Abstract
Background. Spasticity and allodynia are major sequelae that affect the quality of life and daily activities of spinal cord injury (SCI) patients. Although rehabilitation ameliorates spasticity and allodynia, the molecular mechanisms involved in these processes remain elusive. Objective. To investigate the molecular mechanisms by which rehabilitation ameliorates spasticity and allodynia after SCI in rats. Methods. The expression levels of brain-derived neurotrophic factor (BDNF) and potassium-chloride cotransporter-2 (KCC2), as well as the localization of KCC2, were examined in the lumbar enlargements of untrained and treadmill-trained thoracic SCI model rats. Spasticity and allodynia were determined via behavioral and electrophysiological analyses. The effects of BDNF on spasticity, allodynia, and KCC2 activation were determined by inhibition of BDNF signaling via intrathecal administration of TrkB-IgG. The effects of SCI and training on the expression levels of functional phospholipase C-γ in the lumbar enlargement were also examined. Results. Treadmill training after SCI upregulated endogenous BDNF expression and posttranslational modification of KCC2 in the lumbar enlargement significantly. There were also significant correlations between increased KCC2 expression and ameliorated spasticity and allodynia. Administration of TrkB-IgG abrogated the training-induced upregulation of KCC2 and beneficial effects on spasticity and allodynia. The expression level of functional phospholipase C-γ was reduced significantly after SCI, which may have contributed to the change in the function of BDNF, whereby it did not trigger short-term downregulation or induce long-term upregulation of KCC2 expression secondary to training. Conclusions. BDNF-mediated restoration of KCC2 expression underlies the suppression of spasticity and allodynia caused by rehabilitation.
Introduction
Rehabilitation approaches such as treadmill training are utilized widely in the treatment of spinal cord injury (SCI) patients. 1 Spasticity and allodynia are some of the major sequelae that affect the quality of life and daily activities of SCI patients. Although previous studies using SCI animal models have demonstrated that treadmill training ameliorates these sequelae,2,3 the molecular mechanisms underlying this effect remain unclear.
The loss of spinal inhibitory transmission resulting from a collapse of GABAergic activity is a major mechanism of both spasticity and allodynia.4,5 Two independent studies have also implicated potassium-chloride cotransporter 2 (KCC2) in these disorders.6,7 KCC2 is a neuron-specific cotransporter that regulates spinal excitability by maintaining the intracellular chloride concentration of neurons at low levels. 8 In mature neurons with high expression levels of KCC2, GABAergic neurotransmission has inhibitory effects due to the hyperpolarization that occurs when ligand-gated Cl− channels are opened by GABAergic agonists. 9 However, GABA can also induce neuronal depolarization when KCC2 expression is low, as occurs at some developmental stages, during adult neurogenesis, or after SCI.10-12 Hence, SCI-induced downregulation of KCC2 increases neuronal excitability, thereby triggering spasticity and allodynia.7,10,13
In mature and intact neural networks of the spinal cord, brain-derived neurotrophic factor (BDNF) downregulates KCC2 expression through tropomyosin-receptor-kinase-B (TrkB) signaling; 14 by contrast, BDNF upregulates KCC2 in the immature hippocampus by activating the TrkB cascade.15,16 Phospholipase C-γ (PLCγ), which is an effector of the TrkB cascade, is thought to mediate this shift in the action of BDNF; specifically, BDNF induces upregulation of KCC2 in the absence of PLCγ activity. 17 Notably, BDNF also induces upregulation of KCC2 in injured corticospinal neurons. 18 Whereas BDNF expression is reduced after SCI, treadmill training upregulates the expression of this protein and facilitates locomotor recovery.19,20 Remarkably, a recent study showed that exogenous BDNF injection after SCI upregulates KCC2 expression and attenuates spasticity. 7 Furthermore, a separate study demonstrated that a combination of training and exogenous BDNF injection does not exacerbate allodynia. 21 Consequently, a major concern regarding clinical rehabilitation is whether training induced BDNF can trigger downregulation of KCC2 or not. 22 However, to our knowledge, no integrated studies have examined the relationship between rehabilitation-induced endogenous BDNF and KCC2 expression and allodynia/spasticity.
Here, a moderate contusive rat model of thoracic-cord injury was used to investigate the mechanisms involved in the effect of post-SCI treadmill training on endogenous BDNF expression, KCC2 expression and functional posttranslational modification, and spasticity and allodynia.
Methods
Animals
All experiments were approved by the Animal Ethics Committee of Keio University (Tokyo, Japan; No. 12082-(0)) and were performed in full compliance with the Guide for the Care and Use of Laboratory Animals (National Institutes of Health, Bethesda, MD). In total, 136 female Sprague-Dawley rats (8-9 weeks old, 190-220 g; Clea, Tokyo, Japan) were used in the study, as shown in Figure 1. More details are described in the Supplementary Materials.
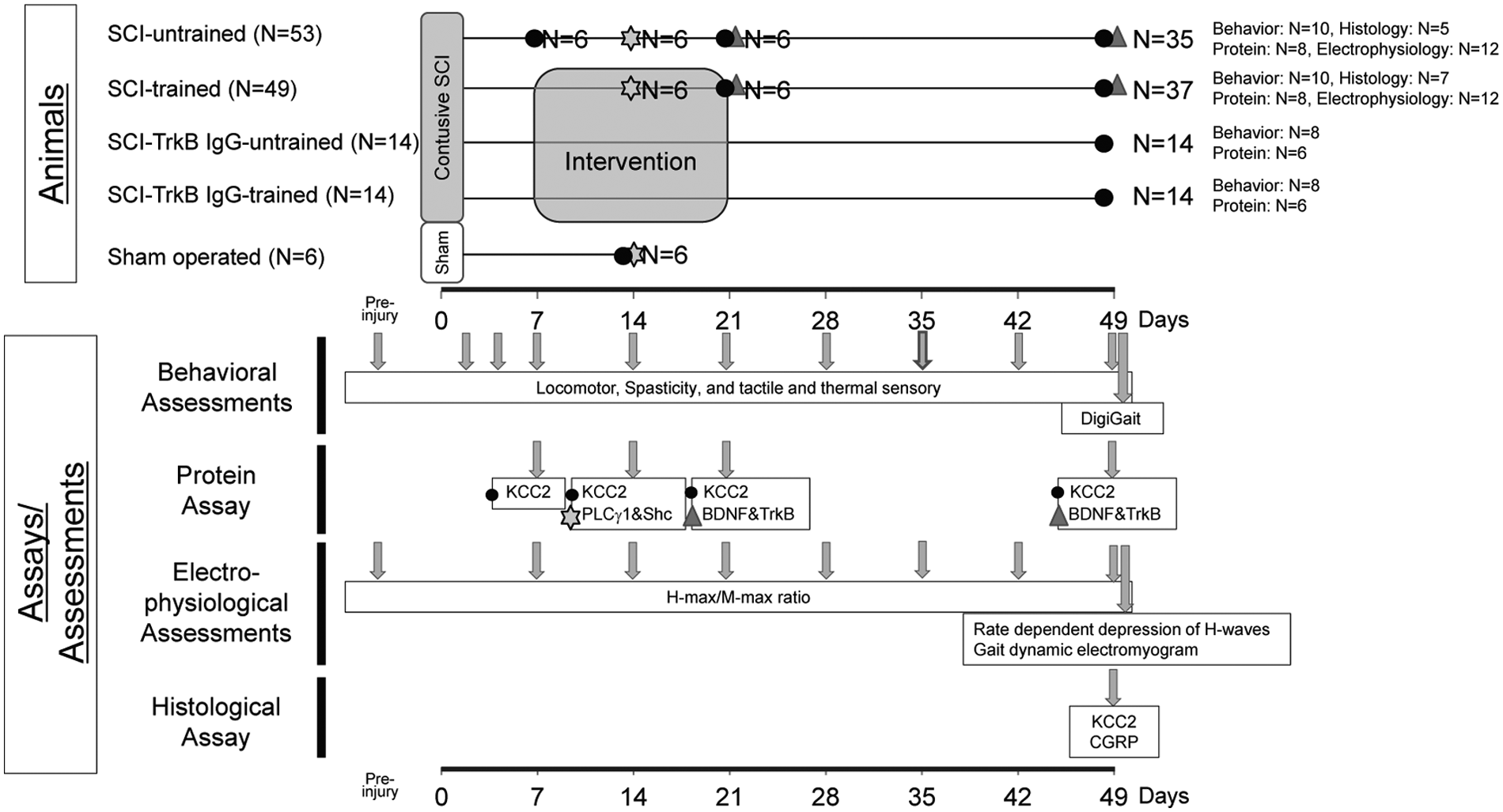
Summary of the experimental timeline and groups.
Spinal Cord Injury
Moderate lower thoracic level contusive SCI was performed as described previously. 23 Briefly, following T10 laminectomy, a 200 kilodyne contusive injury was applied to the exposed dura mater using a commercially available SCI device (IH Impactor, Precision Systems and Instrumentation, Lexington, KY).
Locomotor Training
SCI animals underwent partial body weight–supported bipedal treadmill gait training. The training was conducted 7 days/week for 2 consecutive weeks (days 7-20 after SCI) using a commercially available robotic device (Rodent Robot 3000; Robomedica Inc, Irvine, CA). 24 The training was performed for 20 minutes with 70% to 80% of the animal’s body weight supported. This training device had computer controlled aid arms that were attached to the animals’ ankles to assist the swings; however, to prevent disruption of the voluntary gait, these aid arms were used minimally (particularly at the beginning of the training period). The speed of the treadmill was set to 2.5 to 3.5 cm/s.
Intrathecal Administration of TrkB-IgG
Recombinant human TrkB/Fc chimera (TrkB-IgG; Sigma-Aldrich, St Louis, MO) was used for functional blocking of BDNF. Osmotic minipumps (model 2002; Alzet, Cupertino, CA) filled with phosphate-buffered saline (PBS) alone or TrkB-IgG dissolved in PBS (0.25 µg/µL) were attached to the intrathecal catheters (Alzet). At 7 days postinjury (DPI), the intrathecal catheter was inserted at the laminected 13th thoracic vertebra. TrkB-IgG or PBS was administered continuously at a rate of 3 µg/day for 2 weeks, as described previously.25,26 The pumps were removed at the end of the intervention period (21 DPI).
Behavioral Analyses
The motor function of the hindlimbs was evaluated by the Basso-Beattie-Bresnahan (BBB) scale. 27 Quadrupedal gait dynamics were evaluated using the rats’ footprints and a DigiGait imaging system (Mouse Specifics, Boston, MA). 28 Hindlimb spasticity was assessed as the resistance to full flexion from full extension. The maximum force required was measured using a handheld strain gauge (model FGP-0.5; Nidec-Shimpo, Kyoto, Japan). 29 Tactile hyperalgesia was assessed with harmless mechanical stimuli of the hind paw using a von Frey monofilament (Semmes-Weinstein Anesthesiometer; Stoelting, IL). 30 Thermal hyperalgesia was assessed by the Hargreaves test with the application of radiant heat to the hind paw using a plantar test device (IITC Life Science, Woodland Hills, CA). 31 The detailed protocols are described in Supplementary Materials.
Electrophysiological Analyses
All electrophysiological experiments were performed using a commercially available electromyogram (Neuropack S1 MEB-9402; Nihon Kohden, Tokyo, Japan). Spasticity was assessed via determination of the H-max/M-max value 32 and via the rate-dependent depression (RDD) of the H-reflex. 7 Sciatic nerve was stimulated at the thigh level, and a needle pick-up electrode was inserted into the interosseous muscle. To determine the co-contraction patterns of antagonistic muscles, flat electrodes were secured onto the surfaces of the tibialis anterior and gastrocnemius muscles. Dynamic electromyograms of muscle contractions during bipedal gait were recorded, and the co-activation coefficient of the 2 muscles was determined. 33 The detailed protocols are described in Supplementary Materials.
Primary Antibodies
Supplementary Table 1 lists the primary antibodies used in the study. Specific bands were produced consistently for each antibody, and the size of each band was confirmed using a molecular weight standard (Precision Plus Kaleidoscope standard; Bio-Rad, Hercules, CA).
Immunoblotting
The spinal cords were removed and transversely cut at the lumbar enlargement (L2 to L5). Following mechanical dissociation, the spinal cords were lysed in ice-cold lysis buffer 7 and then centrifuged at 15 000 rpm for 15 minutes. The supernatant was applied for the analysis. Equal amounts of proteins were loaded onto 4% to 12% gradient polyacrylamide gels and then transferred onto polyvinylidene difluoride membranes. 34 After blocking with 2.5% bovine serum albumin, the membranes were probed with primary antibodies at 4°C overnight. After washing, the membranes were incubated with appropriate secondary antibody. The products were detected using an enhanced chemiluminescence reagent (GE Healthcare, Buckinghamshire, UK) and the ImageQuant LAS 4000 instrument (GE Healthcare). Polyacrylamide gel electrophoresis was performed in triplicate for each analysis. The detailed protocols are described in Supplementary Materials.
Immunohistological Assessment of KCC2
Sections (20-µm thick) were dissected from the L2 to L5 level of animal spinal cords and prepared (n = 5 untrained and n = 7 trained). 35 The sections were incubated at 4°C overnight with primary antibodies. After washing, the sections were then incubated with appropriate secondary antibodies. The images were obtained using a confocal laser-scanning microscope (LSM700; Carl Zeiss, Oberkochen, Germany). KCC2 immunoreactivity was examined on the plasma membrane of motoneurons. Randomly selected motoneurons in the lamina IX were captured in axial sections at 630× magnification (total of n = 84 motoneurons from the untrained group and n = 116 motoneurons from the trained group). KCC2 immunoreactivity on the plasma membrane was measured as the index of the mean density of the immunolabeling within the membrane. 7 KCC2 immunoreactivity in the dorsal horn was also measured. 36 The detailed protocols are described in Supplementary Materials.
Immunohistological Assessment of Calcitonin Gene-Related Peptide
To quantify the number of calcitonin gene-related peptide (CGRP)-positive fibers, spinal cord sections of 12 animals (n = 5 untrained and n = 7 trained) were analyzed using 3,3′-diaminobenzidine immunohistochemistry. The bilateral lamina III areas were captured in axial sections of the lumbar enlargement at 400× magnification. CGRP-positive fibers were measured.37-39 The detailed protocols are described in Supplementary Materials.
Statistical Analysis
All data are presented as the mean ± SEM according to the statistical analysis described in Supplementary Materials.
Results
Effects of Treadmill Training on Locomotor Function, BDNF Expression, and TrkB Activation
In clinical settings, to enhance recovery as much as possible, intensive rehabilitation of paralyzed SCI patients is performed chiefly by targeting the phase when locomotor function recovers most efficiently. Based on previous studies and our preliminary data, treadmill training of thoracic-cord-contused rats to promote locomotor function is most effective 1 to 3 weeks after SCI.22,40 Moreover, a previous report demonstrated that at least 1 week of training induces a significant upregulation of BDNF in a rat model. 19 Based on these findings, we chose a 2-week training period, beginning 1 week after SCI. Figure 1 shows an overview of the study timeline and interventions.
Control (untrained) rats underwent SCI but were not subjected to treadmill training. Analyses of the BBB score up to 49 DPI revealed that treadmill training improved the locomotor function of the rats significantly (2-way repeated-measures ANOVA; P = 1.75 × 10−5; Figure 2A). Furthermore, Bonferroni-corrected Mann–Whitney U tests demonstrated a significant benefit of training from 21 DPI until the end of the assessment (Figure 2A). Behavioral analysis using the DigiGait system also revealed that the trained group exhibited significantly better gait performances (longer stride lengths and smaller hindlimb step angles) than the untrained group (Figure 2B).
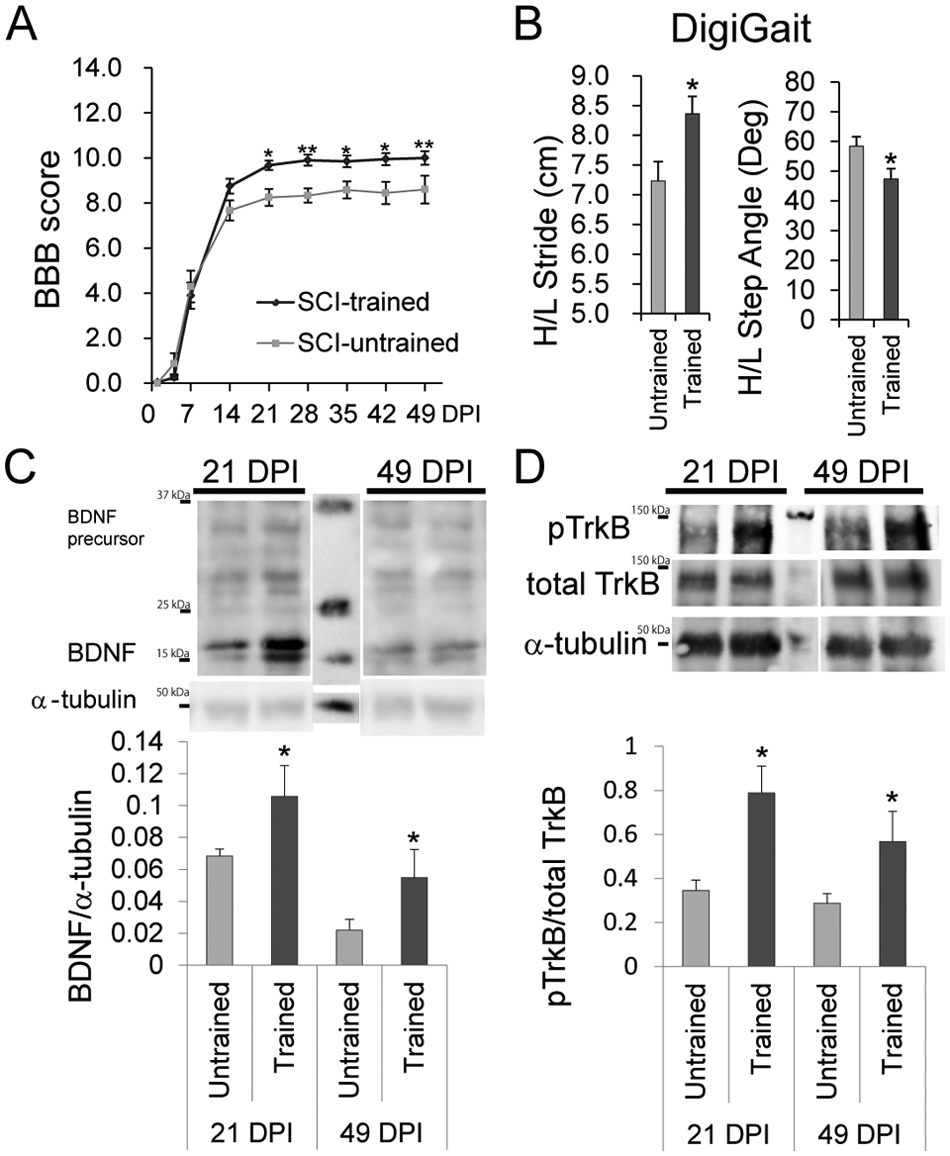
The effect of training on locomotor recovery and activation of the BDNF-TrkB cascade.
To examine the short-term and long-term effects of training on BDNF expression and the activation of its downstream cascade, immunoblot analyses of BDNF, phospho-Y515 TrkB, and total TrkB (full-length form) in the lumbar enlargement, which is the part of the spinal cord (L2-L5) responsible for lower limb function, were performed at 21 and 49 DPI. BDNF protein expression was significantly higher in the trained group than in the untrained group at both time points (Figure 2C). In addition, the ratio of phospho-Y515 TrkB to total TrkB was significantly higher in the trained group than in the untrained group at these time points (Figure 2D). Truncated forms of TrkB with potentially different functions have been reported. 41 Immunoblotting confirmed that training did not affect the relative expression levels of full-length TrkB and its major truncated form (TrkB-T1) at 21 and 49 DPI (Supplementary Figure 1).
Effects of Treadmill Training on Spasticity and Allodynia After SCI
To evaluate the effect of treadmill training on spasticity in the hindlimbs, the hindlimb resistance force at rest was measured using a strain-gauge test and the H-max/M-max ratio was determined up to 49 DPI. Although spasticity increased gradually in the untrained group after injury, training restrained spasticity to almost baseline levels. In the strain-gauge test, the spasticity of the trained group was significantly lower than that of the untrained group (2-way repeated-measures ANOVA; P = 3.13 × 10−11). Bonferroni-corrected Mann–Whitney U tests indicated that the hindlimb resistance force at rest of the trained group was significantly lower than that of the untrained group from 28 DPI until the end of the assessment (Figure 3A). Similarly, the H-max/M-max ratio was significantly lower in the trained group than in the untrained group at 35 DPI (Figure 3B). Furthermore, at higher frequencies, the RDD of the H-reflex at 49 DPI was significantly higher in the untrained group than in the trained group (2-way repeated-measures ANOVA; P = .999, .413, .138, .0276, and .0298 at 0.1, 0.5, 1, 2, and 5 Hz, respectively; Figure 3C), demonstrating that training reduced spasticity.
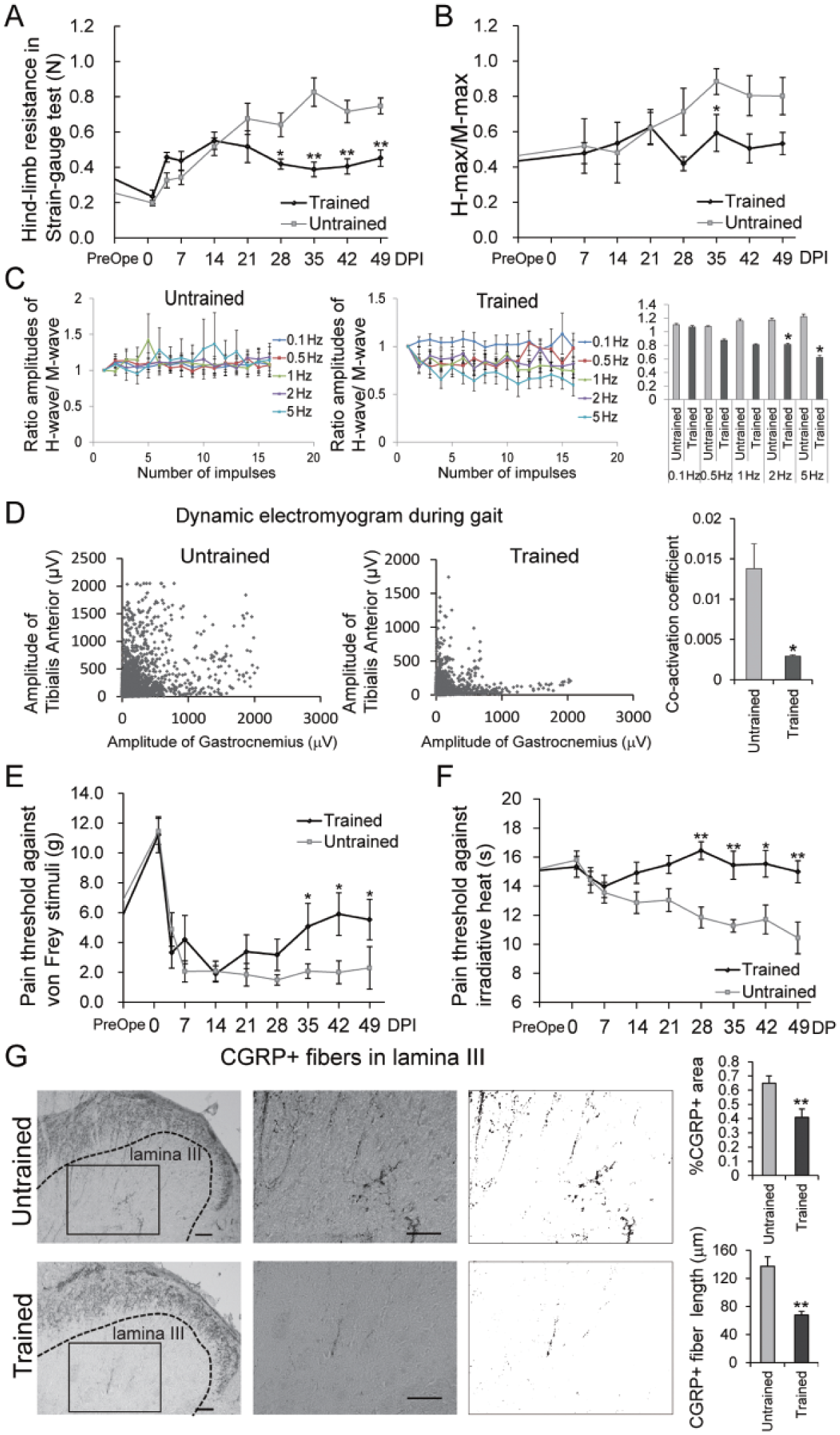
The effect of training on spasticity, motor control, and allodynia of the hindlimb.
Although spasticity is the static aspect of motor control dysfunction, the dynamic aspect relates to the loss of coordinated rhythmic muscle activation. The dynamic control of antagonistic muscles during gait was assessed because coupling of the interlimb homologous muscles and the intralimb flexor muscles is improved by training.42,43 Muscle-surface electrodes were used to collect electromyographic measurements during locomotion at 49 DPI. Compared with that in the untrained group, co-contraction of the ankle flexor and extensor muscles was drastically lower in the trained group (Figure 3D, left and middle panels). The co-activation coefficient of the 2 muscles was also significantly lower in the trained group than in the untrained group (Figure 3D, right panel). These data indicate that training significantly ameliorated spasticity and improved coordinated motor control; notably, the effects became particularly evident at the end of the training period.
Next, to evaluate how treadmill training affects allodynia after SCI, the tactile and thermal hyperalgesia of the hindlimb paws were assessed up to 49 DPI using a von Frey monofilament 30 and Hargreaves tests, 31 respectively. Tactile hyperalgesia was gradually attenuated in the trained group from 28 DPI until the end of the assessment, whereas no remarkable changes were observed in the untrained group (2-way repeated-measures ANOVA; P = .030). A Bonferroni-corrected Mann–Whitney U test demonstrated a significant effect of training starting at 35 DPI (Figure 3E). Thermal hyperalgesia was exacerbated in the untrained group, but not in the trained group, from 21 DPI to the end of the assessment (2-way repeated-measures ANOVA; P = .0044), and Bonferroni-corrected Mann–Whitney U tests demonstrated a significant difference between the 2 groups starting at 28 DPI (Figure 3F). To support these findings, we examined the total length of CGRP-positive fibers, which function in the transmission of pain, in animals from the untrained and trained groups. 37 CGRP in the dorsal horn is mainly of primary afferent origin, 44 and CGRP-positive fibers running between laminae I/II and X are partly considered a product of aberrant sprouting. 45 Quantification of CGRP-positive fibers at lamina III accurately reflects the perception of pain.37-39 Consistent with the behavioral assessments, the CGRP-positive area and length of the fibers in lamina III were significantly smaller in the trained group than in the untrained group at 49 DPI (Figure 3G). These data demonstrate that treadmill training reduced mechanical and thermal hyperalgesia significantly. Consistent with the effect of training on spasticity, the effect on allodynia also became particularly evident at the end of the training period.
Effects of Treadmill Training on KCC2 Expression in the Spinal Motoneurons and the Dorsal Horn of the Lumbar Enlargement
To determine whether treadmill training affects the expression of functional KCC2 in the lumbar enlargement, the expression levels of Ser-940-phosphorylated KCC2 (pKCC2) and total KCC2 were examined at 7, 21, and 49 DPI. As a control, the expression levels of these proteins were also examined in sham-operated rats. The ratio of pKCC2 to KCC2 in the trained group at 49 DPI was significantly greater than those in the pretraining group at 7 DPI, the untrained and trained groups at 21 DPI, and the untrained group at 49 DPI (Figure 4A). There was no significant difference between the pKCC2 to KCC2 ratios in the untrained and trained groups at 21 DPI (Figure 4A). Notably, pKCC2 expression correlated with the results of the strain-gauge test (spasticity), the von Frey test (tactile allodynia), and the Hargreaves test (thermal allodynia), which were conducted in the same animals at 49 DPI (Figure 4B). By contrast, there was no correlation between pKCC2 expression and the BBB score (locomotor function; Figure 4B). To evaluate the distribution of KCC2 in the spinal cord, lumbar enlargement sections of the untrained and trained animals at 49 DPI were immunostained with an anti-KCC2 antibody. A quantitative analysis of the results revealed that KCC2 immunoreactivity on the plasma membranes of motoneurons was significantly higher in the trained group than in the untrained group (Figure 4C). Furthermore, training increased the global KCC2 immunoreactivity in the lamina I/II region significantly (Figure 4D).
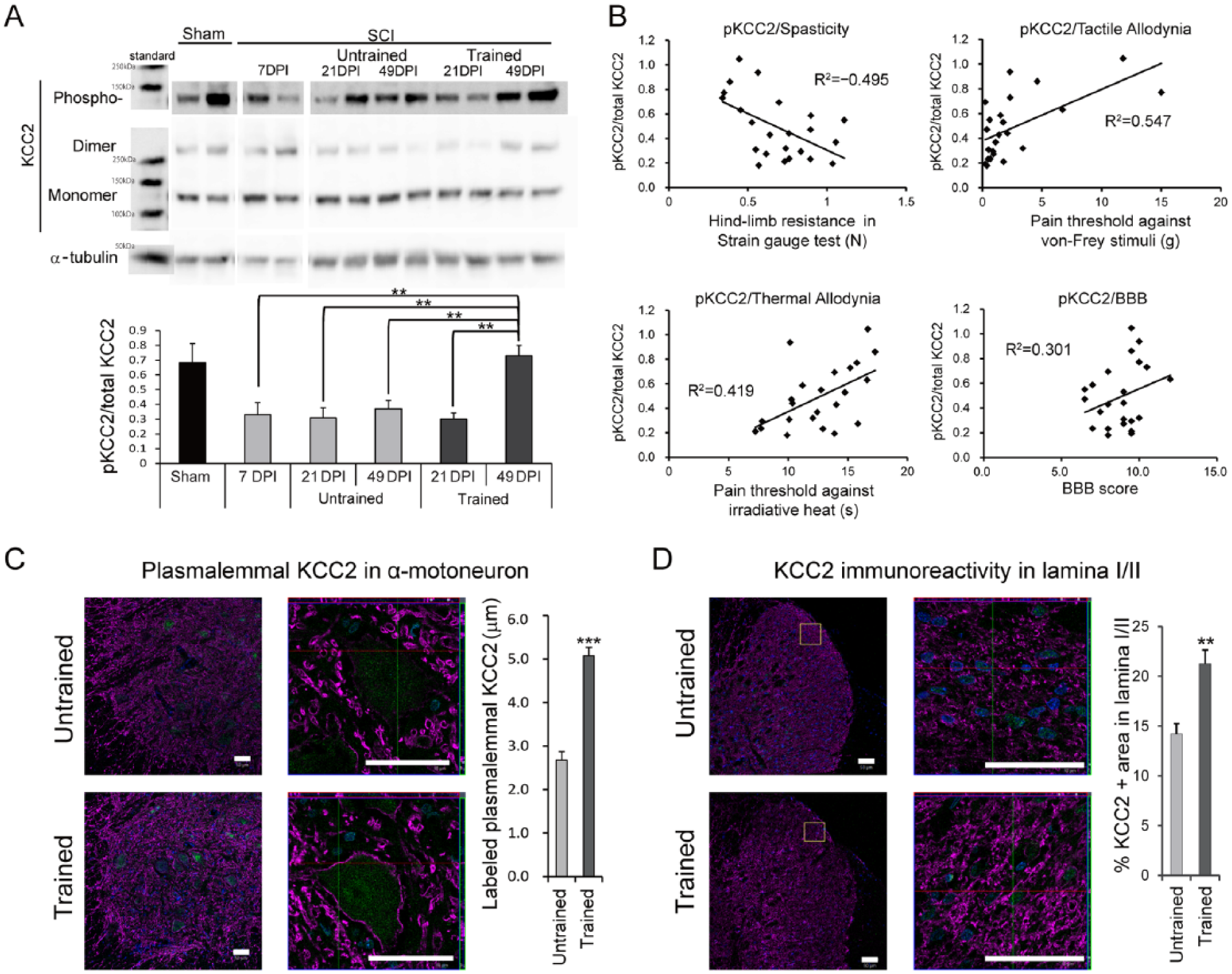
Short-term and long-term changes in KCC2 expression following treadmill training.
Effects of Inhibiting BDNF-Mediated Signaling on Treadmill Training-Induced Behavioral Manifestations and Functional KCC2 Expression
To determine its role in the upregulation of KCC2 expression and the beneficial effects of treadmill training, endogenous BDNF signaling was blocked at 1 to 3 weeks post-SCI (the potential training period) via continuous intrathecal administration of TrkB-IgG, and the BBB scores, hindlimb resistances, pain thresholds, and functional KCC2 expression levels were determined in untrained and trained rats. An overview of the experimental plan is shown in Figure 1. As expected, in the absence of TrkB-IgG, treadmill training improved locomotion, spasticity, and allodynia significantly (Figures 2A, 3A, 3E, and 3F); however, training had no effect on these parameters when endogenous BDNF availability was reduced by intrathecal administration of TrkB-IgG during the training period (Figure 5A-D). Notably, TrkB-IgG had no effect on any of the endpoints in the untrained group, indicating that its effects are not related to nonspecific adverse events.
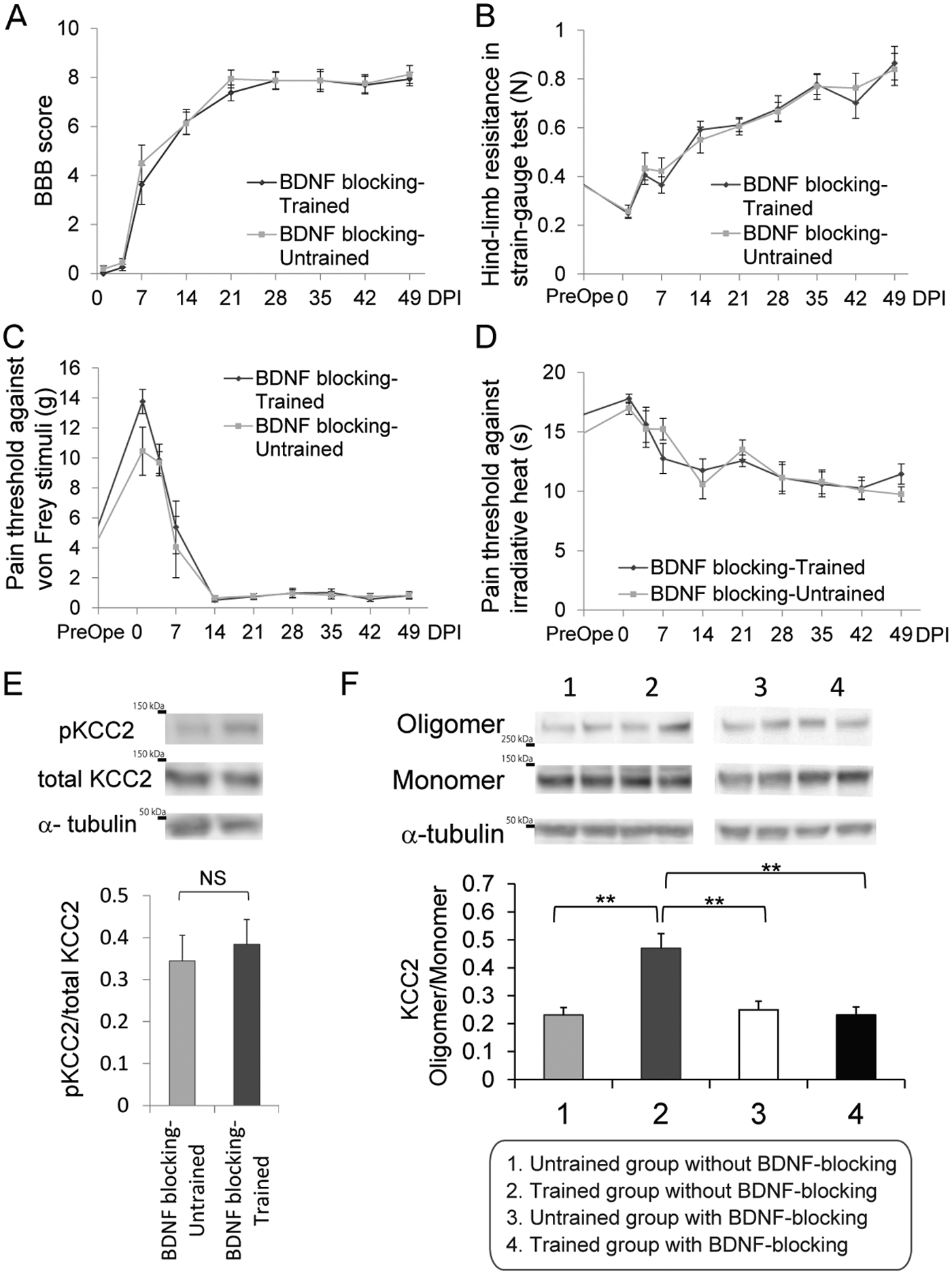
The effect of blocking endogenous BDNF on training-induced changes in locomotor function, spasticity, allodynia, and KCC2 expression.
Immunoblot analyses of pKCC2 expression in spinal cord samples from trained and untrained rats administered TrkB-IgG were also performed at 49 DPI. The administration of TrkB-IgG abrogated the training-induced upregulation of functional pKCC2 expression (Figure 5E) and abolished the training-induced increase in the oligomerization of KCC2 (Figure 5F), which is known to control transporter activity and protein targeting to the plasma membrane. 46 Overall, these results indicate that the improvement in the behavioral tests and the upregulation of functional KCC2 expression caused by treadmill training were abrogated by the blockage of BDNF signaling during the training period.
The Effect of SCI on Phospho-PLCγ Expression in the Spinal Lumbar Enlargement
The role of BDNF/TrkB signaling in the regulation of KCC2 is context-dependent. PLCγ1 is involved in BDNF-TrkB signaling. 47 While the PLCγ and Shc docking sites of TrkB are required for BDNF/TrkB-mediated downregulation of KCC2 in intact neurons, 14 activation of the Shc pathway in the absence of PLCγ activation results in upregulation of KCC2 expression. 17 To characterize the context in which treadmill training-induced endogenous BDNF activity upregulates KCC2 expression, the expression levels of PLCγ1 and Shc in the lumbar enlargement were examined after SCI by immunoblotting. At both 14 DPI, which was in the middle of the training period, and 49 DPI, the ratios of phospho-Y783-PLCγ1 to total PLCγ1 were significantly lower in the injured animals than the sham controls (Figure 6A), indicating prolonged SCI-induced inactivation of PLCγ1. Notably, treadmill training did not modify this inactivation of PLCγ1. Moreover, Shc expression was comparable in sham-operated, SCI-trained, and SCI-untrained rats (Figure 6B).
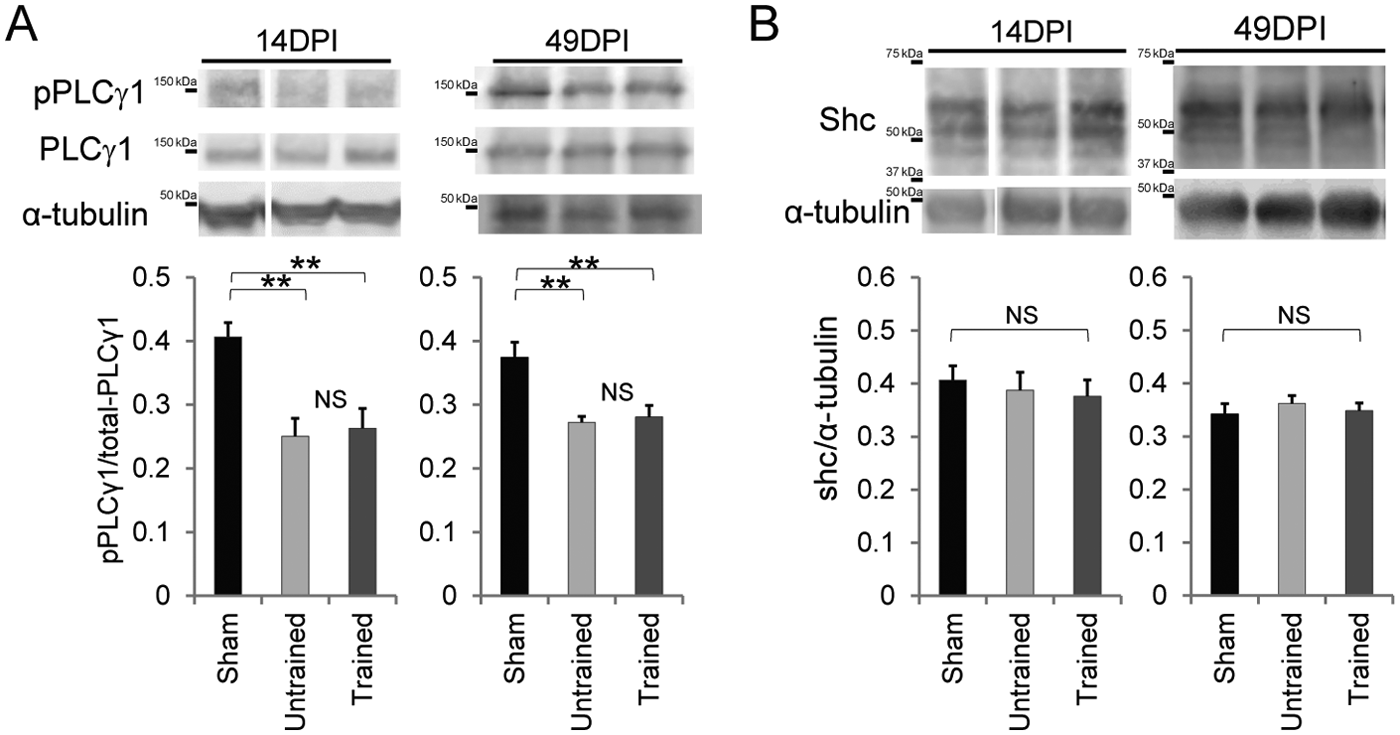
The effect of training on activated PLCγ1 expression and Shc expression in the lumbar enlargement after moderate contusive spinal cord injury (SCI).
Discussion
This study reports 3 novel findings related to the effects of post-SCI treadmill training on recovery after SCI. First, training-induced suppression of spasticity and allodynia is achieved via a common mechanism, namely, the upregulation of BDNF in the lumbar enlargement. Second, training-induced early and long-term increases in endogenous BDNF levels recover the expression of KCC2 and seem to contribute to the amelioration of spasticity and allodynia. Third, a reduction in the activity of PLCγ after SCI may contribute to the beneficial effect of BDNF/TrkB signaling on KCC2 expression.
Notably, the expression of KCC2, rather than BDNF itself, had a similar pattern and correlated significantly with the indices of the 2 different sequelae after the training intervention. SCI typically reduces the expression of KCC2, and this reduction induces spasticity and allodynia.7,10 Previous reports have suggested that upregulation of KCC2 can suppress these sequelae.6,7,10,11 Therefore, the findings presented here provide a link between the basic and therapeutic aspects of the major SCI sequelae; specifically, that spasticity and allodynia are triggered by the downregulation of KCC2,6,7,10,11 whereas training ameliorates these SCI-induced outcomes.2,3,48 However, although functional blocking of BDNF abolished the training-induced increase in pKCC2 at 49 DPI, the possibility that BDNF itself and/or other factors downstream of the BDNF-TrkB signaling pathway participate in the suppression of spasticity and of allodynia cannot be excluded, as described in a recent study. 49 Hence, further investigations are required.
A time lag between the training-induced upregulation of BDNF and the amelioration of spasticity and allodynia, as well as the restoration of KCC2 expression, was observed in this study. At 21 DPI, BDNF expression was higher in trained animals than in untrained animals, whereas KCC2 expression was not upregulated by training until 49 DPI. Previous reports have described both short-term and long-term mechanisms by which BDNF upregulates KCC2 expression.16,50 GABAergic activity, which is increased by BDNF,51,52 contributes to long-term upregulation of KCC2 and functional improvement. 53 Here, consistent with previous studies,5,54 indirect evidence of training-induced increased GABAergic activity was observed at 21 and 49 DPI (Supplementary Figure 2). In addition, we consider a carryover effect of training could affect these longitudinal changes. The average BBB score of trained animals exceed 9 points at the end of training period, whereas it ceased around 8 points in the untrained animals. Thus, SCI animals trained are initiated to walk with weighting on their hindlimbs in their cages. This behavioral change may contribute to the continuous mild BDNF upregulation after the training period. Overall, we consider the longitudinal change is observed regarding the regulation of KCC2 expression after SCI and training.
To date, the mechanism by which increased BDNF expression caused by training or intrathecal injection has beneficial effects on spasticity and allodynia in the context of post-SCI has remained unclear.2,7,21 Although BDNF upregulates KCC2 in injured corticospinal neurons, the underlying mechanism has not been elucidated fully. 18 Here, we attempted to clarify the short-term effect of BDNF on the upregulation of KCC2 after SCI and report the downregulation of activated PLCγ1 in the subacute phase of a SCI model. Because BDNF upregulates KCC2 in the absence of PLCγ, 17 this reduced activation of PLCγ may account for the observed absence of inhibitory effects of BDNF on KCC2 expression during rehabilitation. By contrast, it was reported recently that overexpression of BDNF using an adeno-associated viral vector, not the protein administration or the induction through training, exacerbates allodynia and induces downregulation of KCC2 in a SCI model.55,56 We hypothesize that this discrepancy in the effect of BDNF on KCC2 expression is related to the method of BDNF delivery; therefore, in future investigations, it will be important to confirm that the amount of exogenous BDNF administered via intrathecal injection is equivalent to the amount of BDNF induced by viral vector expression or training. In summary, our finding that training-induced BDNF does not induce KCC2 downregulation leading to spasticity and allodynia is important, because it clearly demonstrates that the voluntary gait training for SCI animals has no harmful effect on spasticity and allodynia. Thus, we argue here that the level of PLCγ activation contributes to the action of BDNF in this context.
In addition to its role in the sequelae of SCI, namely, spasticity and allodynia, downregulation of KCC2 is implicated in the pathogenesis of several neurological disorders, including epilepsy, stress, and schizophrenia.57-59 Therefore, restoring endogenous neuronal inhibition by upregulating KCC2 activity or expression may be a useful therapeutic strategy for these disorders. Although a KCC2-selective analog was developed recently, 60 to our knowledge, no compounds that boost KCC2 function are currently available for clinical use. 61 Overall, the data presented here showing that treadmill training can restore KCC2 levels after SCI is a novel and exciting finding that may have implications for several neurological diseases.
Footnotes
Acknowledgements
We appreciate the help of all the members of the spinal cord research team at the Department of Orthopaedic Surgery, Physiology, and Rehabilitation Medicine at Keio University’s School of Medicine. We also thank Profs J. Nabekura and A. J. Moorhouses for the critical reading of the article and Ms T. Harada and Mr N. Goto for their assistance with the experiments and animal care.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: H. Okano is a scientific consultant for SanBio Co, Ltd (Japan); Eisai Co, Ltd (Japan); and Daiichi Sankyo Co, Ltd (Japan). The remaining authors report no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the following organizations: the Japan Science and Technology-California Institute for Regenerative Medicine collaborative program; the Grants-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (SPS) and the Ministry of Education, Culture, Sports, Science, and Technology of Japan (MEXT); the Project for Realization of Regenerative Medicine; the Support for Core Institutes for iPS Cell Research from MEXT; the Keio Gijuku Academic by a Grant-in-Aid for Scientific Research on Innovative Areas (Comprehensive Brain Science Network) from MEXT; and the General Insurance Association of Japan.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
