Abstract
Background. Transplantation of bone marrow stromal cells (BMSCs) may contribute to functional recovery after stroke. This study was designed to clarify their mechanisms, trophic effects of neurotrophic factors, and neural differentiation. Methods. Mouse neurons exposed to glutamate were cocultured with mouse BMSCs. Either neutralizing antibodies against brain-derived neurotrophic factor (BDNF) or nerve growth factor (NGF) or Trk inhibitor K252a was added to explore the mechanism of their protective effects. Fluorescence in situ hybridization (FISH) was used to assess BDNF or NGF mRNA expression in BMSCs. The mice were subjected to permanent focal ischemia, and 7 days later, either BMSCs or the vehicle was stereotactically transplanted into the ipsilateral striatum. The mouse brains were processed for FISH and immunostaining 2 or 4 weeks after transplantation. Results. BMSCs significantly ameliorated glutamate-induced neuronal death. Treatment with anti-BDNF antibody significantly reduced their protective effects. FISH analysis showed that the majority of BMSCs expressed BDNF and NGF mRNA in vitro. BMSC transplantation significantly improved the survival of neurons in peri-infarct areas. FISH analysis revealed that approximately half of BMSCs expressed BDNF and NGF mRNA 2 weeks after transplantation; however, the percentage of BDNF and NGF mRNA-positive cells decreased thereafter. Instead, the percentage of microtubule-associated protein 2–positive BMSCs gradually increased during 4 weeks after transplantation. Conclusions. These findings strongly suggest that BDNF may be a key factor underlying the trophic effects of BMSCs. BMSCs might exhibit the trophic effect in the early stage of cell therapy and the phenotypic change toward neural cells thereafter.
Keywords
Introduction
Bone marrow stromal cells (BMSCs) are known to hold great potential as a cell source for rescue from ischemic stroke.1-5 When BMSCs were transplanted into animal models of ischemic stroke, the cells could survive in the ischemic brain, migrate toward the cerebral infarct, and promote functional recovery.6-8 Although several hypotheses have been proposed, the exact mechanisms underlying the efficacy of BMSC transplantation remain relatively unknown. 9 First, several reports supported the hypothesis of BMSC differentiation into neural cells in the host brain (transdifferentiation). This mechanism is based on the findings that BMSCs simulate neuronal morphology and express specific neural markers in vitro10,11 or in vivo.1,12 Reportedly, certain BMSC subpopulations can alter their gene expression profiles and differentiate into neural cells in response to the surrounding environment. 13 Second, BMSCs can reportedly fuse with host neurons and simulate neural differentiation (cell fusion). 14 However, the roles and fates of the fused cells remain unknown. 9 Finally, there is increasing evidence that BMSCs can produce neuroprotective or neurotrophic factors and support the survival of host neural cells.7,9,15,16 This “nursing functions” to the damaged brain tissue17,18 may play a key role in the efficacy of BMSC transplantation; however, some questions regarding these mechanisms still remain unanswered. Thus, the present study was designed to clarify the mechanisms of BMSC neural differentiation and the subsequent trophic effect through the nourishing of damaged neurons in ischemic cerebral tissue with neurotrophic factors, particularly brain-derived neurotrophic factor (BDNF) and nerve growth factor (NGF).
Materials and Methods
Isolation and Culture of Mouse BMSCs
BMSCs were harvested from 8-week-old male Balb/c mice (CLEA Japan, Inc, Tokyo, Japan) or transgenic mice expressing enhanced green fluorescent protein (EGFP; The Jackson Laboratory, Bar Harbor, ME) using previously reported procedures. 6 In brief, the mouse femur was dissected, and the marrow was extruded with Dulbecco’s modified Eagle’s medium (DMEM; Sigma-Aldrich, St Louis, MO) containing 10% fetal bovine serum (FBS; Life Technologies, Carlsbad, CA), 100 U/mL of penicillin/streptomycin (Life Technologies), and 10% heparin (Mochida Pharmaceutical Co, Ltd, Tokyo, Japan). Whole marrow cells were placed in a culture flask coated with type I collagen (Becton, Dickinson and Company, Franklin Lakes, NJ) in DMEM/10% FBS. After 24 hours, the nonadherent cells were removed for use in subsequent experiments after 3 passages. All the animal experiments were approved by the Animal Studies Ethical Committee of Hokkaido University Graduate School of Medicine (Sapporo, Japan).
Three-Dimensional (3D) Coculture of BMSCs and Neurons
For the 3D coculture experiments, 1 × 105 mouse BMSCs were plated onto 24-well cell culture inserts (pore size, 1.0 µm; Becton, Dickinson and Company) and incubated for 3 days in DMEM/10% FBS at 37°C in an atmosphere of 5% CO2. The primary-cultured neurons were obtained by dissociating a frozen form of fetal mouse cerebral cortex (Nerve Cell CX <M>; Sumitomo Bakelite Co, Ltd, Tokyo, Japan). A total of 2 × 105 neurons were seeded into each well of 24-well poly-D-lysine-coated plates containing nerve cell culture medium (Sumitomo Bakelite) and cultured for 14 days. Following this, BMSCs were added and cocultured for 24 hours using a previously reported 3D coculture technique. 9 Subsequently, the neurons were exposed to 100 µM glutamate for 10 minutes and again cocultured with BMSCs for 72 hours. To investigate the protective effect of BMSC-secreted BDNFs on neurons against glutamate toxicity, neutralizing antibodies against either BDNF (Promega, Fitchburg, WI)19,20 or NGF (Sigma-Aldrich)21,22 or the Trk tyrosine kinase inhibitor K252a (Millipore Corp, Billerica, MA) were added to the cocultures.
Cell Viability Assay
Cell viability was analyzed using the LIVE/DEAD cell viability assay (Life Technologies) after 72 hours of glutamate exposure. In brief, the neurons were rinsed with phosphate-buffered saline (PBS) and subsequently incubated with viability assay solution containing 2.0 mM calcein AM and 4.0 mM ethidium homodimer I in PBS for 30 minutes at room temperature (RT). After incubation, the cells were washed with PBS and then stained with Hoechst 33342 nucleic acid stain (Life Technologies). Cell viability was analyzed in each group (n = 5) using the IN Cell Analyzer 2000 Imaging System (GE Healthcare, Little Chalfont, UK).
Fluorescence In Situ Hybridization (FISH) of Cultured BMSCs
Mouse BMSCs were seeded onto type I collagen-coated culture slides (Becton, Dickinson and Company) and cultured for 3 days. FISH analysis of cultured BMSCs was performed using the ISHR Starting Kit (Nippon Gene, Tokyo, Japan). Digoxigenin-modified RNA probes for NGF were prepared using complementary DNA included in the ISHR Starting Kit. Meanwhile, probes for BDNF were prepared using plasmid DNA (clone ID: 5730414D07; DNAFORM, Yokohama, Japan). After fixation and acetylation, the slides were incubated in 50% formamide and 2× sodium chloride-sodium citrate solution for 30 minutes at 42°C as a prehybridization step. Hybridization for each probe was performed overnight at 42°C. After hybridization, the slides were washed to clear excess RNA and then blocked with 10% goat serum for 30 minutes. Following this, the slides were sequentially incubated with sheep polyclonal antibody against digoxigenin (dilution 1:100; Roche Diagnostics, Indianapolis, IN) for 2 hours at RT, treated with Alexa Fluor 594-conjugated secondary antibodies (dilution, 1:200; Molecular Probes, Eugene, OR) for 1 hour at RT, and subsequently counterstained with 4′, 6-diamidino-2-phenylindole (DAPI; Sigma-Aldrich). Fluorescence was observed through each appropriate filter using a fluorescence microscope (BX61; Olympus, Tokyo, Japan) and was digitally photographed using a cooled charged-coupled device (CCD) camera (model VB-6000; Keyence Corporation, Osaka, Japan).
Permanent Middle Cerebral Artery (MCA) Occlusion and BMSC Transplantation
Permanent MCA occlusion and stereotactic BMSC transplantation were performed as described previously. 6 In brief, 8-week-old male Balb/c mice (n = 20) were anesthetized, and a skin incision was made between the right eye and ear. Following this, the temporal muscle was mobilized, the skull was exposed, and a burr hole was drilled. The main trunk of MCA was exposed and directly occluded with a bipolar coagulator. The core temperature was maintained between 36.5°C and 37.5°C during the procedures. This method produces a stable infarct lesion only in the ipsilateral neocortex.
BMSCs derived from EGFP mice (EGFP-BMSC) were prepared as a cell suspension in PBS. The mice were fixed under anesthesia to a stereotactic apparatus (model DKI-900; David Kopf Instruments, Tujunga, CA) 7 days after the insult. The cell suspension (2 × 105 cells in 10 µL) or vehicle was injected 2.0 mm to the right of the bregma at a depth of 3.0 mm (n = 10) using an automatic microinjection pump (model KDS-310; Muromachi Kikai Co, Ltd, Tokyo, Japan). All the animals were subcutaneously injected daily with 10 mg/kg of cyclosporine A until they were killed humanely 2 or 4 weeks after cell transplantation (n = 5). The brain was removed and embedded in paraffin, and 8-µm thick coronal sections were prepared for subsequent staining.
FISH and Immunostaining Analyses of Ischemic Brain Tissues After BMSC Transplantation
After the FISH procedure for identification of BDNF or NGF mRNA in the brain sections, we performed fluorescence immunostaining against EGFP on the basis of the procedure reported by Rylski et al. 23 After these procedures, the sections were treated for 1 hour at RT with a mouse monoclonal antibody against EGFP (dilution, 1:100; Santa Cruz Biotechnology, Santa Cruz, CA) that was previously labeled with Zenon Alexa Fluor 488 (Mouse IgG2a Labeling Kit; Molecular Probes).
Fluorescence double staining for neuronal makers and EGFP was also performed. In brief, the sections were treated with a monoclonal antibody against microtubule-associated protein 2 (MAP2; dilution, 1:100; Chemicon International, Inc, Temecula, CA) or neuronal nuclei (NeuN; dilution, 1:400; Chemicon International, Inc.) overnight at 4°C and Alexa Fluor 594-conjugated secondary antibodies (dilution, 1:200) for 1 hour at RT. Subsequently, the sections were immunostained for EGFP and then counterstained with 4′, 6-diamidino-2-phenylindole. The fluorescence was observed using a fluorescence microscope equipped with a cooled CCD camera as described above and concomitantly with a confocal laser-scanning microscope (Fluoview FV10i; Olympus).
Cell Counting for Histological Analysis
To count the NeuN-positive cells, 2 regions of interest (ROIs; 800 × 640 µm2) were set on the ischemic boundary zone and contralateral region in each animal. We also set 4 ROIs (160 × 128 µm2) around the infarct lesion to count the cells coexpressing EGFP and BDNF or NGF and set ROI (160 × 128 µm2) to count cells coexpressing EGFP and MAP2 in each animal. We used Image J image processing software (v 1.32J; National Institutes of Health, Bethesda, MD) to count the cell numbers.
Statistical Analysis
All data are expressed as means ± standard deviations. The data were compared using the unpaired t test for cases involving 2 groups or 1-factor analysis of variance followed by Fisher’s protected least-significant-difference post hoc test for cases with more than 2 groups. A probability (P) value <.05 was considered statistically significant.
Results
Neuroprotective Effect After Coculture With BMSCs
Fetal mouse cortical neurons in culture appeared healthy with few dead cells (Figure 1A), whereas the cultured neurons exposed to 100 mM glutamate appeared injured with an increased rate of mortality 72 hours after exposure. Even the living cells appeared swollen with involvement. After the insult, only 40.7% ± 14.3% (vs control) of the neurons survived at 72 hours (Figure 1B). The coculture with BMSCs significantly ameliorated glutamate-induced neuronal injury. Thus, the neurons maintained normal morphologies, and 104.8% ± 30.4% survived (P < .01, Figure 1B). When a high concentration (20 µg/mL) of neutralizing antibody was used against BDNF, the neuroprotective effect was significantly canceled (Figure 1A). Under these conditions, a significant reduction in cell viability at high concentrations of the neutralizing antibody was observed. The neuronal death rate decreased with a decrease in antibody concentration (20 µg/mL, 49.7% ± 5.0%, P < .05; 2 µg/mL, 83.3% ± 5.8%; Figure 1B). Similarly, when a high concentration of anti-NGF antibody or K252a was used, cell viability also tended to decrease (100 µg/mL of neutralizing antibodies against NGF, 85.7% ± 23.1%; 200 µM K252a, 53.8% ± 19.9%; Figure 1B). These results demonstrated that BMSCs protected the neurons against glutamate toxicity by secreting BDNF.
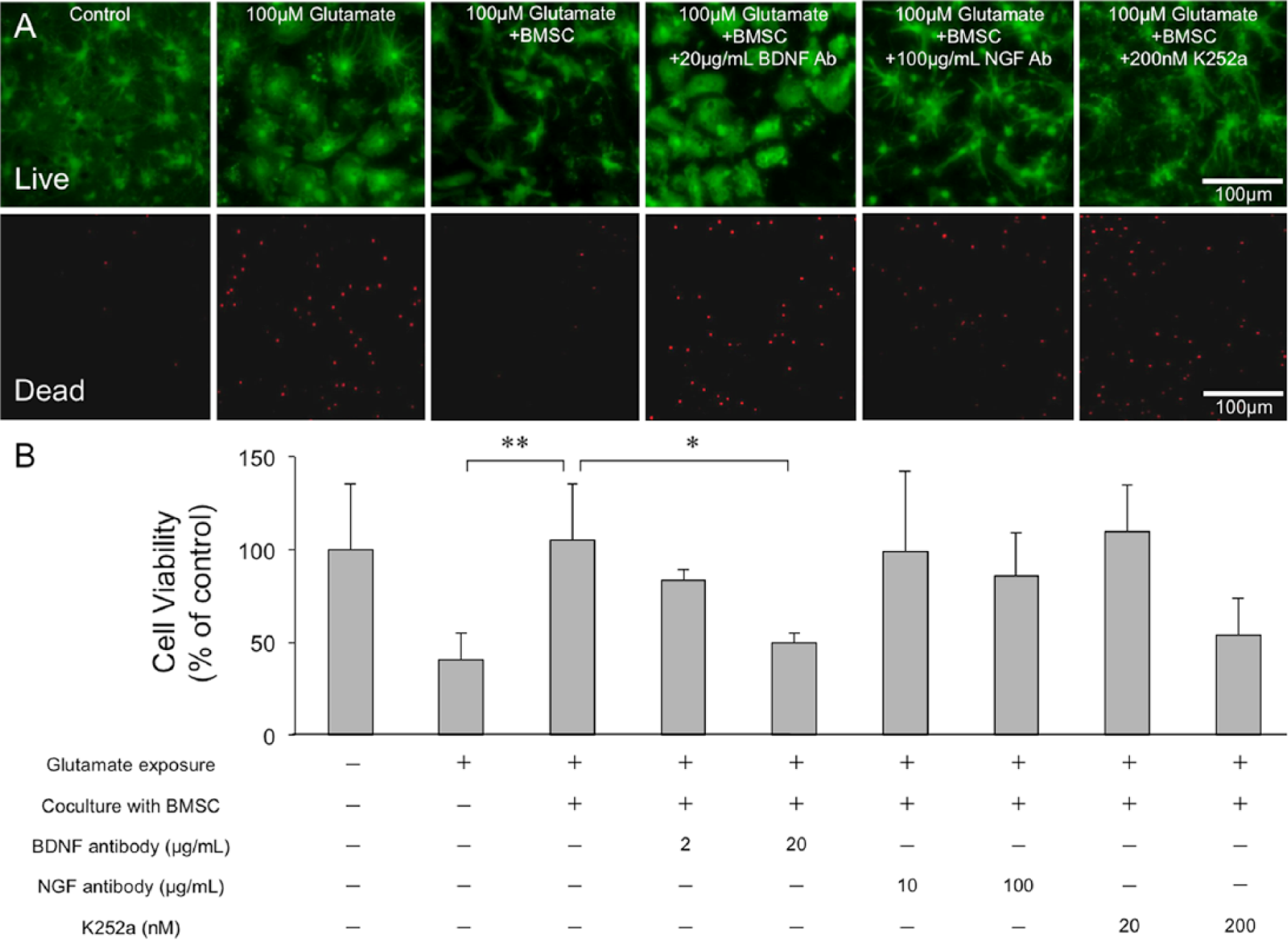
Neuronal cell viability assay after coculturing with BMSCs: Panel A shows a representative photomicrograph from the neuronal cell viability assay. The upper panels show living cells (green), and the lower panels show the nuclei of dead cells (red) in the same areas. Controls are depicted in the left panels. The caption “100 µM glutamate” indicates that the cells were exposed to 100 µM glutamate and “+ BMSC” indicates that the cells were cocultured with BMSCs. Moreover, “+20 ng/mL BDNF Ab,” “+100 ng/mL NGF Ab,” or “+200 nM K252a” indicate cells treated with each neutralizing antibody or drug (scale bar, 100 µm). The bar graph (B) shows the viability of neurons cocultured with BMSCs for 72 hours after glutamate exposure. Values are presented as the percentages of living cells as determined by the cell viability assay. The numerals on the X-axis indicate the concentrations of each additive to the coculture system. Error bars represent standard deviations.
Production of BDNF and NGF by Cultured BMSCs
FISH analysis with antisense RNA probes showed that most cultured BMSCs expressed both BDNF and NGF mRNA (Figure 2). In most BMSCs, these mRNAs were localized to the center of the cell body, adjacent to the nucleus. In contrast, FISH analysis with sense RNA probes for either BDNF or NGF revealed no positive cells (Figure 2). Quantitative analysis indicated that 89.1% ± 2.9% and 82.6% ± 7.3% of the cultured BMSCs expressed BDNF and NGF mRNA, respectively.
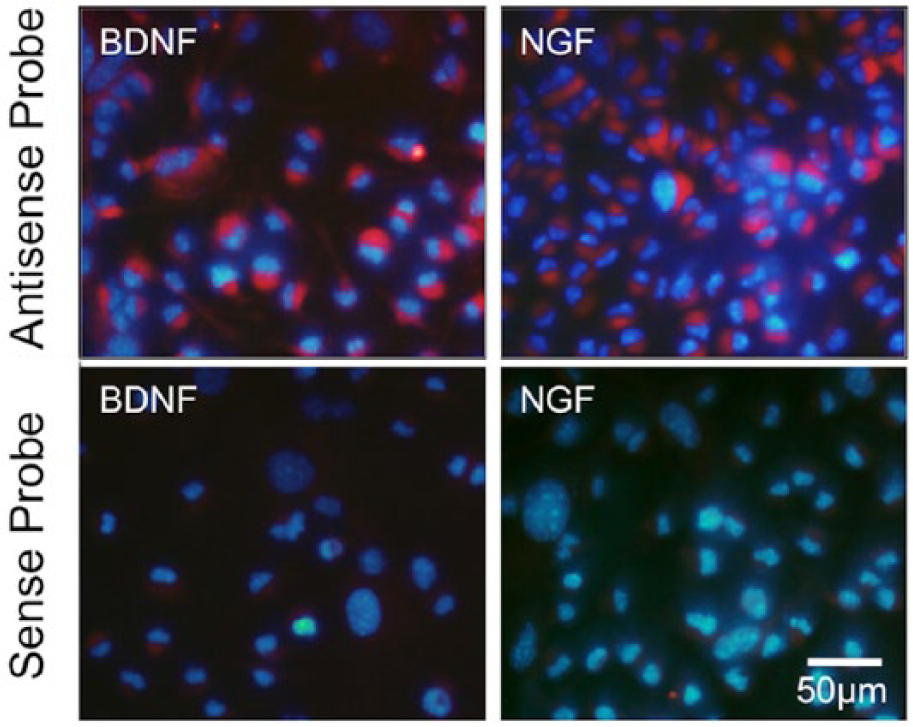
FISH analysis of cultured BMSCs: Panel A contains a representative photomicrograph of cultured BMSCs processed for FISH analysis using an RNA probe for either BDNF (left) or NGF (right). Upper, antisense-RNA probes; lower, sense-RNA probes as negative controls; red, mRNA; blue, 4′, 6-diamidino-2-phenylindole (scale bar: 50 µm).
Neuroprotective Effect Around the Ischemic Boundary Zone
Two weeks after transplantation (3 weeks after ischemia), the necrotic changes were complete, and brain atrophy was observed in the ischemic core lesions of both the groups. There was no difference in the infarct areas between the 2 groups (BMSC, 19.9% ± 6.2%; vehicle, 19.7% ± 3.9%). In the peri-infarct area, some neocortical neurons were clearly damaged (Figure 3A). In the vehicle-transplanted animals, the density of NeuN-positive cells in the peri-infarct area decreased to 73.2% ± 9.2% of the density in the contralateral side. On the other hand, BMSC transplantation significantly ameliorated the neuronal loss in the peri-infarct area (92.4% ± 9.7%, P < .05; Figure 3C).
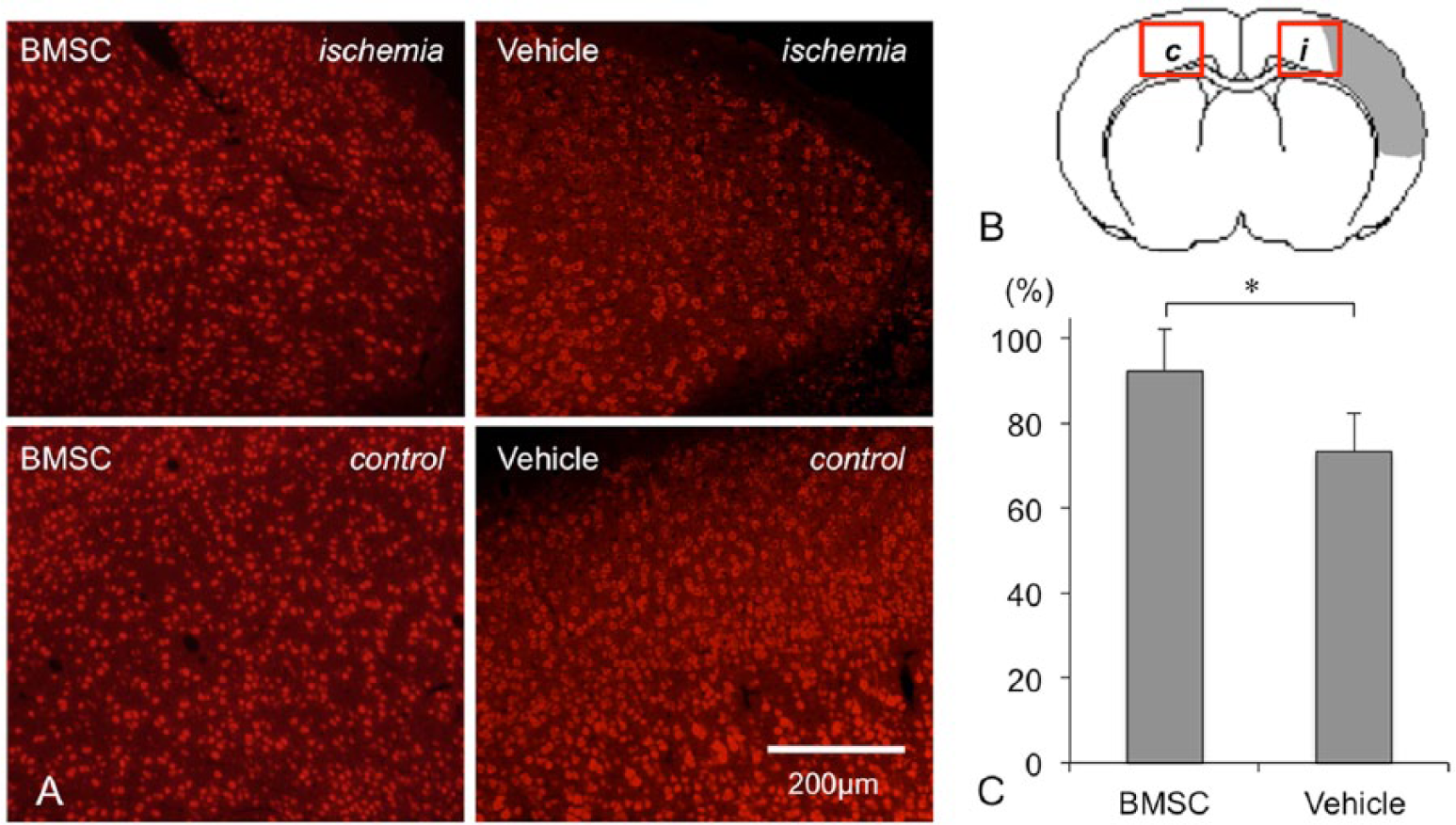
Immunostaining for NeuN in the peri-infarct area: panel A contains a representative photomicrograph of brain sections processed for immunostaining against NeuN in the BMSC group (left) and the vehicle group (right). Upper, ischemic side; lower, contralateral side as controls (scale bar: 200 µm). Panel B shows a schematic image of the host brain. The box on the ischemic boundary zone (i) and the box on the contralateral site (c) indicate the locations of each ROI. The bar graph (C) shows the density of NeuN-positive cells in the peri-infarct area (Y-axis, percentage of positive cells vs control). Error bars represent standard deviations.
Production of BDNF and NGF Around the Ischemic Boundary Zone
Most EGFP-positive cells were observed in the peri-infarct area (Figure 4). In addition, 3D analysis using the confocal laser-scanning microscope showed that some EGFP-positive cells expressed each mRNA in the cell body (BDNF, Figure 5A; NGF, Figure 5B). At 2 weeks after transplantation, 55.9% ± 12.7% of the EGFP-expressing cells in the peri-infarct area also expressed BDNF mRNA (Figure 4A). Similarly, 56.6% ± 14.3% were also positive for NGF mRNA (Figure 4C). At 4 weeks after transplantation, however, the percentage of both BDNF and NGF mRNA-positive cells significantly decreased to 35.0% ± 13.9% and 34.1% ± 9.7%, respectively (Figures 5B and 5D, P < .05).
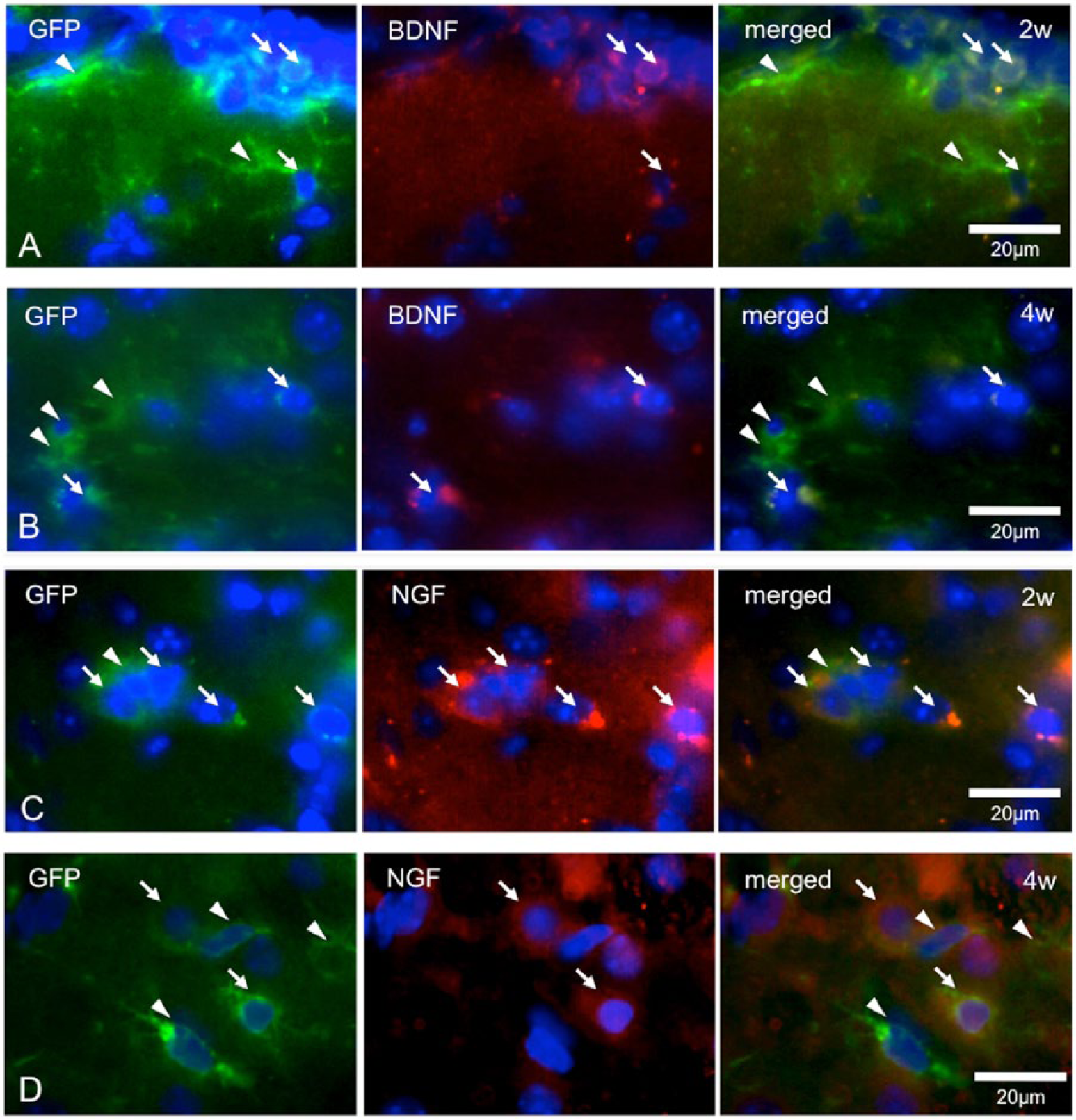
FISH analysis to determine the expression of BDNF or NGF mRNA in vivo: the panels show representative photomicrographs of the sections processed for FISH analysis using an antisense probe for BDNF (A and B) or NGF (C and D). They are fluorescent micrograms of the peri-infarct areas in animals 2 (A and C) and 4 (B and D) weeks after cell transplantation. The left line shows EGFP-positive cells (green), the center shows BDNF-positive or NGF-positive cells (red), and the right shows their merged images. Arrows, cells double positive for EGFP and BDNF; arrowheads, cells positive for EGFP and negative for BDNF (scale bar: 20 µm).
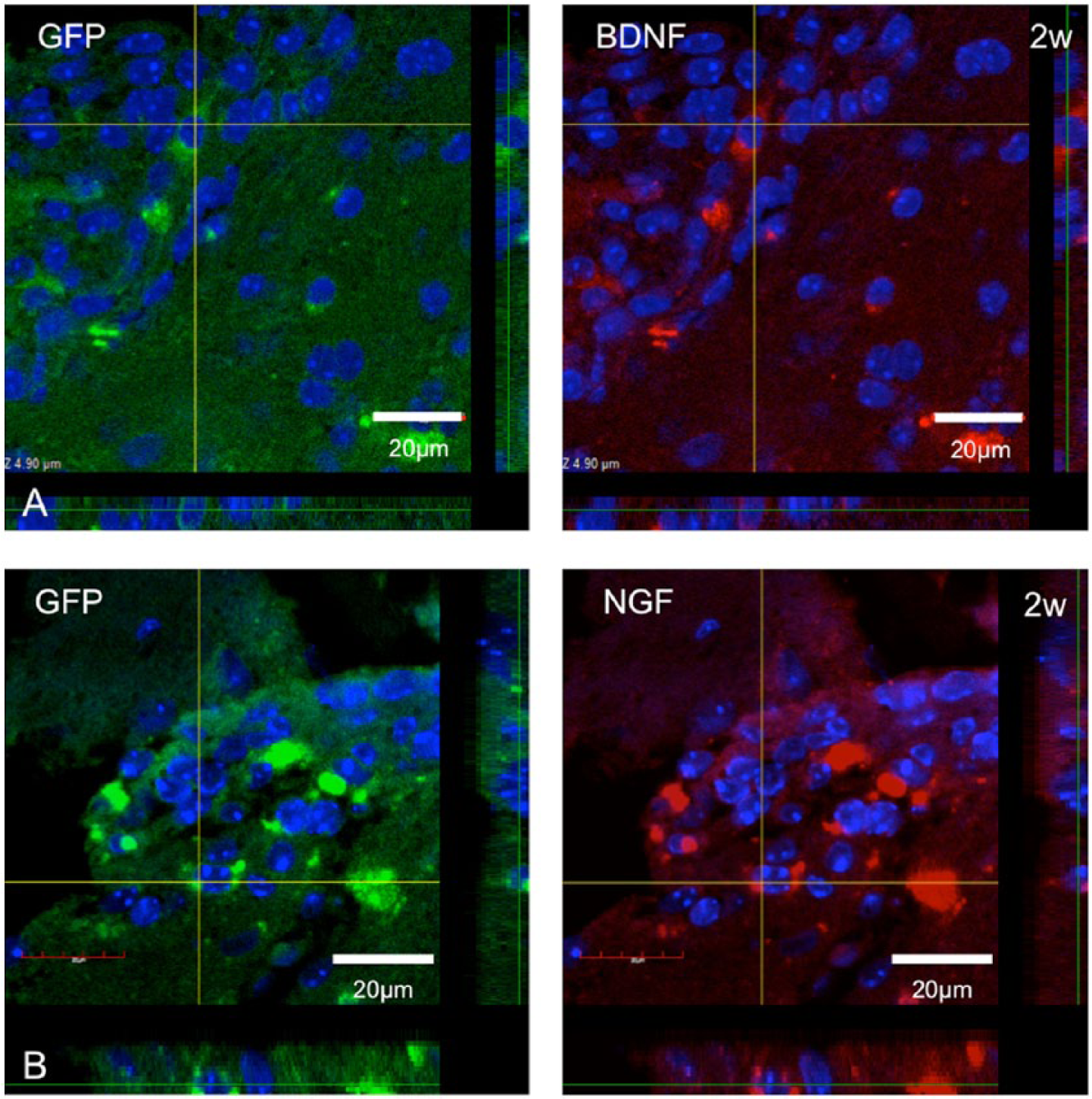
Three-dimensional (3D) imaging of FISH analysis: the panels show the 3D images in the peri-infarct area 2 weeks after transplantation using a confocal laser-scanning microscope (A, EGFP and BDNF; B, EGFP and NGF). The colocalized cells placed on intersections were projected as 3D images inside and under bars of each panel (scale bars: 20 µm).
Phenotypic Change Toward Neural Cells in BMSCs
Double immunostaining for EGFP and NeuN showed that a number of EGFP-positive cells already migrated from the transplantation site to the peri-infarct area 2 weeks after transplantation. Of the migrated donor cells, we observed that some had a tendency for neural differentiation, whereas others did not (Figure 6). Double immunostaining for EGFP and MAP2 also revealed that the number of cells coexpressing the 2 markers in the peri-infarct area gradually increased over a 4-week period after transplantation (Figure 7). Thus, the percentage of cells double positive for GFP and MAP2 significantly increased from 26.2% ± 9.2% at 2 weeks to 73.9% ± 7.2% at 4 weeks (P < .01). Some of these cells exhibited morphological changes to neuronal cells (Figure 7).
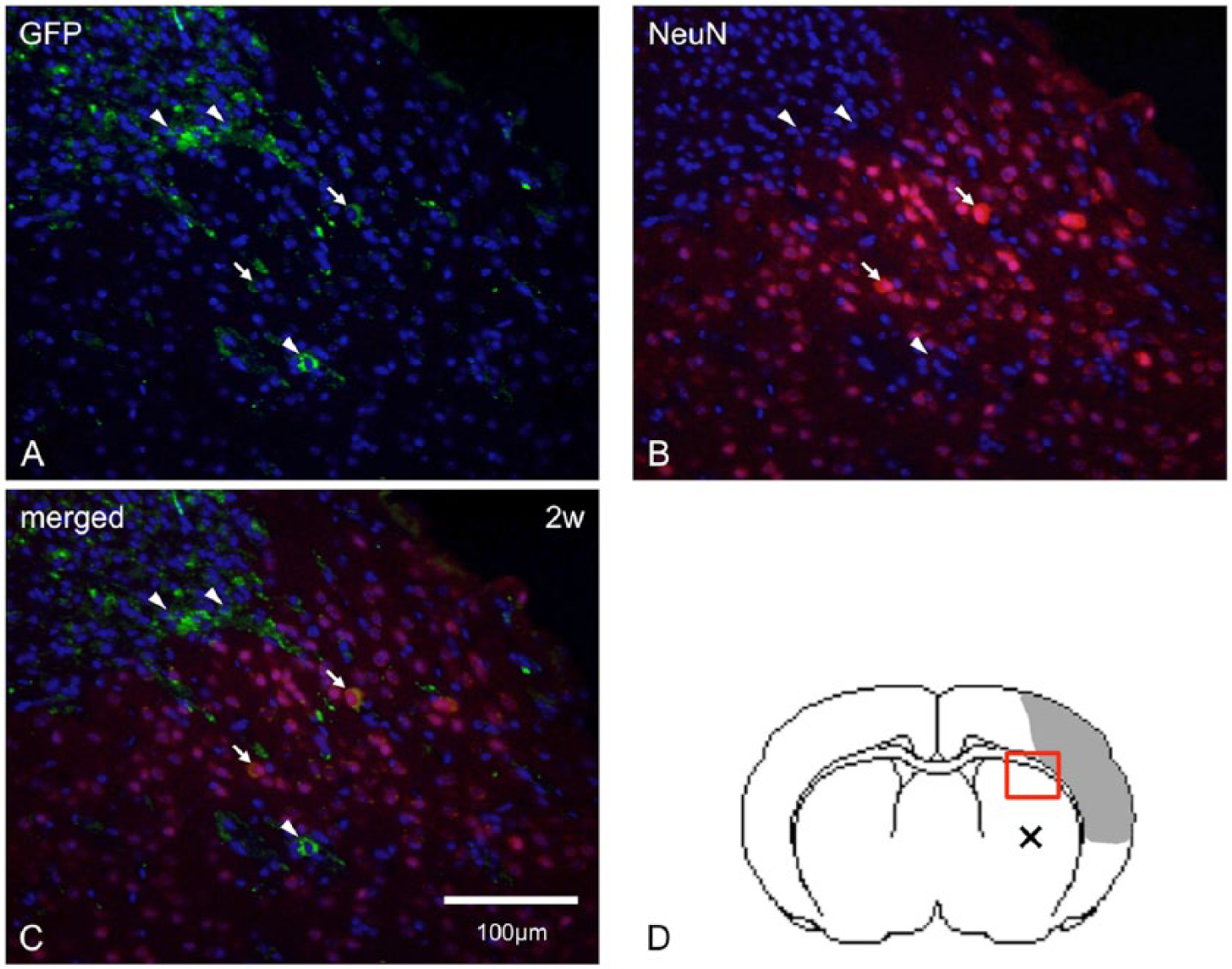
Double immunostaining for EGFP and NeuN: the panels show representative low-power photomicrographs after double immunostaining for EGFP and NeuN of the peri-infarct area 2 weeks after transplantation. EGFP-positive cells (green in A), NeuN-positive cells (red in B), and their merged images (C) are depicted. Panel D shows a schematic image of the host brain. The box and “×” in panel D indicate the locations of the photomicrographs and the cell transplantation sites, respectively. Arrows, cells double positive for EGFP and NeuN; arrowheads, cells positive for EGFP and negative for NeuN (scale bars: 100 µm).
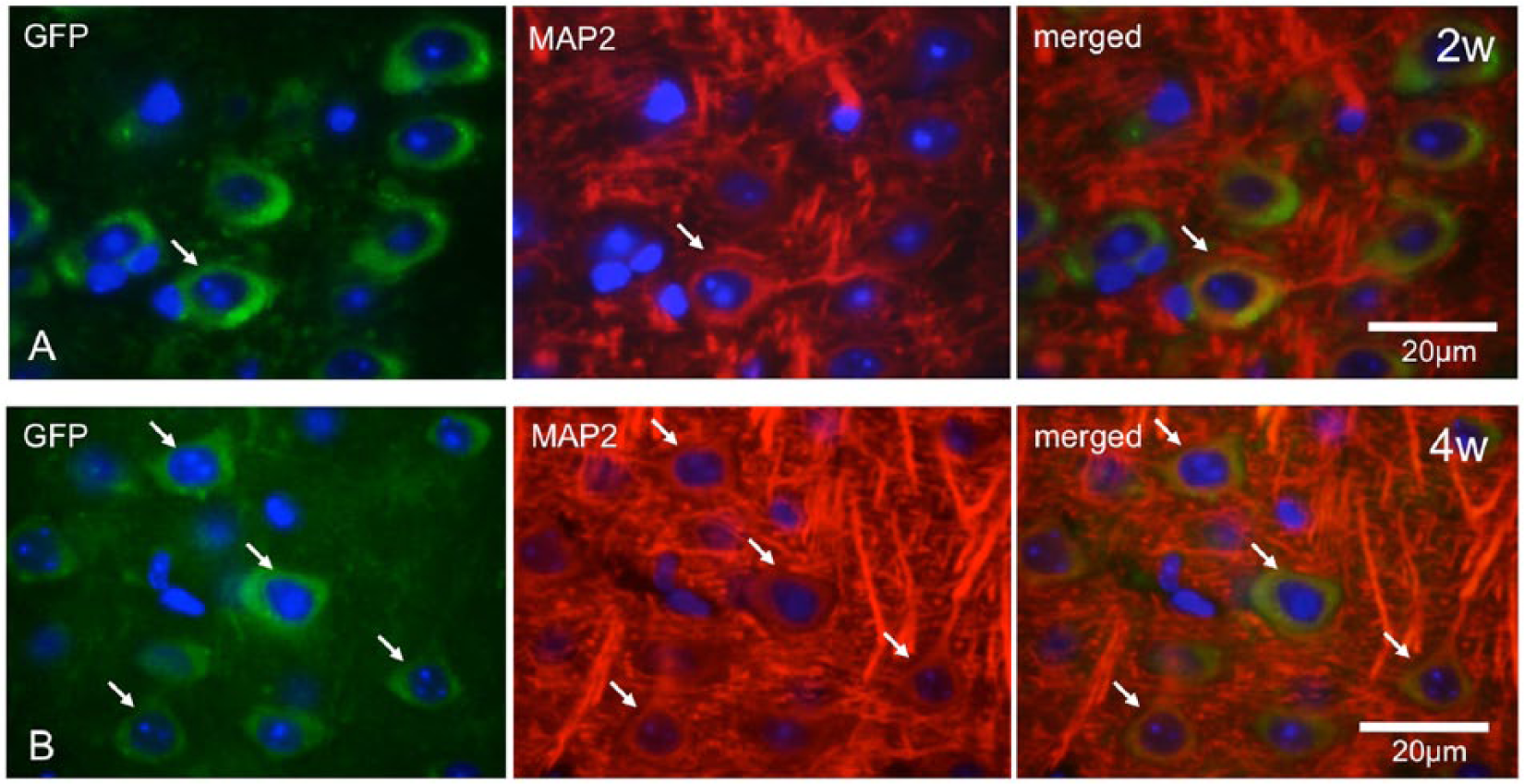
Double immunostaining for EGFP and MAP2: the panels show representative photomicrographs of brain sections processed for double immunostaining for EGFP and MAP2 of the peri-infarct area in animals 2 (A) and 4 (B) weeks after cell transplantation. EGFP-positive cells (green, left), MAP2-positive cells (red, center), and their merged images (right) are depicted. Arrows, cells double positive for EGFP and MAP2 (scale bars: 20 µm).
Discussion
The results of the present study clearly demonstrated that BMSCs can promote the survival of neurons exposed to glutamate by producing Trk-activating factors such as NGF and BDNF in vitro, and their biological activities can be confirmed in the rodent brain. We found that more than half of the engrafted BMSCs expressed NGF and BDNF mRNA, which contributed to neuron survival in the peri-infarct areas during the first 2 weeks after transplantation. However, the number of BMSCs that expressed mRNA for BDNF or NGF decreased over an additional 2 weeks, whereas the number of MAP2-positive BMSCs rapidly increased (Figure 8A). These findings strongly suggested that the engrafted BMSCs were biologically heterogeneous and contributed to functional recovery after ischemic stroke through multiple mechanisms with biphasic temporal profiles (Figure 8B). Thus, a certain BMSC subpopulation may promote the survival of host neurons in the peri-infarct area by BDNF secretion in the early posttransplantation stage (up to 4 weeks). However, BMSC survival in the host brain may be rather short-lived. Subsequently, another BMSC subpopulation may begin to proliferate and differentiate into neural cells, thereby regenerating cells in the infarcted brain around 4 weeks after transplantation. Thus, the ratios of cells double positive for EGFP and MAP2 were 26.2% ± 9.2% and 73.9% ± 7.2% at 2 and 4 weeks after transplantation, respectively. To the best of our knowledge, this is the first study to show that engrafted BMSCs contribute to protect the host brain tissue against cerebral ischemia through a trophic effect in the early stage of cell therapy and “neural differentiation” thereafter.
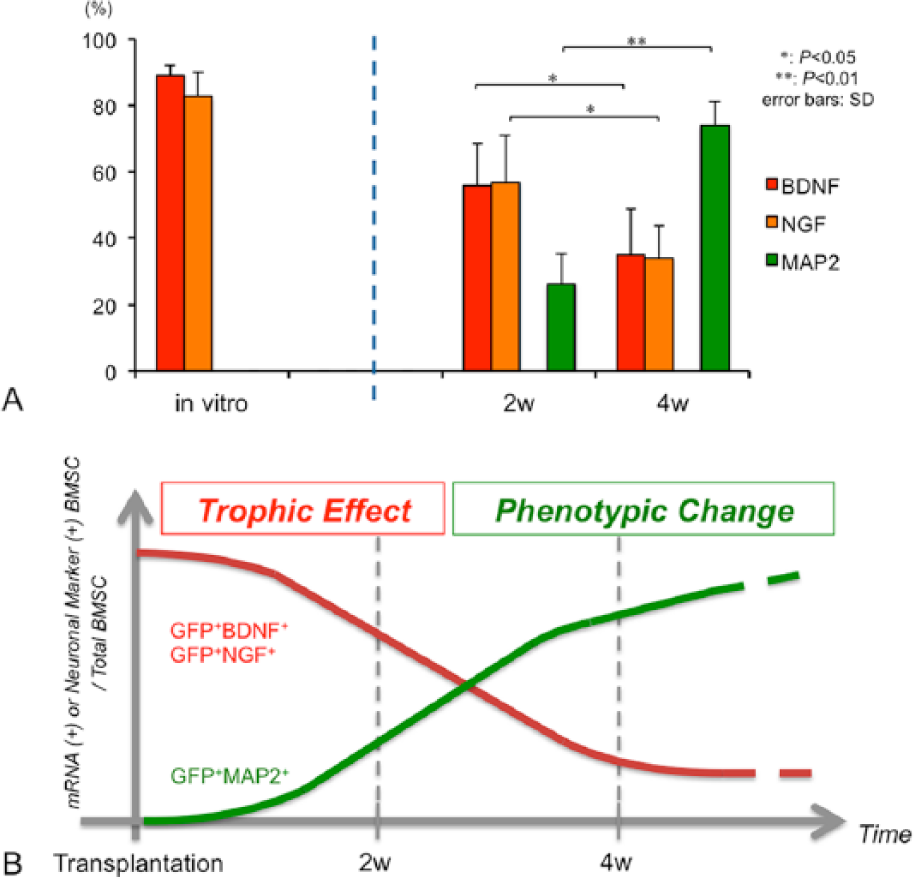
Quantitative analysis for FISH and immunohistochemical analyses and schematic images of therapeutic mechanisms: the bar graph (A) shows the percentage of cells double positive for EGFP and BDNF (red), for EGFP and NGF (orange), and for EGFP and MAP2 (green) in the total EGFP-positive cell population in vitro or in the peri-infarct area of the mouse ischemic brain (2 and 4 weeks after transplantation). The error bars represent standard deviations. Panel B contains a schematic image of the multiple therapeutic mechanisms of BMSC transplantation. More than half of the engrafted BMSCs expressed BDNF and NGF mRNA, which contributed to neuronal survival in the peri-infarct area during the first 2 weeks after transplantation. However, the number of BMSCs expressing mRNA decreased over an additional 2 weeks (trophic effect, red line). On the other hand, the number of MAP2-positive BMSCs rapidly increased (phenotypic change, green line). This schematic image shows that the engrafted BMSCs were biologically heterogeneous and contributed to functional recovery after ischemic stroke through multiple mechanisms with biphasic temporal profiles.
As mentioned above, BMSCs may produce some neuroprotective factors that support the survival of host neural cells. This hypothesis is reasonable because BMSCs per se support the localization and proliferation of hematopoietic cells in the bone marrow by producing various cytokines. BMSCs reportedly release soluble neuroprotective factors, including BDNF, NGF, stromal cell–derived factor 1α, hepatocyte growth factor, transforming growth factor β1, and insulin-like growth factor 1, and significantly ameliorate glutamate-induced neuronal damage. 9 Furthermore, in the present study, the BMSC-conditioned medium activated the phosphorylation of mitogen-activated protein kinase/extracellular signal–regulated protein kinase and/or phosphoinositide 3-kinase/serine/threonine kinase in primary cultures of rat dorsal root ganglion neurons. 24 In 2011, He et al 25 reported that BMSCs significantly increased the expression of basic fibroblast growth factor, BDNF, and vascular endothelial growth factor in the ischemic brain. These findings strongly suggest that BMSCs trigger endogenous signaling pathways of survival and repair in neurons by secreting soluble neurotrophic factors.
BMSCs are thought to differentiate into neural cells in the host brain. Although this hypothesis is quite attractive, several questions still remain unanswered. Recent studies have shown that BMSCs can undergo alterations in gene expression in response to exogenous stimuli and increase the expression of genes related to neural cells.13,26 Using microarray analysis, Yamaguchi et al 13 reported that the expression of mesenchymal cell–related genes was significantly downregulated and that of neuron-related genes was significantly upregulated in BMSCs when chemically treated with basic fibroblast growth factor, retinoic acid, or dimethyl sulfoxide. Thus, BMSCs can acquire the neuronal phenotype under several physiological conditions. Wislet-Gendebien et al 27 reported that nestin-positive BMSCs cocultured with cerebellar granule cells express other neuronal markers and that BMSC-derived neuron-like cells fire single-action potentials in response to neurotransmitters such as glutamate. Hokari et al 9 also demonstrated that a certain BMSC subpopulation morphologically resembles neurons and expresses neuron-specific proteins without any evidence of cell fusion when cocultured with neurons. These findings strongly suggest that certain BMSC subpopulations have the potential to undergo gene expression profile modifications and can differentiate into the neural cells in response to surrounding environmental factors. More important, our present findings indicate that only the subgroup of BMSCs with the potential for neural differentiation can survive in the host brain for periods of more than 4 weeks.
Very recently, Kuroda et al 28 demonstrated the presence of multilineage-differentiating stress-enduring (Muse) cells in BMSC populations that are characterized by stress tolerance, expression of pluripotency markers, self-renewal, and the ability to generate cells with the characteristics of the 3 germ layers from a single cell. When transplanted into immunodeficient mice by local or intravenous injection, these cells integrated into the damaged skin, muscle, or liver and differentiated into cytokeratin 14-, dystrophin-, or albumin-positive cells in the respective tissues. Furthermore, they can be efficiently isolated as stage-specific embryonic antigen 3–positive cells. Unlike authentic embryonic stem cells, the proliferation activity of BMSCs is limited, and they do not form teratomas in the testes of immunodeficient mice. These findings are quite attractive because nontumorigenic stem cells with the ability to generate multiple cell types of the 3 germ layers can be obtained through easily accessible adult human mesenchymal cells without introducing exogenous genes. 28 These results strongly suggest that a certain BMSC subpopulation may possess the biological properties of neural differentiation that can contribute to the regeneration of the infarcted brain.
The present study had some limitations that might weaken the conclusions. First, we evaluated the neuroprotective effects of BMSCs by the secretion of BDNFs only in vitro. Also, the use of appropriate nonspecific IgG controls was an indispensable in vitro assay. Second, although we evaluated the tendency for neural differentiation by the expression of neural markers, we could not utilize strict colocalization of the phenotype markers (NeuN or MAP2) with the GFP label. In future studies, we plan to investigate the in vivo neuroprotective effects of small hairpin RNA interference or knockout BMSCs for BDNF or NGF to elucidate the true neural differentiation in a study on neuronal action potentials.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received the following financial support for the research, authorship, and/or publication of this article: This study was supported by Grant-in-aids from the Ministry of Education, Science and Culture of Japan (No.23390342 and No.24390336).
