Abstract
Background. Crossed cerebellar diaschisis is the disruption of functional connectivity between cerebrum and cerebellum after hemispheric unilateral brain lesions. In adults and to a lesser extent in children, crossed cerebellar diaschisis has been largely investigated by functional connectivity and demonstrated to influence paretic hand function. Objective. We aim to demonstrate a disruption in structural corticopontocerebellar (CPC) connectivity in children with congenital brain lesions and examine its correlation with paretic hand motor function. Methods. Thirty-six children (Manual Ability Classification System: I, n = 21; II, n = 15) with unilateral brain lesions and 18 controls were analyzed in a case-control study, and diffusion magnetic resonance imaging data were acquired at 3T. High angular resolution diffusion imaging probabilistic tractography was employed for the region of interest–based reconstruction of CPC tracts. To identify statistical differences in structural cerebrocerebellar connectivity between case and control groups, an asymmetry index based on the number of streamlines of CPC tracts was used. In the case group, the correlation between asymmetry index and hand function measures was also determined. Results. Projections through the middle cerebellar peduncle to the contralateral cerebral cortex showed greater asymmetry in children with congenital unilateral brain lesion compared to controls (P = .03), thus indicating a disruption of structural cerebrocerebellar connectivity. The degree of asymmetry index showed a correlation (P < .03; r = −0.31) with impaired hand abilities in bimanual tasks. Conclusions. Disruption of structural cerebrocerebellar connectivity is present in patients with congenital unilateral brain injury and might be related to impaired hand function in bimanual skills, with potential implication in tailoring early intervention strategies.
Keywords
The corticopontocerebellar (CPC) projections in human and nonhuman primates take part to a loop system that connects each cerebral hemisphere with the contralateral cerebellar hemisphere and returns via the thalamus.1,2 The CPC tracts consist of corticopontine fibers arising from the cerebral cortex, mostly from the precentral and the postcentral gyrus,1-3 and projecting to the pons, relaying with pontocerebellar fibers that cross the midline to the contralateral middle cerebellar peduncle (MCP). 3
In children and adults with cerebral stroke, an alteration of functional connectivity between the lesioned cerebral hemisphere and the contralateral cerebellar hemisphere has been reported,4-10 reflecting the disruption of CPC connections. 11 This phenomenon has been defined as crossed cerebellar diaschisis (CCD), indicating the focal depression of neuronal activity at a distant site (cerebellar hemisphere) from the original site of injury (contralateral cerebral hemisphere) due to the presence of structural connections. 12 As a long-term effect of CCD, an atrophy of the cerebellar hemisphere contralateral to the side of the supratentorial lesion has been reported on structural magnetic resonance imaging (MRI) in children and adults.10,11,13,14 Less is known on the effects of CCD on structural CPC connectivity.5,15
Lack of structural corticocerebellar connectivity studies might be in part related to the limitation of more traditional diffusion tractography approaches to investigate more complex polysynaptic angulated networks. 16 The availability of novel approaches, such as high angular resolution diffusion imaging (HARDI), improved the resolution of crossing and kissing fiber bundles over diffusion tensor imaging–based approaches and encouraged the study of anatomic crossroads, such as the brainstem and other posterior fossa structures, including the CPC tracts. 16 The characteristics of the cerebellar networks, particularly in the absence of a primary cerebellar lesion, have been very poorly investigated in cerebral palsy (CP) in spite of their potential key clinical role. 17 In fact, as a result of the phylogenetic increasing demands in motor coordination, in humans the cerebellum significantly contributes to motor learning, complex hand movement execution, and intended motor goal prediction. 18
The first purpose of the current study is to validate the use of HARDI with constrained spherical deconvolution and probabilistic tractography to define cerebrocerebellar connectivity through a region of interest (ROI)–based approach in a group of children with typical development. If brain diffusion tractography correctly characterizes CPC, it should be able to detect the known crossed connections between M1 and the contralesional cerebellum, showing no significant differences between right- and left-seeded bundles.
The second purpose of the current study is to determine the pattern of crossed structural connectivity of CPC tracts in children with CP due to unilateral brain lesion, in comparison to typically developing children. This purpose is addressed by studying the asymmetry of the tracts arising from the precentral and postcentral gyrus, via the posterior limb of internal capsule, to the contralateral cerebellum through the MCP. The contribution of relevant variables, such as timing, side, and severity of brain lesion, is also determined.
The third purpose of the study is to determine the relationship between disruption of corticocerebellar connectivity and hand function in the CP cohort. This purpose is addressed by correlating the degree of asymmetry of CPC tracts with quantitative measures of unimanual and bimanual function.
Methods
Participants
Children with congenital hemiplegia were recruited as part of an ongoing cohort study at the Queensland Cerebral Palsy and Rehabilitation Research Centre, which includes structural and diffusion 3T MRI data acquisition.19,20 All children showing evidence of unilateral brain lesion on structural MRI were selected for this study. Typically developing children were also recruited as controls. The University of Queensland and Children’s Health Queensland ethics committees granted ethical approval. Informed parental consent was obtained for all participants.
MRI Acquisition
MRI data were acquired with a 3T Siemens Tim Trio scanner (Siemens, Erlangen, Germany), using a 12-element Tim head array. A T1 high-resolution structural image was acquired using a 0.9-mm isotropic 3-dimension magnetization-prepared rapid gradient echo sequence. The imaging parameters were as follows: field of view, 23 × 23 × 17.3 cm; time to repeat (TR), 1900 ms; time to echo (TE), 2.32 ms; time to invert, 900 ms; flip angle, 9°. Magnetization-prepared rapid gradient echo acquisition time was 4.5 minutes. A T2-weighted turbo inversion recovery magnitude image was acquired for lesion classification (25 axial slices; 4-mm slice thickness with 1.2-mm gap; field of view, 22 × 22 cm; TR, 7000 ms; TE, 79 ms; time to invert, 2500 ms; acquisition matrix 256 × 192 interpolated to 512 × 512). The acquisition time for the turbo inversion recovery magnitude was 2 minutes.
Diffusion images were acquired using a commercial single-shot echo planar multidirection diffusion-weighted sequence, employing a dual bipolar diffusion gradient and a double spin echo. The imaging parameters were as follows: 60 axial slices; 2.5-mm slice thickness; field of view, 30 × 30 cm; TR, 9500 ms; TE, 116 ms; acquisition matrix, 128 × 128, resulting in an in-plane resolution of 2.34 × 2.34 mm. Parallel imaging was employed with an acceleration factor of 2 to reduce susceptibility distortions. Sixty-four diffusion-weighted images were acquired at b = 3000 s/mm2, in which the encoding gradients were distributed in space using the electrostatic approach, 21 along with 1 minimally diffusion-weighted image (b = 0). A field map for diffusion data was acquired using two 2-dimensional gradient-recalled echo images (36 axial slices; 3-mm slice thickness with 0.75-mm gap; field of view, 19.2 × 19.2 cm; TR, 488 ms; TE1, 7.38 ms; TE2, 4.92 ms; acquisition matrix, 64 × 64) to assist in the correction for residual distortions due to susceptibility inhomogeneities. The combined acquisition time for diffusion data and field map was 10 minutes.
Structural Images Analysis
Structural images were assessed by a MRI-trained child neurologist (S.F.). Side, severity, and presumed timing of brain lesions were determined.22,23 In addition, all images were specifically checked for visual asymmetries in cerebellar white or grey matter volumes.
Presumed timing of brain damage was classified according to the Krageloh-Mann qualitative classification system 23 as follows: brain maldevelopments, periventricular white matter lesions, cortical and deep gray matter lesions, or miscellaneous. Severity of brain lesion was systematically assessed by means of a specific visual semiquantitative scale for children with CP. 22 This scale has high interrater reliability (intraclass correlation coefficient, 0.92) and intrarater reliability (intraclass correlation coefficient, 0.91). The hemispheric score for each hemisphere was calculated as the sum of frontal, parietal, temporal, and occipital lesion scores on that side, with a maximum possible hemispheric severity score of 12 (higher scores representing larger injuries). The basal ganglia and brainstem score for each side was calculated as the sum of lenticular, caudate, thalamus, posterior limb of internal capsule, and brainstem lesion score, with a maximum possible severity score of 5 (higher scores representing larger injuries). The hemispheric score and the basal ganglia and brainstem score on each side were included in the analysis as independent determinants of brain lesion severity at the supratentorial and subcortical level, respectively.
dMRI Analysis and Fibers Tracking
An extensive preprocessing procedure was followed to detect and correct for image artifacts caused by involuntary head motion, cardiac pulsation, and image distortions. 24 Image volumes with within-volume movement were detected using the discontinuity index 25 and excluded from further analysis. Image distortions caused by susceptibility inhomogeneities were reduced using the field map, employing tools available with FSL, 26 and intensity inhomogeneities were removed using n3 correction. 27 Subsequently, signal intensity outlier voxels (caused by cardiac pulsation, bulk head motion, and other artifacts) were detected and replaced using detection and replacement of outliers prior to resampling, 28 which was modified from the originally proposed method to employ a higher-order model of the diffusion signal suitable for the detection and replacement of outliers in high b-value diffusion data. 25 Between-volume registration to account for head movement during the scan time performed using “fit the model to all the measurements” 29 with adjustment of the b-matrix. 29 Constrained spherical deconvolution31,32 was employed to estimate the fiber orientation distribution for tractography at maximum harmonic order 8. A short-tracks color-encoded track-density image (TDI) was generated using 5 million streamlines of 2-cm maximum length seeded throughout the entire brain volume. Short-tracks TDIs provide a more balanced image compared to TDIs calculated without length constraint.
To generate the CPC tracts of interest, 2 ROIs were outlined on the color-coded TDI. The CPC originating from the precentral and postcentral areas descend close to the corticospinal tract in the centrum semiovale and in the ipsilateral posterior limb of internal capsule. From the posterior limb of internal capsule, the selected CPC tract passes in the middle portion of the cerebral peduncle and crosses through the contralateral MCP in the basis pontis. For both right and left sides, ROI 1 was traced on the MCP. The MCP was identified as the green area (front-back fibers direction) on the coronal plane of the color-coded TDI, at the level showed in Figure 1A. For both right and left sides, ROI 2 was traced on the posterior limb of internal capsule. This structure was identified as the blue area (top-down fibers direction) on the axial plane of the color-coded TDI, at the level shown in Figure 1B. To extract the CPC tract, ROI 1 on one side was seeded with 1 million streamlines, and the “INCLUDE” operation was applied for the contralateral ROI 2. Some “EXCLUDE” operations were also used to exclude fibers passing through the cerebellar peduncle homolateral to ROI 2 and on the midsagittal plane corresponding to cerebellar vermis. Tracts were checked by 2 experienced raters (S.F. and A.G.) on all subjects to verify trajectory and anatomic landmarks described in atlases of human white matter and to check false-positive streamlines within the CPC pathways. Tracts were excluded from the analysis if the number of streamlines was <10. Examples of the generated pathways in children with CP and controls are shown in Figure 2.
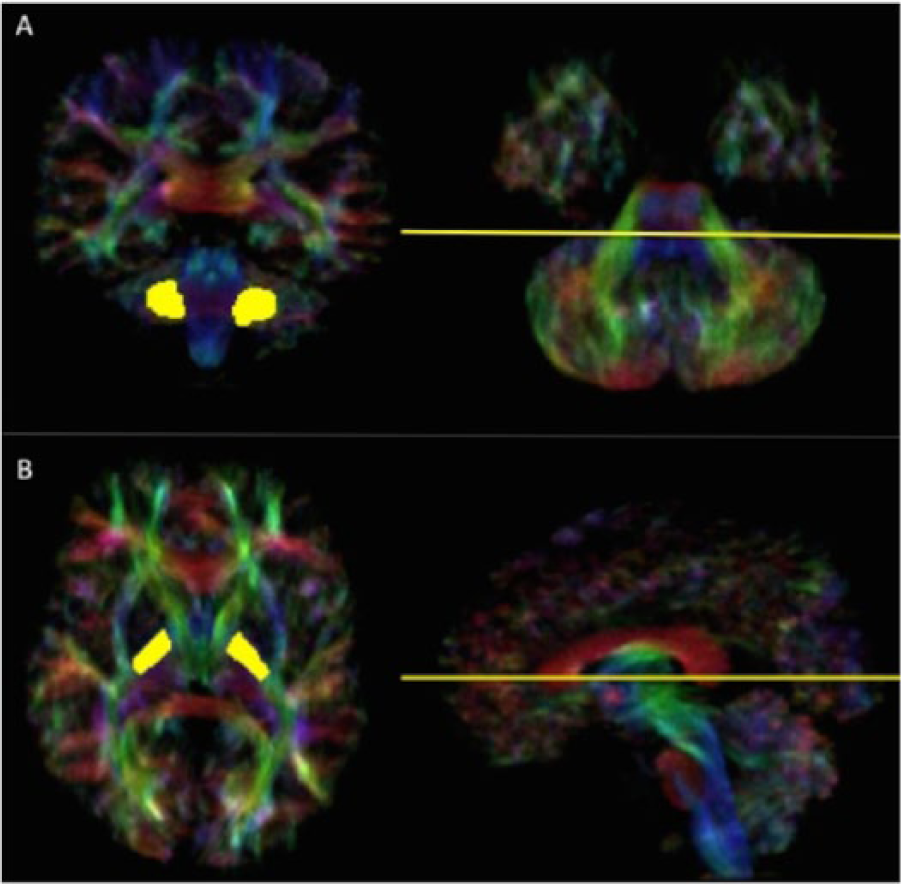
A, Region of interest 1 traced in the coronal plane (left) on the right and left middle cerebellar peduncle at the level identified in the axial plane (right) of the track-density image map. B, Region of interest 2 traced in the axial plane (left) on the right and left posterior limb of internal capsule at the level identified in the sagittal plane (right) of the track-density image map.
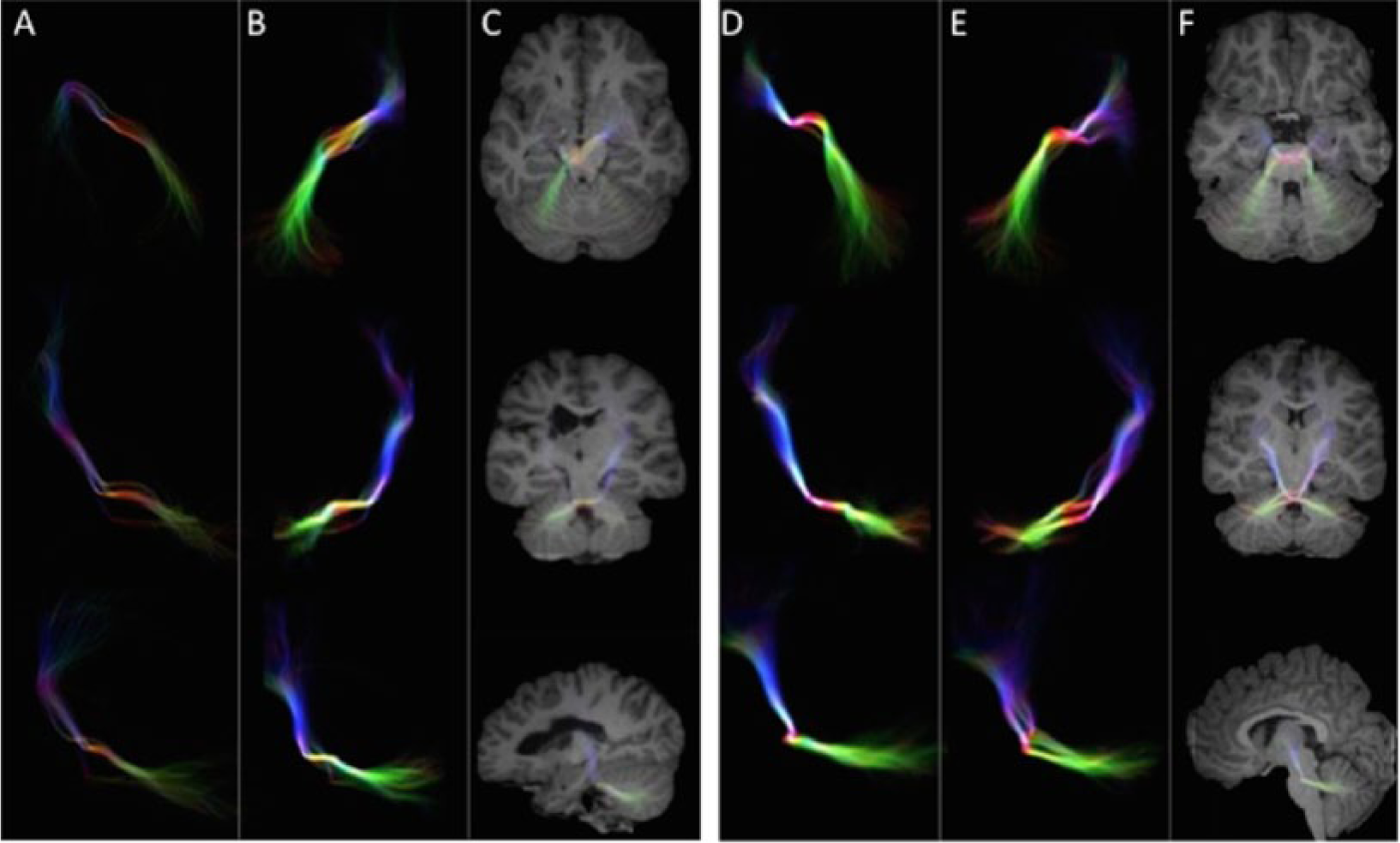
A, From top to bottom, 2-dimensional axial, coronal, and sagittal appearance of corticopontocerebellar tracts arising from the right cerebral hemisphere in a child with cerebral palsy. B, Tract arising from the left cerebral hemisphere in the same patient. C, Two-dimensional synthetic representation of both tracts on the 3 planes superimposed to T1 images in the same subject. D, From top to bottom, 2-dimensional axial, coronal, and sagittal appearance of corticopontocerebellar tracts arising from the right cerebral hemisphere in a child with typical development. E, Tract arising from the left cerebral hemisphere in the same patient. F, Two-dimensional synthetic representation of both tracts on the 3 planes superimposed to T1 images in the same subject. Note the asymmetrical pattern in the patient with cerebral palsy (A-C).
Clinical Testing
Hand function measures were collected for included patients as part of ongoing cohort studies of children with congenital hemiplegia. The use of the hemiplegic hand as an assisting hand was assessed with the Assisting Hand Assessment 33 (AHA) for the effective use of impaired hand in bimanual activities. Unimanual abilities were assessed for the function of the impaired upper limb with the Melbourne Assessment of Unilateral Upper Limb Function 34 (MUUL). Speed and dexterity of impaired limb were assessed with the Jebsen-Taylor Test of Hand Function 35 (JTTHF).
Data Analysis and Statistics
An individual asymmetry index (AI) for the CPC tracts in both case and control groups was generated, since the raw number of streamlines cannot be considered as an absolute quantitative measure for statistical analysis. For the control group, the AI was calculated as follows: (R – L) / (R + L), where R and L represent the number of streamlines from the right and left sides of ROI 1, respectively. The closer to 0 the AI, the more symmetrical the distribution of streamlines arising from one hemisphere compared to the contralateral. For the CP group, the AI was calculated as follows: AI = (C – I) / (C + I), where C and I represent the number of streamlines from the contralesional and ipsilesional hemispheres, respectively, to the contralateral cerebellum. 34 The closer to 1 the AI, the bigger the relative reduction of streamlines arising from the ipsilesional hemisphere compared to the contralesional side for the CPC tract.
For aim 1, the validity of the HARDI ROI-based approach to detect cerebrocerebellar connectivity was tested in the group of children with typical development based on the predicted symmetrical crossed distribution of CPC tracts. 3 We tested the hypothesis that the CPC bundle seeded from ROI 1 and passing through contralateral ROI 2 was not significantly different from the contralateral in terms of number of streamlines. A paired-sample t test was performed to test the null hypothesis that AI in the control group was different from 0.
For aim 2, a 2-tailed t test was used to determine if the AI was significantly different between cases and controls. To minimize type II statistical error, the sign of AI in the control group was removed for this analysis.
For aim 3, the Pearson product-moment correlation was used to determine the relationship between the AI and hand function measures (AHA, MUUL, JTTHF). To assess the contribution of timing and side of the lesion, a t test was used to determine differences within subgroups of periventricular white matter versus cortical or deep gray matter and right versus left, respectively. To assess the contribution of the severity of the lesion, a linear regression was used to determine the correlation of the AI with the hemispheric score and the basal ganglia and brainstem score on the side of brain lesion.
Statistical analysis was performed using SPSS version 20; all P values were 2-tailed; and statistical significance was defined as P < .05.
Results
Forty children with unilateral CP and evidence on structural MRI of unilateral brain lesion were selected, together with 20 controls. Three patients and 2 controls were excluded due to excessive head movement artifacts, signal-to-noise problems on the MRI acquisition, or an inadequate number of fibers identified by the technique. One patient was excluded due to the lack of clinical data. Demographic data, MRI scores, AIs, and functional scores of the children in the final sample are reported in Table 1. Of the 36 CP subjects, 15 presented with right brain lesions and 21 with left brain lesions. Eleven patients presented cortical and deep gray matter lesions, while 25 presented periventricular white matter lesions (ie, venous infarction). No MRI was classified as brain maldevelopment or miscellaneous. No asymmetries in cerebellar white or gray matter volumes were detected on visual inspection.
Participants Characteristics. a
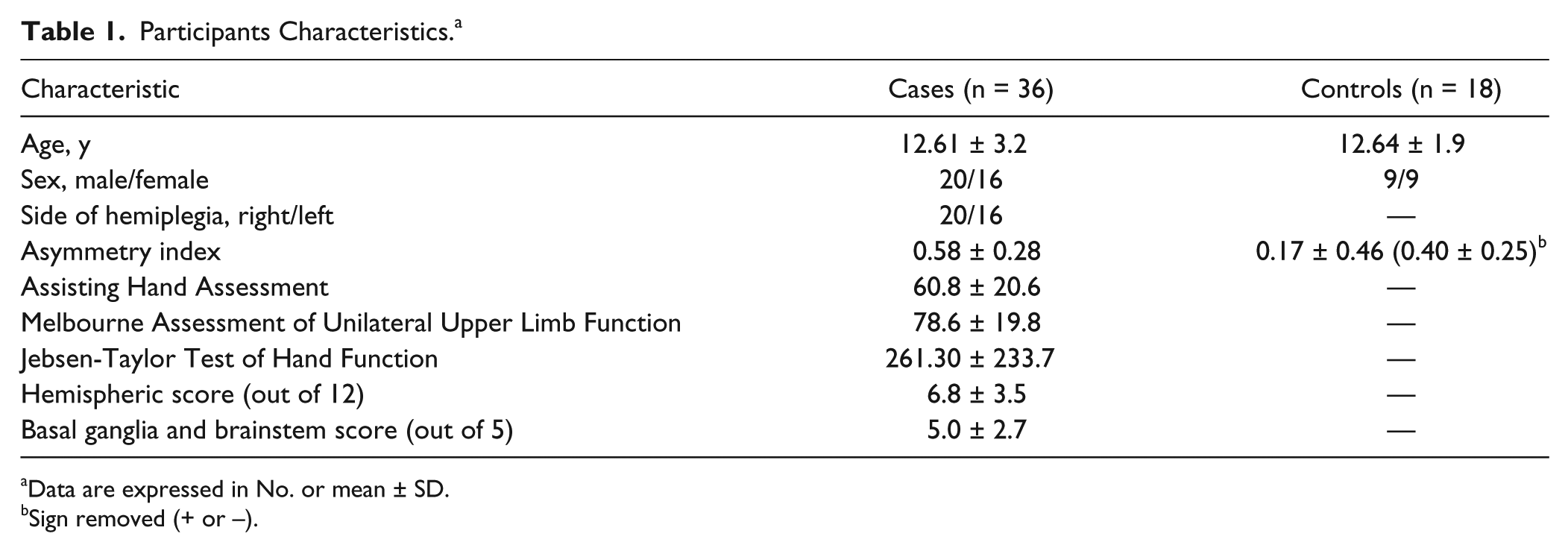
Data are expressed in No. or mean ± SD.
Sign removed (+ or –).
Validation of the Approach
Crossed CPC bundles were extracted in all children with typical development. The paired-samples t test revealed no significant differences, allowing us to reject the null hypothesis that the AI in the control group was significantly different from 0.
Differences in CPC AI Between CP Subjects and Controls
The 2-tailed t-test analysis revealed a difference between AI of cases and controls (P < .03), with a greater degree of asymmetry in the CPC tract found in children with CP. Within the CP group, no significant differences were found when grouping the subjects according to side (P = .246) or timing of brain lesion (P = .955). No correlation was found between CPC AI and the hemispheric scores on the side of brain lesion on the semiquantitative scale (P = .904). A significant correlation was found between CPC AI and the basal ganglia and brainstem score on the side of brain lesion (P = .003), with a greater degree of asymmetry in the CPC tract related to a more severe score.
Correlation Between CPC AI and Hand Function Measures
The correlation analysis between AI and AHA scores in the CP group revealed a significant (P = .034) correlation (r = −0.31). No correlation was found between AI measures and MUUL (P = .118) and JTTHF (P = .208) scoring for any group. Scatter plots are provided in Figure 3.
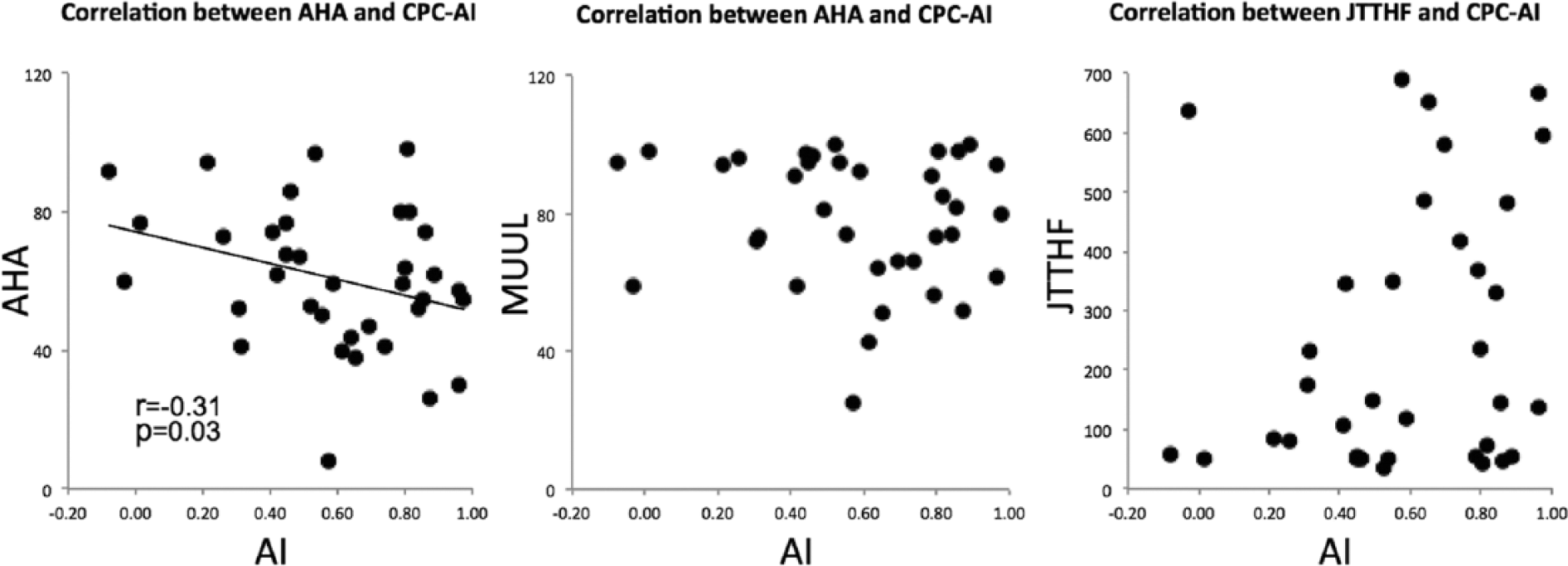
Scatter plots showing the correlation between asymmetry index (AI) of corticopontocerebellar (CPC) tracts—AI = (C – I) / (C + I), where C and I represent the number of streamlines from the contralesional and ipsilesional hemispheres, respectively to the contralateral cerebellum—and hand function measures of the impaired hand as an assisting hand in bimanual tasks (Assisting Hand Assessment [AHA]) and the impaired hand in unimanual tasks (Melbourne Assessment of Unilateral Upper Limb Function [MUUL]; Jebsen-Taylor Test of Hand Function [JTHHF]). A more symmetrical distribution of streamlines arising from one hemisphere compared to the contralateral corresponds to an AI close to 0. The higher the AHA and MUUL scores, the better the hand function.
Discussion
Our study validates the use of the HARDI technique with constrained spherical deconvolution and probabilistic tractography to define the CPC tracts through a ROI-based approach in a cohort of children with typical development. Using this approach, we were able to reproduce the crossed pattern between cerebral and cerebellar cortices, demonstrating a symmetrical distribution of left- and right-seeded bundles, as assessed by the intrasubject measure of the number of streamlines.
To our knowledge, this is the first study that applies diffusion tractography to explore CPC connectivity in children with CP. In adults with stroke, only one study explored CPC tracts’ microstructure by measuring fractional anisotropy of the MCP on color-coded diffusion vector maps. 5 In children, structural CPC abnormalities were only anecdotally reported, consisting of a reduction of contralesional MCP size on the color-coded fractional anisotropy maps following hemispheric stroke. 15 We were able to demonstrate that patients with unilateral brain lesion have a disruption of corticocerebellar connectivity compared to controls, consisting of a thinning of the CPC bundle descending from the lesioned hemisphere to the contralateral cerebellum. This asymmetry was not detected from a visual evaluation of the structural images by an experienced examiner, neither at the level of the cerebellar peduncle nor at the level of the cerebellar hemispheres. This finding is in contrast with that reported by Mah, 8 who found visually detectable crossed cerebellar atrophy in 40% of the children with acquired stroke. Our population differs from that of Mah 10 in that our patients had congenital lesions, occurring either prenatally or perinatally (within 28 days from birth), as opposed to strokes acquired after the first month of life. The simplest explanation for these conflicting findings is that the degree of crossed atrophy in congenital lesions is relatively milder compared to later lesions and the consequent cerebellar atrophy is less obvious and rarely detectable by simple visual assessment, requiring more quantitative approaches. 14 Nevertheless, a more convincing explanation is that other mechanisms of neuroplasticity involving preserved afferent or efferent cerebellar circuits may compensate for the cerebellar hemispheric volumes. This interpretation is compatible with the study of Hamano et al, 4 the only one addressing CPC connectivity in a population including children with CP (ie, congenital lesions). In their article, Hamano and colleagues 4 failed to find evidence of CCD in children with CP, interpreting their findings as the result of an insufficient functional maturation of CPC tract at the time of brain damage. Indeed, morphologic studies have shown that myelination of pontocerebellar fibers appears only during the first postnatal month, when the myelination of the corticospinal tract in the pons is well advanced, and continues at least up until the fourth year of life. 37 All together, previous findings and ours suggest that unilateral lesions occurring pre- or perinatally do not lead to a chronic functional depression of neuronal activity at the distant site of contralateral cerebellum, albeit giving rise to a disruption of structural connectivity of the CPC tract originating from the lesioned hemisphere. The lack of asymmetry in metabolic activity of cerebellar hemispheres, as shown by single-photon emission tomography by Hamano et al 4 in congenital lesions, should then be the result of a functional hemispheric compensatory reorganization targeting cerebellar structures different from the CPC tract. Conversely, the marked reduction of structural connectivity from the lesioned hemisphere to the contralateral cerebellum could be a specific consequence of wallerian degeneration of CPC tracts mainly originating from the sensorimotor cortex. Despite being significantly greater in the CP group, a certain degree of asymmetry of CPC tract has been even found in the control group. We can explain this finding by considering the physiologic asymmetry of motor tract (eg, the corticospinal tract) that has effect on hand dominance; we can hypothesize a similar phenomenon for CPC tracts. Another possibility is due to the technique itself, as based on a visual ROI approach or sometimes generating aberrant fibers.
The evidence of a correlation between CPC connectivity and hand function in our CP subjects would support the significance of the observed structural disruption. There are few studies in adults and one in children exploring the relationship between CCD and motor outcome. In adult stroke studies, CCD has been related with long-term motor outcome and in particular with specific deficits attributable to cerebellar dysfunction such as ataxia,9,38-42 suggesting that secondary damage to crossed cerebrocerebellar pathways is associated to a worse long-term motor outcome. In children with acquired stroke, no evidence of cerebellar dysfunction has been detected with the Paediatric Stroke Outcome Measure, 10 which is, however, a very broad measure of functional outcome. In our cohort, we found a significant but low correlation between degrees of asymmetry of CPC tract and AHA scores for the impaired hand in bimanual abilities. This is a novel finding, as previous studies had shown that bimanual coordination is rather influenced by the involvement of other brain structures, in particular the corpus callosum.43,44 Although never explored in children with CP, anatomic and functional data support the cerebellum as a key structure in coordination of complex bimanual movements, potentially making tests assessing bimanual skills more sensitive to CPC disruption. 41 It is of note that the AHA assesses impaired hand function in bimanual tasks, thus being substantially influenced by unimanual abilities and coordination skills, supporting our finding of a low statistical correlation. Studies on larger samples and specifically exploring cerebellar functions might be expected to unveil stronger correlations between bimanual abilities and CPC asymmetry. Conversely, no correlation was found with unimanual skills (MUUL and JTTHF). This might not be surprising, as unimanual abilities, in terms of both capacity and speed, are more likely to be influenced by spasticity and limitations in range of movement, primarily related to corticospinal and thalamocortical tract connectivity.17,36,45
Our study adds to previous literature in that it is the first to explore the correlation between the quantitative degree of CPC disruption and the severity of different aspects of hand function impairment as assessed by comprehensive quantitative measures of unimanual and bimanual function. These data provide new insights into the understanding of plastic mechanisms of recovery after a brain injury. Should further studies support our findings, a novel variable to target with intervention could be proposed. Potentially, as the structural maturation of cerebellum is more protracted compared to the cerebrum and as the myelination of the CPC via the MCP starts and ends later compared to the corticospinal tract via the posterior limb of internal capsule, 46 a wider window for network plasticity could be targeted for the recovery of bimanual abilities after a prenatal or perinatal injury.
As part of our aims, we assessed the influence of additional factors, including side, timing, and severity of brain damage as to the degree of asymmetry in CPC connectivity. Both side and timing of the lesion did not appear to have an impact on disrupted connectivity of CPC tracts in that all identified subgroups upheld the statistical significance shown by the whole group analysis. The degree of asymmetry was found to correlate with the severity of the lesion as assessed by the basal ganglia and brainstem score. This is not unexpected, as this score consists of the sum of individual scores assessing the integrity of multiple structures, including the posterior limb of the internal capsule and the brainstem, both incorporating fibers from the CPC tract. Interestingly, no correlation was found with the hemispheric score, which is a measure of supratentorial lesion severity. This suggests that the disruption of CPC connectivity is not directly associated with more global measures of lesion size but rather with the involvement of strategic brain structures, thus leading to more specific functional deficits—namely, the impairment of bimanual function. To support this hypothesis, we further explored the correlation between the hemispheric scores and our hand function measures, demonstrating a significant dependence between them. All in all, our findings support the hypothesis that supratentorial lesion severity and CPC disruption are 2 independent determinants of hand motor function.
Our study has several limitations. The spatial resolution of diffusion images was limited by the necessity for shorter acquisition time, as our patients did not receive general anesthesia for MRI acquisition, to improve collaboration and reduce motion artifacts. Also, we do not have information about the type of reorganization of sensorimotor function in these children, which is known to correlate with hand function. Further studies are mandatory to improve the methodology and to consider the impact of other factors as corticospinal reorganization on CPC connectivity and their combined effect on hand function.
In conclusion, our findings for the first time provide evidence of a disruption of cerebrocerebellar structural connectivity in children with congenital unilateral brain damage and hemiplegic CP. While in these patients, the reorganization of corticospinal and thalamocortical sensorimotor pathways correlated with motor abilities,17,36,45 very little is known about the possible functional underpinnings of other relevant motor networks, including the cerebrocerebellar connections. Our data suggest that CPC alterations might have a specific and independent impact on bimanual tasks, thus providing further insights on the structure-function correlation in children with unilateral CP. As the maturational timing of corticocerebellar connectivity is different from that of corticospinal tract, 46 this has potential implications for rehabilitation strategies, as it might contribute to the case-by-case indication for unimanual or bimanual early intervention. Further studies are needed for a better understanding of the role of CPC tract disruption on hand function and the possible effect of an early treatment on cerebrocerebellar connectivity and functional recovery.
Footnotes
Acknowledgements
We thank all the children who took part in this study and their families.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by grants from the National Health and Medical Research Council of Australia and by the Mariani Foundation of Milan (grant R 11-86).
