Abstract
Physical exercise is a cornerstone in the management of many neurodegenerative disorders, such as Parkinson’s disease, dementia, and stroke. However, much of its beneficial effects on improving motor functions and cognition as well as decreasing neurodegeneration and neuroinflammation are not yet well understood. The obvious limitations of studying the protective mechanisms behind exercise, for example, brain plasticity and neurodegeneration, could be overcome by generating novel animal models of neurodegenerative disorders. In this narrative review, we discuss the beneficial effects of exercise performed in animal models of neurodegenerative disorders and how the results from animal studies can be used in clinical settings. From preclinical studies, the positive effects of exercise have been related to increased levels of neurotrophic factors, elevated expression of anti-inflammatory cytokines, and reduced levels of pro-inflammatory cytokines and activated microglia. It is clear that parameters influencing the effect of exercise, such as intensity, still remain to be investigated in animal studies in order to find the optimal program that can be translated into exercise interventions for patients with neurodegenerative diseases.
Introduction
Physical exercise and its effect on mind and body are well established in modern society. With exercise we consider physical activity and training that causes a substantial increase in heart rate that differs significantly from resting heart rate. In animal studies, running wheels and treadmills are typically used, whereas in humans physical exercise relates to a subset of physical activity that is planned, structured, repetitive, and purposeful, where the objective is improvement or maintenance of physical fitness. 1 Thus, in humans, exercise includes different sports, physical training, or planned exercise as well as vigorous leisure activities. Thus, it is mainly the effects of different kinds of running exercise that can be studied in animals and translated into human research. For this purpose, measuring the animals’ heart rate can be used as exercise readouts in order to translate the results into the clinic. There is compelling scientific evidence for disease prevention of not only cardiovascular diseases but also several cancer forms, diabetes, obesity, and osteoporosis. 2 Besides prevention of diseases, physical exercise can also slow down the progression and symptoms of the above-mentioned diseases.
The effects of exercise on the central nervous system in health and in neurodegenerative and cerebrovascular disorders are research areas of recent interest. Neurodegenerative disorders, such as Parkinson’s disease (PD) and Alzheimer’s disease (AD), are estimated to affect up to 1% and 11%, respectively, of the population older than 65 years. 3 Cerebrovascular disorders, such as stroke, are the leading cause of life-long disability. 4 Clinical studies show that physical exercise lower age-related brain atrophy 5 and reduce the risk of dementia independently of the diet. 6 Exercise is known to be an important preventive action in dementia and neurodegenerative diseases, 7 able to slow down progression and ameliorate disability. 8 Although knowledge about the effects of exercise in these conditions has emerged from studies of patients, there are obvious limitations in studying the effects on the human brain in health and disease. Animal models are important in research and could be used to advance our understanding of the effects of exercise in neurodegenerative and cerebrovascular disorders. Over the past decade, an increasing number of animal studies have convincingly showed the positive effects of exercise on areas such as neuroinflammation, neuroplasticity, neurodegeneration, as well as cognitive, motor, and psychiatric behavior. Also, the positive effect of an enriched environment has in some experimental settings been attributed to physical exercise. 9
In this narrative review, we will discuss the effects of different types of exercise on experimental models of neurodegenerative disorders, particularly PD and AD. We will present what is known from animal studies, highlight what still has to be investigated, and how this knowledge can be transferred into clinical settings. Table 1 shows an overview of different setups regarding training intensity, frequency, and duration that have been used in studies that will be discussed in this review.
Setup of Physical Exercise in Animal Experiments.

Abbreviations: AD, Alzheimer disease; MWM, Morris Water Maze; OF, open field; NOR, novel object recognition; TMT, trimethyltin; n/a, not announced; HD, Huntington’s disease; BDNF, brain-derived neurotrophic factor; PD, Parkinson disease; 6-OHDA, 6-hydroxydopamine; MPTP, 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; DA, dopamine; GDNF, glial cell line-derived neurotrophic factor; CUMS, chronic unpredictable mild stress; SPT, sucrose preference test; MCAo, middle cerebral artery occlusion; LPS, lipopolysaccharide.
Method of Literature Search
This review is based on articles searched in PubMed from 1980 using the following terms in different combinations: physical exercise, training, neuroinflammation, neurodegeneration, intensity, high-intensity, interval training, cytokines, behavior, cognition, mice, rodents, and humans. The article search and collection was conducted between January 2013 and August 2014. All articles whose primary study focus was on the effect of exercise on neurodegeneration and/or neuroinflammation and/or behavior in humans or rodents were included. Reports in which the focus was to study the effect of exercise on other diseases of the whole body were also included if the primary focus was to study the effects of different exercise intensities. Both review articles and study reports were included. Additionally, articles were also found by using the references of included study reports and reviews.
Exercise and Neuroinflammation
An increased level of inflammation has been observed in many patients with neurodegenerative diseases. 10 According to observational studies, nonsteroidal anti-inflammatory drugs (NSAIDs) are suggested to reduce the risk to develop neurodegenerative diseases. Several reports suggest that the NSAID ibuprofen could be beneficial and reduce the risk to develop AD by up to 50%. 11 For PD, ibuprofen was found to reduce the risk to develop PD by up to 27%. 12 However, it is important to note that no overall effect of reducing the risk to develop PD by all NSAIDs together was detected and presently no recommendations to use NSAIDs in preventing PD can be made. 12 These clinical data still emphasize a possible contribution of inflammation in brain disorders. Exercise is known to effectively modulate inflammation and has been reported to change the inflammatory state to become anti-inflammatory or neuroprotective.
Current literature also demonstrates that physical training can be a potentially effective strategy to counteract the systemic inflammation and improve motor and cognitive functions in autoimmune neuroinflammatory diseases, such as systemic lupus erythematous (SLE) and multiple sclerosis (MS).13,14 It is therefore tempting to speculate that the immunomodulatory effect of physical training is an important aspect in patients with neurodegenerative and neuroinflammatory diseases.
In this section, we will discuss aspects of inflammation that are related to cytokine response and microglial activation since these areas have been extensively investigated in studies related to physical exercise and neurodegeneration.
Cytokines
Cytokines are immunomodulatory signaling molecules that can be produced by virtually every cell type but are typically produced by immune cells. Cytokines bind to receptors on the surface of different cell types and are the key messengers to orchestrate the cellular immune response. 15 They are crucial for attracting immune cells and for their intracellular signaling and responses to inflammatory stimuli. These responses can be either neurotoxic or neuroprotective, depending on which signals the immune cells receive. The functions of different cytokines are very complex and depend on the context and concentration of cytokines in relation to one another. However, cytokines such as interleukin-6 (IL-6), tumor necrosis factor-α (TNF-α), interferon-γ (IFN-γ), and interleukin-1β (IL-1β) are considered to have profound pro-inflammatory functions. On the contrary, cytokines such as interleukin-4 (IL-4) and interleukin-10 (IL-10) are regarded as anti-inflammatory. It is believed that the inflammatory status in the periphery somewhat corresponds to the level of inflammation in the brain. However, it is suggested that there could be aspects of systemic inflammation that could be beneficial to slow down the progression of neurodegeneration. This is based on the hypothesis that anti-inflammatory treatment could inhibit the recruitment of CD4+ T-cells and monocytes from the blood to the brain and their possibility to modify the destructive neuroinflammation. 16
The role of inflammation in the development of different diseases of the central nervous system (CNS), including AD and PD, is a research area under extensive investigation.10,17 Physical exercise is potent in reducing the risk of developing AD in elderly people, and a follow-up survey demonstrated that physical exercise, and specifically exercise of higher intensity, reduces the risk of developing AD. 6 This reduced risk was independent of the diet.
In line with clinical data, there are numerous studies using AD transgenic mouse models describing the positive effect of physical exercise, nicely summarized by Intlekofer and Cotman. 18 The majority of these AD mouse studies use mice that have a fast-progressing neuropathological phenotype with amyloid plaque formation and cognitive deficits. Typically, exposure to physical exercise enhanced cognitive ability in these AD mouse models and at the same time reduced pathological A-beta levels, either A-beta peptides or fibrils, in the brain. One study showed how 3 weeks of voluntary running exercise effectively normalized the elevated hippocampal levels of IL-1B and TNF-α in the Tg2576 transgenic mice and at the same time lowers the A-beta level. 19 Interestingly, immune response proteins (IFN-γ and MIP-1α) increased as well as markers for antigen presentation (CD40, MHCII), suggesting an immunostimulatory effect and not only a dampening of the neuroinflammation in the running AD mice.
In PD patients, elevated protein levels of pro-inflammatory cytokines, such as TNF-α, IL-6, IFN-γ, and IL-1β, have been found in the striatal brain areas.20-23 Furthermore, in the substantia nigra (SN) increased levels of the master regulator of inflammatory responses, the transcription factor nuclear factor kappa beta (NFκB), has been reported.20-23 Many studies have demonstrated significant effects of exercise on cytokine levels inside and outside of the CNS. Increased levels of pro-inflammatory cytokines have also been shown to be associated with psychiatric symptoms in animals. Depressive-like behavior can be induced together with increased precortical levels of pro-inflammatory cytokines TNF-α and IFN-γ in rats by subjecting them to chronic mild stress. In this rat model of depression, 60 minutes of swimming exercise 5 days a week for 4 weeks decreased the levels of pro-inflammatory cytokines TNF-α and IFN-γ and alleviated the depressive-like behavior. 24 Recently, Agudela and associates showed how the muscle protein PGC-1α1 were involved in resilience of stress-induced depression by modulating kynurenine metabilosm and reducing inflammation in the brain. 25 The effect of exercise on psychiatric symptoms in animal models of neurodegenerative diseases has not yet been studied although exercise seems to protect patients with PD from depressive symptoms. 26 Furthermore, training also reduced the levels of TNF-α and IL-1β in the hippocampus and prevented cognitive impairment in a mouse overexpressing a mutated form of the amyloid precursor protein (APP) 19 or intracerebroventricular injection of beta-amyloid, 27 which leads to formation of amyloid beta (Aβ) plaques and AD pathology. On the contrary, in a brain ischemia study Ding et al found increased expression of TNF-α in cortical as well as subcortical structures of the brain following a few weeks of 30-minute treadmill running per day at a speed of 15 m/min. 28 In this study, the authors suggested that exercise indeed leads to increased levels of TNF-α, but they claimed that this increase is gradual and slower than the increase of TNF-α after an ischemic attack. This slow increase in TNF-α may prevent subsequent extensive immune activation. It was found in the same study that rats subjected to this training preconditioning before cerebral ischemia had a better outcome with less intracellular adhesion molecule 1 (ICAM-1) and leukocyte infiltration in the brain.
It is well established that exercise induces IL-6 in the muscle tissue and that its concentration in the blood can increase up to 100-fold during exercise. IL-6 can suppress the function of pro-inflammatory cytokines such as TNF-α and IL-1β by increasing soluble interleukin-1 receptor antagonist (IL-1RA) and tumor necrosis factor receptor 1 (TNFR1). 29 The systemic increase of IL-6 after exercise is typically followed by an increase in anti-inflammatory response, for example, production of IL-1RA, an endogenous antagonist to IL-1β and IL-10. Anti-inflammatory cytokine IL-10 can cause suppression of pro-inflammatory cytokines as well as a downregulation of adhesion molecules, such as ICAM-1, thereby reducing the infiltration of immune cells. 30 Therefore, muscular IL-6 is rather suggested to have anti-inflammatory functions instead of pro-inflammatory functions. 31 In the brain, IL-6 predominantly comes from activated astrocytes. 32 Under normal conditions, IL-6 is involved in the regulation of appetite and energy homeostasis in the hypothalamus. In contrast, circulating IL-6 increased rapidly during exercise, whereas the production of IL-6 in the brain increases slower.33,34 However, IL-6 levels were found to be increased in CSF 24 hours after exhaustive swimming in rats. 35 In the brain, IL-6 levels are usually low but can increase in response to damage. Funk et al found that neuroprotection is dependent on induction of IL-6 in response to exercise. 36
Some studies have failed to demonstrate any effects of exercise on cytokine levels. It should be mentioned that cytokines are rapidly degraded in tissue and blood, and depending on the specific tissue and analytical instrument, failure in quantification does not necessarily mean unaltered cytokine levels. Many studies show that physical exercise leads to decreased levels of pro-inflammatory cytokines such as TNF-α and IL-1β and increase in anti-inflammatory cytokines such as IL-10.37,38 The effects of physical exercise on the immune system can differ between the CNS and periphery. For example, it has been shown that running for up to 105 minutes per day with speeds up to 28 m/min induced an increase in TNF-α in intestinal lymphocytes in female mice with a simultaneous decrease in the hippocampus. 39 The anti-inflammatory effects of physical exercise can also be an effect of reduced visceral fat in response to exercise since an excess of visceral fat is considered as a source of chronic low-grade systemic inflammation.31,40
Since many cytokines have been shown to have different effects in various contexts, it might be difficult to conclude whether or not a training paradigm is beneficial by simply measuring the levels of specific cytokines. The effects of different training interventions on cytokine levels is important to investigate, but the results must be considered in relation to data regarding the status of other signaling molecules and cells of the nervous system to understand the function of each cytokine in its context. Furthermore, the source and function of specific cytokines sometimes differs between human and mouse, making it even more complicated to draw correct conclusions from studies in mice.41,42 For example, the inflammatory enzyme iNOS, known to be important for microglial/macrophage activation, 43 is suggested to be induced by different cytokines in man (eg, IFN-α/β) compared to mouse (eg, IFN-γ). Also, the expression levels of inflammatory receptors can be different in humans compared with mouse microglia. 42 Possibly, a panel of several cytokines that can easily be measured in blood by multiplex enzyme-linked immunosorbent assays (ELISA) could provide a biomarker to monitor and optimize exercise in neurodegenerative diseases. A future clinical development could be to monitor pro- and anti-inflammatory cytokine profiles in different patient situations. Based on such profiles it might be possible to individualize training programs in order to achieve the best possible outcome in terms of an anti-inflammatory response.
Microglial Activity
In recent years, there has been increased interest in how physical exercise affects microglial cells, the primary immune cells in CNS. Microglia resemble the macrophages found in the periphery. They have many different functions depending on their activation subtype. The pro-inflammatory subtype, referred to as M1, secretes pro-inflammatory cytokines and free radicals that are toxic to the surrounding cells. On the opposite side of the spectrum, alternatively activated macrophages, referred to as M2, are involved in resolving the inflammation by secreting anti-inflammatory cytokines and promote tissue healing by secreting trophic factors. These M1 and M2 phenotypes are typically used in characterizing microglia and macrophages, but recently more phenotypes have been suggested. 44 Different types of toll-like receptors (TLR) are expressed on different subtypes of microglia and harmful molecules, that is, contents of pathogens can bind to these receptors and induce different intracellular signaling through, for example, NFκB.
Already in 1988, McGeer et al found extensive activation of microglia in the SN in patients with PD. 45 An increased level of inducible NO synthase (iNOS), a marker of pro-inflammatory M1 subtype microglia, has also been detected, 46 as well as an upregulation of inflammatory TLR in the striatum 47 that are important pro-inflammatory signaling receptors on the surface of microglia. Some scientists also suggest that inflammatory pathways are hyperactivated with age, which may facilitate neurodegeneration. 48
The effects of physical training on microglia phenotype and activation have been investigated in animal models of neurodegeneration. In the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) mouse model of PD, Sung et al demonstrated that 30 minutes of treadmill exercise 5 days a week at speeds up to 12 m/min could spare nigrostriatal neurons and improve motor behavior and at the same time reduce microglial activation by decreasing the expression of the inflammatory enzyme iNOS involved in microglial oxidative burst. 49 Moreover, treadmill exercise for 30 min/day with an inclination of 10% and a speed of 5 m/min also decreased the microglial activation in the hypothalamus of mice fed with a high-fat diet. 50 There are also studies showing that running exercise for 60 min/day at 12 to 20 m/min might lead to a switch from pro-inflammatory M1 subtype macrophages into alternative M2 subtypes in adipose tissue.40,51 Exercise can also lead to a decrease in TLR expression in macrophages. 52 Treadmill running for 30 min/day at 12 m/min in rats even lead to a reduced overexpression of toll-like receptor 4 (TLR4) and toll-like receptor 2 (TLR2) in the ischemic brain tissue following brain ischemia. 53 The same study also showed that exercise downregulated downstream targets of these pro-inflammatory receptors, such as myeloid differentiation primary response gene 88 (MyD88) and NFκB. Activating these target pathways often lead to the expression of other pro-inflammatory proteins such as cytokines.
Exercise also seems to affect the alternative activation of macrophages. Duman et al suggests that exercise reduces the depressive-like behavior in mice through an increase in the growth hormone insulin growth factor 1 (IGF-1) in the prefrontal cortex and hippocampus.54,55 IGF-1 is suggested to have an anti-inflammatory effect and to be implicated in the neuroprotective activation of macrophages and is connected to the “good” M2 phenotype.54,55
There is evidence that physical exercise affects microglial cells in the brain. So far, very few studies have made profound efforts to investigate the effects on different subtypes of microglia and their activation profiles. From available experimental data, it is tempting to believe that physical exercise can alter the inflammatory mode of microglial cells. Hence, changing their phenotype from a pro-inflammatory mode (M1) to an anti-inflammatory phenotype (M2) will provide the neuropathological brain with trophic support. There is still much to be learnt in this area as to what type of exercise could be specifically beneficial for certain neurodegenerative diseases. Animal models could be valuable to understand which optimal training intervention is useful for patients with brain disorders. Functions and availability of specific immune cells such as macrophage-like cell types sometimes differ between human and mouse as mentioned above. 41 However, most of the immune functions and expression profiles are very similar. 56
The Effect of Adrenergic Signaling on the Immune System
Exercise causes a release of stress hormones, such as adrenaline and cortisol, in the body. Adrenaline and noradrenaline are the fast-acting fight or flight hormones produced by the adrenal glands on direct signaling from the brain in a stressful situation, but they are also released on exercise. Cortisol is a steroid hormone also produced by the adrenal glands. It is not released instantly as adrenalin, but rather takes minutes to be released since it involves step-wise activation of the amygdala. This then activates the hypothalamus to release corticotropin-releasing hormone (CRH), which tells the pituitary gland to release adrenocorticotropic hormone (ACTH) and finally signals to the adrenal glands to produce cortisol. Both adrenaline and cortisol rapidly increases during physical training and these hormones have been suggested to be responsible for the beneficial effects of exercise. 40 The mechanism behind this function could be related to their anti-inflammatory role. Cortisol is a well-known immunosuppressor, and adrenalin can bind to several different adrenergic receptors to exert its functions. Stimulation of the β2-adrenergic receptor (β2AR) on microglia inhibits their activation in both the lipopolysaccharide (LPS, an experimentally well-used inflammatory agent that binds to Toll-like receptor 4) and MPTP mouse models of PD by inhibiting NFκB. 57 These findings were also confirmed in neuron-glia cultures from mice and rats that specifically addressed the effect of microglial cells on neurons. Indeed, exercise affects adrenalin receptor signaling since the levels of β2AR on the cell membrane of macrophages are downregulated following exercise in humans as well as in mice.58,59 Macrophages from mice running at 18 m/min for 30 min/day expressed higher amounts of interleukin-12 (IL-12) following in vitro treatment of LPS, a toxin used to induce expression of IL-12 and other pro-inflammatory cytokines. This increase in IL-12 expression in macrophages from exercised mice was caused by the decreased levels of β2AR. The authors suggest that downregulation of β2AR in response to exercise can improve the pro-inflammatory thymus-derived helper cell type 1(Th1) mediated immune response induced by IL-12. Another study suggested that the downregulation of β2AR only occurred in overtrained subjects and not after moderate exercise. This implies that these receptors still function normally and thus inhibit expression of pro-inflammatory cytokines such as IL-12 after moderate exercise. 60
Although the adrenergic response is only related to the time of exercise bout, the long-term effect on inflammation could be significant. It remains to be investigated if these effects can also be seen in microglia in the brain. The contribution of stress hormones for the beneficial effect of exercise in brain diseases can be addressed in experimental animal models by using genetically modified animals or a pharmacological approach. It is tempting to speculate that the specific effect of high-intensity exercise is more likely to result in a rapid surge in stress hormones compared with low-intensity exercise, and therefore is more beneficial for some clinical conditions.
Exercise and Levels of Neurotrophic Factors
Neurotrophic factors are also highly investigated to understand the molecular mechanisms behind the neurodegeneration observed in diseases such as PD. Neurotrophic factors are naturally secreted from cells and act on receptors on the surface of neurons to promote their survival. Two of the most potent growth factors in the CNS, brain-derived neurotrophic growth factor (BDNF) and glial-derived neurotrophic factor (GDNF), have been extensively studied in animal models of PD and clearly demonstrated a neuroprotective effect, 61 even though clinical trials have not yet been successful. 62
It has been suggested that neurodegeneration might be due to a lack of these trophic factors to support neuronal survival. It has also been proposed that finding ways of increasing the levels of these factors might serve as a good treatment for neurodegeneration.63,64
The 6-hydroxydopamine (6-OHDA) model of dopamine deficiency, where 6-OHDA is injected either into the striatum or the medial forebrain bundle (MFB), is one of the most well-studied rat models of PD. Cohen and coauthors showed that 7 days of forced forelimb use prior to injection of 6-OHDA blocked behavioral deficits, attenuated the striatal dopamine loss, and increased the GDNF levels in the striatum that corresponded to the overused limb. 65
Similar protective effects of exercise have been described when training is initiated after 6-OHDA lesion with training periods for 10 days 66 and up to 4 weeks after the 6-OHDA injection that further showed striatal upregulation of BDNF and GDNF. 67 A study started treadmill training 3 days after 6-OHDA injection and still detected behavioral improvement, but without histological differences in the enzyme thyrosine hydroxylase important for dopamine production. 68 In a recent report using the 6-OHDA rat model, neither pre-treadmill training nor initiation of treadmill training 7 days after 6-OHDA injection provided any behavioral benefit, which the authors claim could be related to the stress of shock-motivated forced running procedure. 69 We have also found that treadmill training in mice can induce a stress response and increase inflammation in the brain.
Huntington’s disease (HD) is another neurodegenerative disease with primarily brain atrophy of striatum caused by a mutation within the Huntingtin gene. A recent report, and the only clinical randomized controlled study looking at physical activity in HD, demonstrated a significant functional benefit of physical activity in patients with HD. 70 Experimental mouse models of HD, with overexpression of the Huntingtin gene, have shown typically mild effects of exercise on motor performance. Amelioration of cognitive impairment after wheel running has been reported, but less/lack of effect on motor coordination and without effects on the BDNF levels in the brain.71,72 Another study shows delay of the onset of motor deficits after wheel running but without improvement in brain pathology. 73 In view of the data showing enhanced HD disease process in marathon runners, 74 it is of great value to address how the exercise administration can be optimized in terms of quantity and intensity to have a beneficial effect.
One study in mice treated with LPS systemically revealed that the beneficial effects of exercise were due to the activation of the BDNF tyrosine-related kinase B (BDNF-TrkB) signaling pathway in neurons. 75 This explanation is also supported by Wu and coauthors who showed that treadmill running of 10 m/min for up to 60 min/day restored LPS-impaired neurogenesis. 76 In another study using the MPTP mouse model of PD, the authors showed that treadmill running for 40 min/day at speeds up to 15 m/min protected the animals from motor deficits where animals also demonstrated increased BDNF levels in the SN as well as mitochondrial dysfunction. 77 However, there are also studies showing that physical exercise has neuroprotective effects without any increase in BDNF or any other neurotrophic factors. 36 The timing of exercise interventions might be of great importance, and if the aim is to prevent a disease through mechanisms of neuroprotection, the intervention should occur in an early phase. Preconditioning with physical exercise is well known to ameliorate acute ischemic brain damage in rodents involving restraining of immunological processes, 78 such as TLR4 signaling, 79 that are known to have a similar protective role in experimental neurodegenerative diseases.80,81 It is therefore reasonable to presume that physical exercise also can delay the onset or progression of neurodegenerative diseases via immunomodulatory mechanisms. However, many studies reveal that programs of higher intensities can be beneficial for neuroprotection in the early phase of a neurodegenerative disease and in the later phase for neuroplasticity and recovery. 82
In summary, it has been shown that physical exercise might affect the levels of BDNF. Nevertheless, it still has to be defined which types of physical exercise cause which effects on BDNF levels, in which regions of the brain, and if there are differences regarding protection and repair. Studies linking the effect of BDNF to other effects of physical exercise such as the inflammatory status are also needed.
Neuroplasticity and Immune Function
In previous sections we have discussed the strong effect of physical exercise on the immune system and how the inflammatory state can be altered. In recent years, it has become clear that the immune system is implicated in neuroplasticity. An early report within this field of brain plasticity showed how peripheral T lymphocytes recognize antigens in the brain (self-specific T cells), opposite to their function in autoimmune diseases, and promote recovery following CNS trauma. 83 For T cells to have this positive effect on the injured brain, the normal naïve T cells could be insufficient, and immune boosting by therapeutic vaccination, for example, modified myelin self-antigen, could mitigate the consequences of CNS injuries. 84 The positive effect of enriched environment on hippocampal neurogenesis has also been linked to the immune system. Ziv et al demonstrated how neurogenesis in the hippocampus, induced by enriched environment, was associated with microglia activation and T cell recruitment. 85 Importantly, they use immune-deficient mice (lacking T- and B-cell populations) and show that the neurogenic effect of enriched environment was lost. In mice where microglia was inhibited or mice lacking T-cells specifically, the CNS antigen myelin basic protein had reduced neurogenesis. Immune deficient mice were also bad in hippocampal learning tasks. Interestingly, both learning ability and neurogenesis correlated positively with BDNF protein expression in naïve and immune deficient mice, suggesting that positive immune mechanisms also involve growth factors as discussed in previous sections. Interestingly, the importance of a specific type of anti-inflammatory monocytes has been shown to be important for the differentiation of naïve T cells into protective and plasticity-prone T cells (Treg and Th2 cells), suggested to be a relevant immunomodulation of multiple sclerosis. 86 A recent study made in healthy men demonstrated that physical exercise direct monocytes into an anti-inflammatory mode (eg, downregulation of TNF and TRL4), 87 which are likely to be a mechanism that support brain plasticity.
It is important to mention that classical pro-inflammatory trigger of the immune system have adverse effect on brain plasticity and neurogenesis 88 and inhibition of this pro-inflammatory response can restore hippocampal neurogenesis. 89 Therefore, it is important to mention that it is not the number of immune cells but their specific phenotype and inflammatory contribution that count for its specific role on brain plasticity and neurogenesis. 90 Physical exercise with its strong effect to increase basal neurogenesis/plasticity is likely to have an additional beneficial effect in the diseased brain by also alter the state of inflammation.
The ageing of the brain and thereby senescence of microglia is believed to be related to increased detrimental activation of microglia. Microglia in the aged brain has reduced ability in normal functions, such as clearance and migration, reduced ability to shift a pro-inflammatory to an anti-inflammatory state, and regulate plasticity and repair mechanisms. 91 The positive effect of physical exercise in elderly people are therefore likely to reduce these age-related processes in the brain and postpone the onset of neurodegenerative diseases. This effect of physical exercise is expected not only to be related to neuroprotection but also neuronal plasticity.
Future Developments and Clinical Implications
In conclusion, there is evidence that exercise affects several important and beneficial processes in the noninjured as well as in the injured brain. The processes concerned in this review can be seen in Figure 1. In summary, exercise can lead to increased levels of neurotrophic factors well as changes in the levels of different cytokines and altered microglial functions in different parts of the brain that could be beneficial for patients with neurodegenerative diseases. Exercise has also been shown to affect cell surface receptors, such as the TLR and adrenergic receptors, as well as intracellular signaling molecules involved in inflammatory pathways. However, more research is needed to determine which type of exercise, in particular the intensity of exercise, results in the most beneficial effects in different animal models and groups of patients.
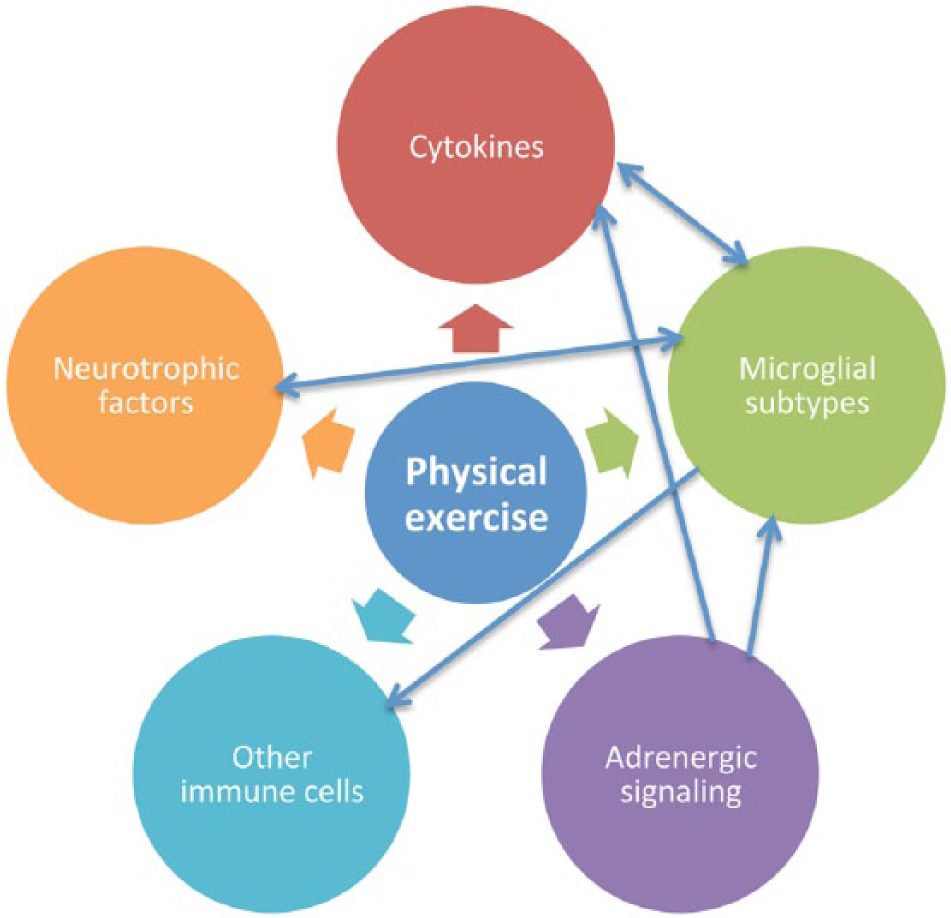
Effects of exercise on several physiological and pathophysiological processes affecting the brain.
One specific area of interest is the impact of intensity in relation to duration and timing of the exercise intervention. For example, it has been shown that exercise was not beneficial in mice with traumatic brain injury when subjected to voluntary running shortly after the trauma. 38 High-intensity exercise consisting of running for 45 minutes at speeds up to 21 m/min in mice has also been shown to cause memory impairments that might be due to the psychological and oxidative stress associated with the forced high intensity.92,93 Studies outside of the nervous system indicate that exercise of various intensities can affect the balance of T cells differently.94,95 High-intensity training consisting of running for 60 min/day at 26 m/min with 10% inclination in mice has been shown to increase anti-inflammatory cytokines and decrease pro-inflammatory cytokines, 96 cytokines that are strongly correlated with neuroprotection. 97 Another study showed that exercise of moderate intensity lowers the levels of pro-inflammatory cytokines more than mild intensity training in diabetic patients. 98 Table 1 displays different setups in terms of intensity, frequency, and duration that have been used in animal experiments discussed in this review.
To our knowledge, no studies have investigated the effect of training intensity on neuroinflammation and neurodegeneration. Thus, studies investigating the effect of training intensity on the neuroinflammatory profiles such as cytokine expression and different subtypes of microglia in different brain regions are highly warranted. Many of the studies of high-intensive training define high intensity in such a way that the mice should be capable of running at a certain speed for 30 to 60 minutes. No studies have investigated the effects on neuroinflammation and neurodegeneration of high-intensity interval training lasting for only a short period of time, say a few minutes. Training setups of this kind have been shown to be very effective in improving insulin sensitivity after only 2 weeks in young men. 99 Such interval training paradigms are possible to be conducted in mice since studies investigating other effects of interval training have been possible to complete in most common laboratory mice.100-103 These studies remain to be tested in mouse models of neurodegenerative disorders.
To be able to translate experimental studies to the clinic, appropriate and available biomarkers associated with exercise-induced neuroprotection and restoration in experimental animals need to be established. These markers should preferably be investigated using noninvasive technology, for example, brain-PET for the direct effect on the brain, or blood samples that could provide reliable information (soluble molecules or blood cell alterations) related to exercise-induced neuroprotection, plasticity, and neurorestoration. To be available for most patients, a simple blood test that could be analyzed at smaller health care centers is of course preferable. If reliable biomarkers for “neuroprotective exercise” can be established in the experimental setting, such candidates can then be validated in the clinic. It is likely that several individual biomarkers will be needed, or global gene/protein assays. Recent data demonstrate that the responsiveness to exercise in terms of aerobic fitness is highly individual. Most likely, personalized medicine also applies to exercise, where different patients will benefit more from specific forms of training.
A great challenge for future research will be to elucidate how changes across studies and animal models should be interpreted for clinical translation. Animal models represent a great opportunity for investigating new and controversial effects of exercise but such studies also poses several limitations. Rodents have a significantly shorter lifespan compared with humans, and a majority of the models used in research studies discussed in this review consists of animals without comorbidities such as diabetes and high blood pressure. Most of the animals are also very young males, which means that results from such studies do not take into account the physiology of females and aging individuals. A common problem using male mice to study behavior and effects of exercise is the fact that males from many commonly occurring experimental stains are rather aggressive after forced exercise and when subjected to stress during behavioral tests. This leads to fighting between the mice, causing increased stress levels, which, in turn, undoubtedly affects the results. Therefore, measuring the stress response is important for correct interpretation of the results. It also remains to be determined how large changes in the target response in animal studies that is required in order to achieve relevant effects in patients. A change in a biomarker in an experimental animal does not necessarily correlate to behavioral improvements of that animal or symptomatic relief in patients. This research field still offers great opportunities for new discoveries, as more studies are needed to elucidate the likely range of intensity, duration, frequency, and type (aerobic or anaerobic, exercise or physiotherapy) of exercise that is required to induce important target responses. An obstacle to exercise research might be to be able to force the animals to run at higher intensities without at the same time inducing negative stress and fear that will affect the target responses studied. It could also be a challenge and a potential health risk to expose older patients to exercise with high intensities. Another challenge is motivation, particularly in older patients or those with lifestyle-related comorbidities such as diabetes and obesity. Despite these obstacles and great challenges we still believe that there is great potential for research of this kind to develop and lead to new pioneering discoveries that could significantly improve the prevention and treatment of neurodegenerative diseases.
Footnotes
Acknowledgements
We are grateful to Philip Rosvall for his input to the article and to Sonia George who kindly proofread the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been supported by the Strategic Research Area MultiPark at Lund University, Lund, Sweden, the Swedish Research Council, Grant No. 2012-2229, by the A.E. Berger Foundation, Gyllenstiernska Krapperup Foundation, Wiberg Foundation, Bergvall Foundation, G&J Kock Foundation, Swedish National Stroke Foundation, Swedish Parkinson Foundation, Stohnes Foundation, and the Royal Physiographic Society.
