Abstract
Background. Recovery from peripheral nerve repair is frequently incomplete. Hence drugs that enhance nerve regeneration are needed clinically. Objectives. To study the effects of nandrolone decanoate in a model of deficient reinnervation in the rat. Methods. In 40 rats, a 40-mm segment of the left median nerve was removed and interposed between the stumps of a sectioned right median nerve. Starting 7 days after nerve grafting and continuing over a 6-month period, we administered nandrolone at a dose of 5 mg/kg/wk to half the rats (n = 20). All rats were assessed behaviorally for grasp function and nociceptive recovery for up to 6 months. At final assessment, reinnervated muscles were tested electrophysiologically and weighed. Results were compared between rats that had received versus not received nandrolone and versus 20 nongrafted controls. Results. Rats in the nandrolone group recovered finger flexion faster. At 90 days postsurgery, they had recovered 42% of normal grasp strength versus just 11% in rats grafted but not treated with nandrolone. At 180 days, the average values for grasp strength recovery in the nandrolone and no-nandrolone groups were 40% and 33% of normal values for controls, respectively. At 180 days, finger flexor muscle twitch strength was 16% higher in treated versus nontreated rats. Thresholds for nociception were not detected in either group 90 days after nerve grafting. At 180 days, nociceptive thresholds were significantly lower in the nandrolone group. Conclusions. Nandrolone decanoate improved functional recovery in a model of deficient reinnervation.
Introduction
Despite progress since the introduction of microsurgical techniques, incomplete recovery following nerve repair is still the rule. 1 It is agreed among surgeons that further improvements in nerve reconstruction outcomes will not be linked to improved techniques of nerve repair but rather to the development of biological factors that have the potential to stimulate nerve regeneration. In this regard, many drugs have been demonstrated to improve nerve regeneration in the laboratory; however, to date, none have found their way into clinical practice. 2
Anabolic-androgenic steroids enhance athletic performance in men and women 3 and are capable of increasing muscle power by up to 20%. 4 Among the various anabolic steroids, nandrolone decanoate has fewer side effects because it is minimally androgenic. 5 Consequently, nandrolone decanoate has been selected to treat several clinical conditions associated with muscle wasting.6-8 In terms of nerve regeneration, Vita et al 9 demonstrated earlier electrophysiological signs of muscle reinnervation in rats given nandrolone decanoate to treat a crush injury to the sciatic nerve. However, crush injuries do not disrupt the endoneurial tubes and therefore represent a model of axonotmesis, 10 a second degree nerve lesion, according to Sunderland’s classification, which progresses to total recovery without requiring surgical repair. 11 Hence the axonotmesis model of nerve injury in the rat does not mimic the real-life clinical scenario of complete nerve transection in humans, in which the potential for nerve regeneration is poor. Isaacs et al 12 recently demonstrated improved muscle contraction following nandrolone administration in a sciatic nerve model in a short-term controlled study. These authors used a short nerve graft that, in rats, invariably ultimately progresses to full or almost full recovery. 13 Moreover, Vita et al 9 and Isaacs et al 12 elected not to use any behavioral evaluation methods, which are an important component of research on neural rehabilitation. In this context, we have developed a method to induce deficient reinnervation in the rat by interposing a long 40-mm nerve graft between the divided stumps of a median nerve. The advantage of using a median nerve model is the potential to evaluate grasp strength behaviorally. 14 In this model of deficient reinnervation, we have observed a long delay in the initiation of recovery (ie, 44 days vs 8 days after crushing) and a maximum of 65% of grasp strength recovery 12 months after surgery. In contrast, after crushing the median nerve, rats only require 1 month for full recovery. 14 Also, in our model of deficient reinnervation, nociceptive recovery thresholds correspond to only 30% that of normal controls. 15
In the present study, we investigated the effects of nandrolone decanoate on motor and sensory recovery in the just-described rat median nerve model of deficient reinnervation, in a 3-arm, randomized controlled study.
Materials and Methods
A total of 60 female Sprague-Dawley rats, weighing an average of 238 g (standard deviation [SD] ± 21), were studied after obtaining necessary approval from our institution’s animal review board, in accordance with international guidelines for research on animals. All rats were housed in groups of 3 per cage in a temperature- and humidity-controlled environment with a 12:12 day-night cycle and were provided food and water ad libitum. Rats were randomly allocated to 3 different groups. In the first and second groups (n = 20 per group), the right median and ulnar nerves were sectioned, and the median nerve was repaired using a 40-mm long nerve graft harvested from the left median nerve. In the left forepaw, a 10-mm segment of the ulnar nerve was also removed (Figures 1 and 2). This produced bilateral finger flexion paralysis. The left forepaw was used as a negative control. Also, the absence of grasping in the left forepaw is necessary for proper assessment of the grafted right median nerve. 14
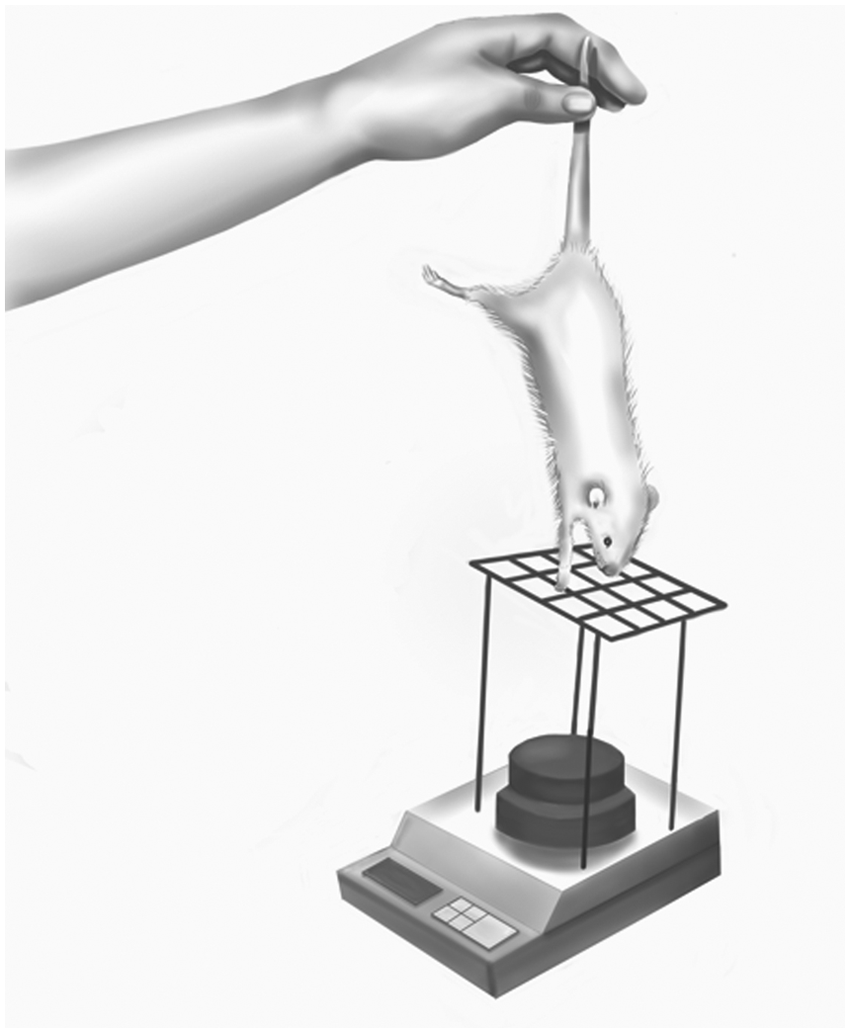
Schematic representation of the grasp strength assess-ment: it is important during this assessment to ensure full elbow and wrist extension, so as to precisely measure finger flexion rather than associated elbow and wrist flexion.
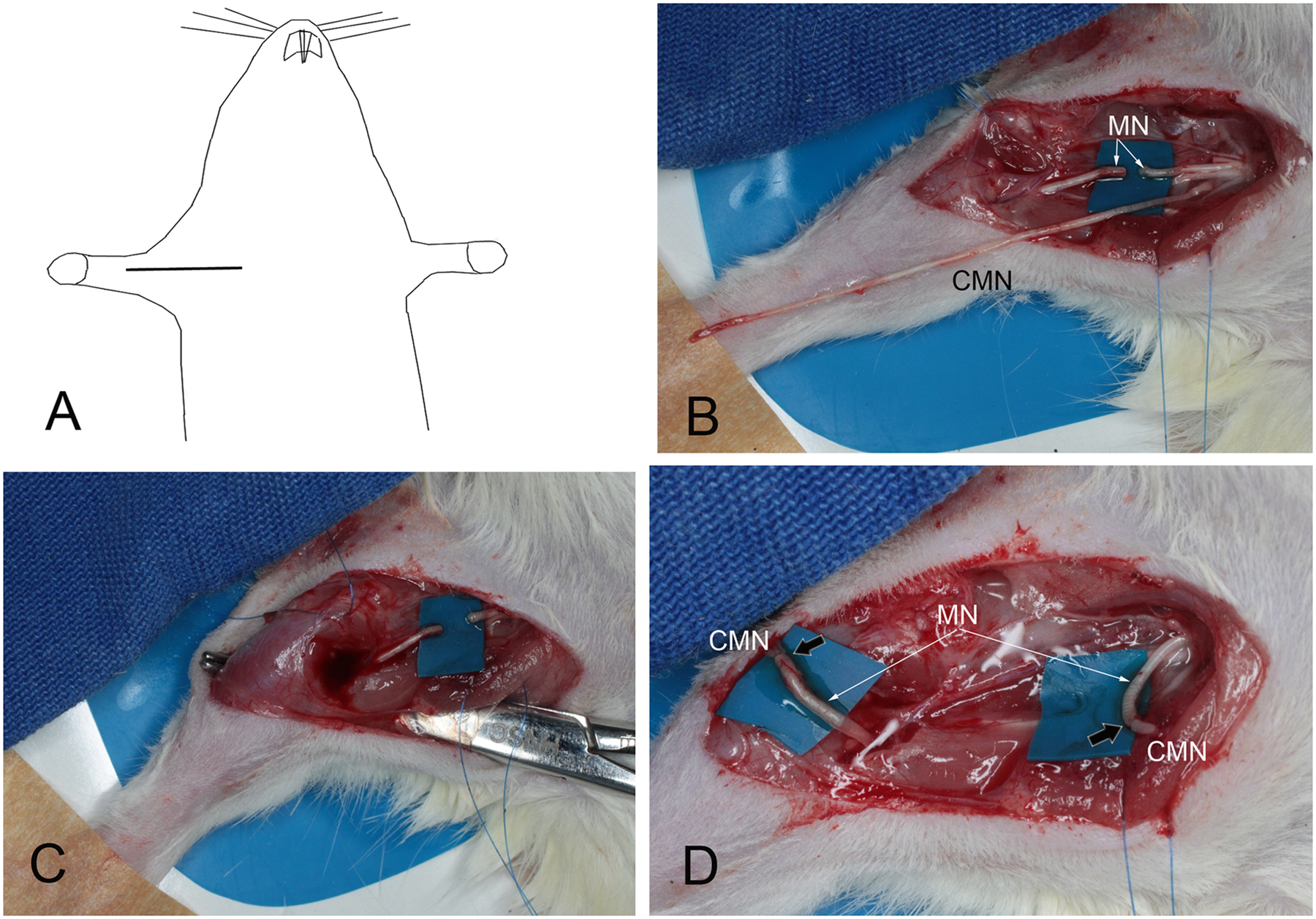
Intraoperative view of the interpositional grafting of the right median nerve (MN) with the entire 40-mm long contralateral median nerve (CMN). In A, we depict the skin incision. In C, we used a forceps to pass the CMN graft posteriorly to the humerus. In D, black arrows indicate nerve coaptation sites.
The 40 grafted rats were subsequently equally divided into 2 groups: 1 group that received nandrolone and 1 group that received groundnut oil, which was the vehicle for the nandrolone. Rats in the nandrolone group received nandrolone decanoate 5 mg/kg intramuscularly once weekly until fine control was achieved. The remaining grafted rats did not receive nandrolone and were treated with weekly intramuscular injection of the oil vehicle. A third group of rats (n = 20) had their left forepaw denervated by removing the ulnar and median nerves, but their right forepaw was left intact because the right paw was the limb tested in all 3 groups; they were thereby used as normal controls.
Surgery
All surgery was performed under general anesthesia with a 7% solution of chloral hydrate (0.6 mL/100 g body weight injected intraperitoneally). The right median nerve was approached through a longitudinal incision running from the anterior aspect of the elbow joint to the upper nipple. The pectoralis major muscle was partially disinserted, leaving the cephalic vein intact. The median and ulnar nerves were dissected and sectioned in the axillary region. The proximal stump was turned back proximally, whereas the distal stump was flipped distally. A 40-mm long median nerve graft was removed from the contralateral upper limb. In the recipient limb, the graft was passed subcutaneously along the posterior region of the arm and forearm. Nerve coaptation was performed using 10-0 mononylon sutures (Figures 1 and 2).
Behavioral Assessment
After grafting, rats were assessed daily by means of the grasp test. 9 For this, they were lifted gently up by their tail but were allowed to grasp the grid of the cage so that their finger flexion movements could be observed. When the first signs of active finger flexion were noted, the day of recovery onset was determined. Grasp strength was assessed at 90 (n = 20 per group) and 180 (n = 9 per group) days after surgery. For this assessment, a wire grid measuring 8 × 14 cm (wire diameter = 1.5 mm) was connected to an ordinary electronic balance by means of 4 sticks, each 15 cm long (Figure 3). A 500-g weight was placed on the balance plate. Rats were allowed to grasp the grid while being lifted by the tail with increasing firmness until they loosened their grip. The value registered by the balance at the precise moment of loosening was noted and the grasp strength calculated (500 g minus the value recorded). To avoid false measurements caused by wrist flexion, the only acceptable scenario was 4 digits grasping the center of the grid.
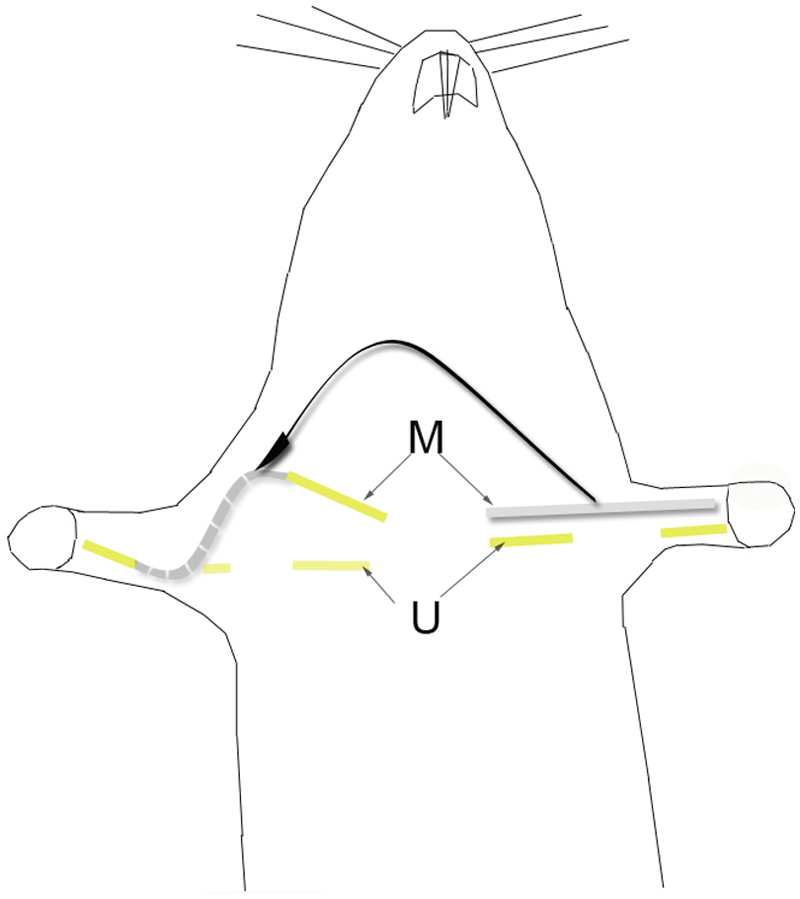
Schematic representation of the surgical paradigm for testing the effects of nandrolone decanoate in a model of deficient reinnervation in a rat median nerve: a 40-mm segment of the left median nerve was removed and interposed between a sectioned right median nerve. For proper accommodation, the graft was passed along the subcutaneous region of the posterior side of the arm and forearm. A 10-mm segment of the ulnar nerve (U) was removed bilaterally.
Nociceptive recovery also was assessed weekly during 90 days after surgery (n = 20 per group). Thereafter, a final evaluation was performed 180 days (n = 9 per group) after surgery. For the assessment of nociceptive recovery, in a quiet room, rats were placed individually over an elevated wire mesh floor and confined underneath individually overturned plastic boxes (4 × 5 cm2). After 1 hour, for rat accommodation, we applied an electrical stimulus using a thin, blunt, coaxial electrode to the palm of the forepaw connected to a nerve stimulator (Cidepe, Sao Paulo, Brazil). Stimulus intensity ranged from 1 to 39 V, with current intensity set to 25 mA. We considered a response positive when the rats removed their forepaw within 3 s of stimulation. With the electrode in contact with the forepaw, we began the test with a low stimulus and then increased it gradually and rapidly to maximal stimulus. If the rat did not remove its forepaw after 3 s of maximal stimulation but its hind paws did respond, sensory recovery was deemed to be absent.
Muscle Twitch Strength Recovery
Muscle strength was assessed 90 (n = 10 per group, half of the rats) and 180 (n = 9 per group) days after surgery. After deep anesthesia, the grafted right median nerve was located, and the median nerve proximal to the graft was sectioned and placed over a latex background. The tendons of the flexor digitorum superficialis and profundus were sectioned and attached to a gauge (PowerLab, Adinstruments, Australia) using mononylon 4-0 sutures. The gauge was connected to a data acquisition system (PowerLab, Adinstruments, Australia). Prior to any muscle force measurement, we calibrated the data acquisition system by applying a 100-mg weight to the gauge. Hence we set our scale for muscle strength measurements in milligrams. A nerve stimulator (Cidepe, Sao Paulo, Brazil) delivered a supramaximal 2-V stimulus to the median nerve proximal to the graft. We confirmed that the 2-V stimulus was supramaximal because increasing stimulus intensity to 3 V did not improve muscle strength. Also, when we used 3 V, the electrical stimulus spread to adjacent nerves and muscles, which complicated the isolated assessment of finger flexion strength. The tension of the muscle was adjusted to produce maximal isometric twitch tension after median nerve stimulation. For this, from a fully relaxed position, we stretched the muscle to its maximal excursion and then stimulated the median nerve and measured muscle strength. This was repeated at 75%, 50%, and 25% of total muscle excursion. There was a resting period of at least 3 minutes between each application of stimulus to avoid fatigue. The highest value of muscle force was considered for statistical analysis.
Muscle Weight Recovery
Muscle weight was assessed 90 (n = 10 per group) and 180 (n = 9 per group) days after surgery. The flexor digitorum profundus and superficialis were removed en bloc and then weighed, again en bloc, using an electronic balance.
Statistical Analysis
All values are reported as means ± SD. Group means were compared using analysis of variance (ANOVA). Where appropriate, Tukey test was used post hoc to identify differences between paired treatment arms. P <.05 was set as the threshold for statistical significance. For the calculation of the percentage of recovery, in grafted treated versus nontreated rats, we attributed 100% to the higher mean value of the day of recovery for grasping strength, twitch strength, muscle weight, and sensory thresholds evaluation. We then calculated the percentage of the lower mean value by multiplying it by 100 and dividing it by the higher mean value. Then, to obtain the percentage difference in recovery, we subtracted the percentage obtained for the lower value from 100% to generate an estimate of effect size.
Results
Over the postoperative period, 1 rat in each of the 3 groups died, leaving 19 rats per group. None of the rats exhibited finger contractures or autotomies. The mean body weight of the rats is shown in Table 1. At 6 months, rats receiving nandrolone were roughly 15% heavier than nontreated and normal control rats.
Variations in Mean Body Weight Throughout the Experiment
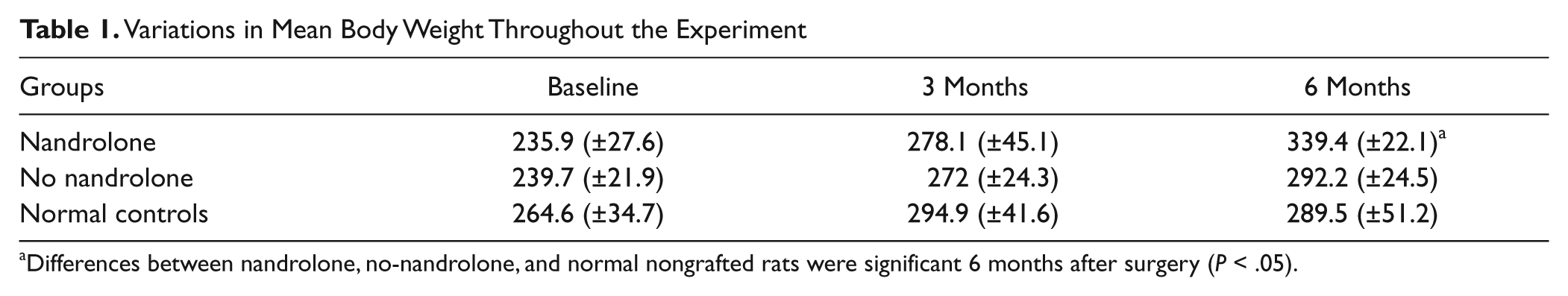
Differences between nandrolone, no-nandrolone, and normal nongrafted rats were significant 6 months after surgery (P < .05).
The average time to finger flexion recovery was 51.1 (SD = ±7.4) and 40.4 (SD = ±5.1) days for the no-nandrolone and nandrolone groups, respectively. Rats in the nandrolone group started recovery earlier (P < .0001). The speed of recovery was increased by 20%. Significant intergroup differences in grasp strength were observed 90 and 180 days after surgery (P < .05). By 90 days, treated rats had recovered 42% of their normal grasp strength, versus only 11% in the no-nandrolone group. At 180 days after surgery, the average values for grasp strength recovery were 40% and 33% of normal for the nandrolone and no-nandrolone groups, respectively (Table 2).
Grasp Recovery
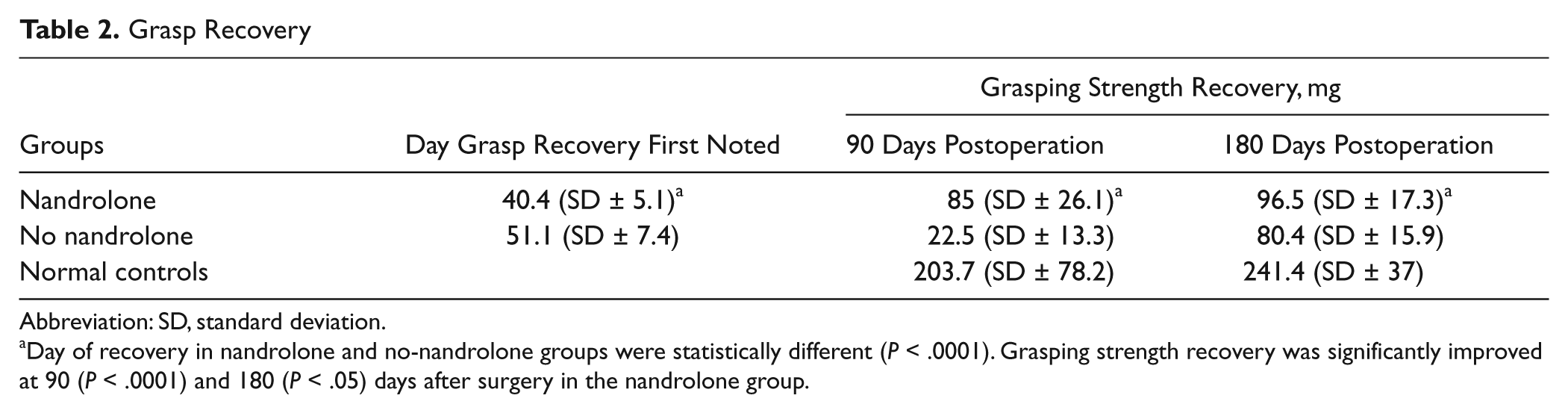
Abbreviation: SD, standard deviation.
Day of recovery in nandrolone and no-nandrolone groups were statistically different (P < .0001). Grasping strength recovery was significantly improved at 90 (P < .0001) and 180 (P < .05) days after surgery in the nandrolone group.
Thresholds for nociception were not detectable in either grafted group 90 days after nerve grafting. At 180 days, rats removed their forepaws from the electrical stimulus at average thresholds of 13.5 V (SD = ±8.6) and 23.5 V (SD = ±11.2) in the nandrolone and no-nandrolone groups, respectively. Differences in recovery were significant (P < .01), achieving 43% improvement in the nandrolone group. Controls removed their forepaws at a significantly (P < .001) lower stimulus intensity, averaging 1.5 V (SD = ±0.7) (Table 3). During cutaneous electrical stimulation, in both grafted and control rats, when nociceptive thresholds were reached, paw withdrawal was immediate.
Nociceptive Thresholds Recovery
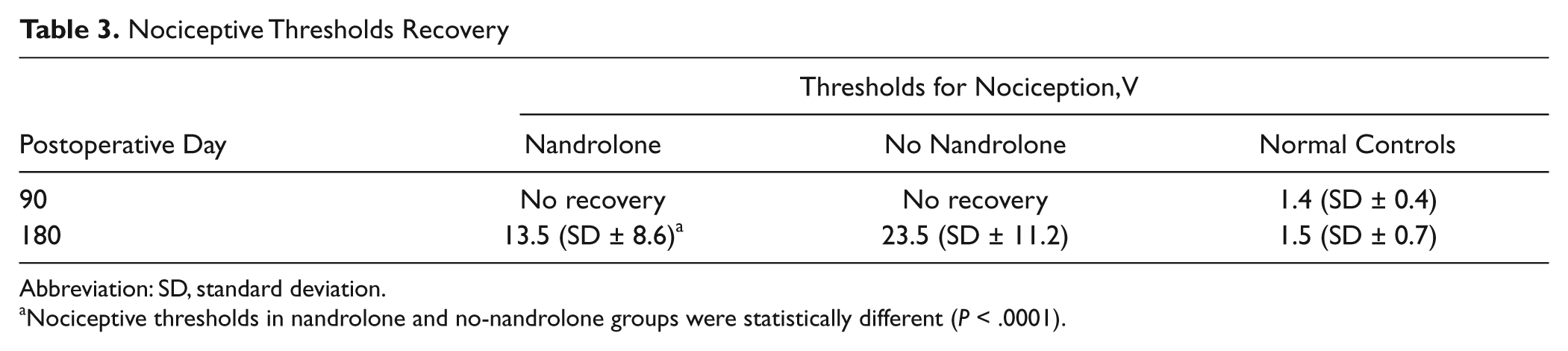
Abbreviation: SD, standard deviation.
Nociceptive thresholds in nandrolone and no-nandrolone groups were statistically different (P < .0001).
Beyond electrical stimulation and muscle strength measurements, we observed differences in finger flexion strength in the grafted nandrolone and no-nandrolone groups only at final evaluation. There was an incremental increase in strength of 16% in treated versus nontreated rats. We noticed increased strength in nandrolone-treated rats versus normal controls. However, differences were not statistically significant (P > .05). Results of muscle strength recordings following electrical stimulation are summarized in Table 4.
Electrophysiological Evaluation of Muscle Strength
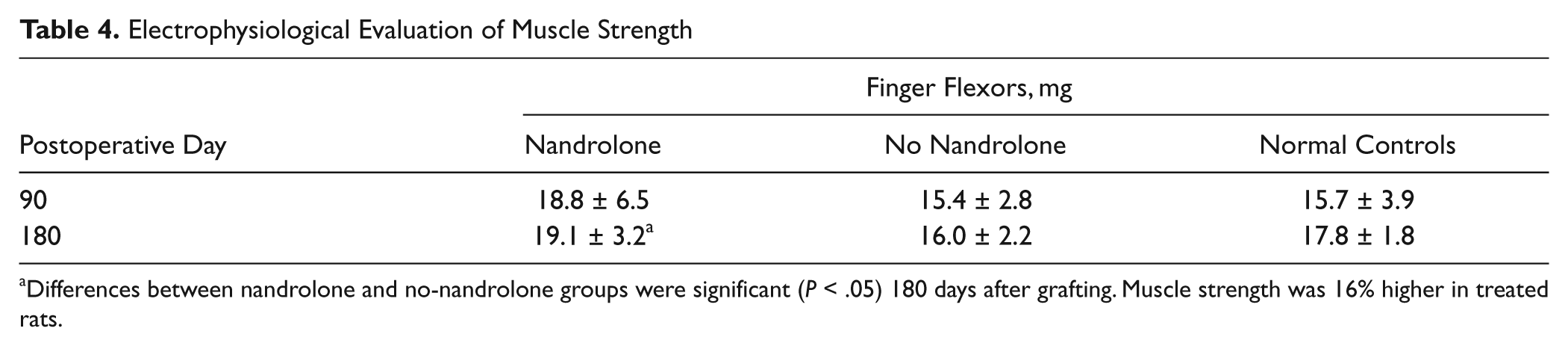
Differences between nandrolone and no-nandrolone groups were significant (P < .05) 180 days after grafting. Muscle strength was 16% higher in treated rats.
The weights of the finger flexor muscles at the different time points are shown in Table 5. No significant differences in weight recovery of the finger flexor muscles were observed in the grafted rats. At 90 days after surgery, the weight of the finger flexors was significantly lower in grafted rats than in normal controls. However, this difference disappeared at final evaluation.
Muscle Weight Recovery a
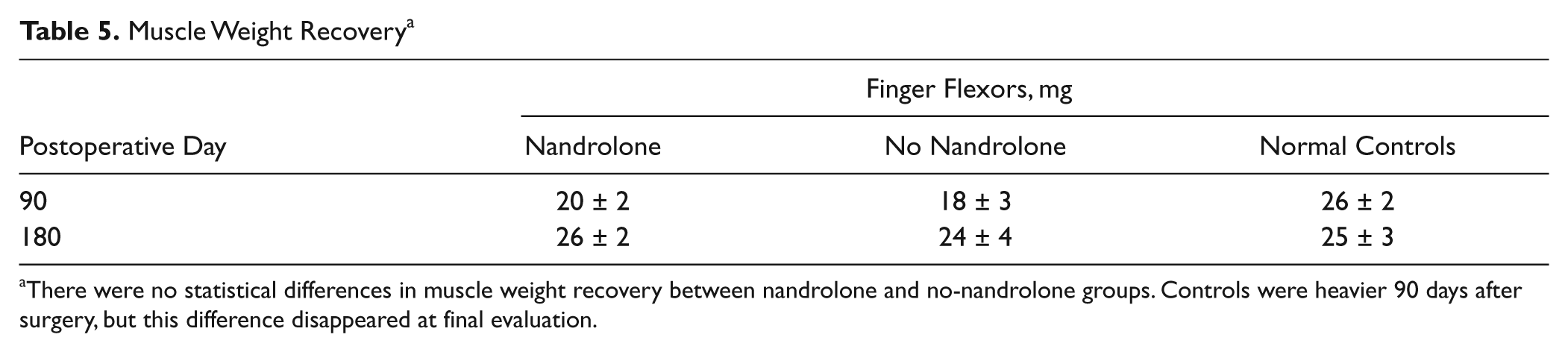
There were no statistical differences in muscle weight recovery between nandrolone and no-nandrolone groups. Controls were heavier 90 days after surgery, but this difference disappeared at final evaluation.
Discussion
In our model of deficient reinnervation, we found that functional recovery was ameliorated by nandrolone. In terms of motor response, we observed a 20% faster onset of recovery and an incremental increase in grasp strength recovery by 31% and 7%, 90 and 180 days postsurgery. The reduction in this difference of recovery from 31% to 7%, at 90 and 180 days, might have resulted from earlier reinnervation and, consequently, earlier improvement in strength in the nandrolone group. This suggests that nandrolone action would be more important for accelerating recovery rather than for ameliorating end results. At final evaluation, electrophysiological measurement demonstrated 16% augmented force in the finger flexors in treated rats. At earlier evaluation, 90 days after surgery, there were differences in mean force recovery between treated and nontreated rats. However, differences were not statistically significant, probably because of the small number of rats per group. We observed differences in the recovery of weight for finger flexors in treated and nontreated rats. However, these differences were not statistically significant. Again, this probably results from the small numbers of animals per group. Alternatively, it is possible that muscle weight assessment is not sensitive to subtle changes in grasp strength. In fact, when weighing muscles, not only muscle fibers but also connective tissues are a determinant of weight. This is the reason that even after prolonged periods of denervation, muscle weight does not decrease below 30% of normal weight. 16
Enhanced recovery in our treated rats could have resulted from nandrolone effects on muscle, nerve, spinal cord, and/or brain. One potential effect improving muscle contraction could have been the induction of satellite cell proliferation 5 and actions on Ca2+ uptake by the sarcoplasmic reticulum pump. 17 In addition, nandrolone could have slowed denervation-related muscle atrophy. The critical period of nandrolone’s effects on muscle denervation-atrophy has been demonstrated to be between 7 and 35 days postdenervation. 18 This might be behind the enhanced muscle performance we observed in grafted and treated animals, in which nandrolone might have protected denervated muscles from atrophy provided that in our experimental model, reinnervation was largely delayed (eg, 40 days). In rats, muscle atrophy has been demonstrated as early as 30 days after denervation. 19 Zhao et al 20 observed that nandrolone has no effect on muscle atrophy if administered on the day of denervation but witnessed protective effects if it was administered 28 days after denervation. Isaacs et al 12 observed that after 3 months of a first surgery to divide the sciatic nerve to produce muscle denervation, a second surgery to graft the previously divided sciatic nerve associated with nandrolone administration, which was started 30 days after repair and continued for an additional 30 days, improved muscle contraction strength but not muscle weight when measured 6 weeks later. In our study, nandrolone was administered 7 days after denervation. The ideal time for nandrolone administration remains undetermined.
A second explanation is that because we observed the onset of functional recovery sooner, in agreement with previous studies by Tanzer and Jones, 21 it is possible that nandrolone also influenced the speed of axonal regrowth through the nerve grafts. This could have been mediated by augmenting circulating neurotrophic factors associated with anabolic steroid administration. For instance, testosterone has been shown to increase the concentration of circulating insulin-like growth factor (IGF) and brain-derived neurotrophic factor (BNDF). 22 IGF and BNDF have known positive effects of accelerating axonal growth. 23
Third, the death of neurons of the spinal cord submitted to axonotomy might be protected by BNDF, 24 which is upregulated in spinal motoneurons following the administration of androgens, as discussed by Verhovshek et al. 22
Fourth, certain effects of androgens on muscle performance might be attributable to their psychological effects, such as drive and mood enhancement and aggressive behavior. This being said, some investigators believe that mood reinforcement is merely a consequence of enhanced muscle performance. 25
Nociceptive threshold measurements have been extensively used in neuroscience and medical research based on the principle that pain intensity can be measured after the application of noxious electrical, thermal, mechanical, or chemical stimuli.26,27 Previously, in the same model of long interpositional nerve grafts employed herein, after application of noxious mechanical stimulus, we observed not only delayed recovery of nociception, around 90 days after grafting, but also increased nociceptive thresholds even 360 days after surgery. 15 When testing nociception, in order to ascertain the absence of recovery, we applied maximal electrical stimulation to the forepaw over a 3-s period. We prolonged the electrical stimulation for 3 s because, in our clinical research, we have observed that patients with long nerve grafts exhibit a delay between painful stimulus application and pain sensation. 28 We have estimated this time to be roughly 3 s. If a shorter-interval stimulus had been applied, a positive response could have been missed.
We could not determine the day of nociceptive recovery because it occurred beyond the 90-day period of our weekly assessment. Hence we could not detect if nandrolone also accelerated sensory recovery. However, at final evaluation, we observed that nandrolone improved sensory recovery by 43%, which has not been reported before. Contrary to our findings, Swallow et al 29 observed no significant effects of testosterone on nociceptive recovery after skin pinch and warm water withdrawal testing in a crush model of sciatic nerve injury. There are several potential explanations for the discrepancy between their results and ours. First, they used testosterone, not nandrolone. Second, the crush model has inherent limitations in terms of demonstrating differences between groups because full recovery is the usual, ultimate outcome after a crush injury (see Introduction). Third, in rats, the saphenous nerve, which was preserved in their experiments, provides sensation to the leg and hind paw; this contributes to the misinterpretation of sensory recovery after a sciatic nerve injury. 30 Finally, the methods they used to quantify nociceptive recovery may have been inadequate, in that they did not measure thresholds. On the other hand, we tested nociception by applying an electrical stimulus to the denervated zone, and electricity produces a thermal injury. 31 This method allowed us to precisely test the zone of reinnervation. At 90 days after surgery, nociception was not detected by our method, probably because of the low amplitudes of electrical stimuli we applied; we elected not to use more intense stimuli, to prevent burns to the rats’ forepaws.
A longer interval was required for sensory recovery than for motor recovery. This probably results from the longer distance between the repair site and target end organs. For the sensory system, cutaneous nociceptors are located on the palm of the forepaw versus the motor end plates of finger flexors, which are located in the proximal forearm. However, motor and sensory reinnervation delays were both much longer than with a crush injury to the median nerve in the arm, which generally recovers within 8 days of injury. 14 Of interest, in the model we used, the entire left median nerve was interposed between the cut stumps of the right median nerve. Hence, for reinnervation, axons should have travelled an increased distance from the proximal stump to the targets. For instance, after division of the median nerve in the axilla, the distance between the proximal stump and motor end plates of finger flexors is approximately 20 mm. After our interpositional 40-mm-long nerve graft, this distance is 60 mm. If we consider forepaw sensory recovery, the ultimate distance for target reinnervation would be roughly 80 mm. In fact, after grafting and successful regeneration, axons doubled their original length. Alternatively, palmar sensory recovery could have originated from collateral reinnervation via sprouting of radial nerve branches. However, this explanation seems unlikely because in denervated forepaws, sensory recovery was demonstrated neither here nor elsewhere. 15
We used 5 mg/kg of nandrolone administered weekly, without interruption. The dosage used in the literature varies from 3.7 to 20 mg/kg. 12 Clearly, the ideal dosage of nandrolone for promoting nerve regeneration still must be determined. In general, the duration of treatment with nandrolone varies from days to weeks.12,17,32 To date, no long-term study has been reported. We studied rats for 6 months and administered nandrolone over this period because nerve regeneration is an ongoing phenomenon that progresses over time. Indeed, 6 months after a freezing injury to the sciatic nerve, regenerating fiber diameters reach only 50% of normal; and 2 years after freezing, fiber diameter still is only 80% of normal. 33 The ideal duration of nandrolone treatment after nerve repair warrants further study.
In summary, with the current study, we have demonstrated that nandrolone administered during the process of nerve regeneration improves functional recovery within both the motor and sensory systems in a model of deficient reinnervation, and this has clinical relevance. Dosage, time of initiation, and duration of treatment with nandrolone are important issues that remain to be evaluated in future studies.
Footnotes
Acknowledgements
We would like to thank Evandro da Cruz Cittadin Soares, Anatomy Laboratory, University of Southern Santa Catarina, for his technical support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
