Abstract
Background. Brain-computer interfaces (BCIs) allow people to control devices by translating brain signals into commands. BCIs represent a concrete solution with regard to communication and motor control disabilities of patients with amyotrophic lateral sclerosis (ALS). Most of the BCIs rely on visual interfaces in which patients must move their eyes to achieve efficient BCI control. This fact represents a limitation of BCI use in ALS patients who are in the final stages of the disease. Objective. We aimed to improve visual interfaces for ALS patients to control the movement of a cursor on a monitor by orienting their covert visuospatial attention (ie, orienting without eye movements). Methods. A total of 10 ALS patients with different levels of impairment used 2 new visual interfaces in an event-related potential (ERP)–based BCI. In the first interface, they were required to use exogenous visuospatial attention orienting (VAO), whereas in the second interface, they were required to use endogenous VAO. Results. ALS patients were able to use the 2 interfaces for controlling the ERP-based BCI system in real time. Nevertheless, better target classification and information transfer rate were associated with the interface that was based on endogenous VAO. Conclusions. ALS patients can exploit their covert VAO to control a BCI that does not require eye movements. The implementation of endogenous VAO in the design of covert visuospatial attention-based interfaces seems to be suitable for designing more ergonomic and efficient BCIs for ALS patients with impaired eye movements.
Keywords
Introduction
Amyotrophic lateral sclerosis (ALS) is a neurodegenerative disease that leads affected patients to paralysis and death typically within 2 to 5 years from the initial diagnosis. 1 ALS patients progressively enter into a condition in which eye muscle control and/or external sphincter control are usually the only spared movements (ie, locked-in state 2 [LIS]). In the later stages of the illness, however, muscle control becomes impossible. In this case, ALS patients enter into the so-called complete locked-in state (CLIS).
By using brain-computer interfaces (BCIs), ALS patients can communicate and interact with their environment. 3 BCIs, indeed, allow users to control external devices such as computers or prostheses without the aid of the users’ peripheral nerves and muscles. ALS patients constitute the most investigated clinical population in BCI studies. 4 Most of the BCIs tested with ALS patients rely on EEG signals (ie, event-related potentials [ERP], slow cortical potentials [SCPs], and sensorimotor rhythms) and on visual interfaces. ALS patients with minor, moderate, or major impairment 5 are able to control online visual BCIs through ERPs,6,7 SCPs,8,9 and sensorimotor rhythms.10,11
Birbaumer et al 12 first described 2 ALS-LIS patients who successfully used the SCPs for communicating through a word spelling system. Efficient control has been reached by other ALS-LIS patients.6,9,10,13-17 In a remarkable longitudinal study, 18 an ALS-LIS patient was described who, for more than 2 years, was able to control the most investigated BCI (ie, the P300-speller 19 ). Nevertheless, in some other studies, ALS-LIS patients were unable to use a BCI.15,20
Less encouraging are the findings of studies on ALS-CLIS patients. Only a few cases have been described in the literature, and none of them was able to reach an acceptable level of BCI control.8,21,22 In their meta-analysis, Kübler and Birbaumer 5 have hypothesized that the reason for this failure might be the extinction of goal-directed thinking in ALS-CLIS patients. An alternative explanation might be that visual BCIs are not suitable for ALS-CLIS patients. The use of auditory23-27 or tactile 28 BCIs can be the solution, but these BCIs have never been tested on ALS-CLIS patients. In summary, visual BCIs for communication seem to be suitable for ALS patients with minor, moderate, and major impairment but not for those who are in the CLIS condition.
Visual BCIs have been successfully used by ALS-LIS patients in some cases6,9,10,13-17 but not in others.15,20 It is clear that the evolution of the illness leads ALS-LIS patients to progressively lose their eye movement control. 29 Recent studies, however, have shown that successful use of the P300-speller requires participants to be able to move their gaze.30,31 Thus, the development of gaze-independent visual BCIs is required for use with ALS-LIS patients before they enter the CLIS condition.
Recently, we have designed and implemented new visual interfaces32,33 in which healthy participants used their covert visuospatial attention orienting (VAO; ie, shift of the focus of spatial attention without eye movements) to control a virtual cursor on a monitor by means of their ERPs (ie, P300 and late negative component [LNC] 33 ). In the aforementioned studies,32,33 our aim was to investigate whether there was an advantage in implementing the principles of covert VAO 34 in our interfaces. Many studies (for review 35 ) have suggested that covert VAO can be oriented by 2 types of cues: peripheral cues, which elicit an exogenous VAO (ExVAO), and central cues, which activate endogenous VAO (EnVAO). The results confirmed that healthy participants can use a BCI in a covert VAO condition.36,37 Moreover, healthy participants reached higher performance with the interface that required EnVAO than with the interfaces that required ExVAO. To date, however, no one has tested covert VAO with ALS patients to investigate whether these patients are able to use visual BCIs, even when their eye movements are impaired.
In the present study, we tested a group of ALS patients who showed different levels of motor impairment. We tested their ability to control a visual BCI without performing eye movements. In addition, we hypothesized that ALS patients, like healthy participants, 32 would control the BCI better by means of EnVAO than by means of ExVAO. Finally, we investigated whether there was an effect of pathology level (measured through the revised ALS functional rating scale: ALSFRS-R 38 ) on participants’ performance.5,7
Methods
Participants
In all, 10 ALS patients gave their informed consent to participate in the study. Their demographic and clinical data are reported in Table 1. Participants were recruited from ALS patients who were in clinical treatment at the IRCCS San Camillo Hospital, Venice-Lido. The study was approved by the ethical committee of the hospital.
Demographic and Clinical Data of ALS Patients.
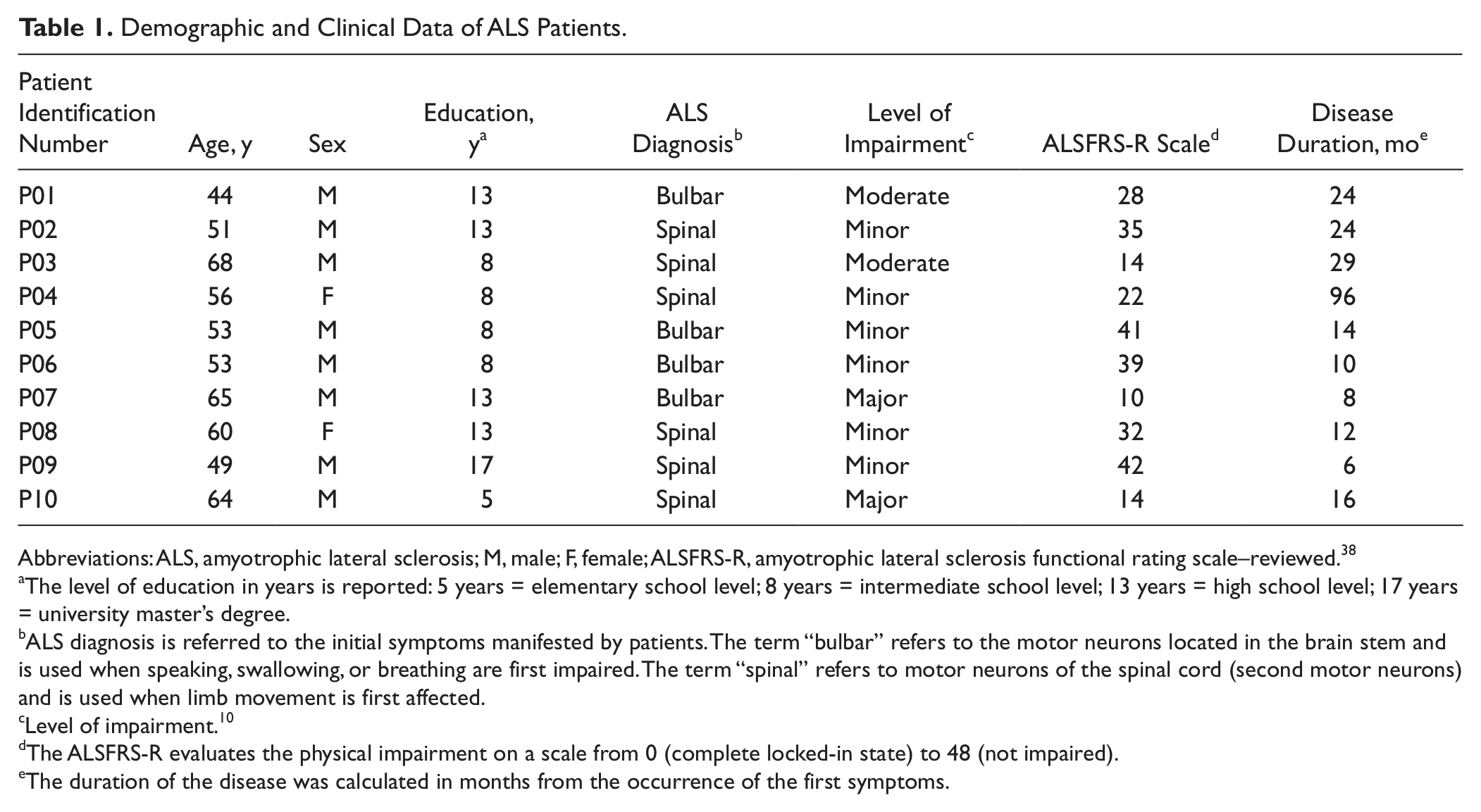
Abbreviations: ALS, amyotrophic lateral sclerosis; M, male; F, female; ALSFRS-R, amyotrophic lateral sclerosis functional rating scale–reviewed. 38
The level of education in years is reported: 5 years = elementary school level; 8 years = intermediate school level; 13 years = high school level; 17 years = university master’s degree.
ALS diagnosis is referred to the initial symptoms manifested by patients. The term “bulbar” refers to the motor neurons located in the brain stem and is used when speaking, swallowing, or breathing are first impaired. The term “spinal” refers to motor neurons of the spinal cord (second motor neurons) and is used when limb movement is first affected.
Level of impairment. 10
The ALSFRS-R evaluates the physical impairment on a scale from 0 (complete locked-in state) to 48 (not impaired).
The duration of the disease was calculated in months from the occurrence of the first symptoms.
Apparatus, Stimuli, and Procedure
Participants sat in front of a monitor (HP L1906T Flat Panel LCD Screen; dimensions: 38 × 30.5 cm; refresh frequency: 60 Hz; resolution: 1024 × 768). The apparatus of the present study was identical to that we had previously used with healthy participants, 32 except for the use of a chin rest. Patient P01 was tracheotomized, and patients P04, P08, P09, and P10 sat in a wheelchair; for these reasons, it was impossible for them to use the chin rest. The monitor was placed at a distance of about 60 cm in front of each participant’s eyes. This experimental setting allowed us to test the use of our BCI system in a more natural situation.
Two interfaces 32 were presented to the participants: the “Auto” interface (Figure 1B) and the “Vol” interface (Figure 1C). Both interfaces were designed for controlling the movement of a virtual cursor on a monitor. The cursor’s control was used for reaching target icons displayed on the monitor. The icons showed everyday activities that might be used by the patients for communicating their needs (eg, the icon of a doctor for requiring his or her assistance; Figure 1A).
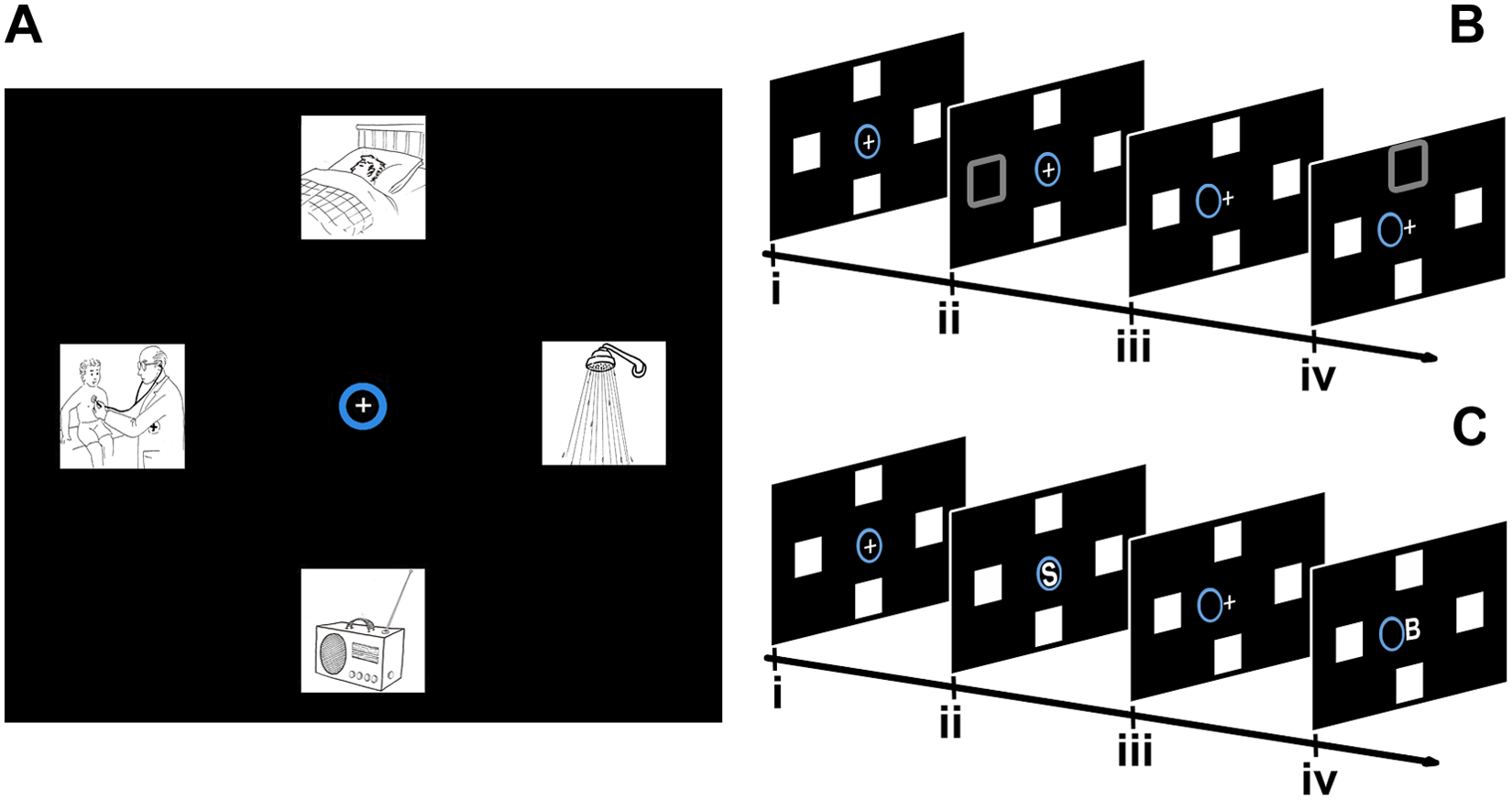
The interfaces: A. Reproduction of the display settings for both the interfaces at the beginning of a session, composed of the cross, the cursor (blue circle), and the 4 icons (adapted from a battery for the assessment of aphasics, 39 used with the kind permission of the authors). B. The “Auto” interface, schematic representation of a trial: (i) initial situation; (ii) offset of an icon for 75 ms; (iii) feedback; if the target ERP was detected, the cursor moved 1 step toward the target icon; and (iv) offset of the next icon. C. The “Vol” interface; schematic representation of a trial: (i) initial situation; (ii) onset of a “directional” letter for 900 ms; (iii) feedback; if the target ERP was detected, the cursor moved 1 step toward the target icon; and (iv) onset of the next letter.
In the “Auto” interface, the principle of ExVAO was implemented. In this, 4 icons were placed in the periphery of the monitor. All icons were presented at a distance of 7 cm from the central cross. During the sessions, the icons disappeared for 75 ms and reappeared at the same spatial position. The offset/onset order of the icons was semirandom. Each participant was asked to pay attention to the icon that was in the target spatial position (ie, the to-be-reached icon that was indicated by the experimenter at the beginning of each session) to guide the cursor toward the target spatial position. Moreover, participants were required to keep their gaze on the cross and ignore the offset of the icons in the nontarget spatial positions.
In the “Vol” interface, the principle of EnVAO was implemented. The 4 icons remained always displayed on the monitor. Four capital letters were presented on the cross, 1 at a time, for 900 ms, in a sequential semirandom order, and then they disappeared. Each one was the initial letter of an Italian directional word and indicated the spatial position of 1 of the icons. Participants were required to pay attention to the letter indicating the direction of the target spatial position (ie, the to-be-reached icon that was indicated by the experimenter at the beginning of each session) and to ignore the others. Moreover, participants were required to keep their gaze fixed on the cross.
Participants’ EEG was recorded during the presentation of the trials with both the interfaces (ie, “Auto”: the offset of an icon for 75 ms; “Vol”: the onset of a letter for 900 ms). The ERPs related to each trial were processed online by an ad hoc classifier (see next paragraph for a description of the online classification procedure).7,32 If participants performed the task correctly, different ERPs were elicited by the target trials than by the nontarget trials. The classifier was trained for detecting the difference between the features in the ERPs related to the target trials and those related to the nontarget trials. When the classifier detected the features of the ERPs related to a target trial, the cursor was moved 1 step toward the cued spatial position (ie, true positive). Otherwise, the cursor was not moved. 32 To reach an icon, participants needed to perform at least 4 steps with the cursor toward the direction of the icon. Note that also when the classifier detected a target ERP following a nontarget cue, the cursor moved 1 step toward the nontarget direction (ie, false positive). A session was defined as the sequence of trials needed to reach the target or 1 of the nontargets, or it ended after 92 trials if neither a target nor a nontarget was reached. The number of trials presented during the BCI sessions was different among the participants and within the sessions performed by each participant (range, 13-92 trials).
The present study took place during 9 consecutive days. On the first day, each participant performed 8 training sessions with each interface. The training sessions were characterized by the automatic movement of the cursor after each target trial occurred. The training sessions were performed to collect a sample of EEG epochs for training the classifier that was used on the first day of the experimental sessions. In the following 8 days, each participant performed 4 testing sessions per day. On each day, each participant used either the “Vol” or the “Auto” interface. For half of the participants, the order of interface presentation during the 8 days was: “Vol”-“Auto”-“Auto”-“Vol”-“Auto”-“Vol”-“Vol”-“Auto.” For the other half of the participants, the order of interface presentation on the 8 days was “Auto”-“Vol”-“Vol”-“Auto”-“Vol”-“Auto”-“Auto”-“Vol.” The target spatial position was different in each of the 4 testing sessions. The order of the target positions was counterbalanced across the 8 days.
Electrophysiological Data Acquisition and Online Processing
The montage of the electrodes was performed according to the International 10-20 System at Fz, Cz, Pz, and Oz. The electro-oculogram was recorded from a pair of electrodes below and laterally to the left eye. All electrodes were referenced to the left earlobe, and the ground was on Fpz. Impedance was lower than 5 kΩ. The 5 channels were amplified, band-pass filtered between 0.15 and 30 Hz, and digitized at 200 Hz sampling rate. Each ERP epoch was synchronized with the occurrence of the stimulus (ie, the offset of the icon in the “Auto” interface and the onset of the letter in the “Vol” interface). Each epoch began 500 ms before the stimulus onset and ended 1000 ms after the stimulus onset. Before each testing day and for each of the 2 interfaces, the classifier was trained and adapted ad personam through a 3-step procedure: independent component analysis decomposition, fixed features extraction, and support vector machine classification.7,32 The 3-step classification procedure was applied online to each single sweep synchronized with each trial. The output of the support vector machine classifier was used to control the movements of the cursor. The average number of trials added after each day of testing sessions was 254 (standard deviation [SD] = 72) and 224 (SD = 59) for the “Auto” and “Vol” interfaces, respectively. The variability between the interfaces and among the ALS patients depended on the classification accuracy that was reached by the participants during the online sessions.
Electrophysiological Data and Offline Processing
ALS patients were required to maintain their gaze fixed on the cross while they were performing the BCI sessions. In this way, we aimed to virtually simulate the condition of ALS-LIS patients, who cannot move their eyes voluntarily while using a visual BCI. Moreover, epochs containing eye movement artifacts were excluded from the statistical analyses. The criterion of ±75 µV was chosen to detect the artifacts that were caused by eye blinks 40 and to detect gaze shifts greater than 5° of visual angle. In fact, a bipolar electro-oculogram recording permits us to detect deflections of about 16 µV for each degree of eye movement. 41 The distance between the fixation point and the center of each icon (ie, “Auto” and “Vol” interfaces) measured 7° of visual angle. The cutoff level of ±75 µV permitted us to detect whether there were artifacts in the epochs because of gaze shifts from the center toward one of the icons displayed in the periphery of the screen (or vice versa). On average, 6.42% of trials were rejected for each participant (ie, “Auto” interface: M = 6.02%; “Vol” interface: M = 6.84%).
Design
Efficiency
The independent variables for testing the effects of VAO on the efficiency of the BCI were the following: interface (“Auto,” “Vol”) and day (Day 1, Day 2, Day 3, Day 4). The dependent variables were the accuracy (correctly classified trials in percentage 42 ), the percentage of error in the classification in the target trials, and the information transfer rate (ITR) measured in bit/min. 42
Event-related potentials
The independent variables for testing the effects of VAO on the ERPs were the following: interface (“Auto,” “Vol”), channel (Fz, Cz, Pz, and Oz), and trial class (target, nontarget). The dependent variables were the amplitude of the P300 and the amplitude of the LNC, taken from the epoch grand average of all the experimental sessions for each ALS patient after having excluded the epochs affected by eye movements (ie, epochs exceeding ±50 µV). The amplitude of the P300 was defined as the average ERP amplitude from 300 to 600 ms. The amplitude of the LNC was defined as the average ERP amplitude from 600 to 995 ms. The time windows used for defining the amplitude were identified through visual inspection of the grand average ERPs.
Disease level
To test whether there was any influence of disease level on the efficiency of BCIs, we performed, for each interface, a linear regression by means of the ALSFRS-R score as predictor of the performance indexes.
Results
We ran analyses of variance (ANOVAs) for repeated measures. The Greenhouse-Geisser correction coefficient is reported when the assumption of sphericity was violated.
Accuracy
There was an improvement in the accuracy (Figure 2A) as a function of Day (day 1: mean [M] = 64.89%, SD = 6.1; day 2: M = 68.64%, SD = 4.9; day 3: M = 70.77%, SD = 5.1; day 4: M = 71.39%, SD = 6.4); F(3, 27) = 12.15, P < .001,
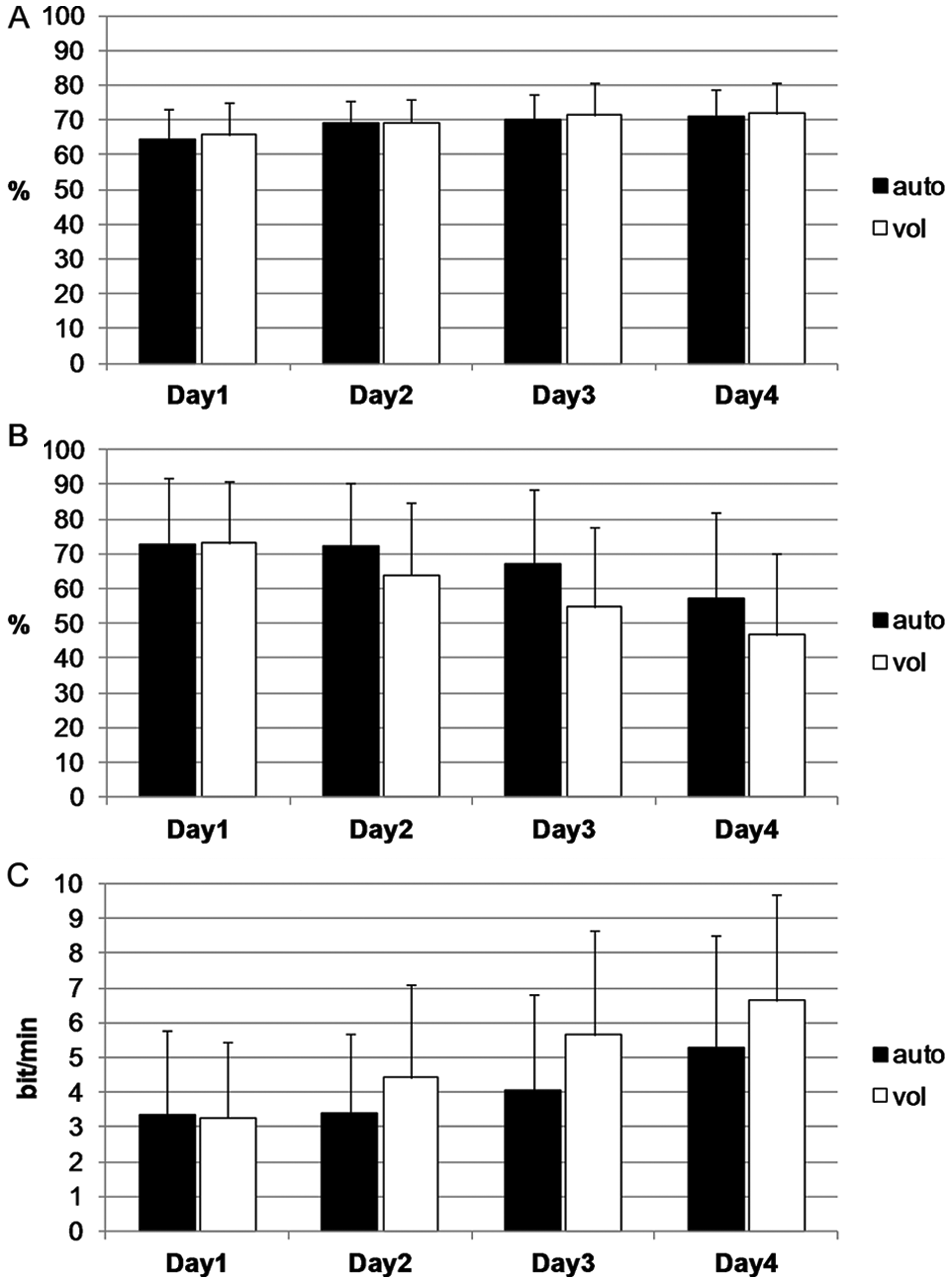
Interface performance: mean percentage and standard deviation of (A) the accuracy (%), (B) the classification errors on target trials (%), and (C) the information transfer rate (in bit/min) for the “Auto” and the “Vol” interfaces for the 4 days.
Classification Errors on Target Trials
The analysis of the classification errors on target trials (Figure 2B) revealed that there were fewer classification errors on target trials in the “Vol” interface (M = 60.39%; SD = 17.7) than in the “Auto” interface (M = 67.24%; SD = 16.9); F(1, 9) = 3.83, P < .05, 1-tailed,
Information Transfer Rate
The analysis of the communication speed data (Figure 2C) showed that patients reached a higher ITR using the “Vol” interface (M = 5.11 bit/min, SD = 2.34) than using the “Auto” interface (M = 4.13 bit/min, SD = 2.28): F(1, 9) = 5.32, P < .05, 1-tailed,
P300 Amplitude
The ANOVA results for the mean amplitude of the P300 are shown in Table 2. For reasons of clarity, only the results that were relevant for our hypotheses are extensively reported within the text below, with particular regard to the Trial Class factor. The P300 amplitude was not differently modulated by the target (M = 1.07 µV, SD = 0.91) and the nontarget trials (M = 1.02 µV, SD = 0.95); F(1, 9) = 2.67, P > .05. This was true for both the interfaces (Interface × Trial Class interaction, F(3, 27) < 1) and for all the channels (Channel × Trial Class, F(3, 27) = 2.59, P > .05). Moreover, the Interface × Channel × Trial Class interaction was not significant: F(3, 27) < 1.
Results of the ANOVAs for the P300 Amplitude and the LNC Amplitude.
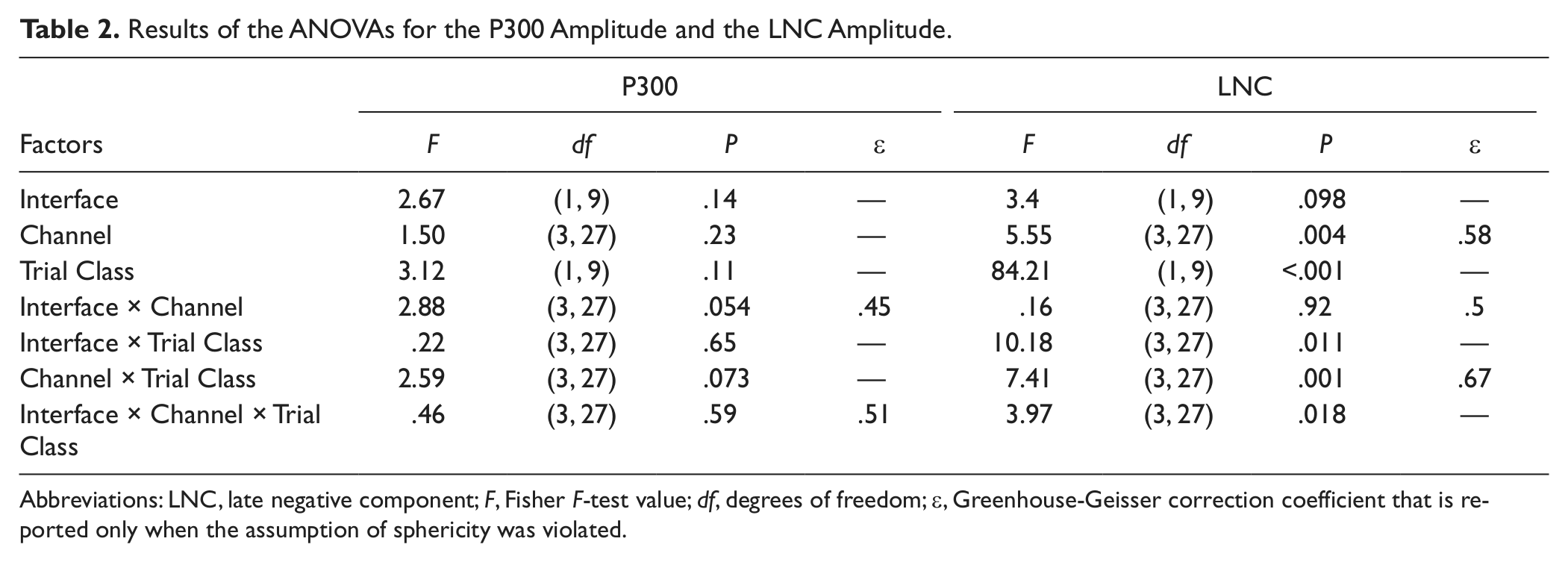
Abbreviations: LNC, late negative component; F, Fisher F-test value; df, degrees of freedom; ε, Greenhouse-Geisser correction coefficient that is reported only when the assumption of sphericity was violated.
LNC Amplitude
The ANOVA results for the mean amplitude are reported in Table 2. As for the P300 amplitude, only the results concerning the modulation of the LNC because of the Trial Class factor are described in detail.
There was a larger negativity in the LNC epoch window on the target (M = −0.44 µV; SD = 0.68) than on the nontarget trials (M = −0.04 µV, SD = 0.59); F(1, 9) = 84.21, P < .001,
Disease Level
The effect of disease level on BCI use was tested by means of linear regression. The ALSFRS-R score was used as the predictor for each of the following dependent variables in both the interfaces: accuracy, classification errors on target trials, and ITR. The disease level measured with the ALSFRS-R scale did not predict ALS patients’ ability to control the BCI. The parameters of the statistical analyses are reported in Table 3.
Disease Level Effects on BCI Use. a
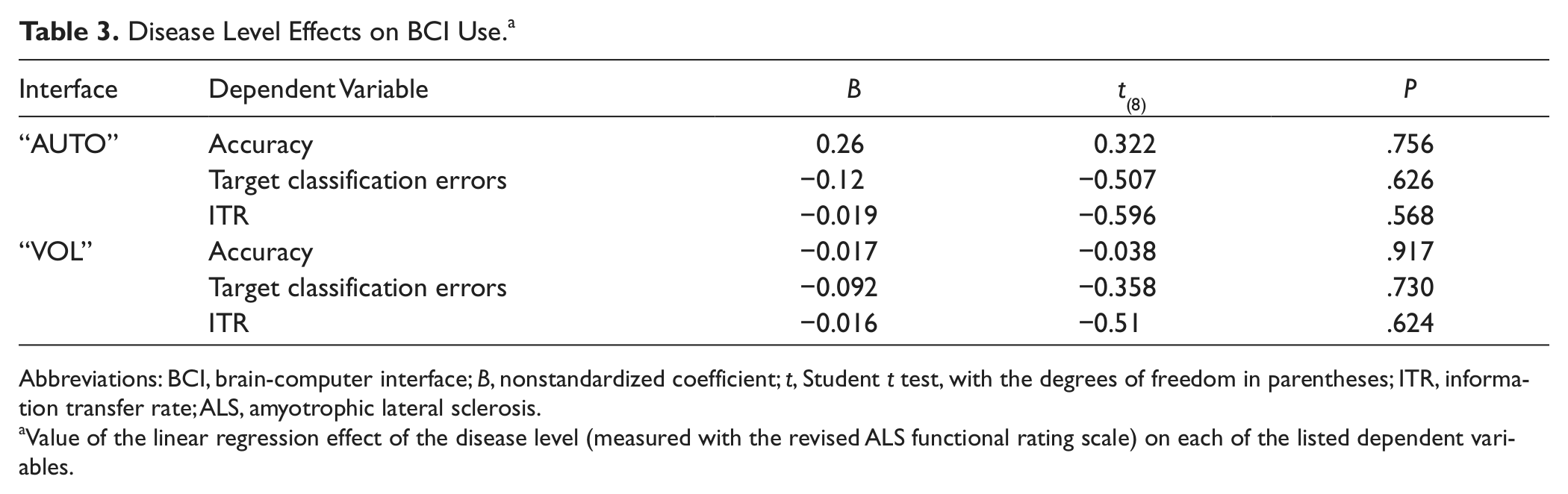
Abbreviations: BCI, brain-computer interface; B, nonstandardized coefficient; t, Student t test, with the degrees of freedom in parentheses; ITR, information transfer rate; ALS, amyotrophic lateral sclerosis.
Value of the linear regression effect of the disease level (measured with the revised ALS functional rating scale) on each of the listed dependent variables.
Furthermore, we investigated whether there was a relation between the disease level of ALS patients and the amplitude of the ERPs elicited by the 2 interfaces (Figure 3). We performed a Pearson correlation between the ALSFRS-R score and the amplitude of both the P300 and the LNC for target and nontarget trials of the “Auto” and “Vol” interfaces and for all the recorded channels (ie, Fz, Cz, Pz, and Oz). There were no significant correlations (all P > .05), even without using any correction for the multiple correlations performed.
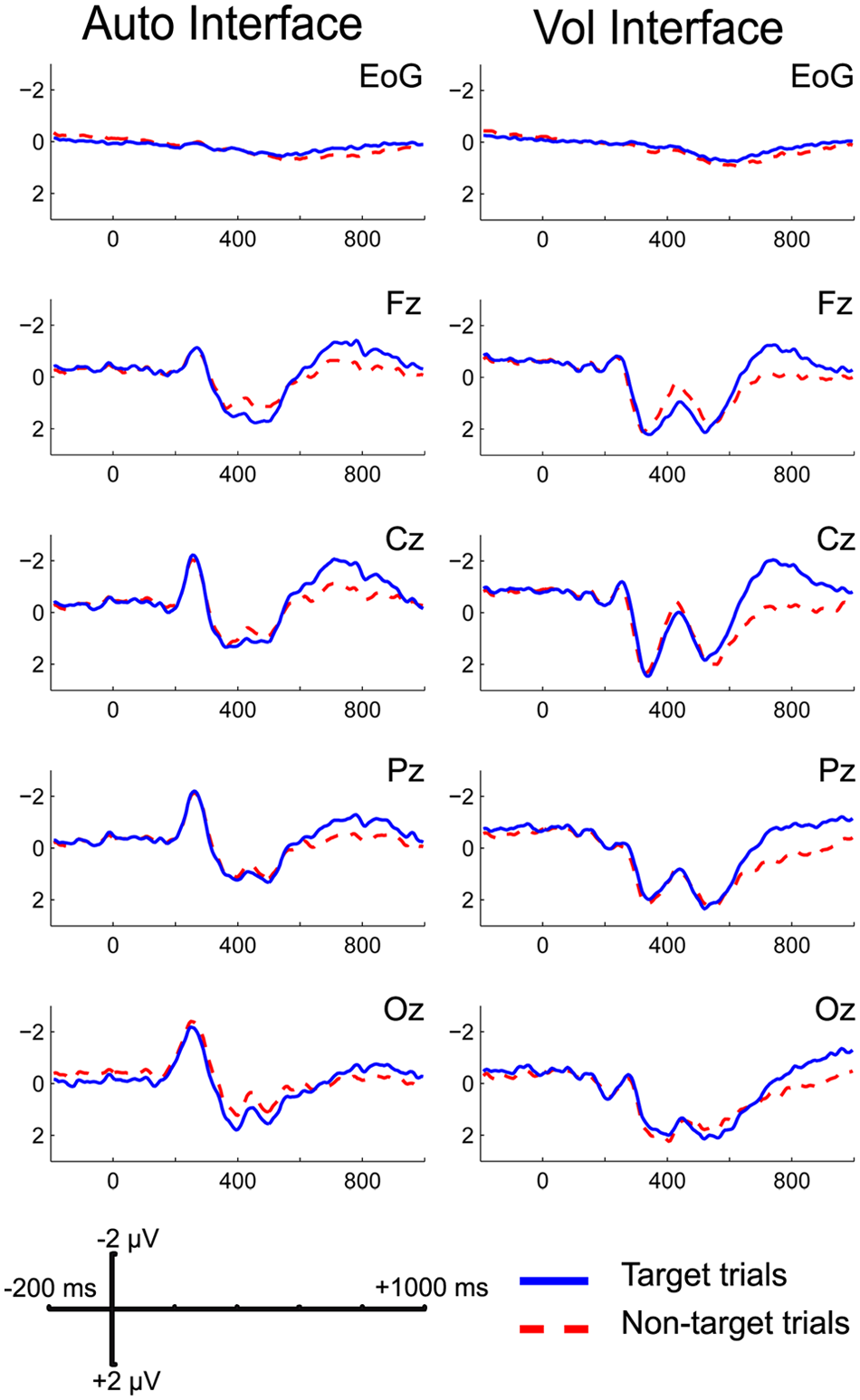
ALS patients’ grand average of the ERPs elicited by the 2 interfaces. Abbreviations: ALS, amyotrophic lateral sclerosis; ERP, event-related potential.
Discussion
We tested the effects of 2 visual interfaces, each based on different principles of covert attention orienting (ie, exogenous and endogenous 34 ), with ALS patients. Participants reached good performance in controlling a cursor with both the interfaces. Even if the mean accuracy of ALS patients was lower than 75% (about 70%), it has to be taken into account that this result might have been affected by our experimental procedures, which were mainly designed for comparing the 2 interfaces and not for ameliorating the overall efficiency of the system. ALS patients were better at using the “Vol” interface than the “Auto” interface. That is, higher ITR and fewer errors in target classification were associated with the use of the “Vol” interface. The better results obtained with the “Vol” interface might be a result of a different effect of the “Vol” interface on the ERPs. In fact, a lower LNC amplitude on nontarget trials was associated with the “Vol” interface. This resulted in a larger difference between target and nontarget trials with the “Vol” interface, which in turn might explain the advantages of ALS patients while using the “Vol” interface. Then, we tested whether there was an effect of ALS patients’ disease level on BCI use (ie, accuracy [in percentage], error in targets classification [in percentage], and ITR [in bit/min]). There was no significant relation between ALS patients’ level of impairment and the results obtained using the 2 interfaces. This finding is in line with previous results.5,7
Visual BCIs are the most used devices for improving communication in ALS patients. Performance with the most used visual BCI (ie, the P300-speller) depends on the possibility of the users to move their gaze.30,31 This makes it difficult for patients with impaired eye muscle control to use the BCIs, such as ALS patients in the final stages of the illness. Thus, new interfaces that do not depend on eye movements might be required. Our results, however, support the idea that ALS patients who have impaired eye movements can use the covert VAO to avoid the problems related to BCIs that require eye movements. Note that our ALS patients were neither in the LIS nor in the CLIS condition. Nevertheless, both our findings and those from other studies5,7 have suggested that there is no relation between disease level and BCI performance, at least after CLIS patients are excluded. This allows us to hypothesize that our new interfaces might be efficient also with ALS-LIS patients. This hypothesis, however, can be definitively tested only by means of a study on ALS-LIS patients.
Other systems for communication with ALS patients in the final stages of their illness (eg, eye-tracking systems and alternative augmentative communication devices) cannot be easily used because these patients’ eye movement control gets progressively worse. We suggest that an efficient BCI system based on a covert VAO interface guided by endogenous orienting might be suitable for permitting ALS patients to communicate their needs, even in the advanced stages of their illness.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
