Abstract
Background. Disturbed arm position sense (APS) is a frequent and debilitating condition in patients with hemiparesis after stroke. Patients with neglect, in particular, show a significantly impaired contralesional APS. Currently, there is no treatment available for this disorder. Galvanic vestibular stimulation (GVS) may ameliorate neglect and extinction by activating the thalamocortical network. Objective. The present study aimed to investigate the immediate effects and aftereffects (AEs; 20 minutes) of subsensory, bipolar GVS (M = 0.6 mA current intensity) on APS in stroke patients with versus without spatial neglect and matched healthy controls. Methods. A novel optoelectronic arm position device was developed, enabling the precise measurement of the horizontal APS of both arms. In all, 10 healthy controls, 7 patients with left-sided hemiparesis and left-spatial neglect, and 15 patients with left hemiparesis but without neglect were tested. Horizontal APS was measured separately for both forearms under 4 experimental conditions (baseline without GVS, left-cathodal/right-anodal GVS, right-cathodal/left-anodal GVS, sham GVS). The immediate effects during GVS and the AEs 20 minutes after termination of GVS were examined. Results. Patients with neglect showed an impaired contralateral APS in contrast to patients without neglect and healthy controls. Left-cathodal/right-anodal GVS improved left APS significantly, which further improved into the normal range 20 minutes poststimulation. GVS had no effect in patients without neglect but right-cathodal/left-anodal GVS worsened left APS in healthy participants significantly. Conclusions. GVS can significantly improve the impaired APS in neglect. Multisession GVS can be tested to induce enduring therapeutic effects.
Introduction
Patients with right-hemisphere stroke often show left-sided spatial neglect. 1 Neglect occurs mostly after right, 1 middle-cerebral-artery infarction,1,2 and its symptoms may persist for years.3,4 Patients with neglect fail to detect or respond to sensory stimuli in their contralesional hemispace and tend to underuse their contralateral limbs. They suffer from greater functional disability than stroke patients without neglect and therefore take longer to recover despite comparable stroke pathology and motor impairments. 5 The associated unawareness aggravates this unfavorable outcome.6-8 By definition, elementary sensory or motor deficits are not causative of neglect but often co-occur with the disorder.
Right Brain Damage, Neglect, and Deficits of Arm Position Sense
In the past, the majority of studies have investigated visual neglect and nonsensory, body-related neglect phenomena (ie, body and motor neglect, proprioceptive deficits,9,10 impaired identification of body parts, and motor extinction) have received less attention (see also review in Punt and Riddoch 11 ). 12 Deficits in arm position sense (APS) are common after stroke (36%-54%)13,14 and were traditionally considered to be primary sensory disorders.6,12,14 Recent studies, however, have found a strong correlation between right-hemisphere stroke, spatial neglect, and impaired APS.9,10 Moreover, impairments in APS were found to occur more often after right than left brain damage. 15 This hemispheric asymmetry fits observations of a higher incidence of neglect following right- versus left-sided stroke.
Furthermore, the cortical representations of each side of the body are mainly located in the contralateral hemisphere with only a minor body representation in the corresponding ipsilateral hemisphere. It is interesting to note that the ipsilateral representation of the left side of the body is less elaborated in the left cerebral hemisphere, 16 rendering this side more susceptible to body-related deficits such as an impaired position sense after a lesion of the right hemisphere. This may explain why patients with neglect perform worse in APS tasks than patients without neglect.9,10 In this context, it is also worth noting that leftward optokinetic stimulation transiently improves contralateral APS by some 12% to 20%,9,10 which points to a neglect-related component in the spatial coding of body parts and shows how this impairment might be modulated.
Clinically, impairments in locating one’s own limbs without visual feedback are associated with imprecise motor functions, reduced spontaneous use of arms, awkward limb positions, reduced safety, and postural instability. 17 In neglect, deficient position sense impedes motor functioning and is related to left motor neglect. 6
One theory hypothesizes that neglect results from an impaired representation/transformation of spatial coordinates into an egocentric reference frame necessary for correct body orientation in space.18-20 Right-cerebral lesions perturb the updating of such egocentric representations for body and space. 9 Recent advances in neglect therapy are based on sensory stimulation techniques that partly rely on these theories 21 and show that optokinetic stimulation,9,22 prism adaptation,23,24 neck muscle vibration, 25 and caloric vestibular stimulation26,27 all reduce visual neglect. 27 Unfortunately, motor 11 and body-related deficits in patients with neglect have been virtually “neglected” in therapy research in the past 2 decades. These types of impairments are not negligible because, for example, 20% of patients with right-hemisphere stroke show motor neglect, 28 and 32% an impaired position sense (see results) without any treatment currently available. In light of the overall negative motor outcome of patients with neglect, 29 their impaired postural control, 29 and the well-known proprioceptive deficits of the left body side,9,10 effective treatments for these nonvisual disorders associated with neglect are urgently required to accelerate a more complete functional recovery beyond the achievements gained in visual neglect. How can this goal be achieved?
Transcutaneous electrical neural stimulation transiently improves neglect-related postural instability selectively in patients with left neglect. 30 Moreover, Dijkerman and colleagues 31 recently demonstrated lasting improvements in proprioception after prism exposure in a patient with left neglect. These studies show a significant modulation of nonvisual disorders in left neglect. Galvanic vestibular stimulation (GVS) is another promising, noninvasive technique activating vestibular, motor, and adjacent cortices as well as subcortical areas involved in postural, spatial, and cognitive functions (review by Lopez and Blanke 32 ). Recently, we showed that one 20-minute session of GVS temporarily reduces the ipsilesional bias in line bisection in patients with visuospatial neglect, 33 and 2 sessions improve tactile extinction permanently by about 40%. 34 Accordingly, caloric vestibular stimulation reduces left motor neglect.26,35 GVS, unlike caloric vestibular stimulation, is easier to use, lacks adverse side effects, and is more appropriate for repetitive treatment without habituation effects.36,37 In practice, weak direct currents are delivered via 2 electrodes of different polarity (anode and cathode) placed on both mastoids behind the ears. 38 GVS activates the whole thalamocortical system up to the parieto-insular-vestibular-cortex,38,39 which is partly damaged in neglect. 40 Bihemispheric activations are obtained by applying left-cathodal/right-anodal GVS (L-GVS), whereas unilateral, right-hemispheric activations are induced by right-cathodal/left-anodal GVS (R-GVS). 41 GVS might thus modulate deficits in APS in a similar way as transcutaneous electrical neural stimulation or prism goggles in the above-mentioned studies. The present study addressed the following issues:
Does GVS modulate APS in both forearms in stroke patients with versus without neglect differentially, and does it lead to an immediate effect (online effect) as well as an aftereffect (AE; 20 minutes post-GVS)? The inclusion of the right brain-damaged patients without left neglect (RBD−N) is clinically important because it may signal the specificity of GVS to a certain subgroup of stroke patients (ie, those with left neglect). Moreover, differential effects might tell the clinician which patient might potentially improve (under GVS) and which not.
Are there polarity-specific effects of GVS on APS similar to those found for tactile extinction 34 or line bisection? 33
Methods
Participants
In all, 7 patients with right-hemisphere stroke and left-sided visuospatial neglect (6 men, RBD+N), 15 patients with right-brain damage but without neglect (14 men, RBD−N), and 10 age-matched, healthy controls (7 men, C) were included (Table 1). Inclusion criteria for all patients were a single, supratentorial, right-hemisphere stroke; right-handedness; and awareness of (left) hemiparesis. Exclusion criteria were bilateral or left-sided lesions, aphasia, and psychiatric disorders in addition to specific GVS exclusion criteria (heart pacemaker, pregnancy, metallic brain implants, epilepsy, or sensitive skin behind the ears). 36 The 3 subject groups did not differ with respect to age (χ2 [df = 2] = 2.57; P = .276) and sex (χ2 [df = 2] = 2.49; P = .288). The 2 patient samples were neither significantly different in the severity of the somatosensory (U = 51.5; z = −0.12; P = .91) and motor impairments (hemiparesis; U = 29.5; z = −1.74; P = .08) nor in time since lesion (U = 28; z = −1.73; P = .08). Proprioception was examined by requiring patients to imitate arm postures during neurological assessment and was found to be normal in all patients. All participants gave their informed written consent before examination. The study was approved by the local ethics committee (Ärztekammer, München, Germany). All participants were right handed, 41 had no history of psychiatric disorders or dementia, showed good awareness of their hemiparesis, and had a corrected visual acuity of at least 0.5 (50%, distance 0.4 m; Table 1).
Clinical and Demographic Data of 7 Patients With Left Neglect Caused by a Single Vascular Lesion of the Right-Cerebral Hemisphere (RBD+N1 to RBD+N7) and 15 Patients With a Single Right-Hemisphere Vascular Lesion Without Neglect (RBD−N1 to RBD−N15). a
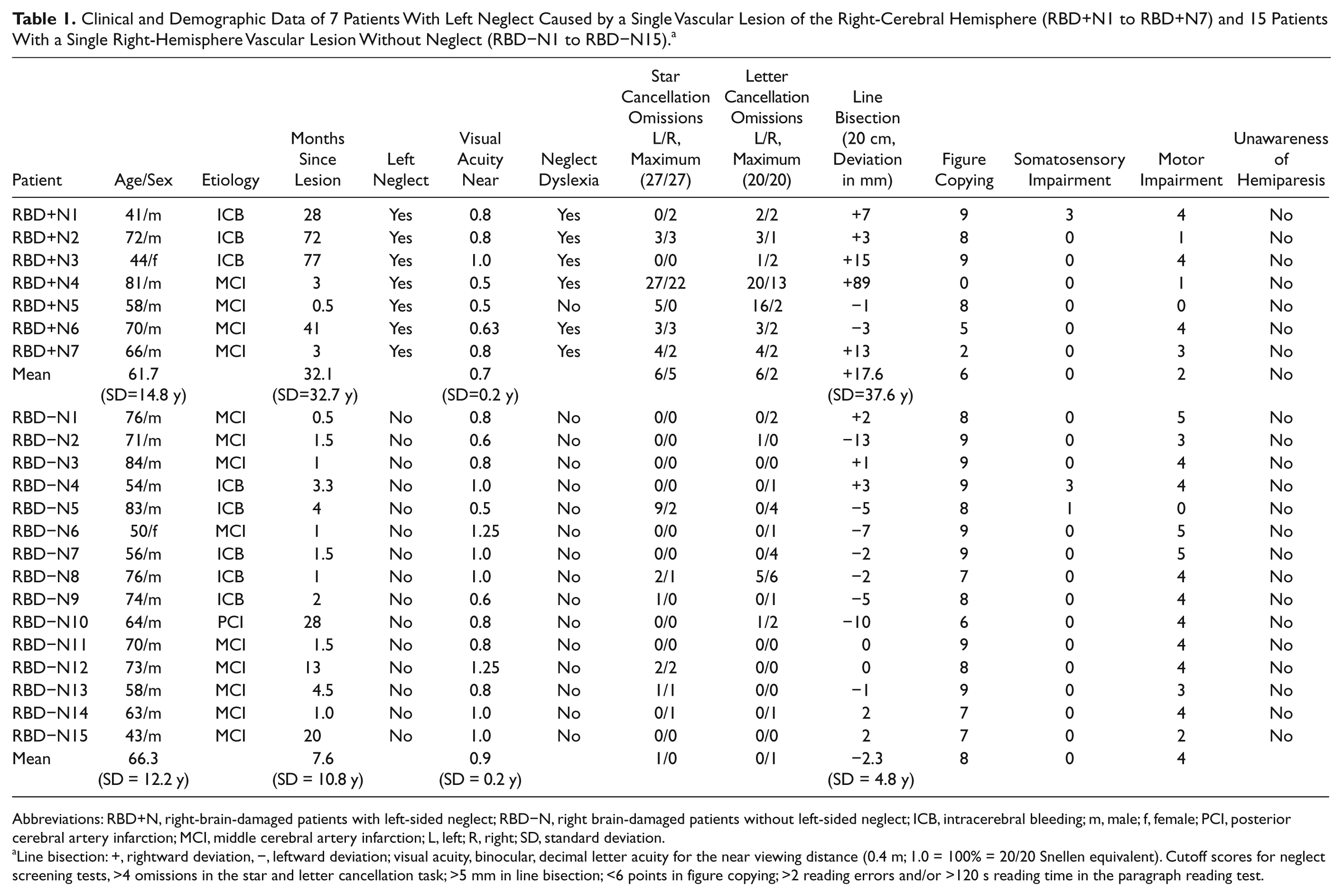
Abbreviations: RBD+N, right-brain-damaged patients with left-sided neglect; RBD−N, right brain-damaged patients without left-sided neglect; ICB, intracerebral bleeding; m, male; f, female; PCI, posterior cerebral artery infarction; MCI, middle cerebral artery infarction; L, left; R, right; SD, standard deviation.
Line bisection: +, rightward deviation, −, leftward deviation; visual acuity, binocular, decimal letter acuity for the near viewing distance (0.4 m; 1.0 = 100% = 20/20 Snellen equivalent). Cutoff scores for neglect screening tests, >4 omissions in the star and letter cancellation task; >5 mm in line bisection; <6 points in figure copying; >2 reading errors and/or >120 s reading time in the paragraph reading test.
Assessment of Somatosensory and Motor Impairment, Unawareness, and Neglect
All patients were assessed with a standard somatosensory function screening of the contralateral (left) upper limb, yielding scores on a 4-point scale ranging from 0 (no defect) to 3 (severe impairment). While the eyes of the patients were closed, the examiner administered with his fingertip 10 single and 10 double, symmetrical, and simultaneous tactile stimuli, as shortly and lightly as possible, on the dorsal surface of the patient’s hands. Tactile extinction was only recorded when more than 90% of unilateral tactile stimuli were correctly reported. Motor impairment of the forearm was measured using the Medical Research Council Scale.42,43 This scale grades muscle strength on a scale from 5 (no defect) to 0 (severe impairment). Unawareness of reduced muscle strength in the contralesional forearm was tested with Cutting’s anosognosia questionnaire, 8 with a rating scale ranging from 0 (spontaneous reporting) to 3 (complete denial of the deficit). Visuospatial hemineglect was assessed by 5 screening tests: star cancellation, letter cancellation, line bisection, and figure copying (subtests of the German BIT44,45) as well as a paragraph reading test (180 words) 22 (for cutoff scores see Table 1). All screening tests were performed with the center of the display positioned perpendicular to the midsagittal plane of the patient’s trunk. The results in all tests were used to calculate a combined neglect severity index (from 0 = normal performance in all 5 tests to 5 = abnormal results in all 5 tests). Patients were classified as showing neglect if they had a score ≥2.
Assessment of APS
The APS in the horizontal plane was measured with an optoelectronic apparatus (arm position device [APD]; see Figure 1). The APD consisted of a 90° circuit table with a manually adjustable LED lamp for optical indication of the required arm position. A movable armrest on a 90° circuit below the table was installed under the circuit of the lamp to locate the participant’s current horizontal arm position with a resolution of 1°. Furthermore, a control panel stored the difference (in degrees) between the target position and the actual arm position during the experiment. The participants were sitting in front of the apparatus with the right or left arm on the armrest. The participant’s forearm was moved manually by the examiner with an average velocity of 4.3 deg/s (range, 3.6 deg/s-5.8 deg/s) toward 2 different angle positions (30° and 60°) from 4 different starting positions: 90° (straight ahead), 60°, 30°, 0° (Figure 1). The experimenter did not see the actual position of the arm support or the patient’s arm because he directed his gaze to the periphery of the APD to avoid experimenter bias. There were 12 trials (3 per position) for each arm and 2 practice trials that were not scored. The sequence of trials varied randomly across participants with the right forearm being tested first in every participant. In each trial, the participants were required to indicate verbally as precisely as possible when their forefinger was directly positioned under the LED. During the experimental session, the arm and shoulder of the individual were covered by a black cape, and the testing room was darkened (1 Lux) to reduce any visual cues. No time constraints were imposed, and no feedback was given. Measurements were performed following a schedule (controlled by a stopwatch), lasting 20 minutes per session. The criterion measure for APS was the unsigned, average positional error, expressed as the absolute difference between the actual arm position and the target arm position as indicated by the LED (in degrees, averaged over 12 trials/condition). The unsigned cutoff scores for healthy controls were 4.6° for the right and 4.2° for the left forearm (based on n = 24 right-handers; age range, 51-71 years).
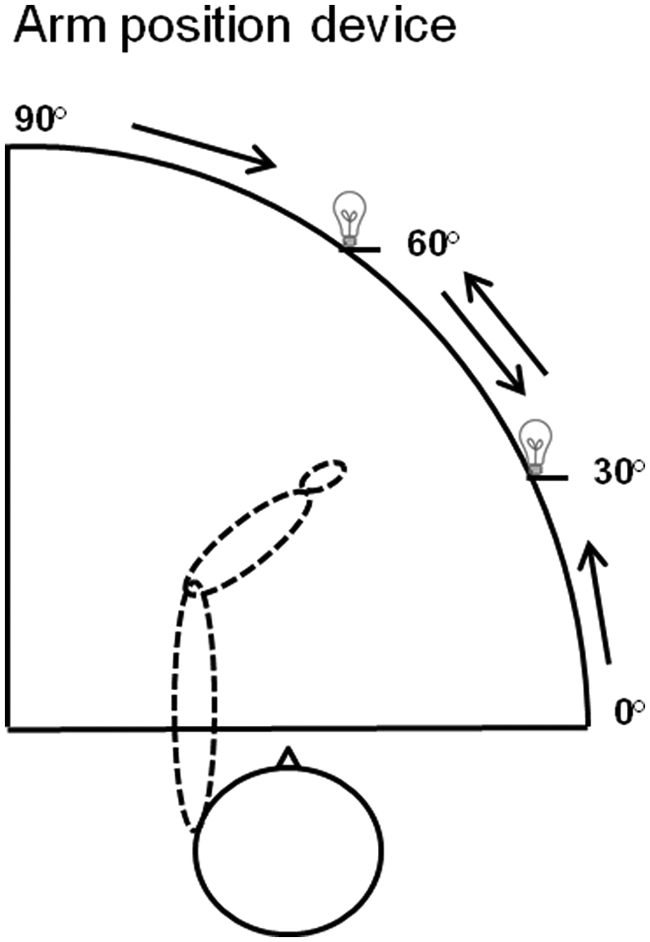
Schematic view of the apparatus used for measuring arm position sense in the study and of the patient’s position when the left forearm was tested—2 different angle positions (30° and 60°) from 4 different starting positions (90°, ie, straight ahead; 60°; 30°; and 0°) are shown.
Galvanic Vestibular Stimulation
Participants were stimulated with a 9-V, battery-driven constant direct current stimulator (neuroConn GmbH, DC-Stimulator, 98693 Ilmenau, Germany). Bilateral bipolar GVS was performed by placing 2 carbon electrodes (anode and cathode) over both mastoids. The electrodes (50 × 35 mm2) were inserted in saline-soaked sponges. The polarity of the applied electric currents was changed for each of the 3 experimental conditions. For L-GVS stimulation, the cathode was placed on the left mastoid and the anode on the right mastoid. This electrode setup was reversed for the R-GVS condition. In the sham condition, the 2 electrodes were positioned as in the L-GVS condition, except that no electric current was applied. For subliminal GVS, the individual sensory threshold for stimulation was determined in session 1 by progressively increasing the current amplitude in steps of 0.1 mA until the participants reported a tingling sensation underneath the electrodes. The current was then decreased until the cutaneous sensation stopped. This procedure was repeated twice, so that a current intensity of 0.1 mA below the mean value at which the tingling sensation occurred was selected for subliminal stimulation. The mean current amplitude used for subliminal GVS in the patients was 0.6 mA. The duration of subsensory GVS was limited to a maximum of 20 minutes in order to conform to established safety guidelines. 37
Design and Procedure
All individuals participated in 4 different experimental sessions (Figure 2). In session 1, all screening assessments (see Assessment of Somatosensory and Motor Impairment, Unawareness, and Neglect) and the first baseline test in horizontal APS were carried out. In sessions 2 to 4, the participants performed the APS task again while receiving either L-GVS, R-GVS, or sham-GVS, respectively, in a pseudorandomized sequence to control for order effects. Participants were blinded to the type of stimulation received. In each stimulation session, the electrodes were removed after 20 minutes of stimulation (alternatively sham stimulation) and the participants paused for 20 minutes in darkness. After 20 minutes, the APS was measured again to evaluate potential AEs. A 2-day interval (minimum, 48 hours) was established between sessions to avoid carryover effects.
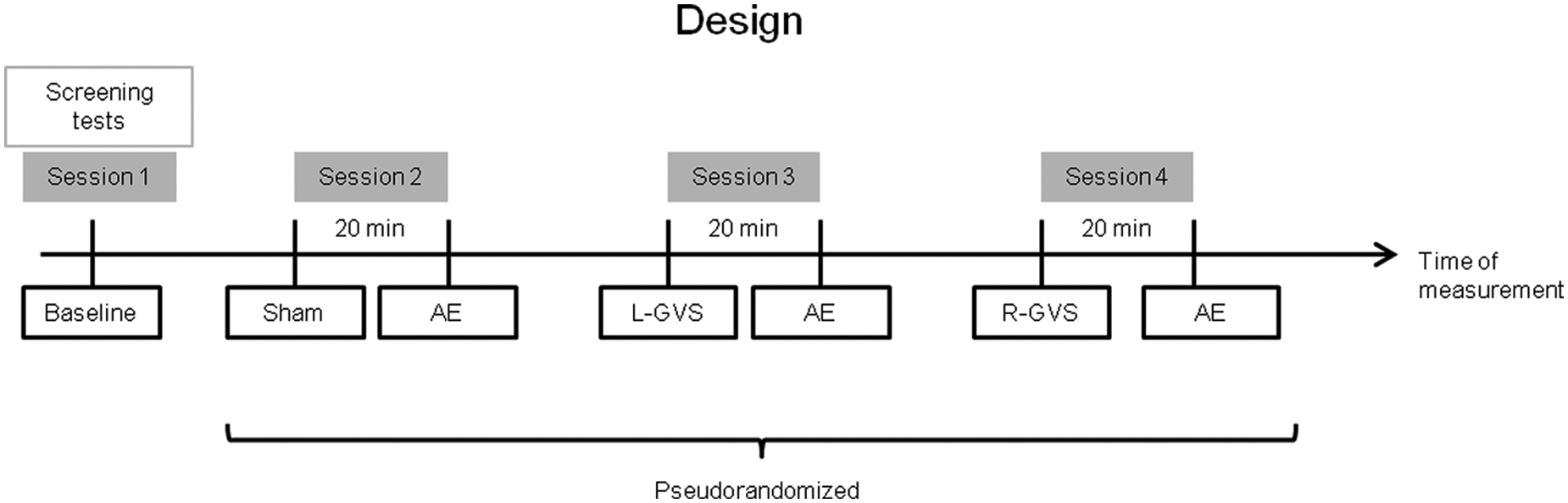
Design of the study with the 4 different experimental sessions.
Statistical Analyses
All analyses were carried out using SPSS, version 19. First, the APS scores of the 12 trials per condition were averaged. analyses of variance (ANOVAs) with the between factor “Group” (C, RBD+N, RBD−N) and the within factor “GVS condition” (baseline, sham, sham-AE, L-GVS, L-GVS-AE, R-GVS, and R-GVS-AE) were carried out separately for the right and left arms. Significant results were followed up with Bonferroni-adjusted t tests for multiple comparisons. 46 The α level was set at P = .05, 2-tailed for all analyses.
Results
Figures 3A and 3B display the online effects and aftereffects of GVS on APS separately for both arms and across all experimental conditions in the 3 subject groups.
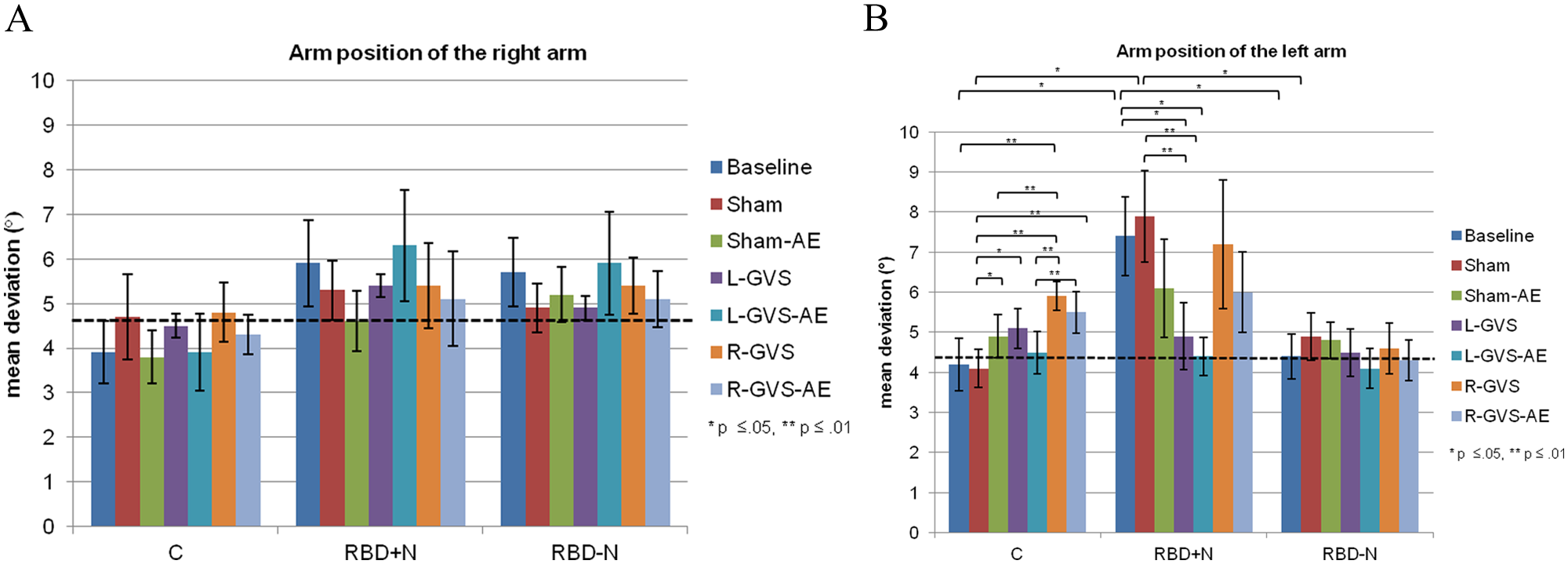
Mean unsigned errors (in degrees) and standard error of the mean (SEM) of the 3 subject groups in the different experimental conditions of right and left arm; Online effects and aftereffects of the right (A) and left (B) arm and the respective cutoff scores of healthy controls (dashed lines, n = 24; right arm = 4.6°, left arm = 4.2°).
Right Arm
The analysis of the performance of the right arm did not show a statistically significant main effect of GVS condition—F(6, 102) = 0.73, P = .555, η2 = .025)—or group—F(2,29) = 0.95, P = .398, η2 = .062—or a significant GVS condition × Group interaction—F(12, 102) = 0.652, P = .713, η2 = .043—see Figure 3A.
Left Arm
In contrast, the analyses of the APS scores of the left arm yielded a significant main effect of GVS condition—F(6, 119) = 3.13, P = .017, η2 = .097—and a significant interaction between GVS condition and Group—F(12, 119) = 2.55, P = .013, η2 = .15. No significant main effect was found for the factor group—F(2, 29) = 2.99, P = .066, η2 = .171. Subsequent t tests analyzing the APS differences between GVS conditions were computed for each group separately. For the control group, the analyses revealed a significant deterioration in APS in the R-GVS condition as compared with baseline—T(9) = −3.78, P = .004—sham—T(9) = −7.82, P = .000—sham-AE—T(9) = 3.24, P = .01—and L-GVS-AE—T(9) = −4.14, P = .003. Moreover, the deviations of the healthy controls were larger in the R-GVS-AE condition than in the sham—T(9) = −4.99, P = .001—and L-GVS-AE conditions—T(9) = −3.46, P = .007. The APS error was also more severe in the sham-AE—T(9) = −2.95, P = .016—and L-GVS conditions—T(9) = −2.26, P = .05—as compared with the sham condition. Furthermore, the RBD+N patients showed significantly smaller deviations in the L-GVS condition—T(6) = 2.45, P = .05—and a further enhancement in the L-GVS-AE condition—T(6) = 3.59, P = .012—as compared with baseline. Additionally, they also improved significantly in both L-GVS—T(6) = 7.39, P = .000—and L-GVS-AE—T(6) = 3.94, P = .008—relative to sham. In the RBD−N-group, however, no significant differences in APS across conditions were found (all P > .127). Subsequent t tests analyzing the GVS condition × Group interaction showed that deviations of the RBD+N patients at baseline and during sham-GVS were significantly larger than those of the control group—baseline, T(11) = −2.69, P = .021, d = −3.21; sham, T(8) = −3.07, P = .015, d = −3.79—and of the RBD−N-group—baseline, T(10) = 2.69, P = .023, d = 3.07; sham, T(9) = 2.32, P = .044, d = 2.99—see Figure 3B.
In summary, RBD+N patients were significantly impaired in their left APS. This deficit improved significantly during L-GVS (P = .05) and further into the normal range 20 minutes after cessation of L-GVS (P = .012). Controls showed a significant deterioration of their left arm’s APS during R-GVS (P = .000). All these effects were significant when compared with baseline and sham conditions, suggesting that these findings do not result from learning, test repetition, or other unspecific effects.
Figure 4 shows the individual results of all 7 patients with neglect of the left arm position as a function of lesion chronicity. All patients showed their best performance during L-GVS and L-GVS-AE (aftereffect), independently of chronicity. Patient 4 showed more variable results and an additional improvement in APS during R-GVS and R-GVS-AE, which was not found in the other 6 patients.
Individual unsigned errors (in degrees, averaged over 12 trials) of the 7 patients with neglect (RBD+N-group) in arm position sense across the different experimental conditions for the left arm and in relation to lesion chronicity (months). For patient codes and lesion chronicity, see Table 1.
There were no significant correlations between the clinical characteristics of patients with neglect and the modulation of APS by GVS (Pearson/Spearman: smallest P = .101/.258, largest rp/rs = 0.36/0.25). Notably, the exception was a significant positive correlation between deficits in left APS and visual neglect severity (Pearson: rp = 0.49, P = .022; Spearman: rs = 0.47, P = .027), which was not found for the right arm (Pearson: rp = 0.19, P = .407; Spearman: rs = 0.09, P = .685).
Discussion
The present study showed the following results: (1) APS of the left (contralesional) but not the right arm was significantly impaired in our mostly chronic RBD+N patients as compared with RBD−N patients and healthy controls; (2) L-GVS selectively improved the accuracy of APS in RBD+N patients during stimulation (significant online effect). Moreover, the left APS in these patients continued to ameliorate for at least 20 minutes after the end of stimulation (significant aftereffect). This improvement was independent of patient characteristics and lesion chronicity; and (3) R-GVS significantly worsened APS for the left arm in healthy individuals.
Contralesional Arm Position Deficit in Spatial Neglect
Consistent with studies in acute patients with neglect, 9 we found a clear contralateral but no ipsilateral deficit in APS in our more chronic patients with neglect. Hence proprioceptive deficits of the contralateral arm are a persistent problem in neglect and correlate significantly with the severity of the syndrome. Despite these minor differences (with respect to the APS deficit in the ipsilesional arm in previous studies9,10), our study confirms that neglect is one contributing factor 9 to this disturbance because the RBD−N group performed at the same level as our healthy controls. Clinically, these findings suggest a nonvisual, neglect-related deficit in patients with visual neglect that impairs the subjective positioning of the contralateral arm in space in the absence of vision. 6
Vestibular Modulation of APS
Only L-GVS significantly improved the accuracy of APS for the left arm in neglect: the beneficial effects on the APS were not only observed during online GVS but seemed to increase further significantly by reaching normal performance 20 minutes poststimulation. All 7 participants with hemineglect showed the above pattern (Figures 3 and 4) independently of their lesion chronicity (see Figure 4).
According to transformational theories of neglect, vestibular stimulation improves the deficient egocentric reference by providing additional sensory input, which in turn may help correct the position sense of the left arm.19,20 This facilitatory effect of GVS may rely on the activation of intact structures within this system that are specifically involved in the recalibration of body and limb position in space. 47 The greater efficiency of L-GVS versus R-GVS in modulating APS probably results from the asymmetry of the cortical vestibular system. 48 Here galvanic inhibition of the left vestibular nerve with excitation of the right vestibular nerve (right-cathodal/left-anodal GVS) results in right vestibular cortex activation, whereas galvanic inhibition of the right vestibular nerve with excitation of the left vestibular nerve (left-cathodal/right-anodal GVS) 49 activates vestibular cortices bilaterally, at least in healthy participants. 47 Thus, it is conceivable that L-GVS leads to a more widespread cerebral activation in both hemispheres, which could partially compensate for the effects of the typically large brain lesions 28 in patients with left neglect. The continuation of the initial improvement 20 minutes after L-GVS stimulation can be explained by long-term potentiation, a well-known phenomenon of neuroplasticity induced by direct-current stimulation. 37
It is important to note that neither R-GVS nor sham-GVS influenced APS, thereby ruling out placebo or unspecific repetition effects as confounding factors of the observed improvements. Moreover, the observed modulating effects are unlikely to result from mere attentional cueing toward the stimulating electrode because the participants could neither feel the stimulation nor discriminate between the different experimental conditions.
Our finding of the lack of any GVS effect in the patients without neglect concurs with findings showing that sensory stimulation maneuvers such as GVS or optokinetic stimulation typically do not modulate performance in these patients, probably because their egocentric reference system is unimpaired. 9
Unexpectedly, R-GVS deteriorated left APS in healthy participants. This is probably caused by R-GVS activating the right (intact) vestibular cortex, which may interfere with regular, intact vestibular activity because of overexcitation in these cortical areas. This, in turn, may induce a temporary disturbance of spatial reference frames, similar to but less pronounced than in neglect. 47 Comparable disruptive effects of vestibular stimulation on cognitive processes in healthy individuals were observed for spatial memory tasks 50 and tactile exploration, 51 suggesting that egocentric frames of reference—even though not lesioned—are susceptible to GVS. The lack of a disturbing effect of R-GVS in our RBD−N participants can be explained by lesion size effects, which generally reduce the influence of GVS (see Sparing et al 52 for related findings in parietal transcranial direct current stimulation).
Some limitations of our study have to be considered: the sample of patients with neglect was small, requiring replication in larger groups. Moreover, because safety standards for GVS limit exposure to 20 minutes, the number of tests and trials possible in 1 session are restricted. Finally, the deterioration of left APS in healthy participants requires further verification in a larger sample.
Summary
APS for the contralesional arm is impaired in patients with left neglect and improves transiently into the normal range after 20 minutes of GVS. Therefore, GVS could be a useful add-on therapy augmenting the effects of other treatments (ie, limb activation, 12 prism exposure, 31 and feedback training 17 ) by ameliorating proprioceptive and other body-related disorders in neglect, 53 as recently observed in a case study of repetitive GVS. 54
Footnotes
Acknowledgements
We are grateful to Dr Igor Schindler, University of Hull, United Kingdom, for critically reading the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Deutsche Forschungsgemeinschaft (DFG) grant to Georg Kerkhoff (IRTG 145, Adaptive minds).
