Abstract
Background. Neglect is associated with disability, unawareness, poor long-term outcome, and dependence from caregivers. No randomized trial has evaluated the effects of smooth pursuit eye movement training (SPT) and visual scanning training (VST) at the bedside on these variables. Objective. To compare the effects of SPT and VST in postacute stroke at 1 month with left neglect. Methods: We carried out an assessor-blinded, randomized controlled trial. The 24 participants were randomly allocated to either SPT or VST (n = 12 each). They received 20 treatment sessions lasting 30 minutes each at the bedside over 4 weeks. Outcome measures included the Functional Neglect Index (FNI) based on 4 tasks: find objects on a tray, stick bisection, picture search, and gaze orientation. In addition, the Unawareness and Behavioral Neglect Index (UBNI) with 6 items about unawareness and 4 about neglect in activities of daily living, the Help index (required assistance in 10 functional activities), the Barthel Index, and the rehabilitation phase were rated by treatment-blinded assessors. Outcome measures were obtained before and immediately after the end of the interventions and at a 2-week follow-up. Results. Significantly greater improvements were obtained after SPT versus VST treatment in the FNI and UBNI, and there were continued improvements selectively in the SPT group 2 weeks later. Conclusions. SPT accelerates recovery from functional neglect and reduces unawareness significantly. Bedside neglect treatment using SPT is effective and feasible early after stroke.
Introduction
Spatial neglect is a challenging disorder, 1 defined as the inability to respond to sensory stimuli in the contralesional hemispace and hemibody of a neurological patient. 1 Despite spontaneous recovery, one-third of all neglect patients develop chronic neglect. 2 Moreover, the associated unawareness impairs recovery, 3 predicts poor functional outcome, 4 and interferes with the treatment of hemiparesis. 5 Unawareness is defined as the impaired insight into left-sided sensory (visual, auditory, haptic, body-related) and motor impairments (hemiparesis) after stroke. As a major source of long-term disability after stroke, neglect and unawareness are associated with an adverse rehabilitation outcome,4,6 especially for functional tasks in peripersonal space, such as finding/grasping objects on a table, finding pictures, perceiving the midline in space, or gaze orientation.7,8 Although neglect rehabilitation is effective, 9 a reduction in the number of sessions by more effective therapies might reduce rehabilitation costs. Moreover, few RCTs have been conducted,10,11 especially in the acute stage. 12 At this stage, patients are immobilized in bed, show an ipsilesional gaze deviation, 13 and unawareness, 14 making treatment difficult. Though much is known about the effects of neglect therapy on test measures, 9 little is known about its effects on disability, 15 functional neglect measures, and (in)dependence in activities of daily living (ADLs). A recent treatment study, however, clearly indicates that theta-burst neglect therapy significantly reduces neglect in ADLs. 16 It is important to note that no RCT has evaluated treatments capable of reducing the unawareness in neglect. 17 Interestingly, caloric-vestibular stimulation 18 transiently reduces unawareness, and galvanic-vestibular stimulation permanently reduces left-hand tactile extinction. 19 This suggests that repetitive application of neglect-effective treatments might cure unawareness. 20
Smooth pursuit eye movement training (SPT) using optokinetic stimulation significantly reduces visual, auditory, and haptic neglect more than other treatments.21-24 Although VST is a well-established treatment, it shows poor transfer to nontrained tasks,25,26, requires numerous treatment sessions (20-40),27,28 does not improve nonvisual neglect, 9 and requires partial awareness by the patient, which is impaired in early neglect. 14 In contrast, SPT requires less voluntary, top-down guided orientation toward contralesional stimuli. Patients follow moving stimuli with their eyes toward the neglected hemispace, which is easier than performing leftward saccades in acute stroke. 29 Although few treatments are feasible in early stroke at the bedside, patients might benefit from treatments adapted to this situation. Moreover, to date, it is unknown whether SPT and VST reduce disability, unawareness, and dependence on caregivers. Disability measures are more sensitive to behavioral neglect than paper-pencil tests, 30 and, therefore, show the patient’s functional (in)capacities in everyday life. In addition, improved awareness would be extremely beneficial because this could increase the unfavorable rehabilitation outcome.3,17
Here, we compared SPT and VST in early stroke rehabilitation as bedside treatments, with a particular emphasis on functional neglect measures, unawareness, and ADLs. Three kinds of measurements were specifically designed for this purpose: First, a functional neglect index (FNI) was derived based on 4 functional tasks assessing the patient’s performance in peripersonal space. These included finding/reaching for objects on a large tray (see Figure 1), picture search, stick bisection, and the patient’s gaze orientation (appendix). Conjugate gaze deviation is a frequent epiphenomenon of acute spatial neglect, which recovers in parallel to the disturbed visual exploration behavior, thus making it a sensitive measure of neglect severity. 31 Second, ratings of unawareness and behavioral neglect in 10 daily activities as well as the help needed with the execution of 10 daily functional tasks were obtained by treatment-blinded caregivers. Third, the patients’ overall rehabilitation status was documented by the Barthel Index (BI) 32 and the current rehabilitation phase (acute, early, or continuing rehabilitation, see the appendix), both rated as well by treatment-blinded medical staff. Based on our recent RCT, which showed significant and large improvements in visual and auditory neglect after SPT but not VST, 22 we expected significantly greater improvements after SPT than following VST also in the functional outcome measures used in the present study. SPT relies, as a bottom-up, sensory stimulation treatment, less on the awareness and compliance of the neglect patient. Because of its greater and multimodal efficacy on neglect (visual, auditory, and haptic), we hypothesized further that SPT might reduce unawareness to a greater extent than VST. Finally, from a practical-clinical viewpoint, we sought to explore whether neglect therapy can be conducted successfully at the patient’s bedside in this early phase of rehabilitation. As a matter of fact, some neglect treatments like visuomotor feedback training are feasible and effective as bedside trainings in acute patients,33,34 whereas others appear more variable in their efficacy in this early stage (prism adaptation, see Turton et al 35 for negative results and Gossmann et al 12 for positive results).
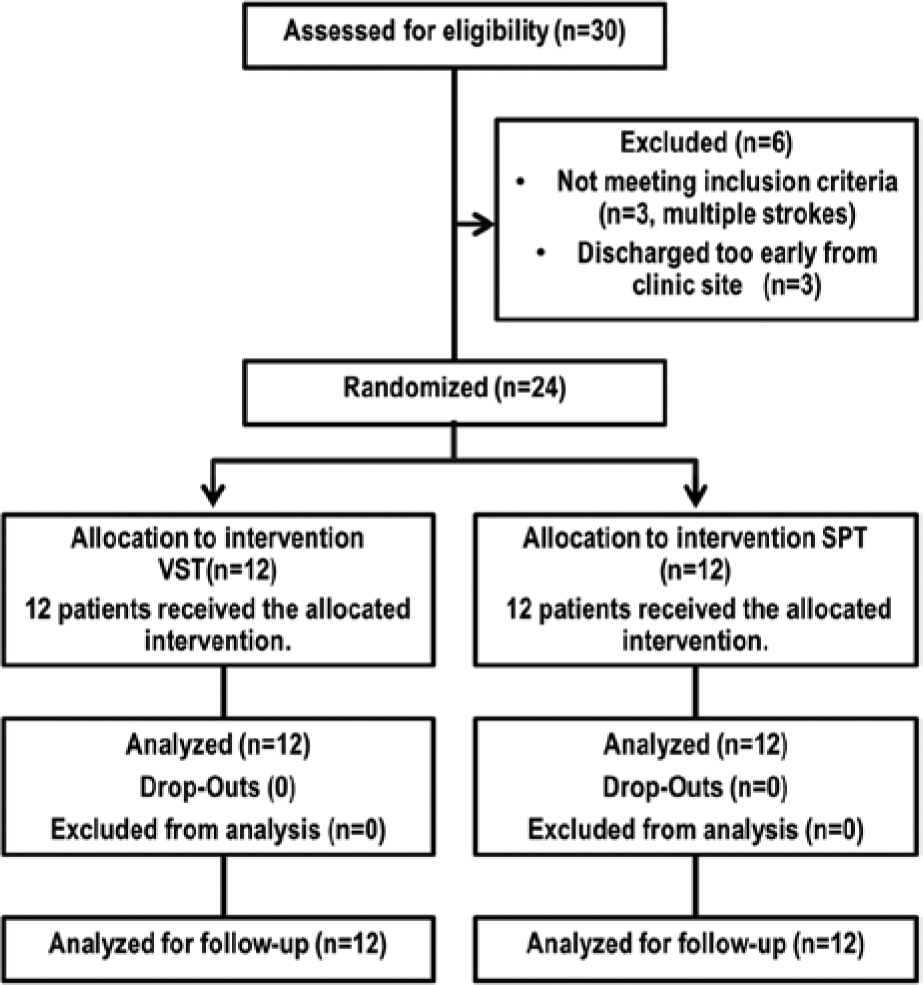
Flow chart of the study with regard to recruitment, allocation, analysis, and follow-up (see text for details).
Methods
Patients
Inclusion criteria were a single right-hemisphere stroke, visual neglect in 2 screening tests (horizontal line bisection of a 200 mm long × 10 mm broad black line, number cancellation), 22 and the ability to execute daily neglect training for 30 minutes. Neglect was diagnosed when bisection deviated >5 mm to the right side and when >1 omissions were found on the left side than on the right side of the cancellation test (see Table 1 for individual data). Exclusion criteria were psychiatric, ophthalmological, or other neurological diseases. From 30 eligible patients with left neglect caused by right-hemisphere stroke, 6 were excluded: 3 because of multiple strokes and 3 because of too early a discharge to complete the trial (Figure 2). Finally, 24 patients with acute visuospatial neglect, as assessed by the 2 screening tests, were randomly allocated to SPT or VST (by a person not involved in the study who drew lots from a sealed envelope). All patients had corrected-to-normal visual acuity, were right-handed, and gave their informed consent to participate in the study according to the declaration of Helsinki II (see Table 1 for clinical/demographic details).
Demographical and Clinical Data of 24 Patients With Left-Sided Spatial Neglect in the 2 Treatment Groups During Baseline Assessments Before Treatment.
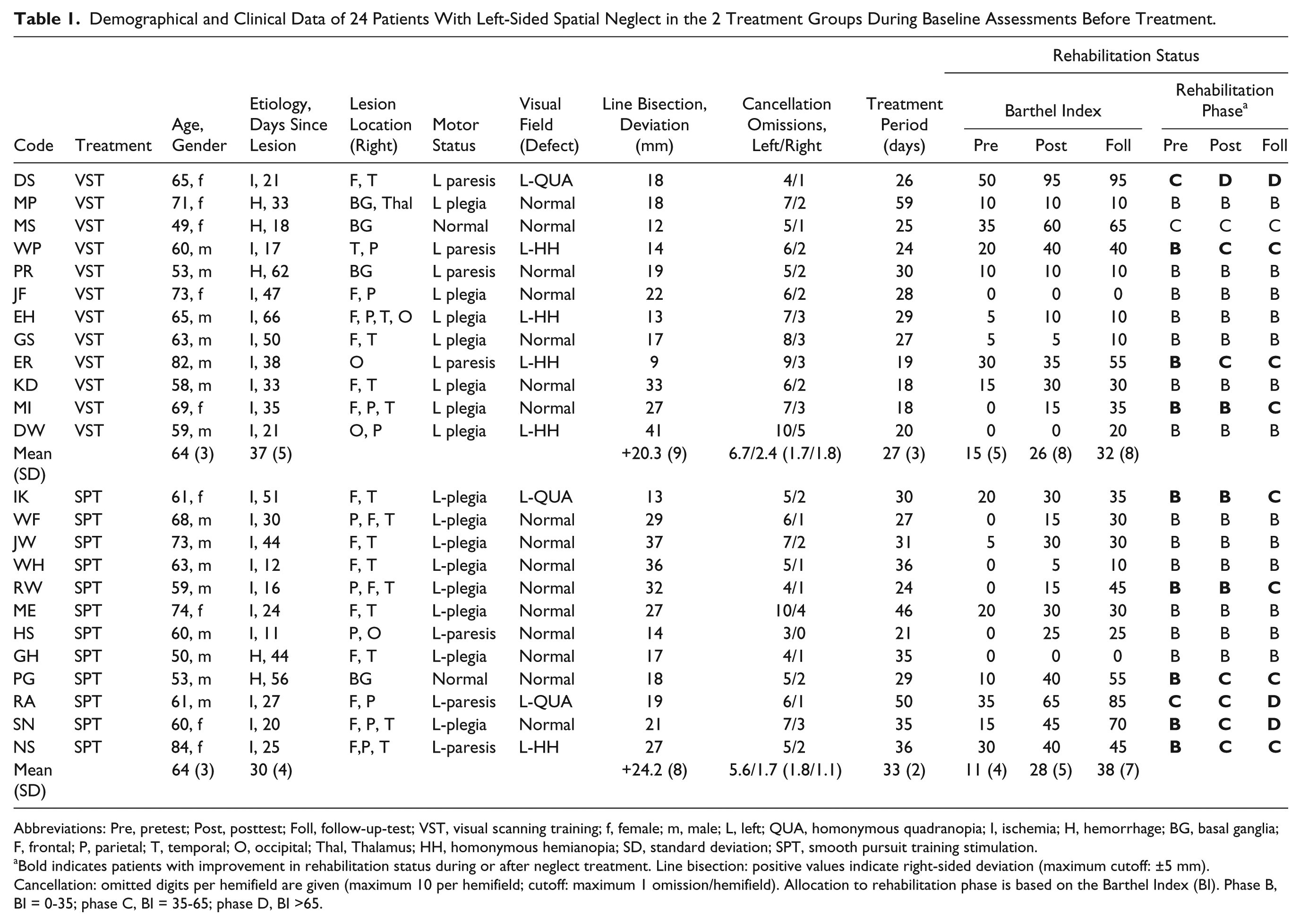
Abbreviations: Pre, pretest; Post, posttest; Foll, follow-up-test; VST, visual scanning training; f, female; m, male; L, left; QUA, homonymous quadranopia; I, ischemia; H, hemorrhage; BG, basal ganglia; F, frontal; P, parietal; T, temporal; O, occipital; Thal, Thalamus; HH, homonymous hemianopia; SD, standard deviation; SPT, smooth pursuit training stimulation.
Bold indicates patients with improvement in rehabilitation status during or after neglect treatment. Line bisection: positive values indicate right-sided deviation (maximum cutoff: ±5 mm). Cancellation: omitted digits per hemifield are given (maximum 10 per hemifield; cutoff: maximum 1 omission/hemifield). Allocation to rehabilitation phase is based on the Barthel Index (BI). Phase B, BI = 0-35; phase C, BI = 35-65; phase D, BI >65.
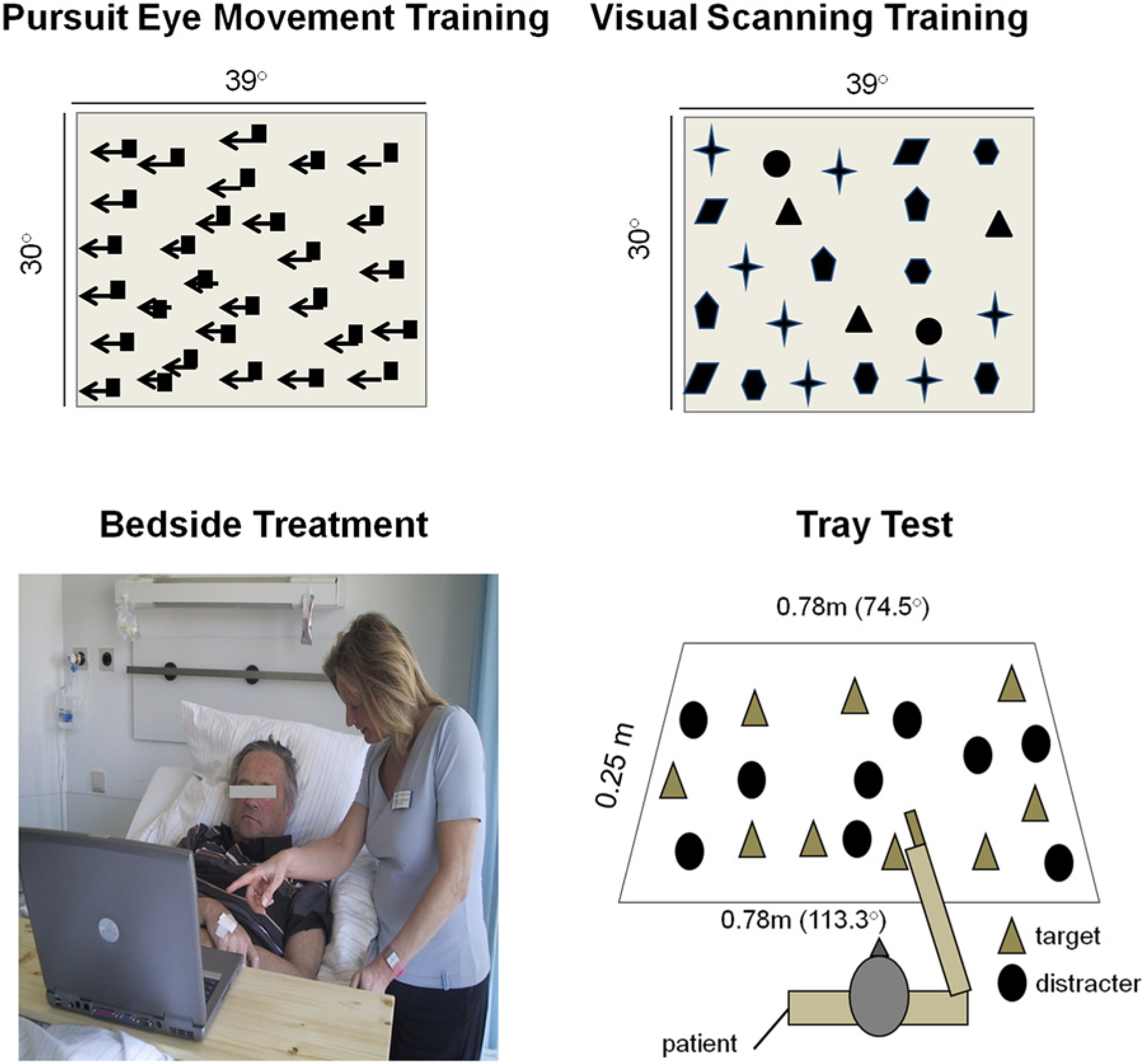
Top: Examples of smooth pursuit eye movement training (SPT) and visual scanning training (VST) designs; Arrows indicate the direction of motion on the screen during VST but were not visible during therapy. Bottom: Layout of the bedside treatment setup and spatial layout of the tray test (see text for details).
Treatments
Patients received 20 treatment sessions (30 minutes each) in total (1 session daily, from Monday to Friday) over a period of 1 month (Table 1). Both treatments were realized as bedside treatments, using a laptop with a 15-inch monitor, centrally placed at a viewing distance of 0.40 m (39.2° vs 30.1° horizontal/vertical size) in front of the patient on a board seated solidly across the patient’s bed, with the patient sitting upright in the bed (Figure 1).
During SPT, stimuli were random displays of 20 to 60 identically colored and sized squares, moving horizontally from right to left at constant velocity, providing a coherent stimulus pattern (see Figure 1 for exemplary display). The color, size (1°-2.5°), and velocity (3.1-12.6 degrees/s) of the stimuli were varied from time to time. The patient was encouraged to conduct smooth pursuit eye movements, repeatedly following the stimulus pattern from right to left, without head movements. During VST, patients viewed stationary displays of stimuli implemented in the same software. Stimuli were geometrical objects (circles, triangles, squares, etc), stars, letters, or digits (see Figure 1 for an example). The number of stimuli (5-60), their size (1°-2.5°), color, and spatial arrangement were varied (systematic versus unordered, with/without line numbering, with/without subsidiary horizontal reference lines, and with/without a red vertical “anchor line” on the leftmost side of the display). Patients were trained to scan systematically from left to right and from top to bottom, naming all objects, or counting certain stimuli. The therapist kept the patient’s head in a straight position in either treatment to promote eye instead of head movements. She watched continuously from the side whether the patient made smooth pursuit eye movements to the contralesional side (SPT) or saccadic eye movements (VST). If patients did not execute the appropriate eye movements, the instructions were repeated. If patients were tired, a short break (2 minutes) was given, adjusted to patients’ individual requirements.
Assessments and Design
The FNI included the following 4 tasks:
Tray test. A total of 18 everyday objects (pen, sponge, tea light, handkerchiefs, etc) were presented on a large ply-wooden tray (size 0.75 m × 0.25 m, 113.3° horizontal eccentricity), at a distance of 0.25 m from the patient (Figure 1). Three target and 3 distractor objects, respectively, were affixed in either of 3 imaginary segments (left, middle, and right segment) of the tray. Target objects were randomly and successively shown to the patient by the experimenter, using a demonstration object that was identical to the respective target object. Patients were instructed to explore for and subsequently grasp the target object with the right hand. The number of target objects found within a cutoff (maximum) duration of 30 s (hits) and the respective exploration duration was stopped with a stopwatch. Objects not found within 30 s were counted as omissions, with the maximum exploration duration of 30 s. Hits (maximum 3 per segment) and exploration duration for the left, middle, and right segments were recorded separately (see the appendix).
Pointing at drawings. A large test sheet (0.42 m × 0.3 m, 80.06° horizontal × 61.93° vertical size) presented 16 color drawings of animals (each 3.5 cm × 3,5 cm = 8.01° × 8.01°), equally spaced in 4 × 4 rows in both hemispaces, at a distance of 0.25 m from the patient. The patient’s task was to point to 10 (5 in each hemispace) drawings as subsequently and randomly announced by the experimenter. The number of animals correctly pointed to within 20 s (recorded by a stopwatch) was counted as hits. Drawings not found within the maximum exploration duration of 20 s were counted as omissions, with the maximum exploration duration of 20 s. Hits (maximum 5 in each hemifield) and respective exploration duration were documented separately for the left and right hemispaces. For the tray test and animal drawings test, 2 parallel versions were developed (A and B), which were alternated across the 3 time points of measurement to reduce memory effects (see the appendix for details).
Horizontal stick bisection. Patients were instructed to assess the midpoint of a wooden bar (length 1 m; height/breadth 1 cm), horizontally and centrally placed at a distance of 0.25 m from the patient. A total of 6 trials (3 visual, 3 tactile) were performed. In the visual condition, patients scanned the bar visually. In the tactile condition, patients were blindfolded, and the right index finger was used to explore the bar haptically. The order of testing was fixed: first visual, then tactile bisection. In both conditions, the right index finger was used to point to the subjective midpoint. The mean deviation from the objective midpoint across all trials was transformed to a score ranging from 0 to 3, reflecting the magnitude of the deviation (see the appendix).
Gaze deviation. The researcher stood at the end of the patient’s bed and scored gaze orientation in a “confrontation” procedure 31 in the following way while watching the patient in bed: Does the patient make normal, spontaneous eye/head movements to either side? (score 0). Does he or she show delayed leftward (contralesional) movement of eyes/head? (score 1). Does he or she show leftward movement of eyes/head only after a verbal command? (score 2). Does he or she show no leftward movement of eyes/head despite a verbal command? (score 3).
The patient’s performance in the tests of the FNI was scored (0-3) by 2 independent raters to calculate a composite score of neglect severity ranging between 0 and 15. Both raters were not involved in the assessments nor in any treatment. The scoring procedure (see the appendix) was defined before the rating was implemented. The aim of this procedure was to rate the left-to-right-gradient of the patient’s spatial attention as a global measure of neglect severity. The scorings of the 2 examiners were identical (Spearman’s ρ; all r = 1.00) for each neglect test, thus showing perfect objectivity and reliability of the scoring procedure.
Unawareness and behavioral neglect index (UBNI): We used a rating similar to the Catherine Bergego Scale (CBS) to assess behavioral neglect in daily life.30,36 In addition and differing from the CBS, the UBNI includes 6 items assessing the degree of unawareness in daily life. The UBNI was rated by the nurses on the ward (who were blinded to the treatment type) on a 4-point rating scale (behavior occurrence: 0, never; 1, rarely; 2, frequent; 3, daily). From the 10 items, 6 items assessed unawareness and 4 behavioral neglect in ADLs (details in the appendix). The mean rating over all 10 items was calculated and used for statistical analyses.
Help Index, Barthel Index, and Rehabilitation Status
The help which the patient needed to accomplish 8 functional ADLs was rated by the nurses on a 5-point scale: 1, no help needed; 2, verbal instruction sufficient for patient to solve the task; 3, little practical help necessary by staff to solve the task; 4, medium help necessary by nurse; 5, patient is completely relying on external help by the nurse to solve the task; see the appendix). The rating nurses were blinded to the treatment type the patients received. The BI includes 10 variables concerning ADL and mobility. 32 Following guidelines of the Federal Rehabilitation Council, patients are allocated to rehabilitation phases (B: early; C: continuing rehabilitation; D, posthospital curative treatment) based on the BI (appendix, Table 1). The BI and rehabilitation phases were scored by treatment-blinded physicians.
Statistical Analyses
The data were analyzed separately for all dependent variables with ANOVAs with the factors time (baseline, posttreatment, and follow-up) and group (SPT, VST). The α level was set at .05 (2-tailed) and corrected for pairwise comparisons according to Holm’s procedure, 37 where the fist comparison is computed with α, the second with α/2, and the third with α/3. The rehabilitation phase data were transferred to a score (B = 1, C = 2, D = 3) and analyzed nonparametrically.
Results
Group Comparison at Baseline
As t tests for independent measurements revealed no significant differences between the groups at the baseline assessment (smallest P = .28; Help Scale), the 2 groups were assumed as comparable in all clinical/demographic variables assessed (Table 1). Moreover, both groups did not differ significantly in horizontal line bisection (t = −1.088; P = .288) and digit cancellation at baseline (left-sided omissions: t = 1.492, P = .15; right-sided omissions: t = 1.704, P = .103; all 2 tailed).
Functional Neglect Index
The analysis revealed a significant effect of time point [F(2, 44) = 36.67, P < .001] and a significant Time Point × Group interaction [F(2, 44) = 5.84, P = .006]. There was no significant effect of group [F(1, 22) = 0.86, P = .37]. Further pairwise comparisons (Figure 3) revealed significant differences between all time points in the SPT group (Baseline − Posttreatment: mean difference = 5.16, P < .001; Baseline − Follow-up: mean difference = 7.00, P < .001; Posttreatment − Follow-up: mean difference = 1.83, P = .021). For the VST group, there were significant differences between baseline and posttreatment (mean difference = 2.83, P = .041) and baseline and follow-up (mean difference = 2.83, P = .010). There was no significant difference between posttreatment and follow-up (mean difference = 0.00, P = 1.00).
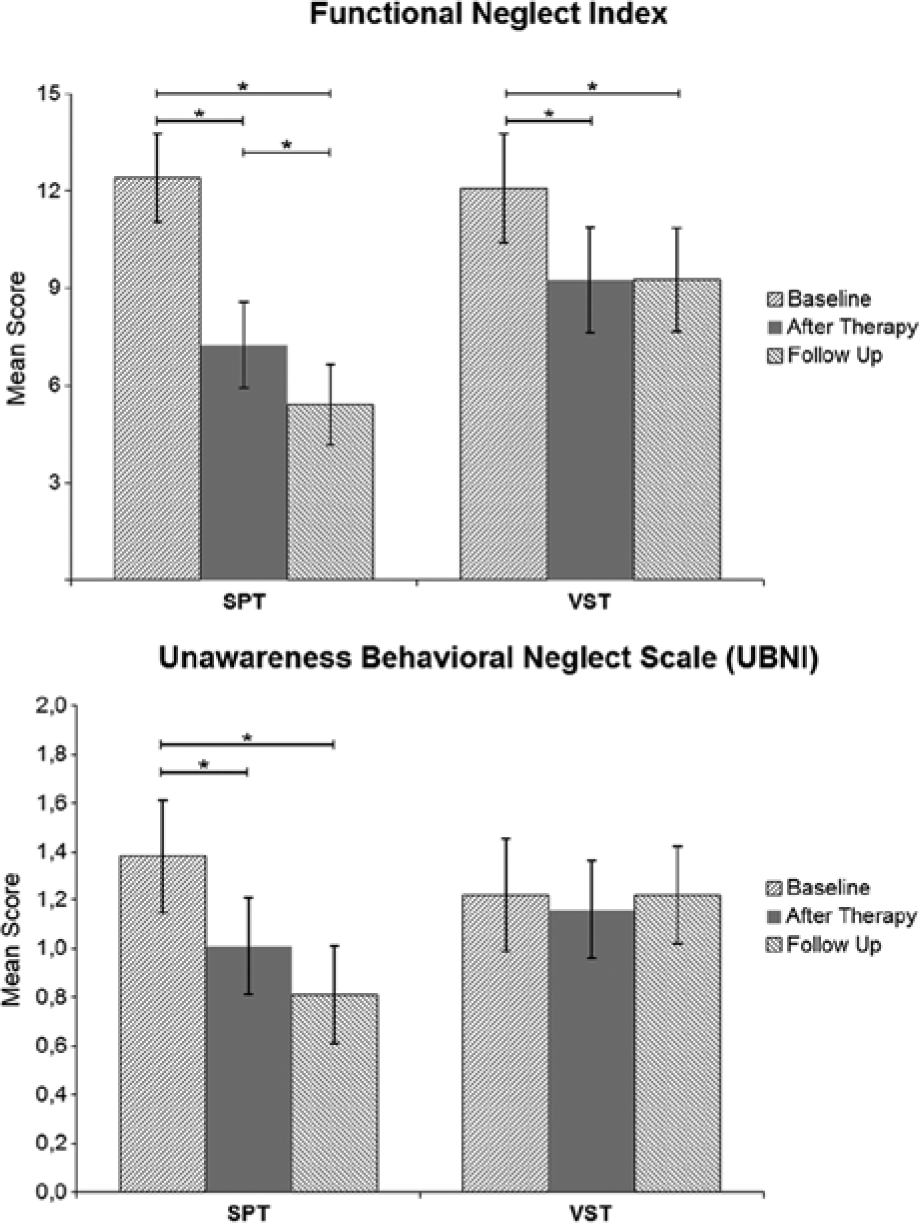
Results of the 2 treatments (SPT, VST) for the Functional Neglect Index and the Unawareness Behavioral Neglect Index. Asterisks indicate significant differences with P < .05, 2-tailed.
Unawareness and Behavioral Neglect Index
For the UBNI, the effect of time [F(2, 44) = 6.87, P = .003] and the Time × Group interaction [F(2, 44) = 6.39, P = .004] were highly significant. There was no significant effect of group [F(1, 22) = 0.25, P = .63]. Pairwise comparisons in the SPT group revealed significant differences between the baseline and posttreatment measurement (mean difference = 0.37, P = .001) and between baseline and follow-up (mean difference = 0.57, P < .002) but no difference between posttreatment and follow-up (mean difference = 0.20, P = .174). For the VST group, all comparisons failed to reach significance: largest mean difference (Baseline − Posttreatment) = 0.06; smallest P = 1.00.
Barthel Index, Rehabilitation Phase, Help Scale
For the BI, there was a significant effect of time [F(2, 44) = 33.66, P < .001] in the ANOVA (Figure 4). Pairwise comparisons were significant for all differences of time (Baseline − Posttreatment: mean difference = −13.95, P < .001; Baseline − Follow-up: mean difference = −21.87, P < .001; Posttreatment − Follow-up: mean difference = −7.91, P = .002). The effect of group [F(1, 22) = 0.46, P = .83] and the Time Point × Group interaction [F(2, 44) = 1.89, P = .16] were not significant.
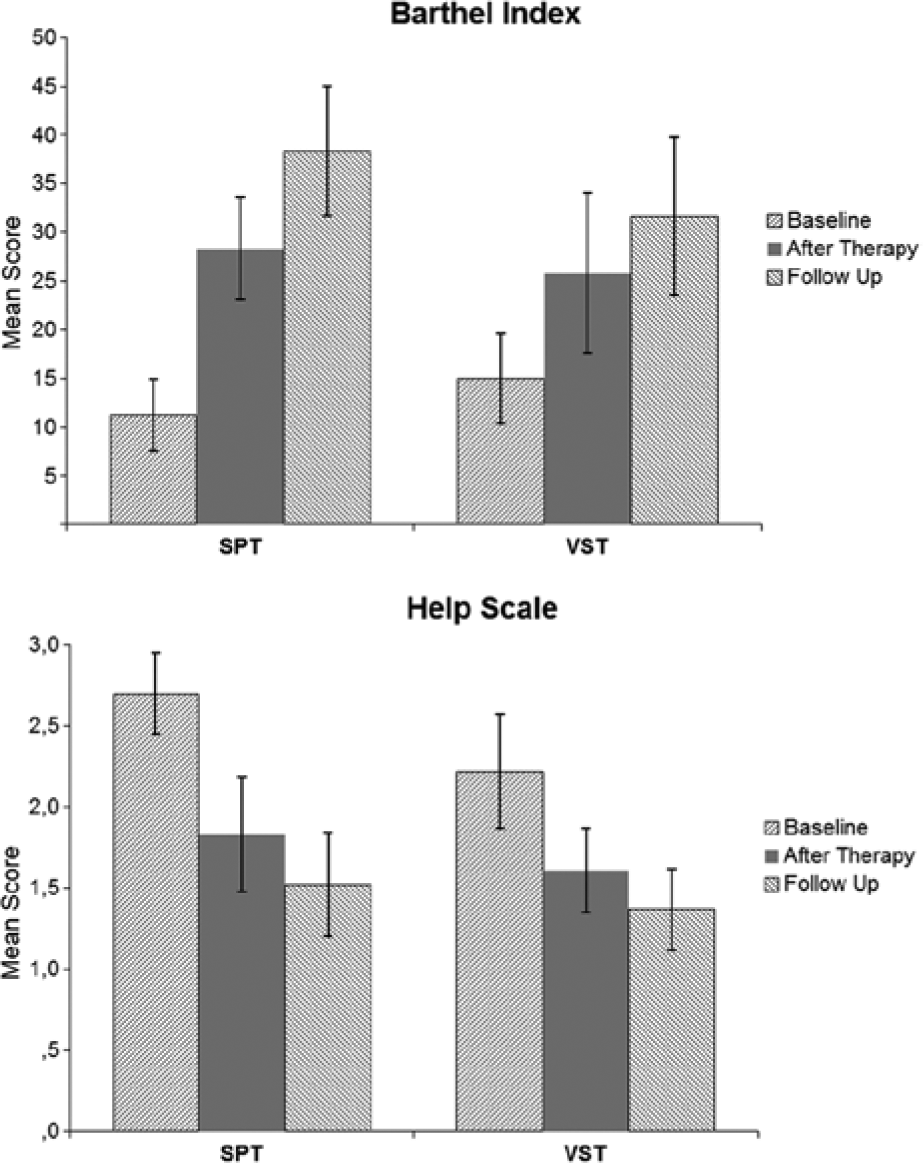
Results of the 2 treatments (SPT, VST) in the Barthel Index and the Help Scale.
Table 1 shows the rehabilitation phases of all patients over the 3 time points of measurements descriptively. Six SPT patients improved in their rehabilitation phase (3 during, 3 after SPT), whereas 4 VST patient improved (3 during, 1 after VST). Friedman tests revealed significant effects of time [χ2(2) = 9.58, P = .008] for the SPT group as well as for the VST group [χ2(2) = 6.50, P = .039]. For the SPT group, pairwise comparisons (Wilcoxon tests) revealed significant differences between baseline and follow-up (Z = 2.33, P = .020) and posttreatment and follow-up (Z = 2.00, P = .046). The comparison baseline-posttreatment failed to reach statistical significance (Z = 1.73, P = .083). For the VST group, only the comparison between baseline and follow-up was significant (Z = 2.00, P = .046). The remaining comparisons were not significant: Baseline − Posttreatment, Z =1.73, P = .083; Posttreatment − Follow-up, Z = 1.00, P = .317. Pairwise comparisons (Mann-Whitney U tests) between both treatment groups were not significant for any time point (all P >.55).
For the Help Scale, the effect of time point [F(2, 44) = 30.05, P < .001] was significant. Pairwise comparisons revealed significant effects between all time points (Baseline − Posttreatment: mean difference = 0.74, P < .001; Baseline − Follow-up: mean difference = 1.01, P < .001; Posttreatment − Follow-up: mean difference = 0.27, P = .004). There was no significant effect of group [F(2, 44) = 0.81, P = .45] and no Time Point × Group interaction [F(2, 44) = 0.81, P = .45].
Discussion
SPT induced significantly greater improvements in the FNI and UBNI than VST. These improvements continued—albeit to a smaller extent—selectively in the SPT group in the posttreatment period, whereas no such effect was observed after VST. Functionally, the progress observed after SPT mainly reflects improvements in finding and grasping objects in the neglected near space, finding more pictures in near space, and improving the perception of the subjective midline (haptically and visually). It is important to note that the ipsilesional gaze deviation, which is so typical and disabling in acute neglect patients, normalized significantly after both treatments, paralleling recent findings after prism treatment in early neglect. 12 These findings in functional neglect tasks were paralleled in significantly greater improvements of the SPT group in the UBNI. Put differently, unawareness and behavioral neglect—as rated by caregivers who were blinded to the treatments and who observed the patients many hours per day on the ward—improved to a significantly greater extent after SPT than VST. After both treatments, the BI and Help Index improved but without a statistical differential treatment effect (as shown by the nonsignificant interaction). However, the percentage of improvements in the BI over the 3 assessments was hugely different: SPT patients improved by 154% during treatment and 35.7 % in the follow-up period. In contrast, VST patients improved by 73% during treatment and by 23 % thereafter. Albeit nonsignificant, this shows that SPT induced more widespread improvements also in the BI, which might reach significance with a larger sample size. Because a patient’s progression in the rehabilitation phase depends on increases in the BI, the greater percentage of patients improving in their rehab phase during or after SPT versus VST (50% vs 33.3%) is encouraging because it shows that SPT accelerates functional recovery from neglect more than conventional VST. It is important to note that the improvements achieved in both groups proved to be stable at follow-up.
SPT as neglect therapy probably relies on the reactivation of a cortico-subcortical network related to optokinetic stimulation with smooth pursuit eye movements. 22 This modulates and finally reduces neglect in multiple ways: first, it provides sensory information necessary to compute egocentric spatial information required for accurate behavior in space. 9 Many of the concerned brain regions are involved in gaze and attention shifts (occipitotemporal, parietal, insular, occipital cortex, basal ganglia, cerebellum, and brain stem; see details in Kerkhoff et al 22 ), which were explicit features of our SPT therapy. Additionally, SPT may activate the vestibulo-ocular system via optokinetic nystagmus. By this mechanism, SPT may correct the pathological, ipsilesional bias in functional visual neglect tasks, enabling the detection of more targets in the tray or animal picture task, and induce a more symmetrical gaze orientation. Second, SPT facilitates multimodal attention shifts to the neglected side. 38 Interestingly, the frontoparietal attention network, which is believed to be involved in neglect, contains a modality-independent priority map, 39 which may be responsible for this multimodal efficacy of SPT. Third, the dynamic features inherent in moving displays that elicit smooth pursuit eye movements 40 may increase perceptual saliency of contralesional targets, and the multiplicity of brain regions involved in visual motion processing 41 may further enhance this superior effect as opposed to the static patterns viewed during VST. It is important to note that the superior effect of SPT was not limited to test scores but was also found for behavioral neglect as rated in relevant ADLs. Fourth, and the most interesting, this is the first study showing that unawareness can be significantly reduced by systematic neglect therapy. This may be also the explanation for the continued improvements after the end of SPT as opposed to VST. Our present findings support empirically the hypothesis put forward by Bottini et al 20 according to which some neglect therapies might also effectively reduce unawareness. Finally, we found that both neglect therapies are feasible as “bedside” treatments early after stroke, albeit with a hugely differential efficacy. This holds true for some other neglect treatments as well (ie, visuomotor feedback training)33,34 but may be more variable for others (ie, prism adaptation: negative 35 and positive 12 results). Early neglect therapy may speed up functional recovery and benefit from processes of brain reorganization occurring during this early phase after stroke.
Our study has limitations: First, it might be argued that improvements over the 3 time points of measurement reflect spontaneous recovery because our study lacks a second baseline. The significantly greater improvements in the FNI and UBNI observed after SPT versus VST argue against this. Moreover, more patients in the SPT than in the VST group showed further improvements in the follow-up period [3 vs 1 patient(s)], although this missed significance. Second, although some patients in the SPT group made quicker progress in their BI and rehabilitation status, both interaction effects were nonsignificant. If this trend can be replicated in larger samples, SPT may accelerate neglect therapy, leading to earlier discharge of patients, thus saving hospital costs.
In conclusion, bedside SPT reduces functional neglect and unawareness significantly more than VST. This corroborates the findings of our recent RCT in a different sample 22 and shows that SPT effectively reduces multimodal neglect and unawareness after stroke.
Footnotes
Appendix
Scoring of FNI. a
| Test | Performance | Score |
|---|---|---|
| Tray test, hits | Omissions in the left segment = Omissions middle = Omissions right/No omissions | 0 |
| Only omissions on the left segment, no omissions in the middle or right segment | 1 | |
| Omissions left > Omissions right and no omissions on the right segment | 2 | |
| Omissions in each segment and a clear left to right gradient (left ≥ middle > right omissions) | 3 | |
| Tray test, search time (ST) | ST left-sided items = ST middle = ST right (±10% tolerance range) | 0 |
| ST left > ST middle > ST right | 1 | |
| ST left > ST middle = ST right (±10% tolerance range); | 2 | |
| ST > 90 s | 3 | |
| Animal picture search, hits | Omissions left = Omissions right/No omissions | 0 |
| Omissions left − Omissions right = 1 | 1 | |
| Omissions left − Omissions right > 1 | 2 | |
| Omission of all left-sided targets | 3 | |
| Animal picture search, search time (ST) | ST left = Right (±10%) | 0 |
| ST left > right (up to 50%) | 1 | |
| ST left > right (more than 50%) | 2 | |
| ST left > 100 s | 3 | |
| Haptic horizontal rod bisection | Right-sided deviation < 5 cm | 0 |
| Right-sided deviation 5-10 cm | 1 | |
| Right-sided deviation 10-30 cm | 2 | |
| Right-sided deviation >30 cm | 3 | |
| Visual horizontal rod bisection | Right-sided deviation <5 cm | 0 |
| Right-sided deviation 5-10 cm | 1 | |
| Right-sided deviation 10-30 cm | 2 | |
| Right-sided deviation >30 cm | 3 | |
| Ipsilesional head and gaze deviation (see De Renzi et al 31 ) | Normal head and gaze deviation | 0 |
| Orientation predominantly normal but preferably deviated toward the right hemispace | 1 | |
| Orientation predominantly to the right hemispace | 2 | |
| Head and gaze completely oriented to the right hemispace | 3 |

Maximum score = 21.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
