Abstract
Background. The slow rate of nerve regeneration following injury can cause extended muscle denervation, leading to irreversible muscle atrophy, fibrosis, and destruction of motor endplates. The immunosuppressant FK506 (tacrolimus) has been shown to accelerate the rate of nerve regeneration and functional recovery. However, the toxic and immunosuppressive properties of FK506 make it undesirable for long-term use. Objective. To take advantage of the regeneration-enhancing effects of FK506 but avoid the potential adverse effects of long-term administration, the current study evaluates and quantifies the efficacy of short-term FK506 treatment in rat models. Methods. Clinically relevant transection and graft models were evaluated, and walking track analysis (WTA) was used to evaluate functional recovery. FK506 was administered for 5 and 10 days post transection injury and 10 and 20 days post graft injury. Both groups involving a short course were compared with the continuous administration group. Results. In the transection model, FK506 was administered for 5 and 10 days postoperatively. WTA demonstrated that 10 days of FK506 administration was sufficient to reduce functional recovery time by 29% compared with negative controls. In the graft model, FK506 was administered for 10 and 20 days postoperatively. Short treatment courses of 10 and 20 days reduced recovery time by 15% and 21%, respectively, compared with negative controls. Analysis of blood–nerve barrier (BNB) integrity demonstrated that FK506 facilitated early reconstitution of the BNB. Conclusions. The results of this study indicate that short-term FK506 delivery following nerve injury imparts a significant therapeutic effect.
Introduction
Although significant advances in nerve repair have been achieved over the past few decades, complete functional recovery following traumatic peripheral nerve injury is rare, and clinical outcomes are often poor.1-3 Complete recovery of muscle function and restoration of protective sensibility, especially discriminative sensibilities, are the ultimate clinical goal for peripheral nerve reconstruction. Successful restoration of muscle function is dependent on reinnervation above a threshold number of motor axons within a critical time period. Muscle denervated for longer than the critical time period will undergo muscle atrophy and irreversible destruction of motor endplates. 4 Conversely, clinical experience indicates that shorter time periods of denervation correlate with improved recovery of muscle function. 5 Laboratory experiments have shown that accelerating nerve regeneration consistently speeds functional recovery and positively affects overall functional outcomes following peripheral nerve injury.6-17 Interventions that speed up nerve regeneration and reinnervation of distal targets have direct applicability to the clinical management of severe peripheral nerve injuries.
FK506 (tacrolimus) is an agent that has the ability to accelerate nerve regeneration. Currently, FK506 is used as an immunosuppressant in clinical solid organ transplantation; it prevents organ rejection by binding calcineurin and inhibiting T-cell proliferation.18-20 In a fortuitous discovery, FK506 was found to augment neural regeneration following peripheral nerve injury. 8
Members of our team began investigating the effects of FK506 on nerve regeneration in the late 1990s and have demonstrated its enhancement of peripheral nerve regeneration in multiple nerve injury models, including crush,12,15 transection, 11 chronic axotomy, 14 isograft, 21 and allograft22,23 models. Despite repeated demonstration of the beneficial effect of FK506 on nerve regeneration, its use in clinical cases of severe nerve injury has been limited by the undesirable side effects associated with its long-term administration. These side effects include increased risk of diabetes mellitus, hypertension, hyperlipidemia, abnormal renal function, gastrointestinal disturbance, leukopenia, cytomegalovirus infection, and opportunistic infections (http://www.prograf.com/index.php).
For successful translation of FK506 to human cases of peripheral nerve injury, it will be necessary to minimize its side effects and maximize its enhancement of nerve regeneration. Previous studies have demonstrated that FK506 accelerates nerve regeneration for as long as it is being administered. 16 However, whether short-term treatment will provide a significant enough benefit to merit clinical translation has yet to be investigated. The current study was designed to comprehensively and quantitatively evaluate the efficacy of short-term FK506 administration on nerve regeneration in rodent models. We hypothesized that short-term administration of FK506 would provide a substantial functional benefit meriting consideration for clinical translation.
Materials and Methods
Experimental Design
All animal procedures were approved by the Animal Studies Committee of Washington University and performed in strict accordance with their guidelines. Rats were housed in a central animal care facility and given a standard rodent diet and water ad libitum. Rats were monitored for weight loss and other signs of distress during the experimental period.
Adult male Lewis rats (of body weight around 250 g) were purchased from Charles River Laboratories International, Inc (Wilmington, Massachusetts). For evaluation of functional recovery, 79 Lewis rats were used—75 for walking track analysis (WTA) and 4 as donors in the graft group. The 75 rats were randomly assigned to undergo one of the following procedures: tibial nerve crush (15 rats), tibial nerve transection (36 rats), or tibial nerve graft (24 rats). Also, 30 other Lewis rats were used for blood–nerve barrier (BNB) continuity analysis—15 rats received FK506 treatment, and 15 rats received no FK506 treatment. The reconstitution of the BNB was evaluated beginning 4 days after nerve crush injury. One rat from each group was evaluated per day for 18 days.
The different surgical models (crush, transection, and graft) provide different levels of regenerative challenge to regenerating axons. It is known that higher degrees of nerve injury require longer periods of time to elicit similar regenerative effects. 24 Similarly, different durations of FK506 administration for the different levels of injury are necessary to observe an effect on regeneration. Although the actual administration times are different for each injury model, the administration time as a percentage of the normal time for functional recovery (ie, recovery without FK605) is similar for each injury model (FK506 administration for ~20%, ~40%, and 100% of the normal recovery time). In addition, the duration of FK506 administration for the 3 models was in part based on previously published data that suggested that we would see an affect.11,12,25
Crush model
A nerve crush model was used in preliminary studies to evaluate the potential for short course administration of FK506 to enhance functional recovery and to help determine appropriate short course durations for FK506 treatment. Rats were divided into 5 subgroups, with 3 rats in each group. All rats received tibial nerve crush 3 mm distal to the trifurcation of the sciatic nerve on the right side. Subgroup A served as a negative control and received no FK506 treatment. Subgroup B served as a positive control and received continuous administration of FK506 (2 mg/kg; SC, LC Laboratories, Woburn, Massachusetts; Cat. No: F-4900) for the duration of the study. Subgroups C, D, and E received short postoperative courses of FK506 treatment for 3, 7, or 14 days, respectively (Figure 1A).

FK506 administration was started 3 days before the operation in all FK506-treated groups in all crush, transection, and graft models. The black triangles indicate the duration of FK506 administration in each group. A. Protocol for tibial nerve crush model with 3 rats in each group. FK506 was withdrawn at t = 3, t = 7, and t = 14 days, except in group B (positive control), in which the FK506 treatment was administered continuously until the end point. B. Protocol for tibial nerve transection and repair model with 9 rats in each group. FK506 was withdrawn at t = 5 and t = 10 days, except in group B (positive control). C. Protocol for tibial nerve graft model with 6 rats in each group. FK506 was withdrawn at t = 10 and t = 20 days, except in group B (positive control).
Transection model
Rats were divided into 4 subgroups with 9 animals in each group. All animals underwent tibial nerve transection and immediate repair 3 mm distal to the sciatic nerve trifurcation on the right side. Subgroup A served as a negative control and received no FK506 treatment. Subgroup B served as a positive control and received continuous administration of FK506 (2 mg/kg) for the entire duration of the study. Subgroups C and D received short postoperative courses of FK506 treatment for 5 or 10 days, respectively (Figure 1B).
Graft model
Four Lewis rats were used as donors to provide nerve isografts for the experimental groups. The remaining rats were divided into 4 subgroups with 6 rats in each group. All animals received a 5-mm nerve isograft inserted 3-mm distal to the sciatic nerve trifurcation. Subgroup A served as a negative control and received no FK506 treatment. Subgroup B served as a positive control and received continuous administration of FK506 (2 mg/kg) for the entire duration of the study. Subgroups C and D received short postoperative courses of FK506 treatment for 10 and 20 days, respectively (Figure 1C).
In our previous studies, we found that FK506 preloading 1 to 3 days prior to nerve injury yielded optimal results compared with administration at the time of and after nerve injury.12,26 At first glance, preloading FK506 does not appear to be clinically relevant because in most cases (with the exception of nerve transfer procedures27,28), it is impossible to predict the exact time of nerve injury. However, during all nerve reconstruction procedures, the nerve undergoes a second injury at the time of reconstruction because the surgeon removes the damaged end of the proximal stump in order to move out of the zone of injury in preparation for surgical repair of the healthy nerve to the distal nerve. This surgical preparation of the proximal stump causes a new injury to the nerve, resulting in the same injury cascade as the primary injury. Thus, clinically the surgeon has the ability to plan for FK506 administration 3 days prior to scheduled nerve reconstruction. In all injury models in this study, FK506 administration was begun 3 days prior to nerve injury and then continued postoperatively for the duration specified for each subgroup. 26 FK506 stock solution (10 mg/mL) was prepared every 3 days by dissolving FK506 in a solution of 80% ethanol and 20% Cremophor EL (Sigma Chemical Co, St Louis, Missouri). Each day, the diluent (75% propylene glycol and 25% deionized water) and stock solution were combined in a 4:1 ratio, and appropriate volumes were drawn into syringes for individual animal administration according to body weight.
Surgical Procedure
Crush model
All surgical procedures were performed aseptically using standard microsurgical techniques under an operating microscope. Rats were anesthetized by subcutaneous administration of 75 mg/kg ketamine hydrochloride (Ketaset, Fort Dodge Animal Health, Fort Dodge, Iowa) and 0.5 mg/kg medetomidine hydrochloride (Dormitor, Orion Corp, Espoo, Finland). The right sciatic nerve and its 3 major branches were exposed through a gluteal muscle–splitting incision. A reliable and reproducible crush injury was created as described previously 12 using a No. 5 jeweler’s forceps for 30 s on the tibial nerve 3-mm distal to the sciatic trifurcation. The crush area was marked with a single 10-0 nylon epineurial suture. After the procedure, the muscle layer and skin were reapproximated with simple interrupted 4-0 nylon sutures. In each animal, the left leg served as a nonoperative control. At the study end point, the animals were again anesthetized and the tibial nerves harvested for histomorphometric evaluation. Animals were then killed with a 0.5-mL intracardiac injection of Euthanasia (Vedco Inc, St Joseph, Missouri).
Transection model
Following anesthesia and exposure, the tibial nerve branch was transected 3-mm distal to the sciatic trifurcation and was immediately reapproximated and repaired with 4 to 5 interupted 10-0 nylon epineurial sutures.
Graft model
Following anesthesia and exposure, the tibial nerve grafts were carefully harvested from donor rats, wrapped in cold saline-soaked gauze, and kept on ice while the recipient rats were prepared. In the recipient rats, a nerve gap was created by transecting the tibial nerve 3-mm distal to the sciatic trifurcation. A 5-mm nerve graft was then reversed in orientation and interposed into the defect. The graft was coapted to the recipient nerve ends with 4 to 5 interrupted 10-0 nylon epineurial sutures.
Functional Assessment of Limb Recovery
A standardized WTA technique was used for the functional assessment of hind limb recovery.11,12,25 The lengths of the footprints on both hind feet (experimental injured and contralateral noninjured control) were measured with a ruler and recorded. The normal side print length (NPL) was compared with the experimental side print length (EPL), and a print length factor (PLF) was derived according to the formula PLF = (EPL − NPL)/NPL. The elongation and subsequent normalization of experimental footprint length correlates with tibial nerve injury and recovery. A PLF for each group was determined by averaging the PLFs of the individual animals within the group. Group PLFs were then compared for statistical significance. Baseline WTAs were performed in all experimental groups prior to surgery.
In the crush model, postsurgery WTA was begun on postoperative day 3 and was performed twice weekly until day 14. After day 14, WTA was performed every day until 19 days after injury (Figure 1A). In the transection model, the WTA was performed on days 5, 10, and 14 postoperatively and then every other day until day 32 (Figure 1B). In the graft model, the WTA was performed on days 1 and 10 postoperatively and then twice weekly until the end time point (Figure 1C). Onset of recovery was determined by sustained (3 days) significant functional improvement (significant change in PLF) relative to the first postoperative walking track. 25
The end point at which all animals were killed was determined based on previous studies evaluating PLF after crush, transection, and nerve graft injuries (no FK506 treatment).
Blood–Nerve Barrier Assessment
BNB integrity was assessed in a rat tibial nerve crush model. In this, 15 rats received FK506 treatment, and 15 rats served as negative controls (no FK506). The crush procedure was performed as described above. The BNB analysis was performed every day from day 4 through day18 postoperatively. Rats in the experimental group received preloading with FK506 (2 mg/kg). The analysis on day 4 postcrush was set as the baseline for BNB integrity assessment. One rat per group was used at each time point from day 14 to day 18. For the BNB assessment, after administration of anesthesia, 2 mL of Evans blue albumin complex (EBA; 1% Evans blue, 5% albumin at 1 mL/100 g) was injected into the left femoral vein with #27G needle. Then, 15 minutes after the injection, the right tibial nerve was harvested and fixed in a 10% phosphate-buffered formalin solution for 24 hours. The integrity of the BNB was evaluated using an N-2 filter fluorescence microscopy system (Olympus BX51) by an observer blinded to the experimental groups. Leakage of the dye from the endoneurial blood vessels into the endoneurium indicated a nonintact BNB. 12
Statistical Analysis
Walking track data that passed the normality and equal variance tests were analyzed using 1-way repeated-measures analysis of variance (ANOVA), followed by a Student-Newman-Keuls post hoc test for pairwise multiple comparison. The level of significance (α) was set at .05, and statistical results were only considered if the calculated power of the performed test was ≥0.8. Functional recovery was defined as significant improvement in the PLF compared with the first day after injury that continues to be significant for the remainder of the study. 25 All results are reported as mean ± standard error of the mean.
Results
Functional Assessment of Limb Recovery
All animals demonstrated complete flaccid paralysis of the operative foot following the crush, transection, and graft procedures. Baseline WTAs performed prior to surgery were similar among animals in all injury models and experimental groups.
In preliminary studies, a nerve crush model was initially used to evaluate the potential for short course administration of FK506 to enhance functional recovery. Rats in all FK506 treatment groups (3-day, 7-day, 14-day, or continuous administration) exhibited a similar trend of functional recovery by postoperative day 14, which was 4 days earlier than the control group (Figure 2). However, the experimental number was not sufficient to obtain statistical significance.
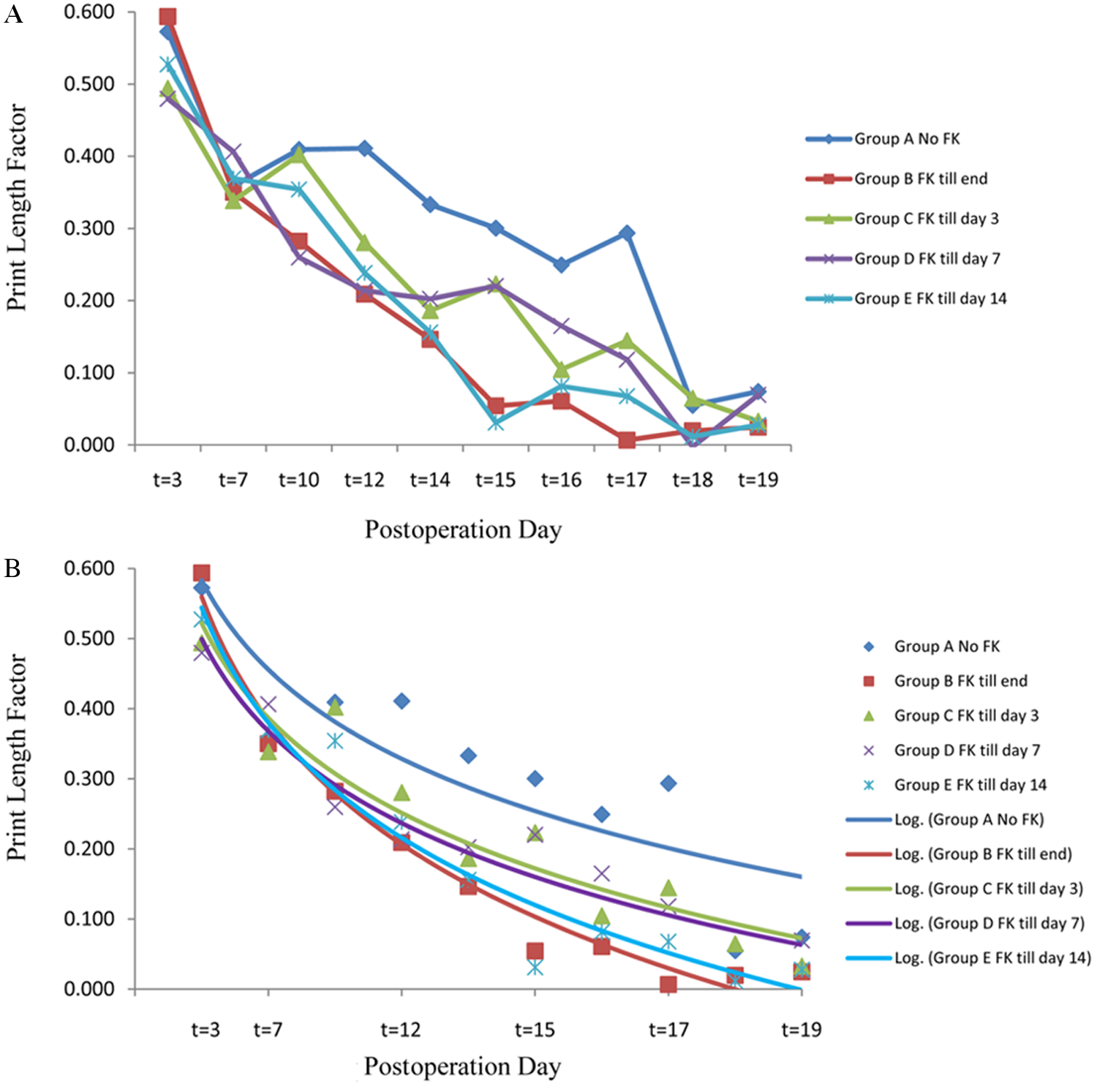
A. Walking track analysis in the tibial nerve crush model: the print length factor (PLF) in all FK506-treated groups recovered 4 days (at t = 14) earlier than the control group (at t = 18). B. Logarithmic trendline analysis with the PLF data (error bars indicate standard error of the mean).
In the nerve transection model, PLF analysis revealed return of hindlimb function on postoperative day 28 in the negative control group. Continuous and 10-day administration groups exhibited return of hindlimb function on postoperative day 20. Return of function was 8 days earlier than in the control group, corresponding to a reduction in recovery time of 29% (Figure 3). Figure 3B shows the data fit to an exponential equation and depicts the trend in return of function.
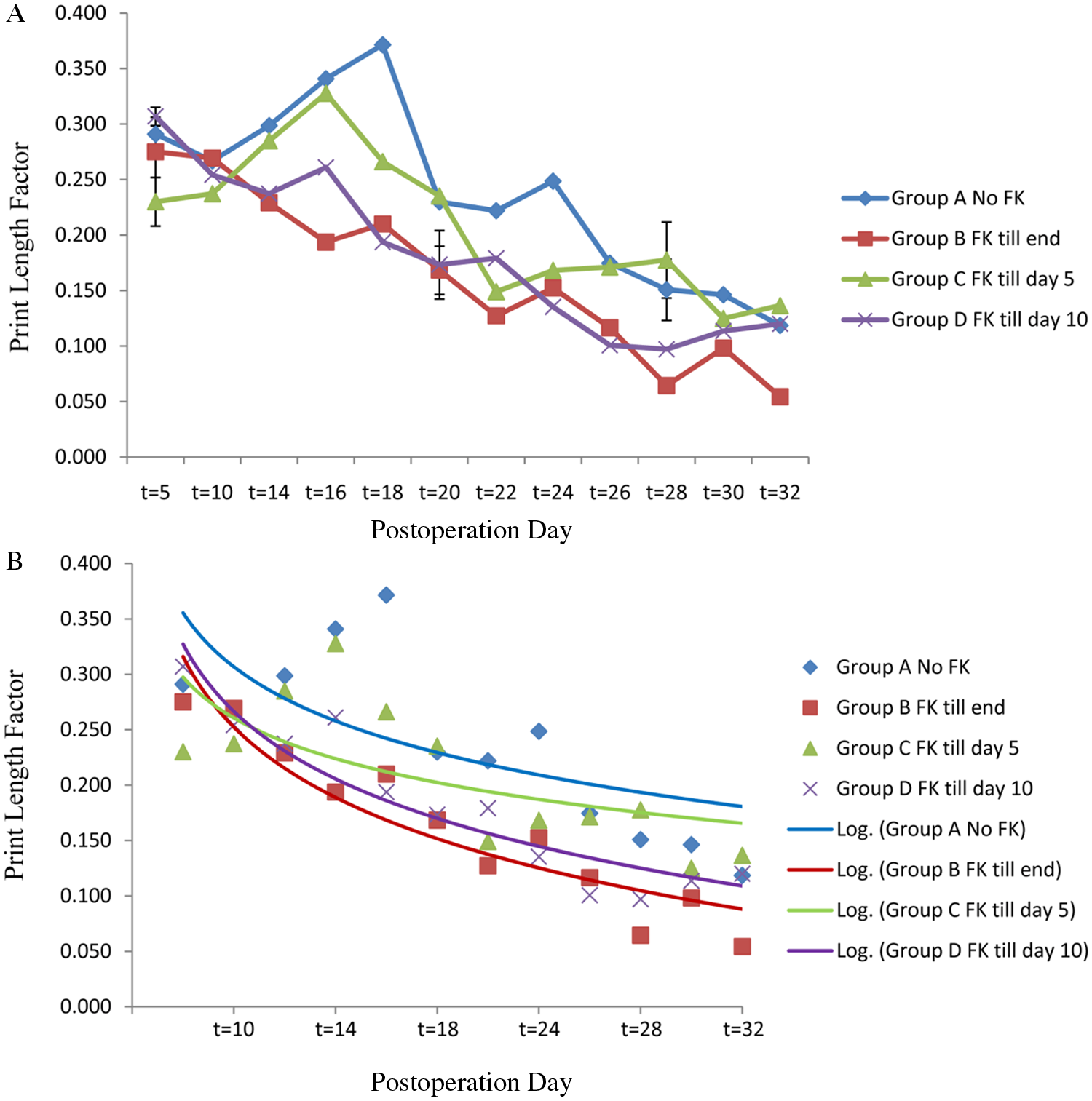
A. Walking track analysis in the tibial nerve transection and repair model. The print length factor (PLF) in the full-term (positive control) FK506-treated group recovered at t = 18. The 10-day course group recovered at t = 18, 10 days earlier than the negative control group (at t = 28). B. Logarithmic trendline analysis with the PLF data (error bars indicate standard error of the mean and are included for significant time points).
In the nerve graft model, significant functional recovery was achieved by control animals by postoperative day 48. The continuous FK506-treated group recovered by postoperative day 34. The 10- and 20-day FK506 treatment yielded functional recovery by postoperative day 41 and 38, respectively, 7 and 10 days earlier than the negative control group (Figure 4). Therefore, continuous FK506 administration was able to reduce the recovery time by 29%, and short-course administration of either 10 or 20 days by 15% and 21%, respectively. Figure 4B shows the trend in functional recovery when the data are fit to an exponential equation.
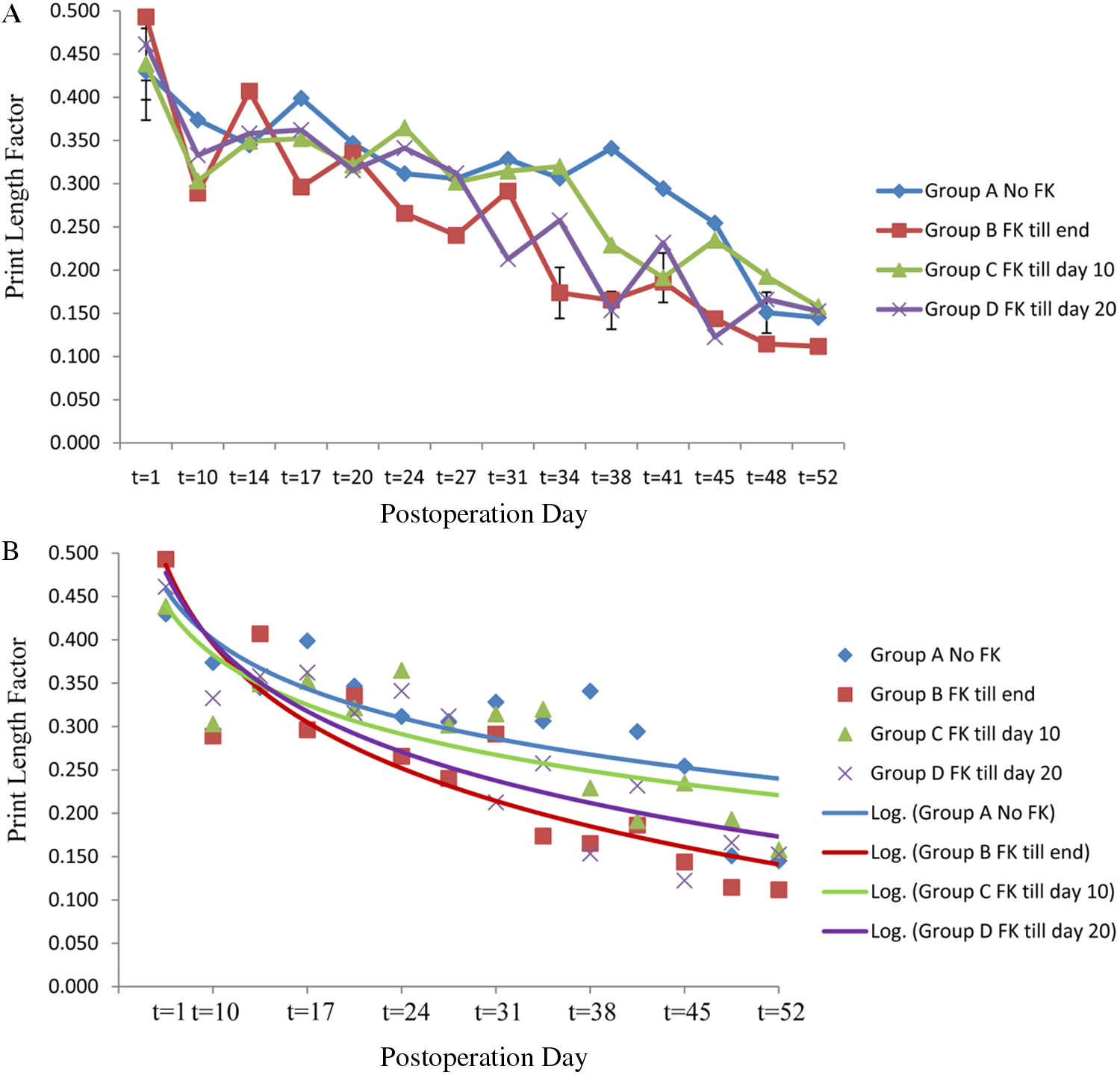
A. Walking track analysis in the tibial nerve graft model: the print length factor (PLF) in the full-term (positive control) FK506-treated group recovered at t = 24, 18 days earlier than the negative control (at t = 42). The 10- and 20-day short-course groups recovered at t = 38 and t = 31 days, respectively, 4 and 11 days earlier than the control group (at t = 42). B. Logarithmic trendline analysis with the PLF data (error bars indicate standard error of the mean and are included for significant time points).
Blood–Nerve Barrier Integrity
Injection of EBA complex, beginning on postoperative day 4 postcrush injury, demonstrated breakdown of the BNB in both FK506-treated and control (no FK506) groups. By postoperative day 9, the BNB had been reestablished in the FK506-treated group (Figure 5). In contrast, an intact BNB was not observed in the control group until day 14. The brightness of EBA that had leaked into the endoneurium was much dimmer in the FK506 treatment group after day 8 than in the control group. Once the BNB had been reestablished in each group, it was consistently maintained at subsequent time points (Table 1).
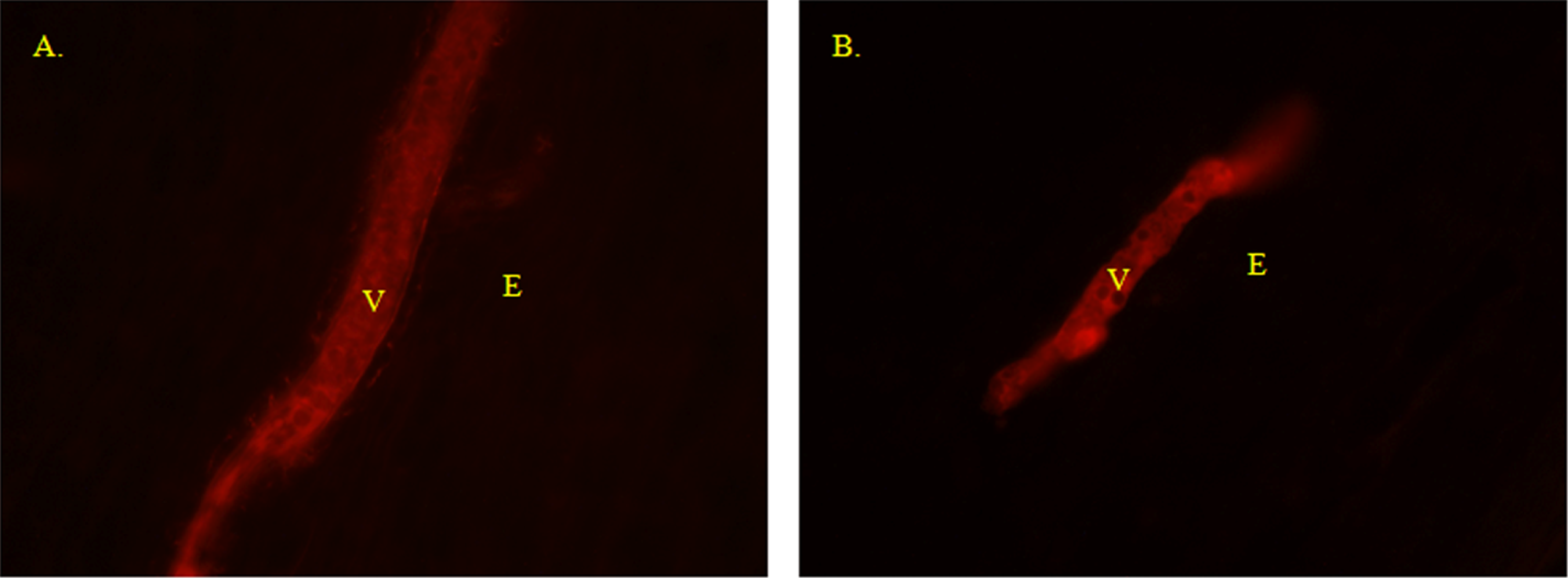
Blood–nerve barrier on postoperative day 9 is leaking in controls (A) but intact in FK506-treated animals (B). Abbreviations: E, endoneurium; V, vessel. Scale bar indicates 10 µm.
Blood–Nerve Barrier Reconstitution a
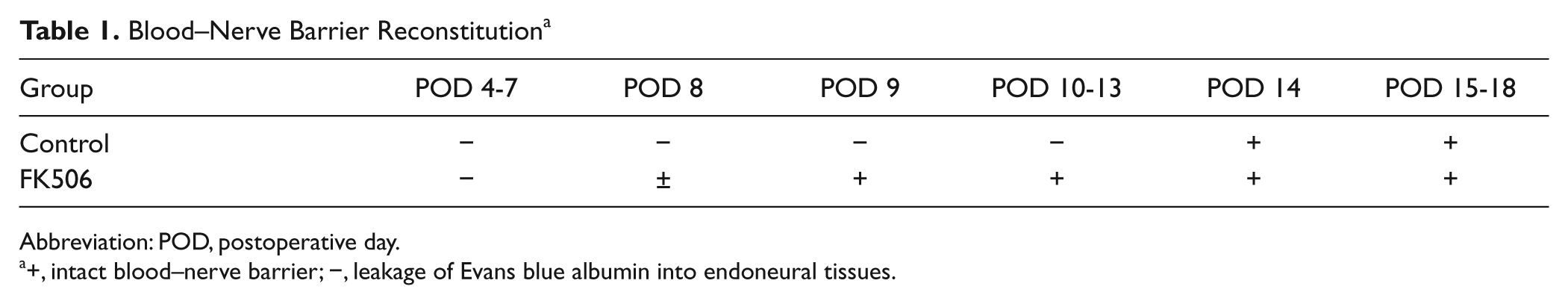
Abbreviation: POD, postoperative day.
+, intact blood–nerve barrier; −, leakage of Evans blue albumin into endoneural tissues.
Nerve Histomorphometry
Nerve histomorphometry was performed on the tibial nerves from each group after the conclusion of the study. Nerve sections distal to crush, transaction, and graft areas were evaluated for fiber density, percentage nerve, and total number of nerve fibers. As expected, there were no significant differences between the groups for all 3 models (data not shown). In all 3 models, the end time point for nerve harvest was after the control group animals fully regained behavioral function. As such, the nerve fiber quantification for all groups was not statistically different.
Discussion
Restoration of function following peripheral nerve injury is severely limited by several factors inherent to normal regeneration. The time delay between injury and reinnervation of denervated muscle has a significant impact on the extent of functional recovery. 4 Prolonged recovery times correlate with decreased quality of recovery. Many factors determine the length of this delay, including the time to therapeutic intervention after injury, the distance from the injury site to the denervated muscle, and the speed of axonal regeneration. Axons are estimated to regenerate approximately 1 to 3 mm per day.20,29-33 With regenerative distances in clinical cases of upper-extremity nerve injury commonly ranging from 10 to 70 cm, the time to regeneration can be up to 24 months. Mackinnon and Dellon 33 have noted in their clinical practice that the maximum time for which proximal muscle can tolerate denervation is 12 months.
Currently, there are no clinically approved interventions to accelerate nerve regeneration following injury and repair. Several therapeutic interventions have been evaluated for their ability to accelerate nerve regeneration, including numerous growth factors34,35 and electrical stimulation. 36 FK506 is the only therapeutic that has been shown to consistently accelerate nerve regeneration and has been comprehensively evaluated to determine ideal parameters for administration.
FK506 has been proven to accelerate nerve regeneration and speed functional recovery in a variety of central and peripheral nerve injury models.6,8,10-15,17,21-23,26,37 The timing of FK506 delivery affects the extent of its regenerative effect; FK506 preloading 1 to 3 days prior to nerve injury yields optimal results, with even further increases in nerve regeneration compared with administration at the time of and after nerve injury.12,26 At first glance, preloading FK506 does not appear to be clinically relevant because in most cases (with the exception of a nerve transfer procedure), it is impossible to predict the time of nerve injury. However, during nerve reconstruction, the nerve undergoes a second injury at the time of surgical repair. Following exposure and prior to reconstruction, the surgeon must remove the damaged tip of the proximal stump to provide an even and scar-free site for surgical anastomosis to the distal stump or artificial graft. This preparation of the proximal stump creates a fresh injury that stimulates the same injury cascade as a primary injury. Because of the fresh injury common to all nerve reconstruction procedures, the surgeon has the ability to plan for FK506 administration 3 days prior to scheduled reconstruction. The scenario does not hold for a crush injury that would likely not undergo surgical management, but in the current study, the crush was used primarily as a guide for the subsequent transection and graft studies.
Despite the staggering amount of data on FK506 and the significant clinical need, FK506 has yet to be implemented as a therapeutic intervention following nerve injury. The undesirable side effects associated with long-term administration represent a significant barrier that has prevented its clinical use. Several studies have been devoted to identifying nonimmunosuppressive alternatives to FK506 that can safely provide the same neuroregenerative benefit.38-42 However, an alternative has yet to make a clinical impact.
A short study by Gold et al 43 previously demonstrated that administration of FK506 for a short course (9 days) following nerve injury provided a regenerative benefit. The study concluded that FK506 accelerated axonal regeneration for as long as it was administered. 43 Clinically, this implies that patients might benefit from short-term exposure to FK506, during which time nerve regeneration might be enhanced and unwanted side effects minimized. The study by Gold and colleagues, 43 however, did not comprehensively evaluate or quantify the magnitude of benefit associated with a given duration of administration. This analysis is needed to determine the risk-to-benefit ratio and decide for what severities of nerve injury FK506 might be clinically indicated. The current study provides this analysis by quantifying the enhancement of functional recovery after short courses of FK506 administration in rat models of nerve crush, transection, and grafting.
In a preliminary study, animals subjected to a tibial nerve crush were treated with FK506 for 3, 7, and 14 days after nerve injury and compared with negative (no FK506 treatment) and positive (continuous FK506) controls. The 3-, 7-, and 14-day regimens were based on previously published data from our group. 12 The preliminary results from the crush suggested that administration of FK506 for ~20% (3 days) of the time normally required for regeneration without treatment (18 days) would result in similar functional benefits as extending the duration of exposure to ~40%, ~80%, or 100% of the regeneration time. These preliminary results suggested that the less-severe tibial nerve crush may not provide a sufficient regenerative challenge to evaluate the benefits of short-duration administration of FK506.
Nerve crush is defined as a second-degree injury (axonotmesis) based on the nerve injury classifications published by Sunderland. 5 Second-degree nerve injury consists of selective injury to axons with conservation of the surrounding endoneurial microstructure; here, subsequent axonal regeneration is more efficient and accurate than more severe injuries. 44 In our preliminary crush study, the smaller magnitude of regenerative challenge associated with crush injuries may be 1 factor responsible for the maximum regenerative effect realized over the short exposure time to FK506 treatment.
Nerve transection, Sunderland’s fifth and most severe degree of injury, results in complete discontinuity of all nerve components. In the transection model of the current study, both continuous and 10-day administration of FK506 resulted in functional recovery 20 days postinjury—8 days earlier than in the control group. FK506 administration for 5 days following injury did not result in any regenerative benefit. Administration for ~35% (10 days) of the normal recovery time resulted in a 29% reduction in the time of recovery after transection. The timeline for recovery of function for both controls and the continuous-FK506 administration group for the transection repair experimental model in the current study is consistent with that seen in previous studies.11,45
We hypothesize that the increased administration time needed to see a therapeutic effect in the transection (10 days) compared with crush (~3 days) is proportional to the increased regenerative challenge posed by transection injuries. Previous studies have demonstrated that the presence of surgical anastomosis in transected nerves significantly slows axonal regeneration to rates below the often quoted 1 to 3 mm/d.36,46,47 The combined results of the preliminary crush and transection study suggested that administration of FK506 need only be performed for a certain threshold of time to obtain the same regenerative benefit as continuous administration.
To further evaluate this threshold hypothesis, we investigated short courses of FK506 treatment (for 10 and 20 days) in a nerve isograft model. PLF analysis revealed that continuous administration of FK506 resulted in functional recovery 34 days after injury, compared with 48 days in control animals (29% reduction). The timeline for recovery of function with continuous FK506 administration for the tibial graft model is consistent with that in previous studies.7,25 We found that 10- and 20-day treatment courses improved the recovery time to 41 and 38 days, respectively—a reduction of 15% and 21%, respectively, compared with control animals. It is interesting to note that exposure to FK506 for only ~42% (20 days) of the normal recovery time (48 days) yielded a significant benefit. The results of the graft model are in line with the conclusion put forth by Gold et al 43 that FK506 accelerates axonal regeneration for as long as it is administered and contradict the threshold hypothesis.
Perineurial cells within the nerve are joined to each other by tight junctions that form the structural basis of BNB. The BNB controls the transfer of serum molecules and water across the nerve fascicle, inactivates complement proteins that can damage nerve fibers, and regulates the endoneurial pressure via its contractile properties.48-50 The BNB is damaged following nerve injury, and restoration of the BNB is required for axonal regeneration and remyelination.4,51 Consistent with what we have previously seen, 12 BNB analyses in the current study suggest that FK506 administration after crush injury promotes earlier recovery of the BNB (Table 1).
Conclusions/Implications
The results of this study suggest that short courses of FK506 administration can provide regenerative benefit for second-degree nerve injuries. It is unlikely that the accelerated functional recovery seen in the current study would result in an increase in the overall functional return in the rodent model. Given enough time in the current rodent model, functional recovery from all groups, treated and untreated, will normalize. In a clinical setting, where regeneration would likely occur over longer times/distances in a human, a reduced time of muscle denervation will decrease the amount of muscular atrophy and irreversible degeneration that accompanies chronic muscular denervation. In the clinical case, the reduction in time of denervation will affect the long-term functional recovery. Although it is difficult to directly extrapolate experimental results and predict response in humans, the rodent model of nerve injury has been used consistently over the past 30 years with good reliability. Many study results have influenced treatment of nerve injury in the clinic. In the case of a proximal upper-extremity transection, FK506 treatment for less than 5 months (~35% of expected recovery time) would reduce recovery time from 12 to ~8 months (~29% reduction). In the case of a proximal upper-extremity graft, FK506 treatment for less than 5 months (~40% of expected recovery time) would reduce recovery time from 12 to ~9 months (~21% reduction). In all cases, 5 or fewer months of exposure would significantly reduce recovery time to well below the 12-month deadline needed to prevent muscle wasting. Short-term administration in cases of proximal nerve injury might, therefore, provide a functional result that was previously unobtainable because of the inherently prolonged course of regeneration.
In summary, treatment with FK506 after peripheral nerve reconstruction will accelerate nerve regeneration, reduce the period of denervation, and likely improve functional recovery. Short-term FK506 administration may provide substantial benefit for nerve injury patients whose chance for viable functional recovery was previously unlikely.
Footnotes
Acknowledgements
We would like to thank Dr Simone Glaus for her assistance with preparation of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Institutes of Health grants 5R-01 NS33406 and a Plastic Surgery Research Council Pilot award to PJJ.
