Abstract
Keywords
Introduction
Stroke is a leading cause of disability, primarily arising from motor impairments. Most of the functional recovery of the upper extremity is thought to occur within 6 months after stroke, and the amount of recovery is very limited. 1 It was demonstrated that electrical stimulation, 2 splint, and combined use of electrical stimulation, 3 and splint alone 4 could reduce spasticity and improve motor function in the paretic upper extremity. Also, the efficacy of electromyography (EMG)-triggered neuromuscular electrical stimulation (NMES)5-7 and functional electrical stimulation (FES)8-14 has been reported. However, EMG-triggered NMES and FES so far reported cannot control electrical stimulation in proportion to voluntary EMG of the target muscle. Hence, their application is limited in activities of daily living settings.
Hybrid assistive neuromuscular dynamic stimulation (HANDS) therapy was developed as a new alternative therapeutic approach to facilitate the use of the affected upper extremity in daily living by combining closed loop EMG-controlled NMES with a wrist splint for patients with insufficient mass or individual extension of their paretic fingers.15,16 For assistive stimulation, the integrated volitional control electrical stimulator (IVES) 17 was applied. The IVES is a portable surface EMG-controlled, single-channel neuromuscular electrical stimulator developed by Muraoka. 17 The IVES continually changes its stimulation intensity in direct proportion to the amplitude of the voluntary EMG of the target muscles. In the HANDS therapy, patients wear a wrist–hand splint and carry a portable IVES in a waste bag for 8 hours during the daytime. The electrodes are placed on the paretic extensor digitorum communis (EDC). When the IVES detects the EMG activity on paretic EDC, electrical stimulation is applied through the same electrodes in proportion to the EMG activity and assists the paretic finger extension. It encourages the patients to use their paretic hand because it becomes easier for patients to extend their paretic fingers and perform pinch and release. Patients can therefore use this stimulator at their will in daily life as long as 8 hours a day. The rationale for combining the stimulation system with a wrist–hand splint was derived from the work of Fujiwara et al, 4 who reported that a wrist–hand splint could reduce spasticity in finger, wrist, and elbow flexors, and facilitate finger extensor muscle activities. It was demonstrated that a 3-week HANDS therapy resulted in both clinical improvement of paretic upper extremity and corticospinal modulation in patients with chronic stroke. 15
To further prove the effectiveness of HANDS therapy, a randomized controlled trial is needed. In addition, it is not known whether HANDS therapy is beneficial to patients within 60 days after stroke, during which their motor function generally improves with conventional therapy. The present study explores the effectiveness of HANDS therapy added to conventional rehabilitation as compared with a splint therapy in addition to a standard inpatient rehabilitation treatment for patients, who cannot fully extend their paretic fingers and cannot perform pinch and release in their daily life, in a randomized controlled trial design.
Methods
Participants
Participants were recruited from stroke inpatients at the Tokyo Metropolitan Rehabilitation Hospital from July 2007 to June 2009. The following inclusion criteria were used: (
Of the 441 stroke patients admitted during the study period, 28 met the study criteria (Figure 1). In total, 24 of the 28 patients gave their informed consent for participation and were included in the trial. A computer-generated list randomly assigned the participants either to the HANDS group or the control group. To confirm the differences in improvements with standard rehabilitation, the HANDS therapy or the splint therapy started at 3 weeks after hospitalization (observation period). We did not calculate the sample size of this study because standard mean of the population was not estimated. The study was performed in accordance with the Declaration of Helsinki, was approved by the Tokyo Metropolitan Rehabilitation Hospital Ethics Committee, and was registered to the UMIN Clinical Trial Registry (UMIN000001971).
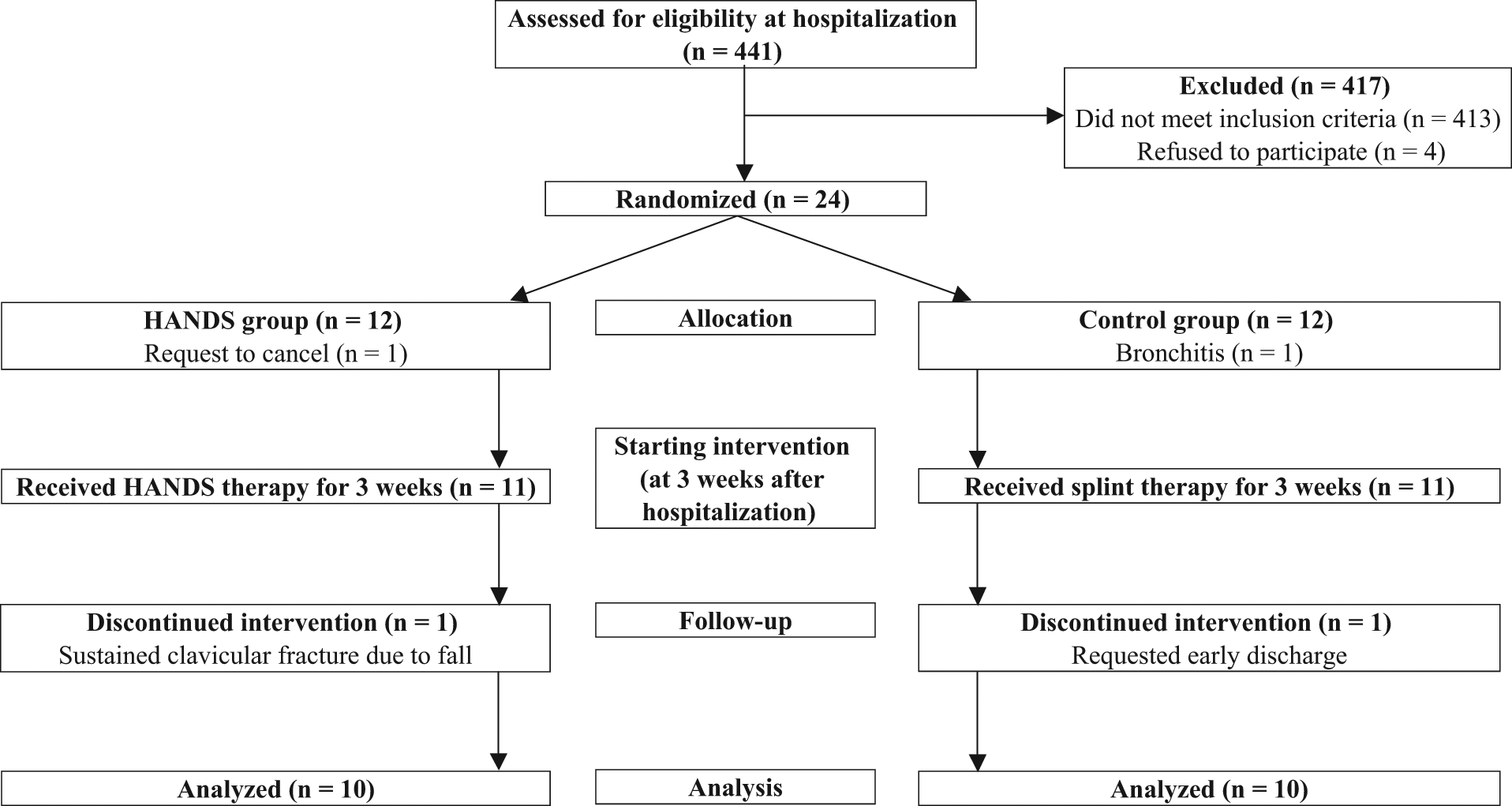
Flow diagram of the randomization procedure. Abbreviation: HANDS, hybrid assistive neuromuscular dynamic stimulation.
Intervention
The HANDS group received 3 weeks of HANDS therapy, which consisted of the IVES and a wrist splint as described in detail by Fujiwara et al. 15 The IVES is a portable, noninvasive (surface), closed loop EMG-controlled, single-channel neuromuscular electrical stimulator. 17 A pair of electrodes for EMG detection and stimulation (30 × 12 mm) placed 5 mm apart, and one electrode (30 × 30 mm) for reference and stimulation were placed on the affected EDC muscle. The stimulator continually adjusts its stimulation intensity in proportion to the amplitude of the voluntary EMG activities. The maximum stimulus intensity was set at a level tolerable by the patients and at which extension of four fingers besides the thumb to 0° was achieved during the voluntary finger extension attempt. The patients also wore a wrist splint and carried a portable IVES in a waist-bag for 8 hours. Their intact upper extremity was not restrained. The control group wore the same wrist splint as the HANDS group for 8 hours a day in addition to the standard rehabilitation program described below. Both groups were instructed to use their affected hand as much as possible while wearing the HANDS system or the splint in their activities of daily living.
Also, all patients participated in a standard rehabilitation program, which consisted of 1 hour of physical therapy and 1 hour of occupational therapy per day, 5 days a week. Speech therapy was added if indicated. All exercises took place in the training rooms. Occupational therapy consisted of gentle stretching exercise of the paretic upper extremity and active muscle reeducation exercise by a therapist’s manual contact. Occupational therapists were directed toward patients’ goals and focused on their particular impairments and disabilities; thus a specific therapy that each patient received was individualized. 15
Outcome Measures
We assessed upper extremity motor function with an upper extremity motor score using the Fugl-Meyer Assessment (FMA). 18 The FMA is a commonly used measure with excellent interrater reliability and construct validity.19,20 The motor score of the upper extremity includes 33 items and ranges from 0 to 66. We separated scores into proximal (shoulder, elbow, and coordination: 42 points, FMA-p) and distal (hand and wrist: 24 points, FMA-d) portions 21 for statistical analysis. The Action Research Arm Test (ARAT) 22 is a frequently used, validated, and reliable measure of upper extremity function with 4 subsections: grip, grasp, pinch, and gross movement.20,23 The maximum summed score is 57. We defined the improvement ratio of the ARAT as the posttreatment score divided by the pretreatment score. The Motor Activity Log (MAL) is a structured interview used to measure upper extremity disability in activities of daily living. The MAL-14 includes 14 items, scored on an 11-point amount of use (AOU) scale (range 0-5) to rate how much the arm is used and an 11-point quality of movement (QOM) scale (range 0-5) to rate how well patients are using their affected upper extremity. 24 High construct validity and reliability have been demonstrated in patients with chronic stroke.24,25 Spasticity was measured with the Modified Ashworth Scale (MAS) 26 of fingers, wrist, and elbow.
All clinical measures were evaluated at hospitalization and pre- and posttreatment. The FMA and MAS were assessed by 2 physiatrists, masked to the therapeutic group assignment of the patient, and ARAT and MAL were assessed by 2 occupational therapists not involved in the treatment of the participants. Prior to the study, we trained the investigators in the use of the FMA, ARAT, and MAL using our manuals, and confirmed good interrater reliability (intraclass correlation coefficients >.95) in 20 inpatients with stroke.
Statistical Analyses
Mann–Whitney
Results
A total of 4 patients dropped out of the study (Figure 1). Two patients were excluded before starting interventions; one chose to drop out, and the other developed bronchitis. Two other patients did not complete the therapy; one (FMA = 24 at pretreatment) sustained a clavicular fracture due to a fall unrelated to the therapy, and the other (FMA = 31 at pretreatment) was discharged at his own request. Consequently, 10 patients in each treatment group completed the interventions. The interventions were not associated with any obvious adverse effects such as shoulder pain.
Demographic data and clinical evaluations of the 20 patients at hospitalization are shown in Table 1. There were no significant differences on any measurements between the HANDS group and the control group at hospitalization. In addition, the changes during an observation period of 3 weeks were not significant in the FMA, FMA-d and FMA-p, ARAT, and MAL-AOU and MAL-QOM between the 2 groups (Mann–Whitney
Patient Characteristics and Clinical Evaluations at Hospitalization a
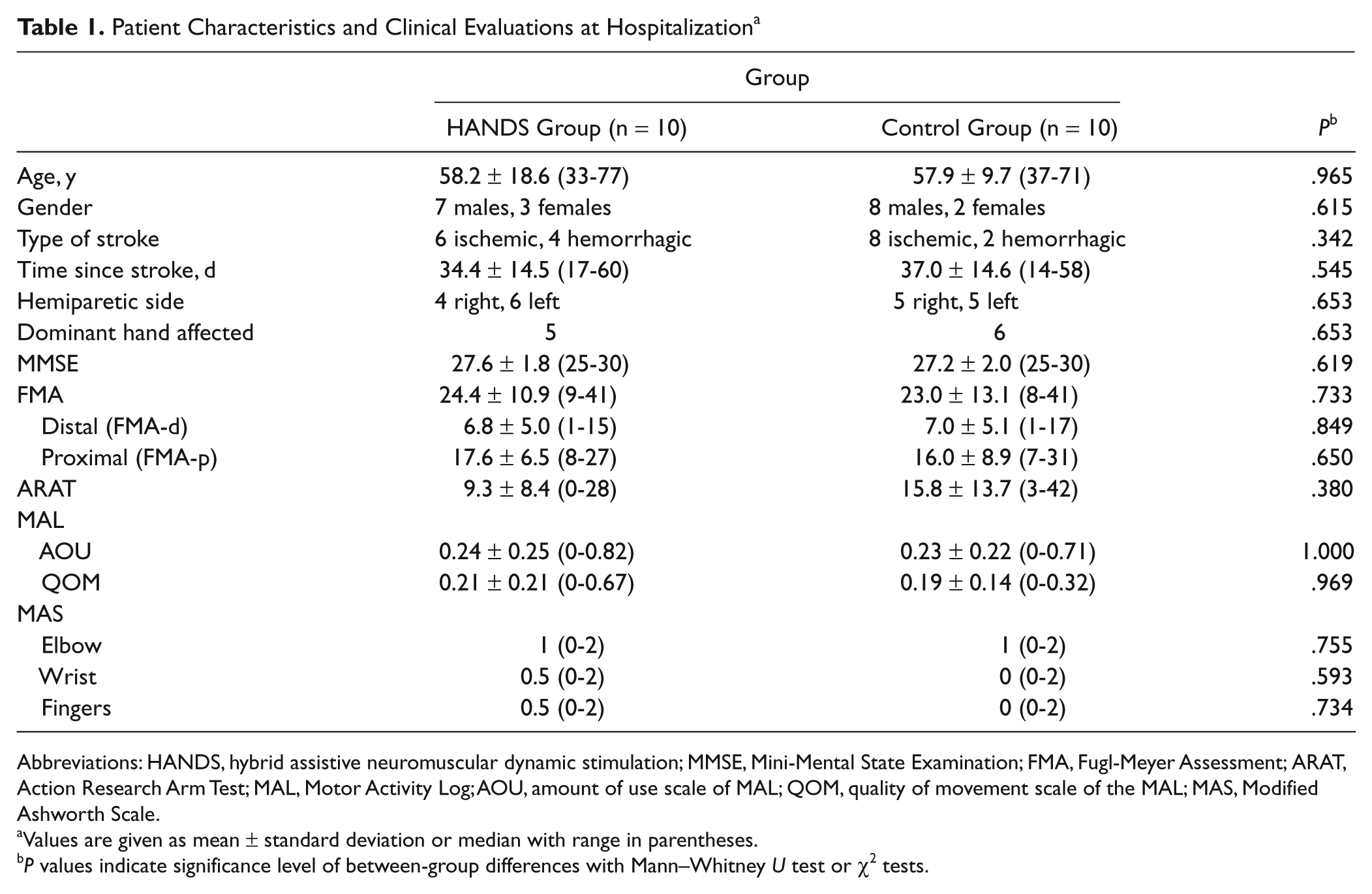
Abbreviations: HANDS, hybrid assistive neuromuscular dynamic stimulation; MMSE, Mini-Mental State Examination; FMA, Fugl-Meyer Assessment; ARAT, Action Research Arm Test; MAL, Motor Activity Log; AOU, amount of use scale of MAL; QOM, quality of movement scale of the MAL; MAS, Modified Ashworth Scale.
Values are given as mean ± standard deviation or median with range in parentheses.
The scores of outcome measures before and after the interventions are shown in Table 2. The HANDS therapy resulted in significantly larger improvements in the FMA, FMA-d, and the improvement ratio of the ARAT than the control group (Mann–Whitney
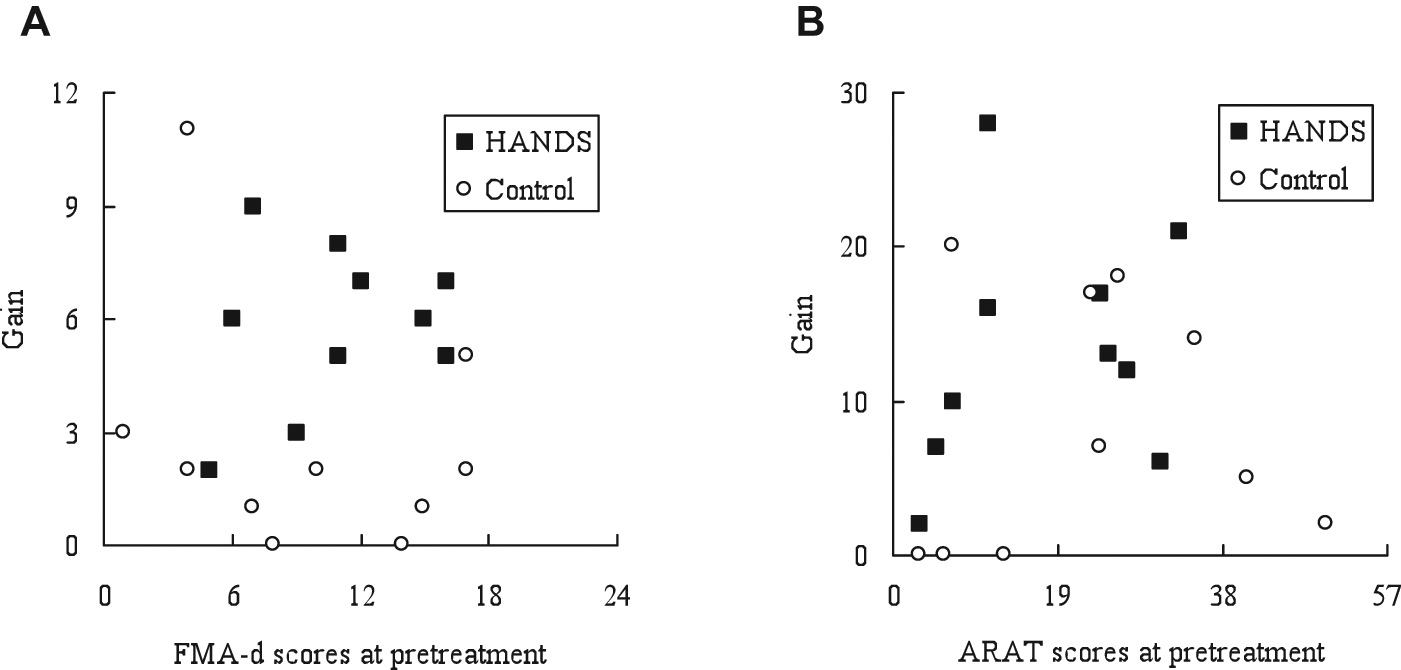
Pretreatment scores and gains after the interventions in distal portion of the FMA and the ARAT. Abbreviations: HANDS, hybrid assistive neuromuscular dynamic stimulation; FMA, Fugl-Meyer Assessment; ARAT, Action Research Arm Test. All participants in the HANDS group showed some improvements in the distal portion item of the FMA (A) and the ARAT (B), but some in the control group did not gain any change.
Pre- and Posttreatment Scores of FMA, ARAT, and MAL in the 2 Groups a
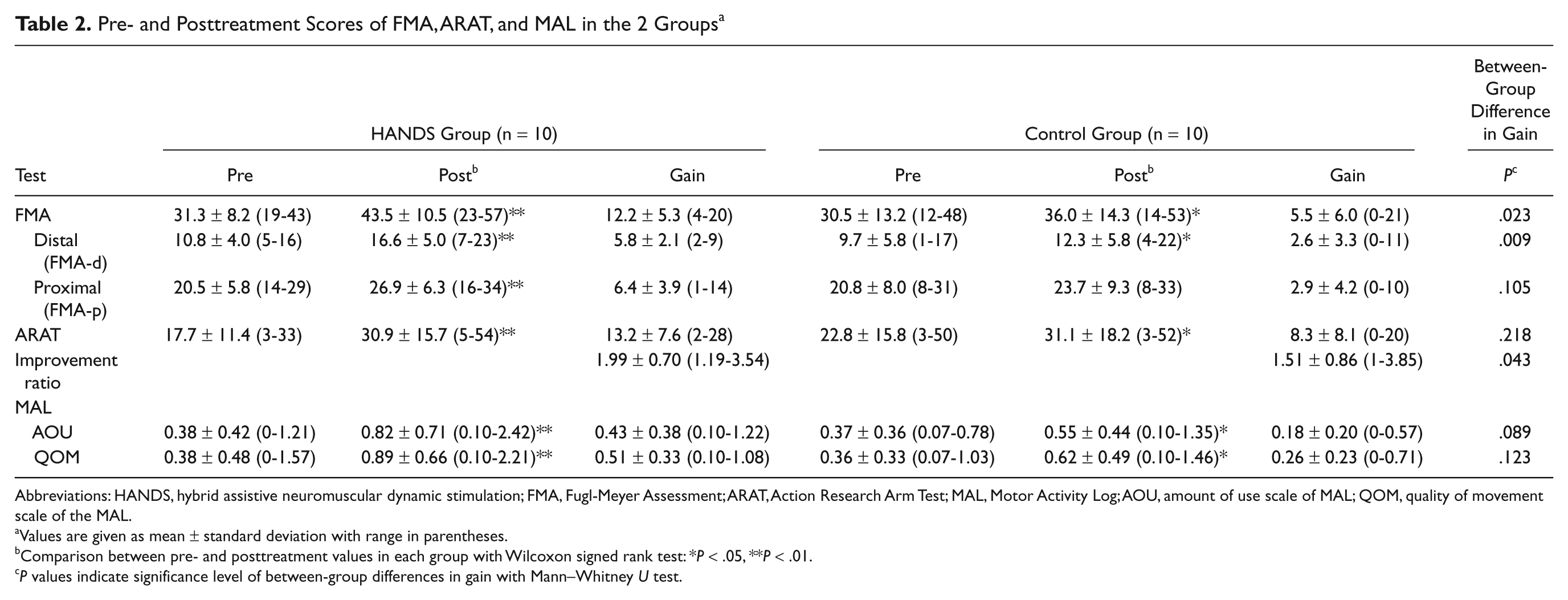
Abbreviations: HANDS, hybrid assistive neuromuscular dynamic stimulation; FMA, Fugl-Meyer Assessment; ARAT, Action Research Arm Test; MAL, Motor Activity Log; AOU, amount of use scale of MAL; QOM, quality of movement scale of the MAL.
Values are given as mean ± standard deviation with range in parentheses.
Comparison between pre- and posttreatment values in each group with Wilcoxon signed rank test: *
Discussion
We found that HANDS therapy in addition to standard rehabilitation produced significantly larger improvements in the FMA-d and ARAT compared with the control group among patients with stroke from 8 (pretreatment) to 11 weeks (posttreatment) after stroke onset. Our findings support the effectiveness of the HANDS therapy, and these results were consistent with the findings of our previous study. 15 Especially, we found significant improvement of hand function with the HANDS therapy though a recent systematic review 27 suggested that none of the interventions identified showed a consistent pattern of improvement in hand function.
The improvements induced with HANDS therapy are thought to be brought about by 3 components: (
In our study, patients in the control group wore the wrist splint during their daily activities. Fujiwara et al 4 reported that a wrist–hand splint could reduce spasticity in finger, wrist, and elbow flexors and facilitate finger extensor muscle activities. Therefore, the wrist splint can make it easier to detect finger extensor activities with surface electrodes. It can also help reduce flexor muscle spasticity, probably through repetitive finger flexion–extension movement in daily activities. In our study, all patients with mild to moderate spasticity in their fingers showed reduced spasticity, or at least a stable level of spasticity.
Dose-dependent use is known to induce reorganization of the damaged motor cortex. 30 HANDS therapy, as well as constraint-induced movement therapy (CIMT), 31 is supposed to induce dose-dependent plasticity. The HANDS system allows patients to use it for 8 hours during the daytime and increase the use of their paretic hand in their daily activities without feeling mental stress. The HANDS therapy has the advantage of controlling electrical stimulation in direct proportion to the voluntary drive of the target muscle, different from EMG-triggered NMES 6 and FES8-12 that produce stereotyped electrical stimulations of the paretic forearm and finger muscles. The HANDS system makes it easier for the patients to perform grip and release, and pinch and release with their paretic hand. The MAL scores improved significantly after the interventions in each group; however, the difference in the gains in the MAL between the HANDS and the control groups was not statistically significant, though the gain in the HANDS group was slightly larger. The MAL scores tend to change more easily in cases of milder paresis, as reported in the CIMT studies.31,32 In patients with more severe paresis, however, gains in motor function may not be easily reflected in changes in the MAL scores. In our previous study, 15 the improvement of motor performance of the affected limb brought about by the HANDS therapy was sustained at 3 months after the intervention without further treatment, indicating the existence of the kind of threshold described by Han et al. 33 In the present study, it is also likely that HANDS therapy may have helped bring performance above the threshold with increased daily usage of the paretic upper extremity, but we could not address this possibility because we did not perform follow-up evaluation. This is an important area for future research.
Compared with a previously reported study using a conventional therapy, HANDS therapy apparently induced greater improvements in motor function. Duncan et al 1 assessed the FMA in 459 patients with stroke from 2 weeks to 6 months after stroke onset. The scores of the FMA in patients with moderate stroke severity increased by only 4 points—from 37 points at 4 weeks to 41 points at 12 weeks—after the stroke onset in their study. In contrast, our data demonstrated that the HANDS therapy improved the FMA score from 31.3 at 8 weeks (pretreatment) to 43.5 at 11 weeks (posttreatment). The average gain in the FMA after the HANDS therapy was 12.2 in 3 weeks. These results suggest that the HANDS therapy has the potential to facilitate greater motor recovery of the upper extremity when compared with conventional therapy, though there might be inconsistent baseline levels and ceiling effects between the 2 studies. Similarly it is difficult to compare the present work directly with other studies6,8-14,32,33 because of the differences in the severity of motor impairment, intensity and duration of intervention, and phase following stroke. Fortunately, the changes during an observation period of 3 weeks were not significant in clinical evaluations between the HANDS and the control groups, though the scores of the ARAT in the HANDS group at hospitalization and pretreatment seemed lower than those in the control group. Motor function of the paretic upper extremity might be influenced by differences in combinations of proximal and distal impairments, as the ARAT evaluated not only hand function but activities requiring reaching and raising of the arm. 13
One limitation of this trial is the relatively small sample size, which prevented us from examining the effect of differences in severity of paresis using a subgroup analysis. Another limitation is the lack of follow-up evaluation after the interventions; after finishing the 3 weeks of therapy reported here, patients in the control group also received the HANDS therapy. Because there was a possibility that the HANDS therapy was simply speeding up recovery, it will be needed to compare overall recovery between the 2 interventions in a longer term study. Also, we did not measure the amount of realistic activity in the affected upper extremity during the interventions for 8 hours a day, as well as the quantity of stimulation through the HANDS system. To further clarify whether the improvements observed in this study were caused by the effects of HANDS therapy or by the increased activity of the paretic limb, an intensity-matched control study is needed to measure the activities of the paretic limb. HANDS therapy may be an effective adjunct to standard rehabilitation in patients with impaired motor control of the hand early after stroke.
Footnotes
Acknowledgements
The authors wish to thank Yoshihiro Muraoka for technical support.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
This study was partially supported by Research Funds from Tokyo Metropolitan Rehabilitation Hospital and the Strategic Research Program for Brain Science (SRPBS) from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
