Abstract
Background. Action observation influences motor performance in healthy subjects and persons with motor impairments. Objective. To understand the effects of action observation on the spontaneous rate of finger movements in patients with Parkinson disease (PD). Methods. Participants, 20 with PD and 14 healthy controls, were randomly divided into 2 groups. Those in the VIDEO group watched video clips showing repetitive finger movements paced at 3 Hz, whereas those in the ACOUSTIC group listened to an acoustic cue paced at 3 Hz. All participants performed a finger sequence at their spontaneous pace at different intervals (before, at the end of, 45 minutes after, and 2 days after training); 8 participants with PD were recruited for a sham intervention, watching a 6-minute video representing a static hand. Finally, 10 patients participated in the same protocol used for the VIDEO group but were tested in the on and off medication states. Results. Both VIDEO and ACOUSTIC training increased the spontaneous rate in all participants. VIDEO intervention showed a greater effect over time, improving the spontaneous rate and reducing the intertapping interval to a larger extent than ACOUSTIC 45 minutes and 2 days after training. Action observation significantly influenced movement rate in on and off conditions, but 45 minutes after training, the effect was still present only in the on condition. No effect was observed after sham intervention. Conclusions. These findings suggest that the dopaminergic state contributes to the effects of action observation, and this training may be a promising approach in the rehabilitation of bradykinesia in PD.
Introduction
Bradykinesia is one of the cardinal manifestations of Parkinson disease (PD) and is characterized by reduced speed when initiating and executing a single movement and progressive reduction of its amplitude, up to complete cessation during repetitive, simple movements.1-4 Generally, patients with PD experience major problems in performing and maintaining repetitive/rhythmic voluntary movements such as finger tapping or steady gait with severe consequences on daily living.5-7 However, PD patients may exhibit greater impairment of speed rather than amplitude (or vice versa) that may differently respond to treatment. Indeed, amplitude, but not speed, of finger movements improves after pallidotomy,8,9 whereas only speed increases after dopaminergic medications. 10
In the past decade, physical therapy programs combined with various sensory cues have been used to improve movement speed and/or amplitude in PD patients.11-13 Cueing strategies, defined as applying temporal or spatial external stimuli associated with the initiation and ongoing facilitation of motor activity, are often used to bypass the defective function of basal ganglia, possibly by using alternative unaffected pathways.11,14,15 Among cueing techniques, external rhythmic auditory cues and visual cues have been applied in PD to improve both speed and amplitude.
Recently, it has been shown that action observation has beneficial effects in athletes 16 and in patients suffering from neurological diseases, 17 including PD.18,19 Furthermore, recent evidence suggests that action observation can improve the consolidation of early motor memories and that this effect is consistent when observed movements are congruent with practiced movements.20,21 In accordance, Bove and coworkers 20 demonstrated that in healthy individuals, the mere observation of rhythmical actions influences the spontaneous rate of finger movements with changes in motor performance that are maintained over time (ie, till 2 days after the session of action observation). After watching a video of repetitive finger movements paced at 1 or 3 Hz for 10 minutes, the participants shifted their spontaneous movement rate (SMR) from around 2 Hz to a rate close to that of the video. These findings raised the possibility that action observation can influence the motor performance in terms of temporal organization of movement. We hypothesized that if action observation can influence the motor performance in terms of temporal organization, then it could be of some clinical utility in treating specific aspects of bradykinesia, such as speed and rhythm.
The specific aims of the experiment reported here were the following: (1) to investigate whether the observation of rapid rhythmical finger movements can influence the SMR of the same movements in PD over time; (2) to compare the effect of action observation with that obtained with external rhythmic auditory cues, and (3) to elucidate whether the dopaminergic state of the patients influences the effect of action observation on motor performance.
Methods
Study Design
For this single-center, randomized controlled trial, 55 participants (PD patients = 40; normal controls [NCs] = 15) were initially screened for eligibility. Two PD patients and 1 NC were excluded because they declined informed consent.
PD patients were randomly assigned (Figure 1) to the following: (1) main experiment (comparing the effects of VIDEO and ACOUSTIC training), (2) control experiment 1 (sham intervention), and (3) control experiment 2 (comparing the effect of VIDEO training in the on and off medication states). Healthy participants were recruited as NCs only for the main experiment. The randomization sequence was created using computer-generated random number tables with 1:1:1:1 allocation of PD patients and 1:1 allocation of NCs. An independent researcher did the randomization and advised the therapists about group allocation. The referring neurologist and the researcher conducting the assessments were kept blind to group allocation. Informed consent was obtained prior to the experiments according to our institutions’ policy and to the Declaration of Helsinki. The study was approved by the local ethics committee of the University of Genoa.
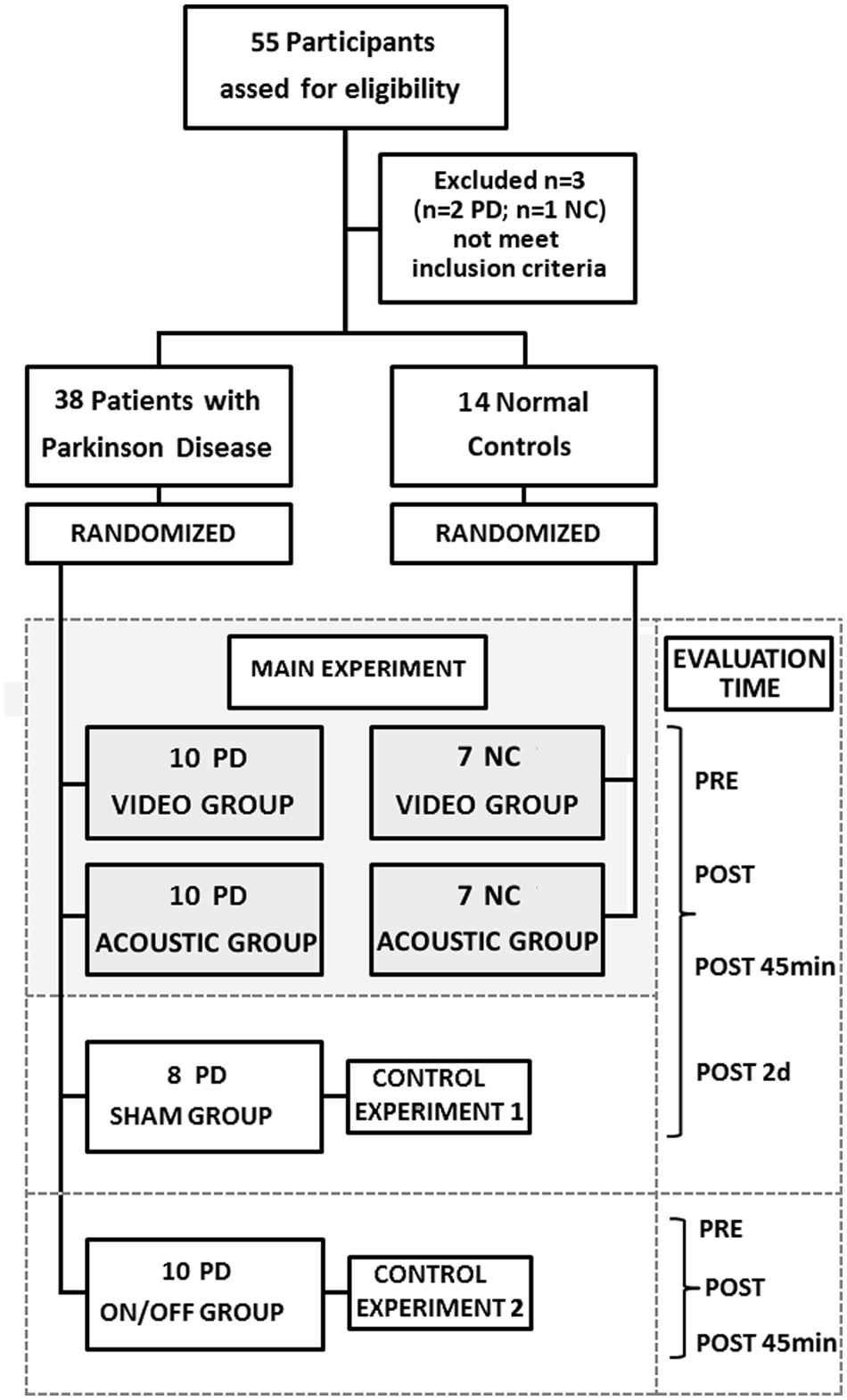
Experimental design: the flow chart reports the study design and the randomization process.
Participants
In all, 38 PD patients (17 male, 21 female; mean age ± standard deviation = 67.4 ± 7.4 years), according to the United Kingdom Parkinson Disease Society Brain Bank criteria were recruited from the outpatient Movement Disorders Clinic of the University of Genoa. Disease severity was determined by means of the Unified Parkinson Disease Rating Scale (UPDRS; Part III Motor). All patients were in Hoehn and Yahr stages 1 to 3 and on a stable medication regimen. The following exclusion criteria were applied: (1) past history of neurological conditions other than PD, (2) deep brain stimulation, (3) Mini-Mental State Examination score <24, (4) visual or acoustic limitations, and (5) severe orthopedic problems of the upper limb. All patients suffered from more severe symptoms on one side of their body than on the other at the time of symptom onset. This side is referred to as the more affected side. The designation of more affected side was determined from the history of the limb that was initially affected and confirmed by clinical evaluation. Only patients in whom the more affected side was the right one were included in the study.
A total of 14 aged-matched individuals (8 male, 6 female; mean age = 65.9 ± 7.6 years) with normal neurological examination and no history of neurological disorders were recruited as NCs from the hospital staff or relatives of the patients. They participated only in the main experiment. Demographic and clinical features of PD patients and NCs are summarized in Table 1. All participants were right handed as measured by the Edinburgh Handedness Inventory Questionnaire 22 and naïve to the purpose of the study.
Demographic Features of Patients With PD and NCs. a
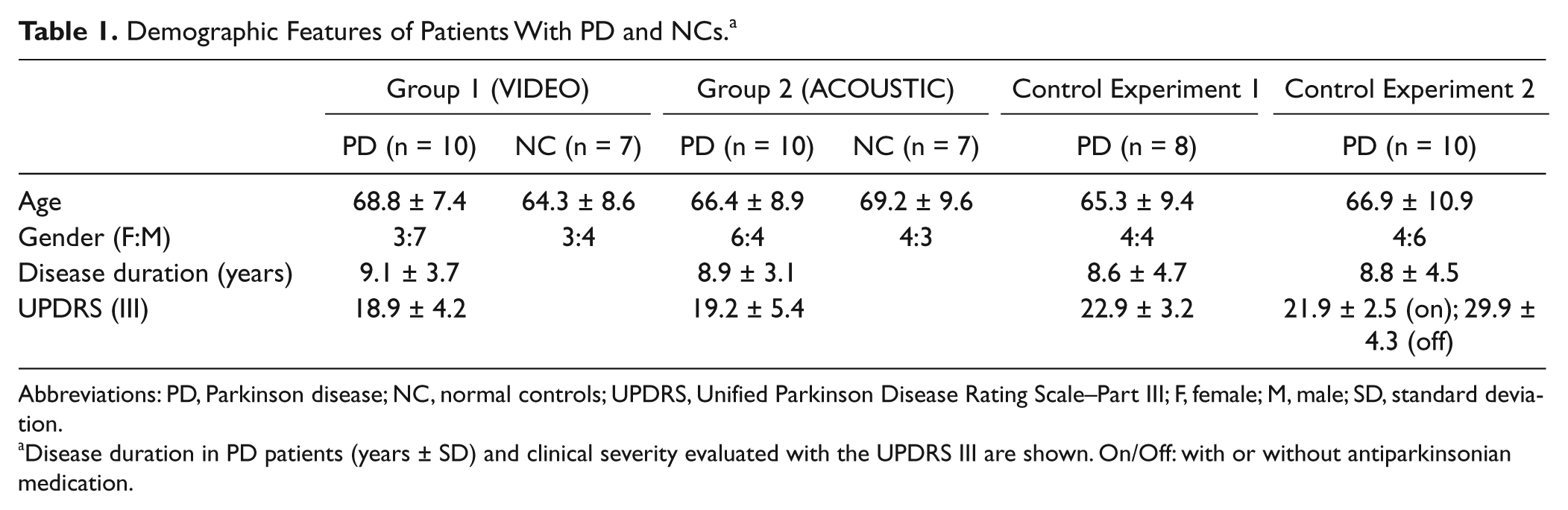
Abbreviations: PD, Parkinson disease; NC, normal controls; UPDRS, Unified Parkinson Disease Rating Scale–Part III; F, female; M, male; SD, standard deviation.
Disease duration in PD patients (years ± SD) and clinical severity evaluated with the UPDRS III are shown. On/Off: with or without antiparkinsonian medication.
Main Experiment
The experimental design of the main experiment is summarized in Figure 2. Participants enrolled in the group VIDEO (PD, n = 10; NC, n = 7) were asked to watch a 6-minute video of repetitive finger movements consisting of the opposition of thumb to index, medium, ring, and little fingers, paced at 3 Hz. The video showed the right hand of a trained actor from a third-person perspective. Participants enrolled in the group ACOUSTIC (PD, n = 10; NC, n = 7) were asked to listen for 6 minutes to an acoustic cue (generated by a metronome) paced at 3 Hz. Participants in the VIDEO group were instructed to concentrate on how the actions were performed, whereas participants in the ACOUSTIC group were instructed to focus their attention on the rhythmical cues produced by the metronome. PD patients were tested while in the on medication state.
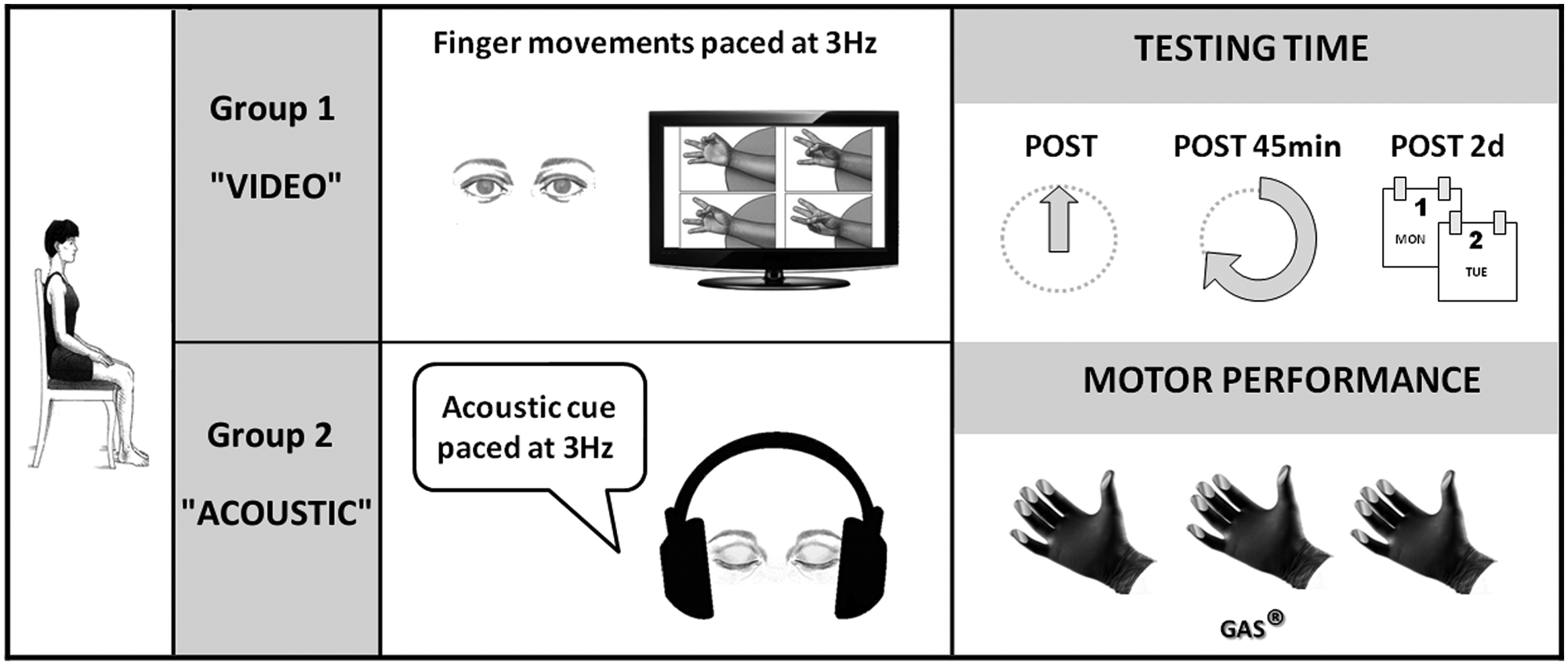
Experimental paradigm of the main experiment.
Control Experiments
Before each session, the motor disability of the patients was assessed by means of the UPDRS–Motor Section. PD patients recruited for control experiment 1 (n = 8) were asked to watch a 6-minute video representing a static hand from a third-person perspective. This session required the same amount of attention for participants and was therefore chosen as control treatment for the video intervention. PD patients were tested while in the on medication state.
PD patients recruited for control experiment 2 (n = 10) were asked to participate in the same protocol used for the VIDEO group, but they were tested twice, with an at least 3-week interval, while in the on and off medication states. Patients were randomly assigned to either of these sessions (cross-over design): on state corresponded to 90 minutes after taking the scheduled antiparkinsonian medication, whereas off state corresponded to withdrawal of any dopaminergic medication for at least 18 hours.
Outcome Measures
The main outcome measure in all the experiments was the SMR of self-paced finger movements. Kinematic parameters describing motor performance (intertapping interval and touch duration) were secondary outcome measures.
In the main experiment and in control experiment 1, motor performance was evaluated at different testing times with respect to the intervention: before the intervention (baseline: PRE), immediately after (POST), 45 minutes later (POST45min), and 2 days later (POST2d). In control experiment 2 (on/off state), patients were tested only 3 times (PRE, POST, and POST45min) because it was not possible to prolong the off state till 2 days. The motor task consisted in the execution of 2 repetitions of the same sequence of finger opposition movements (opposition of thumb to index, medium, ring, and little fingers) lasting 1 minute, separated by a 1-minute rest. Participants wore a sensor-engineered glove on their right hand (Glove analyzer System [GAS], eTT s.r.l., Italy). Data were acquired at 1 kHz (National Instrument board 800008B-01).
For each trial, we computed the duration (in milliseconds) of both the transition phase from 1 finger to the subsequent one within the sequence (intertapping interval) and the finger touching phase (touch duration). Then, we calculated the SMR as 1/(touch duration + intertapping interval) and expressed this value in Hertz. The mean value obtained from the 2 trials recorded in every session was used for the analysis.
Statistical Analysis
The sample size for the VIDEO group of the main experiment was determined based on the results of our previous pilot study on NCs. 20 Using a t test and α error probability set at .05, 6 participants would be required to detect differences within the group related to action observation at 90% of study power (G-power statistical analysis).
To avoid any attrition bias, demographic (age and gender) and clinical (UPDRS–Motor Score and disease duration) characteristics of the different groups of PD patients were compared at baseline. Furthermore, demographic characteristics of PD patients and NCs enrolled in the main experiment were also compared. The χ2 statistic test was used to assess for differences in gender, and 1-way ANOVA was used to analyze the continuous variables (age, UPDRS–Motor Score, and disease duration). The motor performance parameters (SMR, intertapping interval, and touch duration) were entered separately in a mixed repeated-measure (RM) ANOVA with the factor Time (PRE, POST, POST45min, POST2d) as a within-subject factor and Modality of Cue (VIDEO and ACOUSTIC) and Group (PD and NC) as between-subjects factors.
For the control experiments, motor performance parameters were entered separately in a RM ANOVA with the factor Time (PRE, POST, POST45min, POST2d)—control experiment 1—and Time (PRE, POST, POST45min) and Condition (on and off state)—control experiment 2—as within-subject main factors. When ANOVA gave a significant result (P < .05), post hoc analysis was performed using t tests and applying the Bonferroni correction for multiple comparisons where necessary. All statistical analyses were performed with SPSS 18.0.
Results
No significant differences in the demographic and clinical characteristics were detected between the different groups of PD patients and between PD patients and NCs enrolled in the main experiment (P always >.05).
Effect of Video and Acoustic Training (Main Experiment)
Both video and acoustic training increased the SMR, but training with video was more effective than acoustic training in modifying the motor performance at longer intervals (45 minutes and 2 days after training). The SMR in PD patients and NCs is presented in Figure 3A. RM ANOVA showed a main effect of Time—F(3, 90) = 80.35, P < .0001—and a significant interaction, Time × Modality of Cue—F(3, 90) = 7.54, P < .0001. Post hoc analysis revealed that starting from comparable SMRs at baseline (PRE: VIDEO vs ACOUSTIC P = .35), at POST, the increase of SMR was similar across the different cue modalities (VIDEO vs ACOUSTIC P = .21), whereas at POST45min and POST2d, video observation induced a larger increase of SMR than acoustic training (POST45m P = .007; POST2d P = .004). No significant interaction was found for Time × Group × Modality of Cue. However, a significant effect of Group was found—Group F(1, 30) = 124.67, P < .0001—and post hoc analysis revealed that patients with PD were always slower than NCs in performing finger opposition movements (P < .05).
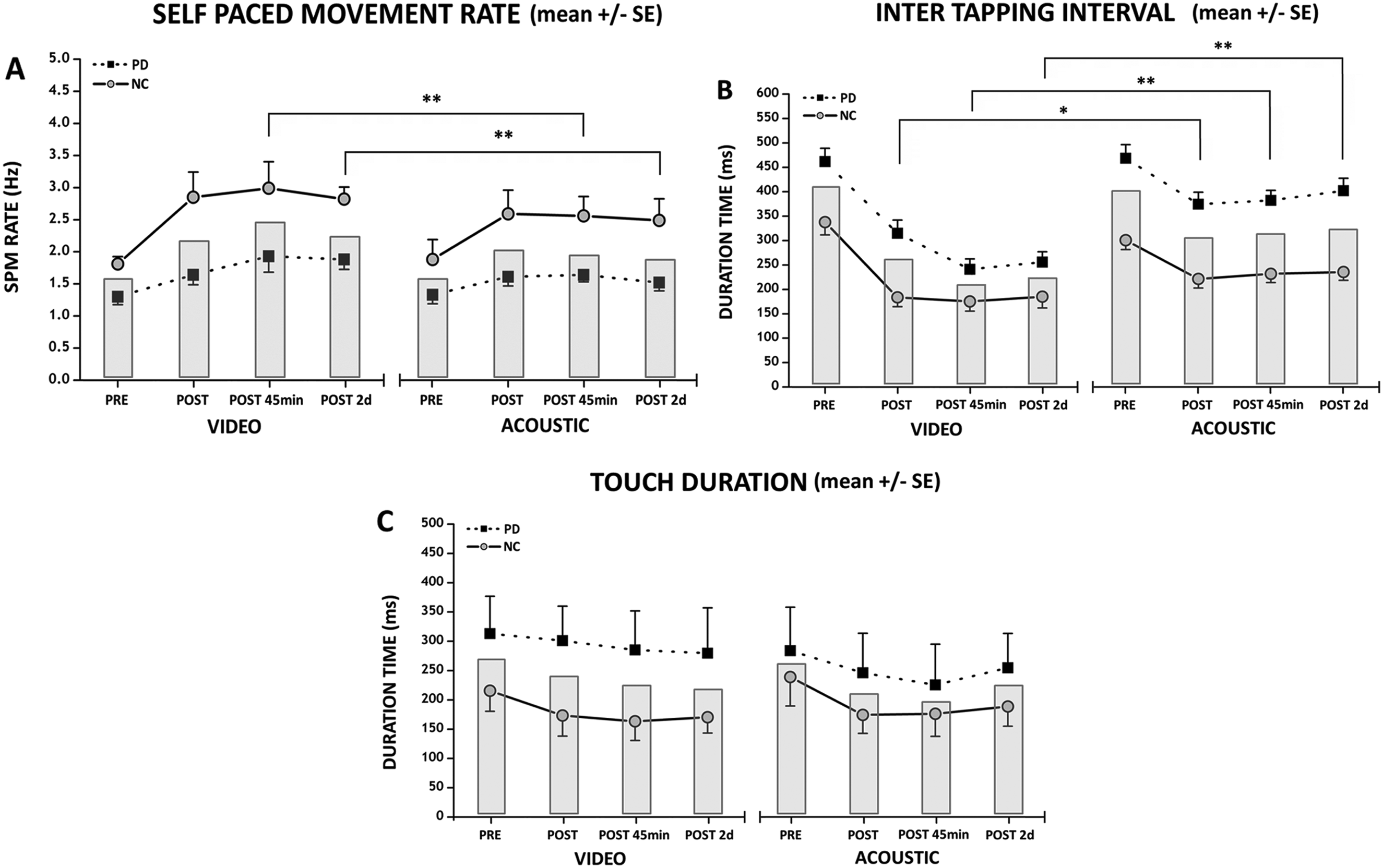
Effects of training sessions (VIDEO and ACOUSTIC) on motor performance at the different testing times in PD patients and NCs. The training modalities at the various testing times are reported in the abscissa. Data recorded at baseline (PRE) and after the training session (immediately after, POST; 45 minutes after, POST45min; 2 days after, POST 2d) are reported. The ordinates indicate the values of (A) self paced movement (SPM) rate, expressed in Hertz; (B) intertapping interval; and (C) touch duration expressed in milliseconds. Both data from patients with PD (dotted line and black squares) and data from NCs (solid line and gray circles) are reported. Columns represent mean values across the participants. Asterisks indicate significant differences between VIDEO and ACOUSTIC interventions when there was a significant interaction, Time × Modality of Cue (*P < .05, **P < .01). Vertical bars indicate SE.
Intertapping interval data are reported in Figure 3B. RM-ANOVA showed a main effect of Time—F(3, 90) = 64.10, P < .0001—and a significant interaction, Time × Modality of Cue—F(3, 90) = 11.74, P < .0001. Post hoc analysis revealed that video observation induced a decrease of intertapping interval larger than acoustic modality at all the testing times (PRE: VIDEO vs ACOUSTIC P = .39; POST: VIDEO vs ACOUSTIC P = .019; POST45min: VIDEO vs ACOUSTIC P < .001; POST2d: VIDEO vs ACOUSTIC P < .001). No significant interaction was found for Time × Group × Modality of Cue. However, a significant effect of Group was found—F(1, 30) = 124.26, P < .0001—and post hoc analysis revealed that PD patients showed a mean intertapping interval value longer than that of NCs at all testing times (always P < .05).
Touch duration data are reported in Figure 3C. RM-ANOVA showed a main effect of Time—F(3, 90) =13.37, P < .0001—and post hoc analysis revealed that PRE values of touch duration were significantly higher than those recorded at POST, POST45min, and POST2d (P always <.0001).
Time × Modality of Cue and Time × Group × Modality of Cue interactions were not significant. However, a significant effect of Group was found—Group F(1, 30) = 47.03, P < .0001—and post hoc analysis revealed that touch duration was significantly longer in PD than in NC at all testing times (P < .05).
Effect of Observation of a Static Hand Video (Control Experiment 1)
Control experiment 1 showed that the mere observation of a static image did not induce any modifications in motor performance in PD patients. Accordingly, RM-ANOVA showed no effect of Time for SMR—F(3, 18) = 0.65, P = .59—intertapping interval—F(3, 18) = 2.10, P = .13—and touch duration—F(3, 18) = 2.01, P = .15. Thus, the effect was not simply a result of video exposure nor of repeating the motor task.
Influence of the Dopaminergic State (Control Experiment 2)
At baseline, before training, significant differences were found in motor performance of PD patients between the on and off conditions (Figures 4A-4C). In the off condition, participants were slower in performing finger opposition movements—SMR, Condition F(1, 9) = 5.12, P = .040. Accordingly, the intertapping interval value was longer in off with respect to the on condition—Condition F(1, 9) = 9.01, P = .015—whereas touch duration value did not differ between the on and off states—Condition F(1, 9) = 0.43, P = .53.
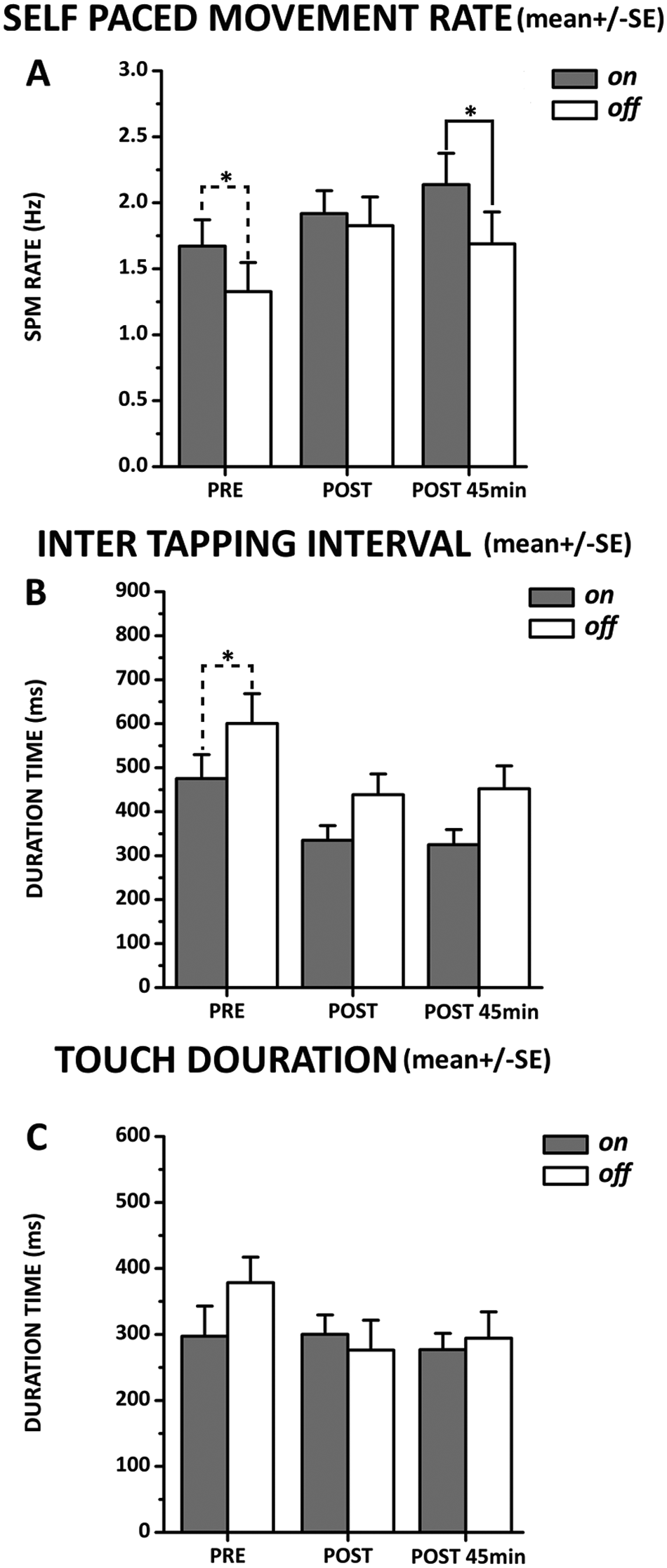
Comparison between on and off conditions. Testing times are indicated in the abscissa. Data recorded at baseline (PRE) and after the video training session (immediately after, POST, and 45 minutes after, POST45min) are reported. Ordinate indicates the mean values of (A) self pace movement (SPM) rate, expressed in Hertz; (B) intertapping interval; and (C) touch duration expressed in milliseconds. Data measured in 10 PD patients in on (gray columns) and off (white columns) states are shown. Asterisks indicate differences between on and off values when the interaction of Time × Condition was statistically significant (*P < .05, ** P < .01). Vertical bars indicate standard error (SE).
The SMR increased after action observation training in both conditions but with a different effect over time. RM-ANOVA showed a significant Time × Condition interaction—F(2, 18) = 4.02, P = .036—and post hoc analysis (Figure 4A) revealed that SMR was greater in the on than in the off condition at baseline (P = .045) and at POST45min (P = .031), whereas no significant difference was found at POST (P > .05).
Training with action observation induced a similar decrease in touch duration—Time F(2, 18) = 3.68, P = .045—and intertapping interval—Time F(2, 18) = 22.80, P < .0001—over time in the on and off conditions. Indeed, a nonsignificant Time × Condition interaction was found for touch duration—F(2, 18) = 2.64, P = .12—and intertapping interval—F(2, 18) = 1.03, P = .38.
Discussion
Our main findings were the following: (1) the observation of rapid rhythmical finger movements was able to implicitly induce an increase of spontaneous finger movement rate in PD patients similarly to NCs; (2) when compared with acoustic training, the effect of action observation was longer lasting over time thanks to a different effect on kinematic aspects of the motor performance; (3) in PD patients, the effect of action observation training was partially dependent on the dopaminergic state of the patients.
Training with both the observation of rhythmical finger movements executed at 3 Hz and external auditory cues paced at 3 Hz proved effective in increasing the spontaneous rate of repetitive finger movements, thus improving the bradykinesia of patients with PD. We can exclude the argument that this effect was a result of the fact that the participants were just repeating a motor task several times within a few days because when participants (control experiment 1) were engaged in watching a static image of a hand for 6 minutes, no significant changes in motor performance were recorded over time.
Bradykinesia may be associated with disturbances in the execution of normal motor rhythms because of dysfunction of a putative “internal clock” but can be modulated by dopaminergic medications.23-25 Rehabilitation treatment, in addition to dopaminergic therapy, can also improve bradykinesia in PD. In particular, cueing strategies may increase stride time and walking speed26,27 and reduce the variability of finger tapping. 28 Here we demonstrated that training with action observation, similarly to external auditory cues, can act on temporal processing and motor timing, leading to modifications in the spontaneous movement tempo controlled by the “internal clock” in PD.
Recent contributions have demonstrated that the basal ganglia might indeed be involved in the “mirror neuron” system in humans.29-31 For example, changes in local field potentials recorded from the subthalamic nucleus in patients with PD can be detected during the observation of a movement performed by another participant, whereas they are not present during the observation of a static image.30,31 These changes were present in both “off” and “on” motor states and were similar to those observed during movement execution.
It was reported that action observation combined with the repetition of the observed actions has a positive effect in terms of retention of information but only when observed movements are congruent with practiced movements, as in our experimental paradigm.18,20,21 The continuous implicit comparison occurring between the action performed and that observed in the video at the beginning of the training session might be responsible for this retention effect. The present results suggest that a comparison between 2 sensory representations of the movement (the “observed” or visual and the “experienced” or somatosensory) is necessary to consolidate a motor learning task. Indeed, somatosensory afferents and the somatosensory cortex play an important role in memory formation, as suggested by earlier human and animal studies.32,33 Consistently, the retention of improved performance was significantly better with action observation: training with video was more effective than acoustic training in modifying the performance of repetitive finger movements at longer intervals (45 minutes and 2 days after training).
Furthermore, our results suggest that consolidation of motor performance is modulated by dopaminergic therapy. Consolidation of memory traces depends on neuroplasticity mechanisms. Experimental studies, both in animals and humans, underscored the importance of the dopaminergic system for stabilizing plasticity processes34-36 and learning. 37 In addition, some recent works provided direct evidence of the modulator effect of dopamine therapy on different aspects of learning in the PD patients population.38,39
Another intriguing finding of the present study was that action observation influenced the kinematic aspects of motor performance differently from acoustic training. Specifically, the reduction of intertapping interval was larger than that induced by the acoustic training at all the testing times, whereas touch duration was similarly modified at all the testing times. Whereas intertapping interval is likely to represent a pure motor phase, touch duration may be regarded as the combination of a sensory phase and a motor preparation phase in which the subsequent movement is correctly planned prior to execution. When trained with the acoustic cue at 3 Hz, participants increased their spontaneous tempo by reducing similarly the duration of the time devoted to movement execution (intertapping interval) as well as the time devoted to sensorimotor preparation (touch duration). When participants were trained with the video showing finger opposition movements at a rate around 3 Hz, however, they increased their spontaneous tempo by reducing the duration of the time devoted to movement execution. This finding might suggest that action observation is able to provide information mainly dealing with the dynamic part of the movement (transition between a finger to the successive one of the sequence). Therefore, even if both types of training (action observation and acoustic cues) similarly influenced the spontaneous movement tempo, the effects on the parameters describing motor performance (intertapping interval and touch duration) were different. This result is potentially relevant in the context of neurorehabilitation, opening up the possibility of designing an ad hoc training strategy on the basis of individual motor performance impairment.
Our study has certain limitations. First, the observed positive effect of action observation was obtained in a relatively small sample that is not necessarily representative of the whole PD population. A larger group of patients needs to be tested to verify the generalizability of our results. In addition, in this pilot study, we verified the effect of a single training session in improving finger movements; a longer period of training must be examined.
Conclusions
Our preliminary results show that action observation seems to be particularly effective in terms of retention of the acquired information. The maintenance of motor improvements is likely to reflect dopaminergic-associated training-induced neuroplasticity. The findings represent a promising approach for the implementation of training with action observation in clinical practice.
Footnotes
Authors’ Note
EP and MB contributed equally to this work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received any financial support for the research, authorship, and/or publication of this article.
