Abstract
Background. Episodic memory and semantic abilities deteriorate early in Alzheimer disease (AD). Since the cognitive system includes interconnected and reciprocally influenced neuronal networks, the authors hypothesized that stimulation of lexical–semantic abilities may benefit semantically structured episodic memory. Objective. To investigate the effects of lexical–semantic stimulation (LSS) on verbal communication and episodic memory in early AD. Methods. Forty AD participants were randomized to LSS or unstructured cognitive stimulation (UCS) as control condition. Treatments lasted 3 months, 2 sections a week. The primary outcome measures were the Mini-Mental State Examination (MMSE), Boston Naming Test (BNT), Verbal Naming Test (VNT), Phonemic and Semantic Fluency, Story Recall, and Rey Auditory Verbal Learning (RAVL). Secondary outcome measures were neuropsychological tests assessing cognitive functions not stimulated by the intervention, such as attention, executive functions, and visual–spatial abilities, and the instrumental activities of daily living scale. A 6-month follow-up assessment was administered to the LSS group. Results. LSS treatment yielded significant improvements of the MMSE, BNT, VNT, Brief Story Recall, and RAVL delayed recall mean scores. Among secondary outcome measures, only working memory and the speed of a task assessing executive functions (Stroop test) improved after LSS. Unstructured cognitive stimulation intervention did not improve any cognitive domain. Six months after LSS discontinuation, the MMSE mean score remains significantly higher than the baseline value. Conclusion. LSS treatment may improve episodic memory in AD patients and may be regarded as a clinical option to counteract the cognitive decline typical of the disease.
Keywords
Introduction
Episodic memory is the most impaired cognitive function in the early stage of sporadic late-onset Alzheimer disease (AD), representing the main distressing problem experienced by patients. Language disturbances in early AD are also frequently observed. In contrast to what happens in aphasia due to focal lesions where the difficulty mainly lies in accessing lexical–semantic information, the main problem in AD is the deterioration of semantic memory, in both general knowledge and lexical components. 1 Deficits of semantic abilities are common in mild AD and may exist prior to the clinical diagnosis. A decline of semantic memory accelerates many years before the diagnosis of AD. 2 A neuroanatomic correlate of the semantic memory impairment is mesial temporal lobe atrophy. 3 Subregions of the hippocampus are engaged in both episodic and semantic memory processes. 3 Thus, episodic and semantic memory networks seem to share temporal and regional patterns of alterations.
Given the mild efficacy of drugs approved for AD, 4 nonpharmacological interventions, such as cognitive rehabilitation, are emerging as a potential approach to improve or stabilize cognitive functions.5-7 However, the clinical impact on memory of both generalized cognitive stimulation and specific memory training are not conclusively proven,5-7 making the use of these strategies not yet widely applied.
The brain can be viewed as a plastic “computational machine” consisting of different functional systems, hierarchically organized in a highly overlapping fashion. 8 For example, one specific cognitive function, such as episodic memory, may be the final result of the contribution of several interconnected and reciprocally influenced neural networks, encoding different cognitive competencies. Therefore, our working hypothesis was that interventions oriented at stimulating lexical–semantic abilities and strengthening semantic representations may produce an improvement of verbal communication and semantic memory, which could reshape other verbal-related memory networks, thus stimulating impaired brain capacities.
Within this theoretical framework, we designed an observer-blinded randomized controlled pilot study comparing the effects on memory of a focused lexical–semantic stimulation (LSS) intervention with those of an unstructured cognitive stimulation (UCS) treatment in participants with early AD.
Materials and Methods
Participants
A total of 213 participants with mild memory decline, referred to the Memory Unit of the Department of Neurosciences at Padova University, were screened from July 2007 to December 2008. They were evaluated for their eligibility by the neurologists and neuropsychologists of the Neurosciences Department in Padova and the Rehabilitation Hospital San Camillo in Venice. The inclusion criteria were diagnosis of probable AD according to National Institute of Neurological and Communicative Disorders and Stroke–AD and Related Disorders Association criteria 9 ; early stage of the disease, that is, Clinical Dementia Rating 10 score ranging from 0.5 to 1; and stable psychoactive medication in the previous 3 months. In addition, participants were not on antidementia drug therapy (cholinesterase inhibitors or memantine) for the following reasons: presence of contraindications related to heart arrhythmia, lack of compliance, and Mini-Mental State Examination (MMSE) 11 score cutoff >26 out of 30 (a score below this threshold is required by the Italian Public Health regulatory system for eligibility for prescription of antidementia drugs). The exclusion criteria were presence of brain lesions with computed tomography/magnetic resonance imaging studies, severe systemic diseases, severe depression assessed by means of the Center For Epidemiologic Studies–Depression Scale (CES-D) 12 score (range = 0-60), disturbing behavioral symptoms evaluated with the Neuropsychiatric Inventory (NPI) 13 questionnaire (range of score = 0-144), and severe lexical–semantic deficits that may interfere with the rehabilitation therapy, established with the verbal naming test (VNT) 14 score ≥2 standard deviations below normal values.
Forty participants, meeting the selection criteria, entered the study. By using a simple computerized randomization technique, they were divided into 2 treatment groups, 20 participants each, receiving LSS or UCS treatments. The study was approved by the local ethical committee and supported by a grant from the Italian Ministry of Health. Informed consents were obtained from the participants and their closest relatives.
Interventions
The LSS treatment consisted of focused lexical–semantic rehabilitation exercises, modified from those used by Visch-Brink and colleagues, 15 who first applied this technique in poststroke aphasic patients. The LSS protocol contains a wide range of lexical tasks aimed at enhancing semantic verbal processing. The exercises are focused on the interpretation of written words, sentences, and stories and are divided into 8 main parts: semantic categories, syntagmatic and paradigmatic relationships, level of semantic affinity between words, adequacy of adjectives to the context of the text, part–whole relationship, recognition of non-sense sentences, identification of semantic definition and context of a short story. We report here a brief description of the type of exercises for each part of the LSS.
Semantic categories: A number of semantically related words are given to the participant, together with 1 word that belongs to a different semantic category. The participant is required to choose the semantically unrelated word.
Syntagmatic and paradigmatic relationships: The participant is asked to match words or sentences respecting syntagmatic or paradigmatic relationships.
Semantic classification: A list of words needs to be paired with 1 of 2 predefined semantic categories.
Sentences with correct/wrong adjectives and exclamations: The participant is asked to choose, among alternative adjectives and exclamations, the one that is semantically related with a target sentence.
Part–whole relationship: A target sentence gives an example of a part–whole relationship between 2 words. The participant needs to choose among alternative words which one has the same relationship with one of the target words.
Anomalous sentences: Sentences are presented as either semantically correct or incorrect and a yes/no decision about correct meaning is requested.
Semantic definition: A target word is given that can also be used in compound words. The participant is asked to decide whether the combination makes an appropriate or non-appropriate compound.
Semantic context: Identify semantic anomalies in text. Exercises are structured in multiple choice or right/wrong format, with different degrees of difficulty.
The difficulty level was determined by factors such as the number of distracters, the strength of the semantic relation, the frequency, and abstractness/ambiguity of the word. The participants were asked to choose a written response from alternatives or to determine whether a response was accurate or inappropriate. Group discussion about response choices was encouraged to stimulate verbal competencies. Participants of the UCS intervention group were engaged in completely different exercises, consisting of creative work such as practicing manual skills, manufacturing external memory aids, stimulating fantasy and creativeness, reading the newspaper with active participation and discussion on current news, and improving verbal communication.
Throughout the duration of the trial, the same neuropsychologist (NJ) performed the LSS and UCS therapy. The intervention protocol consisted of 2 weekly sessions of LSS or UCS exercises, lasting 1 hour each in the morning, over a period of 3 months. Both treatments were administered to small subgroups of 4 participants.
Outcome Measures
Extensive neuropsychological assessment addressing multiple cognitive domains was given to each subject at study entry and postintervention, after 3 months of treatments. In addition, the LSS therapy group underwent a follow-up evaluation, 6 months after treatment discontinuation. All these assessments were carried out by an experienced neuropsychologist (EM), blinded to the treatment group to which each patient was allocated.
Primary outcome measures were (a) global cognitive function, assessed with MMSE; (b) lexical–semantic abilities, assessed with Boston Naming Test (BNT), 16 Verbal Naming Test (VNT), 14 and Phonemic and Semantic Fluency14,17; and (c) episodic verbal memory, assessed with Brief Story Recall 18 and Rey Auditory Verbal Learning (RAVL) 19 tests. Secondary outcome measures were (a) working memory, assessed with the Forward Digit Span 20 test; (b) visual–spatial memory, assessed with the Rey–Osterrieth Complex Figure (ROCF) 21 delayed recall test; (c) attention and executive functions, assessed with the Stroop test, 22 Attentive Matrices,17,23 and Trail Making Test (TMT A and B)17,24; (d) visual–spatial abilities, evaluated with the ROCF 21 Copy and the Clock Drawing Test (CDT) 25 ; and (e) the instrumental activities of daily living (IADL). 26
Statistical Analysis
The Kolmogorov–Smirnov method was used to test for the normality of variables. The Student t test was used for normally distributed variables, whereas for ordinal variables (ie, test scores), a non-parametric statistic was performed. The Wilcoxon test for paired data was used for within-group difference, whereas the Mann–Whitney U test was performed for between-group difference by computing for every test and for every patient the score difference before and after therapy. For categorical variables the χ2 test, or the Fisher exact test when required, were performed. All P values reported are uncorrected for multiple comparisons. The significance level was set at P < .05.
Bonferroni correction for multiple comparisons was also applied to the tests involving primary outcome measures, with a significance level set at P ≤ .008.
Results
The overall experimental design is depicted in Figure 1.
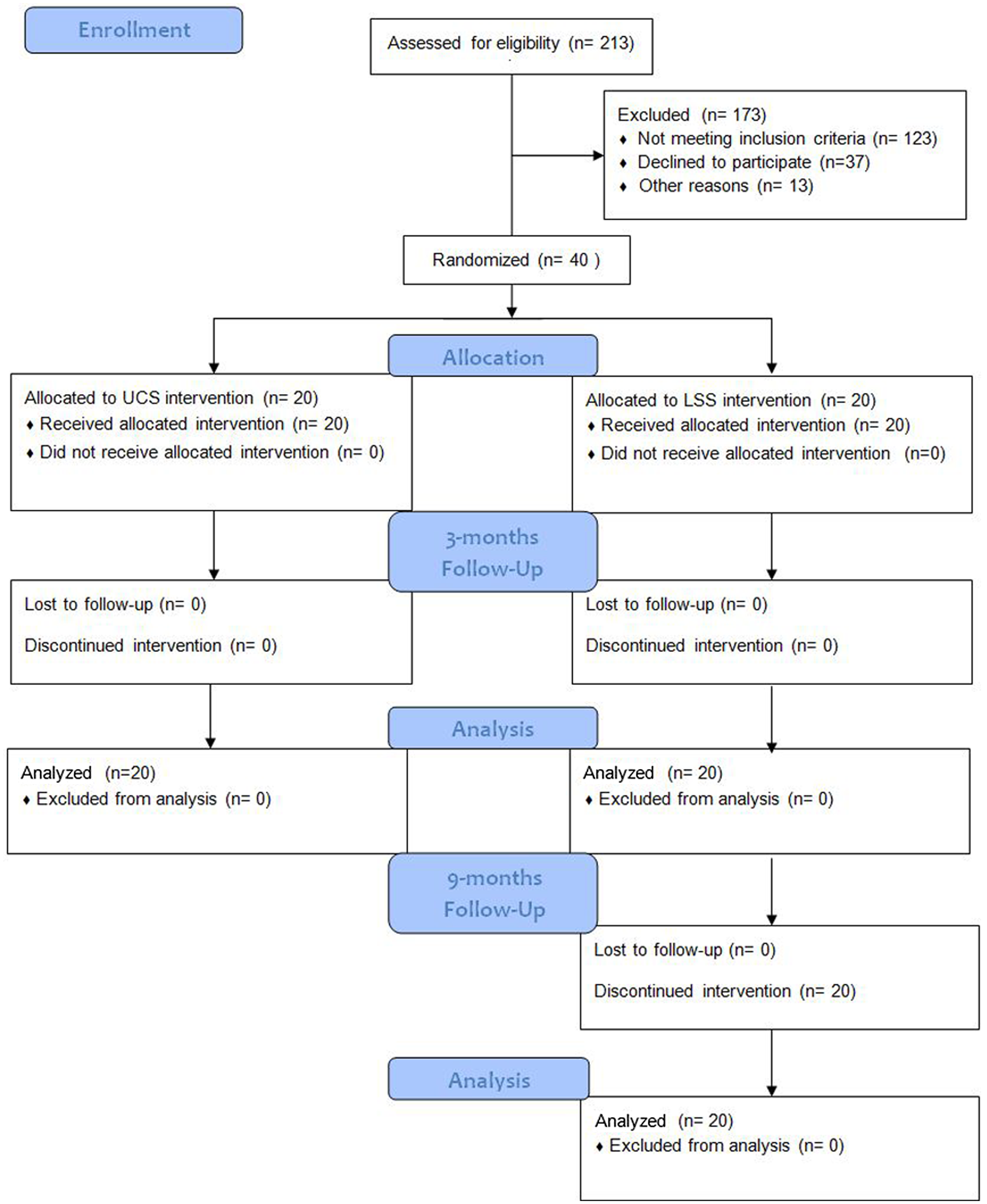
Flow diagram of the trial.
At study entry, the 2 groups were comparable in terms of sex (UCS: M/F [male/female] = 5/15; LSS: M/F = 2/18), mean age (UCS: 81.8 ± 5.5 years; LSS: 82.9 ± 3.6 years), educational level (UCS: 8.25 ± 3.6 years; LSS: 6.7 ± 2.9 years of schooling), and severity of cognitive impairment (UCS: MMSE = 25 ± 2.6; LSS: MMSE = 24.4 ± 2.8). In addition, no significant differences were found between the UCS group and the LSS in NPI mean scores, 4.5 ± 5.0 and 5.1 ± 9.7; CES-D mean scores, 18.4 ± 12 and 17.3 ± 10.3.
As shown in Table 1, mean MMSE scores significantly improved in the LSS group (P < .001), although there was a decrease of about 4% in the UCS group (P < .01), as compared with the baseline mean values. Given this opposite trend, the between-group analysis showed a statistically significant improvement of the MMSE test score in the LSS compared with the UCS group (P < .001). Moreover, each subject in the LSS group showed an improvement of the MMSE score compared with the baseline value. The items of the MMSE test that improved the most in the LSS group were working memory and time orientation.
Effects of Unstructured Cognitive and Lexical–Semantic Stimulation Therapy on Global Cognition, Language, and Verbal-Related Memory Domains
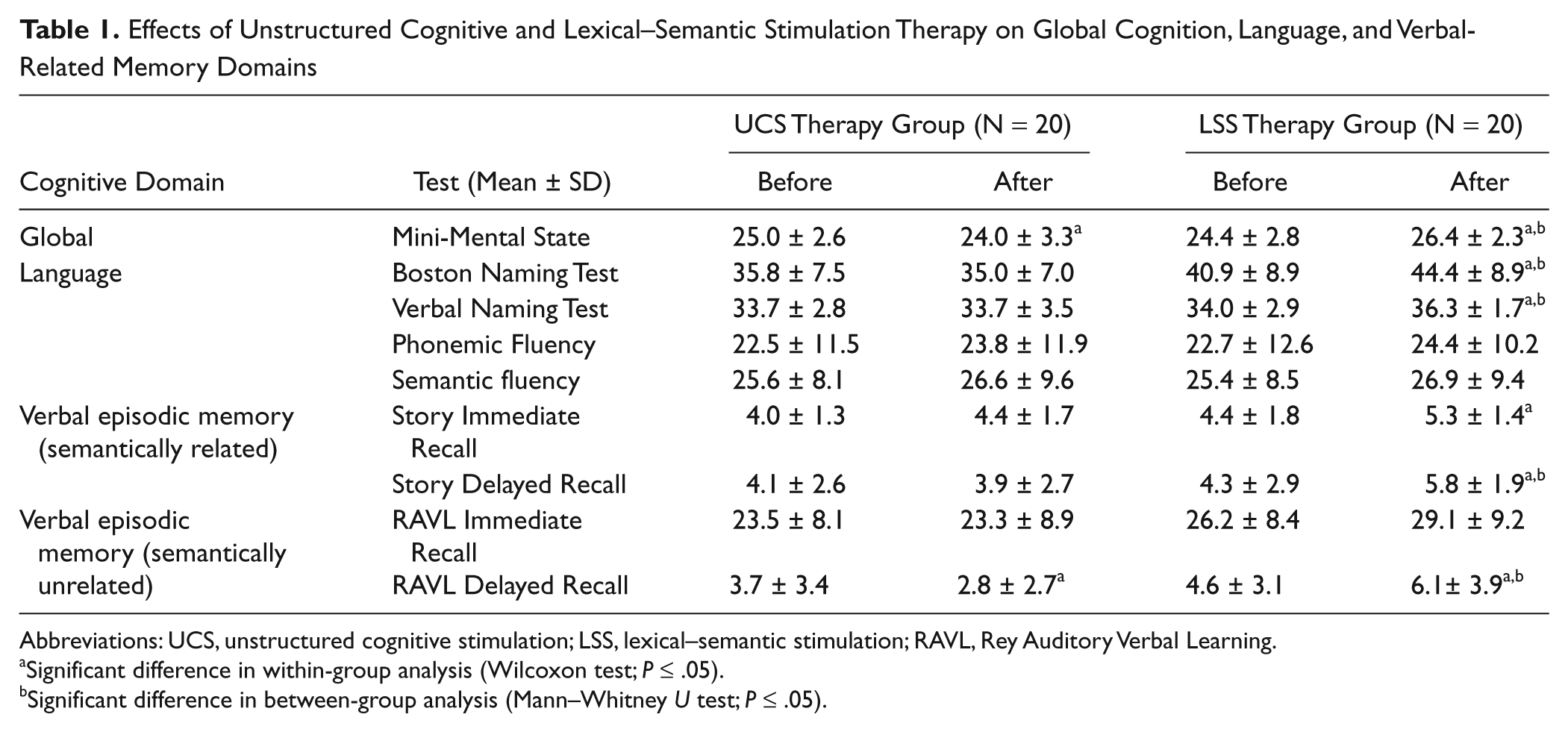
Abbreviations: UCS, unstructured cognitive stimulation; LSS, lexical–semantic stimulation; RAVL, Rey Auditory Verbal Learning.
Significant difference in within-group analysis (Wilcoxon test; P ≤ .05).
Significant difference in between-group analysis (Mann–Whitney U test; P ≤ .05).
Language abilities were affected differently by the treatments. LSS therapy significantly improved BNT (P = .007) and VNT (P = .004) mean scores by 7% to 8%, whereas UCS therapy had no effect on both test scores, leading to a significant between-group difference. Phonemic and semantic fluency test scores improved by 3% to 5% at the end of both therapies, without reaching significant within-group and between-group differences.
Verbal episodic memory significantly changed after LSS treatment with improvements (ranging from 20% to 35%) in the Story Immediate Recall (P = .007) and Story Delayed Recall (P = .008) and in the RAVL delayed recall test mean scores (P = .03). In the UCS group, there was a 24% significant decline in the RAVL delayed recall test (P = .03), without any substantial changes in the other tests exploring verbal episodic memory. Mean values of Story Delayed Recall and RAVL delayed recall tests survived the between-group analysis, in favor of LSS therapy. MMSE, BNT, VNT, and Story Delayed Recall tests remained significant after Bonferroni correction for multiple comparisons.
As described in Table 2, at baseline working memory, assessed with the Forward Digit Span, was similar in the 2 groups. The within-group comparison showed a small (4%) but not significant improvement of working memory after LSS treatment and a significant (8%) decline of the test mean score after UCS therapy. As a consequence, between-group analysis showed a significant improvement of working memory in the LSS group (P = .009). Performance on visual– spatial memory task, as measured with ROCF delayed recall test, was not different in between-group analysis.
Effects of Unstructured Cognitive and Lexical–Semantic Stimulation Therapy on Verbal-Unrelated Memory Domains, Attention and Executive Functions, Visual–Spatial Abilities, and Functional Activities
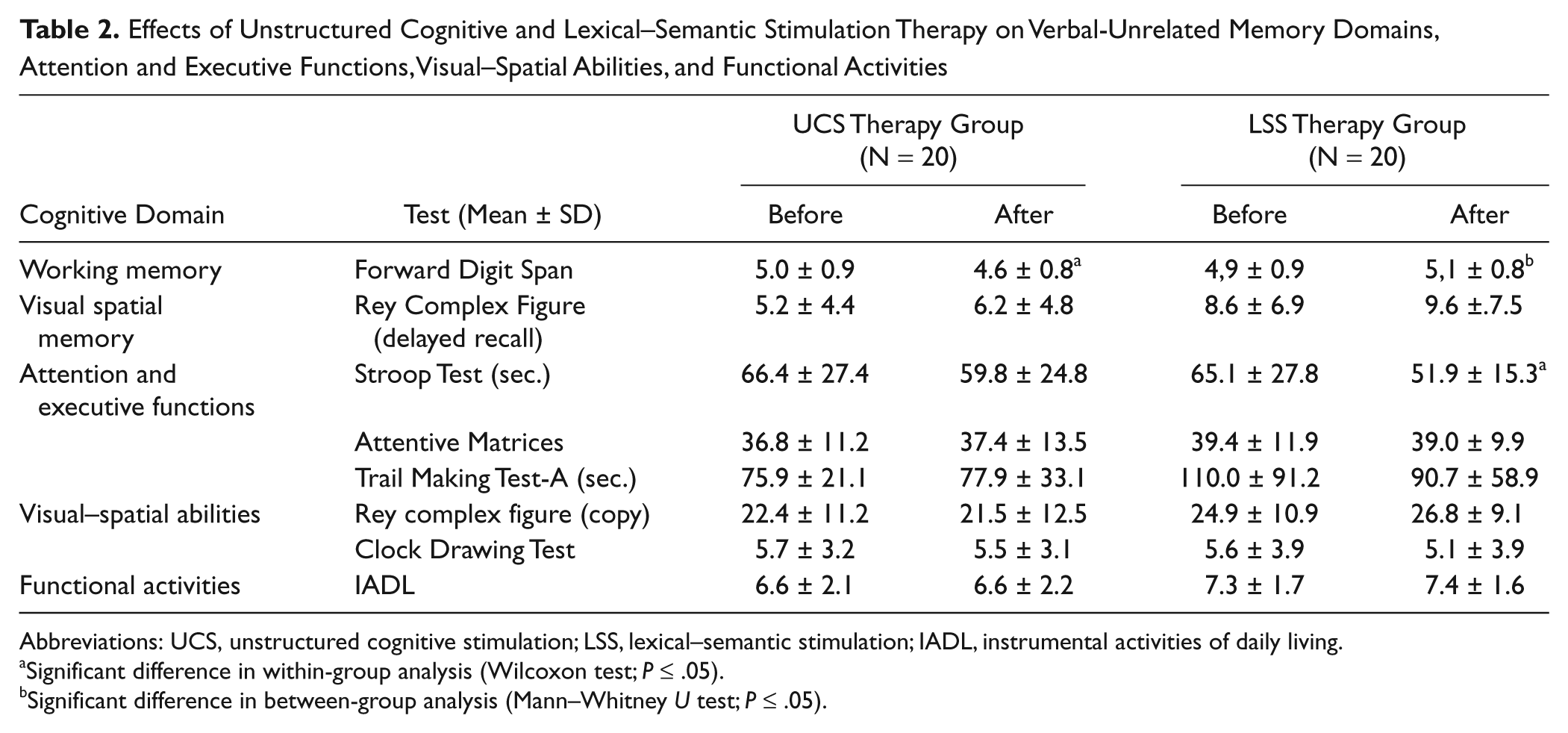
Abbreviations: UCS, unstructured cognitive stimulation; LSS, lexical–semantic stimulation; IADL, instrumental activities of daily living.
Significant difference in within-group analysis (Wilcoxon test; P ≤ .05).
Significant difference in between-group analysis (Mann–Whitney U test; P ≤ .05).
With regard to attention and executive functions, only the Stroop test significantly improved after LSS (P = .004) but not after UCS therapy. At baseline, only 9 participants of the LSS and 4 of the UCS group were able to complete the TMT-B test within the required time, thus in the analysis this variable was not included. No therapy-related significant modifications of visual–spatial abilities (scored by means of ROCF copy and CDT) and IADL were found.
The long-lasting effects of LSS therapy were evaluated comparing the percent changes from baseline mean values with those after 3 months of treatment and 6 months after discontinuation (Figure 2). To this end, we considered only the neuropsychological tests that showed a treatment effect, namely, MMSE, Story Immediate and Delayed Recall, RAVL delayed recall, BNT, VNT, and Stroop tests. Only the MMSE mean score did not decay, remaining significantly improved from the baseline value. On the other hand, in all the other neuropsychological tests that showed a treatment effect after 3 months, the scores declined by 50% to 70% with respect to the values observed after intervention. Notably, the above test mean values remained higher, even though not significantly, than those observed at baseline, particularly in the Story Delayed Recall test exploring semantic-related verbal memory.
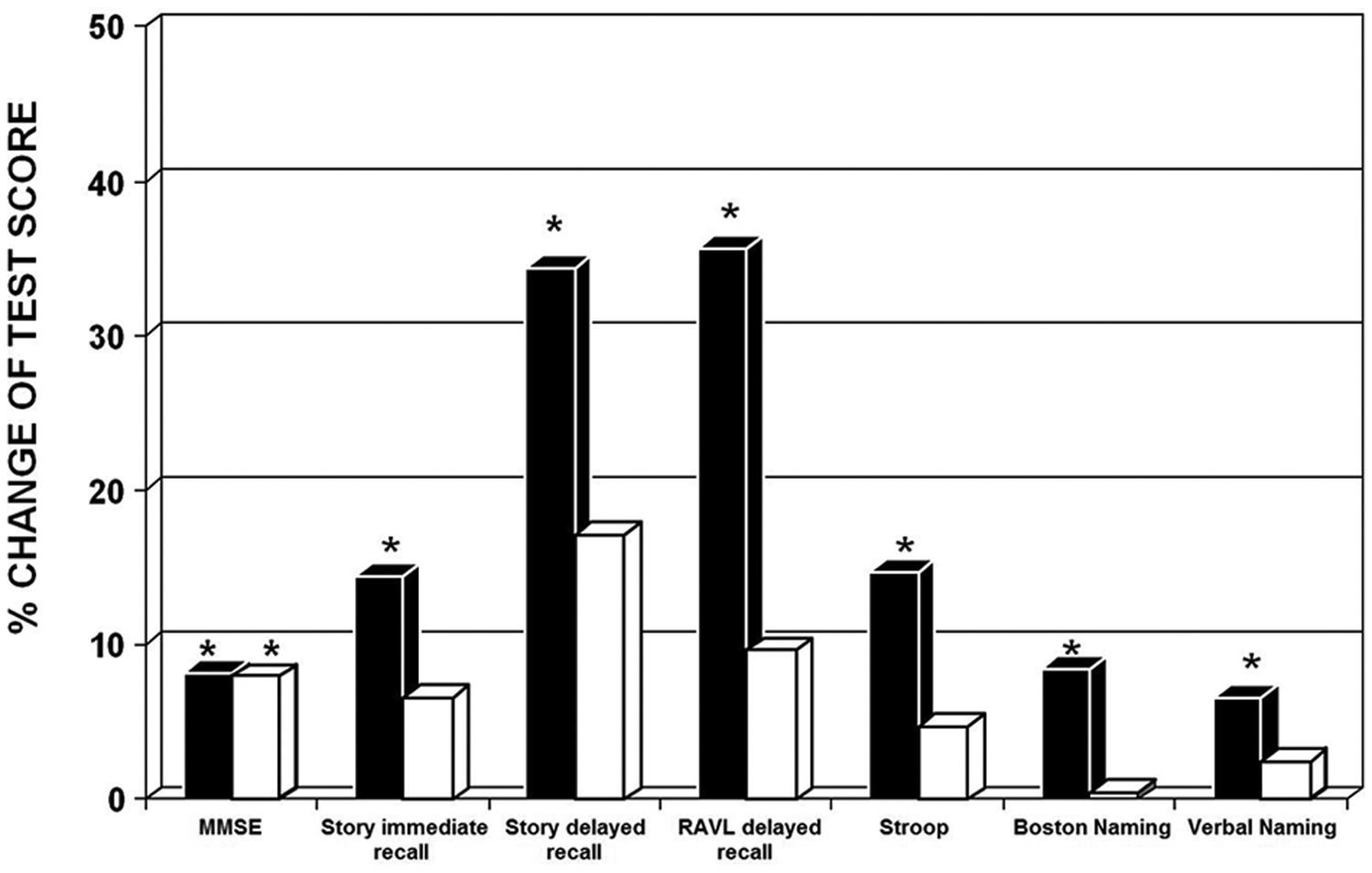
Bar graph shows the percentage of the mean scores improvement from baseline at the end of lexical–semantic stimulation therapy (black box ■) and 6 months after treatment discontinuation (white box □) of the following tests: Mini-Mental State Examination (MMSE), Story Immediate Recall and Delayed Recall, Rey Auditory Verbal Learning (RAVL) delayed recall, Stroop, Boston Naming, and verbal Naming. *Significantly different from baseline values (χ2 test, P ≤ .05).
Discussion
This study assessed the effects of LSS against UCS intervention on episodic memory and other cognitive abilities in 2 well-balanced groups of subjects in the early stage of AD. At the end of 3 months of treatment, within-group and between-group comparisons showed that stimulation of lexical–semantic abilities improved global cognitive performance, lexical–semantic abilities, and verbal episodic memory. LSS effects did not yield a clinical meaningful improvement of attention and executive functions. In fact, among attentional tasks, only working memory improved, whereas performance of executive functions was faster but not more accurate.
The lack of a substantial improvement of attentional/executive functions (a) confirms the validity of the intervention protocol focused at stimulating a specific cognitive domain and (b) excludes that improvements of episodic memory is caused by an increased attentional component. Finally, the effects of LSS treatment did not benefit nonverbal-related memory and visual–spatial abilities, probably because of a less tight connection with the stimulated lexical–semantic network. We cannot exclude, however, that LSS treatment could have induced a more widespread cognitive improvement if administered more frequently or for a longer period. On the other hand, UCS-treated participants showed impairments or no changes of the investigated cognitive domains, confirming the slowly progressive deterioration, typical of the clinical course of AD. After a 6-month period of LSS therapy discontinuation, the MMSE mean score remained significantly higher than the baseline evaluation and comparable with the value recorded at the end of treatment. Specific cognitive functions, which improved after LSS therapy, declined after treatment discontinuation. Nevertheless, at follow-up many neuropsychological test scores were still higher, although not significant, compared with baseline. In our opinion, LSS therapy may yield modest but persistent improvements in different cognitive domains, which, summing-up, may contribute to the long-lasting significant improvement of the MMSE.
The results indicate that an intervention aimed at stimulating lexical–semantic abilities may be effective in improving verbal communication and semantic memory, then reconfiguring other verbal-related memory domains such as episodic memory. From the neuropsychological background of language deterioration in AD, 1 it was expected that lexical–semantic exercises administered in LSS would succeed in obtaining an improvement of verbal communication by reorganizing and reinforcing semantic memory. Instead, the finding that improvement of lexical–semantic organization results in improvement of test scores tapping episodic memory is somehow surprising, since semantic and episodic memory are known to be dissociated. Moreover, as a general rule, category cueing does not have positive effects on episodic memory in AD. 27 It may be speculated, however, that simple semantic cueing may not have enough strength to facilitate the performance on episodic recall. LSS training tasks, instead, require more articulated exercises leading to a reorganization of semantic memory. This reorganization could then help the encoding phase of declarative memory by efficiently capturing new information with a richer semantic network. The semantic encoding phase is thought to be instrumental for efficient functioning of episodic memory, which is suggested by our study. In fact, RAVL delayed recall test scores, where the information has more time to be encoded in long-term memory, improved significantly whereas RAVL immediate recall test scores did not.
These “cascade” effects may be related to the anatomo-functional organization of the brain, as thoroughly studied by Agnati and Fuxe 28 since 1984. According to these investigators, the brain is a plastic system composed of highly interconnected networks, hierarchically organized in a fractal-like way, from the molecular to the neuronal level.29,30 Therefore, the cognitive system may be viewed as an ensemble of nested networks each regulating functions such as memory, attention, language, and so on. The high degree of plasticity and connectivity allows the neuronal networks to reshape themselves in response to adequate stimulations or lesions and to convey new information to other anatomo-functional systems. Within this theoretical framework, this study showed that LSS treatment, targeted to lexical–semantic verbal function, improves the efficiency of the corresponding semantically related domains and reshapes other connected cognitive networks. The reasons why only some networks are involved in this reorganization are still unknown. This phenomenon may depend on several factors, including the hierarchical organization of the brain (such as strength of connections), the intensity and specificity of brain stimulation, and the degree and spatial extent of neurodegenerative brain lesions.
Conclusion
We demonstrate that focused cognitive stimulation aimed at improving lexical–semantic abilities in early AD subjects spreads its effects beyond the direct target of the intervention, involving memory and other cognitive abilities. Its effect on global cognitive performance lasted beyond the end of therapy. LSS treatment may therefore be regarded as one of the clinical options to apply in the attempt to counteract the progressive cognitive decline typical of the disease. Further testing of stimulation of other cognitive components 31 can contribute to rehabilitation strategies for cognitive impairment.
Footnotes
Acknowledgements
We thank Prof Carlo Semenza and Dr Serena De Pellegrin (Department of Neurosciences in Padova) for their suggestions on lexical–semantic stimulation and useful discussion on study design and results interpretation.
Authors’ Note
Nela Jelcic and Annachiara Cagnin contributed equally to the research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the Italian Ministry of Health within the Finalized Research Projects 2006-2007 (Type I, RFNMC 2005- 626001).
