI (SCI; n = 15), or T3 complete SCI plus hind-limb cycling (SCI-EX; n = 15). At the end of the experimental period (day 32), rodents from each group were randomly assigned to stream 1 (n = 24) or stream 2 (n = 21; see also Figure 1). Stream 1 rodents were assessed for the blood pressure response to colorectal distension (ie, AD severity) and were then perfused for tissue collection and immunohistochemistry. Stream 2 animals underwent excision of the superior mesenteric artery for in vitro myography assessments.
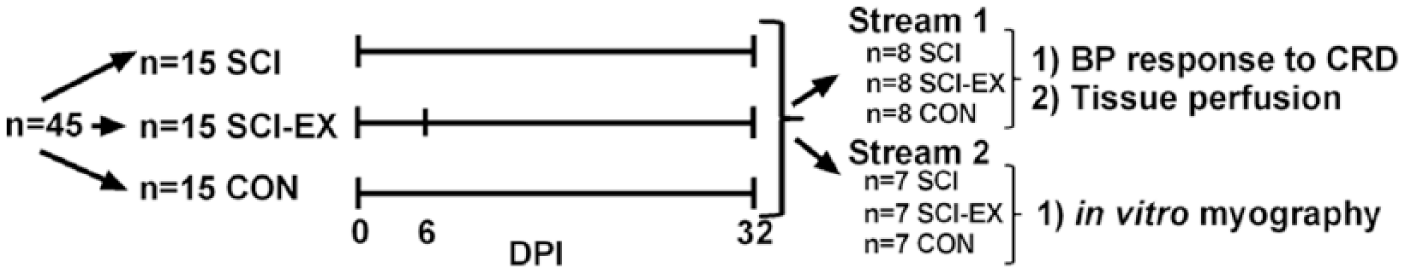
Experimental timeline. Animals were randomly assigned to T3 complete spinal cord injury (SCI; n = 15), T3 complete SCI plus hind-limb cycling exercise (SCI-EX; n = 15; beginning day 6 post-SCI), or uninjured control (CON; n = 15). At the end of the experimental period (day 32), rodents from each group were randomly assigned to stream 1 (n = 24) or stream 2 (n = 21). Stream 1 rodents were assessed for the blood pressure (BP) response to colorectal distension (CRD) and were then perfused for tissue collection and immunohistochemistry. Stream 2 animals underwent excision of the superior mesenteric artery for in vitro myography assessments. DPI, days postinjury.
Surgery and Animal Care
Rats were anaesthetized with ketamine hydrochloride (70 mg/kg, ip; Vetalar; AVP) and medetomidine hydrochloride (0.5 mg/kg, ip; Domitor; AVP). A dorsal midline incision was made in the superficial muscle overlying the C8-T3 vertebrae. The dura was opened at the T2-T3 intervertebral gap and the spinal cord was completely transected using microscissors. Complete transection was confirmed by 2 surgeons via visual separation of the rostral and caudal stumps, and Gelfoam (Pharmacia & Upjohn Company, Pfizer, NY) was placed between the stumps to minimize bleeding. Immediately following surgery, anesthetic was reversed with atipamezole hydrochrloride (1 mg/kg, sc; Antiseden; Novartis, Mississauga, ON) and animals were provided with 5 mL of warmed Lactated Ringer’s solution before being moved to a temperature-controlled chamber (Animal Intensive Care Unit, HotSpot for Birds, Los Angeles, CA). Buprenorphine (0.02 mg/kg, sc) and Ketoprofen (5 mg/kg, sc; Anafen; AVP) were administered daily for 3 days postinjury to manage postoperative pain. A dose of antibiotic (enroflaxcin; 10 mg/kg, sc) was also administered daily to reduce the incidence of bladder infection. During the first week postinjury, manual bladder expression was performed 4 times per day. After the first week this was reduced to 2 times per day until spontaneous bladder function returned. Animals were housed in groups of 3 to 4 per cage and provided with an enriched standardized diet 3 times per day as described previously. 23
Passive Hind-Limb Cycling Intervention
The SCI-EX group completed 4 weeks of hind-limb cycling, 5 days/week (beginning day 6 post-SCI, 2 × 30 minute sessions/day, 10 minutes rest between sessions), using a customized cycle ergometer. In each training session, rats were suspended in a horizontal (prone) position on a sling, with the hind-limbs hanging through large holes cut into the sling. The hind feet were secured to pedals with surgical tape and rats were cycled at a frequency of 0.5 Hz. Cycling was commenced after 5 days of recovery following SCI. 19 The acute cardiovascular and locomotor responses to passive hind-limb cycling have been published elsewhere, along with a depiction of the bike setup.10,19 The choice of timing for the initiation of cycling along with the frequency and duration of the cycling were based on previous experiments, which demonstrated that earlier initiation of cycling in rodents with SCI (ie, day 2 post-SCI) did not elicit the beneficial changes gene expression that cycling started at day 5 post-SCI did. 24
Blood Pressure Assessment
Carotid cannulation was performed in stream 1 animals (n = 8 per group) under gas anesthetic (isoflurane; 5% induction, 1% to 3% maintenance in 100% O2) as described previously. 25 The catheter was tunneled subcutaneously and externalized via a small incision at the base of the skull. Two hours following carotid artery cannulation, the catheter was connected to a fluid-filled pressure transducer (SP 844, MEMScAP; Skoppum, Norway). Rats were placed into their home cages and allowed to move freely during the blood pressure (BP) recordings. Following a 10-minute period of recording, a 5-minute baseline BP was obtained. Heart rate (HR) was estimated from the beat-to-beat BP interval. In our follow-up experiment (separate stream to those listed above) to track the temporal development of AD in animals that did (n = 4) or did not (n = 4) undergo hind-limb cycling, BP was assessed using a telemetry device inserted into the abdominal aorta (just distal to renal arteries). The procedure for aortic implantation is reported elsewhere. 10
Severity of AD was assessed via 2 bouts of colorectal distension (CRD), a procedure that is well characterized in our laboratory.25-27 CRD was not performed in uninjured controls. Animals were unrestrained and freely moving within their cage during the CRD procedure. To initiate CRD, a small, deflated plastic balloon (the balloon tip of a Swan-Ganz catheter; 10 mm in length) was inserted rectally for a distance of 1.5 cm. When BP and HR had stabilized (~5 minutes), the balloon was infused with 2 mL of air over 10 seconds and distension was maintained for 1 minute. CRD was performed twice with a minimum interval of 10 minutes. Beat-to-beat arterial pressure from either the carotid cannulation or telemetric system was monitored using PowerLab and Chart 7 for Windows (ADInstruments, Colorado Springs, CO). Raw, unfiltered beat-by-beat BP and HR data were averaged over 1-second intervals and the maximum increase in systolic BP (SBP) and maximum decrease in HR was averaged over the 2 trials.
Histological Analysis
Rats in stream 1 were transcardially perfused with phosphate-buffered saline (PBS), followed by 4% paraformaldehyde in a 0.1 M phosphate buffer. The lumbosacral spinal cord was removed, postfixed in 4% paraformaldehyde, and subsequently transferred to 20% sucrose. The L4/L5 spinal cord was cryosectioned at 10 µm (coronal sections, n = 5 per group). Slides were incubated in blocking medium (10% normal donkey serum) in PBS plus Triton X-100 (0.1%) for 20 minutes. CGRP primary antibodies (Sigma-Aldrich [catalogue number c8198], St Louis, MO; 1:1000) were applied in PBS plus Triton X overnight. Secondary antibodies raised in donkey and conjugated to Alexa 488 (Invitrogen [catalogue number a21202]) were applied at 1:100, in PBS–Triton X for 2 hours. Images were obtained with an epifluorescent microscope (Axioplan 2, Zeiss, Jena, Germany), equipped with a digital camera (Q Imaging, Burnaby, Canada). Image files were analyzed with Sigma Scan Pro 5 (SPSS, Inc, Chicago, IL). Identical imaging settings were used for each antigen. The density of CGRP+ axon terminals was measured as a function of depth at 2 sites in the dorsal horn of the L4/L5 cord (medial and lateral) as described previously. 28 Briefly, two 100-µm wide strips were placed between the uppermost border of gray matter and Lamina V either immediately below Lissauer’s tract (medial) or more laterally (lateral) where superficial lamina curl ventrally. Intensity measurements were captured every 10 µm until 300 µm deep and expressed as the density of CGRP+ axons within a given area (ie, proportional area). Images were passed through a Laplacian omnidirectional edge-detection filter and terminal profiles were selected with a threshold overlay. Density measures for each animal represent an average across 12 sections. Each section was separated by 100 µm. For statistical purposes, the average density measure for each animal was treated as one observation.
In Vitro Myography
Sections from the proximal end of the superior mesenteric arteries were dissected from the gut of deeply anaesthetized rats in stream 2. Arteries were immersed in physiological salt solution (composition: NaCl [119 mM], KCl [4.7 mM], KH2PO4 [1.18 mM], MgSO4 [1.17 mM], NaHCO3 [24.9 mM], EDTA [0.023 mM], CaCl2 [1.6 mM], and dextrose [11.1 mM]), cut into 2-mm segments and mounted isometrically onto a 4-channel wire myograph (JP Trading, Aarhus, Denmark), such that vascular responses represent the average of 4 segments. Segments were prepared and equilibrated as described previously, 25 after which they were constricted with potassium chloride (KCl) to ensure viability and allow normalization of developed force. Following PBS wash, phenylephrine (PE) was added in a cumulative manner (1 nM to 10 µM) to generate concentration–response curves. Force generated was normalized to contractile force induced by KCl. To examine endothelium-dependent vasodilatation, arterial segments were treated with 1 µM PE to establish a stable contraction and then exposed to cumulative addition of acetylcholine (ACh; 1 nM to 10 mM). Vasodilator responses were expressed as percent relaxation of PE-induced contraction. Maximal contraction/dilatation and half-maximal effective concentration (EC50) of PE and ACh were determined for each group, along with the slope of the Hill equation.
Locomotor Function
Hind-limb locomotion was scored by the open-field locomotor scale described by Basso, Beattie, and Bresnahan (BBB). 29 Briefly, the BBB is a 21-point scale in which rats are scored based on hind-limb movements made in an open field during a 4-minute interval while freely exploring the surroundings. The BBB assessment was conducted by 2 individuals who were experienced with this assessment and who were blinded to the study groups and objectives. The 2 investigators independently scored each rodent and the mean was used for analyses.
Statistical Analyses
Between-group differences for density of CGRP+ axons in the L4/L5 spinal cord and telemetric analyses of induced AD severity were assessed by 2-way repeated-measures ANOVA, with one factor for group and one factor for distance into the dorsal horn/time of distension. Post hoc testing was conducted with a Bonferroni correction. All other cardiovascular variables were assessed using a 1-way ANOVA with Bonferroni-corrected pairwise comparisons, Kruskal–Wallis, or independent-samples t tests where appropriate. For in vitro myography data, GraphPad V5.0 was used for curve fitting and calculation of half-effective concentration (EC50). Statistical analyses were conducted using STATA v12.0. Significance was set at P < .05.
Results
Passive Hind-Limb Cycling Attenuates the Severity of AD
Compared to SCI rats, SCI-EX rats had a significant reduction in the pressor response to CRD (50 ± 5 vs 27 ± 3 mm Hg, P < .001) and a concomitant reduction in CRD-induced bradycardia (−62 ± 6 vs −14 ± 3 bpm, P < .001). There were no between-group differences in resting SBP or HR at pre-CRD baseline. To examine the temporal development of AD in both the SCI and SCI-EX groups, we repeated the current study (n = 4 SCI, n = 4 SCI-EX; separate stream of animals) and additionally placed telemetry devices into the descending aorta. We then assessed the severity of AD on a weekly basis from 1 week to 1 month postinjury. Compared to SCI, we found that the blood pressure response to CRD (ie, AD severity) was markedly reduced in SCI-EX at weeks 2, 3, and 4 postinjury (all P < .01; Figure 2A-D).
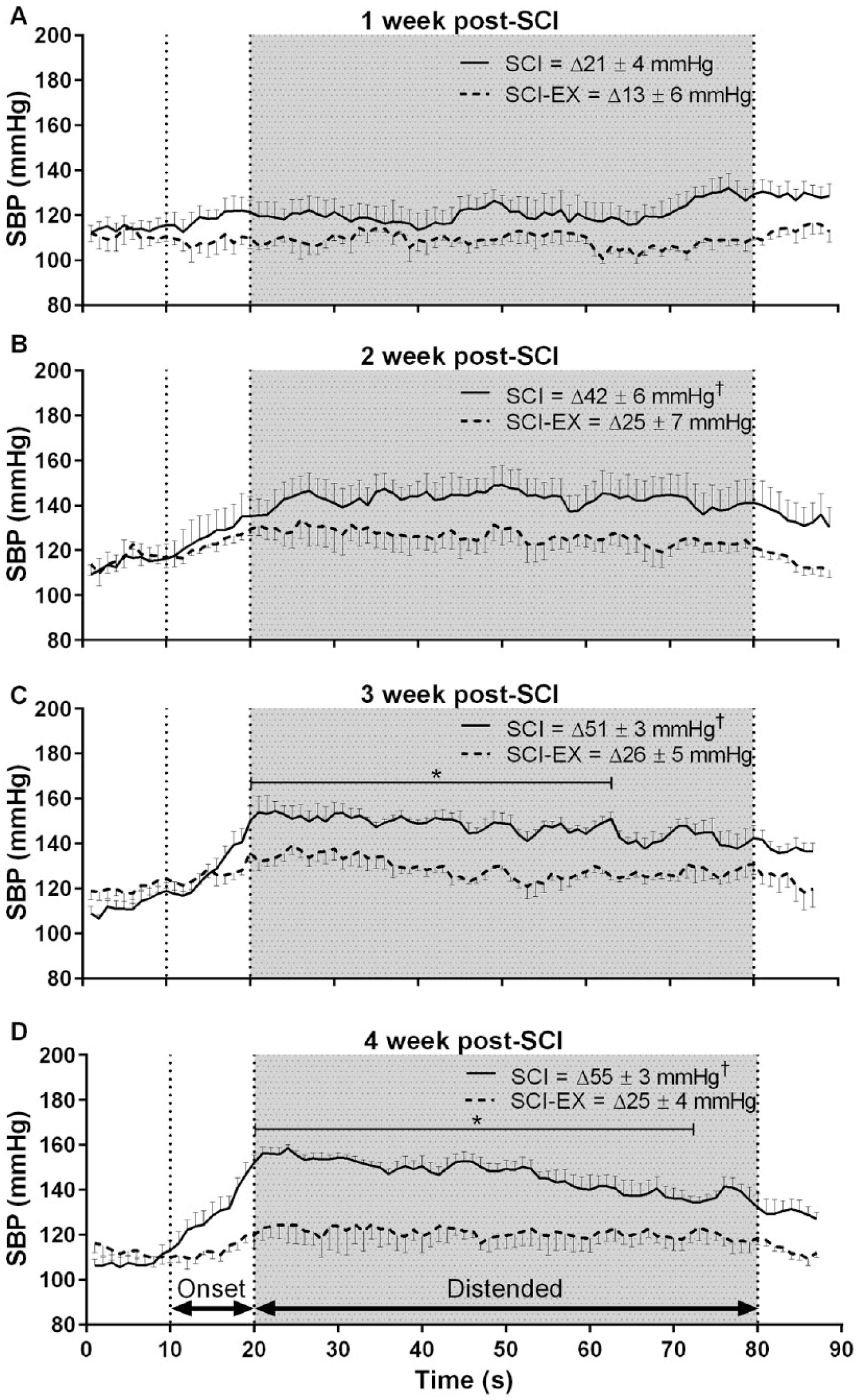
Group mean ± SEM averaged (1 second) systolic blood pressure (SBP) responses to colorectal distension in SCI (n = 4, solid lines) and SCI-EX (n = 4, dotted lines) by time postinjury. Note that autonomic dysreflexia was most pronounced at 3 and 4 weeks post-SCI. Note also that the blood pressure response to distension was significantly attenuated in SCI-EX versus SCI at 2, 3, and 4 weeks postinjury. *P < .05 between-group difference at that time-point during distension. †P < .05 between-group difference for the maximal change (1-second average) in SBP during distension. SCI, spinal cord injury.
Passive Hind-Limb Cycling Prevents SCI-Induced Aberrant Plasticity of CGRP+ Axons
SCI caused increased CGRP+ afferent density in superficial laminae (I-IIo), and invasion of CGRP+ afferents into lamina IV compared to CON (Figure 3A-C; all P < .05). For SCI-EX, however, the density of CGRP+ afferents was significantly less in superficial laminae (I-IIo) and virtually absent in lamina IV/V compared to SCI (P = .026).
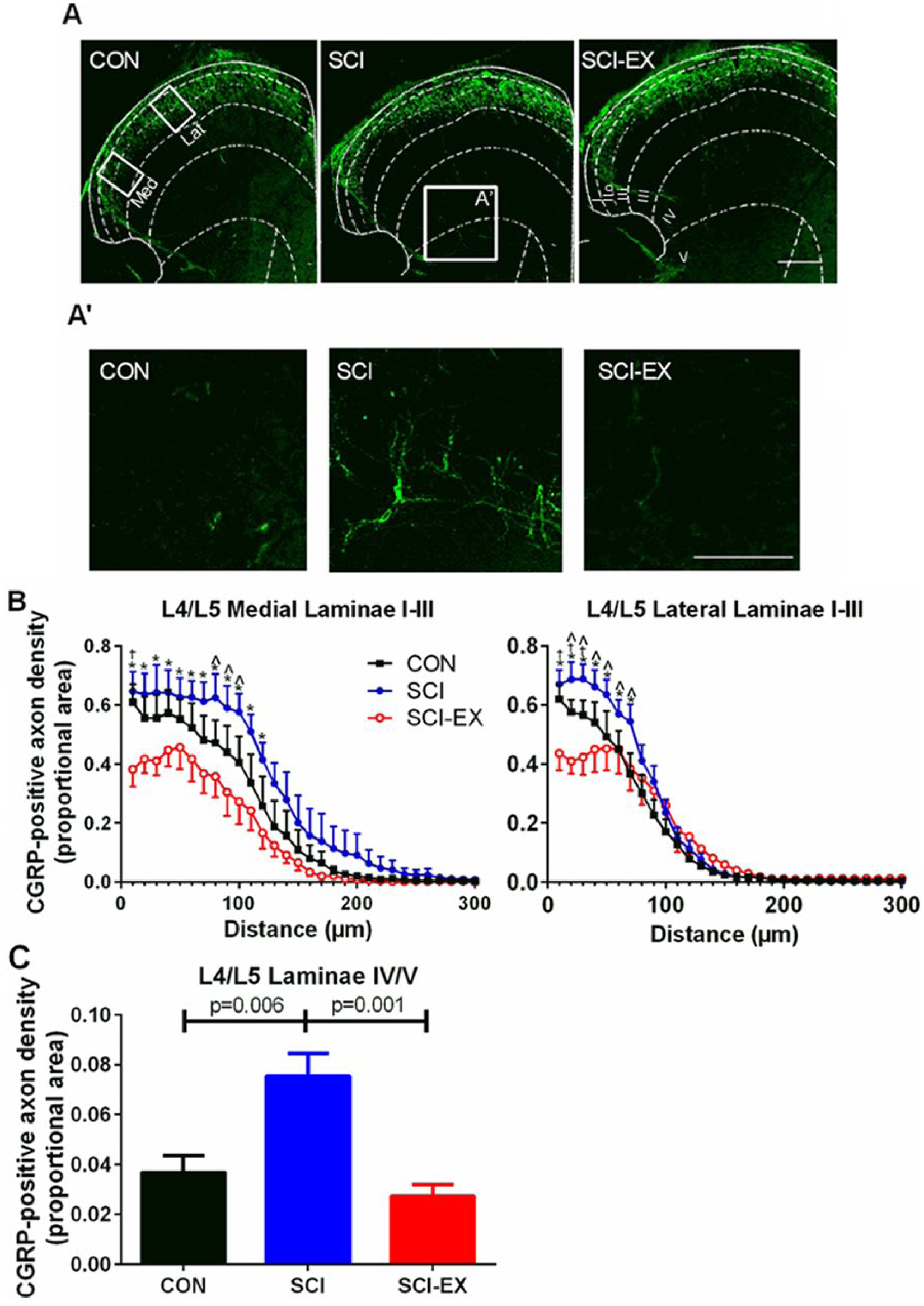
Calcitonin gene-related peptide+ (CGRP+) afferent density in the L4/L5 dorsal horn. (A) Example CGRP+ immunostaining in laminae I-V of the dorsal horn. White dotted lines denote stencil outline of the location of each spinal laminae. med = location of medial measurements in panel B. Lat = location of lateral measurements in panel B. (A′) Higher magnification of the box outlined in panel A showing CGRP+ afferent density in lamina IV/V. (B) Density of CGRP+ afferents by distance into the dorsal horn.*P < .05 between spinal cord injury (SCI) and SCI-EX at that distance. †P < .05 between SCI-EX and CON at that distance. ^P < .05 between SCI and CON at that distance. (C) Group mean ± SEM of CGRP+ afferents in lamina IV/V of the spinal cord. Scale bar for A and A′ = 100 µm.
SCI Induces Vascular Dysfunction That Is Not Reversible With Passive Hind-Limb Cycling
SCI elicited a leftward shift of the PE concentration response curve (Figure 4A); hence, half maximal effective concentration (EC50) of PE was lower in SCI versus CON vessels (P = .041; Figure 4B). Importantly, we demonstrate that SCI-EX also exhibited a leftward shift in the PE concentration–response curve (Figure 4A); hence, EC50 is also lower in SCI-EX versus CON vessels (P = .007) but no different versus SCI vessels (Figure 4B). Maximal force in response to potassium and maximal contraction in response to PE were not different between groups (see also Figure 4C). Further to changes in PE sensitivity, we also found that SCI elicited a rightward shift of the ACh concentration–response curve (Figure 5A) and a consequent increase in EC50 compared to CON (P = .008; Figure 5B). Interestingly, there was an almost identical rightward shift in the ACh response curve in SCI-EX; hence, EC50 was increased in SCI-EX versus CON vessels (P = .003; Figure 5B) but not different from SCI (P = 1.00). Maximal relaxation did not differ between groups (P = .14; Figure 5C).
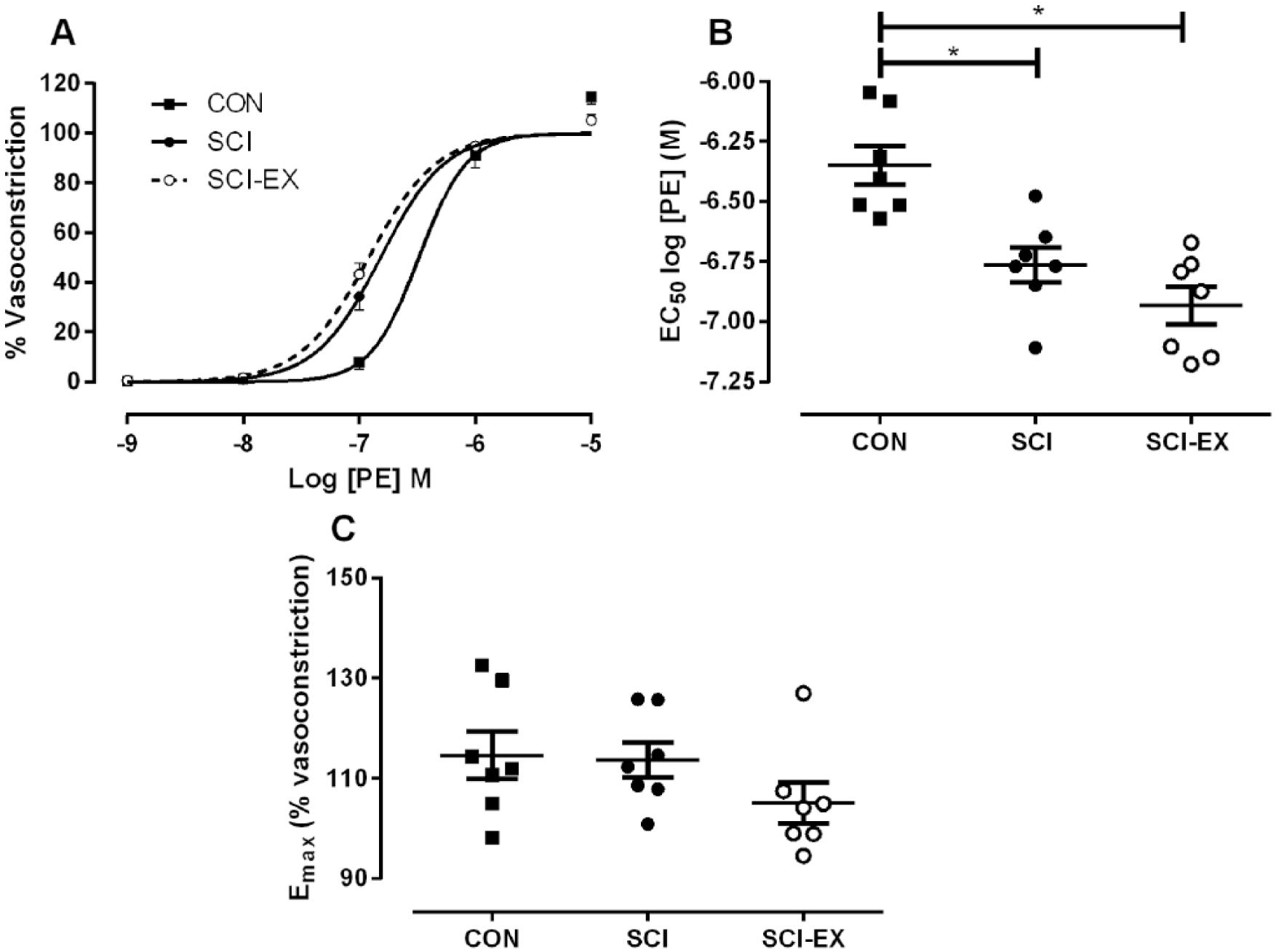
Mesenteric vascular responses. (A) Spinal cord injury (SCI) caused a significant leftward shift in the concentration–response curve of mesenteric arteries to phenylephrine (PE) compared to uninjured control (CON) that is not reversible with hind-limb cycling (SCI-EX). (B) Half-maximal effective concentration (EC50) of PE was reduced after SCI and SCI-EX versus CON. *P < .05. (C) Maximal vasoconstriction (Emax; in response to 10–5 M PE) was not different between groups. Symbols represent individual animals. Group mean ± SEM also shown.
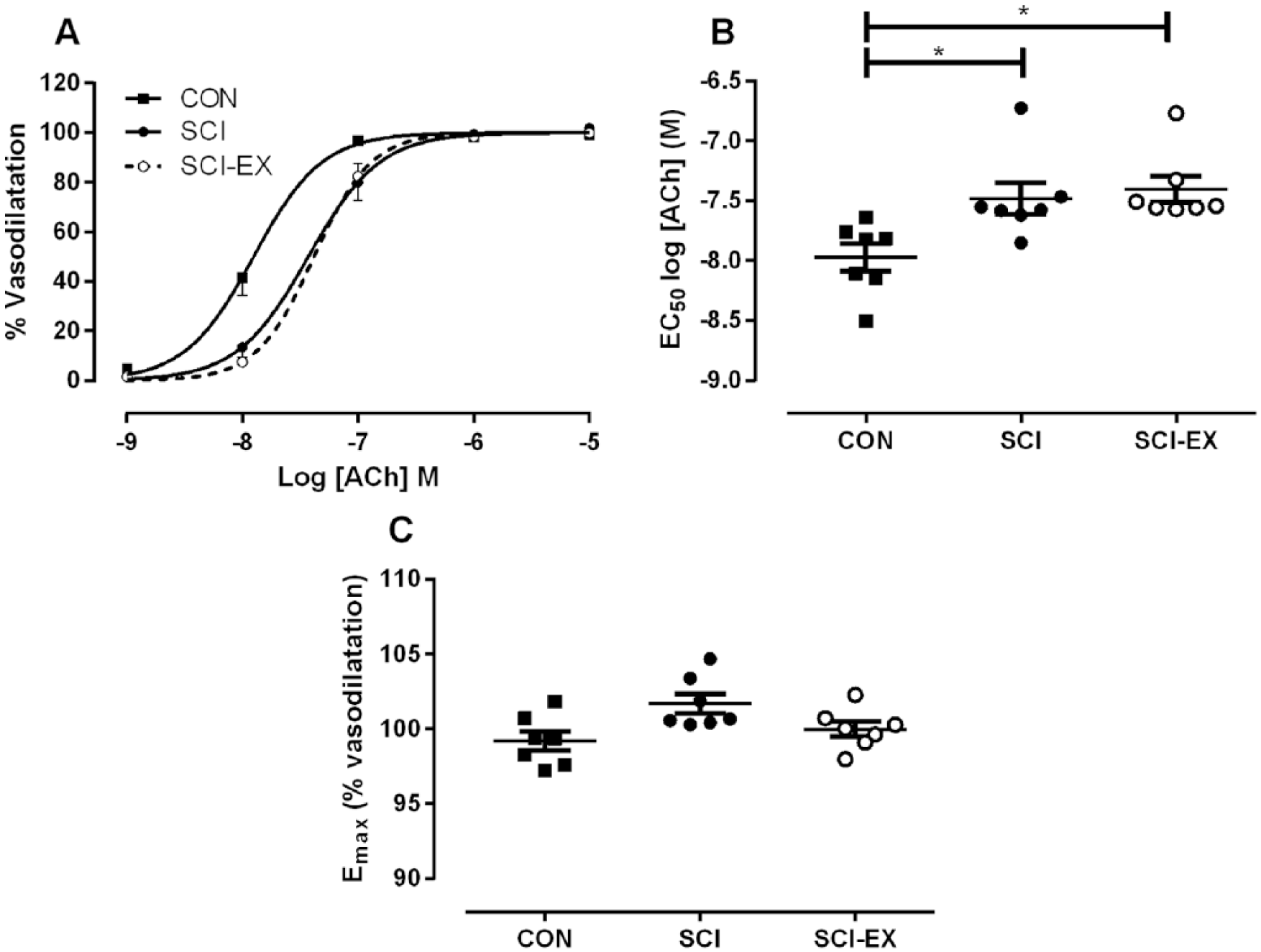
Mesenteric endothelial function. (A) Spinal cord injury (SCI) caused a significant rightward shift in the concentration–response curve of mesenteric arteries to acetylcholine (ACh) compared to uninjured control (CON) that is not reversible with hind-limb cycling exercise (SCI-EX). †P < .05 between SCI-EX and CON. ^P < .05 between SCI and CON. (B) EC50 was increased after SCI and SCI-EX versus CON. (C) Maximal vasodilatation (Emax; in response to 10−6M ACh) was not different between groups. Symbols represent individual animals. Group mean ± SEM also shown.
Hind-Limb Cycling Exercise Does Not Improve Locomotor Ability
We assessed locomotor ability at the terminal experimental time-point using the BBB score. There was no significant difference in BBB score between SCI and SCI-EX (2.1 ± 0.6 vs 1 ± 0.1, P > .05). The slightly higher value in the SCI group was due to 1 rat that scored a 6, all other animals in both groups scored between 0 and 1.5.
Discussion
We report for the first time that passive hind-limb cycling significantly reduces the severity of AD in rodents with high-thoracic SCI. Accompanying the cycling-induced reduction in AD severity are statistically significant reductions in the density of CGRP+ afferents in superficial and deep lumbar spinal laminae. Conversely, SCI-induced peripheral adrenoceptor hypersensitivity and endothelial dysfunction persist after cycling, suggesting that passive cycling has limited effects on the peripheral vasculature.
Passive hind-limb cycling reduced AD severity from week 2 onwards. At 1 week post-SCI (at which point SCI-EX had only undergone 2 days of cycling) neither group displayed a characteristic pressor response to distension, most likely due to the presence of neurogenic shock and acute changes within spinal-sympathetic circuitry after SCI.30,31 This finding is in agreement with our recent report that characteristic AD only begins to occur at approximately 2 weeks post-SCI, 32 most likely because this is when plasticity within sympathetic circuitry that appear necessary for AD begins to develop. 30 Thus, we believe the timing of cycling initiation is critical to reduce the severity of AD. On the one hand, exercise needs to start early enough before maladaptive plasticity within spinal and sympathetic circuitry begins to occur, but on the other hand, if exercise is started too early then it may exacerbate secondary damage around the SCI lesion and impair outcomes. 33 Thus, future studies that initiate exercise at varying time point post-SCI are urgently required to identify the optimum “AD-prevention time point” for exercise initiation.
A reduction in AD severity in response to cycling contradicts the only previous study that examined the effect of exercise on AD severity in rodents with SCI, whereby treadmill training was shown to exacerbate AD severity (see also Introduction). 18 Interestingly, our finding of a cycling-induced reduction in CGRP+ afferents entering lamina IV/V of the dorsal horn (or a prevention of the de novo expression of CGRP in axons that were previously CGRP−) and reduced density of CGRP+ afferents in superficial laminae I and II is also contradictory to that of Laird et al, 18 who reported increased CGRP+ immunoreactivity in the lumbar cord. It is thought that reduced sprouting of CGRP+ axons into deeper spinal laminae prevents/reduces sensory transmission to interneurons located within laminae IV that likely mediate spinal sympathetic reflexes that ultimately present as AD.34,35 The mechanism(s) responsible for the differential plasticity of the spinal cord in response to treadmill training and hind-limb cycling is not entirely clear, but may stem from modality-dependent changes in growth factors and/or sprouting-inhibiting factors. In this respect, a number of neurotrophic factors have been shown to be differentially regulated in response to treadmill training and passive hind-limb cycling following low-thoracic SCI. 36 Although no study has examined the neurotrophic response to cycling following high-thoracic SCI, a cycling-induced modulation of neurotrophic factors is certainly an attractive candidate for reduced AD severity given the known effect that altering nerve growth factor levels has on reducing AD severity post high-thoracic SCI.8,9 An intriguing alternative hypotheses to explain the differential effects of treadmill training and cycling training on AD severity may relate to the type of sensory stimulation achieved with each. In treadmill training, sensory stimulation will be intermittent and unpredictable based on the timing of foot placement by the experimenter, whereas cycle training will elicit regular and predictable stimulation. Previous research that investigated regular and unpredictable electrical stimulation of the spinal cord in the absence of supraspinal control reported that regular stimulation promoted beneficial spinal plasticity that is both N-methyl-D-aspartate receptor and brain-derived neurotrophic factor dependent. 37 The authors also suggested that regular stimulation may reduce the negative implications of uncontrolled noiciceptive input post-SCI, which appears to be supported by the findings of the present experiment.
In the peripheral vasculature, we expected that the hyperemia associated with hind-limb cycling would improve endothelial function due to the associated increase in shear stress and that a reduction in AD severity with cycling may negate α-adrenoceptor hyperresponsiveness. Contrary to this, we found that cycling was unable to reverse SCI-induced mesenteric endothelial dysfunction or PE hypersensitivity. In the non-SCI population, endothelial function is improved following both active 38 and passive exercise. 39 The reason for the lack of improvement in endothelial function with hind-limb cycling in our study is unclear. We previously reported that the same intervention normalizes the SCI-induced reduction in mean arterial pressure, offsets reductions in cardiac contractility, and improves stroke volume 10 ; thus, we are confident that cycling elicits hyperemia. It is possible that cycling did induce moderate changes in endothelial function that were not of a sufficient magnitude to chronically alter ACh sensitivity, or that cycling may have induced changes in vascular beds that are more local to the stimulus. That cycling did not reduce mesenteric PE responsiveness in the face of attenuated AD severity was also unexpected. Previously, both human and animal studies have found exaggerated responsiveness of the peripheral α-adrenoceptors to sympathomimetics40,41 and have implicated such responses as a key component of AD.42,43 It is of interest that a previous study in our laboratory found that repetitively inducing AD for 2 weeks post-SCI exacerbates mesenteric PE responses but reduces the pressor response to a single bout of colorectal distension. 25 We believe these previous findings in conjunction with those from the current study imply that exaggerated responses of the peripheral vasculature to symapthomimetics may purely be coincidental with AD rather than a direct cause. The reason for the lack of a cycling-induced effect on PE responsiveness remains unclear. Acute treadmill training in SCI rodents has been shown to reduce PE responsiveness in the immediate postexercise period, 17 whereas chronic exercise training moderately exacerbates the sublesional vascular (renal) response to PE. 18 It is possible that passive hind-limb cycling produced an insufficient release of catecholamines to stimulate adrenergic receptors to elicit adaptation. In this respect, clinical evidence suggests that SCI individuals with loss of descending sympathetic control over the splanchnic vasculature exhibit only a minor exercise-induced increase in circulating catecholamines during lower-limb functional electrical stimulation cycle exercise. 16
The present study revealed no improvement in locomotor outcomes as evidenced by a similar BBB score between SCI and SCI-EX. Both groups scored between 1 and 2 on the BBB scale, which is consistent with other studies investigating locomotor outcomes following complete thoracic SCI. 18 The lack of a beneficial effect of cycling on locomotor function was not surprising since recovery of locomotor function in SCI likely occurs only in response to task-specific training, such as treadmill training, which is thought to activate the central pattern generator in the lumbar cord.44,45 Moreover, previous research that investigated the effect of passive hind-limb cycling on locomotor function in rats with unilateral cervical contusion SCI reported no beneficial locomotor effects. 21
Conclusion
We provide evidence that passive hind-limb cycling reduces the severity of AD in rats with high-thoracic SCI. That the reduction in AD severity was accompanied by changes in sensory afferents but not changes in vascular hyperresponsiveness provides an important contribution to the long-standing debate on the mechanism(s) responsible for the development of AD in SCI. The remarkable similarity in the cardio-autonomic profile of our animal model of SCI to the clinical population suggests that further research should be directed toward lower-limb passive cycling in humans with SCI. Passive lower-limb cycling requires little assistance, is low cost, and is remarkably easy to perform both during rehabilitation and in the home.
Footnotes
Acknowledgements
The authors would like to acknowledge the insight and expertise of Professor John Houle in conducting hind-limb cycling in rodents with SCI. The authors would also like to acknowledge the assistance of Dr Leanne Ramer and Ms Jess Inskip for assistance with blood pressure measurements, and Ms Peggy Assinck and Mr Greg Duncan for assistance with locomotor assessments.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Canadian Institute for Health Research (Grant Number TCA 118348). Research in the laboratory of AK is supported by the Canadian Foundation for Innovation, BC Knowledge Translation Foundation, Canadian Institute for Health Research, and Craig Neilsen Foundation. CW is the recipient of Craig Neilsen Foundation and Michael Smith Foundation Postdoctoral Fellowships.
