Abstract
Background. Our ability to sense movement is essential for motor control; however, the impact of kinesthesia deficits on functional recovery is not well monitored in the spinal cord injury (SCI) population. One problem is the lack of accurate and reliable tools to measure kinesthesia. Objective. The purpose of this study was to establish the validity and reliability of a quantitative robotic assessment tool to measure lower limb kinesthesia in people with SCI. Methods. Seventeen individuals with an incomplete SCI and 17 age-matched controls completed 2 robotic-based assessments of lower limb kinesthesia sense, separated by at least 1 week. The Lokomat, a lower limb robotic exoskeleton, was used to quantify the movement detection score bilaterally for the hip and knee joints. Four passive movement speeds (0.5, 1.0, 2.0, and 4.0 deg/s) were applied in both flexion and extension directions. Participants responded via pressing a joystick button when movement was felt. Results. The movement detection score was significantly greater in people with SCI compared with the control group, particularly at the slowest movement speed. The difference between groups was more pronounced among those classified as ASIA (American Spinal Injury Association) Impairment Scale B. Our measure showed high test-retest reliability and good internal consistency for the hip and knee joints. Conclusions. Our findings demonstrated that lower limb kinesthesia deficits are common in the SCI population and highlighted the importance of valid and reliable tools to monitor sensory function. Future studies need to examine changes in sensory function in response to therapy.
Introduction
Proprioception is the ability to sense the relative positions of our body segments (ie, joint position sense) and detect that movement of a body segment has occurred (ie, movement sense or kinesthesia). 1 This sensory function is critical for behaviors that require coordinated movements between body segments, 2 such as balance, 3 walking, 4 visual-motor tasks, 5 and functional activities of daily living. 6 For individuals with a spinal cord injury (SCI), damage to the dorsal column or spinocervical thalamic tract (ie, Morin’s tract) can lead to impaired transmission of proprioceptive signals from below the injury level,7,8 While proprioceptive deficits are recognized as important and present among people with SCI,2,9 there has been limited ability to accurately identify the incidence and severity.
Although intact proprioception is important for functional recovery, current standardized assessment tools of sensory function in the SCI population have been limited to measures of light touch, 10 pain,10,11 vibration, 12 and temperature. 13 In clinical settings, assessments of proprioception are not objective and lack sensitivity to detect changes in function. 14 Examples of assessing proprioceptive sense include the ability to accurately discriminate the direction of a passive movement, 12 and actively replicating a joint angle in the same or opposite limb as initially positioned by a clinician. 12 In the latter case, the response accuracy is usually determined by visual estimation or manual goniometer measurement. Thus, there is a need to develop easy and accurate tools to quantify features of proprioceptive function.
Recent technological advances in robotics have the potential to offer better precision and reliability to measure proprioceptive deficits. As well, these tools have the ability to move a limb through a specified range of motion, which eliminates the severity of motor deficits as a potential confounder. Robotic-based protocols for assessing joint position sense have been developed to assess the degree of upper limb impairment among people with stroke,15-17 traumatic brain injury 18 and cerebral palsy, 19 and lower limb impairment in people with SCI.20,21 These tools have been helpful to characterize deficits in joint position sense due to neurological injury, and develop a better understanding of the relationship between proprioception and motor impairment. 6 However, fewer studies have focused on developing techniques for measuring kinesthesia deficits, and have been limited to the upper extremity.22,23
Here we present a novel robotic-based protocol to assess lower limb kinesthesia among people with SCI. Compared with clinical tools, our approach uses precise and reliable measurements from robotic sensors and allows standardized delivery of the assessment, while limiting variability from other factors, such as the degree of motor impairment. Developing better assessment tools has important clinical implications, as the recovery of motor function can be highly influenced by sensory deficits. Current studies have suggested that sensory function (ie, light touch and pin prick sensation) does not contribute to improved locomotor function after training in SCI. 24 However, it is possible that we are not measuring the appropriate component of sensory function to track changes in recovery. By developing techniques to quantify and evaluate proprioception, we will be better able to monitor the effects of locomotor training strategies, and design therapies directed at improving proprioceptive deficits to maximize recovery. Thus, it is important to establish an objective, accurate, and reliable tool for assessing lower limb kinesthesia.
The aim of this study was to (a) quantify lower limb kinesthesia deficits among people with SCI and (b) evaluate the reliability and validity of a robotic-based assessment tool to measure lower limb kinesthesia sense. We hypothesized that the robotic-based assessment will demonstrate strong internal consistency and test-retest reliability, and that the assessment of lower limb joint kinesthesia will show validity through the ability to discriminate between able-bodied controls and people with SCI.
Methods
Subjects
Seventeen individuals with a chronic incomplete SCI (American Spinal Injury Association [ASIA] Impairment Scale B, C, or D), who were in stable medical condition, had adequate lower limb range of motion (at least 40° of hip flexion and 30° of knee flexion), could tolerate an upright posture, and able to follow instructions participated in this study. The International Standards for Neurological Classification of Spinal Cord Injury (ISNCSCI) were used to classify impairment level.10,25 Subjects were excluded if they had any other neurological injury (eg, brain injury, stroke) or musculoskeletal disease (eg, muscle pain, broken or weak bones), or exceeded the capacity limits of the Lokomat (weight more than 136 kg or height greater than 1.85 m). Adults with no neurological, cardiovascular, or musculoskeletal injuries participated as age- and sex-matched controls. The University of British Columbia Clinical Research Ethics Board approved all procedures, and all subjects gave their written informed consent.
Protocol
Participants completed 2 assessments of lower limb joint kinesthesia, separated by at least 1 week. During the first session, the ISNCSCI light touch and pin prick examination was also conducted to provide a score out of 36 (sum of L1 and below). In addition, a manual assessment of kinesthesia was performed bilaterally for the hip and knee joints. Participants lay supine on a mat with their eyes closed while a researcher moved each joint separately through a randomized order of 10° of flexion or extension. The starting positions of the hip and knee joints were 20° and 30° of flexion, respectively. Subjects were instructed which joint would be tested, that there would be a delay before the movement starts, and that catch trials with no movement may occur. At the end of each movement, participants were instructed to indicate if they felt a movement and in what direction did the movement occur. One point was given for each correct response for a total out of 10 per joint.
For the robot-based kinesthesia assessment, participants were suspended in the air with an overhead harness system and attached to the Lokomat robotic gait orthosis (Hocoma AG, Volketswil, Switzerland; Figure 1). The ankle joints were held in a neutral position with 90° between the foot and shank by passive foot lifter straps. The untested foot was placed onto a platform so that the participant could bear some weight on that leg for comfort, reducing weight supported by the harness. Vision of the lower limbs was blocked throughout the testing procedure to prevent visual cues. Kinesthesia was tested bilaterally for the hip and knee joints using custom software control of the Lokomat. Rest breaks were provided at the end of testing for each joint. Participants were instructed to keep their legs passive throughout the experiment, and allow the Lokomat to perform the movement. Electromyography was used to verify that muscle activity of the tibialis anterior, medial gastrocnemius, rectus femoris, and semitendinosus remained at a resting level (Delsys Inc, Natick, MA, USA). Data were sampled at 1000 Hz. Trials with any burst of muscle activity were re-collected. Similarly, if spasticity was observed or felt by the subject, the trial was discarded and re-collected.
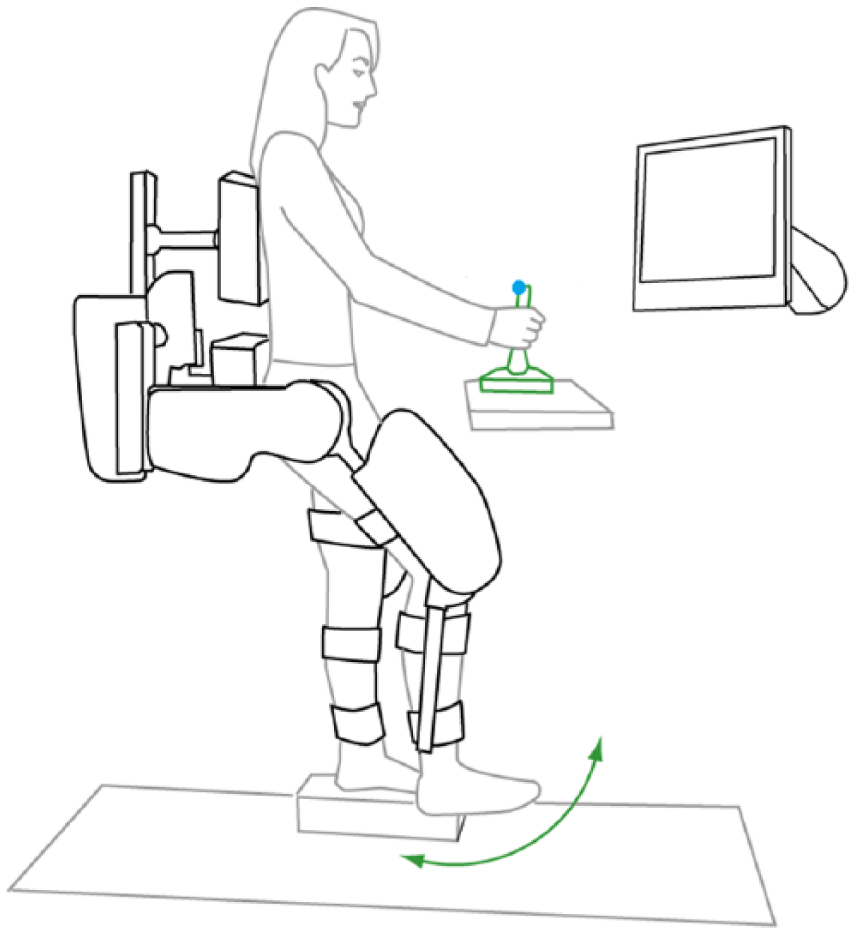
The experimental setup (vision of the legs was blocked with a curtain—not shown).
Twenty-seven trials were performed for each joint with the Lokomat in a randomized order of movement speeds (0.5, 1.0, 2.0, and 4.0 deg/s), directions (flexion and extension), and catch trials (no movement). Only one joint was tested at a time. Before each set of trials, participants were informed (a) which joint would be tested, (b) that there would be a random delay (eg, within 10 seconds) before each movement starts, (c) that there would be catch trials with no movement, and (d) that they should push the joystick button when they detect a change in their joint position and then state which direction they felt the movement. The Lokomat joints were programmed to move from the start position (10° for the hip and 30° for the knee) through 10° of motion, or until the joystick button was pressed. Joint angles from the Lokomat joint encoders and reaction time data from the joystick were collected using custom software written in LabView (National Instruments, Austin, TX, USA), sampled at 40 Hz.
The amount of movement prior to detection and whether the subject perceived the correct direction was used to calculate the movement detection score. The movement detection score is calculated by dividing the joint displacement prior to detection by 10, which scales the maximum value to 1, then adding 1 or 0 for an incorrect or correct direction response, respectively. The maximum possible score is 2, with higher values indicating poorer kinesthesia sense.
Because of possible effects of upper extremity motor impairment across participants with a SCI, reaction time to a cutaneous stimuli delivered to the cheek was also recorded. A peripheral nerve stimulator was used to randomly elicit an electrical impulse at an intensity just above their sensory threshold. Participants were instructed to push the joystick button as fast as possible when a pulse was detected. The test was conducted for 1 minute with 15 pulses delivered. Data was sampled at 1000 Hz in a LabView program to determine average reaction time between the stimuli and response onset times.
Data and Statistical Analysis
Data recorded were analyzed in Matlab (Natick, MA, USA) to determine the amount of joint displacement from the start of each trial to the button press or maximum joint excursions of 10°. If no response was indicated, the maximum displacement was recorded. The movement detection score was used as the primary outcome measure for statistical analyses. Descriptive statistics (mean ± standard deviation) were used to summarize the movement detection score across each speed tested for the hip and knee joints (left and right sides combined). A Pearson correlation was used to determine if the response to the cutaneous stimuli at the cheek was related to the movement detection score. Individuals in the SCI group were subdivided into their AIS classification to quantify kinesthesia deficits across impairment level. A one-factor analysis of variance was performed to compare the total movement detection score based on group (AIS B, C, D, and controls) separately for the hip and knee joints. Tukey post hoc analysis was performed to adjust for multiple comparisons.
A separate analysis was performed for the known groups’ validation design to evaluate the validity of the Lokomat kinesthesia measure. A mixed factorial analysis of variance, with 1 between-subject factor (group) and 3 within-subject factors (visit, joint, Lokomat speed), was performed to compare average movement detection score. Reaction time to the electrical stimulus at the cheek was added as a covariate to the model to control for differences in upper extremity motor function between subjects. The factors had the following levels: group (SCI and controls), joint (left hip, right hip, left knee, right knee), Lokomat speed (0.5, 1.0, 2.0, 4.0 deg/s), and visit (session 1 and 2). Pairwise contrasts examined differences within factors that had multiple levels. Post hoc analysis was performed with the least significant differences test was used to adjust for multiple comparisons. Normality was verified within each group using the Shapiro-Wilk test. Significant differences were assessed at an α of .05.
To determine the relationship between the robotic and manual tests of kinesthesia in the SCI group, Spearman’s rank correlation coefficients were calculated using the movement detection scores for each joint from the first session. We also tested the correlation between the total movement detection score from the robotic kinesthesia test and the ISNCSCI sensory scores for light touch and pin prick (sum from L1 and below) using the Spearman test.
Pearson correlation coefficients were computed between each speed. Cronbach’s α was calculated to assess internal consistency between the speeds tested. An α within .7 and .9 indicates good consistency between items, while values greater than .9 were checked for redundancy. If a movement speed was repetitive, it would be removed. This analysis was conducted for each joint and visit separately. A composite score was derived to represent kinesthesia sense by computing the average movement detection score across speeds. Test-retest reliability was used to establish the consistency of participant’s kinesthesia sense between sessions. Intraclass correlation coefficients (ICCs) of the composite scores were calculated for all joints. Test-retest reliability was considered strong if the ICC was >0.90. All statistical analysis was performed using SPSS 22.0 (IBM Corp, Armonk, NY, USA).
Results
Demographic and clinical characteristics of participants with SCI are summarized in Table 1. The mean (± SD) age of the control participants was 39.5 ± 9.7 years. Mean (± SD) reaction time to the cutaneous stimuli delivered to the cheek was 334 ± 100 ms for the SCI group and 268 ± 34 ms for the control group (P = .016). Reaction time to the cutaneous stimuli at the cheek was not correlated with the movement detection score for each joint (P = .147). Table 2 presents the average movement detection score across each speed and for the SCI groups based on ISNCSCI classification and controls. Participants classified as AIS B showed greater kinesthesia deficits at the hip and knee joints compared with individuals classified as AIS D (P < .001), and able-bodied controls (P < .001). No difference was found between AIS B compared with AIS C (P > .05). The AIS C group showed greater kinesthesia deficits compared with the AIS D group for the hip and knee joints (P < .05). The average score for AIS D participants was not different from the control group at the hip (P = .307) but was significantly higher at the knee (P = .022).
Summary of Participant Demographic and Clinical Data. a
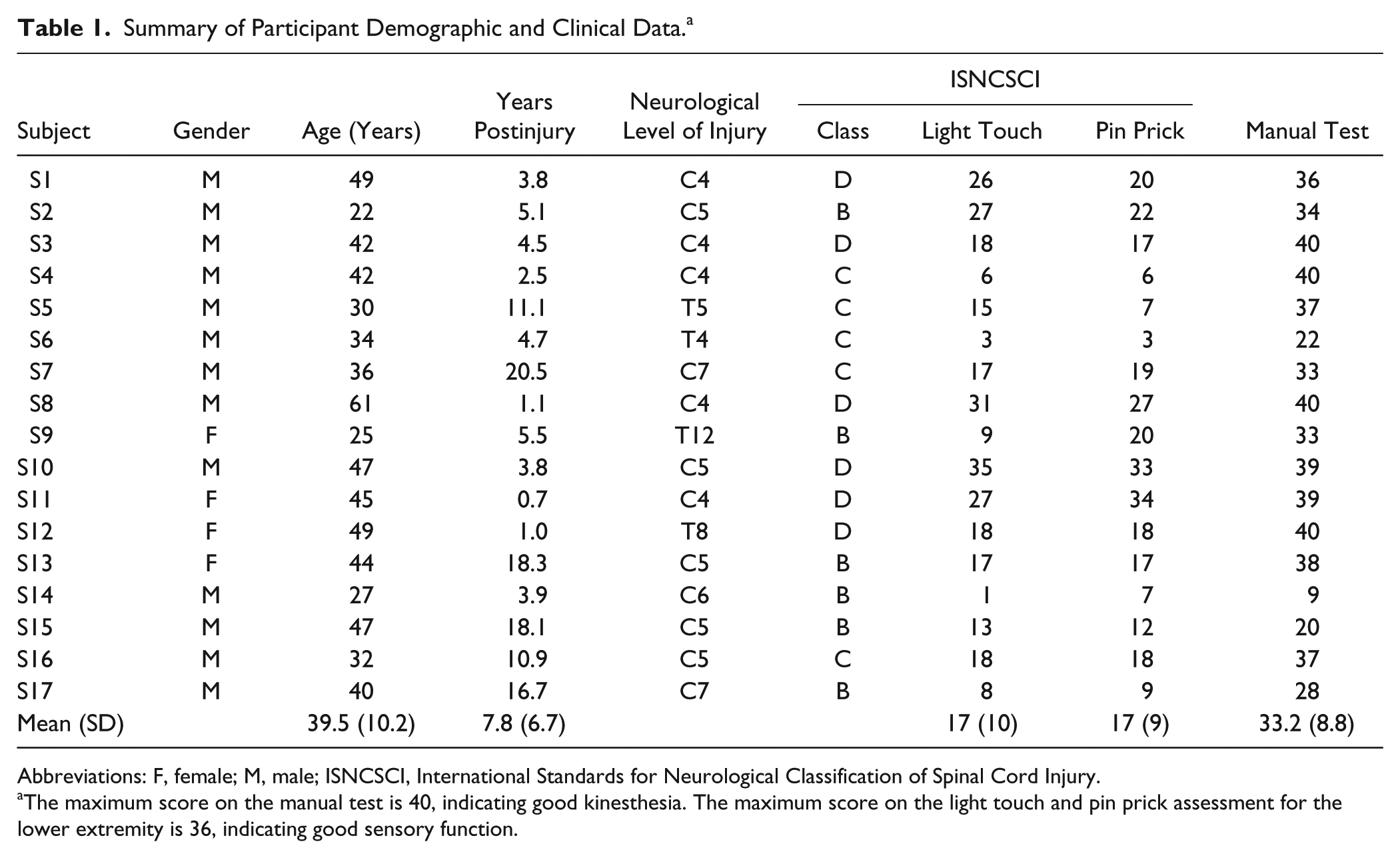
Abbreviations: F, female; M, male; ISNCSCI, International Standards for Neurological Classification of Spinal Cord Injury.
The maximum score on the manual test is 40, indicating good kinesthesia. The maximum score on the light touch and pin prick assessment for the lower extremity is 36, indicating good sensory function.
Summary of the Movement Detection Scores Across AIS Levels. a
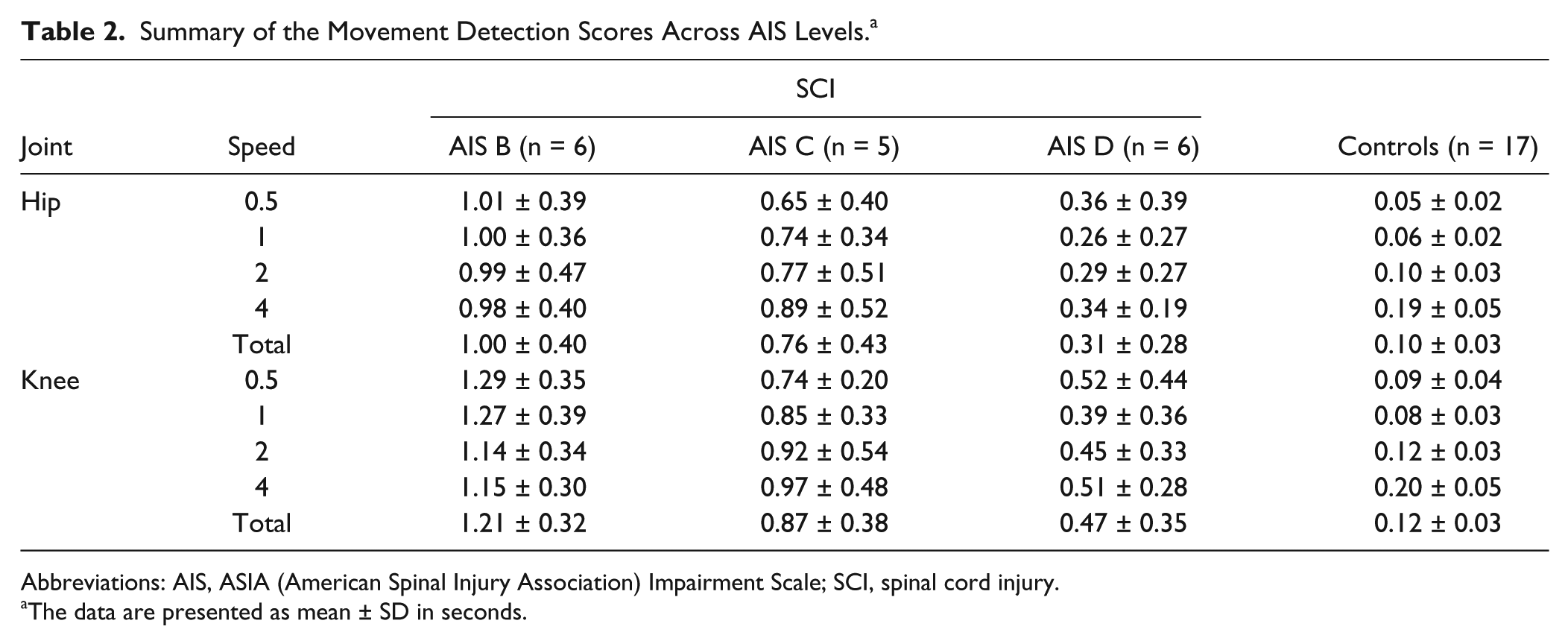
Abbreviations: AIS, ASIA (American Spinal Injury Association) Impairment Scale; SCI, spinal cord injury.
The data are presented as mean ± SD in seconds.
Validity
Average movement detection scores across the Lokomat speeds for the hip and knee joints are shown in Figure 2. There was a significant main effect of group, joint and speed. Reaction time to the cheek stimulus was not correlated with the movement detection score (r = 0.574, P = .147), and did not show a significant effect as a covariate (P = .541); therefore, it was removed from the model. The SCI group (0.751 ± 0.084) had larger movement detection scores compared with the control group (0.127 ± 0.086; P < .001). The movement detection score at the hip joints (0.393 ± 0.058) were less than those at the knee joints (0.486 ± 0.058; P < .001) for both groups. Contrasts show no difference between the left and right sides for both the hip and knee joints (P < .05). As movement speed decreased, the movement detection score significantly decreased from 0.495 ± 0.058 at 4.0 deg/s to 0.419 ± 0.058 at 0.5 deg/s (P < .001). The movement detection score at 0.5, 1.0, and 2.0 deg/s speeds were significantly lower than the fastest speed (P < .001). There was no significant difference between visits for both groups (P = .169).
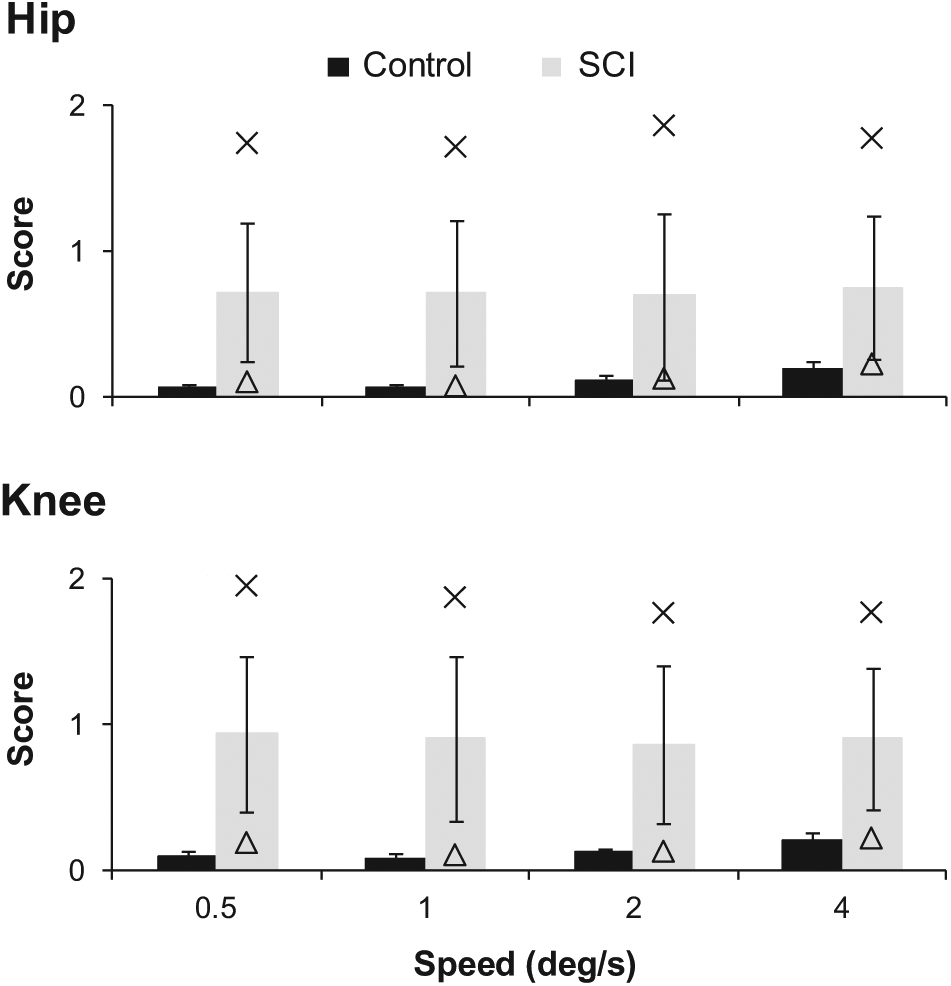
Average movement detection scores are presented for the spinal cord injury (SCI) and control groups across each speed tested at the hip and knee joints. Individual data representing the lowest (triangle) and highest (×) scores in the SCI group is also plotted. Error bars represent standard deviations (SD). Results from the repeated-measures analysis of variance show significant main effects for group, joint, and speed (P < .001).
The group * joint interaction effect showed a larger difference in the movement detection score between the hip and knee joints in the SCI group compared with the control group (average difference between joints is 0.022 for controls and 0.164 for SCI, P < .001). A significant group * speed interaction effect demonstrates a larger difference in the movement detection score between the fastest and slowest speeds for the SCI group (0.781 ± 0.084 at 4.0 deg/s and 0.75 ± 0.084 at 0.5 deg/s) compared with control (0.208 ± 0.087 at 4.0 deg/s and 0.087 ± 0.087 at 0.5 deg/s; P < .001). At the fastest speed (4.0 deg/s), there was no difference between groups (P > .05). The interaction effect between joint and speed was not significant (P = .732). All 3-way interactions and 2-way interaction effects with visit were not significant (P > .05).
Relationship Between Robotic and Manual Kinesthesia Tests
The movement detection scores were negatively related to the manual kinesthesia scores for all the joints (left hip, r = −0.71, P = .002; left knee, r = −0.86, P < .001; right hip, r = −0.47, P = .056; right knee, r = −0.57, P = .017; Figure 3). The plots also reveal a ceiling effect in the manual test. Eight to 9 subjects (47% to 53% of the participants) scored the maximum possible score of 10/10 during manual testing of the hip and knee joints.
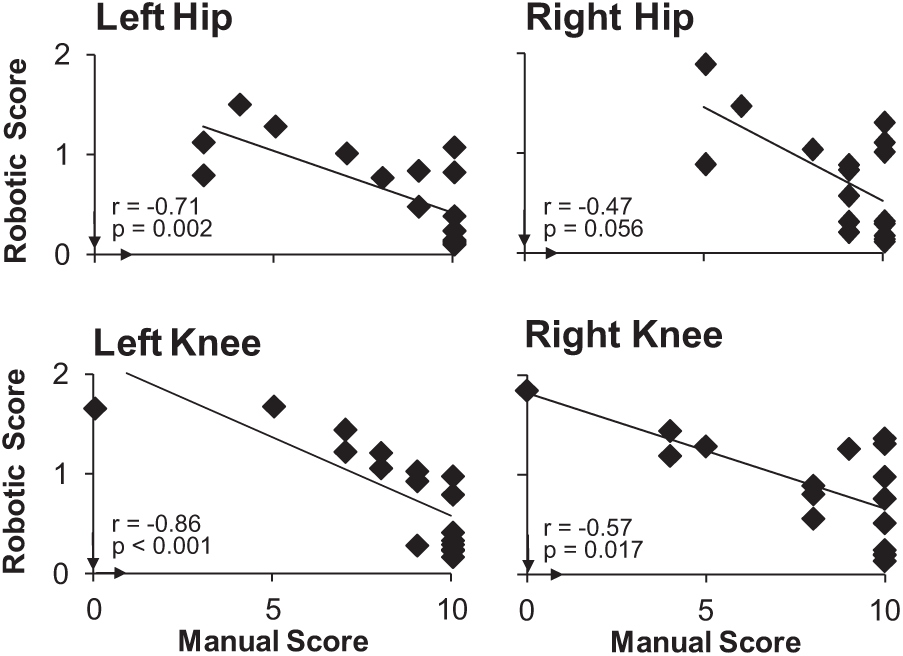
Movement detection scores measured by the robotic kinesthesia test were plotted against the corresponding manual proprioceptive test scores at each joint (number of correct responses out of 10 trials indicating the direction of movement). The arrows on the x- and y-axis represent the direction of better kinesthesia sense. A ceiling effect was noted for all the manual test scores.
Relationship Between Robotic Test and ISNCSCI Sensory Score
Correlations between total movement detection score and ISNCSCI lower extremity sensory score showed a negative relationship for light touch (r = −0.48, P = .048), and pin prick (r = −0.54, P = .026).
Internal Consistency
The movement detection score showed excellent internal consistency between the speeds tested for the hip and knee joints of both groups (Table 3). Removal of the 4.0 deg/s speed would slightly decrease the α value and reduce the redundancy between speeds for all joints in the SCI group (Table 3) since the movement detection score at this speed was not significantly different compared with the control group. Interitem correlation coefficients ranged from 0.48 to 0.97, indicating a moderate to strong relationship between the speeds tested.
Internal Consistency Statistics Based on Cronbach’s α. a
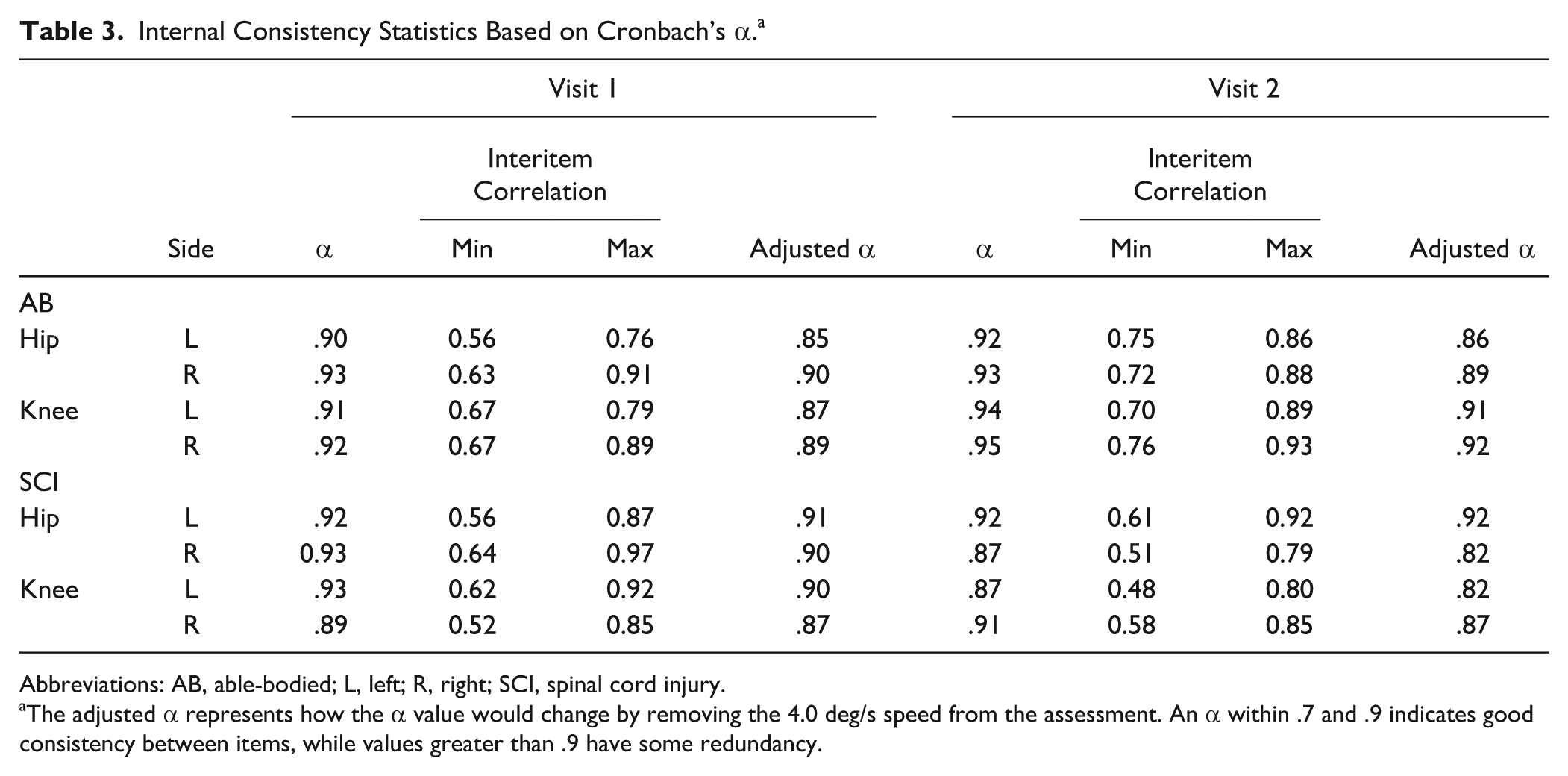
Abbreviations: AB, able-bodied; L, left; R, right; SCI, spinal cord injury.
The adjusted α represents how the α value would change by removing the 4.0 deg/s speed from the assessment. An α within .7 and .9 indicates good consistency between items, while values greater than .9 have some redundancy.
Test-Retest Reliability
Very high test-retest reliability was found in the control group for the composite movement detection score of the hip (ICC = 0.88 left, ICC = 0.94 right; P < .001) and knee (ICC = 0.90 left, ICC = 0.91 right; P < .001; Figure 4). The SCI group also showed high test-retest reliability at the hip (ICC = 0.97 left, ICC = 0.96 right; P < .001) and knee (ICC = 0.95 left, ICC = 0.96 right, P < .001; Figure 4).
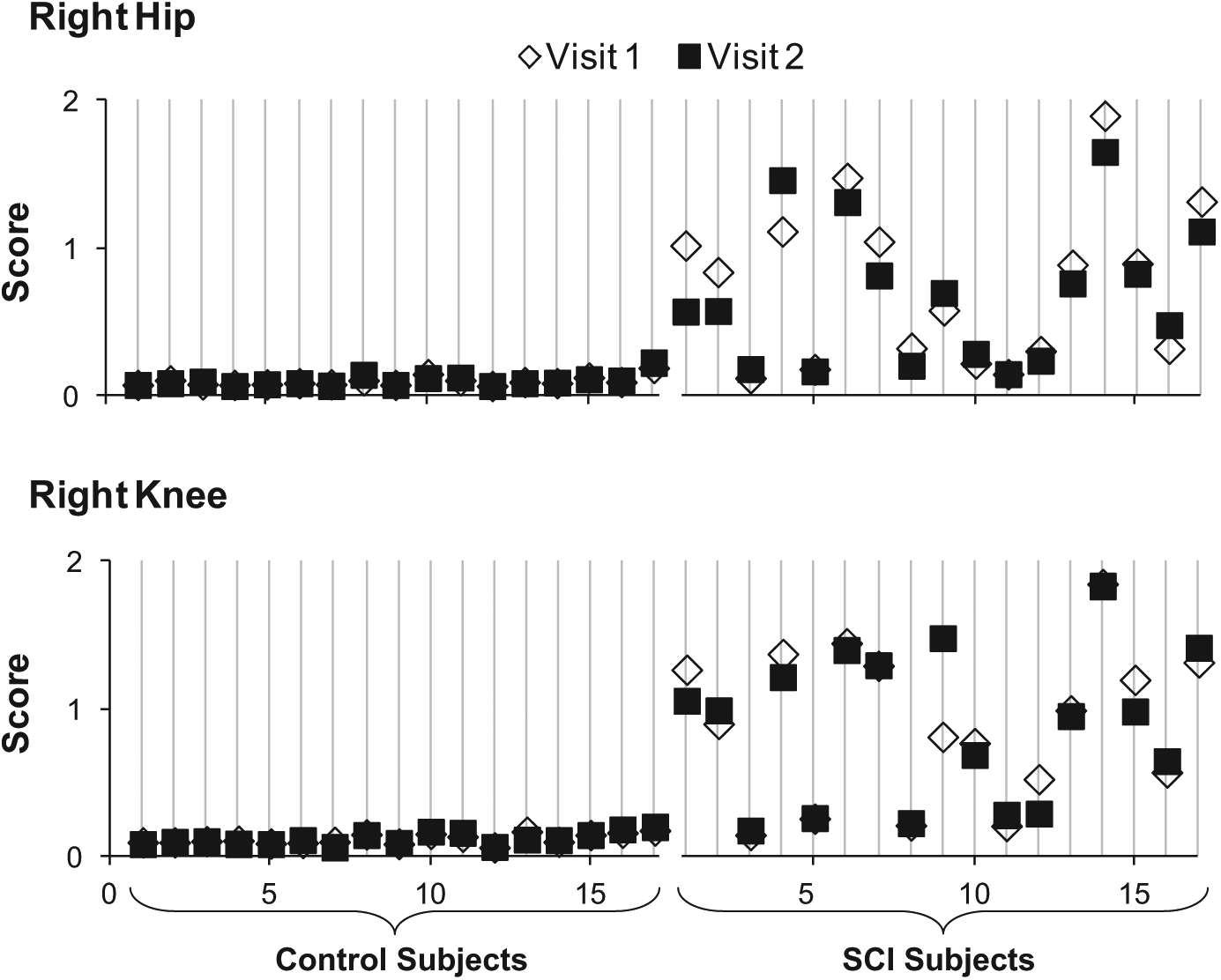
The data presented show an example of the test-retest reliability for the right hip and knee joint. Average movement detection score is plotted for both visits; conducted at least 1 week apart. Both groups showed high test-retest reliability at the hip (control, ICC = 0.94; SCI, ICC = 0.96; P < .001) and knee (control, ICC = 0.91; SCI, ICC = 0.96; P < .001) joints. ICC, intraclass correlation coefficient; SCI, spinal cord injury.
Discussion
We found that many of the participants with SCI we tested have impaired lower limb kinesthesia sense, and that these deficits were more pronounced among those with motor-complete injuries (AIS B) and lower ISNCSCI sensory scores. Our assessment also indicates greater sensory impairment at the knee joint compared with hip joint. Importantly, these findings were repeatable as no differences were found between visits. These findings indicate that our novel robotic-based assessment is a valid and reliable tool to quantify lower limb kinesthesia deficits in the SCI population. Furthermore, our robotic-based tool may be more sensitive compared with the manual test of kinesthesia due to the better degree of precision available and lack of a ceiling effect. Development of a reliable and valid assessment tool represents an important step in rehabilitation to improve our ability to objectively monitor and track changes in sensory function following SCI.
The use of robotic technology for functional assessments in addition to training can offer many benefits to neurological rehabilitation.26-28 We were able to take advantage of the Lokomat’s features to implement a passive movement at a controlled speed at one joint while stabilizing all other lower limb joints. This is important as active movements are influenced by the degree of motor impairment and may engage synergistic movements at other joints. In addition, responses to the slowest speed show a greater difference between individuals with SCI and able-bodied controls. While it appears that the slowest speed may be more sensitive to differences in kinesthesia abilities and possibly better to detect changes in response to therapy, faster speeds such as 1.0 and 2.0 deg/s may be useful indicators of change for individuals with severe impairments that otherwise might be overlooked if only the 0.5 deg/s speed was used. For instance, one participant was unable to detect any movements at the slowest speed, whereas movement was felt at the higher speeds. Even though these speeds are slower compared with those that occur in a gait cycle for able-bodied adults, we were able to measure with fine precision the level of kinesthesia abilities in able-bodied adults and individuals with SCI. Considerations in regards to the movement speed also has important clinical implications as current manual assessments cannot control for speed, therefore limiting how much the results can provide valid and meaningful information. The lack of ability to regulate speed when performing a manual test limits the sensitivity of the measure to detect changes that may occur with recovery. 14 Since robotic-based measures are inherently objective, they are better able to deliver results that can lead to improved clinical interpretations and guide decision-making for treatment.
While previous studies have used robotic technology to conduct proprioceptive assessments,6,15-20 this is the first time a kinesthesia-based approach has been applied to the lower limb in a neurological population. Upper extremity robotic devices, such as the KINARM and InMotion, have been used to evaluate joint position and kinesthesia sense among individuals with stroke,15,22 traumatic brain injury, 18 and children with cerebral palsy. 19 We have also previously demonstrated that the Lokomat can be used as an objective tool to characterize static joint position sense in the lower limb among people with SCI.20,21 Other approaches to measure lower limb kinesthesia have found that the threshold to detection of passive motion paradigm is valid and reliable at the knee joint for people with orthopedic injuries and matched uninjured controls.29,30 Much of the orthopedic literature has focused on kinesthetic assessments at the knee because it has been found to be predictive of impairment and functional performance in patients with knee injury or disease. 29
We have found that lower limb kinesthesia is moderately associated with other aspects of impaired sensory function, particularly light touch and pinprick sensation. Sensory testing is routinely performed as a part of the ISNCSCI examination. Together with a motor examination the level of completeness is then classified with the ASIA Impairment Scale. 10 However, proprioceptive sense represents only an optional component of the ISNCSCI, measured as joint position sense at the great toe. 10 Current clinical sensory evaluations may overlook critical information as several studies point to the critical contribution of lower limb proprioceptive sense on balance and gait function.3,4 Recent studies have also indicated an association between impaired proprioception and upper limb functional recovery following neurological injury.6,31-33 Dukelow et al 6 reported a relationship between upper limb joint position sense and functional abilities in individuals with stroke, thus indicating that impaired joint position sense impacts performance of daily activities. New robotic-based approaches have many advantages, including objectivity and reliability to provide meaningful information on functional recovery that may otherwise be overlooked with coarse ordinal scales.
As robotic technology becomes more available to rehabilitation centers, we have promising opportunities to establish objective measures of sensory function for people with neurological injuries. Our lower limb kinesthesia assessment was feasible to conduct with individuals who had a wide range sensorimotor impairment. A robotic-based assessment of kinesthesia can address limitations of manual testing, such as a difficulty in standardizing the speed of movement, variations in points of contact on the individual, and quantification of the response. However, there are some practical drawbacks to implementing our robotic assessment in clinical practice, such as time requirements for set-up and the availability of devices. Nevertheless, the concepts and methodological techniques developed by studies investigating robotic-based assessments will be useful for developing alternative lower-cost options for clinical practice.
Implementing a robotic-based kinesthesia assessment in a clinical setting may also lead to the development of new therapies and validation of current therapies to enhance sensory function. For example, task-specific gait training that provides repeated practice of walking movements has been shown to improve walking function34-36; however, the contribution of kinesthesia to functional recovery has yet to be considered. Given the key role of proprioception in locomotor control, 2 it likely influences the extent of improvement possible with gait training. In addition, recent reviews have yet to identify therapies that specifically target kinesthesia.37,38 Another important research directions will be to track long-term changes in kinesthesia ability during rehabilitation after SCI, and understand the relationship to changes in motor function.
This robotic-based proprioceptive assessment is not without limitations. The straps from the leg cuffs and body-weight support harness may provide additional cutaneous feedback to the participant about movement at the joint. In particular, differences observed between the hip and knee joints may partially reflect more cutaneous input from the harness straps around the groin and sacral pad. However, we ensured that the cuffs were tightly secured around the legs to minimize movement between the cuff and skin. Participants were provided with rest breaks between testing each joint to reduce any effects from being suspended for a long period of time. We also used a platform to allow the participant to bear weight on the opposite leg being tested to reduce weight supported by the harness. Although auditory stimuli were not blocked during the assessment, we do not believe it provided subjects with additional cues, as the measured noise level did not change when the Lokomat legs were moving. We did not use a quantitative value in the manual test to measure the degree of movement. However, the method of scoring we used for the manual tests closely reflects clinical practice that typically uses a verbal response from a selection of choices (eg, “up” or “down”).
Conclusion
The present study established a valid and reliable robotic-based approach to quantify lower limb kinesthesia deficits in the SCI population. A measure of kinesthesia may provide additional information not captured in standard evaluations of sensory function to guide the selection of therapeutic interventions. This study also points to the importance of developing tools to better characterize sensory deficits and track recovery after SCI.
Footnotes
Acknowledgements
Thank you to our volunteers (Raed Alamro, Megan Brousseau, and Rachel Cote) who helped with data collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the International Foundation for Research in Paraplegia. TL was supported by a Canadian Institutes of Health Research New Investigator Award.
